Vibration Sensing Systems Based on Poly(Vinylidene Fluoride) and Microwave-Assisted Synthesized ZnO Star-Like Particles with Controllable Structural and Physical Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of ZnO Star-Like Particles
2.3. Preparation of the PVDF/ZnO Composite Films
2.4. Poling of the Prepared Spin-Coated Films
2.5. General Characterization
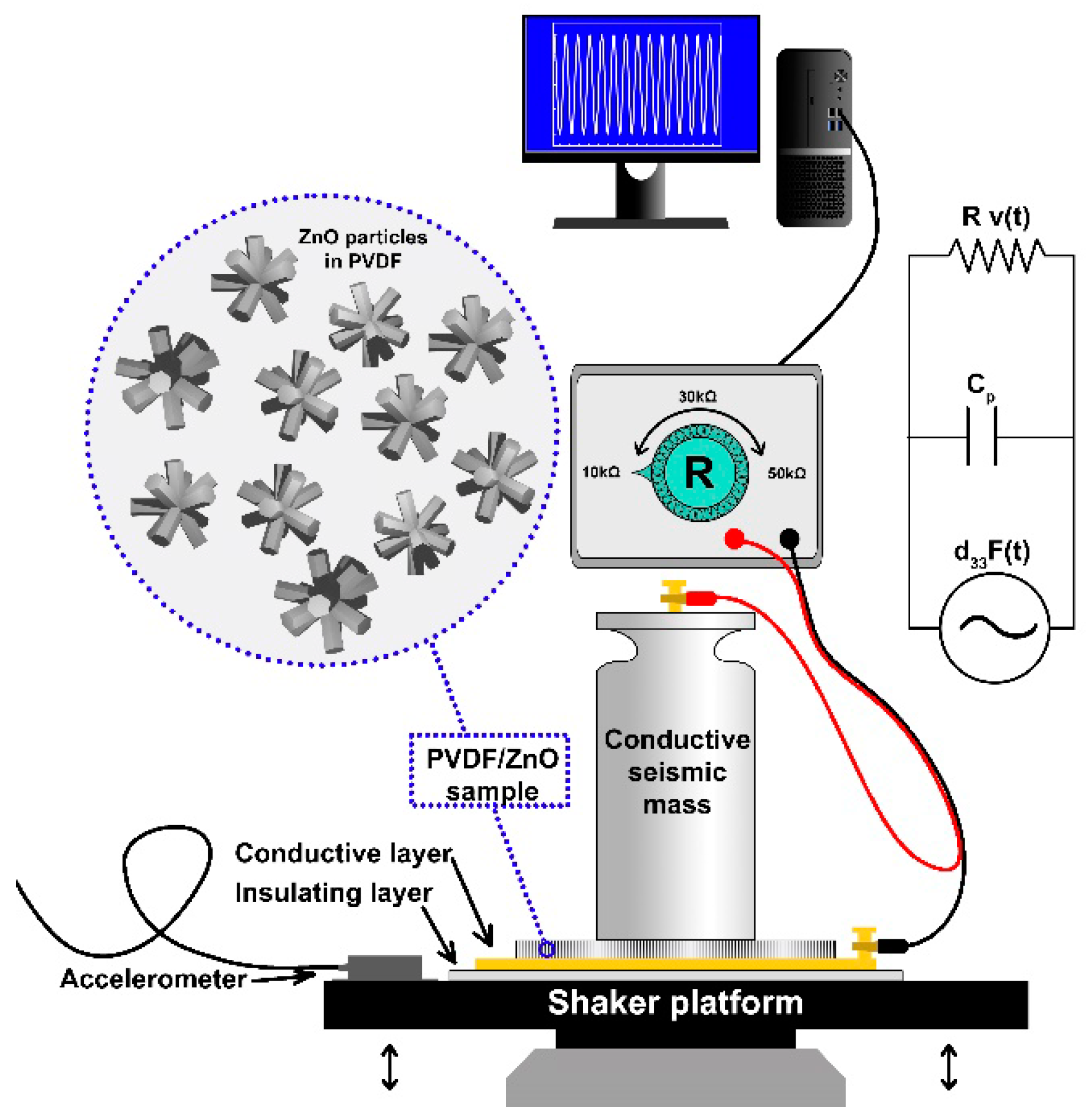
2.6. Vibration Sensing under Mechanical Excitation
3. Results and Discussion
3.1. Synthesis of ZnO Star-Like Particles
3.2. Structural Characterization of the ZnO Star-Like/PVDF Composite Films
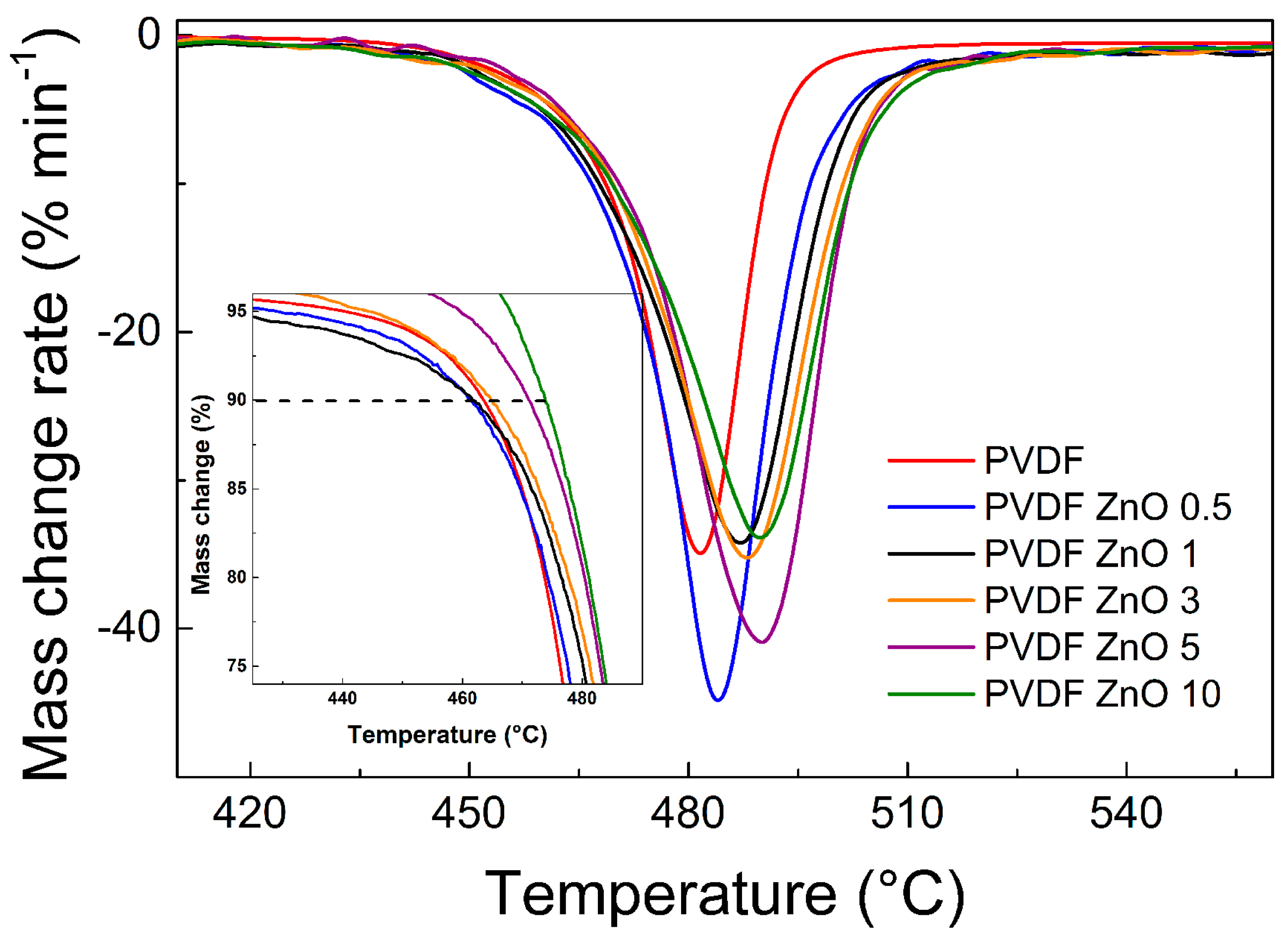
Thermal Properties of the ZnO Star-Like/PVDF Composite Films
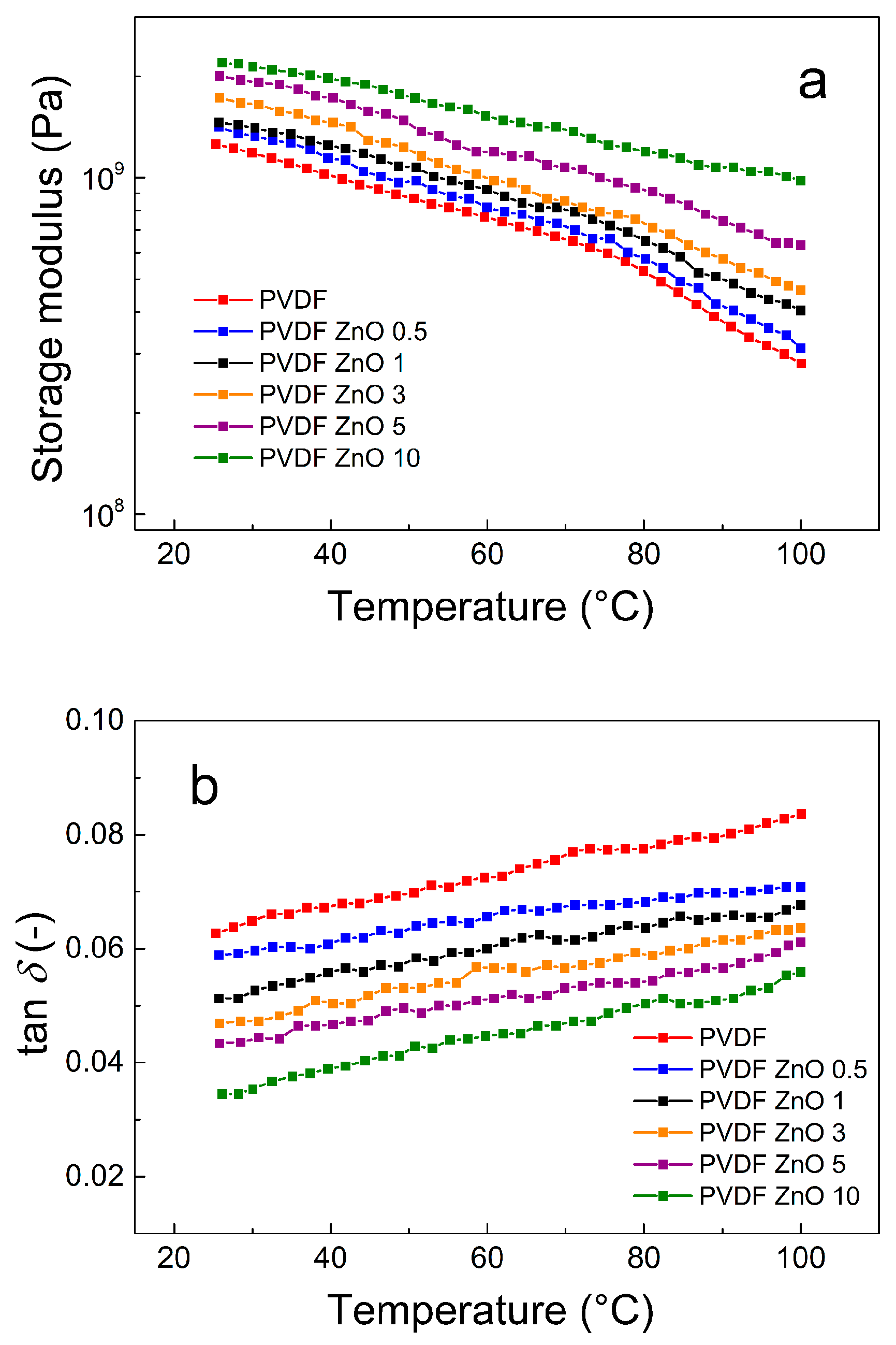
3.3. Dynamic Mechanical Properties of the ZnO Star-Like/PVDF Composite Films
3.4. Vibration Sensing Capability upon Mechanical Excitation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Feng, C.-X.; Huang, T.; Chen, H.-M.; Yang, J.-H.; Zhang, N.; Wang, Y.; Zhang, C.-L.; Zhou, Z.-W. Carbon nanotubes induced poly(vinylidene fluoride) crystallization from a miscible poly(vinylidene fluoride)/poly(methyl methacrylate) blend. Colloid Polym. Sci. 2014, 292, 3279–3290. [Google Scholar] [CrossRef]
- Huang, L.; Lu, C.; Wang, F.; Wang, L. Preparation of PVDF/graphene ferroelectric composite films by in situ reduction with hydrobromic acids and their properties. RSC Adv. 2014, 4, 45220–45229. [Google Scholar] [CrossRef]
- Chang, C.; Tran, V.H.; Wang, J.; Fuh, Y.-K.; Lin, L. Direct-Write Piezoelectric Polymeric Nanogenerator with High Energy Conversion Efficiency. Nano Lett. 2010, 10, 726–731. [Google Scholar] [CrossRef]
- Zhang, Z.; Xu, X.-L.; Yang, J.; Huang, T.; Zhang, N.; Wang, Y.; Zhou, Z. High thermal conductivity of poly(vinylidene fluoride)/carbon nanotubes nanocomposites achieved by adding polyvinylpyrrolidone. Compos. Sci. Technol. 2015, 106, 1–8. [Google Scholar] [CrossRef]
- Lee, C.; Tarbutton, J.A. Electric poling-assisted additive manufacturing process for PVDF polymer-based piezoelectric device applications. Smart Mater. Struct. 2014, 23, 7. [Google Scholar] [CrossRef]
- Saravanakumar, B.; SoYoon, S.; Kim, S.-J. Self-Powered pH Sensor Based on a Flexible Organic–Inorganic Hybrid Composite Nanogenerator. ACS Appl. Mater. Interfaces 2014, 6, 13716–13723. [Google Scholar] [CrossRef] [PubMed]
- Shao, H.; Fang, J.; Wang, H.; Lin, T. Effect of electrospinning parameters and polymer concentrations on mechanical-to-electrical energy conversion of randomly-oriented electrospun poly(vinylidene fluoride) nanofiber mats. RSC Adv. 2015, 5, 14345–14350. [Google Scholar] [CrossRef]
- Zelenika, S.; Hadas, Z.; Bader, S.; Becker, T.; Gljušćić, P.; Hlinka, J.; Janak, L.; Kamenar, E.; Ksica, F.; Kyratsi, T.; et al. Energy Harvesting Technologies for Structural Health Monitoring of Airplane Components—A Review. Sensors 2020, 20, 6685. [Google Scholar] [CrossRef]
- Kanik, M.; Aktas, O.; Sen, H.S.; Durgun, E.; Bayindir, M. Spontaneous High Piezoelectricity in Poly(vinylidene fluoride) Nanoribbons Produced by Iterative Thermal Size Reduction Technique. ACS Nano 2014, 8, 9311–9323. [Google Scholar] [CrossRef] [PubMed]
- Dalui, A.; Sarkar, P.K.; Aggarwal, L.; Ghosh, S.K.; Mandal, D.; Sheet, G.; Acharya, S. Self-oriented β-crystalline phase in the polyvinylidene fluoride ferroelectric and piezo-sensitive ultrathin Langmuir–Schaefer film. Phys. Chem. Chem. Phys. 2015, 17, 8159–8165. [Google Scholar] [CrossRef]
- Martins, P.; Lopes, A.C.; Lanceros-Mendez, S. Electroactive phases of poly(vinylidene fluoride): Determination, processing and applications. Prog. Polym. Sci. 2014, 39, 683–706. [Google Scholar] [CrossRef]
- Li, L.; Zhang, M.; Rong, M.; Ruan, W. Studies on the transformation process of PVDF from α to β phase by stretching. RSC Adv. 2014, 4, 3938–3943. [Google Scholar] [CrossRef]
- Liu, G.; Schneider, K.; Zheng, L.; Zhang, X.; Li, C.; Stamm, M.; Wang, D. Stretching induced phase separation in poly(vinylidene fluoride)/poly(butylene succinate) blends studied by in-situ X-ray scattering. Polymer 2014, 55, 2588–2596. [Google Scholar] [CrossRef]
- Sharma, M.; Madras, G.; Bose, S. Process induced electroactive β-polymorph in PVDF: Effect on dielectric and ferroelectric properties. Phys. Chem. Chem. Phys. 2014, 16, 14792–14799. [Google Scholar] [CrossRef]
- Kim, G.H.; Hong, S.M.; Seo, Y. Piezoelectric properties of poly(vinylidene fluoride) and carbon nanotube blends: β-phase development. Phys. Chem. Chem. Phys. 2009, 11, 10506–10512. [Google Scholar] [CrossRef]
- Lei, T.; Cai, X.; Wang, X.; Yu, L.; Hu, X.; Zheng, G.; Lv, W.; Wang, L.; Wu, D.; Sun, D.; et al. Spectroscopic evidence for a high fraction of ferroelectric phase induced in electrospun polyvinylidene fluoride fibers. RSC Adv. 2013, 3, 24952–24958. [Google Scholar] [CrossRef]
- Fang, J.; Niu, H.; Wang, H.; Wang, X.; Lin, T. Enhanced mechanical energy harvesting using needleless electrospun poly(vinylidene fluoride) nanofibre webs. Energy Environ. Sci. 2013, 6, 2196–2202. [Google Scholar] [CrossRef]
- Fang, J.; Wang, X.; Lin, T. Electrical power generator from randomly oriented electrospun poly(vinylidene fluoride) nanofibre membranes. J. Mater. Chem. 2011, 21, 11088–11091. [Google Scholar] [CrossRef]
- Lund, A.; Gustafsson, C.; Bertilsson, H.; Rychwalski, R.W. Enhancement of β phase crystals formation with the use of nanofillers in PVDF films and fibres. Compos. Sci. Technol. 2011, 71, 222–229. [Google Scholar] [CrossRef]
- Mofokeng, T.G.; Luyt, A.S.; Pavlović, V.P.; Pavlović, V.B.; Dudić, D.; Vlahović, B.; Djoković, V. Ferroelectric nanocomposites of polyvinylidene fluoride/polymethyl methacrylate blend and BaTiO3 particles: Fabrication of β-crystal polymorph rich matrix through mechanical activation of the filler. J. Appl. Phys. 2014, 115, 084109. [Google Scholar] [CrossRef]
- Zhang, Y.Y.; Jiang, S.L.; Yu, Y.; Zeng, Y.; Zhang, G.Z.; Zhang, Q.F.; He, J.G. Crystallization behavior and phase-transformation mechanism with the use of graphite nanosheets in poly(vinylidene fluoride) nanocomposites. J. Appl. Polym. Sci. 2012, 125, E314–E319. [Google Scholar] [CrossRef]
- Thangavel, E.; Ramasundaram, S.; Pitchaimuthu, S.; Hong, S.W.; Lee, S.Y.; Yoo, S.-S.; Kim, D.-E.; Ito, E.; Kang, Y.S. Structural and tribological characteristics of poly(vinylidene fluoride)/functionalized graphene oxide nanocomposite thin films. Compos. Sci. Technol. 2014, 90, 187–192. [Google Scholar] [CrossRef]
- Jia, N.; Xing, Q.; Xia, G.; Sun, J.; Song, R.; Huang, W. Enhanced β-crystalline phase in poly(vinylidene fluoride) films by polydopamine-coated BaTiO3 nanoparticles. Mater. Lett. 2015, 139, 212–215. [Google Scholar] [CrossRef]
- Guan, X.; Zhang, Y.; Li, H.; Ou, J. PZT/PVDF composites doped with carbon nanotubes. Sens. Actuators A Phys. 2013, 194, 228–231. [Google Scholar] [CrossRef]
- Jaleh, B.; Fakhri, P.; Noroozi, M.; Muensit, N. Influence of Copper Nanoparticles Concentration on the Properties of Poly(vinylidene fluoride)/Cu Nanoparticles Nanocomposite Films. J. Inorg. Organomet. Polym. Mater. 2012, 22, 878–885. [Google Scholar] [CrossRef]
- Vasundhara, K.; Mandal, B.P.; Tyagi, A. Enhancement of dielectric permittivity and ferroelectricity of a modified cobalt nanoparticle and polyvinylidene fluoride based composite. RSC Adv. 2015, 5, 8591–8597. [Google Scholar] [CrossRef]
- Miranda, D.; Sencadas, V.; Sánchez-Iglesias, A.; Pastorizasantos, I.; Liz-Marzán, L.M.; Ribelles, J.L.G.; Lanceros-Mendez, S. Influence of Silver Nanoparticles Concentration on the α- to β-Phase Transformation and the Physical Properties of Silver Nanoparticles Doped Poly(vinylidene fluoride) Nanocomposites. J. Nanosci. Nanotechnol. 2009, 9, 2910–2916. [Google Scholar] [CrossRef]
- Indolia, A.P.; Gaur, M.S. Investigation of structural and thermal characteristics of PVDF/ZnO nanocomposites. J. Therm. Anal. Calorim. 2012, 113, 821–830. [Google Scholar] [CrossRef]
- Ourry, L.; Marchesini, S.; Bibani, M.; Mercone, S.; Ammar, S.; Mammeri, F. Influence of nanoparticle size and concentration on the electroactive phase content of PVDF in PVDF-CoFe2O4-based hybrid films. Phys. Status Solidi (a) 2014, 212, 252–258. [Google Scholar] [CrossRef]
- An, N.; Liu, H.; Ding, Y.; Zhang, M.; Tang, Y. Preparation and electroactive properties of a PVDF/nano-TiO2 composite film. Appl. Surf. Sci. 2011, 257, 3831–3835. [Google Scholar] [CrossRef]
- Jaleh, B.; Jabbari, A. Evaluation of reduced graphene oxide/ZnO effect on properties of PVDF nanocomposite films. Appl. Surf. Sci. 2014, 320, 339–347. [Google Scholar] [CrossRef]
- Loh, K.J.; Chang, D. Zinc oxide nanoparticle-polymeric thin films for dynamic strain sensing. J. Mater. Sci. 2011, 46, 228–237. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, X.; Li, G. In situ ZnO nanowire growth to promote the PVDF piezo phase and the ZnO–PVDF hybrid self-rectified nanogenerator as a touch sensor. Phys. Chem. Chem. Phys. 2014, 16, 5475–5479. [Google Scholar] [CrossRef] [PubMed]
- Fang, L.; Wu, W.; Huang, X.; He, J.; Jiang, P. Hydrangea-like zinc oxide superstructures for ferroelectric polymer composites with high thermal conductivity and high dielectric constant. Compos. Sci. Technol. 2015, 107, 67–74. [Google Scholar] [CrossRef]
- Zheng, Y.; Zheng, L.; Zhan, Y.; Lin, X.; Zheng, A.Q.; Wei, K. Ag/ZnO Heterostructure Nanocrystals: Synthesis, Characterization, and Photocatalysis. Inorg. Chem. 2007, 46, 6980–6986. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Chen, X.; Wang, X.; Zhang, Y.; Wei, W.; Sun, Y.; Antonietti, M.; Titirici, M.-M. One-Step Solvothermal Synthesis of a Carbon@TiO2 Dyade Structure Effectively Promoting Visible-Light Photocatalysis. Adv. Mater. 2010, 22, 3317–3321. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Ge, M.; Liu, L.; Gao, G.; Feng, Y.; Wang, Y. Directed Synthesis of Mesoporous TiO2 Microspheres: Catalysts and Their Photocatalysis for Bisphenol A Degradation. Environ. Sci. Technol. 2010, 44, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Bazant, P.; Kuritka, I.; Munster, L.; Machovsky, M.; Kozakova, Z.; Saha, P. Hybrid nanostructured Ag/ZnO decorated powder cellulose fillers for medical plastics with enhanced surface antibacterial activity. J. Mater. Sci. Mater. Med. 2014, 25, 2501–2512. [Google Scholar] [CrossRef]
- Sedlačík, M.; Mrlik, M.; Kozáková, Z.; Pavlínek, V.; Kuritka, I. Synthesis and electrorheology of rod-like titanium oxide particles prepared via microwave-assisted molten-salt method. Colloid Polym. Sci. 2012, 291, 1105–1111. [Google Scholar] [CrossRef]
- Machovsky, M.; Kuritka, I.; Kozakova, Z. Microwave assisted synthesis of nanostructured Fe3O4/ZnO microparticles. Mater. Lett. 2012, 86, 136–138. [Google Scholar] [CrossRef]
- Plachý, T.; Mrlik, M.; Kozáková, Z.; Suly, P.; Sedlačík, M.; Pavlínek, V.; Kuritka, I. The Electrorheological Behavior of Suspensions Based on Molten-Salt Synthesized Lithium Titanate Nanoparticles and Their Core–Shell Titanate/Urea Analogues. ACS Appl. Mater. Interfaces 2015, 7, 3725–3731. [Google Scholar] [CrossRef]
- Pan, X.; Wang, Z.; Cao, Z.; Zhang, S.; He, Y.; Zhang, Y.; Chen, K.; Hu, Y.; Gu, H. A self-powered vibration sensor based on electrospun poly(vinylidene fluoride) nanofibres with enhanced piezoelectric response. Smart Mater. Struct. 2016, 25, 105010. [Google Scholar] [CrossRef]
- Liu, Z.; Pan, C.; Su, C.-Y.; Lin, L.; Chen, Y.; Tsai, J. A flexible sensing device based on a PVDF/MWCNT composite nanofiber array with an interdigital electrode. Sens. Actuators A Phys. 2014, 211, 78–88. [Google Scholar] [CrossRef]
- Ram, F.; Gudadhe, A.; Vijayakanth, T.; Aherrao, S.; Borkar, V.; Boomishankar, R.; Shanmuganathan, K. Nanocellulose Reinforced Flexible Composite Nanogenerators with Enhanced Vibrational Energy Harvesting and Sensing Properties. ACS Appl. Polym. Mater. 2020, 2, 2550–2562. [Google Scholar] [CrossRef]
- Zhao, S.; Erturk, A. Deterministic and band-limited stochastic energy harvesting from uniaxial excitation of a multilayer piezoelectric stack. Sens. Actuators A Phys. 2014, 214, 58–65. [Google Scholar] [CrossRef]
- Shehata, N.; Kandas, I.; Hassounah, I.; Sobolciak, P.; Krupa, I.; Mrlik, M.; Popelka, A.; Steadman, J.; Lewis, R.V. Piezoresponse, Mechanical, and Electrical Characteristics of Synthetic Spider Silk Nanofibers. Nanomaterials 2018, 8, 585. [Google Scholar] [CrossRef] [PubMed]
- Byzynski, G.; Melo, C.; Volanti, D.P.; Ferrer, M.M.; Gouveia, A.F.; Ribeiro, C.; Andrés, J.; Longo, E. The interplay between morphology and photocatalytic activity in ZnO and N-doped ZnO crystals. Mater. Des. 2017, 120, 363–375. [Google Scholar] [CrossRef]
- Machovsky, M.; Mrlik, M.; Kuritka, I.; Pavlinek, V.; Babayan, V. Novel synthesis of core–shell urchin-like ZnO coated carbonyl iron microparticles and their magnetorheological activity. RSC Adv. 2014, 4, 996–1003. [Google Scholar] [CrossRef]
- Issa, A.A.; Al-Maadeed, M.; Luyt, A.S.; Mrlik, M.; Hassan, M.K. Investigation of the physico-mechanical properties of electrospun PVDF/cellulose (nano)fibers. J. Appl. Polym. Sci. 2016, 133, 12. [Google Scholar] [CrossRef]
- Částková, K.; Kastyl, J.; Sobola, D.; Petruš, J.; Stastna, E.; Riha, D.; Tofel, P. Structure–Properties Relationship of Electrospun PVDF Fibers. Nanomaterials 2020, 10, 1221. [Google Scholar] [CrossRef]
- Florczak, S.; Lorson, T.; Zheng, T.; Mrlik, M.; Hutmacher, D.W.; Higgins, M.J.; Luxenhofer, R.; Dalton, P.D. Melt electrowriting of electroactive poly(vinylidene difluoride) fibers. Polym. Int. 2019, 68, 735–745. [Google Scholar] [CrossRef]
- Salimi, A.; Yousefi, A. Analysis Method. Polym. Test. 2003, 22, 699–704. [Google Scholar] [CrossRef]
- Liu, J.; Lu, X.; Wu, C.; Zhao, C. Effect of preparation conditions on the morphology, polymorphism and mechanical properties of polyvinylidene fluoride membranes formed via thermally induced phase separation. J. Polym. Res. 2013, 20, 10. [Google Scholar] [CrossRef]
- Mrlik, M.; Leadenham, S.; Almaadeed, M.A.; Erturk, A. Figure of merit comparison of PP-based electret and PVDF-based piezoelectric polymer energy harvesters. In Proceedings of the SPIE 9799: Active and Passive Smart Structures and Integrated Systems, Las Vegas, NE, USA, 21–24 March 2016; p. 979923. [Google Scholar] [CrossRef]






| Sample Code | Neat PVDF | PVDF ZnO 0.5 | PVDF ZnO 1 | PVDF ZnO 3 | PVDF ZnO 5 | PVDF ZnO 10 |
|---|---|---|---|---|---|---|
| Ratio | 1.9 | 8.3 | 8.7 | 14.4 | 18.9 | 22.5 |
| F (β) | 44.1 | 46.2 | 47.3 | 53.8 | 60.9 | 66.4 |
| Sample Code | Tm (°C) | ΔHm (J g−1) | Tc (°C) | ΔHc (J g−1) | Xc (%) |
|---|---|---|---|---|---|
| neat PVDF | 171.73 | 41.96 | 134.09 | −51.72 | 40.2 |
| PVDF ZnO 0.5 | 170.44 | 43.16 | 134.47 | −53.95 | 41.3 |
| PVDF ZnO 1 | 169.69 | 44.48 | 134.65 | −54.99 | 42.6 |
| PVDF ZnO 3 | 168.77 | 47.16 | 135.02 | −56.84 | 45.1 |
| PVDF ZnO 5 | 168.59 | 50.45 | 135.58 | −59.42 | 48.3 |
| PVDF ZnO 10 | 167.84 | 52.56 | 135.76 | −61.36 | 50.3 |
| Sample Code | T10% (°C) | Tmax (°C) |
|---|---|---|
| neat PVDF | 463.4 | 481.5 |
| PVDF ZnO 0.5 | 461.3 | 483.9 |
| PVDF ZnO 1 | 461.7 | 486.9 |
| PVDF ZnO 3 | 465.3 | 488.1 |
| PVDF ZnO 5 | 471.1 | 489.1 |
| PVDF ZnO 10 | 473.8 | 489.9 |
| Sample Name | 10 kΩ (pC/N) | 30 kΩ (pC/N) | 50 kΩ (pC/N) | Avg. (pC/N) |
|---|---|---|---|---|
| pure PVDF | 16.4 | 16.6 | 16.01 | 16.3 ± 0.3 |
| PVDF ZnO 0.5 | 18.7 | 19.00 | 19.1 | 18.9 ± 0.2 |
| PVDF ZnO 1 | 18.8 | 19.4 | 19.3 | 19.1 ± 0.3 |
| PVDF ZnO 3 | 23.2 | 23.9 | 22.5 | 23.2 ± 0.7 |
| PVDF ZnO 5 | 26.2 | 25.7 | 25.5 | 25.8 ± 0.4 |
| PVDF ZnO 10 | 28.9 | 29.3 | 29.4 | 29.2 ± 0.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chamakh, M.M.; Mrlík, M.; Leadenham, S.; Bažant, P.; Osička, J.; AlMaadeed, M.A.A.; Erturk, A.; Kuřitka, I. Vibration Sensing Systems Based on Poly(Vinylidene Fluoride) and Microwave-Assisted Synthesized ZnO Star-Like Particles with Controllable Structural and Physical Properties. Nanomaterials 2020, 10, 2345. https://doi.org/10.3390/nano10122345
Chamakh MM, Mrlík M, Leadenham S, Bažant P, Osička J, AlMaadeed MAA, Erturk A, Kuřitka I. Vibration Sensing Systems Based on Poly(Vinylidene Fluoride) and Microwave-Assisted Synthesized ZnO Star-Like Particles with Controllable Structural and Physical Properties. Nanomaterials. 2020; 10(12):2345. https://doi.org/10.3390/nano10122345
Chicago/Turabian StyleChamakh, Mariem M., Miroslav Mrlík, Stephen Leadenham, Pavel Bažant, Josef Osička, Mariam Al Ali AlMaadeed, Alper Erturk, and Ivo Kuřitka. 2020. "Vibration Sensing Systems Based on Poly(Vinylidene Fluoride) and Microwave-Assisted Synthesized ZnO Star-Like Particles with Controllable Structural and Physical Properties" Nanomaterials 10, no. 12: 2345. https://doi.org/10.3390/nano10122345
APA StyleChamakh, M. M., Mrlík, M., Leadenham, S., Bažant, P., Osička, J., AlMaadeed, M. A. A., Erturk, A., & Kuřitka, I. (2020). Vibration Sensing Systems Based on Poly(Vinylidene Fluoride) and Microwave-Assisted Synthesized ZnO Star-Like Particles with Controllable Structural and Physical Properties. Nanomaterials, 10(12), 2345. https://doi.org/10.3390/nano10122345






