Carbon-Based Quantum Dots for Supercapacitors: Recent Advances and Future Challenges
Abstract
1. Introduction
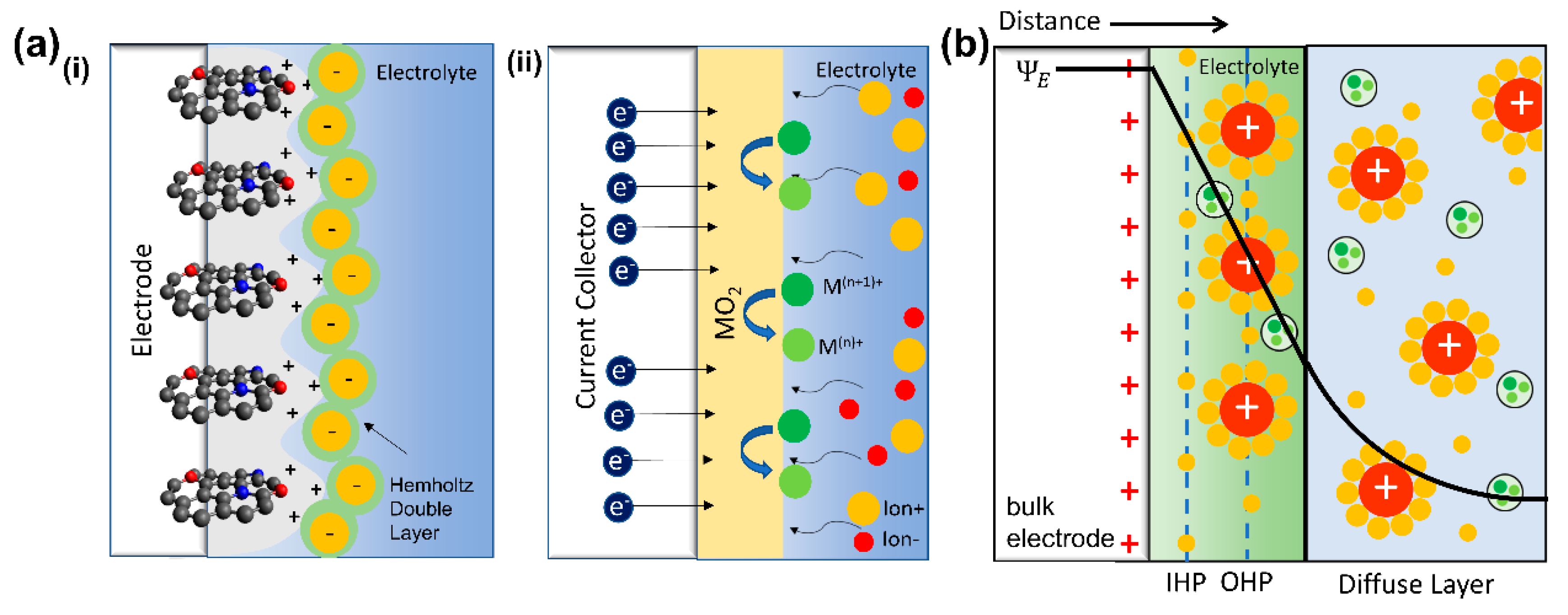
2. Fundamental of Supercapacitor
2.1. Electric Double Layer Capacitor
2.2. Pseudocapacitor
3. Properties and Preparation of Carbon-Based Quantum Dots (C-QDs)
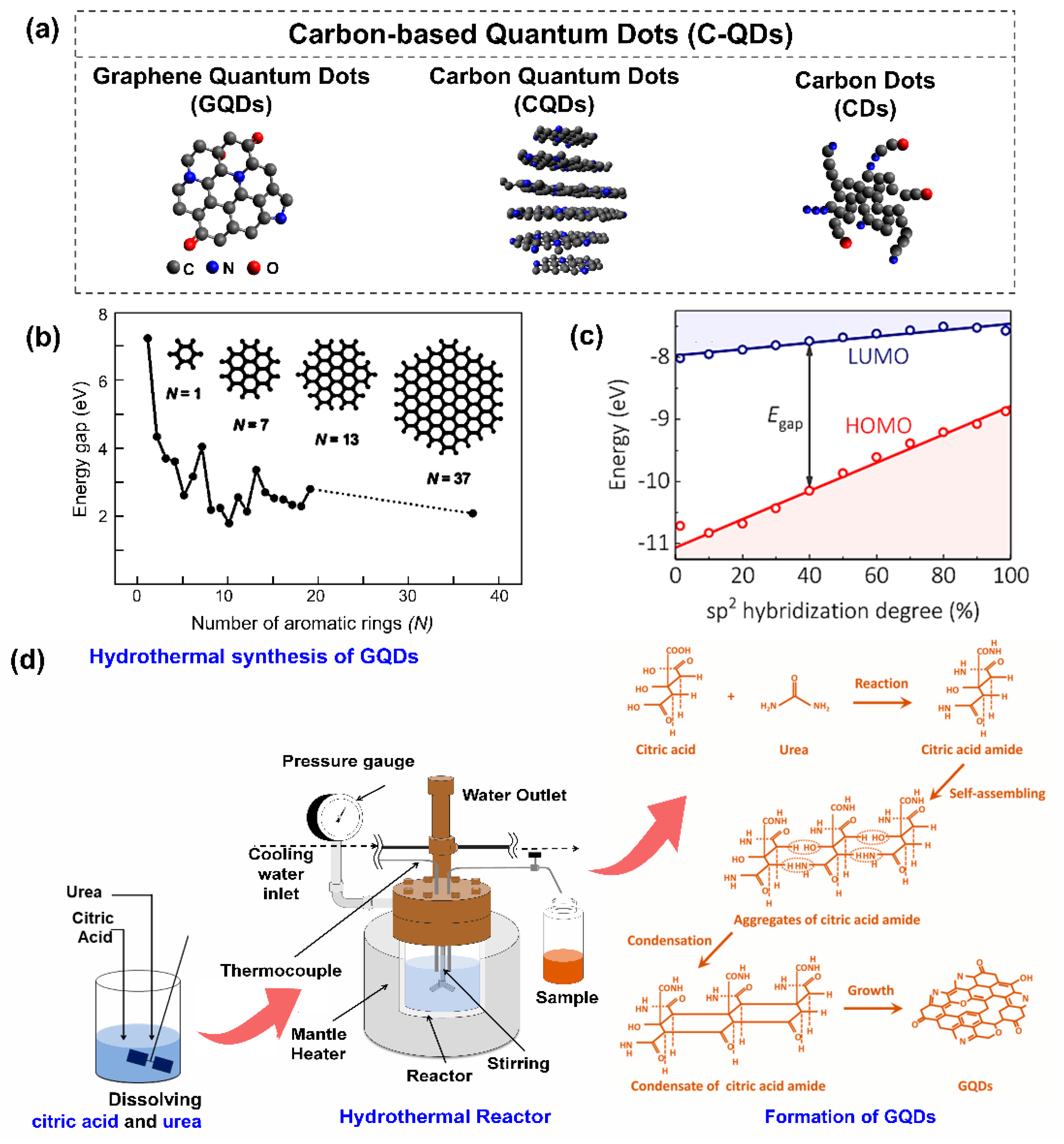
3.1. Structural Properties
3.1.1. Carbon Dots (CDs)
3.1.2. Carbon Quantum Dots (C-QDs)
3.1.3. Graphene Quantum Dots (GQDs)
3.2. Electronic Properties
3.3. Preparation of Carbon-Based Quantum Dots
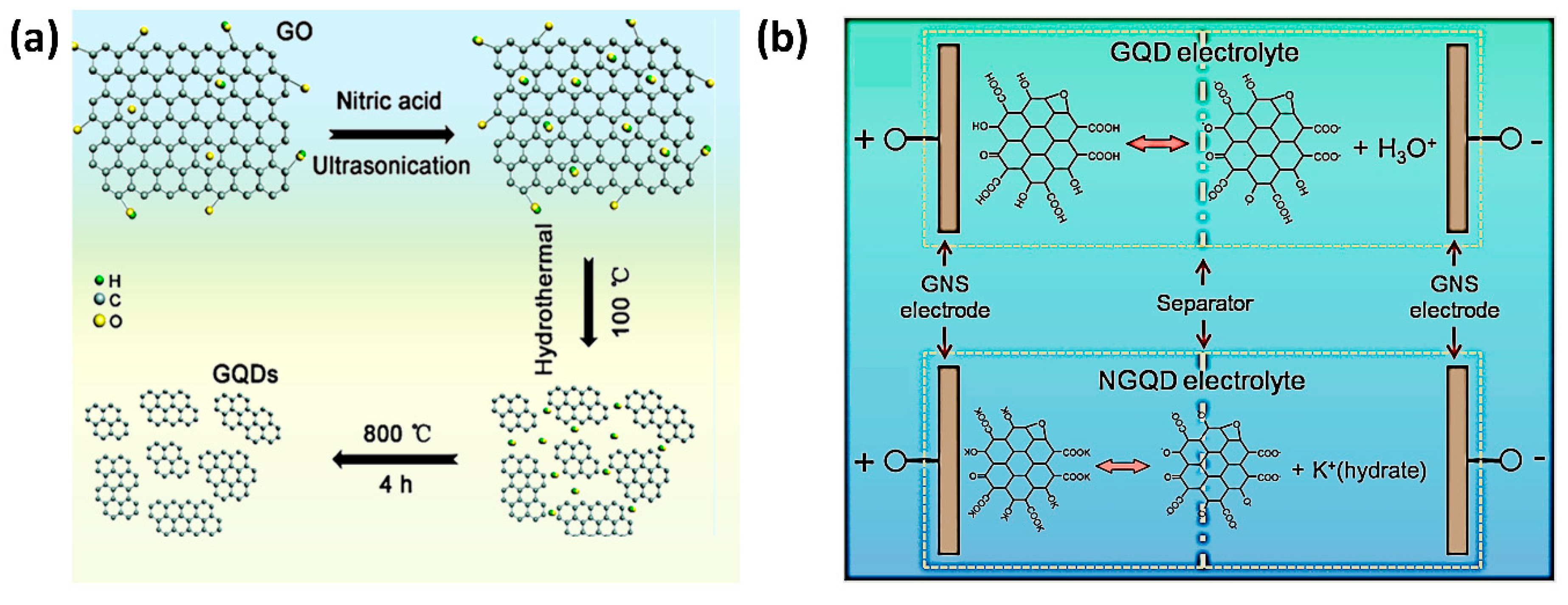
3.3.1. Top-Down Approach
3.3.2. Bottom-Up Approach
4. Recent Progress of Bare Carbon-Based Quantum Dots for Supercapacitors
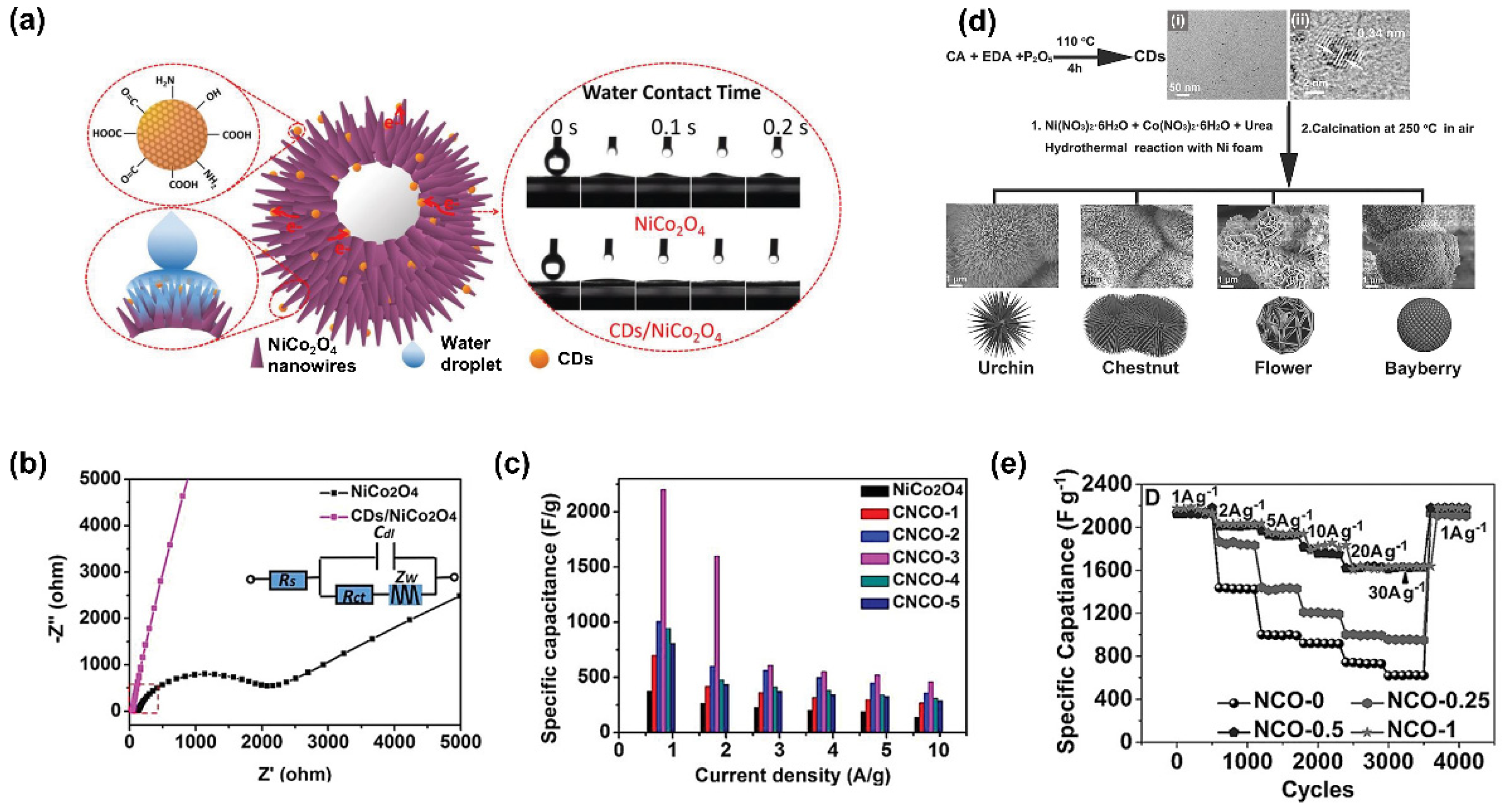
5. Recent Progress of Carbon-Based QD Composites
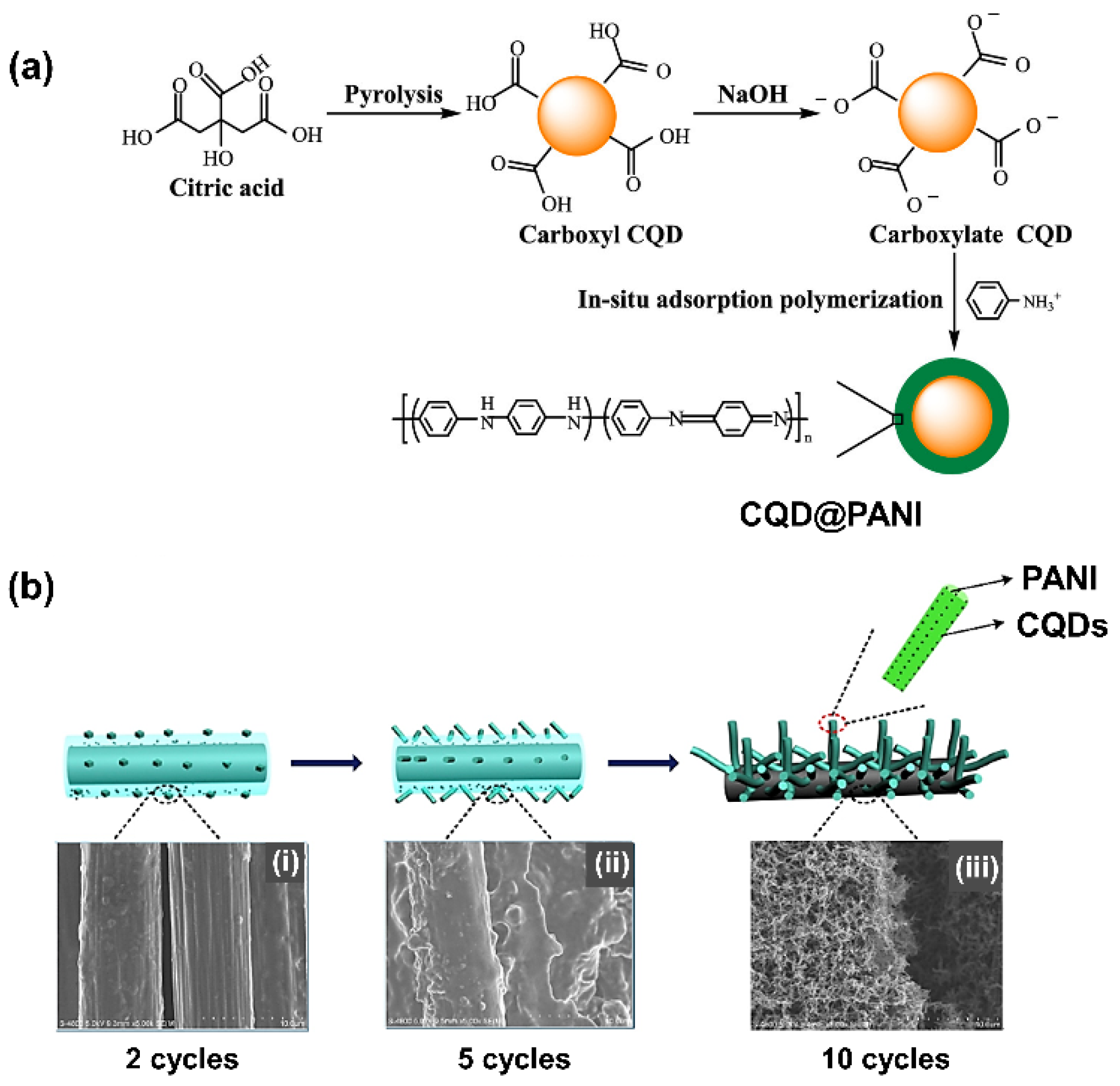
5.1. Carbon Dot Composites
5.2. Carbon Quantum Dot Composites
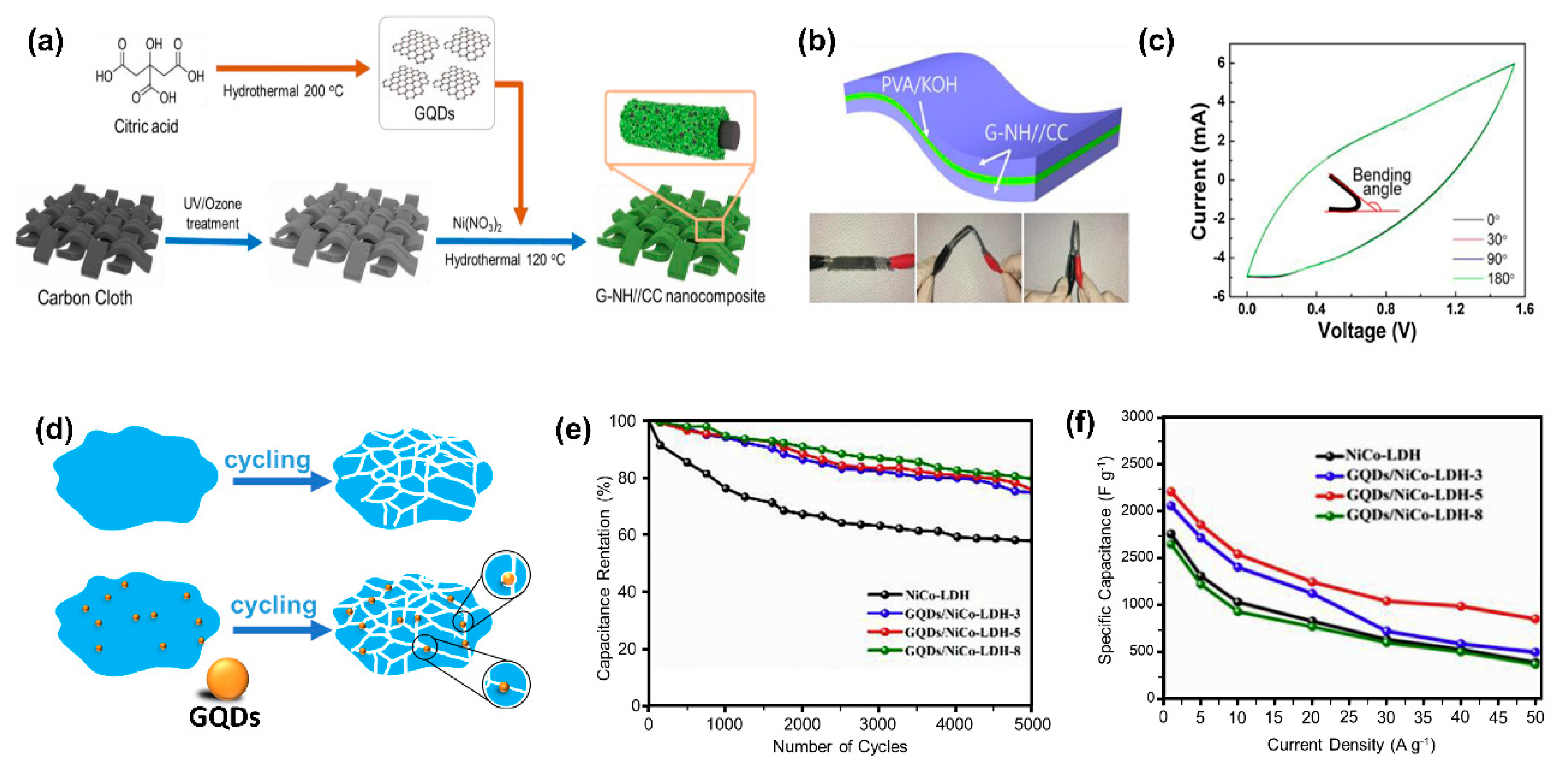
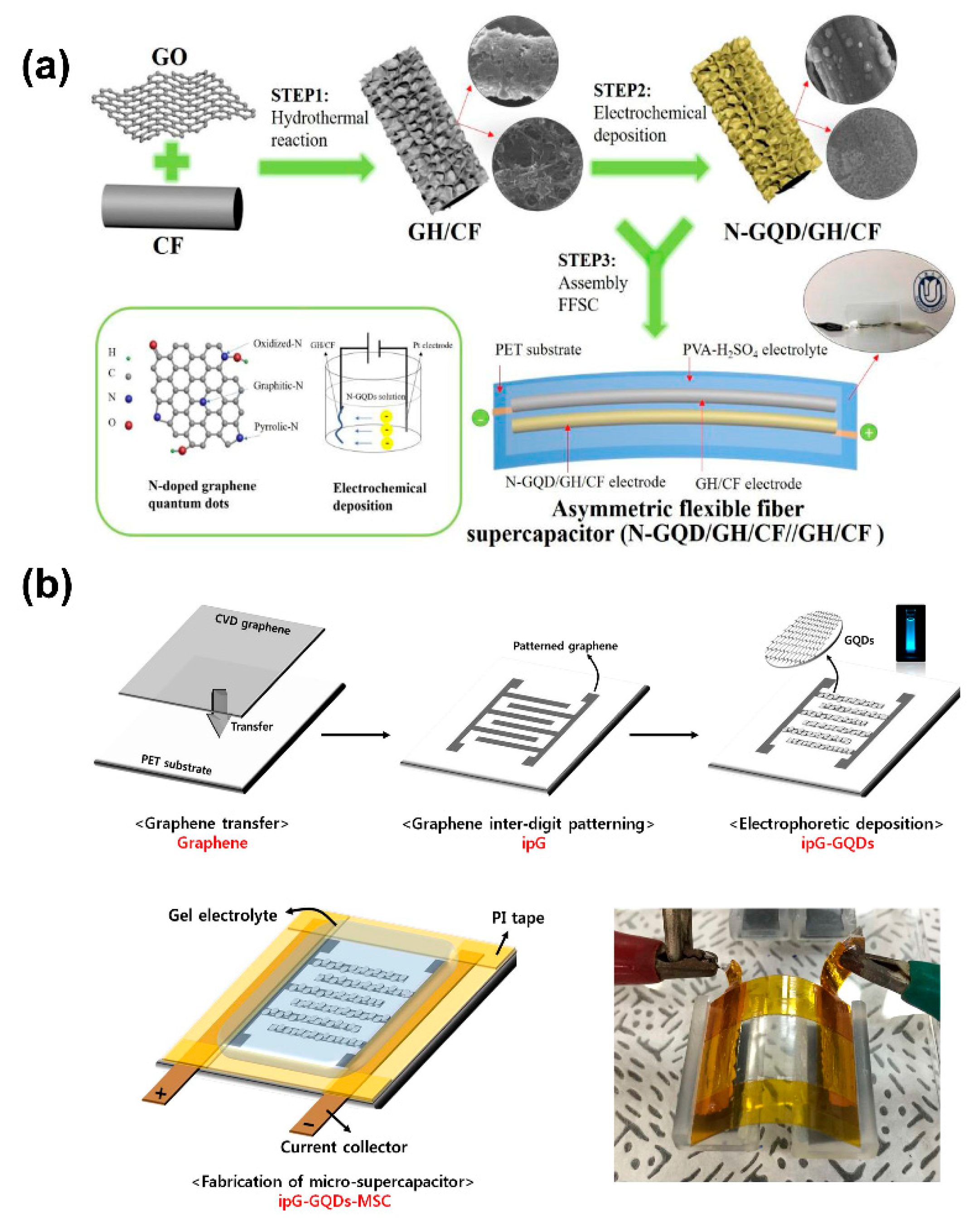
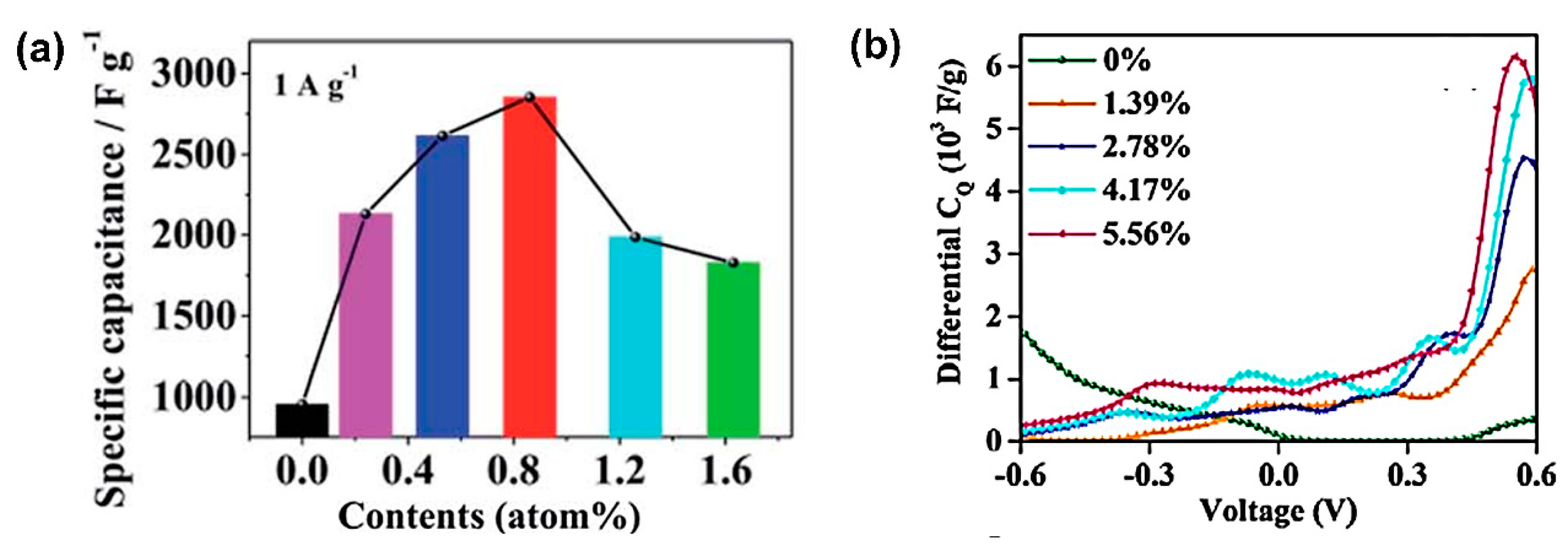
5.3. Graphene Quantum Dot Composites
6. Challenges and Future Perspectives
6.1. Wearable Supercapacitors
6.2. Quantum Capacitance Supercapacitor
6.3. Self-Charging Supercapacitor
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chen, D.; Wang, Q.; Wang, R.; Shen, G. Ternary oxide nanostructured materials for supercapacitors: A review. J. Mater. Chem. A 2015, 3, 10158–10173. [Google Scholar] [CrossRef]
- Meng, Q.; Cai, K.; Chen, Y.; Chen, L. Research progress on conducting polymer based supercapacitor electrode materials. Nano Energy 2017, 36, 268–285. [Google Scholar] [CrossRef]
- Sun, J.; Wu, C.; Sun, X.; Hu, H.; Zhi, C.; Hou, L.; Yuan, C. Recent progresses in high-energy-density all pseudocapacitive-electrode-materials-based asymmetric supercapacitors. J. Mater. Chem. A 2017, 5, 9443–9464. [Google Scholar] [CrossRef]
- Becker, H.I. Worldwide Applications. U.S. Patent 2,800,616, 23 July 1957. to General Electric. [Google Scholar]
- Winter, M.; Brodd, R.J. What Are Batteries, Fuel Cells, and Supercapacitors? Chem. Rev. 2004, 104, 4245–4270. [Google Scholar] [CrossRef]
- Kouchachvili, L.; Yaïci, W.; Entchev, E. Hybrid battery/supercapacitor energy storage system for the electric vehicles. J. Power Sources 2018, 374, 237–248. [Google Scholar] [CrossRef]
- Zhang, L.; Hu, X.; Wang, Z.; Sun, F.; Dorrell, D.G. A review of supercapacitor modeling, estimation, and applications: A control/management perspective. Renew. Sustain. Energy Rev. 2018, 81, 1868–1878. [Google Scholar] [CrossRef]
- Sharma, K.; Arora, A.; Tripathi, S.K. Review of supercapacitors: Materials and devices. J. Energy Storage 2019, 21, 801–825. [Google Scholar]
- Muzaffar, A.; Ahamed, M.B.; Deshmukh, K.; Thirumalai, J. A review on recent advances in hybrid supercapacitors: Design, fabrication and applications. Renew. Sustain. Energy Rev. 2019, 101, 123–145. [Google Scholar] [CrossRef]
- González, A.; Goikolea, E.; Barrena, J.A.; Mysyk, R. Review on supercapacitors: Technologies and materials. Renew. Sustain. Energy Rev. 2016, 58, 1189–1206. [Google Scholar] [CrossRef]
- Conway, B.E.; Supercapacitors, E. Scientific Fundamentals and Technological Applications; KluwerAcademic/Plenum Publisher: New York, NY, USA, 1999; p. 528. [Google Scholar]
- Zhong, C.; Deng, Y.; Hu, W.; Qiao, J.; Zhang, L.; Zhang, J. A review of electrolyte materials and compositions for electrochemical supercapacitors. Chem. Soc. Rev. 2015, 44, 7484–7539. [Google Scholar] [CrossRef]
- Chen, Z.; Wen, J.; Yan, C.; Rice, L.; Sohn, H.; Shen, M.; Cai, M.; Dunn, B.; Lu, Y. High-performance supercapacitors based on hierarchically porous graphite particles. Adv. Energy Mater. 2011, 1, 551–556. [Google Scholar] [CrossRef]
- Zhang, M.; He, L.; Shi, T.; Zha, R. Nanocasting and direct synthesis strategies for mesoporous carbons as supercapacitor electrodes. Chem. Mater. 2018, 30, 7391–7412. [Google Scholar] [CrossRef]
- Wu, Z.; Wang, D.; Ren, W.; Zhao, J.; Zhou, G.; Li, F.; Cheng, H. Anchoring hydrous RuO2 on graphene sheets for high-performance electrochemical capacitors. Adv. Funct. Mater. 2010, 20, 3595–3602. [Google Scholar] [CrossRef]
- Liu, S.; Yin, Y.; Wu, M.; Hui, K.S.; Hui, K.N.; Ouyang, C.; Jun, S.C. Phosphorus-Mediated MoS2 Nanowires as a High-Performance Electrode Material for Quasi-Solid-State Sodium-Ion Intercalation Supercapacitors. Small 2019, 15, 1803984. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Tetard, L.; Zhai, L.; Thomas, J. Supercapacitor electrode materials: Nanostructures from 0 to 3 dimensions. Energy Environ. Sci. 2015, 8, 702–730. [Google Scholar] [CrossRef]
- Bak, S.; Kim, D.; Lee, H. Graphene quantum dots and their possible energy applications: A review. Curr. Appl. Phys. 2016, 16, 1192–1201. [Google Scholar] [CrossRef]
- Lim, S.Y.; Shen, W.; Gao, Z. Carbon quantum dots and their applications. Chem. Soc. Rev. 2015, 44, 362–381. [Google Scholar] [CrossRef]
- Sun, Y.-P.; Zhou, B.; Lin, Y.; Wang, W.; Fernando, K.A.S.; Pathak, P.; Meziani, M.J.; Harruff, B.A.; Wang, X.; Wang, H. Quantum-sized carbon dots for bright and colorful photoluminescence. J. Am. Chem. Soc. 2006, 128, 7756–7757. [Google Scholar] [CrossRef]
- Wang, Y.; Hu, A. Carbon quantum dots: Synthesis, properties and applications. J. Mater. Chem. C 2014, 2, 6921–6939. [Google Scholar] [CrossRef]
- Habiba, K.; Makarov, V.I.; Avalos, J.; Guinel, M.J.F.; Weiner, B.R.; Morell, G. Luminescent graphene quantum dots fabricated by pulsed laser synthesis. Carbon N. Y. 2013, 64, 341–350. [Google Scholar] [CrossRef]
- Hu, C.; Li, M.; Qiu, J.; Sun, Y.-P. Design and fabrication of carbon dots for energy conversion and storage. Chem. Soc. Rev. 2019, 48, 2315–2337. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Rui, M.; Song, J.; Shen, Z.; Zeng, H. Carbon and graphene quantum dots for optoelectronic and energy devices: A review. Adv. Funct. Mater. 2015, 25, 4929–4947. [Google Scholar] [CrossRef]
- Yan, Y.; Gong, J.; Chen, J.; Zeng, Z.; Huang, W.; Pu, K.; Liu, J.; Chen, P. Recent advances on graphene quantum dots: From chemistry and physics to applications. Adv. Mater. 2019, 31, 1808283. [Google Scholar] [CrossRef] [PubMed]
- Dager, A.; Uchida, T.; Maekawa, T.; Tachibana, M. Synthesis and characterization of Mono-disperse carbon Quantum Dots from fennel Seeds: Photoluminescence analysis using Machine Learning. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Yuan, F.; Li, S.; Fan, Z.; Meng, X.; Fan, L.; Yang, S. Shining carbon dots: Synthesis and biomedical and optoelectronic applications. Nano Today 2016, 11, 565–586. [Google Scholar] [CrossRef]
- Yang, S.; Sun, J.; Li, X.; Zhou, W.; Wang, Z.; He, P.; Ding, G.; Xie, X.; Kang, Z.; Jiang, M. Large-scale fabrication of heavy doped carbon quantum dots with tunable-photoluminescence and sensitive fluorescence detection. J. Mater. Chem. A 2014, 2, 8660–8667. [Google Scholar] [CrossRef]
- Bisri, S.Z.; Shimizu, S.; Nakano, M.; Iwasa, Y. Endeavor of Iontronics: From Fundamentals to Applications of Ion-Controlled Electronics. Adv. Mater. 2017, 29, 1607054. [Google Scholar] [CrossRef]
- Helmholtz, H.V. Studien über electrische Grenzschichten. Annalen der Physik 1879, 243, 337–382. [Google Scholar] [CrossRef]
- Gouy, M. Sur La Constitution De La Charge Électrique À La Surface D’un Électrolyte. J. Phys. Theor. Appl. 1910, 9, 457–468. [Google Scholar] [CrossRef]
- Chapman, D.L. A Contribution to the Theory of Electrocapillarity. Lond. Edinb. Dublin Philos. Mag. J. Sci. 1913, 25, 475–481. [Google Scholar] [CrossRef]
- Stern, O. Zur Theorie der Elektrolytischen Doppelschicht. Z. Elektrochem. Angew. Phys. Chemie 1924, 30, 508–516. [Google Scholar]
- Dai, G.; Zhang, L.; Liao, Y.; Shi, Y.; Xie, J.; Lei, F.; Fan, L. Multi-Scale Model for Describing the Effect of Pore Structure on Carbon-Based Electric Double Layer. J. Phys. Chem. C 2020, 124, 3952–3961. [Google Scholar] [CrossRef]
- Hsieh, W.; Horng, T.-L.A.; Huang, H.-C.; Teng, H. Facile simulation of carbon with wide pore size distribution for electric double-layer capacitance based on Helmholtz models. J. Mater. Chem. 2015, 3, 16535–16543. [Google Scholar] [CrossRef]
- Huang, J.; Sumpter, B.G.; Meunier, V. Theoretical model for nanoporous carbon supercapacitors. Angew. Chemie Int. Ed. 2008, 47, 520–524. [Google Scholar] [CrossRef]
- Bandaru, P.R.; Yamada, H.; Narayanan, R.; Hoefer, M. Charge transfer and storage in nanostructures. Mater. Sci. Eng. R Rep. 2015, 96, 1–69. [Google Scholar] [CrossRef]
- Mei, B.-A.; Munteshari, O.; Lau, J.; Dunn, B.; Pilon, L. Physical interpretations of Nyquist plots for EDLC electrodes and devices. J. Phys. Chem. C 2018, 122, 194–206. [Google Scholar] [CrossRef]
- Stoller, M.D.; Park, S.; Zhu, Y.; An, J.; Ruoff, R.S. Graphene-based ultracapacitors. Nano Lett. 2008, 8, 3498–3502. [Google Scholar] [CrossRef]
- Peng, X.; Peng, L.; Wu, C.; Xie, Y. Two dimensional nanomaterials for flexible supercapacitors. Chem. Soc. Rev. 2014, 43, 3303–3323. [Google Scholar] [CrossRef]
- Xiao, X.; Beechem, T.E.; Brumbach, M.T.; Lambert, T.N.; Davis, D.J.; Michael, J.R.; Washburn, C.M.; Wang, J.; Brozik, S.M.; Wheeler, D.R. Lithographically defined three-dimensional graphene structures. ACS Nano 2012, 6, 3573–3579. [Google Scholar] [CrossRef]
- Sun, J.; Iakunkov, A.; Rebrikova, A.T.; Talyzin, A. V Exactly matched pore size for the intercalation of electrolyte ions determined using the tunable swelling of graphite oxide in supercapacitor electrodes. Nanoscale 2018, 10, 21386–21395. [Google Scholar] [CrossRef]
- Na, W.; Jun, J.; Park, J.W.; Lee, G.; Jang, J. Highly porous carbon nanofibers co-doped with fluorine and nitrogen for outstanding supercapacitor performance. J. Mater. Chem. A 2017, 5, 17379–17387. [Google Scholar] [CrossRef]
- Kou, T.; Yao, B.; Liu, T.; Li, Y. Recent advances in chemical methods for activating carbon and metal oxide based electrodes for supercapacitors. J. Mater. Chem. A 2017, 5, 17151–17173. [Google Scholar] [CrossRef]
- Purkait, T.; Singh, G.; Singh, M.; Kumar, D.; Dey, R.S. Large area few-layer graphene with scalable preparation from waste biomass for high-performance supercapacitor. Sci. Rep. 2017, 7, 15239. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.-H. The Influence of the interlayer distance on the performance of thermally reduced graphene oxide supercapacitors. Materials 2018, 11, 263. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, Z.; Lei, Y.; Chen, Y.; Zhang, Z.; Jiang, Z.; Hu, J.; Lin, Y. Preparation and Specific Capacitance Properties of Sulfur, Nitrogen Co-Doped Graphene Quantum Dots. Nanoscale Res. Lett. 2019, 14, 219. [Google Scholar] [CrossRef]
- Hasegawa, G.; Deguchi, T.; Kanamori, K.; Kobayashi, Y.; Kageyama, H.; Abe, T.; Nakanishi, K. High-Level Doping of Nitrogen, Phosphorus, and Sulfur into Activated Carbon Monoliths and Their Electrochemical Capacitances. Chem. Mater. 2015, 27, 4703–4712. [Google Scholar] [CrossRef]
- Jiang, Y.; Liu, J. Definitions of pseudocapacitive materials: A brief review. Energy Environ. Mater. 2019, 2, 30–37. [Google Scholar] [CrossRef]
- Long, J.W.; Bélanger, D.; Brousse, T.; Sugimoto, W.; Sassin, M.B.; Crosnier, O. Asymmetric electrochemical capacitors—Stretching the limits of aqueous electrolytes. Mrs Bull. 2011, 36, 513–522. [Google Scholar] [CrossRef]
- Lee, J.-S.M.; Briggs, M.E.; Hu, C.-C.; Cooper, A.I. Controlling electric double-layer capacitance and pseudocapacitance in heteroatom-doped carbons derived from hypercrosslinked microporous polymers. Nano Energy 2018, 46, 277–289. [Google Scholar] [CrossRef]
- Augustyn, V.; Simon, P.; Dunn, B. Pseudocapacitive oxide materials for high-rate electrochemical energy storage. Energy Environ. Sci. 2014, 7, 1597–1614. [Google Scholar] [CrossRef]
- Allagui, A.; Freeborn, T.J.; Elwakil, A.S.; Maundy, B.J. Reevaluation of Performance of Electric Double-layer Capacitors from Constant-current Charge/Discharge and Cyclic Voltammetry. Sci. Rep. 2016, 6, 38568. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Qi, L.; Lu, L.; Wang, H. Facile preparation and performance of mesoporous manganese oxide for supercapacitors utilizing neutral aqueous electrolytes. RSC Adv. 2012, 2, 3298–3308. [Google Scholar] [CrossRef]
- Kwon, H.-N.; Jang, S.-J.; Kang, Y.C.; Roh, K.C. The effect of ILs as co-salts in electrolytes for high voltage supercapacitors. Sci. Rep. 2019, 9, 1180. [Google Scholar] [CrossRef] [PubMed]
- Chi, K.; Zhang, Z.; Lv, Q.; Xie, C.; Xiao, J.; Xiao, F.; Wang, S. Well-ordered oxygen-deficient CoMoO4 and Fe2O3 nanoplate arrays on 3D graphene foam: Toward flexible asymmetric supercapacitors with enhanced capacitive properties. ACS Appl. Mater. Interfaces 2017, 9, 6044–6053. [Google Scholar] [CrossRef]
- Patil, A.M.; Lokhande, A.C.; Chodankar, N.R.; Kumbhar, V.S.; Lokhande, C.D. Engineered morphologies of β-NiS thin films via anionic exchange process and their supercapacitive performance. Mater. Des. 2016, 97, 407–416. [Google Scholar] [CrossRef]
- Li, Z.; Zhao, D.; Xu, C.; Ning, J.; Zhong, Y.; Zhang, Z.; Wang, Y.; Hu, Y. Reduced CoNi2S4 nanosheets with enhanced conductivity for high-performance supercapacitors. Electrochim. Acta 2018, 278, 33–41. [Google Scholar] [CrossRef]
- Dhibar, S.; Bhattacharya, P.; Hatui, G.; Sahoo, S.; Das, C.K. Transition metal-doped polyaniline/single-walled carbon nanotubes nanocomposites: Efficient electrode material for high performance supercapacitors. ACS Sustain. Chem. Eng. 2014, 2, 1114–1127. [Google Scholar] [CrossRef]
- Okubo, M.; Sugahara, A.; Kajiyama, S.; Yamada, A. MXene as a charge storage host. Acc. Chem. Res. 2018, 51, 591–599. [Google Scholar] [CrossRef]
- Kim, J.; Young, C.; Lee, J.; Heo, Y.-U.; Park, M.-S.; Hossain, M.S.A.; Yamauchi, Y.; Kim, J.H. Nanoarchitecture of MOF-derived nanoporous functional composites for hybrid supercapacitors. J. Mater. Chem. A 2017, 5, 15065–15072. [Google Scholar] [CrossRef]
- Abdah, M.A.A.M.; Azman, N.H.N.; Kulandaivalu, S.; Sulaiman, Y. Review of the use of transition-metal-oxide and conducting polymer-based fibres for high-performance supercapacitors. Mater. Des. 2020, 186, 108199. [Google Scholar] [CrossRef]
- Sari, F.N.I.; So, P.; Ting, J. MnO2 with controlled phase for use in supercapacitors. J. Am. Ceram. Soc. 2017, 100, 1642–1652. [Google Scholar] [CrossRef]
- Murugesan, D.; Prakash, S.; Ponpandian, N.; Manisankar, P.; Viswanathan, C. Two dimensional α-MoO3 nanosheets decorated carbon cloth electrodes for high-performance supercapacitors. Colloids Surfaces A Physicochem. Eng. Asp. 2019, 569, 137–144. [Google Scholar] [CrossRef]
- Borenstein, A.; Hanna, O.; Attias, R.; Luski, S.; Brousse, T.; Aurbach, D. Carbon-based composite materials for supercapacitor electrodes: A review. J. Mater. Chem. A 2017, 5, 12653–12672. [Google Scholar] [CrossRef]
- Zhang, X.; Samorì, P. Graphene/polymer nanocomposites for supercapacitors. ChemNanoMat 2017, 3, 362–372. [Google Scholar] [CrossRef]
- Sahoo, S.; Satpati, A.K.; Sahoo, P.K.; Naik, P.D. Incorporation of Carbon Quantum Dots for Improvement of Supercapacitor Performance of Nickel Sulfide. ACS Omega 2018, 3, 17936–17946. [Google Scholar] [CrossRef]
- Xia, J.; Chen, F.; Li, J.; Tao, N. Measurement of the quantum capacitance of graphene. Nat. Nanotechnol. 2009, 4, 505–509. [Google Scholar] [CrossRef]
- Kim, C.-H.; Frisbie, C.D. Determination of quantum capacitance and band filling potential in Graphene transistors with dual Electrochemical and field-effect gates. J. Phys. Chem. C 2014, 118, 21160–21169. [Google Scholar] [CrossRef]
- Septianto, R.D.; Liu, L.; Iskandar, F.; Matsushita, N.; Iwasa, Y.; Bisri, S.Z. On-demand tuning of charge accumulation and carrier mobility in quantum dot solids for electron transport and energy storage devices. NPG Asia Mater. 2020, 12, 33. [Google Scholar] [CrossRef]
- Zhan, C.; Lian, C.; Zhang, Y.; Thompson, M.W.; Xie, Y.; Wu, J.; Kent, P.R.C.; Cummings, P.T.; Jiang, D.; Wesolowski, D.J. Computational insights into materials and interfaces for capacitive energy storage. Adv. Sci. 2017, 4, 1700059. [Google Scholar] [CrossRef]
- Cayuela, A.; Soriano, M.L.; Carrillo-Carrion, C.; Valcarcel, M. Semiconductor and carbon-based fluorescent nanodots: The need for consistency. Chem. Commun. 2016, 52, 1311–1326. [Google Scholar] [CrossRef]
- Essner, J.B.; Baker, G.A. The emerging roles of carbon dots in solar photovoltaics: A critical review. Environ. Sci. Nano 2017, 4, 1216–1263. [Google Scholar] [CrossRef]
- Zhu, S.; Song, Y.; Zhao, X.; Shao, J.; Zhang, J.; Yang, B. The photoluminescence mechanism in carbon dots (graphene quantum dots, carbon nanodots, and polymer dots): Current state and future perspective. Nano Res. 2015, 8, 355–381. [Google Scholar] [CrossRef]
- Wang, B.; Yu, J.; Sui, L.; Zhu, S.; Tang, Z.; Yang, B.; Lu, S. Rational Design of Multi-Color-Emissive Carbon Dots in a Single Reaction System by Hydrothermal. Adv. Sci. 2020, 2001453. [Google Scholar] [CrossRef]
- Tepliakov, N.V.; Kundelev, E.V.; Khavlyuk, P.D.; Xiong, Y.; Leonov, M.Y.; Zhu, W.; Baranov, A.V.; Fedorov, A.V.; Rogach, A.L.; Rukhlenko, I.D. sp2–sp3-Hybridized Atomic Domains Determine Optical Features of Carbon Dots. ACS Nano 2019, 13, 10737–10744. [Google Scholar] [CrossRef]
- Zhou, J.; Booker, C.; Li, R.; Zhou, X.; Sham, T.K.; Sun, X.; Ding, Z. An electrochemical avenue to blue luminescent nanocrystals from multiwalled carbon nanotubes (MWCNTs). J. Am. Chem. Soc. 2007, 129, 744–745. [Google Scholar] [CrossRef]
- Chen, G.; Wu, S.; Hui, L.; Zhao, Y.; Ye, J.; Tan, Z.; Zeng, W.; Tao, Z.; Yang, L.; Zhu, Y. Assembling carbon quantum dots to a layered carbon for high-density supercapacitor electrodes. Sci. Rep. 2016, 6, 19028. [Google Scholar] [CrossRef]
- Liu, Y.; Li, W.; Wu, P.; Ma, C.; Wu, X.; Xu, M.; Luo, S.; Xu, Z.; Liu, S. Hydrothermal synthesis of nitrogen and boron co-doped carbon quantum dots for application in acetone and dopamine sensors and multicolor cellular imaging. Sens. Actuators B Chem. 2019, 281, 34–43. [Google Scholar] [CrossRef]
- Li, M.; Chen, T.; Gooding, J.J.; Liu, J. Review of carbon and graphene quantum dots for sensing. ACS Sens. 2019, 4, 1732–1748. [Google Scholar] [CrossRef]
- Permatasari, F.A.; Aimon, A.H.; Iskandar, F.; Ogi, T.; Okuyama, K. Role of C–N configurations in the photoluminescence of graphene quantum dots synthesized by a hydrothermal route. Sci. Rep. 2016, 6, 21042. [Google Scholar] [CrossRef]
- Moghimian, S.; Sangpour, P. One-step hydrothermal synthesis of GQDs-MoS2 nanocomposite with enhanced supercapacitive performance. J. Appl. Electrochem. 2020, 50, 71–79. [Google Scholar] [CrossRef]
- Eda, G.; Lin, Y.; Mattevi, C.; Yamaguchi, H.; Chen, H.; Chen, I.; Chen, C.; Chhowalla, M. Blue photoluminescence from chemically derived graphene oxide. Adv. Mater. 2010, 22, 505–509. [Google Scholar] [CrossRef] [PubMed]
- Ogi, T.; Iwasaki, H.; Aishima, K.; Iskandar, F.; Wang, W.-N.; Takimiya, K.; Okuyama, K. Transient nature of graphene quantum dot formation via a hydrothermal reaction. RSC Adv. 2014, 4, 55709–55715. [Google Scholar] [CrossRef]
- Yuan, W.; Zhou, Y.; Li, Y.; Li, C.; Peng, H.; Zhang, J.; Liu, Z.; Dai, L.; Shi, G. The edge-and basal-plane-specific electrochemistry of a single-layer graphene sheet. Sci. Rep. 2013, 3, 2248. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Shim, J.; Rivera, J.; Jin, X.; Estrada, D.; Solovyeva, V.; You, X.; Pak, J.; Pop, E.; Aluru, N. Electrochemistry at the edge of a single graphene layer in a nanopore. ACS Nano 2013, 7, 834–843. [Google Scholar] [CrossRef] [PubMed]
- Kalita, H.; Shinde, D.B.; Pillai, V.K.; Aslam, M. Hysteresis and charge trapping in graphene quantum dots. Appl. Phys. Lett. 2013, 102, 143104. [Google Scholar] [CrossRef]
- Zhou, G.; Wang, D.-W.; Yin, L.-C.; Li, N.; Li, F.; Cheng, H.-M. Oxygen bridges between NiO nanosheets and graphene for improvement of lithium storage. ACS Nano 2012, 6, 3214–3223. [Google Scholar] [CrossRef]
- Shen, J.; Zhu, Y.; Yang, X.; Li, C. Graphene quantum dots: Emergent nanolights for bioimaging, sensors, catalysis and photovoltaic devices. Chem. Commun. 2012, 48, 3686–3699. [Google Scholar] [CrossRef]
- Chen, W.; Lv, G.; Hu, W.; Li, D.; Chen, S.; Dai, Z. Synthesis and applications of graphene quantum dots: A review. Nanotechnol. Rev. 2018, 7, 157–185. [Google Scholar] [CrossRef]
- Wu, J.; Pisula, W.; Müllen, K. Graphenes as potential material for electronics. Chem. Rev. 2007, 107, 718–747. [Google Scholar] [CrossRef]
- Lv, H.; Gao, X.; Xu, Q.; Liu, H.; Wang, Y.-G.; Xia, Y. Carbon quantum dot-induced MnO2 nanowire formation and construction of a binder-free flexible membrane with excellent superhydrophilicity and enhanced supercapacitor performance. ACS Appl. Mater. Interfaces 2017, 9, 40394–40403. [Google Scholar] [CrossRef]
- Li, Z.; Qin, P.; Wang, L.; Yang, C.; Li, Y.; Chen, Z.; Pan, D.; Wu, M. Amine-enriched graphene quantum dots for high-pseudocapacitance supercapacitors. Electrochim. Acta 2016, 208, 260–266. [Google Scholar] [CrossRef]
- Qie, L.; Chen, W.; Xu, H.; Xiong, X.; Jiang, Y.; Zou, F.; Hu, X.; Xin, Y.; Zhang, Z.; Huang, Y. Synthesis of functionalized 3D hierarchical porous carbon for high-performance supercapacitors. Energy Environ. Sci. 2013, 6, 2497–2504. [Google Scholar] [CrossRef]
- Li, L.; Dong, T. Photoluminescence tuning in carbon dots: Surface passivation or/and functionalization, heteroatom doping. J. Mater. Chem. C 2018, 6, 7944–7970. [Google Scholar] [CrossRef]
- Yan, X.; Cui, X.; Li, L. Synthesis of large, stable colloidal graphene quantum dots with tunable size. J. Am. Chem. Soc. 2010, 132, 5944–5945. [Google Scholar] [CrossRef]
- Tian, P.; Tang, L.; Teng, K.S.; Lau, S.P. Graphene quantum dots from chemistry to applications. Mater. Today Chem. 2018, 10, 221–258. [Google Scholar] [CrossRef]
- Zheng, C.; Qian, W.; Cui, C.; Zhang, Q.; Jin, Y.; Zhao, M.; Tan, P.; Wei, F. Hierarchical carbon nanotube membrane with high packing density and tunable porous structure for high voltage supercapacitors. Carbon N. Y. 2012, 50, 5167–5175. [Google Scholar] [CrossRef]
- Fan, Z.; Yan, J.; Zhi, L.; Zhang, Q.; Wei, T.; Feng, J.; Zhang, M.; Qian, W.; Wei, F. A three-dimensional carbon nanotube/graphene sandwich and its application as electrode in supercapacitors. Adv. Mater. 2010, 22, 3723–3728. [Google Scholar] [CrossRef]
- Ma, Y.; Yuan, W.; Bai, Y.; Wu, H.; Cheng, L. The toughening design of pseudocapacitive materials via graphene quantum dots: Towards enhanced cycling stability for supercapacitors. Carbon N. Y. 2019, 154, 292–300. [Google Scholar] [CrossRef]
- Kwon, W.; Do, S.; Won, D.C.; Rhee, S.-W. Carbon quantum dot-based field-effect transistors and their ligand length-dependent carrier mobility. ACS Appl. Mater. Interfaces 2013, 5, 822–827. [Google Scholar] [CrossRef]
- Zhang, R.Q.; Bertran, E.; Lee, S.-T. Size dependence of energy gaps in small carbon clusters: The origin of broadband luminescence. Diam. Relat. Mater. 1998, 7, 1663–1668. [Google Scholar] [CrossRef]
- Xu, Q.; Li, W.; Ding, L.; Yang, W.; Xiao, H.; Ong, W.-J. Function-driven engineering of 1D carbon nanotubes and 0D carbon dots: Mechanism, properties and applications. Nanoscale 2019, 11, 1475–1504. [Google Scholar] [CrossRef] [PubMed]
- Calabro, R.L.; Yang, D.S.; Kim, D.Y. Controlled Nitrogen Doping of Graphene Quantum Dots through Laser Ablation in Aqueous Solutions for Photoluminescence and Electrocatalytic Applications. ACS Appl. Nano Mater. 2019, 2, 6948–6959. [Google Scholar] [CrossRef]
- Kuo, N.J.; Chen, Y.S.; Wu, C.W.; Huang, C.Y.; Chan, Y.H.; Chen, I.W.P. One-Pot Synthesis of Hydrophilic and Hydrophobic N-Doped Graphene Quantum Dots via Exfoliating and Disintegrating Graphite Flakes. Sci. Rep. 2016, 6, 30426. [Google Scholar] [CrossRef] [PubMed]
- Ahirwar, S.; Mallick, S.; Bahadur, D. Electrochemical Method to Prepare Graphene Quantum Dots and Graphene Oxide Quantum Dots. ACS Omega 2017, 2, 8343–8353. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Jeong, Y.K.; Ryu, J.H.; Son, Y.; Kim, W.R.; Lee, B.; Jung, K.H.; Kim, K.M. Pulsed laser ablation based synthetic route for nitrogen-doped graphene quantum dots using graphite flakes. Appl. Surf. Sci. 2020, 506, 144998. [Google Scholar] [CrossRef]
- Kang, S.; Jeong, Y.K.; Jung, K.H.; Son, Y.; Kim, W.R.; Ryu, J.H.; Kim, K.M. One-step synthesis of sulfur-incorporated graphene quantum dots using pulsed laser ablation for enhancing optical properties. Opt. Express 2020, 28, 21659. [Google Scholar] [CrossRef] [PubMed]
- Iskandar, F.; Hikmah, U.; Stavila, E.; Aimon, A.H. Microwave-assisted reduction method under nitrogen atmosphere for synthesis and electrical conductivity improvement of reduced graphene oxide (rGO). RSC Adv. 2017, 7, 52391–52397. [Google Scholar] [CrossRef]
- Iskandar, F.; Abdillah, O.B.; Stavila, E.; Aimon, A.H. The influence of copper addition on the electrical conductivity and charge transfer resistance of reduced graphene oxide (rGO). New J. Chem. 2018, 42, 16362–16371. [Google Scholar] [CrossRef]
- Hess, S.C.; Permatasari, F.A.; Fukazawa, H.; Schneider, E.M.; Balgis, R.; Ogi, T.; Okuyama, K.; Stark, W.J. Direct synthesis of carbon quantum dots in aqueous polymer solution: One-pot reaction and preparation of transparent UV-blocking films. J. Mater. Chem. A 2017, 5, 5187–5194. [Google Scholar] [CrossRef]
- Lai, C.W.; Hsiao, Y.H.; Peng, Y.K.; Chou, P.T. Facile synthesis of highly emissive carbon dots from pyrolysis of glycerol; Gram scale production of carbon dots/mSiO 2 for cell imaging and drug release. J. Mater. Chem. 2012, 22, 14403–14409. [Google Scholar] [CrossRef]
- Ogi, T.; Aishima, K.; Permatasari, F.A.; Iskandar, F.; Tanabe, E.; Okuyama, K. Kinetics of nitrogen-doped carbon dot formation via hydrothermal synthesis. New J. Chem. 2016, 40, 5555–5561. [Google Scholar] [CrossRef]
- Permatasari, F.A.; Fukazawa, H.; Ogi, T.; Iskandar, F.; Okuyama, K. Design of pyrrolic-N-rich carbon dots with absorption in the first near-infrared window for photothermal therapy. ACS Appl. Nano Mater. 2018, 1, 2368–2375. [Google Scholar] [CrossRef]
- Li, Q.; Cheng, H.; Wu, X.; Wang, C.-F.; Wu, G.; Chen, S. Enriched carbon dots/graphene microfibers towards high-performance micro-supercapacitors. J. Mater. Chem. A 2018, 6, 14112–14119. [Google Scholar] [CrossRef]
- Zhang, L.L.; Zhao, X.; Ji, H.; Stoller, M.D.; Lai, L.; Murali, S.; Mcdonnell, S.; Cleveger, B.; Wallace, R.M.; Ruoff, R.S.; et al. Nitrogen doping of graphene and its effect on quantum capacitance, and a new insight on the enhanced capacitance of N-doped. Carbon 2012, 5, 9618–9625. [Google Scholar] [CrossRef]
- Liu, W.; Feng, Y.; Yan, X.; Chen, J.; Xue, Q. Superior micro-supercapacitors based on graphene quantum dots. Adv. Funct. Mater. 2013, 23, 4111–4122. [Google Scholar] [CrossRef]
- Hassan, M.; Haque, E.; Reddy, K.R.; Minett, A.I.; Chen, J.; Gomes, V.G. Edge-enriched graphene quantum dots for enhanced photo-luminescence and supercapacitance. Nanoscale 2014, 6, 11988–11994. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Sui, L.; Dong, H.; He, W.; Dong, L.; Yu, L. High-performance supercapacitor of graphene quantum dots with uniform sizes. ACS Appl. Mater. Interfaces 2018, 10, 12983–12991. [Google Scholar] [CrossRef]
- Zhang, S.; Li, Y.; Song, H.; Chen, X.; Zhou, J.; Hong, S.; Huang, M. Graphene quantum dots as the electrolyte for solid state supercapacitors. Sci. Rep. 2016, 6, 19292. [Google Scholar] [CrossRef]
- Li, Z.; Bu, F.; Wei, J.; Yao, W.; Wang, L.; Chen, Z.; Pan, D.; Wu, M. Boosting the energy storage densities of supercapacitors by incorporating N-doped graphene quantum dots into cubic porous carbon. Nanoscale 2018, 10, 22871–22883. [Google Scholar] [CrossRef]
- Li, Z.; Wei, J.; Ren, J.; Wu, X.; Wang, L.; Pan, D.; Wu, M. Hierarchical construction of high-performance all-carbon flexible fiber supercapacitors with graphene hydrogel and nitrogen-doped graphene quantum dots. Carbon N. Y. 2019, 154, 410–419. [Google Scholar] [CrossRef]
- Qing, Y.; Jiang, Y.; Lin, H.; Wang, L.; Liu, A.; Cao, Y.; Sheng, R.; Guo, Y.; Fan, C.; Zhang, S. Boosting the supercapacitor performance of activated carbon by constructing overall conductive networks using graphene quantum dots. J. Mater. Chem. A 2019, 7, 6021–6027. [Google Scholar] [CrossRef]
- Wei, J.-S.; Song, T.-B.; Zhang, P.; Zhu, Z.-Y.; Dong, X.-Y.; Niu, X.-Q.; Xiong, H.-M. Integrating Carbon Dots with Porous Hydrogels to Produce Full Carbon Electrodes for Electric Double-Layer Capacitors. ACS Appl. Energy Mater. 2020, 3, 6907–6914. [Google Scholar] [CrossRef]
- Tjandra, R.; Liu, W.; Zhang, M.; Yu, A. All-carbon flexible supercapacitors based on electrophoretic deposition of graphene quantum dots on carbon cloth. J. Power Sources 2019, 438, 227009. [Google Scholar] [CrossRef]
- Li, Z.; Li, Y.; Wang, L.; Cao, L.; Liu, X.; Chen, Z.; Pan, D.; Wu, M. Assembling nitrogen and oxygen co-doped graphene quantum dots onto hierarchical carbon networks for all-solid-state flexible supercapacitors. Electrochim. Acta 2017, 235, 561–569. [Google Scholar] [CrossRef]
- Zhang, S.; Zhu, J.; Qing, Y.; Wang, L.; Zhao, J.; Li, J.; Tian, W.; Jia, D.; Fan, Z. Ultramicroporous carbons puzzled by graphene quantum dots: Integrated high gravimetric, volumetric, and areal capacitances for supercapacitors. Adv. Funct. Mater. 2018, 28, 1805898. [Google Scholar] [CrossRef]
- Luo, P.; Guan, X.; Yu, Y.; Li, X.; Yan, F. Hydrothermal synthesis of graphene quantum dots supported on three-dimensional graphene for supercapacitors. Nanomaterials 2019, 9, 201. [Google Scholar] [CrossRef]
- Tian, W.; Zhu, J.; Dong, Y.; Zhao, J.; Li, J.; Guo, N.; Lin, H.; Zhang, S.; Jia, D. Micelle-induced assembly of graphene quantum dots into conductive porous carbon for high rate supercapacitor electrodes at high mass loadings. Carbon 2020, 161, 89–96. [Google Scholar] [CrossRef]
- Xu, L.; Cheng, C.; Yao, C.; Jin, X. Flexible supercapacitor electrode based on lignosulfonate-derived graphene quantum dots/graphene hydrogel. Org. Electron. 2020, 78, 105407. [Google Scholar] [CrossRef]
- Theerthagiri, J.; Karuppasamy, K.; Durai, G.; Rana, A.U.H.S.; Arunachalam, P.; Sangeetha, K.; Kuppusami, P.; Kim, H.-S. Recent advances in metal chalcogenides (MX.; X= S, Se) nanostructures for electrochemical supercapacitor applications: A brief review. Nanomaterials 2018, 8, 256. [Google Scholar] [CrossRef]
- Wang, J.; Fang, Z.; Li, T.; ur Rehman, S.; Luo, Q.; Chen, P.; Hu, L.; Zhang, F.; Wang, Q.; Bi, H. Highly Hydrophilic Carbon Dots’ Decoration on NiCo2O4 Nanowires for Greatly Increased Electric Conductivity, Supercapacitance, and Energy Density. Adv. Mater. Interfaces 2019, 6, 1900049. [Google Scholar] [CrossRef]
- Wei, J.S.; Ding, H.; Zhang, P.; Song, Y.F.; Chen, J.; Wang, Y.G.; Xiong, H.M. Carbon Dots/NiCo2O4Nanocomposites with Various Morphologies for High Performance Supercapacitors. Small 2016, 12, 5927–5934. [Google Scholar] [CrossRef] [PubMed]
- Kandra, R.; Bajpai, S. Synthesis, mechanical properties of fluorescent carbon dots loaded nanocomposites chitosan film for wound healing and drug delivery. Arab. J. Chem. 2020, 13, 4882–4894. [Google Scholar] [CrossRef]
- Ji, Z.; Liu, K.; Li, N.; Zhang, H.; Dai, W.; Shen, X.; Zhu, G.; Kong, L.; Yuan, A. Nitrogen-doped carbon dots anchored NiO/Co3O4 ultrathin nanosheets as advanced cathodes for hybrid supercapacitors. J. Colloid Int. Sci. 2020, 579, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Ji, Z.; Dai, W.; Zhang, S.; Wang, G.; Shen, X.; Liu, K.; Zhu, G.; Kong, L.; Zhu, J. Bismuth oxide/nitrogen-doped carbon dots hollow and porous hierarchitectures for high-performance asymmetric supercapacitors. Adv. Powder Tech. 2020, 31, 632–638. [Google Scholar] [CrossRef]
- Narayanan, R. Single step hydrothermal synthesis of carbon nanodot decorated V2O5 nanobelts as hybrid conducting material for supercapacitor application. J. Solid State Chem. 2017, 253, 103–112. [Google Scholar] [CrossRef]
- Kumar, V.B.; Borenstein, A.; Markovsky, B.; Aurbach, D.; Gedanken, A.; Talianker, M.; Porat, Z. Activated carbon modified with carbon nanodots as novel electrode material for supercapacitors. J. Phys. Chem. C 2016, 120, 13406–13413. [Google Scholar] [CrossRef]
- Ji, Z.; Li, N.; Xie, M.; Shen, X.; Dai, W.; Liu, K.; Xu, K.; Zhu, G. High-performance hybrid supercapacitor realized by nitrogen-doped carbon dots modified cobalt sulfide and reduced graphene oxide. Electrochim. Acta 2020, 334, 135632. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, J.; Liu, J.; Wu, J.; Chen, H.; Bi, H. Design and preparation of a ternary composite of graphene oxide/carbon dots/polypyrrole for supercapacitor application: Importance and unique role of carbon dots. Carbon N. Y. 2017, 115, 134–146. [Google Scholar] [CrossRef]
- Moreno Araújo Pinheiro Lima, R.; de Oliveira, H.P. Carbon dots reinforced polypyrrole/graphene nanoplatelets on flexible eggshell membranes as electrodes of all-solid flexible supercapacitors. J. Energy Storage 2020, 28, 101284. [Google Scholar] [CrossRef]
- Zhao, T.; Peng, X.; Zhao, X.; Hu, J.; Jiang, T.; Lu, X.; Zhang, H.; Li, T.; Ahmad, I. Compounds Preparation and performance of carbon dot decorated copper sulphide/carbon nanotubes hybrid composite as supercapacitor electrode materials. J. Alloys Compd. 2020, 817, 153057. [Google Scholar] [CrossRef]
- Zhu, Y.; Wu, Z.; Jing, M.; Hou, H.; Yang, Y.; Zhang, Y.; Yang, X.; Song, W.; Jia, X.; Ji, X. Porous NiCo2O4 spheres tuned through carbon quantum dots utilised as advanced materials for an asymmetric supercapacitor. J. Mater. Chem. A 2015, 3, 866–877. [Google Scholar] [CrossRef]
- Zhu, Y.; Ji, X.; Pan, C.; Sun, Q.; Song, W.; Fang, L.; Chen, Q.; Banks, C.E. A carbon quantum dot decorated RuO2 network: Outstanding supercapacitances under ultrafast charge and discharge. Energy Environ. Sci. 2013, 6, 3665–3675. [Google Scholar] [CrossRef]
- Prasath, A.; Athika, M.; Duraisamy, E.; Selva Sharma, A.; Sankar Devi, V.; Elumalai, P. Carbon Quantum Dot-Anchored Bismuth Oxide Composites as Potential Electrode for Lithium-Ion Battery and Supercapacitor Applications. ACS Omega 2019, 4, 4943–4954. [Google Scholar] [CrossRef] [PubMed]
- Wei, G.; Du, K.; Zhao, X.; Wang, Z.; Liu, M.; Li, C.; Wang, H.; An, C.; Xing, W. Carbon quantum dot-induced self-assembly of ultrathin Ni (OH) 2 nanosheets: A facile method for fabricating three-dimensional porous hierarchical composite micro-nanostructures with excellent supercapacitor performance. Nano Res. 2017, 10, 3005–3017. [Google Scholar] [CrossRef]
- Wei, G.; Zhao, X.; Du, K.; Wang, Z.; Liu, M.; Zhang, S.; Wang, S.; Zhang, J.; An, C. A general approach to 3D porous CQDs/MxOy (M= Co, Ni) for remarkable performance hybrid supercapacitors. Chem. Eng. J. 2017, 326, 58–67. [Google Scholar] [CrossRef]
- Lv, H.; Yuan, Y.; Xu, Q.; Liu, H.; Wang, Y.-G.; Xia, Y. Carbon quantum dots anchoring MnO2/graphene aerogel exhibits excellent performance as electrode materials for supercapacitor. J. Power Sources 2018, 398, 167–174. [Google Scholar] [CrossRef]
- Li, L.; Li, M.; Liang, J.; Yang, X.; Luo, M.; Ji, L.; Guo, Y.; Zhang, H.; Tang, N.; Wang, X. Preparation of Core–Shell CQD@ PANI Nanoparticles and Their Electrochemical Properties. ACS Appl. Mater. Interfaces 2019, 11, 22621–22627. [Google Scholar] [CrossRef]
- Jian, X.; Yang, H.; Li, J.; Zhang, E.; Liang, Z. Flexible all-solid-state high-performance supercapacitor based on electrochemically synthesized carbon quantum dots/polypyrrole composite electrode. Electrochim. Acta 2017, 228, 483–493. [Google Scholar] [CrossRef]
- Wei, G.; Zhao, X.; Du, K.; Huang, Y.; An, C.; Qiu, S.; Liu, M.; Yao, S.; Wu, Y. Flexible asymmetric supercapacitors made of 3D porous hierarchical CuCo2O4@CQDs and Fe2O3@CQDs with enhanced performance. Electrochim. Acta 2018, 283, 248–259. [Google Scholar] [CrossRef]
- Ganganboina, A.B.; Dutta Chowdhury, A.; Doong, R. Engineering New avenue for appendage of graphene quantum dots on halloysite nanotubes as anode materials for high performance supercapacitors. ACS Sustain. Chem. Eng. 2017, 5, 4930–4940. [Google Scholar] [CrossRef]
- Hong, Y.; Xu, J.; Chung, J.S.; Choi, W.M. Graphene quantum dots/Ni(OH)2 nanocomposites on carbon cloth as a binder-free electrode for supercapacitors. J. Mater. Sci. Technol. 2020, 58, 73–79. [Google Scholar] [CrossRef]
- Huang, Y.; Lin, L.; Shi, T.; Cheng, S.; Zhong, Y.; Chen, C.; Tang, Z. Graphene quantum dots-induced morphological changes in CuCo2S4 nanocomposites for supercapacitor electrodes with enhanced performance. Appl. Surf. Sci. 2019, 463, 498–503. [Google Scholar] [CrossRef]
- Zhang, M.; Liu, W.; Liang, R.; Tjandra, R.; Yu, A. Graphene quantum dot induced tunable growth of nanostructured MnCo 2 O 4.5 composites for high-performance supercapacitors. Sustain. Energy Fuels 2019, 3, 2499–2508. [Google Scholar] [CrossRef]
- Jia, H.; Cai, Y.; Lin, J.; Liang, H.; Qi, J.; Cao, J.; Feng, J.; Fei, W. Heterostructural Graphene Quantum Dot/MnO2 Nanosheets toward High-Potential Window Electrodes for High-Performance Supercapacitors. Adv. Sci. 2018, 5, 1700887. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Wang, J.; Liu, S.; Wu, W.; Jia, T.; Yang, Z.; Mu, S.; Huang, Y. Graphene quantum dots encapsulated tremella-like NiCo2O4 for advanced asymmetric supercapacitors. Carbon 2019, 146, 1–8. [Google Scholar] [CrossRef]
- Huang, Y.; Shi, T.; Zhong, Y.; Cheng, S.; Jiang, S.; Chen, C.; Liao, G.; Tang, Z. Graphene-quantum-dots induced NiCo2S4 with hierarchical-like hollow nanostructure for supercapacitors with enhanced electrochemical performance. Electrochim. Acta 2018, 269, 45–54. [Google Scholar] [CrossRef]
- Li, H.; Yuan, J.; Zha, L.; Wang, L.; Chen, H.; Che, J. Soft conducting polymer hydrogels in situ doped by sulfonated graphene quantum dots for enhanced electrochemical activity. J. Mater. Sci. Mater. Electron. 2020, 31, 2153–2161. [Google Scholar] [CrossRef]
- Syed Zainol Abidin, S.N.J.; Mamat, S.; Abdul Rasyid, S.; Zainal, Z.; Sulaiman, Y. Fabrication of poly (vinyl alcohol)-graphene quantum dots coated with poly (3, 4-ethylenedioxythiophene) for supercapacitor. J. Polym. Sci. Part A Polym. Chem. 2018, 56, 50–58. [Google Scholar] [CrossRef]
- Wang, S.; Shen, J.; Wang, Q.; Fan, Y.; Li, L.; Zhang, K.; Yang, L.; Zhang, W.; Wang, X. High-Performance Layer-by-Layer Self-Assembly PANI/GQD-rGO/CFC Electrodes for a Flexible Solid-State Supercapacitor by a Facile Spraying Technique. ACS Appl. Energy Mater. 2019, 2, 1077–1085. [Google Scholar] [CrossRef]
- Zhao, Z.; Xie, Y. Enhanced electrochemical performance of carbon quantum dots-polyaniline hybrid. J. Power Sources 2017, 337, 54–64. [Google Scholar] [CrossRef]
- Privitera, A.; Righetto, M.; Mosconi, D.; Lorandi, F.; Isse, A.A.; Moretto, A.; Bozio, R.; Ferrante, C.; Franco, L. Boosting carbon quantum dots/fullerene electron transfer via surface group engineering. Phys. Chem. Chem. Phys. 2016, 18, 31286–31295. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Xie, Y. Enhanced electrochemical stability of carbon quantum dots-incorporated and ferrous-coordinated polypyrrole for supercapacitor. J. Solid State Electrochem. 2018, 22, 2515–2529. [Google Scholar] [CrossRef]
- Chang, H.-W.; Fu, J.-X.; Huang, Y.-C.; Lu, Y.-R.; Kuo, C.-H.; Chen, J.-L.; Chen, C.-L.; Lee, J.-F.; Chen, J.-M.; Tsai, Y.-C. NiCo2O4/graphene quantum dots (GQDs) for use in efficient electrochemical energy devices: An electrochemical and X-ray absorption spectroscopic investigation. Catal. Today 2019, 348, 290–298. [Google Scholar] [CrossRef]
- Savekar, A.; Tewari, D.; Kumar, V. Supercapacitor Market; Allied Market Research: Pune, India, 2020; Ch.1-2. [Google Scholar]
- Mazumdar, S. Supercapacitor Market Report: Trends, Forecast and Competitive Analysis; Lucintel: Irving, TX, USA, 2020; Ch.3. [Google Scholar]
- Li, Y.; Wang, Q.; Wang, Y.; Bai, M.; Shao, J.; Ji, H.; Feng, H.; Zhang, J.; Ma, X.; Zhao, W. A flexible and conductive metallic paper-based current collector with energy storage capability in supercapacitor electrodes. Dalt. Trans. 2019, 48, 7659–7665. [Google Scholar] [CrossRef]
- Tang, Z.; Jia, C.; Wan, Z.; Zhou, Q.; Ye, X.; Zhu, Y. Facile preparation of CoNi2S4@NiSe nano arrays on compressed nickel foam for high performance flexible supercapacitors. RSC Adv. 2016, 6, 112307–112316. [Google Scholar] [CrossRef]
- Lee, K.; Lee, H.; Shin, Y.; Yoon, Y.; Kim, D.; Lee, H. Highly transparent and flexible supercapacitors using graphene-graphene quantum dots chelate. Nano Energy 2016, 26, 746–754. [Google Scholar] [CrossRef]
- Soram, B.S.; Thangjam, I.S.; Dai, J.Y.; Kshetri, T.; Kim, N.H.; Lee, J.H. Flexible transparent supercapacitor with core-shell Cu@Ni@NiCoS nanofibers network electrode. Chem. Eng. J. 2020, 395, 125019. [Google Scholar] [CrossRef]
- Ilani, S.; Donev, L.A.K.; Kindermann, M.; McEuen, P.L. Measurement of the quantum capacitance of interacting electrons in carbon nanotubes. Nat. Phys. 2006, 2, 687–691. [Google Scholar] [CrossRef]
- Zhang, H.; Berthod, C.; Berger, H.; Giamarchi, T.; Morpurgo, A.F. Band Filling and Cross Quantum Capacitance in Ion-Gated Semiconducting Transition Metal Dichalcogenide Monolayers. Nano Lett. 2019, 19, 8836–8845. [Google Scholar] [CrossRef]
- John, D.L.; Castro, L.C.; Pulfrey, D.L. Quantum capacitance in nanoscale device modeling. J. Appl. Phys. 2004, 96, 5180–5184. [Google Scholar] [CrossRef]
- Rosenblatt, S.; Yaish, Y.; Park, J.; Gore, J.; Sazonova, V.; McEuen, P.L. High Performance Electrolyte Gated Carbon Nanotube Transistors. Nano Lett. 2002, 2, 869–872. [Google Scholar] [CrossRef]
- Jiang, L.; Fu, W.; Birdja, Y.Y.; Koper, M.T.M.; Schneider, G.F. Quantum and electrochemical interplays in hydrogenated graphene. Nat. Commun. 2018, 9, 793. [Google Scholar] [CrossRef] [PubMed]
- Mousavi-Khoshdel, M.; Targholi, E.; Momeni, M.J. First-Principles Calculation of Quantum Capacitance of Codoped Graphenes as Supercapacitor Electrodes. J. Phys. Chem. C 2015, 119, 26290–26295. [Google Scholar] [CrossRef]
- Su, F.; Huo, L.; Kong, Q.; Xie, L.; Chen, C. Theoretical study on the quantum capacitance origin of graphene cathodes in lithium ion capacitors. Catalysts 2018, 8, 444. [Google Scholar] [CrossRef]
- Yang, G.M.; Zhang, H.Z.; Fan, X.F.; Zheng, W.T. Density Functional Theory Calculations for the Quantum Capacitance Performance of Graphene-Based Electrode Material. J. Phys. Chem. C 2015, 119, 6464–6470. [Google Scholar] [CrossRef]
- Chuai, M.; Yang, T.; Zhang, M. Quantum capacitance of CuS:Ce3+ quantum dots as high-performing supercapacitor electrodes. J. Mater. Chem. A 2018, 6, 6534–6541. [Google Scholar] [CrossRef]
- Zeng, W.; Zhang, Y.; Liu, X.; Qi, L.; Kang, W.; Fang, L.; Zhou, M. B/N-doped graphdiyne as superior supercapacitor electrode with record high quantum capacitance. Appl. Surf. Sci. 2020, 523, 146468. [Google Scholar] [CrossRef]
- Xu, Q.; Yang, G.M.; Zheng, W.T. DFT calculation for stability and quantum capacitance of MoS2 monolayer-based electrode materials. Mater. Today Commun. 2020, 22, 100772. [Google Scholar] [CrossRef]
- Narayanan, R.; Yamada, H.; Karakaya, M.; Podila, R.; Rao, A.M.; Bandaru, P.R. Modulation of the electrostatic and quantum capacitances of few layered graphenes through plasma processing. Nano Lett. 2015, 15, 3067–3072. [Google Scholar] [CrossRef]
- Chen, J.; Han, Y.; Kong, X.; Deng, X.; Park, H.J.; Guo, Y.; Jin, S.; Qi, Z.; Lee, Z.; Qiao, Z. The Origin of Improved Electrical Double-Layer Capacitance by Inclusion of Topological Defects and Dopants in Graphene for Supercapacitors. Angewandte 2016, 55, 13822–13827. [Google Scholar] [CrossRef]
- Parida, K.; Bhavanasi, V.; Kumar, V.; Wang, J.; Lee, P.S. Fast charging self-powered electric double layer capacitor. J. Power Sources 2017, 342, 70–78. [Google Scholar] [CrossRef]
- Gao, D.; Liu, R.; Yu, W.; Luo, Z.; Liu, C.; Fan, S. Gravity-Induced Self-Charging in Carbon Nanotube/Polymer Supercapacitors. J. Phys. Chem. C 2019, 123, 5249–5254. [Google Scholar] [CrossRef]
- Ramadoss, A.; Saravanakumar, B.; Lee, S.W.; Kim, Y.-S.; Kim, S.J.; Wang, Z.L. Piezoelectric-driven self-charging supercapacitor power cell. ASC Nano 2015, 9, 4337–4345. [Google Scholar] [CrossRef] [PubMed]
- Kundu, A.; Fisher, T.S. Harnessing the thermogalvanic effect of the ferro/ferricyanide redox couple in a thermally chargeable supercapacitor. Electrochim. Acta 2018, 281, 357–369. [Google Scholar] [CrossRef]
- Jiao, F.; Naderi, A.; Zhao, D.; Schlueter, J.; Shahi, M.; Sundström, J.; Granberg, H.; Edberg, J.; Ail, U.; Brill, J. Ionic thermoelectric paper. J. Mater. Chem. A 2017, 5, 16883–16888. [Google Scholar] [CrossRef]
- Wang, H.; Ail, U.; Gabrielsson, R.; Berggren, M.; Crispin, X. Ionic Seebeck effect in conducting polymers. Adv. Engery Mater. 2015, 5, 1500044. [Google Scholar] [CrossRef]
- Zhao, D.; Wang, H.; Khan, Z.U.; Chen, J.C.; Gabrielsson, R.; Jonsson, M.P.; Berggren, M.; Crispin, X. Ionic thermoelectric supercapacitors. Engery Environ. Sci. 2016, 9, 1450–1457. [Google Scholar] [CrossRef]
- Wang, H.; Zhao, D.; Khan, Z.U.; Puzinas, S.; Jonsson, M.P.; Berggren, M.; Crispin, X. Ionic Thermoelectric Figure of Merit for charging of supercapacitors. Adv. Electron. Mater. 2017, 3, 1700013. [Google Scholar] [CrossRef]
- Kim, S.L.; Lin, H.T.; Yu, C. Thermally Chargeable Solid-State Supercapacitor. Adv. Energy Mater. 2016, 6, 1600546. [Google Scholar] [CrossRef]
- Aji, A.S.; Nishi, R.; Ago, H.; Ohno, Y. High output voltage generation of over 5 V from liquid motion on single-layer MoS2. Nano Energy 2020, 68, 104370. [Google Scholar] [CrossRef]




| Material | Size (nm) | Initial Capacity | Current Density | Capacity Retention | Power Density | Energy Density | Ref |
|---|---|---|---|---|---|---|---|
| Bare GQDs | 1–5 | 296 F/g | 1 A/g | 97.6% (5000×) | 10,000 W/kg | 22.2 Wh/kg | [119] |
| CDs/Graphene | 2–5 | 91.9 F/g | 0.1 A/cm2 | 96% (10,000×) | 15 mw/cm2 | 46.67 Wh/cm2 | [115] |
| ONCDs/Porous Hydrogels | 3–10 | 483 F/g | 1 A/g | 100% (10,000×) | 11,000 W/kg | 13.5 Wh/kg | [124] |
| N-GQDs/Carbon Fibre/Graphene Hydrogel | 1.5–7 | 93.7 F/cm3 | 20 mA/cm3 | 84.2% (10,000×) | 200 W/kg | 20.5 Wh/kg | [122] |
| GQDs/Carbon Cloth | 10–20 | 77.5 mF/cm2 | 0.2 mA/cm2 | NP | 0.288 W/cm2 | 24,8 mWh/cm2 | [125] |
| GQDs/Activated Carbon | 2.9 | 388 F/g | 1 A/g | 100% (10,000×) | 12.500 W/kg | 7.99 Wh/kg | [123] |
| N,O-GQDs/CNT/Carbon Cloth | 4–5 | 461 F/cm3 | 0.5 mA/cm2 | 87.5% (2000×) | 0.1 mW/cm2 | 41.7 Wh/cm2 | [126] |
| Amine-enriched GQDs | 2.9 | 595 F/g | 1 A/g | 90% (10,000×) | 250 W/kg | 21.8 Wh/kg | [93] |
| S-GQDs | 20 | 362 F/g | 5 mV/s | NP | NP | NP | [47] |
| GQDs/Ultra microporous Carbon | 2.96 | 270 F/g | 1 A/g | 100% (50,000×) | 70 W/kg | 9.38 Wh/kg | [127] |
| GQDs/3DG | NP | 242 F/g | 1.17 A/g | 93% (10,000×) | NP | NP | [128] |
| GQDs/Conductive Porous Carbon | NP | 315 F/g | 1 A/g | 100% (10,000×) | 247.75 W/kg | 9.21 Wh/kg | [129] |
| GH-GQD | 8–10 | 451.7 F/g | 0.5 A/g | 89% (10,000×) | NP | NP | [130] |
| Material | C-QDs Type | Size (nm) | Initial Capacity (F/g @ 1 A/g) | Capacity Retention | Power Density (W/kg) | Energy Density (Wh/kg) | Ref. | |||
|---|---|---|---|---|---|---|---|---|---|---|
| M | C | without C-QDs | with C-QDs | without C-QDs | with C-QDs | |||||
| NiCo2O4 nanowire | CDs | 100 | 2.56 | 699 | 2202 | NP | 95.3% (1100×) | 499.8 | 73.5 | [132] |
| Polypyrrole | CDs | NP | 8.7 | 267 | 563 | 82.9% (1000×) | 97% (1000×) | 250.1 | 30.1 | [140] |
| GNP-PPy | CDs | NP | 2.9 | NP | 173 mF/cm2 | NP | 96% (2000×) | NP | NP | [141] |
| CoS/rGO | CDs | NP | NP | NP | 697 | NP | 85.9% (10,000×) | 16.000 | 36.6 | [139] |
| NiCo2O4 | CDs | 20–2000 | 2–4 | NP | 2168 | NP | 99.96% (5000×) | 216 | 62 | [133] |
| V2O5 | CDs | 1000 | 200–220 | 60 | 270 | 67% (5000×) | 87% (5000×) | 4100 | 60 | [137] |
| CuS | CDs | 300 | 4 | NP | 736.1 | NP | 92% (5000×) | NP | NP | [142] |
| NiO/Co3O4 | CDs | NP | NP | NP | 1775 | NP | 95.7% (10,000) | 16000 | 21.3 | [135] |
| Bi2O3 | CDs | NP | NP | 849 | 1046 | NP | 85.3% (1500×) | 770.9 | 79.9 | [136] |
| NiCo2O4 | C-QDs | 250 | 3–6 | NP | 856 | NP | 98.7% (10,000×) | 10240 | 27.8 | [143] |
| RuO2 | C-QDs | 50–100 | < 10 | 642 | 594 | 39.6% (5000×) | 95.9 (5000×) | NP | NP | [144] |
| Bi2O3 | C-QD | NP | NP | NP | 125 | NP | ~97% (2500×) | 8400 | 32 | [145] |
| NiOH | C-QD | NP | NP | 1414.3 | 2750 @20 A/g | 26% (2000×) | 64.1% (2000×) | 683.7 | 57.4 | [146] |
| MnO2 nanowire | C-QDs | 1–4 | NP | 232 | 340 | 61.1% (10,000×) | 76.4% (10,000×) | 10000 | 12.5 | [92] |
| Co3O4 | C-QDs | 9 | 3 | 1446 | 1603 | 96% (2000×) | 97.6% (2000×) | 810 | 78.4 | [147] |
| MnO2 | C-QDs | NP | 3 | 527 | 721 | 70.3% (100,00×) | 92% (10,000×) | 1000 | 38.2 | [148] |
| NiS | C-QDs | 800 | 1–2 | 710 | 880 | NP | NP | 33000 | 30 | [67] |
| PANI | C-QDs | 4 | 10 | 182.5 | 222.7 | ~95% (500×) | ~90% (500×) | NP | NP | [149] |
| Polypyrrole | C-QDs | NP | NP | 215.6 | 424.6 | NP | 85.7% (2000×) | NC | NC | [150] |
| CuCo2O4 | C-QDs | 3 | NP | 464.4 | 779.8 | NP | 86.4% (5000×) | 1203.7 | 39.5 | [151] |
| Halloysite Nanotube | GQDs | NP | 5–10 | NP | 338 | NP | 88% (5000× | 230 | 50.03 | [152] |
| Ni(OH)2 | GQDs | NP | 7 | NP | 1825 | 55.2% (8000×) | 83.5% (8000×) | 2021 | 80.8 | [153] |
| CuCo2S4 | GQDs | NP | NP | 908 | 1725 | NP | 90% (10,000×) | NP | NP | [154] |
| MnCo2O4.5 | GQDs | ~100 | NP | 368 | 1625 | NP | 77% (5000×) | 2800 | 24 | [155] |
| MnO2 | GQDs | 10 | 1 | 400 | 1170 | 43.1% (10,000×) | 92.7% (10,000×) | 12351 | 118 | [156] |
| MOF | GQDs | NP | 3 | 188 | 780 @10 mV/s | NP | 80.2% (10,000×) | 5,000 | 6.81 | [121] |
| MoS2 | GQDs | NP | 15 | 100 | 323.5 | NP | 92.3% (500×) | 489.96 | 38.47 | [82] |
| NiCo-LDH | GQDs | NP | 2 | 1769 | 2220 | 55.7% (10,000×) | 75.3% (10,000×) | 8000 | 50.84 | [100] |
| NiCo2O4 | GQDs | 800 | 2–4.5 | 1190 | 1382 | 99% (4000×) | 99% (4000×) | 800 | 38 | [157] |
| NiCoS | GQDs | 200 | NP | 562 | 678 @0.2 A/g | 70% (5000×) | 94% (10,000×) | NP | NP | [158] |
| Polypyrrole | GQDs | NP | 5 | 437 | 560 | ~37% (1000×) | 60.5% (1000×) | NP | NP | [159] |
| PVA-PEDOT | GQDs | NP | 55.6 | 161.48 | 291.6 | 64% (1000×) | 98% (1000×) | NP | 16.95 | [160] |
| PANI/rGO/CFC | GQDs | NP | NP | NP | NP | 78% (10,000×) | 97.7% (10,000×) | 424 | 34.2 | [161] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Permatasari, F.A.; Irham, M.A.; Bisri, S.Z.; Iskandar, F. Carbon-Based Quantum Dots for Supercapacitors: Recent Advances and Future Challenges. Nanomaterials 2021, 11, 91. https://doi.org/10.3390/nano11010091
Permatasari FA, Irham MA, Bisri SZ, Iskandar F. Carbon-Based Quantum Dots for Supercapacitors: Recent Advances and Future Challenges. Nanomaterials. 2021; 11(1):91. https://doi.org/10.3390/nano11010091
Chicago/Turabian StylePermatasari, Fitri Aulia, Muhammad Alief Irham, Satria Zulkarnaen Bisri, and Ferry Iskandar. 2021. "Carbon-Based Quantum Dots for Supercapacitors: Recent Advances and Future Challenges" Nanomaterials 11, no. 1: 91. https://doi.org/10.3390/nano11010091
APA StylePermatasari, F. A., Irham, M. A., Bisri, S. Z., & Iskandar, F. (2021). Carbon-Based Quantum Dots for Supercapacitors: Recent Advances and Future Challenges. Nanomaterials, 11(1), 91. https://doi.org/10.3390/nano11010091





