An Overview of the Recent Progress in Modifications of Carbon Nanotubes for Hydrogen Adsorption
Abstract
1. Introduction
2. Factors Affecting the Hydrogen Adsorption Properties of CNTs
2.1. Effect of Internal Factors on Hydrogen Adsorption Capacity of CNTs
2.2. Effect of External Factors on Hydrogen Adsorption Capacity of CNTs
3. Modification of CNTs
3.1. Activation of CNTs
3.1.1. Physical Treatment
3.1.2. Chemical Treatment
3.2. Loading Metals
3.3. Loading Hetero-Atoms
3.4. Other Doping
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Li, W.; Wang, H.; Ren, Z.; Wang, G.; Bai, J. Co-production of hydrogen and multi-wall carbon nanotubes from ethanol decomposition over Fe/Al2O3 catalysts. Appl. Catal. B-Environ. 2008, 84, 433–439. [Google Scholar] [CrossRef]
- Eswaramoorthi, I.; Sundaramurthy, V.; Dalai, A. Partial oxidation of methanol for hydrogen production over carbon nanotubes supported Cu-Zn catalysts. Appl. Catal. A-Gen. 2006, 313, 22–34. [Google Scholar] [CrossRef]
- Ioannatos, G.E.; Verykios, X.E. H2 storage on single- and multi-walled carbon nanotubes. Int. J. Hydrog. Energy 2010, 35, 622–628. [Google Scholar] [CrossRef]
- Aydın, R.; Köleli, F. Hydrogen evolution on conducting polymer electrodes in acidic media. Prog. Org. Coat. 2006, 56, 76–80. [Google Scholar] [CrossRef]
- Kiliç, E.Ö.; Koparal, A.S.; Öğütveren, Ü.B. Hydrogen production by electrochemical decomposition of formic acid via solid polymer electrolyte. Fuel Process. Technol. 2009, 90, 158–163. [Google Scholar] [CrossRef]
- Jukic, A.; Metikoš-Hukovic, M. The hydrogen evolution reaction on pure and polypyrrole-coated GdNi4Al electrodes. Electrochim. Acta 2003, 48, 3929–3937. [Google Scholar] [CrossRef]
- Sherif, S.A.; Barbir, F.; Veziroglu, T.N. Wind energy and the hydrogen economy-review of the technology. Sol. Energy 2005, 78, 647–660. [Google Scholar] [CrossRef]
- Zhang, K.; Liu, S.; Liu, M.; Zhang, H.; Lu, J.; Cao, Z.; Zhang, H. Research developments of hydrogen energy. Mater. Rev. 2011, 25, 116–119. (In Chinese) [Google Scholar]
- Mao, Z. Moving towards hydrogen energy. Sci. Technol. Rev. 2005, 23, 34–38. (In Chinese) [Google Scholar] [CrossRef]
- Liu, M.; Li, D.; Qiao, J.; Li, W.; Wu, Z. The use of hydrogen energy and hydrogen adsorption storage technology of carbonaceous materials. Chem. Ind. Times 2013, 27, 35–38. (In Chinese) [Google Scholar] [CrossRef]
- Chen, Y.L.; Liu, B.; Wu, J.; Huang, Y.; Jiang, H.; Hwang, K.C. Mechanics of hydrogen storage in carbon nanotubes. J. Mech. Phys. Solids 2008, 56, 3224–3241. [Google Scholar] [CrossRef]
- Barghi, S.H.; Tsotsis, T.T.; Sahimi, M. Chemisorption, physisorption and hysteresis during hydrogen storage in carbon nanotubes. Int. J. Hydrog. Energy 2014, 39, 1390–1397. [Google Scholar] [CrossRef]
- Huang, Z. Review of hydrogen storage by carbon nanotubes. Chin. J. Rare Met. 2005, 29, 890–897. (In Chinese) [Google Scholar] [CrossRef]
- Lyu, J.; Lider, A.; Kudiiarov, V. Using ball milling for modification of the hydrogenation/dehydrogenation process in magnesium-based hydrogen storage materials: an overview. Metals 2019, 9, 768. [Google Scholar] [CrossRef]
- Jinzhe, L.; Lider, A.M.; Kudiiarov, V.N. An overview of progress in Mg-based hydrogen storage films. Chin. Phys. B 2019, 28, 98801. [Google Scholar] [CrossRef]
- Dillon, A.C.; Jones, K.M.; Bekkedahl, T.A.; Kiang, C.H.; Bethune, D.S.; Heben, M.J. Storage of hydrogen in single-walled carbon nanotubes. Nature 1997, 386, 377–379. [Google Scholar] [CrossRef]
- Li, S.; Yang, X.D.; Zhu, H.Y.; Liu, Y.; Liu, Y.N. Hydrogen storage alloy and carbon nanotubes mixed catalyst in a direct borohydride fuel cell. J. Mater. Sci. Technol. 2011, 27, 1089–1093. [Google Scholar] [CrossRef]
- Yamaguchi, H.; Nejoh, Y. Numerical simulation of hydrogen storage into a single-walled carbon nanotube in a plasma. IEEJ Trans. Electr. Electron. Eng. 2008, 3, 596–598. [Google Scholar] [CrossRef]
- Sankaran, M.; Viswanathan, B.; Murthy, S.S. Boron substituted carbon nanotubes—How appropriate are they for hydrogen storage? Int. J. Hydrog. Energy 2008, 33, 393–403. [Google Scholar] [CrossRef]
- Reyhani, A.; Mortazavi, S.Z.; Golikand, A.N.; Moshfegh, A.Z.; Mirershadi, S. The effect of various acids treatment on the purification and electrochemical hydrogen storage of multi-walled carbon nanotubes. J. Power Sources 2008, 183, 539–543. [Google Scholar] [CrossRef]
- Cheng, J.R.; Zhang, L.B.; Ding, R.; Ding, Z.F.; Wang, X.; Wang, Z.; Fang, X. Influence of chemical potential on the computer simulation of hydrogen storage in single-walled carbon nanotube array. Comput. Mater. Sci. 2008, 44, 601–604. [Google Scholar] [CrossRef]
- Zacharia, R.; Kim, K.Y.; Hwang, S.W.; Nahm, K.S. Intrinsic linear scaling of hydrogen storage capacity of carbon nanotubes with the specific surface area. Catal. Today 2007, 120, 426–431. [Google Scholar] [CrossRef]
- Xie, L.; Li, X.Q. Study on electrochemical hydrogen storage of multi-walled carbon nanotubes. Adv. Mater. Res. 2007, 26–28, 831–834. [Google Scholar] [CrossRef]
- Weng, C.I.; Ju, S.P.; Fang, K.C.; Chang, F.P. Atomistic study of the influences of size, VDW distance and arrangement of carbon nanotubes on hydrogen storage. Comput. Mater. Sci. 2007, 40, 300–308. [Google Scholar] [CrossRef]
- Sankaran, M.; Viswanathan, B. Hydrogen storage in boron substituted carbon nanotubes. Carbon 2007, 45, 1628–1635. [Google Scholar] [CrossRef]
- Kuc, A.; Zhechkov, L.; Patchkovskii, S.; Seifert, G.; Heine, T. Hydrogen sieving and storage in fullerene intercalated graphite. Nano Lett. 2007, 7, 1–5. [Google Scholar] [CrossRef]
- Li, J.; Furuta, T.; Goto, H.; Ohashi, T.; Fujiwara, Y.; Yip, S. Theoretical evaluation of hydrogen storage capacity in pure carbon nanostructures. J. Chem. Phys. 2003, 119, 2376–2385. [Google Scholar] [CrossRef]
- Yang, Z.; Xia, Y.; Mokaya, R. Enhanced hydrogen storage capacity of high surface area zeolite-like carbon materials. J. Am. Chem. Soc. 2007, 129, 1673–1679. [Google Scholar] [CrossRef]
- Dimitrakakis, G.K.; Tylianakis, E.; Froudakis, G.E. Pillared graphene: A new 3-D network nanostructure for enhanced hydrogen storage. Nano Lett. 2008, 8, 3166–3170. [Google Scholar] [CrossRef]
- Assfour, B.; Leoni, S.; Seifert, G.; Baburin, I.A. Packings of carbon nanotubes—New materials for hydrogen storage. Adv. Mater. 2011, 23, 1237–1241. [Google Scholar] [CrossRef]
- Mahdizadeh, S.J.; Goharshadi, E.K. Hydrogen storage on silicon, carbon, and silicon carbide nanotubes: A combined quantum mechanics and grand canonical Monte Carlo simulation study. Int. J. Hydrog. Energy 2014, 39, 1719–1731. [Google Scholar] [CrossRef]
- Minami, D.; Ohkubo, T.; Kuroda, Y.; Sakai, K.; Sakai, H.; Abe, M. Structural optimization of arranged carbon nanotubes for hydrogen storage by grand canonical Monte Carlo simulation. Int. J. Hydrog. Energy 2010, 35, 12398–12404. [Google Scholar] [CrossRef]
- Gayathri, V.; Devi, N.R.; Geetha, R. Hydrogen storage in coiled carbon nanotubes. Int. J. Hydrog. Energy 2010, 35, 1313–1320. [Google Scholar] [CrossRef]
- Shalabi, A.S.; Halim, W.S.A.; Aal, S.A.; Soliman, K.A. Tuning hydrogen storage of carbon nanotubes by mechanical bending: theoretical study. Mol. Simul. 2016, 42, 709–714. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, Q.; Yan, Y.; Cheng, X.; Liu, Y. Influence of sample cell physisorption on measurements of hydrogen storage of carbon materials using a Sieverts apparatus. Carbon 2010, 48, 714–720. [Google Scholar] [CrossRef]
- García-García, F.R.; Pérez-Cabero, M.; Nevskaia, D.M.; Rodríguez-Ramos, I.; Guerrero-Ruiz, A. Improving the synthesis of high purity carbon nanotubes in a catalytic fluidized bed reactor and their comparative test for hydrogen adsorption capacity. Catal. Today 2008, 133–135, 815–821. [Google Scholar] [CrossRef]
- Liu, C.; Chen, Y.; Wu, C.Z.; Xu, S.T.; Cheng, H.M. Hydrogen storage in carbon nanotubes revisited. Carbon 2010, 48, 452–455. [Google Scholar] [CrossRef]
- Sudibandriyo, M.; Wulan, P.P.D.K.; Prasodjo, P. Adsorption capacity and its dynamic behavior of the hydrogen storage on carbon nanotubes. Int. J. Technol. 2015, 6, 1128–1136. [Google Scholar] [CrossRef]
- Yang, R.T. Hydrogen storage by alkali-doped carbon nanotubes-revisited. Carbon 2000, 38, 623–626. [Google Scholar] [CrossRef]
- Gao, L.Z.; Yoo, E.; Nakamura, J.; Zhang, W.K.; Chua, H.T. Hydrogen storage in Pd-Ni doped defective carbon nanotubes through the formation of CHx (x=1, 2). Carbon 2010, 48, 3250–3255. [Google Scholar] [CrossRef]
- Bianco, S.; Giorcelli, M.; Musso, S.; Castellino, M.; Agresti, F.; Khandelwal, A.; Lo Russo, S.; Kumar, M.; Ando, Y.; Tagliaferro, A. Hydrogen adsorption in several types of carbon nanotubes. J. Nanosci. Nanotechnol. 2010, 10, 3860–3866. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, K.; Roth, S.; Hirscher, M.; Grünwald, W. Carbon nanostructures: An efficient hydrogen storage medium for fuel cells. Fuel Cells Bulletin 2001, 4, 9–12. [Google Scholar] [CrossRef]
- Zhu, H.; Li, X.; Ci, L.; Xu, C.; Mao, Z.; Liang, J.; Wu, D. Hydrogen storage characteristics of carbon nanotubes after graphitization by floating catalytic. Sci. China Ser. E–Technol. Sci. 2002, 32, 152–157. (In Chinese) [Google Scholar] [CrossRef]
- Zheng, H.; Wang, Z.; Cheng, H. Effect of chemical potential on simulation calculationhydrogen storage of single-walled carbon nanotubes. Sci. China Ser. B 2003, 33, 467–473. (In Chinese) [Google Scholar] [CrossRef]
- Zhou, Z.; Wu, X.; Wang, Y.; Lin, G.; Zhang, H. Characteristics of adsorption and desorption of hydrogen on multi-walled carbon nanotubes. Acta Phys.-Chim. Sin. 2002, 18, 692–698. (In Chinese) [Google Scholar]
- Yang, F.H.; Yang, R.T. Ab initio molecular orbital study of adsorption of atomic hydrogen on graphite:: Insight into hydrogen storage in carbon nanotubes. Carbon 2002, 40, 437–444. [Google Scholar] [CrossRef]
- Reyhani, A.; Mortazavi, S.Z.; Mirershadi, S.; Moshfegh, A.Z.; Parvin, P.; Golikand, A.N. Hydrogen storage in decorated multiwalled carbon nanotubes by Ca, Co, Fe, Ni, and Pd nanoparticles under ambient conditions. J. Phys. Chem. C 2011, 115, 6994–7001. [Google Scholar] [CrossRef]
- Cheng, J.; Yan, H.; Chen, Y.; Zhang, L.; Zhao, L.; Huang, D.; Tang, R. Computer simulations of hydrogen storage in single–walled carbon nanotubes. Chin. J. Comput. Phys. 2003, 20, 255–258. (In Chinese) [Google Scholar] [CrossRef]
- Shen, C.; Hu, Y.T.; Zhou, S.; Ma, X.L.; Li, H. The grand canonical Monte Carlo simulation of hydrogen physisorption on single-walled carbon nanotubes at the low and normal temperatures. Acta Phys. Sin. 2013, 10.7498/aps.62.038801, 462–471. (In Chinese) [Google Scholar] [CrossRef]
- Oriňáková, R.; Oriňák, A. Recent applications of carbon nanotubes in hydrogen production and storage. Fuel 2011, 90, 3123–3140. [Google Scholar] [CrossRef]
- Zheng, Q.; Gu, A.; Lu, X.; Lin, W. Determination of isosteric heat of adsorption of hydrogen on carbon-based adsorbents with nonlocal density functional theory approach. J. Chem. Ind. Eng. (China Chin. Ed.) 2003, 54, 995–1000. (In Chinese) [Google Scholar] [CrossRef]
- Zheng, Q.; Gu, A.; Lin, W.; Li, M.; Lu, X. Theoretical study on the adsorbed hydrogen molecules on multi walled carbon nanotubes. Acta Phys.-Chim. Sin. 2003, 19, 139–143. (In Chinese) [Google Scholar]
- Ferre–Vilaplana, A. Ab initio computational investigation of physisorption of molecular hydrogen on achiral single-walled carbon nanotubes. J. Chem. Phys. 2005, 122, 214724. [Google Scholar] [CrossRef] [PubMed]
- Arellano, J.S.; Molina, L.M.; Rubio, A.; López, M.J.; Alonso, J.A. Interaction of molecular and atomic hydrogen with (5,5) and (6,6) single-wall carbon nanotubes. J. Chem. Phys. 2002, 117, 2281–2288. [Google Scholar] [CrossRef]
- Dinadayalane, T.C.; Leszczynski, J. Toward understanding of hydrogen storage in single-walled carbon nanotubes by investigations of chemisorption mechanism. Pract. Aspects Comput. Chem. Methods Concepts Appl. 2009, 297–313. [Google Scholar] [CrossRef]
- Brieno-Enriquez, K.M.; Ledesma-Garcia, J.; Perez-Bueno, J.J.; Godinez, L.A.; Terrones, H.; Angeles-Chavez, C. Bonding titanium on multi-walled carbon nanotubes for hydrogen storage: An electrochemical approach. Mater. Chem. Phys. 2009, 115, 521–525. [Google Scholar] [CrossRef]
- Yan, S.Y.; Yang, Z.Q. The mechanism of hydrogen storage in single-walled carbon nanotube. J. At. Mol. Phys. 2012, 29, 941–947. (In Chinese) [Google Scholar] [CrossRef]
- Zhang, H.Y.; Fu, X.J.; Chen, Y.M.; Yi, S.P.; Li, S.H.; Zhu, Y.L.; Li, W. The electrochemical hydrogen storage of multi-walled carbon nanotubes synthesized by chemical vapor deposition using a lanthanum nickel hydrogen storage alloy as catalyst. Physica B 2004, 352, 66–72. [Google Scholar] [CrossRef]
- Yürüm, Y.; Taralp, A.; Veziroglu, T.N. Storage of hydrogen in nanostructured carbon materials. Int. J. Hydrog. Energy 2009, 34, 3784–3798. [Google Scholar] [CrossRef]
- Reddy, A.L.M.; Ramaprabhu, S. Hydrogen adsorption properties of single-walled carbon nanotube-nanocrystalline platinum composites. Int. J. Hydrog. Energy 2008, 33, 1028–1034. [Google Scholar] [CrossRef]
- Panella, B.; Hirscher, M.; Roth, S. Hydrogen adsorption in different carbon nanostructures. Carbon 2005, 43, 2209–2214. [Google Scholar] [CrossRef]
- Challet, S.; Azaïs, P.; Pellenq, R.J.M.; Isnard, O.; Soubeyroux, J.L.; Duclaux, L. Hydrogen adsorption in microporous alkali-doped carbons (activated carbon and single wall nanotubes). J. Phys. Chem. Solids 2004, 65, 541–544. [Google Scholar] [CrossRef]
- Dogan, E.E.; Tokcan, P.; Kizilduman, B.K. Storage of hydrogen in activated carbons and carbon nanotubes. Adv. Mater. Sci. 2018, 18, 5–16. [Google Scholar] [CrossRef]
- Wen, Y.W.; Liu, H.J.; Pan, L.; Tan, X.J.; Lv, H.Y.; Shi, J.; Tang, X.F. A triplet form of (5,0) carbon nanotube with higher hydrogen storage capacity. J. Phys. Chem. C 2011, 115, 9227–9231. [Google Scholar] [CrossRef]
- Rather, S.U. Trimetallic catalyst synthesized multi-walled carbon nanotubes and their application for hydrogen storage. Korean J. Chem. Eng. 2016, 33, 1551–1556. [Google Scholar] [CrossRef]
- Barghi, S.H.; Tsotsis, T.T.; Sahimi, M. Hydrogen sorption hysteresis and superior storage capacity of silicon-carbide nanotubes over their carbon counterparts. Int. J. Hydrog. Energy 2014, 39, 21107–21115. [Google Scholar] [CrossRef]
- Yang, W.Y.; Chen, Y.; Wang, J.F.; Peng, T.J.; Xu, J.H.; Yang, B.C.; Tang, K. Reduced graphene oxide/carbon nanotube composites as electrochemical energy storage electrode applications. Nanoscale Res. Lett. 2018, 13, 181. [Google Scholar] [CrossRef]
- Wang, J.G.; Liu, H.Z.; Zhang, X.Y.; Li, X.; Liu, X.R.; Kang, F.Y. Green synthesis of hierarchically porous carbon nanotubes as advanced materials for high-efficient energy storage. Small 2018, 14, 1703950. [Google Scholar] [CrossRef]
- Kumar, S.; Nehra, M.; Kedia, D.; Dilbaghi, N.; Tankeshwar, K.; Kim, K.H. Carbon nanotubes: A potential material for energy conversion and storage. Prog. Energy Combust. Sci. 2018, 64, 219–253. [Google Scholar] [CrossRef]
- Gao, Z.; Song, N.N.; Zhang, Y.Y.; Schwab, Y.; He, J.J.; Li, X.D. Carbon nanotubes derived from yeast-fermented wheat flour and their energy storage application. ACS Sustain. Chem. Eng. 2018, 6, 11386–11396. [Google Scholar] [CrossRef]
- Banerjee, D.; Ghorai, U.K.; Das, N.S.; Das, B.; Thakur, S.; Chattopadhyay, K.K. Amorphous carbon nanotubes-nickel oxide nanoflower hybrids: A low cost energy storage material. ACS Omega 2018, 3, 6311–6320. [Google Scholar] [CrossRef]
- Sun, L.M.; Wang, X.H.; Wang, Y.R.; Zhang, Q. Roles of carbon nanotubes in novel energy storage devices. Carbon 2017, 122, 462–474. [Google Scholar] [CrossRef]
- Rana, M.; Asim, S.; Hao, B.; Yang, S.D.; Ma, P.C. Carbon nanotubes on highly interconnected carbonized cotton for flexible and light-weight energy storage. Adv. Sustain. Syst. 2017, 1, 9. [Google Scholar] [CrossRef]
- Lu, Z.; Chao, Y.F.; Ge, Y.; Foroughi, J.; Zhao, Y.; Wang, C.Y.; Long, H.R.; Wallace, G.G. High-performance hybrid carbon nanotube fibers for wearable energy storage. Nanoscale 2017, 9, 5063–5071. [Google Scholar] [CrossRef]
- Lee, J.S.; Jun, J.; Cho, S.; Kim, W.; Jang, J. Electrospun three-layered polymer nanofiberbased porous carbon nanotubes for high-capacity energy storage. RSC Adv. 2017, 7, 201–207. [Google Scholar] [CrossRef]
- Hong, S.; Lee, J.; Do, K.; Lee, M.; Kim, J.H.; Lee, S.; Kim, D.H. Stretchable electrode based on laterally combed carbon nanotubes for wearable energy harvesting and storage devices. Adv. Funct. Mater. 2017, 27, 1704353. [Google Scholar] [CrossRef]
- Sankaran, M.; Viswanathan, B. The role of heteroatoms in carbon nanotubes for hydrogen storage. Carbon 2006, 44, 2816–2821. [Google Scholar] [CrossRef]
- Liu, F.; Zhang, X.; Cheng, J.; Tu, J.; Kong, F.; Huang, W.; Chen, C. Preparation of short carbon nanotubes by mechanical ball milling and their hydrogen adsorption behavior. Carbon 2003, 41, 2527–2532. [Google Scholar] [CrossRef]
- Lee, S.Y.; Park, S.J. Effect of chemical treatments on hydrogen storage behaviors of multi-walled carbon nanotubes. Mater. Chem. Phys. 2010, 124, 1011–1014. [Google Scholar] [CrossRef]
- Lee, S.Y.; Rhee, K.Y.; Nahm, S.H.; Park, S.J. Effect of p-type multi-walled carbon nanotubes for improving hydrogen storage behaviors. J. Solid State Chem. 2014, 210, 256–260. [Google Scholar] [CrossRef]
- Yan, H.; Chen, Y.; Cheng, J. The distribution of hydrogen storage in carbon nanotubes. J. Anhui Univ. (Nat. Sci.) 2002, 26, 71–73. (In Chinese) [Google Scholar] [CrossRef]
- Bénard, P.; Chahine, R. Storage of hydrogen by physisorption on carbon and nanostructured materials. Scr. Mater. 2007, 56, 803–808. [Google Scholar] [CrossRef]
- Yan, X.; Guo, X.; Wang, D.; Sun, B. The Study on Mechanism of Hydrogen Storage of Carbon Nanotubes. Lab. Sci. 2007, 69–70. (In Chinese) [Google Scholar] [CrossRef]
- Ghosh, S.; Padmanabhan, V. Hydrogen storage capacity of bundles of single-walled carbon nanotubes with defects. Int. J. Energy Res. 2017, 41, 1108–1117. [Google Scholar] [CrossRef]
- Cheng, J.; Yuan, X.; Zhao, L.; Huang, D.; Zhao, M.; Dai, L.; Ding, R. GCMC simulation of hydrogen physisorption on carbon nanotubes and nanotube arrays. Carbon 2004, 42, 2019–2024. [Google Scholar] [CrossRef]
- Shen, K.; Xu, H.; Jiang, Y.; Pietraß, T. The role of carbon nanotube structure in purification and hydrogen adsorption. Carbon 2004, 42, 2315–2322. [Google Scholar] [CrossRef]
- Cheng, J.; Dai, L.; Zhao, M.; Ding, R.; Huang, D. The properties of hydrogen physisorption in carbon nanotube arrays. J. Anhui Univ. (Nat. Sci.) 2004, 28, 32–36. (In Chinese) [Google Scholar] [CrossRef]
- Zhang, L.; Cheng, J.; Zhao, L.; Huang, D. Computer simulation study on the hydrogen storage in carbon nanotubes advanced materials research. J. Anhui Inst. Archit. Ind. (Nat. Sci.) 2004, 12, 34–39. (In Chinese) [Google Scholar] [CrossRef]
- Muniz, A.R.; Meyyappan, M.; Maroudas, D. On the hydrogen storage capacity of carbon nanotube bundles. Appl. Phys. Lett. 2009, 95, 162111. [Google Scholar] [CrossRef]
- Yuan, X.; Cheng, J.; Huang, D.; Zhao, L.; Zhang, L.; Zhao, M.; Dai, L. The potential effect and space effect of hydrogen physisorption in carbon nanotubes. J. Anhui Univ. (Nat. Sci.) 2004, 28, 38–42. (In Chinese) [Google Scholar] [CrossRef]
- Zhang, L.; Cheng, J.; Huang, D.; Zhao, L. Study on Distribution of Potential Field in Carbon Nanotubes before and after Storing Hydrogen. J. Anhui Tech. Teach. Coll. 2004, 18, 50–52. (In Chinese) [Google Scholar] [CrossRef]
- Brzhezinskaya, M.; Belenkov, E.A.; Greshnyakov, V.A.; Yalovega, G.E.; Bashkin, I.O. New aspects in the study of carbon-hydrogen interaction in hydrogenated carbon nanotubes for energy storage applications. J. Alloy. Compd. 2019, 792, 713–720. [Google Scholar] [CrossRef]
- Wang, Y.J.; Wang, L.Y.; Wang, S.B.; Wu, L.; Jiao, Q.Z. Hydrogen storage of Carbon nanotubes: Theoretical Studies. Adv. Mater. Res. 2011, 179–180, 722–727. [Google Scholar] [CrossRef]
- Tang, C.Y.; Man, C.Z.; Chen, Y.G.; Yang, F.; Luo, L.S.; Liu, Z.F.; Mei, J.; Lau, W.M.; Wong, K.W. Realizing the Storage of Pressurized Hydrogen in Carbon Nanotubes Sealed with Aqueous Valves. Energy Technol. 2013, 1, 309–312. [Google Scholar] [CrossRef]
- Zhao, M. Molecular Dynamics Study on Hydrogen Storage Characteristics in Carbon Nanotubes. J. Hefei Norm. Univ. 2014, 3, 7. (In Chinese) [Google Scholar]
- Darkrim, F.L.; Malbrunot, P.; Tartaglia, G.P. Review of hydrogen storage by adsorption in carbon nanotubes. Int. J. Hydrog. Energy 2002, 27, 193–202. [Google Scholar] [CrossRef]
- Zhao, T.K.; Ji, X.L.; Jin, W.B.; Yang, W.B.; Li, T.H. Hydrogen storage capacity of single-walled carbon nanotube prepared by a modified arc discharge. Fuller. Nanotub. Carbon Nanostruct. 2017, 25, 355–358. [Google Scholar] [CrossRef]
- Kaskun, S.; Kayfeci, M. The synthesized nickel-doped multi-walled carbon nanotubes for hydrogen storage under moderate pressures. Int. J. Hydrog. Energy 2018, 43, 10773–10778. [Google Scholar] [CrossRef]
- Safa, S.; Larijani, M.M.; Fathollahi, V.; Kakuee, O.R. Investigating Hydrogen Storage Behavior of Carbon Nanotubes at Ambient Temperature and above by Ion Beam Analysis. Nano 2010, 5, 341–347. [Google Scholar] [CrossRef]
- Ströbel, R.; Garche, J.; Moseley, P.T.; Jörissen, L.; Wolf, G. Hydrogen storage by carbon materials. J. Power Sources 2006, 159, 781–801. [Google Scholar] [CrossRef]
- Liu, Z.L.; Xue, Q.Z.; Ling, C.C.; Yan, Z.F.; Zheng, J.T. Hydrogen storage and release by bending carbon nanotubes. Comput. Mater. Sci. 2013, 68, 121–126. [Google Scholar] [CrossRef]
- Seenithurai, S.; Pandyan, R.K.; Kumar, S.V.; Saranya, C.; Mahendran, M. Al-decorated carbon nanotube as the molecular hydrogen storage medium. Int. J. Hydrog. Energy 2014, 39, 11990–11998. [Google Scholar] [CrossRef]
- Zhou, L.; Zhou, Y.; Sun, Y. Studies on the mechanism and capacity of hydrogen uptake by physisorption-based materials. Int. J. Hydrog. Energy 2006, 31, 259–264. [Google Scholar] [CrossRef]
- Sudan, P.; Züttel, A.; Mauron, P.; Emmenegger, C.; Wenger, P.; Schlapbach, L. Physisorption of hydrogen in single-walled carbon nanotubes. Carbon 2003, 41, 2377–2383. [Google Scholar] [CrossRef]
- Lee, S.Y.; Park, S.J. Influence of the pore size in multi-walled carbon nanotubes on the hydrogen storage behaviors. J. Solid State Chem. 2012, 194, 307–312. [Google Scholar] [CrossRef]
- Rakhi, R.B.; Sethupathi, K.; Ramaprabhu, S. Synthesis and hydrogen storage properties of carbon nanotubes. Int. J. Hydrog. Energy 2008, 33, 381–386. [Google Scholar] [CrossRef]
- Adeniran, B.; Mokaya, R. Low temperature synthesized carbon nanotube superstructures with superior CO2 and hydrogen storage capacity. J. Mater. Chem. A 2015, 3, 5148–5161. [Google Scholar] [CrossRef]
- Silambarasan, D.; Surya, V.J.; Iyakutti, K.; Asokan, K.; Vasu, V.; Kawazoe, Y. Gamma (gamma)-ray irradiated multi-walled carbon nanotubes (MWCNTs) for hydrogen storage. Appl. Surf. Sci. 2017, 418, 49–55. [Google Scholar] [CrossRef]
- Geng, H.Z.; Kim, T.H.; Lim, S.C.; Jeong, H.K.; Jin, M.H.; Jo, Y.W.; Lee, Y.H. Hydrogen storage in microwave-treated multi-walled carbon nanotubes. Int. J. Hydrog. Energy 2010, 35, 2073–2082. [Google Scholar] [CrossRef]
- Lin, K.Y.; Chang, J.K.; Chen, C.Y.; Tsai, W.T. Effects of heat treatment on materials characteristics and hydrogen storage capability of multi-wall carbon nanotubes. Diam. Relat. Mat. 2009, 18, 553–556. [Google Scholar] [CrossRef]
- Lee, J.H.; Rhee, K.Y.; Park, S.J. Effects of cryomilling on the structures and hydrogen storage characteristics of multi-walled carbon nanotubes. Int. J. Hydrog. Energy 2010, 35, 7850–7857. [Google Scholar] [CrossRef]
- Zhao, T.K.; Li, G.M.; Liu, L.H.; Du, L.; Liu, Y.N.; Li, T.H. Hydrogen Storage Behavior of Amorphous Carbon Nanotubes at Low Pressure and Room Temperature. Fuller. Nanotub. Carbon Nanostruct. 2011, 19, 677–683. [Google Scholar] [CrossRef]
- Onishchenko, D.V.; Reva, V.P. Use of multilayer carbon nanotubes made from plant materials as sorption-based hydrogen storage containers. Chem. Tech. Fuels Oils 2013, 49, 281–286. [Google Scholar] [CrossRef]
- Uvarova, I.Y.; Basnukaeva, R.M.; Dolbin, A.V.; Danilchenko, B.A.; IEEE. Hydrogen Storage Capacity of Carbon Nanotubes gamma—Irradiated in Hydrogen and Deuterium Media. In 2013 Proceedings of the 36th International Spring Seminar on Electronics Technology; IEEE: New York, NY, USA, 2013; pp. 309–313. [Google Scholar]
- Mirershadi, S.; Reyhani, A.; Mortazavi, S.Z.; Safibonab, B.; Esfahani, M.K. The effects of bromine treatment on the hydrogen storage properties of multi-walled carbon nanotubes. Int. J. Hydrog. Energy 2011, 36, 15622–15631. [Google Scholar] [CrossRef]
- Im, J.S.; Kang, S.C.; Bai, B.C.; Suh, J.K.; Lee, Y.S. Effect of thermal fluorination on the hydrogen storage capacity of multi-walled carbon nanotubes. Int. J. Hydrog. Energy 2011, 36, 1560–1567. [Google Scholar] [CrossRef]
- Wu, H.M.; Wexler, D.; Ranjbartoreh, A.R.; Liu, H.K.; Wang, G.X. Chemical processing of double-walled carbon nanotubes for enhanced hydrogen storage. Int. J. Hydrog. Energy 2010, 35, 6345–6349. [Google Scholar] [CrossRef]
- Elyassi, M.; Rashidi, A.; Hantehzadeh, M.R.; Elahi, S.M. Hydrogen Storage Behaviors by Adsorption on Multi-Walled Carbon Nanotubes. J. Inorg. Organomet. Polym. Mater. 2017, 27, 285–295. [Google Scholar] [CrossRef]
- Uvarova, I.Y.; Basnukaeva, R.M.; Dolbin, A.V.; Danilchenko, B.A. Hydrogen Storage Capacity of Carbon Nanotubes gamma—Irradiated in Hydrogen and Deuterium Media. Int Spr Sem Elect Te 2013, 309–313. [Google Scholar] [CrossRef]
- Lee, S.Y.; Park, S.J. Effect of temperature on activated carbon nanotubes for hydrogen storage behaviors. Int. J. Hydrog. Energy 2010, 35, 6757–6762. [Google Scholar] [CrossRef]
- Dolbin, A.V.; Esel’son, V.B.; Gavrilko, V.G.; Manzhelii, V.G.; Vinnikov, N.A.; Yaskovets, I.I.; Uvarova, I.Y.; Tripachko, N.A.; Danilchenko, B.A. Hydrogen sorption by the bundles of single-wall carbon nanotubes, irradiated in various gas media. Low Temp. Phys. 2013, 39, 610–617. [Google Scholar] [CrossRef]
- Park, S.J.; Lee, S.Y. Hydrogen storage behaviors of platinum–supported multi-walled carbon nanotubes. Int. J. Hydrog. Energy 2010, 35, 13048–13054. [Google Scholar] [CrossRef]
- Lee, S.Y.; Park, S.J. Influence of CO2 activation on hydrogen storage behaviors of platinum-loaded activated carbon nanotubes. J. Solid State Chem. 2010, 183, 2951–2956. [Google Scholar] [CrossRef]
- Rashidi, A.M.; Nouralishahi, A.; Khodadadi, A.A.; Mortazavi, Y.; Karimi, A.; Kashefi, K. Modification of single wall carbon nanotubes (SWNT) for hydrogen storage. Int. J. Hydrog. Energy 2010, 35, 9489–9495. [Google Scholar] [CrossRef]
- Xiao, H.; Li, S.H.; Cao, J.X. First-principles study of Pd-decorated carbon nanotube for hydrogen storage. Chem. Phys. Lett. 2009, 483, 111–114. [Google Scholar] [CrossRef]
- Chen, J.; Wang, X.G.; Zhang, H.Y. Hydrogen Storage in Carbon Nanotubes Prepared by Using Copper Nanoparticles as Catalyst. New Trends Mech. Eng. Mater. 2013, 251–342. [Google Scholar] [CrossRef]
- Mosquera, E.; Diaz-Droguett, D.E.; Carvajal, N.; Roble, M.; Morel, M.; Espinoza, R. Characterization and hydrogen storage in multi-walled carbon nanotubes grown by aerosol-assisted CVD method. Diam. Relat. Mat. 2014, 43, 66–71. [Google Scholar] [CrossRef]
- Hirscher, M.; Becher, M.; Haluska, M.; Dettlaff-Weglikowska, U.; Quintel, A.; Duesberg, G.S.; Choi, Y.M.; Downes, P.; Hulman, M.; Roth, S.; et al. Hydrogen storage in sonicated carbon materials. Appl. Phys. A-Mater. Sci. Process. 2001, 72, 129–132. [Google Scholar] [CrossRef]
- Erunal, E.; Ulusal, F.; Aslan, M.Y.; Guzel, B.; Uner, D. Enhancement of hydrogen storage capacity of multi-walled carbon nanotubes with palladium doping prepared through supercritical CO2 deposition method. Int. J. Hydrog. Energy 2018, 43, 10755–10764. [Google Scholar] [CrossRef]
- Das, T.; Banerjee, S.; Dasgupta, K.; Joshi, J.B.; Sudarsan, V. Nature of the Pd-CNT interaction in Pd nanoparticles dispersed on multi-walled carbon nanotubes and its implications in hydrogen storage properties. RSC Adv. 2015, 5, 41468–41474. [Google Scholar] [CrossRef]
- Chen, C.H.; Huang, C.C. Effect of surface characteristics and catalyst loaded amount on hydrogen storage in carbon nanotubes. Microporous Mesoporous Mat. 2008, 112, 553–560. [Google Scholar] [CrossRef]
- Anikina, E.; Banerjee, A.; Beskachko, V.; Ahuja, R. Li-Functionalized Carbon Nanotubes for Hydrogen Storage: Importance of Size Effects. ACS Appl. Nano Mater. 2019, 2, 3021–3030. [Google Scholar] [CrossRef]
- Lu, J.L.; Xiao, H.; Cao, J.X. Mechanism for high hydrogen storage capacity on metal-coated carbon nanotubes: A first principle analysis. J. Solid State Chem. 2012, 196, 367–371. [Google Scholar] [CrossRef]
- Tian, Z.Y.; Liu, Y.T.; Wu, W.W.; Jiang, L.N.; Dong, S.L. Hydrogen storage of capped single-walled carbon nanotube via transition-metal doping. EPL 2013, 104, 36002. [Google Scholar] [CrossRef]
- Chang, J.K.; Chen, C.Y.; Tsai, W.T. Decorating carbon nanotubes with nanoparticles using a facile redox displacement reaction and an evaluation of synergistic hydrogen storage performance. Nanotechnology 2009, 20, 495603. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.; Lin, K.Y.; Tsai, W.T.; Chang, J.K.; Tseng, C.M. Electroless deposition of Ni nanoparticles on carbon nanotubes with the aid of supercritical CO2 fluid and a synergistic hydrogen storage property of the composite. Int. J. Hydrog. Energy 2010, 35, 5490–5497. [Google Scholar] [CrossRef]
- Yang, L.; Yu, L.L.; Wei, H.W.; Li, W.Q.; Zhou, X.; Tian, W.Q. Hydrogen storage of dual-Ti-doped single-walled carbon nanotubes. Int. J. Hydrog. Energy 2019, 44, 2960–2975. [Google Scholar] [CrossRef]
- Silambarasan, D.; Surya, V.J.; Vasu, V.; Iyakutti, K. Investigation of single-walled carbon nanotubes-titanium metal composite as a possible hydrogen storage medium. Int. J. Hydrog. Energy 2013, 38, 14654–14660. [Google Scholar] [CrossRef]
- Lin, K.Y.; Tsai, W.T.; Chang, J.K. Decorating carbon nanotubes with Ni particles using an electroless deposition technique for hydrogen storage applications. Int. J. Hydrog. Energy 2010, 35, 7555–7562. [Google Scholar] [CrossRef]
- Wu, H.M.; Wexler, D.; Liu, H.K. Effect of different reductants for palladium loading on hydrogen storage capacity of double-walled carbon nanotubes. Int. J. Hydrog. Energy 2011, 36, 9032–9036. [Google Scholar] [CrossRef]
- Wenelska, K.; Michalkiewicz, B.; Chen, X.C.; Mijowska, E. Pd nanoparticles with tunable diameter deposited on carbon nanotubes with enhanced hydrogen storage capacity. Energy 2014, 75, 549–554. [Google Scholar] [CrossRef]
- Kumar, R.; Oh, J.H.; Kim, H.J.; Jung, J.H.; Jung, C.H.; Hong, W.G.; Kim, H.J.; Park, J.Y.; Oh, I.K. Nanohole-Structured and Palladium–Embedded 3D Porous Graphene for Ultrahigh Hydrogen Storage and CO Oxidation Multifunctionalities. ACS Nano 2015, 9, 7343–7351. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Corral, I.; Irigoyen, B.; Juan, A. Bonding in PdH2 and Pd2H2 systems adsorbed on carbon nanotubes: Implications for hydrogen storage. Int. J. Hydrog. Energy 2014, 39, 8780–8790. [Google Scholar] [CrossRef]
- Wang, Y.; Li, A.; Wang, K.A.; Guan, C.; Deng, W.Q.; Li, C.M.; Wang, X. Reversible hydrogen storage of multi-wall carbon nanotubes doped with atomically dispersed lithium. J. Mater. Chem. 2010, 20, 6490–6494. [Google Scholar] [CrossRef]
- Tian, Z.Y.; Dong, S.L. Yttrium dispersion on capped carbon nanotube: Promising materials for hydrogen storage applications. Int. J. Hydrog. Energy 2016, 41, 1053–1059. [Google Scholar] [CrossRef]
- Chakraborty, B.; Modak, P.; Banerjee, S. Hydrogen Storage in Yttrium-Decorated Single Walled Carbon Nanotube. J. Phys. Chem. C 2012, 116, 22502–22508. [Google Scholar] [CrossRef]
- Verdinelli, V.; Juan, A.; German, E. Ruthenium decorated single walled carbon nanotube for molecular hydrogen storage: A first-principle study. Int. J. Hydrog. Energy 2019, 44, 8376–8383. [Google Scholar] [CrossRef]
- Panigrahi, P.; Naqvi, S.R.; Hankel, M.; Ahuja, R.; Hussain, T. Enriching the hydrogen storage capacity of carbon nanotube doped with polylithiated molecules. Appl. Surf. Sci. 2018, 444, 467–473. [Google Scholar] [CrossRef]
- Mananghaya, M.; Yu, D.; Santos, G.N.; Rodulfo, E. Scandium and Titanium Containing Single-Walled Carbon Nanotubes for Hydrogen Storage: a Thermodynamic and First Principle Calculation. Sci Rep 2016, 6, 27370. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zhao, Y.H.; Li, Y.; Jiang, Q.; Lavernia, E.J. Enhanced Hydrogen Storage on Li-Dispersed Carbon Nanotubes. J. Phys. Chem. C 2009, 113, 2028–2033. [Google Scholar] [CrossRef]
- Mehrabi, M.; Parvin, P.; Reyhani, A.; Mortazavi, S.Z. Hydrogen storage in multi-walled carbon nanotubes decorated with palladium nanoparticles using laser ablation/chemical reduction methods. Mater. Res. Express 2017, 4, 095030. [Google Scholar] [CrossRef]
- Banerjee, S.; Dasgupta, K.; Kumar, A.; Ruz, P.; Vishwanadh, B.; Joshi, J.B.; Sudarsan, V. Comparative evaluation of hydrogen storage behavior of Pd doped carbon nanotubes prepared by wet impregnation and polyol methods. Int. J. Hydrog. Energy 2015, 40, 3268–3276. [Google Scholar] [CrossRef]
- Sheng, Q.; Wu, H.M.; Wexler, D.; Liu, H.K. Effects of Reducing Temperatures on the Hydrogen Storage Capacity of Double-Walled Carbon Nanotubes with Pd Loading. J. Nanosci. Nanotechnol. 2014, 14, 4706–4709. [Google Scholar] [CrossRef] [PubMed]
- Mortazavi, S.Z.; Parvin, P.; Reyhani, A.; Malekfar, R.; Mirershadi, S. Hydrogen storage property of laser induced Pd-nanoparticle decorated multi-walled carbon nanotubes. RSC Adv. 2013, 3, 1397–1409. [Google Scholar] [CrossRef]
- Wu, H.M.; Wexler, D.; Liu, H. Effects of different palladium content loading on the hydrogen storage capacity of double-walled carbon nanotubes. Int. J. Hydrog. Energy 2012, 37, 5686–5690. [Google Scholar] [CrossRef]
- Singh, P.; Kulkarni, M.V.; Gokhale, S.P.; Chikkali, S.H.; Kulkarni, C.V. Enhancing the hydrogen storage capacity of Pd-functionalized multi-walled carbon nanotubes. Appl. Surf. Sci. 2012, 258, 3405–3409. [Google Scholar] [CrossRef]
- Lin, K.S.; Mai, Y.J.; Li, S.R.; Shu, C.W.; Wang, C.H. Characterization and Hydrogen Storage of Surface-Modified Multiwalled Carbon Nanotubes for Fuel Cell Application. J. Nanomater. 2012, 2012. [Google Scholar] [CrossRef]
- Chen, C.Y.; Chang, J.K.; Tsai, W.T. Improved hydrogen storage performance of defected carbon nanotubes with Pd spillover catalysts dispersed using supercritical CO2 fluid. Int. J. Hydrog. Energy 2012, 37, 3305–3312. [Google Scholar] [CrossRef]
- Vermisoglou, E.C.; Labropoulos, A.; Romanos, G.E.; Kouvelos, E.; Papageorgiou, S.; Karanikolos, G.N.; Katsaros, F.; Kanellopoulos, N.K. Hydrogen Storage in Polymer-Functionalized Pd-Decorated Single Wall Carbon Nanotubes. J. Nanosci. Nanotechnol. 2010, 10, 5971–5980. [Google Scholar] [CrossRef]
- Shen, C.Q.; Aguey-Zinsou, K.F. Nanosized Magnesium Electrochemically Deposited on a Carbon Nanotubes Suspension: Synthesis and Hydrogen Storage. Front. Energy Res. 2017, 5, 27. [Google Scholar] [CrossRef]
- Reyhani, A.; Mortazavi, S.Z.; Mirershadi, S.; Golikand, A.N.; Moshfegh, A.Z. H2 adsorption mechanism in Mg modified multi-walled carbon nanotubes for hydrogen storage. Int. J. Hydrog. Energy 2012, 37, 1919–1926. [Google Scholar] [CrossRef]
- Pandyan, R.K.; Seenithurai, S.; Mahendran, M. Hydrogen storage in MgH2 coated single walled carbon nanotubes. Int. J. Hydrog. Energy 2011, 36, 3007–3015. [Google Scholar] [CrossRef]
- Mehrabi, M.; Reyhani, A.; Parvin, P.; Mortazavi, S.Z. Surface structural alteration of multi-walled carbon nanotubes decorated by nickel nanoparticles based on laser ablation/chemical reduction methods to enhance hydrogen storage properties. Int. J. Hydrog. Energy 2019, 44, 3812–3823. [Google Scholar] [CrossRef]
- Lee, S.Y.; Park, S.J. Hydrogen Storage Behaviors of Ni-loaded Activated Carbon Nanotubes. Adv. Mater. Res. 2010, 123–125, 695–698. [Google Scholar] [CrossRef]
- Ghosh, S.; Padmanabhan, V. Hydrogen storage in Titanium-doped single-walled carbon nanotubes with Stone-Wales defects. Diam. Relat. Mat. 2017, 77, 46–52. [Google Scholar] [CrossRef]
- Shevlin, S.A.; Guo, Z.X. High-Capacity Room-Temperature Hydrogen Storage in Carbon Nanotubes via Defect-Modulated Titanium Doping. J. Phys. Chem. C 2008, 112, 17456–17464. [Google Scholar] [CrossRef]
- Ni, M.Y.; Huang, L.F.; Guo, L.J.; Zeng, Z. Hydrogen storage in Li-doped charged single-walled carbon nanotubes. Int. J. Hydrog. Energy 2010, 35, 3546–3549. [Google Scholar] [CrossRef]
- Wang, H.L.; Cheng, X.L.; Zhang, H.; Tang, Y.J. Very High Hydrogen Storage Capacity of Al-Adsorbed Single-Walled Carbon Nanotube (Swcnt): Multi-Layered Structure of Hydrogen Molecules. Int. J. Mod. Phys. B 2013, 27, 1350061. [Google Scholar] [CrossRef]
- Iyakutti, K.; Kawazoe, Y.; Rajarajeswari, M.; Surya, V.J. Aluminum hydride coated single-walled carbon nanotube as a hydrogen storage medium. Int. J. Hydrog. Energy 2009, 34, 370–375. [Google Scholar] [CrossRef]
- Mei, F.; Ma, X.; Bie, Y.; Xu, G. Probing hydrogen adsorption behaviors of Ti- and Ni-decorated carbon nanotube by density functional theory. J. Theor. Comput. Chem. 2017, 16, 1750065. [Google Scholar] [CrossRef]
- Bhowmick, R.; Rajasekaran, S.; Friebel, D.; Beasley, C.; Jiao, L.; Ogasawara, H.; Dai, H.; Clemens, B.; Nilsson, A. Hydrogen spillover in Pt-single-walled carbon nanotube composites: formation of stable C—H bonds. J. Am. Chem. Soc. 2011, 133, 5580–5586. [Google Scholar] [CrossRef]
- Cheng, J.; Yuan, X.; Fang, X.; Zhang, L. Computer simulation of hydrogen physisorption in a Li-doped single walled carbon nanotube array. Carbon 2010, 48, 567–570. [Google Scholar] [CrossRef]
- Tylianakis, E.; Dimitrakakis, G.K.; Martin–Martinez, F.J.; Melchor, S.; Dobado, J.A.; Klontzas, E.; Froudakis, G.E. Designing novel nanoporous architectures of carbon nanotubes for hydrogen storage. Int. J. Hydrog. Energy 2014, 39, 9825–9829. [Google Scholar] [CrossRef]
- Suttisawat, Y.; Rangsunvigit, P.; Kitiyanan, B.; Williams, M.; Ndungu, P.; Lototskyy, M.V.; Nechaev, A.; Linkov, V.; Kulprathipanja, S. Investigation of hydrogen storage capacity of multi-walled carbon nanotubes deposited with Pd or V. Int. J. Hydrog. Energy 2009, 34, 6669–6675. [Google Scholar] [CrossRef]
- Chen, L.; Xia, K.S.; Huang, L.Z.; Li, L.W.; Pei, L.B.; Fei, S.X. Facile synthesis and hydrogen storage application of nitrogen-doped carbon nanotubes with bamboo-like structure. Int. J. Hydrog. Energy 2013, 38, 3297–3303. [Google Scholar] [CrossRef]
- Viswanathan, B.; Sankaran, M. Hetero-atoms as activation centers for hydrogen absorption in carbon nanotubes. Diam. Relat. Mat. 2009, 18, 429–432. [Google Scholar] [CrossRef]
- Ariharan, A.; Viswanathan, B.; Nandhakumar, V. Nitrogen-incorporated carbon nanotube derived from polystyrene and polypyrrole as hydrogen storage material. Int. J. Hydrog. Energy 2018, 43, 5077–5088. [Google Scholar] [CrossRef]
- Sharma, A.; Dasgupta, K.; Banerjee, S.; Patwardhan, A.; Srivastava, D.; Joshi, J.B. In-situ nitrogen doping in carbon nanotubes using a fluidized bed reactor and hydrogen storage behavior of the doped nanotubes. Int. J. Hydrog. Energy 2017, 42, 10047–10056. [Google Scholar] [CrossRef]
- Cho, J.H.; Yang, S.J.; Lee, K.; Park, C.R. Si-doping effect on the enhanced hydrogen storage of single walled carbon nanotubes and graphene. Int. J. Hydrog. Energy 2011, 36, 12286–12295. [Google Scholar] [CrossRef]
- Xiu-Ying, L.; Chao-Yang, W.; Yong-Jian, T.; Wei-Guo, S.; Wei-Dong, W.; Jia-Jing, X. Theoretical studies on hydrogen adsorption of single-walled boron-nitride and carbon nanotubes using grand canonical Monte Carlo method. Physica B 2009, 404, 1892–1896. [Google Scholar] [CrossRef]
- Rafiee, M.A. The Study of Hydrogen Storage in Carbon Nanotubes Using Calculated Nuclear Quadrupole Coupling Constant (NQCC) Parameters (A Theoretical Ab Initio Study). J. Comput. Theor. Nanosci. 2012, 9, 2021–2026. [Google Scholar] [CrossRef]
- Liu, P.; Liang, J.W.; Xue, R.H.; Du, Q.P.; Jiang, M.R. Ruthenium decorated boron–doped carbon nanotube for hydrogen storage: A first–principle study. Int. J. Hydrog. Energy 2019, 44, 27853–27861. [Google Scholar] [CrossRef]
- Zhou, J.G.; Williams, Q.L. Hydrogen Storage on Platinum-Decorated Carbon Nanotubes with Boron, Nitrogen Dopants or Sidewall Vacancies. J. Nano Res. 2011, 15, 29–40. [Google Scholar] [CrossRef]
- Wang, Y.S.; Li, M.; Wang, F.; Sun, Q.; Jia, Y. Li and Na Co-decorated carbon nitride nanotubes as promising new hydrogen storage media. Phys. Lett. A 2012, 376, 631–636. [Google Scholar] [CrossRef]
- Silambarasan, D.; Vasu, V.; Iyakutti, K.; Surya, V.J.; Ravindran, T.R. Reversible hydrogen storage in functionalized single-walled carbon nanotubes. Physica E 2014, 60, 75–79. [Google Scholar] [CrossRef]
- Surya, V.J.; Iyakutti, K.; Venkataramanan, N.S.; Mizuseki, H.; Kawazoe, Y. Single walled carbon nanotubes functionalized with hydrides as potential hydrogen storage media: A survey of intermolecular interactions. Phys. Status Solidi B-Basic Solid State Phys. 2011, 248, 2147–2158. [Google Scholar] [CrossRef]
- Surya, V.J.; Iyakutti, K.; Venkataramanan, N.; Mizuseki, H.; Kawazoe, Y. The role of Li and Ni metals in the adsorbate complex and their effect on the hydrogen storage capacity of single walled carbon nanotubes coated with metal hydrides, LiH and NiH2. Int. J. Hydrog. Energy 2010, 35, 2368–2376. [Google Scholar] [CrossRef]
- Silambarasan, D.; Surya, V.J.; Vasu, V.; Iyakutti, K. Experimental investigation of hydrogen storage in single walled carbon nanotubes functionalized with borane. Int. J. Hydrog. Energy 2011, 36, 3574–3579. [Google Scholar] [CrossRef]
- Surya, V.J.; Iyakutti, K.; Rajarajeswari, M.; Kawazoe, Y. First-Principles Study on Hydrogen Storage in Single Walled Carbon Nanotube Functionalized with Ammonia. J. Comput. Theor. Nanosci. 2010, 7, 552–557. [Google Scholar] [CrossRef]
- Lavanya, R.; Surya, V.J.; Lakshmi, I.; Iyakutti, K.; Vasu, V.; Mizuseki, H.; Kawazoe, Y. Hydrogen storage in TiO2 functionalized (10,10) single walled carbon nanotube (SWCNT)—First principles study. Int. J. Hydrog. Energy 2014, 39, 4973–4980. [Google Scholar] [CrossRef]
- Kanmani, M.; Lavanya, R.; Silambarasan, D.; Iyakutti, K.; Vasu, V.; Kawazoe, Y. First principles studies on hydrogen storage in single-walled carbon nanotube functionalized with TiO2. Solid State Commun. 2014, 183, 1–7. [Google Scholar] [CrossRef]
- Konni, M.; Mukkamala, S.B. Synthesis and hydrogen storage performance of Al2O3 nanoparticle decorated functionalized multi-walled carbon nanotubes (Al2O3@f-MWCNTs). J. Indian Chem. Soc. 2019, 96, 269–274. [Google Scholar]
- Salehabadi, A.; Salavati-Niasari, M.; Ghiyasiyan-Arani, M. Self-assembly of hydrogen storage materials based multi-walled carbon nanotubes (MWCNTs) and Dy3Fe5O12 (DFO) nanoparticles. J. Alloy. Compd. 2018, 745, 789–797. [Google Scholar] [CrossRef]
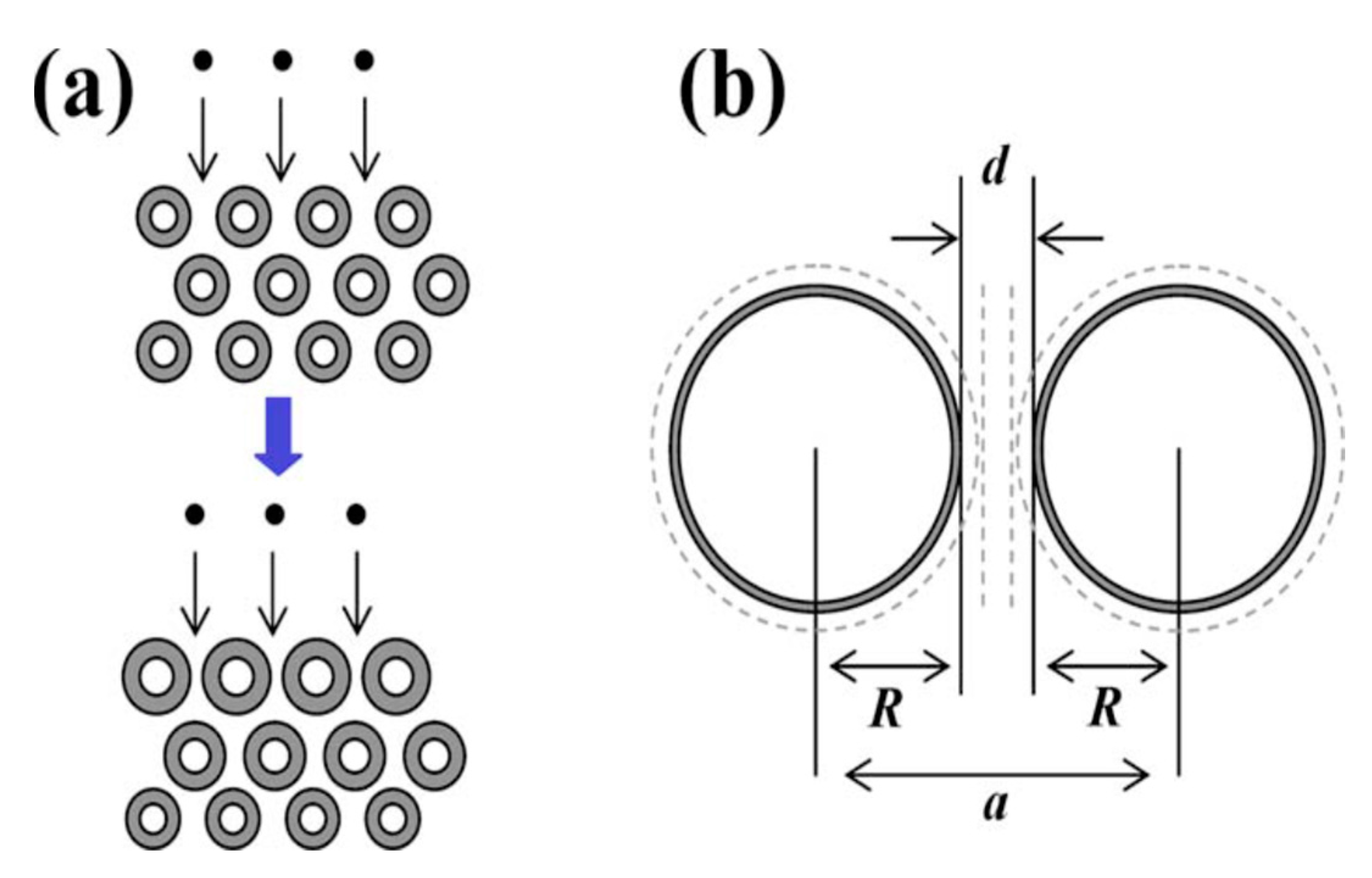
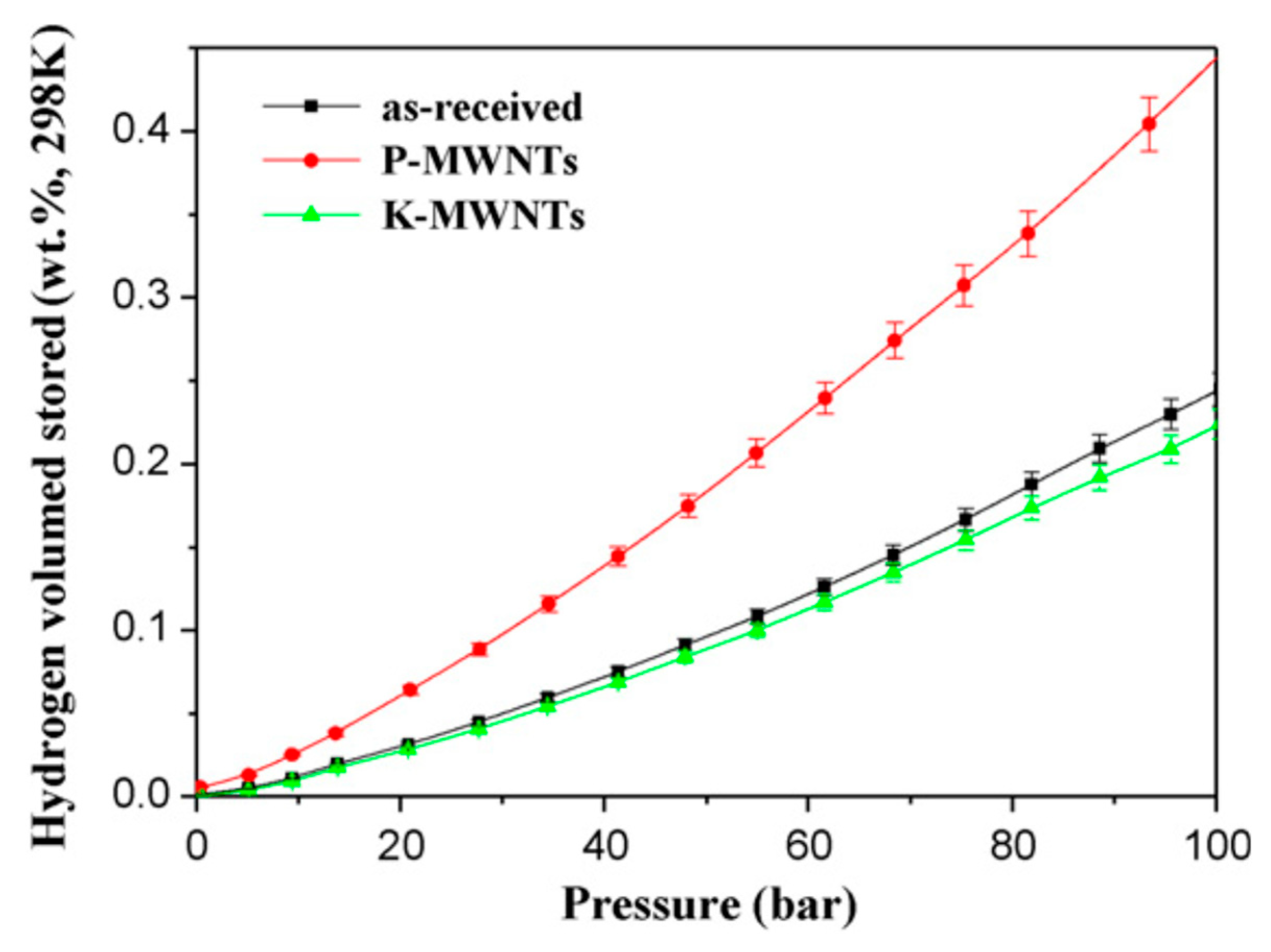
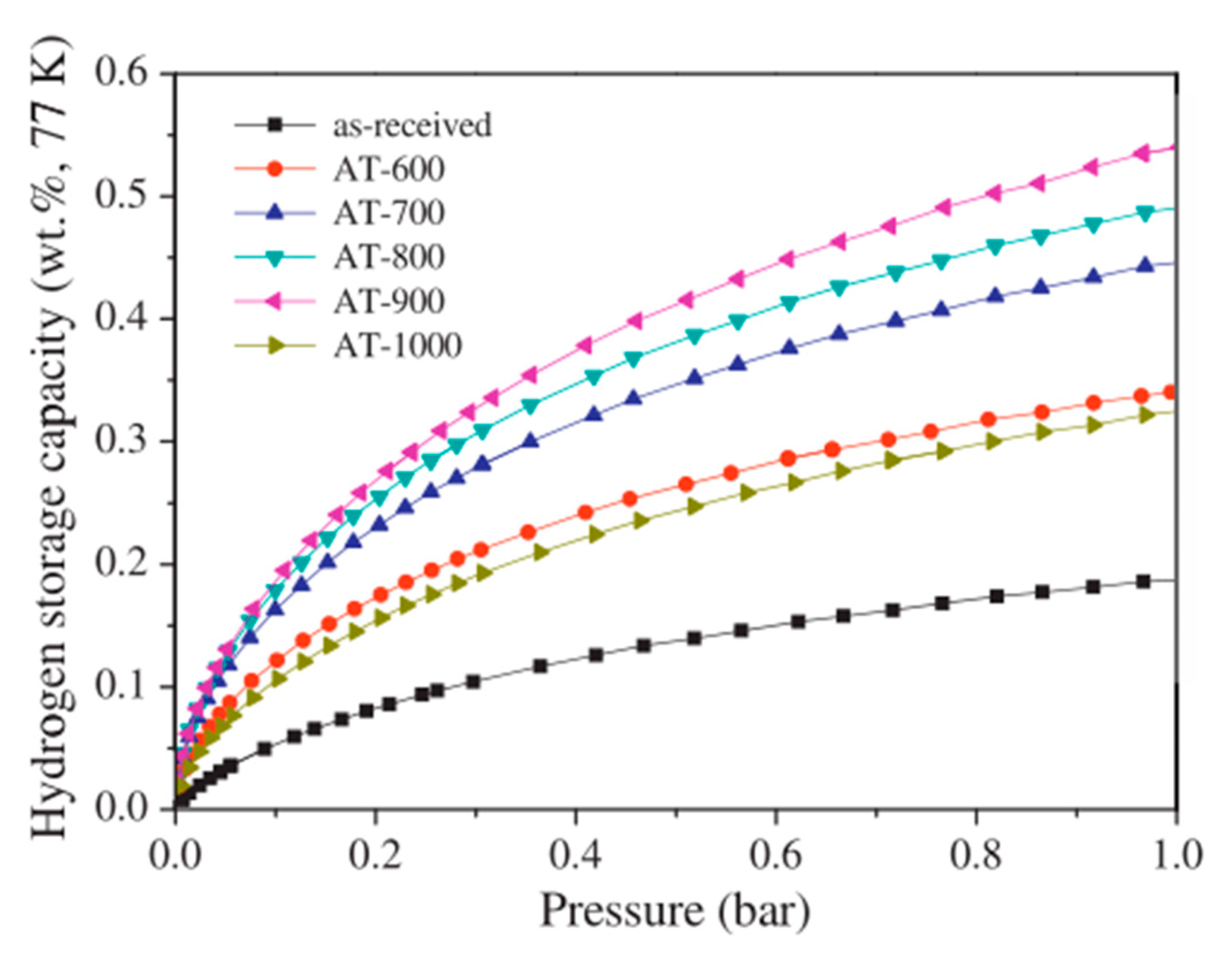
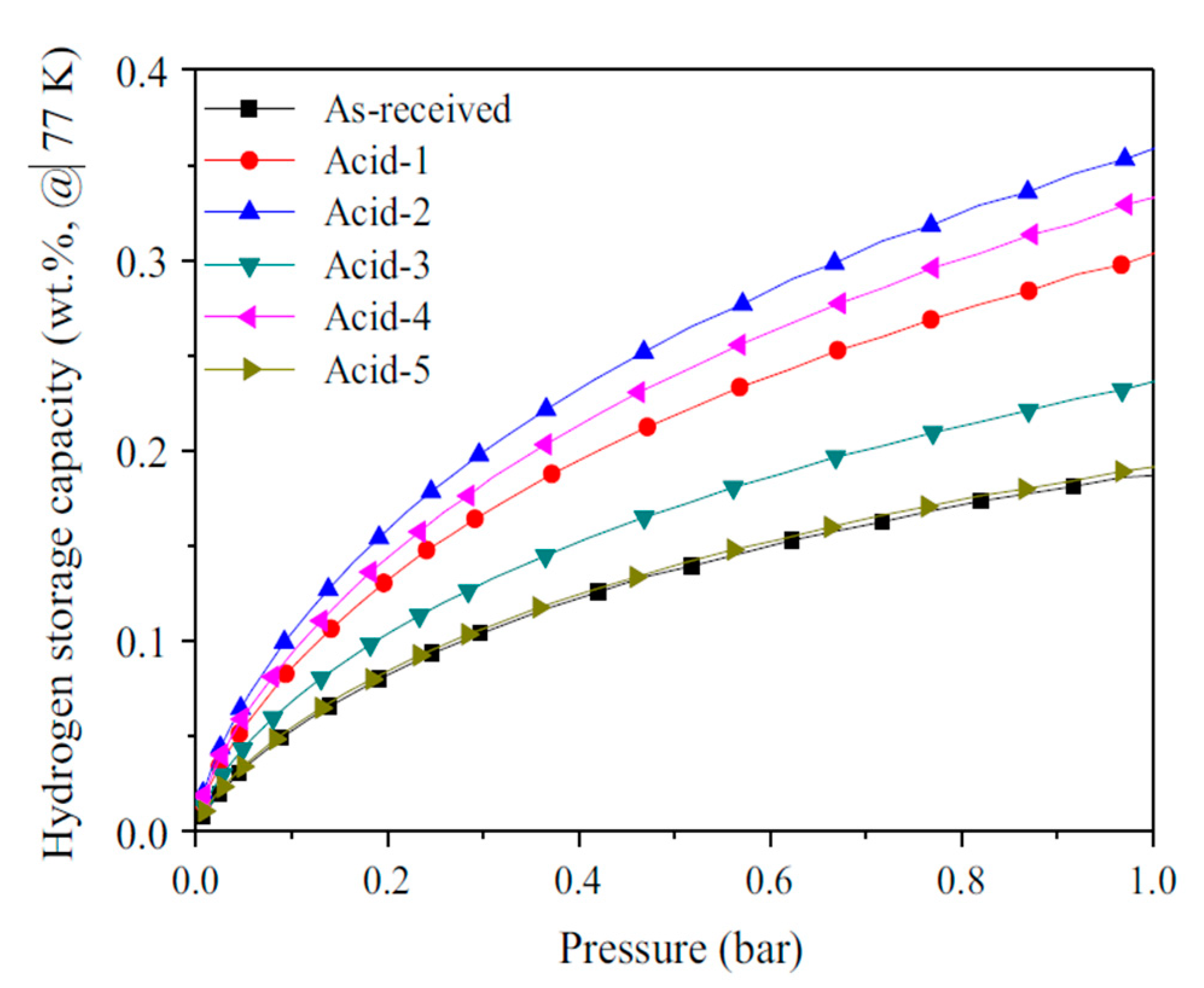
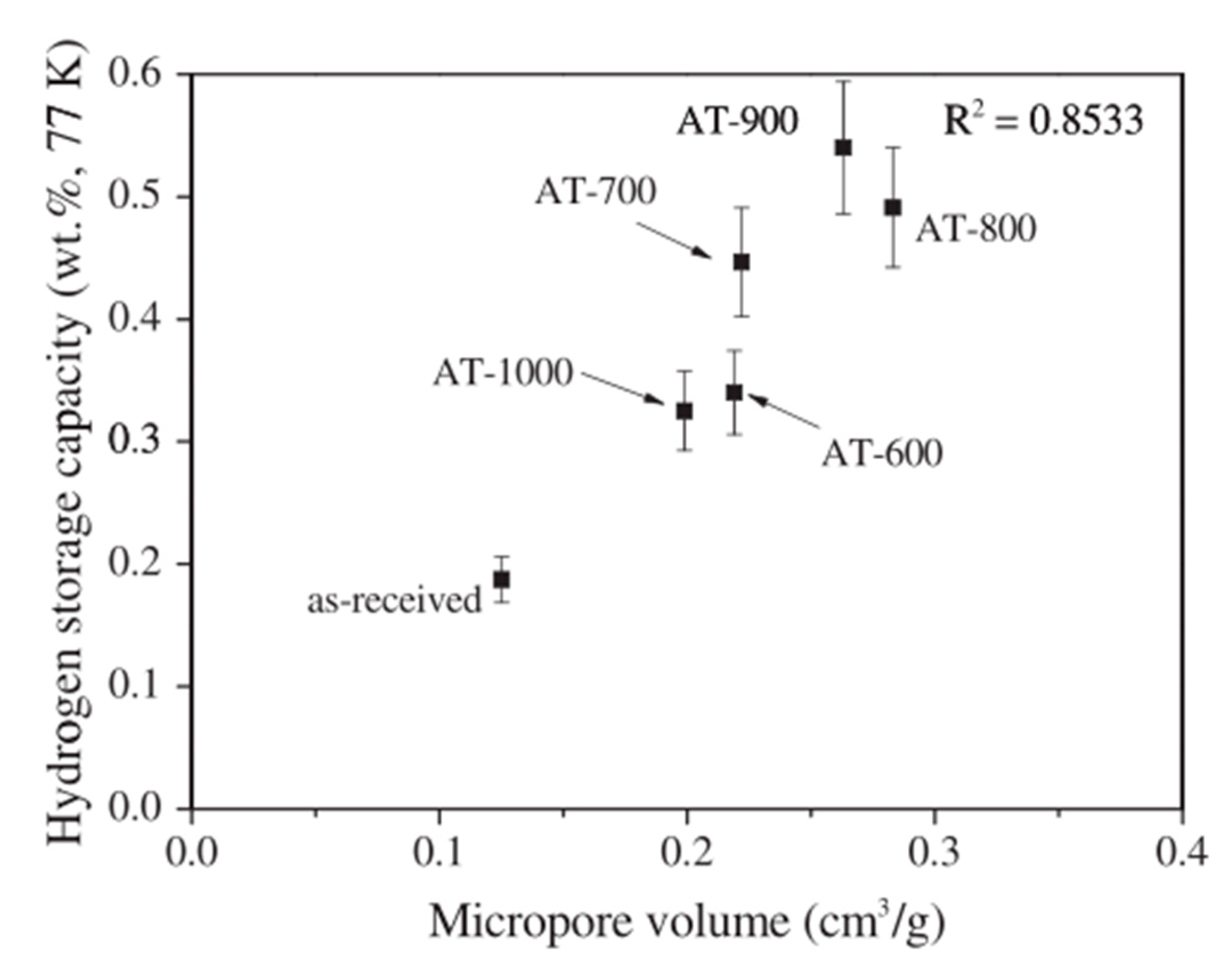
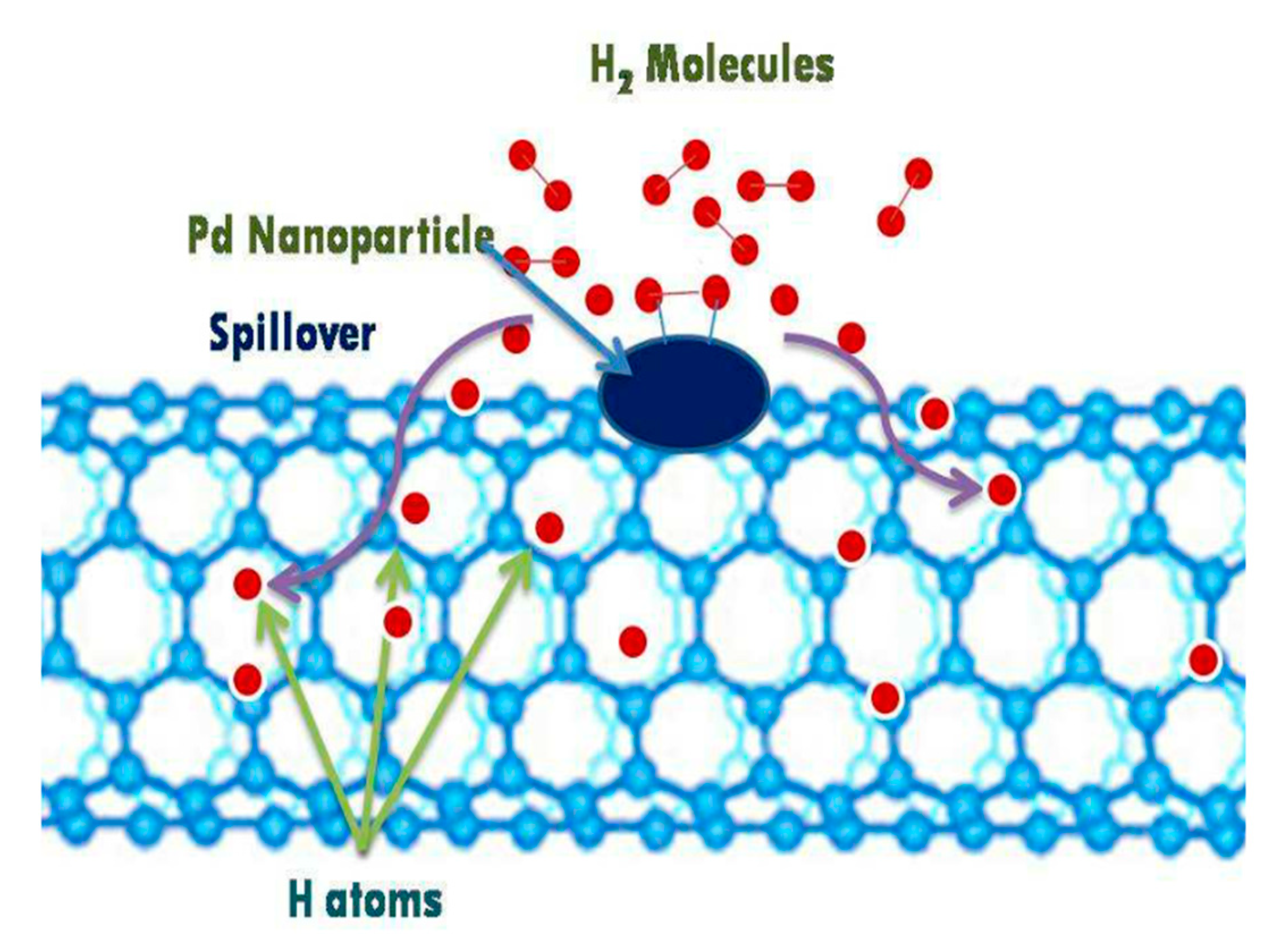
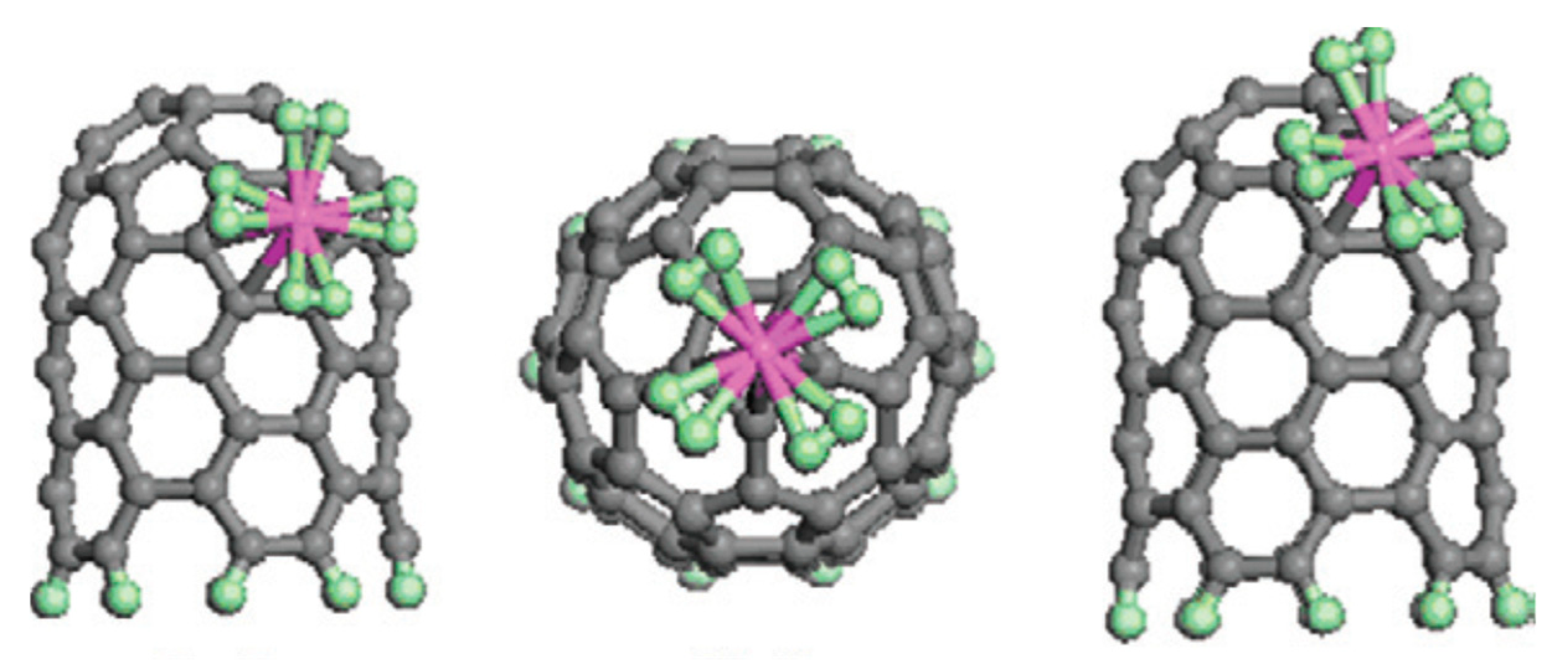
| Carbon Material | CNTs Synthetic Method | Research Method | Method/Device for Measuring Hydrogen Amount | Hydrogen Amount | Refs. |
|---|---|---|---|---|---|
| SWNTs (triangular array) | - | GCMC | - | Ads: 6 wt%/45 kg/m/77 K/1 MPa | [32] |
| KOH-activated MWCNT | chemicalvapor deposition (CVD) | experimentally | volumetric method | Ads: 1.2 wt%/12 MPa/298 K | [37] |
| CNT | CVD | experimentally | Hidden IMI PSI gas storage device | Ads: 1.14 wt%/80 bar/77 K | [63] |
| Triplet form of (5,0) CNT | - | DFT | - | Ads: 10.4 wt% (physisorption 4.4 wt% + chemisorption 6 wt%) | [64] |
| MWCNT milled without MgO for 2 h | the catalytic decomposition of acetylene | experimentally | volumetric method | Ads: 0.22 wt%/8–9 MPa/298 K | [78] |
| MWCNT milled without MgO for 10 h | the catalytic decomposition of acetylene | experimentally | volumetric method | Ads: 0.65 wt%/8–9 MPa/ 298 K | [78] |
| MWCNT milled with MgO for 1 h (The weight ratio of MWNT and MgO was 1:5) | the catalytic decomposition of acetylene | experimentally | volumetric method | Ads: 0.69 wt%/8–9 MPa/298 K | [78] |
| SWCNT (square array) | - | MD | - | Ads: 1.8 wt%/14 MPa/298 K | [84] |
| SWCNT (triangular array) | - | MD | - | Ads: 1.6 wt%/14 MPa/298 K | [84] |
| SWCNT | - | MD | - | Ads: 1.4 wt%/14 MPa/298 K | [84] |
| SWCNT | arc discharge | experimentally | volumetric method | Ads: 1.73 wt%/10 MPa/77 K Des: 1.23 wt%/1 atm/77 K Ads: 0.95 wt%/10 MPa/203 K Des: 0.62 wt%/1 atm/203 K Ads: 0.67 wt%/10 MPa/303 K Des: 0.42 wt%/1 atm/303 K | [97] |
| CNT | thermal chemical vapor deposition (TCVD) | experimentally | Elastic Recoil Detection Analysis | Ads: 0.175 wt% /5 bar/30 °C | [99] |
| KOH and heat-activated CNT (Mass radio of KOH/carbon = 2, activation temperature 800 °C) | - | experimentally | gravimetric method | Ads: 5.8 wt%/20 bar/77 K | [107] |
| KOH and heat-activated CNT (Mass radio of KOH/carbon = 4, activation temperature 800 °C) | - | experimentally | gravimetric method | Ads: 7.3 wt%/20 bar/77 K | [107] |
| MWCNT (150 kGy γ-irradiation in air) | - | experimentally | - | Ads:1.2 wt%/1 atm/100 °C | [108] |
| microwave-treated MWCNT | CVD | experimentally | - | Ads: 0.35 wt%/298 K | [109] |
| Microwave and heat treated MWCNTs | CVD | experimentally | - | Ads: 0.4 wt%/298 K | [109] |
| MWCNT ball milled for 6 h at −180 °C with the milling speed of 300 rpm | catalytic chemical vapor deposition (CCVD) | experimentally | volumetric method | Ads: 1.815 mg/g /100 kPa/77 K | [111] |
| MWCNT ball milled for 6 h at –180 °C with the milling speed of 700 rpm | CCVD | experimentally | volumetric method | Ads: 2.215 mg/g /100 kPa/77 K | [111] |
| Ball milled CNT | arc discharge | experimentally | volumetric method | Ads: 0.9 wt%/2.47 MPa/290 K/4000 s | [112] |
| MWCNT | TCVD | experimentally | volumetric method | Ads: 0.35 wt%/1 atm/298 K | [115] |
| Bromine activated MWCNT | TCVD | experimentally | volumetric method | Ads: 1.15 wt.%/1 atm/298 K | [115] |
| H2SO4 activated MWCNT | TCVD | experimentally | volumetric method | Ads: 0.41 wt%/1 atm/298 K | [115] |
| HCl activated MWCNT | TCVD | experimentally | volumetric method | Ads: 0.62 wt%/1 atm/298 K | [115] |
| HNO3 activated MWCNT | TCVD | experimentally | volumetric method | Ads: 0.85 wt%/1 atm/298 K | [115] |
| MWCNT | - | experimentally | volumetric method | Ads: 0.42 wt%/10 MPa/30 °C | [116] |
| MWCNT activated by Fluorine at 350 °C | - | experimentally | volumetric method | Ads: 1.69 wt%/10 MPa/30 °C | [116] |
| MWCNT activated by KOH at 900 °C | CCVD | experimentally | volumetric method | Ads: 1.24 wt%/34 bar/298 K | [118] |
| As purified MWCNT | CCVD | experimentally | volumetric method | Ads: 0.67 wt%/34 bar/298 K | [118] |
| MWCNT activated by H2SO4:HNO3 = 3:1 | CCVD | experimentally | volumetric method | Ads: 0.40 wt%/34 bar/298 K | [118] |
| SWCNT with 80% purity | CVD | experimentally | - | Ads: 0.4 wt% | [124] |
| SWCNT with 90% purity | CVD | experimentally | - | Ads: 0.5 wt% | [124] |
| SWCNT | pyrolysis method | experimentally | Ads: 8 wt%/2 MPa/290 K | [126] |
| Carbon Material | CNTs Synthetic Method | Doping Method | Research Method | Method/Device for Measuring Hydrogen Amount | Hydrogen Amount | Refs. |
|---|---|---|---|---|---|---|
| 100 mg MWCNT-1 mol Pd | TCVD | solution method | experimentally | volumetric method | Abs: 7 wt%/1 atm/298 K | [47] |
| 100 mg MWCNT-1 mol Ni | TCVD | solution method | experimentally | volumetric method | Abs: 0.4 wt%/1 atm/298 K | [47] |
| 100 mg MWCNT-1 mol Fe | TCVD | solution method | experimentally | volumetric method | Abs: 0.75 wt%/1 atm/298 K | [47] |
| 100 mg MWCNT-1 mol Co | TCVD. | solution method | experimentally | volumetric method | Abs: 1.5 wt%/1 atm/298 K | [47] |
| 100 mg MWCNT-1 mol Ca | TCVD | solution method | experimentally | volumetric method | Abs: 1.05 wt%/1 atm/298 K | [47] |
| MWCNT | TCVD | solution method | experimentally | volumetric method | Abs: 0.3 wt%/1 atm/298 K | [47] |
| (8,0) SWCNT-8(Al + 4) | - | - | DFT | - | Abs: 6.15 wt% Ead = 0.214 eV/H2 | [102] |
| MWCNT-3.72 wt% Pt | CVD | chemical reduction | experimentally | volumetric method | Abs: 18 cm3/g /100 bar/298 K | [122] |
| Nitric-activated MWCNT-1 wt% Pd | CVD | reverse micro-emulsion method | experimentally | volumetric method | Abs: 0.91 wt%/50 bar/123 K; Abs: 0.45 wt%/50 bar/223 K; Abs: 0.24 wt% /50 bar/303 K | [130] |
| Nitric-activated MWCNT-5 wt% Pd | CVD | reverse micro-emulsion method | experimentally | volumetric method | Abs: 1.16 wt%/50 bar/123 K; Abs: 0.81 wt%/50 bar/223 K; Abs: 0.49 wt% /50 bar/303 K | [130] |
| Nitric-treated MWCNT-10 wt% Pd | CVD | reverse micro-emulsion method | experimentally | volumetric method | Abs: 1.25 wt%/50 bar/123 K; Abs:1.05 wt%/50 bar/223 K; Abs: 0.64 wt% /50 bar/303 K | [130] |
| Sc-doped capped-SWCNT C30(Sc)6(H2)24 | - | - | DFT | - | Abs: 7.08 wt% Ead = 0.93 eV/H2 | [134] |
| Chemical-activated MWCNT | catalyzed vapor decomposition | electroless deposition | experimentally | gravimetric method | Abs: 0.35 wt%/6.89 MPa/298 K | [139] |
| Chemical-activated MWCNT-9.2 wt% Ni | catalyzed vapor decomposition | electroless deposition | experimentally | gravimetric method | Abs: 1.02 wt%/6.89 MPa/298 K | [139] |
| MWCNT + 1.2 wt% Li | - | experimentally | volumetric method | Abs: 3.9 wt%/106.66 kPa/77 K | [144] | |
| Capped CNT-6(Y + 6) | - | - | DFT | - | Abs: 7.51wt% Ead = 0.48eV/H2 | [145] |
| SWCNT-4(Y + 6) | - | - | DFT | - | Abs: 6.1 wt%/300 K Ead = 0.31eV/H2 | [146] |
| Li-doped CNT with the configuration of eight Li dispersed at the hollow sites above the hexagonal carbon rings | - | - | DFT | - | Abs: 13.45 wt% Ead = 0.17 eV/H2 | [150] |
| Nitric-activated MWCNT-11.54 wt% Pd | - | Chemical reduction | experimentally | volumetric method | Abs: 1.1 wt% /1.5 bar/298 K | [151] |
| Nitric-activated MWCNT-57.7 wt% Pd | - | laser ablation | experimentally | volumetric method | Abs: 6 wt%/1.5 bar/298 K | [151] |
| Nitric-activated MWCNT + 5 wt% Pd | CVD | polyol methods | experimentally | IMI analyzer | Abs: 6 wt%/50 atm/123 K | [152] |
| Nitric-activated MWCNT + 5 wt% Pd | CVD | wet impregnation | experimentally | IMI analyzer | Abs: 0.7 wt% /50 atm/123 K | [152] |
| DWCNT-2 wt% Pd | - | Chemical reduction at 300 under H2 atmosphere | experimentally | Sieverts method | Abs: 1.85 wt% /1 atm/298 K | [153] |
| DWCNT-2 wt% Pd | - | Chemical reduction at 400 under H2 atmosphere | experimentally | Sieverts method | Abs: 2 wt% /1 atm/298 K | [153] |
| DWCNT-2 wt% Pd | - | Chemical reduction at 500 under H2 atmosphere | experimentally | Sieverts method | Abs: 1.93 wt% /1 atm/298 K | [153] |
| DWCNT | - | Chemical reduction | experimentally | AMC Gas Reactor Controller | Abs: 1.7 wt%1 atm/298 K | [155] |
| DWCNT-1 wt% Pd | - | Chemical reduction | experimentally | AMC Gas Reactor Controller | Abs: 1.85 wt%1 atm/298 K | [155] |
| DWCNT-2 wt% Pd | - | Chemical reduction | experimentally | AMC Gas Reactor Controller | Abs: 3 wt%1 atm/298 K | [155] |
| DWCNT-3 wt% Pd | - | Chemical reduction | experimentally | AMC Gas Reactor Controller | Abs: 2 wt%/1 atm/298 K | [155] |
| MWCNT | TCVD | - | experimentally | volumetric method | Abs: 1.4 wt% | [161] |
| MWCNT-10.4 wt% Mg | TCVD | - | experimentally | volumetric method | Abs: 1.8 wt% | [161] |
| Nitric-activated MWCNT-12.3 wt% Ni | - | Chemical reduction | experimentally | volumetric method | Abs:0.6 wt% /1.5 bar/30 °C | [163] |
| Nitric-activated MWCNT-12.3 wt% Ni | - | laser ablation | experimentally | volumetric method | Abs: 1 wt%/1.5 bar/30 °C | [163] |
| KOH-activated CNT | CVD | Chemical reduction | experimentally | volumetric method | Abs: 0.44 wt%/100 bar/298 K | [164] |
| KOH-activated CNT-1.2 wt% Ni | CVD | Chemical reduction | experimentally | volumetric method | Abs: 0.65 wt%/100 bar/298 K | [164] |
| KOH-activated CNTs-2.2 wt% Ni | CVD | Chemical reduction | experimentally | volumetric method | Abs: 0.74 wt%/100 bar/298 K | [164] |
| KOH-activated CNTs + 4.1 wt% Ni | CVD | Chemical reduction | experimentally | volumetric method | Abs: 0.48 wt%/100 bar/298 K | [164] |
| Ti-doped CNTs with the configuration where two carbon atoms of the 6-member rings are substituted by a Ti atom | - | - | DFT and MD | - | Abs: 7.75 wt% (209 g H2/L)/650 atm/298 K Ead = 0.649 eV/H2 | [165] |
| Al-(7, 7) SWCNT Al7C70 | - | - | DFT | - | Abs: 28 wt% Ead = 0.131 eV/H2 | [168] |
| AlH3-(5, 5) SWCNT | - | - | DFT | - | Abs: 8.3 wt% Ead = 0.1–0.2 eV/H2 | [169] |
| Super Diamond CNT with 67.8 Ǻ distance between adjacent centers of CNT junctions | - | - | ab-initio and GCMC | - | Ads: 8.35 wt%(9.8 g/L)/100 bar/300 K | [173] |
| HNO3-activated MWCNT + 10 wt% Pd | - | reflux method | experimentally | Sievert’s apparatus | Abs: 0.125 wt% /65 bar/20 °C | [174] |
| HNO3-activated MWCNT + 10 wt% V | - | - | experimentally | Sievert’s apparatus | Abs: 0.1 wt% /65 bar/20 °C | [174] |
| Carbon Material | Carbon Source | Research Method | Method/Device for Measuring Hydrogen Amount | Hydrogen Amount | Refs. |
|---|---|---|---|---|---|
| Si MWCNT | methane | experimentally | gravimetric method | Abs: 0.3 wt%/100 bar/298 K | [66] |
| CNT-1.5 at% N | melamine | experimentally | volumetric method | Abs: 0.17 wt% 19 bar/298 K | [175] |
| CNT | polyphenylacetylene polymer | experimentally | - | Abs: 0.61 wt% | [176] |
| CNT-6.4 at% N | polypyrrole | experimentally | - | Abs: 1.2 wt% | [176] |
| BCNT | 1,4- divinylbenzene and diborane | experimentally | - | Abs: 2.03 wt% | [176] |
| CNT-8.5 at% N | polystyrene and polypyrrole | experimentally | volumetric method | Abs: 2 wt%/100 bar/298 K | [177] |
| CNT-5.4 at% N | imidazole | experimentally | IMI analyzer | Abs: 0.8 wt%/50 bar/163 K | [178] |
| SWCNT | - | GCMC | - | Abs: 1.4 wt%/100 bar/298 K | [179] |
| SWCNT-10 atom% Si | - | GCMC | - | Abs: 2.5 wt%/100 bar/298 K | [179] |
| Carbon Material | Research Method | Method/Device for Measuring Hydrogen Amount | Hydrogen Amount | Refs. |
|---|---|---|---|---|
| BCNT-1Ru- 4H2 | DFT | - | Ead = 1.151 eV/H2 | [182] |
| (4ND)10-NCNT-10(Sc + 5H2) | generalized gradient approximation (GGA), DFT and MD | - | Ads: 5.85 wt%/300 K Ead = 0.166 eV/H2 | [149] |
| SWCNT-BH3 | experimentally | CHN-elemental analysis | Ads: 4.77 wt%/50 °C Des temperature: 90–125 °C | [185] |
| (5,5) SWCNT-5(LiH + H2) | DFT | - | Ads: 1.90 wt% Ead = 0.20 eV/H2 | [187] |
| (5,5) SWCNT-5(LiH + 5H2) | DFT | - | Ads: 7.36 wt% Ead = 0.14 eV/H2 | [187] |
| (5,5) SWCNT-10(LiH + H2) | DFT | - | Ads: 3.48 wt% Ead = 0.15 eV/H2 | [187] |
| (5,5) SWCNT-10(NiH2 + H2) | DFT | - | Ads: 0.73 wt% Ead = 0.19 eV/H2 | [187] |
| (5,5) SWCNT-10(NiH2 + 5H2) | DFT | - | Ads: 2.44 wt% Ead = 0.06 eV/H2 | [187] |
| (5,5) SWCNT-5(NiH2 + H2) | DFT | - | Ads: 1.27 wt% | [187] |
| SWCNT-BH3 | experimentally | CHNS elemental analysis | Ads: 1.5 wt% | [188] |
| (5,5) SWCNT-5(NH3 + 5H2) | DFT | - | Ads: 8.18 wt% Ead = 0.091eV/H2 Desorption temperature: 115 K | [189] |
| (5,5) SWCNT-10(NH3 + 5H2) | DFT | - | Ads: 13.2 wt% Ead = 0.092eV/H2 Desorption temperature: 115 K | [189] |
| (10,10) SWCNT-8(TiO2 + 7H2) | DFT | - | Ads: 6.6 wt% | [190] |
| (10,10) SWCNT-4(TiO2 + 6H2) | DFT | - | Ads: 3.64 wt% Ead = 0.25eV/H2 Desorption temperature: 332 K | [191] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lyu, J.; Kudiiarov, V.; Lider, A. An Overview of the Recent Progress in Modifications of Carbon Nanotubes for Hydrogen Adsorption. Nanomaterials 2020, 10, 255. https://doi.org/10.3390/nano10020255
Lyu J, Kudiiarov V, Lider A. An Overview of the Recent Progress in Modifications of Carbon Nanotubes for Hydrogen Adsorption. Nanomaterials. 2020; 10(2):255. https://doi.org/10.3390/nano10020255
Chicago/Turabian StyleLyu, Jinzhe, Viktor Kudiiarov, and Andrey Lider. 2020. "An Overview of the Recent Progress in Modifications of Carbon Nanotubes for Hydrogen Adsorption" Nanomaterials 10, no. 2: 255. https://doi.org/10.3390/nano10020255
APA StyleLyu, J., Kudiiarov, V., & Lider, A. (2020). An Overview of the Recent Progress in Modifications of Carbon Nanotubes for Hydrogen Adsorption. Nanomaterials, 10(2), 255. https://doi.org/10.3390/nano10020255






