Interface Chemical Modification between All-Inorganic Perovskite Nanocrystals and Porous Silica Microspheres for Composite Materials with Improved Emission
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Setup
2.2. Synthesis
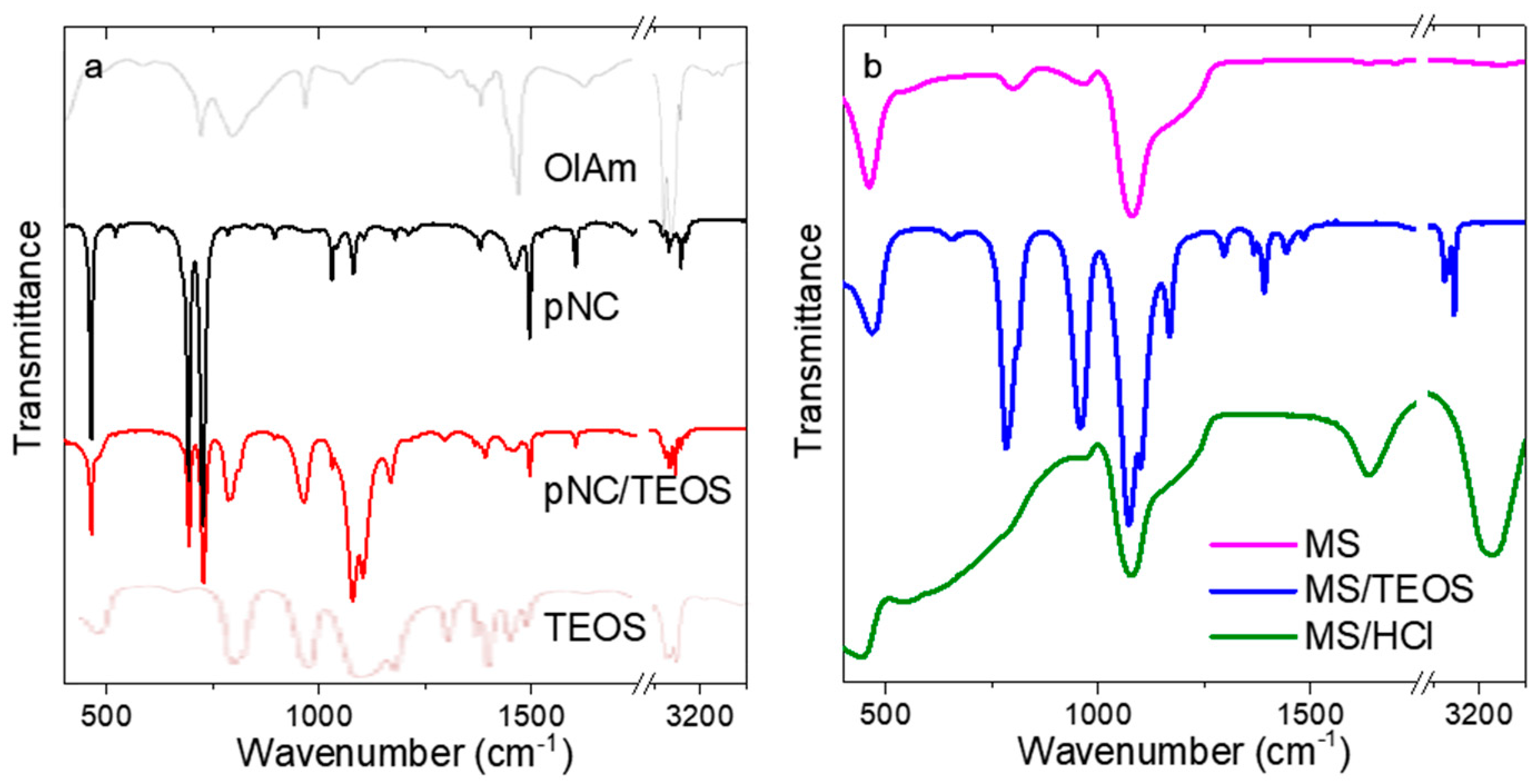
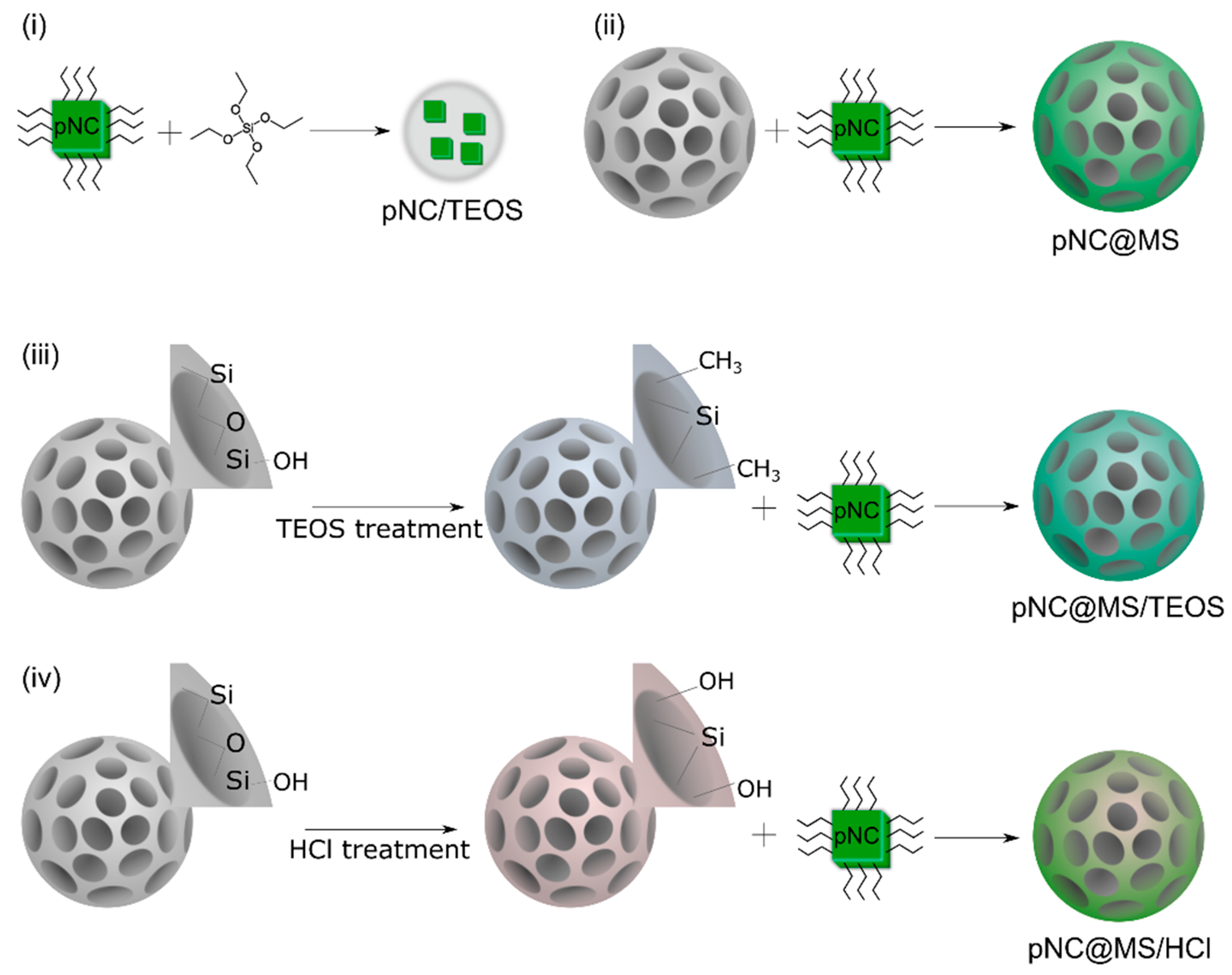
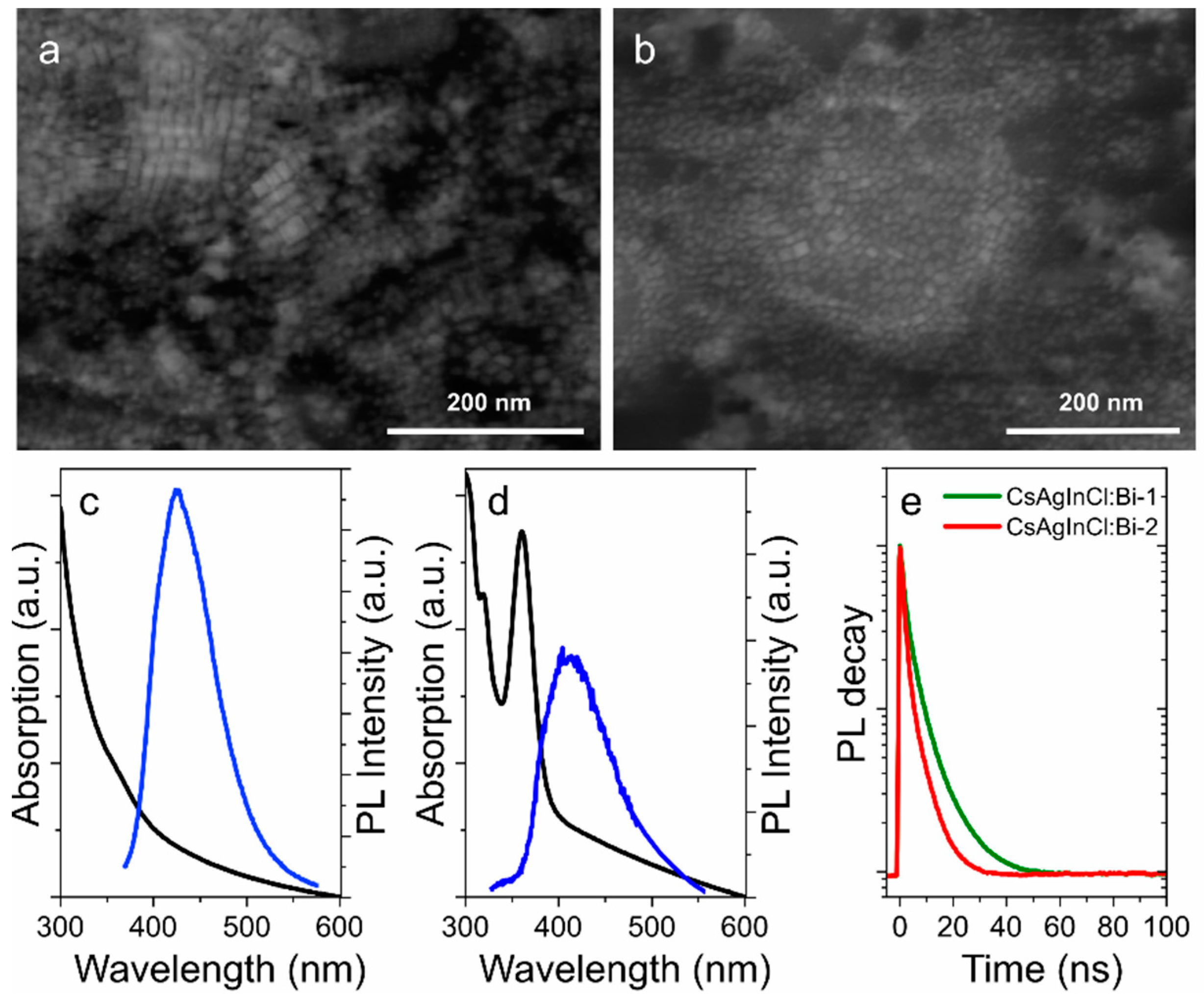
3. Results and Discussion
TEOS-Treated Lead-Free pNCs
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shamsi, J.; Urban, A.S.; Imran, M.; De Trizio, L.; Manna, L. Metal halide perovskite nanocrystals: Synthesis, post-synthesis modifications, and their optical properties. Chem. Rev. 2019, 119, 3296–3348. [Google Scholar] [CrossRef] [PubMed]
- Jena, A.K.; Kulkarni, A.; Miyasaka, T. Halide perovskite photovoltaics: Background, status, and future prospects. Chem. Rev. 2019, 119, 3036–3103. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, X.; Huang, H.; Kershaw, S.V.; Rogach, A.L. Advances in metal halide perovskite nanocrystals: Synthetic strategies, growth mechanisms, and optoelectronic applications. Mater. Today 2020, 32, 204–221. [Google Scholar] [CrossRef]
- Krieg, F.; Ochsenbein, S.T.; Yakunin, S.; Brinck, S.T.; Aellen, P.; Süess, A.; Clerc, B.; Guggisberg, D.; Nazarenko, O.; Shynkarenko, Y.; et al. Colloidal CsPbX3 (X = Cl, Br, I) nanocrystals 2.0: Zwitterionic capping ligands for improved durability and stability. ACS Energy Lett. 2018, 3, 641–646. [Google Scholar] [CrossRef] [PubMed]
- Liang, P.; Zhang, P.; Pan, A.; Yan, K.; Zhu, Y.; Yang, M.; He, L. Unusual stability and temperature-dependent properties of highly emissive CsPbBr3 perovskite nanocrystals obtained from in situ crystallization in poly (vinylidene difluoride). ACS Appl. Mater. Interfaces 2019, 11, 22786–22793. [Google Scholar] [CrossRef]
- Ushakova, E.V.; Matuhina, A.I.; Sokolova, A.V.; Cherevkov, S.A.; Dubavik, A.; Medvedev, O.S.; Litvin, A.P.; Kurdyukov, D.A.; Golubev, V.G.; Baranov, A.V. Enhanced stability of the optical responses from all-inorganic perovskite nanocrystals embedded in a synthetic opal matrix. Nanotechnology 2019, 30, 405206. [Google Scholar] [CrossRef]
- Pan, A.; Li, Y.; Wu, Y.; Yan, K.; Jurow, M.J.; Liu, Y.; He, L. Stable luminous nanocomposites of CsPbX3 perovskite nanocrystals anchored on silica for multicolor anti-counterfeit ink and white-LEDs. Mater. Chem. Front. 2019, 3, 414–419. [Google Scholar] [CrossRef]
- Qiu, L.; Yang, H.; Dai, Z.; Sun, F.; Hao, J.; Guan, M.; Dang, P.; Yan, C.; Linab, J.; Li, G. Highly efficient and stable CsPbBr3 perovskite quantum dots by encapsulation in dual-shell hollow silica spheres for WLEDs. Inorg. Chem. Front. 2020, 7, 2060–2071. [Google Scholar] [CrossRef]
- Zhong, Q.; Cao, M.; Hu, H.; Yang, D.; Chen, M.; Li, P.; Wu, L.; Zhang, Q. One-pot synthesis of highly stable CsPbBr3@SiO2 core-shell nanoparticles. ACS Nano 2018, 12, 8579–8587. [Google Scholar] [CrossRef]
- Liu, S.; Chen, T.-M. Synthesis and luminescent properties of polymer-silica multilayer-encapsulated perovskite quantum dots for optoelectronics. J. Chin. Chem. Soc. 2020, 67, 109–115. [Google Scholar] [CrossRef]
- Huang, S.; Li, Z.; Kong, L.; Zhu, N.; Shan, A.; Li, L. Enhancing the stability of CH3NH3PbBr3 quantum dots by embedding in silica spheres derived from tetramethyl orthosilicate in “waterless” toluene. J. Am. Chem. Soc. 2016, 138, 5749–5752. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Li, F.; Qiu, L.; Lin, F.; Lai, Z.; Wang, S.; Lin, L.; Zhu, Y.; Wang, Y.; Jiang, Y.; et al. Enhancing the stability of CH3NH3PbBr3 nanoparticles using double hydrophobic shells of SiO2 and poly(vinylidene fluoride). ACS Appl. Mater. Interfaces 2019, 11, 26384–26391. [Google Scholar] [CrossRef] [PubMed]
- Ushakova, E.V.; Cherevkov, S.A.; Sokolova, A.V.; Li, Y.; Azizov, R.R.; Baranov, M.A.; Kurdyukov, D.A.; Stovpiaga, E.Y.; Golubev, V.G.; Rogach, A.L.; et al. Stable luminescent composite microspheres based on porous silica with embedded CsPbBr3 perovskite nanocrystals. ChemNanoMat 2020, 6, 1080–1085. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, A.; Liu, T.; Zhou, L.; Zheng, J.; Zuo, Y.; He, Y.; Li, J. A facile method for preparing Yb3+-doped perovskite nanocrystals with ultra-stable near-infrared light emission. RSC Adv. 2020, 10, 17635–17641. [Google Scholar] [CrossRef]
- Li, W.; Deng, W.; Fan, X.; Chun, F.; Xie, M.; Luo, C.; Yang, S.; Osman, H.; Liu, C.; Zheng, X.; et al. Low toxicity antisolvent synthesis of composition-tunable luminescent all-inorganic perovskite nanocrystals. Ceram. Int. 2018, 44, 18123–18128. [Google Scholar] [CrossRef]
- Hu, Z.; Liu, Z.; Bian, Y.; Li, S.; Tang, X.; Du, J.; Zang, Z.; Zhou, M.; Hu, W.; Tian, Y.; et al. Enhanced two-photon-pumped emission from in situ synthesized nonblinking CsPbBr3/SiO2 nanocrystals with excellent stability. Adv. Opt. Mater. 2018, 6, 6. [Google Scholar] [CrossRef]
- Bai, Y.; Lin, Y.; Ren, L.; Shi, X.; Strounina, E.; Deng, Y.; Wang, Q.; Fang, Y.; Zheng, X.; Lin, Y.; et al. Oligomeric silica-wrapped perovskites enable synchronous defect passivation and grain stabilization for efficient and stable perovskite photovoltaics. ACS Energy Lett. 2019, 4, 1231–1240. [Google Scholar] [CrossRef]
- Tang, X.; Chen, Y.; Liu, Z.; Du, J.; Yao, Z.; Huang, Y.; Chen, C.; Yang, Z.; Shi, T.; Hu, W.; et al. Ultrathin, core–shell structured SiO2 Coated Mn2+ -doped perovskite quantum dots for bright white light-emitting diodes. Small 2019, 15, e1900484. [Google Scholar] [CrossRef]
- Zheng, Z.; Liu, L.; Yi, F.; Zhao, J. Significantly improving the moisture-, oxygen- and thermal-induced photoluminescence in all-inorganic halide perovskite CsPbI3 crystals by coating the SiO2 layer. J. Lumin. 2019, 216, 116722. [Google Scholar] [CrossRef]
- Ushakova, E.V.; Cherevkov, S.; Kuznetsova, V.; Baranov, A. Lead-free perovskites for lighting and lasing applications: A minireview. Materials 2019, 12, 3845. [Google Scholar] [CrossRef]
- Stylianakis, M.M.; Maksudov, T.; Panagiotopoulos, A.; Kakavelakis, G.; Petridis, K. Inorganic and hybrid perovskite based laser devices: A review. Materials 2019, 12, 859. [Google Scholar] [CrossRef]
- Wang, Z.; He, H.; Liu, S.; Wang, H.; Zeng, Q.; Liu, Z.; Xiong, Q.; Fan, H.J. Air stable organic-inorganic perovskite nanocrystals@polymer nanofibers and waveguide lasing. Small 2020, 16, e2004409. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Chen, B.; Hua, Y.; Liu, Z.; Wei, Y.; Liu, S.; Hu, A.; Shen, X.; Zhang, Y.; Gao, Y.; et al. CMOS compatible high-performance nanolasing based on perovskite-SiN hybrid integration. Adv. Opt. Mater. 2020, 8, 2000453. [Google Scholar] [CrossRef]
- Price, M.B.; Lewellen, K.J.; Hardy, J.; Lockwood, S.M.; Zemke-Smith, C.; Wagner, I.; Gao, M.; Grand, J.; Chen, K.; Hodgkiss, J.M.; et al. Whispering-gallery mode lasing in perovskite nanocrystals chemically bound to silicon dioxide microspheres. J. Phys. Chem. Lett. 2020, 11, 7009–7014. [Google Scholar] [CrossRef] [PubMed]
- Yan, D.; Shi, T.; Zang, Z.; Zhao, S.; Du, J.; Leng, Y. Stable and low-threshold whispering-gallery-mode lasing from modified CsPbBr3 perovskite quantum dots@SiO2 sphere. Chem. Eng. J. 2020, 401, 126066. [Google Scholar] [CrossRef]
- Trofimova, E.Y.; Kurdyukov, D.A.; Yakovlev, S.A.; Kirilenko, D.A.; Kukushkina, Y.A.; Nashchekin, A.V.; Sitnikova, A.A.; Yagovkina, M.A.; Golubev, V. Monodisperse spherical mesoporous silica particles: Fast synthesis procedure and fabrication of photonic-crystal films. Nanotechnology 2013, 24, 155601. [Google Scholar] [CrossRef]
- National Institute of Advanced Industrial Science and Technology (AIST) Spectral Database for Organic Compounds SDBS. Available online: https://sdbs.db.aist.go.jp/sdbs/cgi-bin/landingpage?sdbsno=10418 (accessed on 1 December 2020).
- Yan, F.; Jiang, J.; Chen, X.; Tian, S.; Li, K. Synthesis and characterization of silica nanoparticles preparing by low-temperature vapor-phase hydrolysis of SiCl4. Ind. Eng. Chem. Res. 2014, 53, 11884–11890. [Google Scholar] [CrossRef]
- Liu, Y.; Jing, Y.; Zhao, J.; Liu, Q.; Xia, Z. Design optimization of lead-free perovskite Cs2AgInCl6:Bi nanocrystals with 11.4% photoluminescence quantum yield. Chem. Mater. 2019, 31, 3333–3339. [Google Scholar] [CrossRef]
- Li, S.; Lei, D.; Ren, W.; Guo, X.; Wu, S.; Zhu, Y.; Rogach, A.L.; Chhowalla, M.; Jen, A.K.-Y. Water-resistant perovskite nanodots enable robust two-photon lasing in aqueous environment. Nat. Commun. 2020, 11, 1192–1198. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cherevkov, S.; Azizov, R.; Sokolova, A.; Nautran, V.; Miruschenko, M.; Arefina, I.; Baranov, M.; Kurdyukov, D.; Stovpiaga, E.; Golubev, V.; et al. Interface Chemical Modification between All-Inorganic Perovskite Nanocrystals and Porous Silica Microspheres for Composite Materials with Improved Emission. Nanomaterials 2021, 11, 119. https://doi.org/10.3390/nano11010119
Cherevkov S, Azizov R, Sokolova A, Nautran V, Miruschenko M, Arefina I, Baranov M, Kurdyukov D, Stovpiaga E, Golubev V, et al. Interface Chemical Modification between All-Inorganic Perovskite Nanocrystals and Porous Silica Microspheres for Composite Materials with Improved Emission. Nanomaterials. 2021; 11(1):119. https://doi.org/10.3390/nano11010119
Chicago/Turabian StyleCherevkov, Sergei, Ruslan Azizov, Anastasiia Sokolova, Valeriia Nautran, Mikhail Miruschenko, Irina Arefina, Mikhail Baranov, Dmitry Kurdyukov, Ekaterina Stovpiaga, Valery Golubev, and et al. 2021. "Interface Chemical Modification between All-Inorganic Perovskite Nanocrystals and Porous Silica Microspheres for Composite Materials with Improved Emission" Nanomaterials 11, no. 1: 119. https://doi.org/10.3390/nano11010119
APA StyleCherevkov, S., Azizov, R., Sokolova, A., Nautran, V., Miruschenko, M., Arefina, I., Baranov, M., Kurdyukov, D., Stovpiaga, E., Golubev, V., Baranov, A., & Ushakova, E. (2021). Interface Chemical Modification between All-Inorganic Perovskite Nanocrystals and Porous Silica Microspheres for Composite Materials with Improved Emission. Nanomaterials, 11(1), 119. https://doi.org/10.3390/nano11010119








