Hexagonal and Monoclinic Phases of La2O2CO3 Nanoparticles and Their Phase-Related CO2 Behavior
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Characterizations
3. Results and Discussion
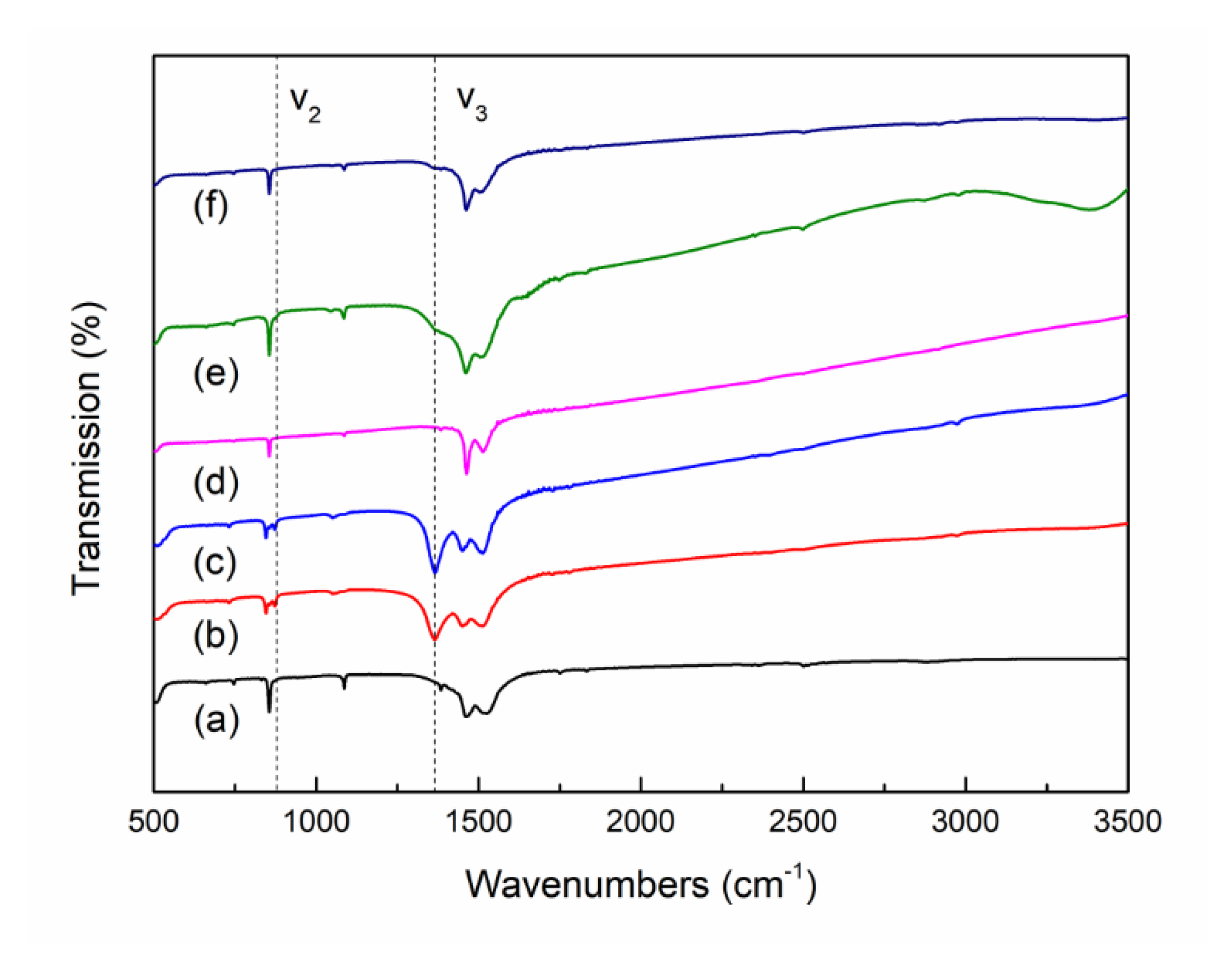
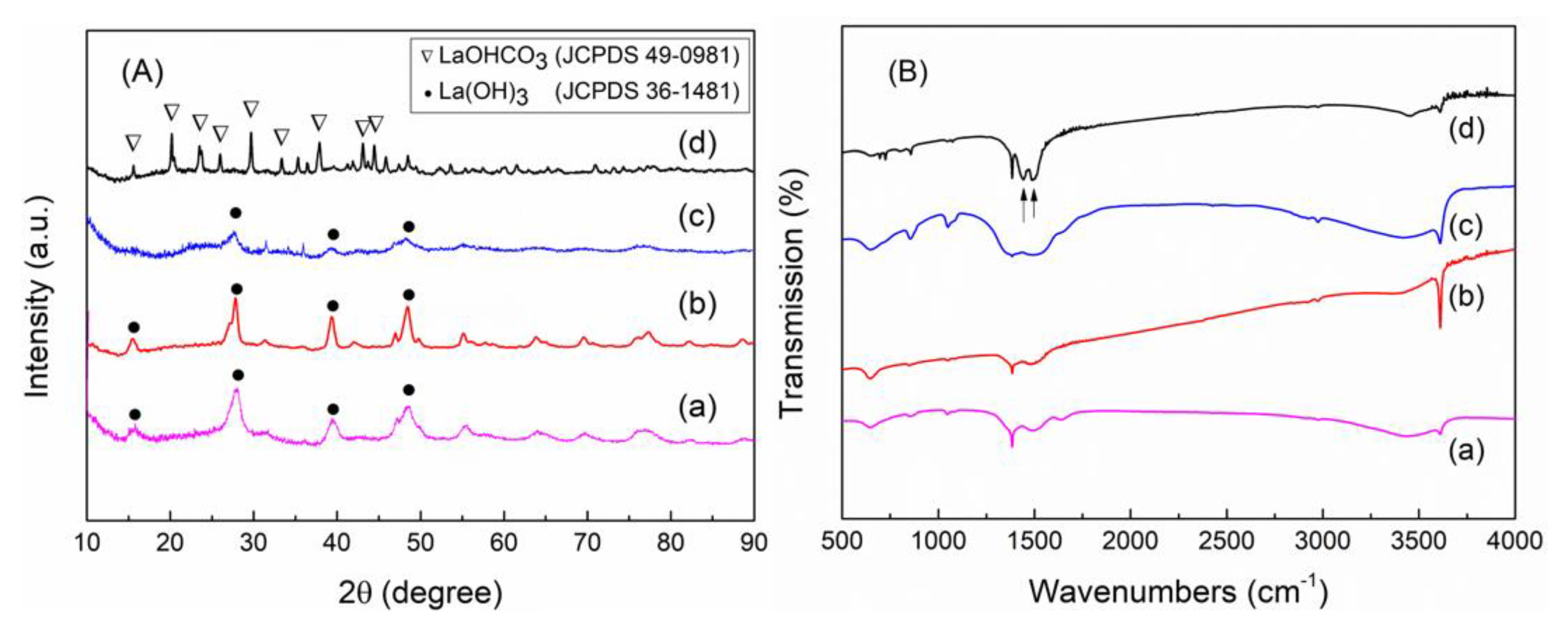
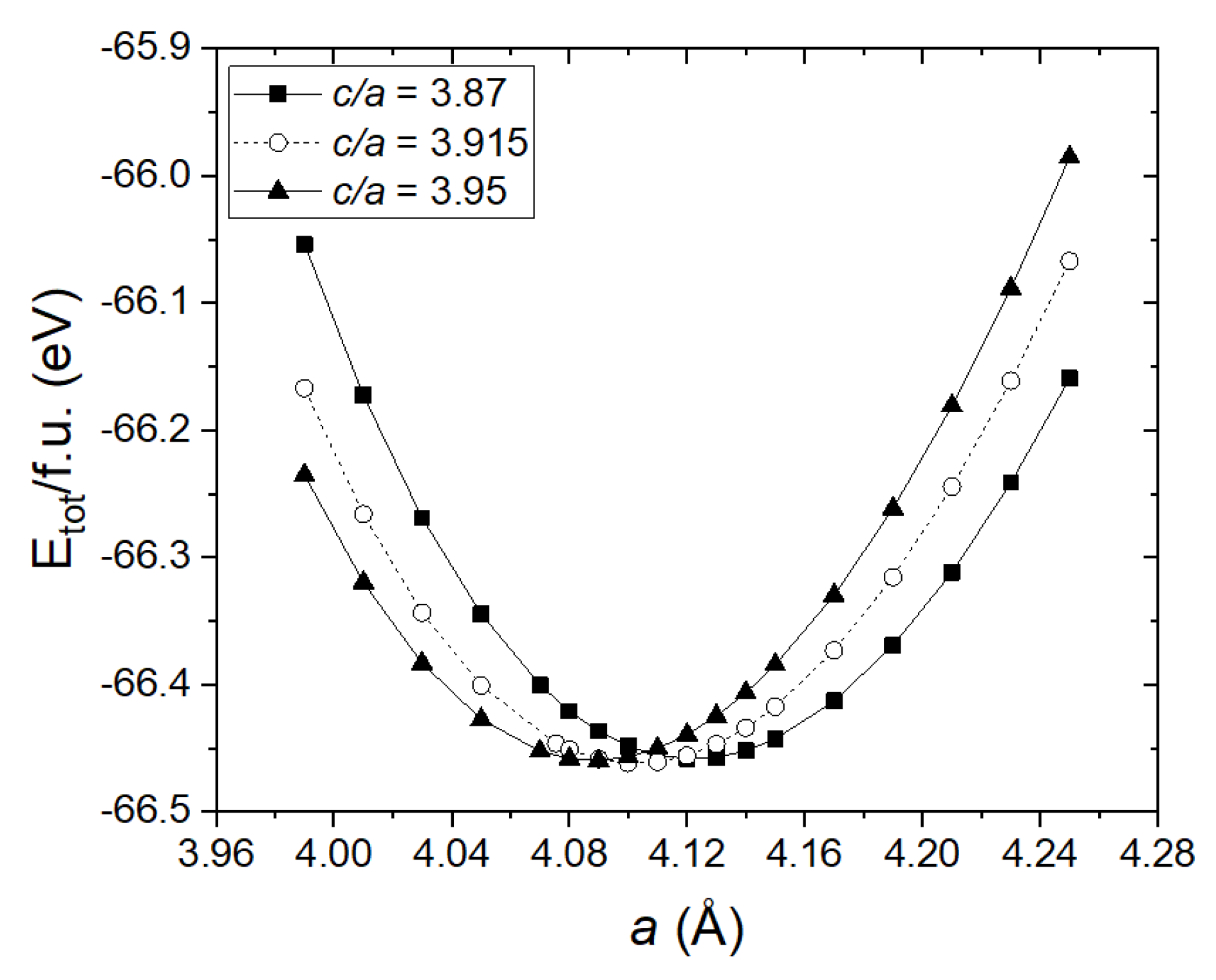
3.1. Synthesis of Monoclinic and Hexagonal La2O2CO3 Nanoparticles
3.2. CO2 Behavior on La2O2CO3 Nanoparticles
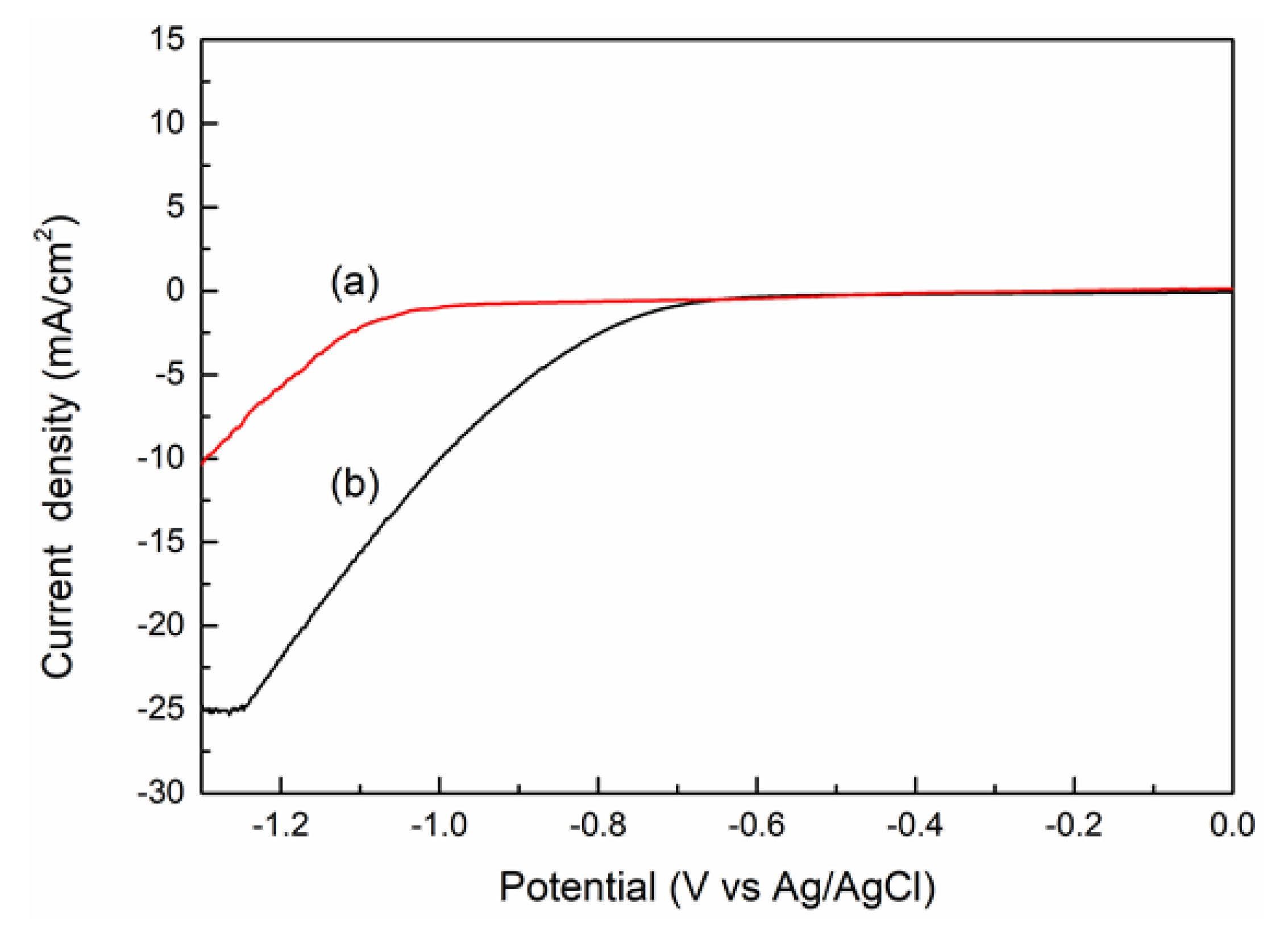
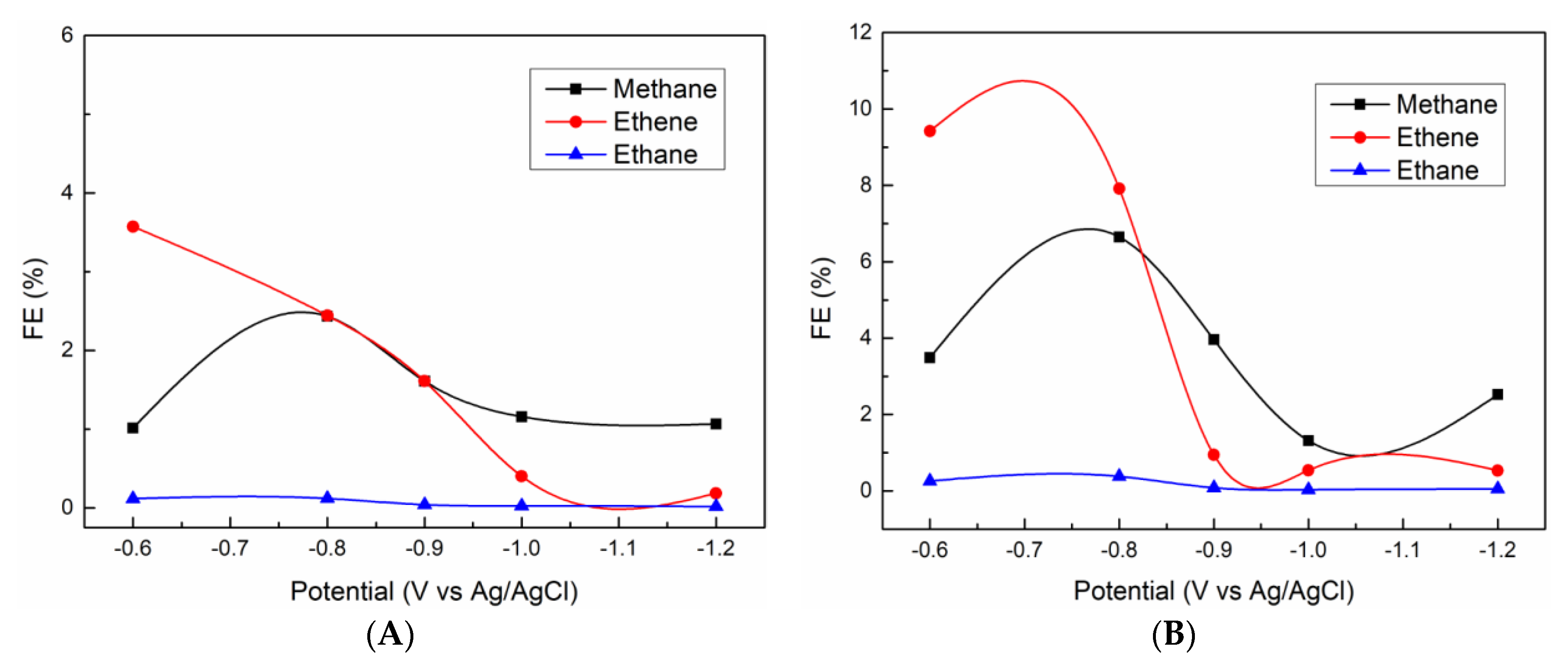
3.3. CO2 Electrochemical Reduction
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ding, D.; Lu, W.; Xiong, Y.; Pan, X.; Zhang, J.; Ling, C.; Du, Y.; Xue, Q. Facile synthesis of La2O2CO3 nanoparticle films and its CO2 sensing properties and mechanisms. Appl. Surf. Sci. 2017, 426, 725–733. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, Z.; Wei, X.; Fang, Q.; Jiang, X. The shape effect of La2O2CO3 in Pd/La2O2CO3 catalyst for selective hydrogenation of cinnamaldehyde. Appl. Catal. A Gen. 2017, 543, 196–200. [Google Scholar] [CrossRef]
- Li, X.; Li, D.; Tian, H.; Zeng, L.; Zhao, J.W.; Gong, J. Dry reforming of methane over Ni/La2O3 nanorod catalysts with stabilized Ni nanoparticles. Appl. Catal. B Environ. 2017, 202, 683–694. [Google Scholar] [CrossRef]
- Park, C.Y.; Nguyen, P.H.; Shin, E.W. Glycerol carbonation with CO2 and La2O2CO3/ZnO catalysts prepared by two different methods: Preferred reaction route depending on crystalline structure. Mol. Catal. 2017, 435, 99–109. [Google Scholar] [CrossRef]
- Bosch, C.E.; Copley, M.P.; Eralp, T.; Bilbé, E.; Thybaut, J.W.; Marin, G.B.; Collier, P. Tailoring the physical and catalytic properties of lanthanum oxycarbonate nanoparticles. Appl. Catal. A Gen. 2017, 536, 104–112. [Google Scholar] [CrossRef]
- Mu, Q.; Wang, Y. Synthesis, characterization, shape-preserved transformation, and optical properties of La(OH)3, La2O2CO3, and La2O3 nanorods. J. Alloys Compd. 2011, 509, 396–401. [Google Scholar] [CrossRef]
- Sun, C.; Sun, J.; Xiao, G.; Zhang, H.; Qiu, X.; Li, H.; Chen, L. Mesoscale organization of nearly monodisperse flowerlike ceria microspheres. J. Phys. Chem. B 2006, 110, 13445–13452. [Google Scholar] [CrossRef] [PubMed]
- Yi, G.; Lu, H.; Zhao, S.; Ge, Y.; Yang, W. Synthesis, characterization, and biological application of size-controlled nanocrystalline NaYF 4: Yb, Er phosphors. Nano Lett. 2004, 4, 2191–2196. [Google Scholar] [CrossRef]
- Li, G.; Peng, C.; Zhang, C.; Xu, Z.; Shang, M.; Yang, D.; Kang, X.; Wang, W.; Li, C.; Cheng, Z.; et al. Eu3+/Tb3+-Doped La2O2CO3/La2O3 nano/microcrystals with multiform morphologies: Facile synthesis, growth mechanism, and luminescence properties. Inorg. Chem. 2010, 49, 10522–10535. [Google Scholar] [CrossRef]
- Lin, J. Multiform oxide optical materials via the versatile pechini-type sol-gel process. Proc. Int. Meet. Inf. Disp. 2008, 8, 1247–1250. [Google Scholar] [CrossRef]
- Van Le, T.; Che, M.; Kermarec, M.; Louis, C.; Tatibouët, J.M. Structure sensitivity of the catalytic oxidative coupling of methane on lanthanum oxide. Catal. Lett. 1990, 6, 395–400. [Google Scholar] [CrossRef]
- Wang, F.; Ta, N.; Li, Y.; Shen, W. La(OH)3 and La2O2CO3 nanorod catalysts for Claisen-Schmidt condensation. Chin. J. Catal. 2014, 35, 437–443. [Google Scholar] [CrossRef]
- Lacombe, S.; Geantet, C.; Mirodatos, C. Oxidative coupling of methane over lanthana catalysts. I. Identification and role of specific active-sites. J. Catal. 1995, 151, 439–452. [Google Scholar] [CrossRef]
- Chen, H.; Yu, H.; Peng, F.; Wang, H.; Yang, J.; Pan, M. Efficient and stable oxidative steam reforming of ethanol for hydrogen production: Effect of in situ dispersion of Ir over Ir/La2O3. J. Catal. 2010, 269, 281–290. [Google Scholar] [CrossRef]
- Wang, H.; Fang, Y.; Liu, Y.; Bai, X. Perovskite LaFeO3 supported bi-metal catalyst for syngas methanation. J. Nat. Gas Chem. 2012, 21, 745–752. [Google Scholar] [CrossRef]
- Turcotte, R.P.; Sawyer, J.O.; Eyring, L. On the rare earth dioxymonocarbonates and their decomposition. Inorg. Chem. 1969, 8, 238–246. [Google Scholar] [CrossRef]
- Olafscn, A.; Fjcllvåg, H. Synthesis of rare earth oxide carbonates and thermal stability of Nd2O2CO3 II. J. Mater. Chem. 1999, 9, 2697–2702. [Google Scholar] [CrossRef]
- Hirsch, O.; Kvashnina, K.O.; Luo, L.; Süess, M.J.; Glatzel, P.; Koziej, D. High-energy resolution X-ray absorption and emission spectroscopy reveals insight into unique selectivity of La-based nanoparticles for CO2. Proc. Natl. Acad. Sci. USA 2015, 112, 15803–15808. [Google Scholar] [CrossRef]
- Hölsä, J.; Turkki, T. Preparation, thermal stability and luminescence properties of selected rare earth oxycarbonates. Thermochim. Acta 1991, 190, 335–343. [Google Scholar] [CrossRef]
- Koyabu, K.; Masui, T.; Tamura, S.; Imanaka, N. Synthesis of a new phosphor based on rare earth oxycarbonate. J. Alloys Compd. 2006, 408–412, 867–870. [Google Scholar] [CrossRef]
- Yu, H.; Men, Y.; Shin, E.W. Structural properties of disordered macroporous La2O2CO3/ZnO materials prepared by a solution combustion method. Korean J. Chem. Eng. 2019, 36, 522–528. [Google Scholar] [CrossRef]
- Kresse, G.; Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 1996, 54, 11169–11186. [Google Scholar] [CrossRef] [PubMed]
- Kresse, G.; Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 1996, 6, 15–50. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef] [PubMed]
- Monkhorst, H.J.; Pack, J.D. Special points for Brillouin-zone integrations. Phys. Rev. B 1976, 13, 5188–5192. [Google Scholar] [CrossRef]
- Hou, Y.H.; Han, W.C.; Xia, W.S.; Wan, H.L. Structure sensitivity of La2O2CO3 catalysts in the oxidative coupling of methane. ACS Catal. 2015, 5, 1663–1674. [Google Scholar] [CrossRef]
- Fleming, P.; Farrell, R.A.; Holmes, J.D.; Morris, M.A. The rapid formation of La(OH)3 from La2O3 powders on exposure to water vapor. J. Am. Ceram. Soc. 2010, 93, 1187–1194. [Google Scholar] [CrossRef]
- Fedorov, P.P.; Nazarkin, M.V.; Zakalyukin, R.M. On polymorphism and morphotropism of rare earth sesquioxides. Crystallogr. Rep. 2002, 47, 281–286. [Google Scholar] [CrossRef]
- Bakiz, B.; Guinneton, F.; Arab, M.; Benlhachenu, A.; Gavarria, J.-R. Elaboration, characterization of LaOHCO3, La2O2CO3 and La2O3 phases and their gas solid interactions with CH4 and CO gases. Moroc. J. Condens. Matter Soc. 2010, 12, 60–67. [Google Scholar]
- Rémiás, R.; Kukovecz, Á.; Darányi, M.; Kozma, G.; Varga, S.; Kónya, Z.; Kiricsi, I. Synthesis of zinc glycerolate microstacks from a ZnO nanorod sacrificial template. Eur. J. Inorg. Chem. 2009, 2009, 3622–3627. [Google Scholar] [CrossRef]
- Manoilova, O.V.; Podkolzin, S.G.; Tope, B.; Lercher, J.; Stangland, E.E.; Goupil, J.M.; Weckhuysen, B.M. Surface acidity and basicity of La2O3, LaOCl, and LaCl3 characterized by IR spectroscopy, TPD, and DFT calculations. J. Phys. Chem. B 2004, 108, 15770–15781. [Google Scholar] [CrossRef]
- Di Cosimo, J.I.; Díez, V.K.; Xu, M.; Iglesia, E.; Apesteguía, C.R. Structure and surface and catalytic properties of Mg-Al basic oxides. J. Catal. 1998, 178, 499–510. [Google Scholar] [CrossRef]
- Sholl, D.S.; Steckel, J.A. Density Functional Theory: A Practical Introduction; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2009. [Google Scholar]
- Olafsen, A.; Larsson, A.K.; Fjellvåg, H.; Hauback, B.C. On the crystal structure of Ln2O2CO3 II (Ln=La and Nd). J. Solid State Chem. 2001, 158, 14–24. [Google Scholar] [CrossRef]
- Attfield, J.P. Structure determinations of La2O2CO3-II and the unusual disordered phase La2O2.52(CO3)0.74 Li0.52 using powder diffraction. J. Solid State Chem. 1989, 82, 132–138. [Google Scholar] [CrossRef]
- Zhang, B.; Zhang, J.; Hua, M.; Wan, Q.; Su, Z.; Tan, X.; Liu, L.; Zhang, F.; Chen, G.; Tan, D.; et al. Highly electrocatalytic ethylene production from CO2 on nanodefective Cu nanosheets. J. Am. Chem. Soc. 2020, 142, 13606–13613. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Yin, R.; Shao, Q.; Zhu, T.; Huang, X. Oxygen vacancies in amorphous InOx nanoribbons enhance CO2 adsorption and activation for CO2 electroreduction. Angew. Chem. Int. Ed. 2019, 58, 5609–5613. [Google Scholar] [CrossRef] [PubMed]
- Horiuchi, T.; Hidaka, H.; Fukui, T.; Kubo, Y.; Horio, M.; Suzuki, K.; Mori, T. Effect of added basic metal oxides on CO2 adsorption on alumina at elevated temperatures. Appl. Catal. A Gen. 1998, 167, 195–202. [Google Scholar] [CrossRef]


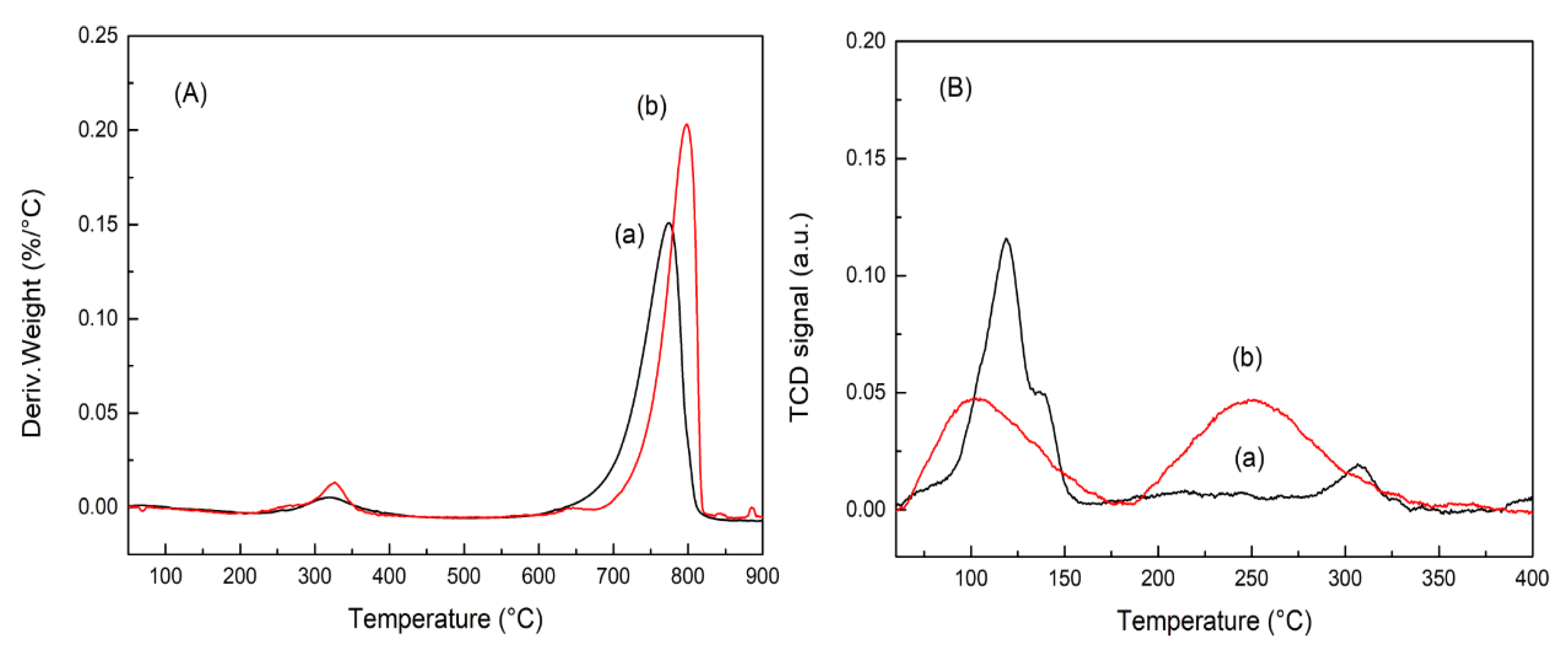

| Samples | Temperature at Maximum (°C) | Quantity (cm3/g STP) |
|---|---|---|
| PL-12h | 119 | 31.7 |
| 306 | 3.38 | |
| HL-12h | 109 | 24.6 |
| 241 | 29.0 |
| La2O2CO3 | DFT Calculated Data | Experimental Data [33,34] |
|---|---|---|
| Monoclinic | a = 12.286 Å | a = 12.239 Å |
| b = 7.097 Å | b = 7.067 Å | |
| c = 16.531 Å | c = 16.465 Å | |
| β = 75.677 | β = 75.690 | |
| Hexagonal | a = 4.100 Å | a = 4.076 Å |
| b = 4.100 Å | b = 4.076 Å | |
| c = 16.053 Å | c = 16.465 Å | |
| γ = 120 | γ = 120 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, H.; Jiang, K.; Kang, S.G.; Men, Y.; Shin, E.W. Hexagonal and Monoclinic Phases of La2O2CO3 Nanoparticles and Their Phase-Related CO2 Behavior. Nanomaterials 2020, 10, 2061. https://doi.org/10.3390/nano10102061
Yu H, Jiang K, Kang SG, Men Y, Shin EW. Hexagonal and Monoclinic Phases of La2O2CO3 Nanoparticles and Their Phase-Related CO2 Behavior. Nanomaterials. 2020; 10(10):2061. https://doi.org/10.3390/nano10102061
Chicago/Turabian StyleYu, Hongyan, Kaiming Jiang, Sung Gu Kang, Yong Men, and Eun Woo Shin. 2020. "Hexagonal and Monoclinic Phases of La2O2CO3 Nanoparticles and Their Phase-Related CO2 Behavior" Nanomaterials 10, no. 10: 2061. https://doi.org/10.3390/nano10102061
APA StyleYu, H., Jiang, K., Kang, S. G., Men, Y., & Shin, E. W. (2020). Hexagonal and Monoclinic Phases of La2O2CO3 Nanoparticles and Their Phase-Related CO2 Behavior. Nanomaterials, 10(10), 2061. https://doi.org/10.3390/nano10102061






