CeO2 Nanomaterials from Diesel Engine Exhaust Induce DNA Damage and Oxidative Stress in Human and Rat Sperm In Vitro
Abstract
1. Introduction
2. Material and Methods
2.1. Physical-Chemical Characterisation
2.1.1. Ageing of the Diesel Fuel Additive
2.1.2. Aged CeO2 NM Dissolution in FertiCult® Medium
2.2. Gamete Collection
2.3. Ethical Authorization
2.4. Sperm Exposure
2.5. DNA Damage Evaluation by Comet Assay
2.6. Oxidative Stress Analysis on Human Sperm
2.7. Imaging of Human Sperm Cells after In Vitro Exposure
3. Results
3.1. No Detectable Dissolution of Aged CeO2 NMs in Abiotic Conditions
3.2. Higher DNA Damage Detected at the Lowest Concentration Exposure
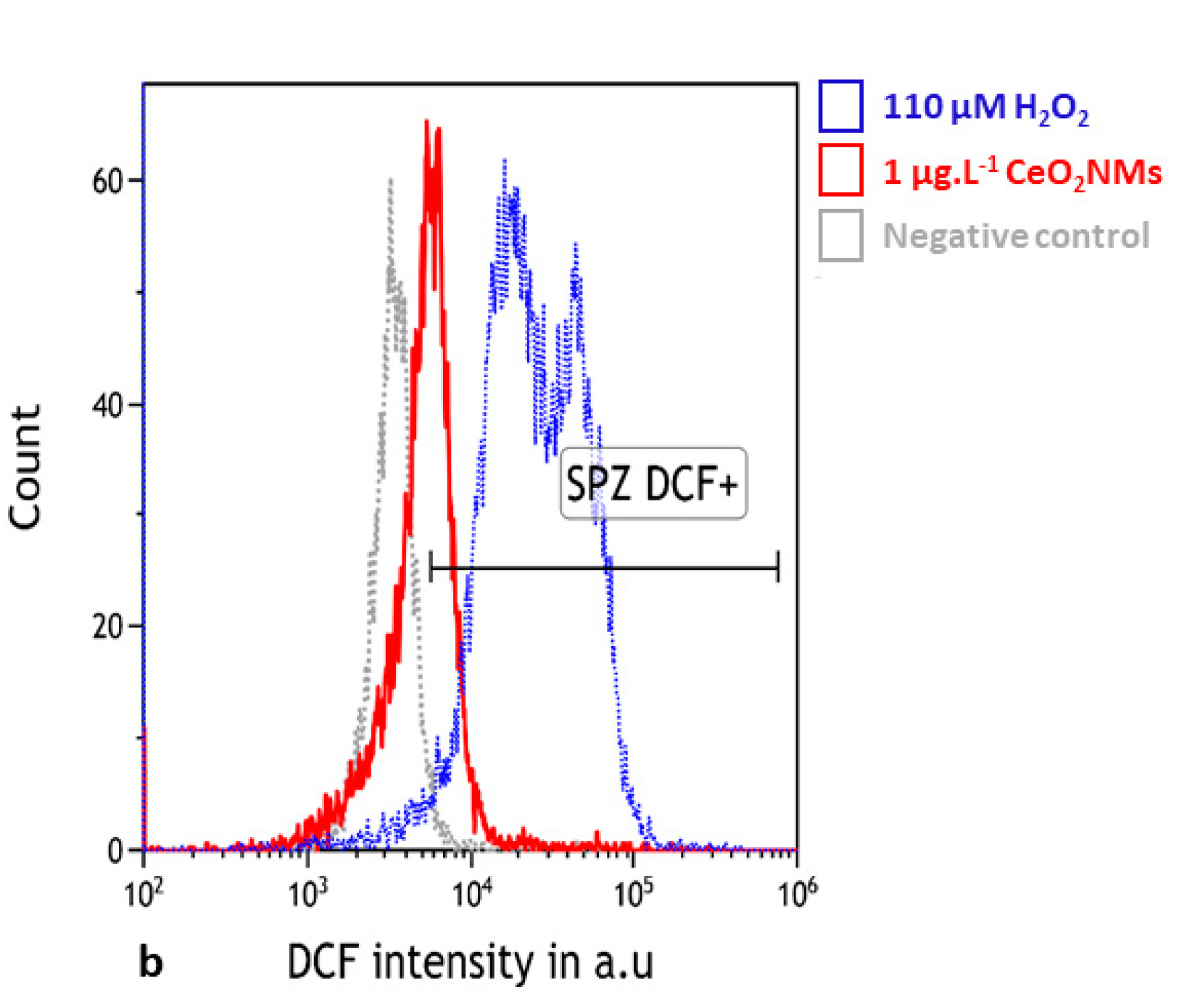
3.3. Oxidative Stress Detected in Human Sperm
3.4. Aged CeO2 NMs Detected by TEM on the Plasma Membrane of Human Sperm
4. Discussion
4.1. Exposure to Low Doses of Aged CeO2 NMs Induced Higher DNA Damage
4.2. DNA Damage in Human Sperm Was Associated with Oxidative Stress
4.3. Lifecycle Stage-Dependent (Geno)Toxicity of CeO2 NMs
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lee, J.; Jeong, J.-S.; Kim, S.Y.; Lee, S.-J.; Shin, Y.-J.; Im, W.-J.; Kim, S.-H.; Park, K.; Jeong, E.J.; Nam, S.-Y.; et al. Safety assessment of cerium oxide nanoparticles: Combined repeated-dose toxicity with reproductive/developmental toxicity screening and biodistribution in rats. Nanotoxicology 2020, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Oberdörster, G.; Oberdörster, E.; Oberdörster, J. Nanotoxicology: An Emerging Discipline Evolving from Studies of Ultrafine Particles. Environ. Health Perspect. 2005, 113, 823–839. [Google Scholar] [CrossRef] [PubMed]
- Graczyk, H.; Riediker, M. Occupational exposure to inhaled nanoparticles: Are young workers being left in the dust? J. Occup. Health 2019, 61, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Auffan, M.; Tella, M.; Liu, W.; Pariat, A.; Cabié, M.; Borschneck, D.; Angeletti, B.; Landrot, G.; Mouneyrac, C.; Giambérini, L.; et al. Structural and physical–chemical behavior of a CeO2 nanoparticle based diesel additive during combustion and environmental release. Environ. Sci. Nano 2017, 4, 1974–1980. [Google Scholar] [CrossRef]
- Masion, A.; Auffan, M.; Rose, J. Monitoring the Environmental Aging of Nanomaterials: An Opportunity for Mesocosm Testing? Materials 2019, 12, 2447. [Google Scholar] [CrossRef] [PubMed]
- Gaiser, B.K.; Fernandes, T.F.; Jepson, M.; Lead, J.R.; Tyler, C.R.; Stone, V. Assessing exposure, uptake and toxicity of silver and cerium dioxide nanoparticles from contaminated environments. Environ. Health 2009, 8, S2. [Google Scholar] [CrossRef] [PubMed]
- Park, B.; Martin, P.; Harris, C.; Guest, R.; Whittingham, A.; Jenkinson, P.; Handley, J. Initial in vitro screening approach to investigate the potential health and environmental hazards of EnviroxTM—A nanoparticulate cerium oxide diesel fuel additive. Part. Fibre Toxicol. 2007, 4, 12. [Google Scholar] [CrossRef]
- Jung, H.; Kittelson, D.B. Measurement of Electrical Charge on Diesel Particles. Aerosol Sci. Technol. 2005, 39, 1129–1135. [Google Scholar] [CrossRef]
- Zhang, J.; Nazarenko, Y.; Zhang, L.; Calderon, L.; Lee, K.-B.; Garfunkel, E.; Schwander, S.; Tetley, T.D.; Chung, K.F.; Porter, A.E.; et al. Impacts of a Nanosized Ceria Additive on Diesel Engine Emissions of Particulate and Gaseous Pollutants. Environ. Sci. Technol. 2013, 47, 13077–13085. [Google Scholar] [CrossRef]
- Park, B.; Donaldson, K.; Duffin, R.; Tran, L.; Kelly, F.; Mudway, I.; Morin, J.-P.; Guest, R.; Jenkinson, P.; Samaras, Z.; et al. Hazard and Risk Assessment of a Nanoparticulate Cerium Oxide-Based Diesel Fuel Additive—A Case Study. Inhal. Toxicol. 2008, 20, 547–566. [Google Scholar] [CrossRef]
- Johnson, A.C.; Park, B. Predicting contamination by the fuel additive cerium oxide engineered nanoparticles within the United Kingdom and the associated risks. Environ. Toxicol. Chem. 2012, 31, 2582–2587. [Google Scholar] [CrossRef] [PubMed]
- Gantt, B.; Hoque, S.; Willis, R.D.; Fahey, K.M.; Delgado-Saborit, J.M.; Harrison, R.M.; Erdakos, G.B.; Bhave, P.V.; Zhang, K.M.; Kovalcik, K.; et al. Near-Road Modeling and Measurement of Cerium-Containing Particles Generated by Nanoparticle Diesel Fuel Additive Use. Environ. Sci. Technol. 2014, 48, 10607–10613. [Google Scholar] [CrossRef] [PubMed]
- Batley, G.E.; Halliburton, B.; Kirby, J.K.; Doolette, C.L.; Navarro, D.; McLaughlin, M.J.; Veitch, C. Characterization and ecological risk assessment of nanoparticulate CeO2 as a diesel fuel catalyst: Characterization and risk of nano-CeO2 as a diesel catalyst. Environ. Toxicol. Chem. 2013, 32, 1896–1905. [Google Scholar] [CrossRef] [PubMed]
- Dale, J.G.; Cox, S.S.; Vance, M.E.; Marr, L.C.; Hochella, M.F. Transformation of Cerium Oxide Nanoparticles from a Diesel Fuel Additive during Combustion in a Diesel Engine. Environ. Sci. Technol. 2017, 51, 1973–1980. [Google Scholar] [CrossRef] [PubMed]
- Cassee, F.R.; van Balen, E.C.; Singh, C.; Green, D.; Muijser, H.; Weinstein, J.; Dreher, K. Exposure, Health and Ecological Effects Review of Engineered Nanoscale Cerium and Cerium Oxide Associated with its Use as a Fuel Additive. Crit. Rev. Toxicol. 2011, 41, 213–229. [Google Scholar] [CrossRef] [PubMed]
- OECD’s Guidelines OECD’s Meeting on Safety Testing of Manufactured Nanomaterials and Test Guidelines . March 2011. Available online: http://www.oecd.org/science/nanosafety/48291037.pdf (accessed on 15 May 2020).
- Haase, A.; Luch, A. Genotoxicity of nanomaterials in vitro: Treasure or trash? Arch. Toxicol. 2016, 90, 2827–2830. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dejmek, J.; Jelínek, R.; Solansky’, I.; Benes, I.; Srám, R.J. Fecundability and parental exposure to ambient sulfur dioxide. Environ. Health Perspect. 2000, 108, 647–654. [Google Scholar] [CrossRef]
- Nieuwenhuijsen, M.J.; Basagaña, X.; Dadvand, P.; Martinez, D.; Cirach, M.; Beelen, R.; Jacquemin, B. Air pollution and human fertility rates. Environ. Int. 2014, 70, 9–14. [Google Scholar] [CrossRef]
- Slama, R.; Bottagisi, S.; Solansky, I.; Lepeule, J.; Giorgis-Allemand, L.; Sram, R. Short-Term Impact of Atmospheric Pollution on Fecundability. Epidemiology 2013, 24, 871–879. [Google Scholar] [CrossRef]
- Legro, R.S.; Sauer, M.V.; Mottla, G.L.; Richter, K.S.; Li, X.; Dodson, W.C.; Liao, D. Effect of air quality on assisted human reproduction. Hum. Reprod. 2010, 25, 1317–1324. [Google Scholar] [CrossRef]
- Perin, P.M.; Maluf, M.; Czeresnia, C.E.; Nicolosi Foltran Januário, D.A.; Nascimento Saldiva, P.H. Effects of exposure to high levels of particulate air pollution during the follicular phase of the conception cycle on pregnancy outcome in couples undergoing in vitro fertilization and embryo transfer. Fertil. Steril. 2010, 93, 301–303. [Google Scholar] [CrossRef] [PubMed]
- Falchi, L.; Khalil, W.A.; Hassan, M.; Marei, W.F.A. Perspectives of nanotechnology in male fertility and sperm function. Int. J. Vet. Sci. Med. 2018, 6, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Geraets, L.; Oomen, A.G.; Schroeter, J.D.; Coleman, V.A.; Cassee, F.R. Tissue Distribution of Inhaled Micro- and Nano-sized Cerium Oxide Particles in Rats: Results From a 28-Day Exposure Study. Toxicol. Sci. 2012, 127, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Qin, F.; Shen, T.; Li, J.; Qian, J.; Zhang, J.; Zhou, G.; Tong, J. SF-1 mediates reproductive toxicity induced by Cerium oxide nanoparticles in male mice. J. Nanobiotechnol. 2019, 17. [Google Scholar] [CrossRef] [PubMed]
- Préaubert, L.; Courbiere, B.; Achard, V.; Tassistro, V.; Greco, F.; Orsiere, T.; Bottero, J.-Y.; Rose, J.; Auffan, M.; Perrin, J. Cerium dioxide nanoparticles affect in vitro fertilization in mice. Nanotoxicology 2015, 10, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Préaubert, L.; Tassistro, V.; Auffan, M.; Sari-Minodier, I.; Rose, J.; Courbiere, B.; Perrin, J. Very low concentration of cerium dioxide nanoparticles induce DNA damage, but no loss of vitality, in human spermatozoa. Toxicol. In Vitro 2018, 50, 236–241. [Google Scholar] [CrossRef]
- Benbrahim-Tallaa, L.; Baan, R.A.; Grosse, Y.; Lauby-Secretan, B.; El Ghissassi, F.; Bouvard, V.; Guha, N.; Loomis, D.; Straif, K. Carcinogenicity of diesel-engine and gasoline-engine exhausts and some nitroarenes. Lancet Oncol. 2012, 13, 663–664. [Google Scholar] [CrossRef]
- OECD’s Guidelines, Report of the OECD Expert Meeting on the Physical Chemical Properties of Manufactured Nanomaterials and Test Guidelines 2014. Available online: http://www.oecd.org/env/ehs/nanosafety/publications-series-safety-manufactured-nanomaterials.htm (accessed on 15 May 2020).
- Flynn, P.F.; Durrett, R.P.; Hunter, G.L.; zur Loye, A.O.; Akinyemi, O.C.; Dec, J.E.; Westbrook, C.K. Diesel Combustion: An Integrated View Combining Laser Diagnostics, Chemical Kinetics, and Empirical Validation; SAE International: Warrendale, PA, USA, 1999. [Google Scholar]
- Einaudi, L.; Courbiere, B.; Tassistro, V.; Prevot, C.; Sari-Minodier, I.; Orsiere, T.; Perrin, J. In vivo exposure to benzo(a)pyrene induces significant DNA damage in mouse oocytes and cumulus cells. Hum. Reprod. 2014, 29, 548–554. [Google Scholar] [CrossRef]
- Perrin, J.; Tassistro, V.; Mandon, M.; Grillo, J.-M.; Botta, A.; Sari-Minodier, I. Tobacco consumption and benzo(a)pyrene-diol-epoxide-DNA adducts in spermatozoa: In smokers, swim-up procedure selects spermatozoa with decreased DNA damage. Fertil. Steril. 2011, 95, 2013–2017. [Google Scholar] [CrossRef]
- Singh, N.P.; McCoy, M.T.; Tice, R.R.; Schneider, E.L. A simple technique for quantitation of low levels of DNA damage in individual cells. Exp. Cell Res. 1988, 175, 184–191. [Google Scholar] [CrossRef]
- Baumgartner, A.; Cemeli, E.; Anderson, D. The comet assay in male reproductive toxicology. Cell Biol. Toxicol. 2009, 25, 81–98. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhong, Z.; Xu, Z.; Chen, L.; Wang, Y. 2′,7′-Dichlorodihydrofluorescein as a fluorescent probe for reactive oxygen species measurement: Forty years of application and controversy. Free Radic. Res. 2010, 44, 587–604. [Google Scholar] [CrossRef] [PubMed]
- Aitken, R.J.; Smith, T.B.; Lord, T.; Kuczera, L.; Koppers, A.J.; Naumovski, N.; Connaughton, H.; Baker, M.A.; De Iuliis, G.N. On methods for the detection of reactive oxygen species generation by human spermatozoa: Analysis of the cellular responses to catechol oestrogen, lipid aldehyde, menadione and arachidonic acid. Andrology 2013, 1, 192–205. [Google Scholar] [CrossRef] [PubMed]
- Gallo, A.; Manfra, L.; Boni, R.; Rotini, A.; Migliore, L.; Tosti, E. Cytotoxicity and genotoxicity of CuO nanoparticles in sea urchin spermatozoa through oxidative stress. Environ. Int. 2018, 118, 325–333. [Google Scholar] [CrossRef]
- WHO Laboratory Manual for the Examination and Processing of Human Semen, 5th ed.; World Health Organization: Geneva, Switzerland, 2010; ISBN 978-92-4-154778-9.
- Kuhlbusch, T.A.J. Nanomaterial exposures for worker, consumer and the general public. NanoImpact 2018, 10, 11–25. [Google Scholar] [CrossRef]
- Modrzynska, J.; Berthing, T.; Ravn-Haren, G.; Kling, K.; Mortensen, A.; Rasmussen, R.R.; Larsen, E.H.; Saber, A.T.; Vogel, U.; Loeschner, K. In vivo-induced size transformation of cerium oxide nanoparticles in both lung and liver does not affect long-term hepatic accumulation following pulmonary exposure. PLoS ONE 2018, 13, e0202477. [Google Scholar] [CrossRef]
- Hougaard, K.S.; Jackson, P.; Jensen, K.A.; Sloth, J.J.; Löschner, K.; Larsen, E.H.; Birkedal, R.K.; Vibenholt, A.; Boisen, A.-M.Z.; Wallin, H.; et al. Effects of prenatal exposure to surface-coated nanosized titanium dioxide (UV-Titan). A study in mice. Part. Fibre Toxicol. 2010, 7, 16. [Google Scholar] [CrossRef]
- Jacobsen, N.R.; Møller, P.; Jensen, K.A.; Vogel, U.; Ladefoged, O.; Loft, S.; Wallin, H. Lung inflammation and genotoxicity following pulmonary exposure to nanoparticles in ApoE−/− mice. Part. Fibre Toxicol. 2009, 6, 2. [Google Scholar] [CrossRef]
- Li, D.; Morishita, M.; Wagner, J.G.; Fatouraie, M.; Wooldridge, M.; Eagle, W.E.; Barres, J.; Carlander, U.; Emond, C.; Jolliet, O. In vivo biodistribution and physiologically based pharmacokinetic modeling of inhaled fresh and aged cerium oxide nanoparticles in rats. Part. Fibre Toxicol. 2016, 13, 45. [Google Scholar] [CrossRef]
- Fu, P.P.; Xia, Q.; Hwang, H.-M.; Ray, P.C.; Yu, H. Mechanisms of nanotoxicity: Generation of reactive oxygen species. J. Food Drug Anal. 2014, 22, 64–75. [Google Scholar] [CrossRef]
- Rocco, L.; Santonastaso, M.; Mottola, F.; Costagliola, D.; Suero, T.; Pacifico, S.; Stingo, V. Genotoxicity assessment of TiO2 nanoparticles in the teleost Danio rerio. Ecotoxicol. Environ. Saf. 2015, 113, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Mottola, F.; Iovine, C.; Santonastaso, M.; Romeo, M.L.; Pacifico, S.; Cobellis, L.; Rocco, L. NPs-TiO2 and Lincomycin Coexposure Induces DNA Damage in Cultured Human Amniotic Cells. Nanomaterials 2019, 9, 1511. [Google Scholar] [CrossRef] [PubMed]
- Mohanraj, V.J.; Chen, Y. Nanoparticles—A review. Trop. J. Pharm. Res. 2006, 5, 561–573. [Google Scholar] [CrossRef]
- Sager, T.M.; Porter, D.W.; Robinson, V.A.; Lindsley, W.G.; Schwegler-Berry, D.E.; Castranova, V. Improved method to disperse nanoparticles for in vitro and in vivo investigation of toxicity. Nanotoxicology 2007, 1, 118–129. [Google Scholar] [CrossRef]
- Aitken, R.J. Reactive oxygen species as mediators of sperm capacitation and pathological damage. Mol. Reprod. Dev. 2017, 84, 1039–1052. [Google Scholar] [CrossRef]
- Agarwal, A. Oxidative Stress and Male Infertility: From Research Bench to Clinical Practice. J. Androl. 2002, 23, 737–752. [Google Scholar] [CrossRef]
- Aitken, R.J.; Gordon, E.; Harkiss, D.; Twigg, J.P.; Milne, P.; Jennings, Z.; Irvine, D.S. Relative Impact of Oxidative Stress on the Functional Competence and Genomic Integrity of Human Spermatozoa. Biol. Reprod. 1998, 59, 1037–1046. [Google Scholar] [CrossRef]
- Aitken, R.J.; Curry, B.J. Redox regulation of human sperm function: From the physiological control of sperm capacitation to the etiology of infertility and DNA damage in the germ line. Antioxid. Redox Signal. 2011, 14, 367–381. [Google Scholar] [CrossRef]
- Das, M.; Patil, S.; Bhargava, N.; Kang, J.-F.; Riedel, L.M.; Seal, S.; Hickman, J.J. Auto-catalytic Ceria Nanoparticles Offer Neuroprotection to Adult Rat Spinal Cord Neurons. Biomaterials 2007, 28, 1918–1925. [Google Scholar] [CrossRef]
- Niu, J.; Azfer, A.; Rogers, L.M.; Wang, X.; Kolattukudy, P.E. Cardioprotective effects of cerium oxide nanoparticles in a transgenic murine model of cardiomyopathy. Cardiovasc. Res. 2007, 73, 549–559. [Google Scholar] [CrossRef]
- Auffan, M.; Rose, J.; Wiesner, M.R.; Bottero, J.-Y. Chemical stability of metallic nanoparticles: A parameter controlling their potential cellular toxicity in vitro. Environ. Pollut. 2009, 157, 1127–1133. [Google Scholar] [CrossRef] [PubMed]
- Sozarukova, M.M.; Proskurnina, E.V.; Baranchikov, A.E.; Ivanov, V.K. CeO2 nanoparticles as free radical regulators in biological systems. Nanosyst. Phys. Chem. Math. 2020, 11, 324–332. [Google Scholar] [CrossRef]
- Pulido-Reyes, G.; Rodea-Palomares, I.; Das, S.; Sakthivel, T.S.; Leganes, F.; Rosal, R.; Seal, S.; Fernández-Piñas, F. Untangling the biological effects of cerium oxide nanoparticles: The role of surface valence states. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Hayes, S.A.; Yu, P.; O’Keefe, T.J.; O’Keefe, M.J.; Stoffer, J.O. The Phase Stability of Cerium Species in Aqueous Systems. Part 1. E-pH Diagram for the Ce—HClO4—H2O System. ChemInform 2002, 34. [Google Scholar] [CrossRef]
- Guo, T.; Cui, L.; Shen, J.; Wang, R.; Zhu, W.; Xu, Y.; Qian, X. A dual-emission and large Stokes shift fluorescence probe for real-time discrimination of ROS/RNS and imaging in live cells. Chem. Commun. 2013, 49, 1862. [Google Scholar] [CrossRef] [PubMed]
- Myhre, O.; Andersen, J.M.; Aarnes, H.; Fonnum, F. Evaluation of the probes 2′,7′-dichlorofluorescin diacetate, luminol, and lucigenin as indicators of reactive species formation. Biochem. Pharmacol. 2003, 65, 1575–1582. [Google Scholar] [CrossRef]
- Mahfouz, R.; Sharma, R.; Lackner, J.; Aziz, N.; Agarwal, A. Evaluation of chemiluminescence and flow cytometry as tools in assessing production of hydrogen peroxide and superoxide anion in human spermatozoa. Fertil. Steril. 2009, 92, 819–827. [Google Scholar] [CrossRef]
- Tang, R.M.Y.; Cheah, I.K.-M.; Yew, T.S.K.; Halliwell, B. Distribution and accumulation of dietary ergothioneine and its metabolites in mouse tissues. Sci. Rep. 2018, 8, 1–15. [Google Scholar] [CrossRef]
- Benoff, S. Receptors and channels regulating acrosome reactions. Hum. Fertil. (Camb.) 1999, 2, 42–55. [Google Scholar] [CrossRef]
- Darszon, A.; Labarca, P.; Nishigaki, T.; Espinosa, F. Ion channels in sperm physiology. Physiol. Rev. 1999, 79, 481–510. [Google Scholar] [CrossRef]
- Benoff, S.; Hurley, I.; Cooper, G.W.; Mandel, F.S.; Rosenfeld, D.L.; Hershlag, A. Head-specific mannose-ligand receptor expression in human spermatozoa is dependent on capacitation-associated membrane cholesterol loss. Hum. Reprod. 1993, 8, 2141–2154. [Google Scholar] [CrossRef] [PubMed]
- Benoff, S.; Cooper, G.W.; Centola, G.M.; Jacob, A.; Hershlag, A.; Hurley, I.R. Metal ions and human sperm mannose receptors. Andrologia 2000, 32, 317–329. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Song, B.; Wu, J.; Zhang, Y.; Chen, A.; Shao, L. Potential adverse effects of nanoparticles on the reproductive system. Int. J. Nanomed. 2018, 13, 8487–8506. [Google Scholar] [CrossRef]
- Lansman, J.B. Blockade of current through single calcium channels by trivalent lanthanide cations. Effect of ionic radius on the rates of ion entry and exit. J. Gen. Physiol. 1990, 95, 679–696. [Google Scholar] [CrossRef]
- Thill, A.; Zeyons, O.; Spalla, O.; Chauvat, F.; Rose, J.; Auffan, M.; Flank, A.M. Cytotoxicity of CeO2 Nanoparticles for Escherichia coli. Physico-Chemical Insight of the Cytotoxicity Mechanism. Environ. Sci. Technol. 2006, 40, 6151–6156. [Google Scholar] [CrossRef] [PubMed]
- Zeyons, O.; Thill, A.; Chauvat, F.; Menguy, N.; Cassier-Chauvat, C.; Oréar, C.; Daraspe, J.; Auffan, M.; Rose, J.; Spalla, O. Direct and indirect CeO2 nanoparticles toxicity for Escherichia coli and Synechocystis. Nanotoxicology 2009, 3, 284–295. [Google Scholar] [CrossRef]
- Plakhova, T.V.; Romanchuk, A.Y.; Yakunin, S.N.; Dumas, T.; Demir, S.; Wang, S.; Minasian, S.G.; Shuh, D.K.; Tyliszczak, T.; Shiryaev, A.A.; et al. Solubility of Nanocrystalline Cerium Dioxide: Experimental Data and Thermodynamic Modeling. J. Phys. Chem. C 2016, 120, 22615–22626. [Google Scholar] [CrossRef]
- Lishko, P.V.; Botchkina, I.L.; Fedorenko, A.; Kirichok, Y. Acid extrusion from human spermatozoa is mediated by flagellar voltage-gated proton channel. Cell 2010, 140, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Mehrabi, K.; Nowack, B.; Arroyo Rojas Dasilva, Y.; Mitrano, D.M. Improvements in Nanoparticle Tracking Analysis To Measure Particle Aggregation and Mass Distribution: A Case Study on Engineered Nanomaterial Stability in Incineration Landfill Leachates. Environ. Sci. Technol. 2017, 51, 5611–5621. [Google Scholar] [CrossRef]
- Xia, T.; Kovochich, M.; Brant, J.; Hotze, M.; Sempf, J.; Oberley, T.; Sioutas, C.; Yeh, J.I.; Wiesner, M.R.; Nel, A.E. Comparison of the abilities of ambient and manufactured nanoparticles to induce cellular toxicity according to an oxidative stress paradigm. Nano Lett. 2006, 6, 1794–1807. [Google Scholar] [CrossRef]
- Singh, N.; Manshian, B.; Jenkins, G.J.S.; Griffiths, S.M.; Williams, P.M.; Maffeis, T.G.G.; Wright, C.J.; Doak, S.H. NanoGenotoxicology: The DNA damaging potential of engineered nanomaterials. Biomaterials 2009, 30, 3891–3914. [Google Scholar] [CrossRef] [PubMed]
- Ema, M.; Kobayashi, N.; Naya, M.; Hanai, S.; Nakanishi, J. Reproductive and developmental toxicity studies of manufactured nanomaterials. Reprod. Toxicol. 2010, 30, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Blum, J.L.; Xiong, J.Q.; Hoffman, C.; Zelikoff, J.T. Cadmium Associated With Inhaled Cadmium Oxide Nanoparticles Impacts Fetal and Neonatal Development and Growth. Toxicol. Sci. 2012, 126, 478–486. [Google Scholar] [CrossRef] [PubMed]
- Gao, G.; Ze, Y.; Li, B.; Zhao, X.; Zhang, T.; Sheng, L.; Hu, R.; Gui, S.; Sang, X.; Sun, Q.; et al. Ovarian dysfunction and gene-expressed characteristics of female mice caused by long-term exposure to titanium dioxide nanoparticles. J. Hazard. Mater. 2012, 243, 19–27. [Google Scholar] [CrossRef]
- Tassinari, R.; Cubadda, F.; Moracci, G.; Aureli, F.; D’Amato, M.; Valeri, M.; De Berardis, B.; Raggi, A.; Mantovani, A.; Passeri, D.; et al. Oral, short-term exposure to titanium dioxide nanoparticles in Sprague-Dawley rat: Focus on reproductive and endocrine systems and spleen. Nanotoxicology 2014, 8, 654–662. [Google Scholar] [CrossRef]
- Pietroiusti, A.; Massimiani, M.; Fenoglio, I.; Colonna, M.; Valentini, F.; Palleschi, G.; Camaioni, A.; Magrini, A.; Siracusa, G.; Bergamaschi, A.; et al. Low doses of pristine and oxidized single-wall carbon nanotubes affect mammalian embryonic development. ACS Nano 2011, 5, 4624–4633. [Google Scholar] [CrossRef]
- Williams, R.; Sparacino, C.; Petersen, B.; Bumgarner, J.; Jungers, R.H.; Lewtas, J. Comparative characterization of organic emissions from diesel particles, coke oven mains, roofing tar vapors and cigarette smoke condensate. Int. J. Environ. Anal. Chem. 1986, 26, 27–49. [Google Scholar] [CrossRef]
- Scheepers, P.T.J.; Bos, R.P. Combustion of diesel fuel from a toxicological perspective. Int. Arch. Occup. Environ. Heath 1992, 64, 149–161. [Google Scholar] [CrossRef]
- Brevik, A.; Lindeman, B.; Rusnakova, V.; Olsen, A.-K.; Brunborg, G.; Duale, N. Paternal Benzo[a]pyrene Exposure Affects Gene Expression in the Early Developing Mouse Embryo. Toxicol. Sci. 2012, 129, 157–165. [Google Scholar] [CrossRef]
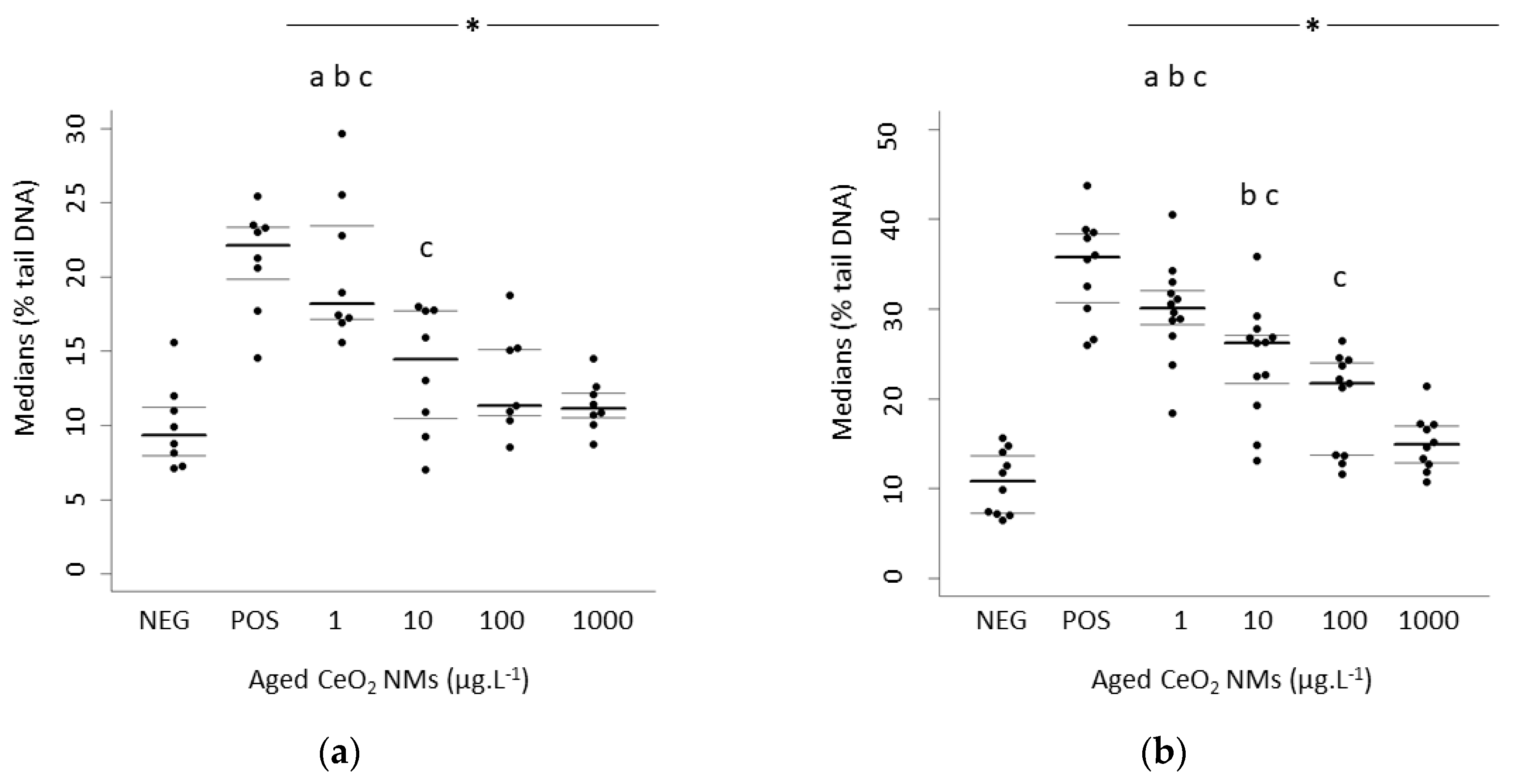

| Rat Sperm Cells | MEDIAN Values | 1st Quartile | 3rd Quartile | Human Sperm Cells | MEDIAN values | 1st Quartile | 3rd Quartile |
|---|---|---|---|---|---|---|---|
| Negative control | 9.34 | 7.94 | 11.24 | Negative control | 10.8 | 7.26 | 13.7 |
| 1 µg·L−1 CeO2 | 18.2 | 17.17 | 23.47 | 1 µg·L−1 CeO2 | 30.1 | 28.28 | 32.06 |
| 10 µg·L−1 CeO2 | 14.46 | 10.48 | 17.72 | 10 µg·L−1 CeO2 | 26.24 | 21.69 | 27.09 |
| 100 µg·L−1 CeO2 | 11.31 | 10.64 | 15.13 | 100 µg·L−1 CeO2 | 21.74 | 13.72 | 23.99 |
| 1000 µg·L−1 CeO2 | 11.15 | 10.53 | 12.19 | 1000 µg·L−1 CeO2 | 14.91 | 12.88 | 17.02 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cotena, M.; Auffan, M.; Robert, S.; Tassistro, V.; Resseguier, N.; Rose, J.; Perrin, J. CeO2 Nanomaterials from Diesel Engine Exhaust Induce DNA Damage and Oxidative Stress in Human and Rat Sperm In Vitro. Nanomaterials 2020, 10, 2327. https://doi.org/10.3390/nano10122327
Cotena M, Auffan M, Robert S, Tassistro V, Resseguier N, Rose J, Perrin J. CeO2 Nanomaterials from Diesel Engine Exhaust Induce DNA Damage and Oxidative Stress in Human and Rat Sperm In Vitro. Nanomaterials. 2020; 10(12):2327. https://doi.org/10.3390/nano10122327
Chicago/Turabian StyleCotena, Martina, Mélanie Auffan, Stéphane Robert, Virginie Tassistro, Noémie Resseguier, Jérôme Rose, and Jeanne Perrin. 2020. "CeO2 Nanomaterials from Diesel Engine Exhaust Induce DNA Damage and Oxidative Stress in Human and Rat Sperm In Vitro" Nanomaterials 10, no. 12: 2327. https://doi.org/10.3390/nano10122327
APA StyleCotena, M., Auffan, M., Robert, S., Tassistro, V., Resseguier, N., Rose, J., & Perrin, J. (2020). CeO2 Nanomaterials from Diesel Engine Exhaust Induce DNA Damage and Oxidative Stress in Human and Rat Sperm In Vitro. Nanomaterials, 10(12), 2327. https://doi.org/10.3390/nano10122327





