Towards the Continuous Hydrothermal Synthesis of ZnO@Mg2Al-CO3 Core-Shell Composite Nanomaterials
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Chemicals
2.2. Preparation of Composite Nanomaterials
2.3. Materials Characterisation
3. Results and Discussion
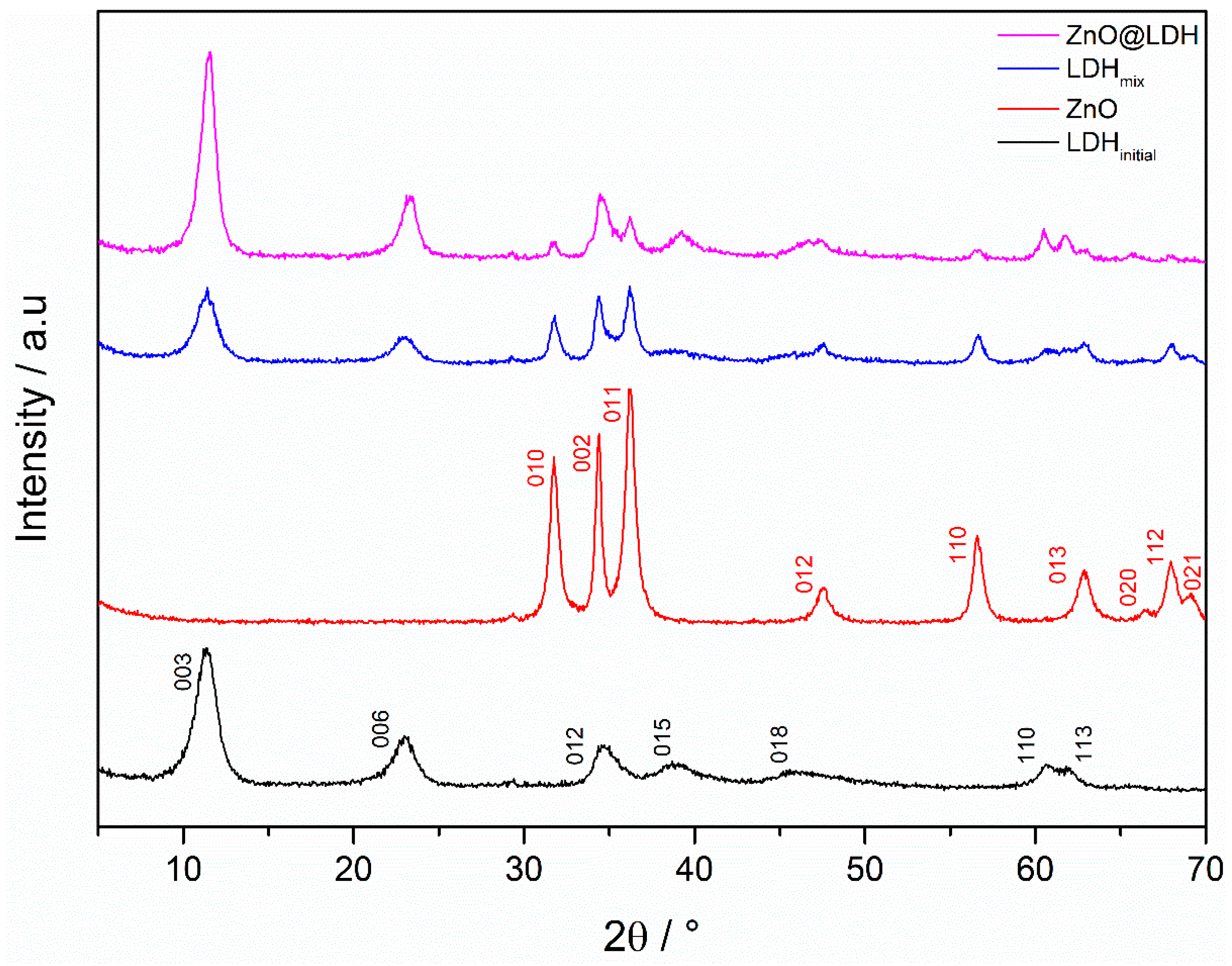
3.1. Crystal Characterisation of Composite and Single-Phase Materials
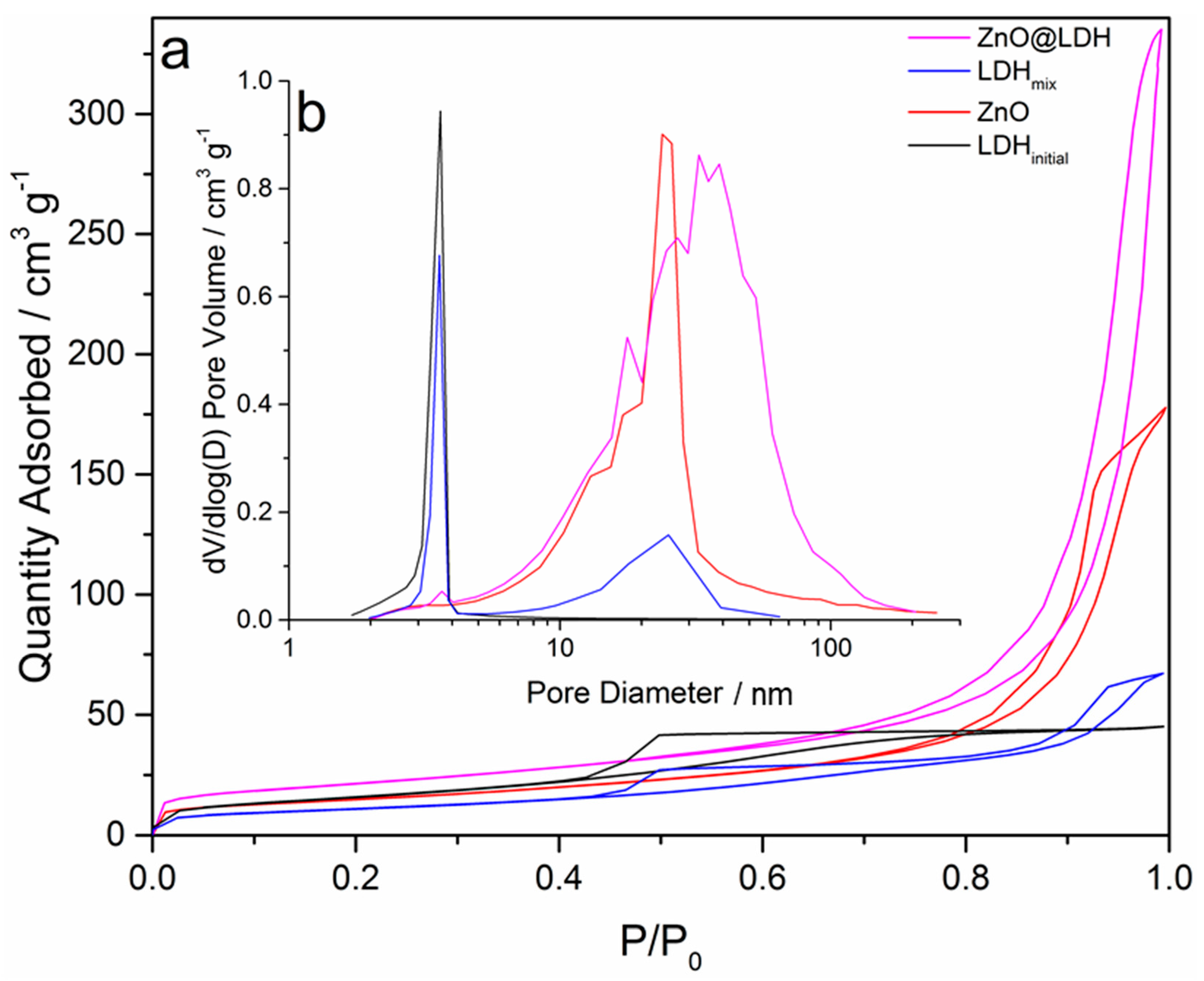
3.2. Specific Surface Area Analysis
3.3. Chemical Characterisation
3.4. Thermal Stability
3.5. Electronic Structure Characterisation
3.6. The Impact of Reactor 1 vs. Reactor 2
3.7. LDH Formation in Reactor 1 and in Reactor 2
3.8. The Impact of Residual Ions from ZnO Synthesis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Auerbach, S.M.; Carrado, K.A.; Dutta, P.K. Handbook of Layered Materials; M. Dekker: New York, NY, USA, 2004; pp. 1–664. [Google Scholar]
- Xu, R.; Pang, W.; Huo, Q. Modern Inorganic Synthetic Chemistry; Elsevier Science: Amsterdam, The Netherlands, 2011; pp. 1–590. [Google Scholar]
- Bukhtiyarova, M.V. A review on effect of synthesis conditions on the formation of layered double hydroxides. J. Solid State Chem. 2019, 269, 494–506. [Google Scholar] [CrossRef]
- Extremera, R.; Pavlovic, I.; Perez, M.R.; Barriga, C. Removal of acid orange 10 by calcined Mg/Al layered double hydroxides from water and recovery of the adsorbed dye. Chem. Eng. J. 2012, 213, 392–400. [Google Scholar] [CrossRef]
- Lv, L.; He, J.; Wei, M.; Evans, D.G.; Duan, X. Factors influencing the removal of fluoride from aqueous solution by calcined Mg-Al-CO3 layered double hydroxides. J. Hazard. Mater. 2006, 133, 119–128. [Google Scholar] [CrossRef]
- Lv, L.; He, J.; Wei, M.; Evans, D.G.; Zhou, Z.L. Treatment of high fluoride concentration water by MgAl-CO3 layered double hydroxides: Kinetic and equilibrium studies. Water Res. 2007, 41, 1534–1542. [Google Scholar] [CrossRef] [PubMed]
- Lv, L.A.; Wang, W.; Wei, M.; Cheng, H.J. Bromide ion removal from contaminated water by calcined and uncalcined MgAl-CO3 layered double hydroxides. J. Hazard. Mater. 2008, 152, 1130–1137. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.I.; Lei, L.X.; Norquist, A.J.; O’Hare, D. Intercalation and controlled release of pharmaceutically active compounds from a layered double hydroxide. Chem. Commun. 2001, 22, 2342–2343. [Google Scholar] [CrossRef]
- Gao, Y.S.; Wu, J.W.; Wang, Q.; Wilkie, C.A.; O’Hare, D. Flame retardant polymer/layered double hydroxide nanocomposites. J. Mater. Chem. A 2014, 2, 10996–11016. [Google Scholar] [CrossRef]
- Wang, Q.; Undrell, J.P.; Gao, Y.S.; Cai, G.P.; Buffet, J.C.; Wilkie, C.A.; O’Hare, D. Synthesis of Flame-Retardant Polypropylene/LDH-Borate Nanocomposites. Macromolecules 2013, 46, 6145–6150. [Google Scholar] [CrossRef]
- Shao, M.F.; Han, J.B.; Wei, M.; Evans, D.G.; Duan, X. The synthesis of hierarchical Zn-Ti layered double hydroxide for efficient visible-light photocatalysis. Chem. Eng. J. 2011, 168, 519–524. [Google Scholar] [CrossRef]
- Zhao, Y.F.; Li, B.; Wang, Q.; Gao, W.; Wang, C.L.J.; Wei, M.; Evans, D.G.; Duan, X.; O’Hare, D. NiTi-Layered double hydroxides nanosheets as efficient photocatalysts for oxygen evolution from water using visible Light. Chem. Sci. 2014, 5, 951–958. [Google Scholar] [CrossRef]
- Alejandre, A.; Medina, F.; Rodriguez, X.; Salagre, P.; Cesteros, Y.; Sueiras, J.E. Cu/Ni/Al layered double hydroxides as precursors of catalysts for the wet air oxidation of phenol aqueous solutions. Appl. Catal. B 2001, 30, 195–207. [Google Scholar] [CrossRef]
- Coq, B.; Tichit, D.; Ribet, S. Co/Ni/Mg/Al layered double hydroxides as precursors of catalysts for the hydrogenation of nitriles: Hydrogenation of acetonitrile. J. Catal. 2000, 189, 117–128. [Google Scholar] [CrossRef]
- Zhang, H.; Pan, D.K.; Zou, K.; He, J.; Duan, X. A novel core-shell structured magnetic organic-inorganic nanohybrid involving drug-intercalated layered double hydroxides coated on a magnesium ferrite core for magnetically controlled drug release. J. Mater. Chem. 2009, 19, 3069–3077. [Google Scholar] [CrossRef]
- Zhang, H.; Pan, D.K.; Duan, X. Synthesis, Characterization, and Magnetically Controlled Release Behavior of Novel Core-Shell Structural Magnetic Ibuprofen-Intercalated LDH Nanohybrids. J. Phys. Chem. C 2009, 113, 12140–12148. [Google Scholar] [CrossRef]
- Chen, C.P.; Felton, R.; Buffet, J.C.; O’Hare, D. Core-shell SiO2@LDHs with tuneable size, composition and morphology. Chem. Commun. 2015, 51, 3462–3465. [Google Scholar] [CrossRef]
- Chen, C.P.; Wang, P.H.; Lim, T.T.; Liu, L.H.; Liu, S.M.; Xu, R. A facile synthesis of monodispersed hierarchical layered double hydroxide on silica spheres for efficient removal of pharmaceuticals from water. J. Mater. Chem. A 2013, 1, 3877–3880. [Google Scholar] [CrossRef]
- Chen, C.P.; Byles, C.F.H.; Buffet, J.C.; Rees, N.H.; Wu, Y.; O’Hare, D. Core-shell zeolite@aqueous miscible organic-layered double hydroxides. Chem. Sci. 2016, 7, 1457–1461. [Google Scholar] [CrossRef]
- Guo, L.; Zhang, X.; Chen, Q.; Ruan, C.; Leng, Y. Enhanced removal performance by the core-shell zeolites/MgFe-layered double hydroxides (LDHs) for municipal wastewater treatment. Environ. Sci. Pollut. Res. 2016, 23, 6749–6757. [Google Scholar] [CrossRef] [PubMed]
- Ning, F.Y.; Shao, M.F.; Zhang, C.L.; Xu, S.M.; Wei, M.; Duan, X. Co3O4@layered double hydroxide core/shell hierarchical nanowire arrays for enhanced supercapacitance performance. Nano Energy 2014, 7, 134–142. [Google Scholar] [CrossRef]
- Li, X.; Yang, Z.C.; Qi, W.; Li, Y.T.; Wu, Y.; Zhou, S.X.; Huang, S.M.; Wei, J.; Li, H.J.; Yao, P. Binder-free Co3O4@NiCoAl-layered double hydroxide core-shell hybrid architectural nanowire arrays with enhanced electrochemical performance. Appl. Surf. Sci. 2016, 363, 381–388. [Google Scholar] [CrossRef]
- Shao, M.F.; Ning, F.Y.; Wei, M.; Evans, D.G.; Duan, X. Hierarchical Nanowire Arrays Based on ZnO Core-Layered Double Hydroxide Shell for Largely Enhanced Photoelectrochemical Water Splitting. Adv. Funct. Mater. 2014, 24, 580–586. [Google Scholar] [CrossRef]
- Trang, N.T.H.; Ngoc, H.V.; Lingappan, N.; Kang, D.J. A comparative study of supercapacitive performances of nickel cobalt layered double hydroxides coated on ZnO nanostructured arrays on textile fibre as electrodes for wearable energy storage devices. Nanoscale 2014, 6, 2434–2439. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.S.; Zhang, Q.H.; Wang, H.Z.; Liu, Z.F.; Zhao, Z. Core-shell structured ZnO@Cu-Zn-Al layered double hydroxides with enhanced photocatalytic efficiency for CO2 reduction. Catal. Commun. 2016, 77, 118–122. [Google Scholar] [CrossRef]
- Hadnadjev-Kostic, M.; Vulic, T.; Marinkovic-Neducin, R. Solar light induced rhodamine B degradation assisted by TiO2-Zn-Al LDH based photocatalysts. Adv. Powder Technol. 2014, 25, 1624–1633. [Google Scholar] [CrossRef]
- Dou, Y.B.; Zhang, S.T.; Pan, T.; Xu, S.M.; Zhou, A.W.; Pu, M.; Yan, H.; Han, J.B.; Wei, M.; Evans, D.G.; et al. TiO2@ Layered Double Hydroxide Core-Shell Nanospheres with Largely Enhanced Photocatalytic Activity Toward O-2 Generation. Adv. Funct. Mater. 2015, 25, 2243–2249. [Google Scholar] [CrossRef]
- Seftel, E.M.; Niarchos, M.; Mitropoulos, C.; Mertens, M.; Vansant, E.F.; Cool, P. Photocatalytic removal of phenol and methylene-blue in aqueous media using TiO2@LDH clay nanocomposites. Catal. Today 2015, 252, 120–127. [Google Scholar] [CrossRef]
- Yan, R.-Q.; Liu, G.-H.; Wang, Q.-F.; Liu, W.; Song, C.-L. Fast Photodegradation of Malachite Green using Nano-ZnO on Ceramic MgAl Carbonate Layered Double Hydroxides Support. Chin. J. Chem. Phys. 2016, 29, 241–244. [Google Scholar] [CrossRef]
- Hosseini, S.A.; Akbari, M. ZnO/Mg-Al Layered Double Hydroxides as a Photocatalytic Bleaching of Methylene Orange - A Black Box Modeling by Artificial Neural Network. Bull. Chem. React. Eng. Catal. 2016, 11. [Google Scholar] [CrossRef]
- Chen, D.; Li, Y.; Zhang, J.; Zhou, J.Z.; Guo, Y.; Liu, H. Magnetic Fe3O4/ZnCr-layered double hydroxide composite with enhanced adsorption and photocatalytic activity. Chem. Eng. J. 2012, 185, 120–126. [Google Scholar] [CrossRef]
- Lester, E.; Blood, P.; Denyer, J.; Giddings, D.; Azzopardi, B.; Poliakoff, M. Reaction engineering: The supercritical water hydrothermal synthesis of nano-particles. J. Supercrit. Fluids 2006, 37, 209–214. [Google Scholar] [CrossRef]
- Cheng, B.; Samulski, E.T. Hydrothermal synthesis of one-dimensional ZnO nanostructures with different aspect ratios. Chem. Commun. 2004, 986–987. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Li, F.; Zhang, R.; Evans, D.G.; Duan, X. Preparation of layered double-hydroxide nanomaterials with a uniform crystallite size using a new method involving separate nucleation and aging steps. Chem. Mater. 2002, 14, 4286–4291. [Google Scholar] [CrossRef]
- Wang, Q.; Tang, S.; Lester, E.; O’Hare, D. Synthesis of ultrafine layered double hydroxide (LDHs) nanoplates using a continuous-flow hydrothermal reactor. Nanoscale 2013, 5, 114–117. [Google Scholar] [CrossRef] [PubMed]
- Miyata, S.; Okada, A. SYNTHESIS OF HYDROTALCITE-LIKE COMPOUNDS AND THEIR PHYSICOCHEMICAL PROPERTIES - SYSTEMS MG2+-AL3+-SO42- AND MG2+-AL3+-CRO42. Clays Clay Miner. 1977, 25, 14–18. [Google Scholar] [CrossRef]
- Lee, K.M.; Lai, C.W.; Ngai, K.S.; Juan, J.C. Recent developments of zinc oxide based photocatalyst in water treatment technology: A review. Water Res. 2016, 88, 428–448. [Google Scholar] [CrossRef] [PubMed]
- Marsalek, R. Particle Size and Zeta Potential of ZnO. APCBEE Proc. 2014, 9, 13–17. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Clark, I. Continuous Synthesis and Characterisation of Layered Double Hydroxide Nanomaterials for their Application for Dye Wastewater Remediation; University of Nottingham: Nottingham, UK, 2018. [Google Scholar]
- Rives, V. Layered Double Hydroxides: Present and Future; Nova Science Publishers: Huntington, NY, USA, 2001; pp. 1–439. [Google Scholar]
- Islam, M.; Patel, R. Synthesis and physicochemical characterization of Zn/Al chloride layered double hydroxide and evaluation of its nitrate removal efficiency. Desalination 2010, 256, 120–128. [Google Scholar] [CrossRef]
- Ogawa, M.; Kaiho, H. Homogeneous precipitation of uniform hydrotalcite particles. Langmuir 2002, 18, 4240–4242. [Google Scholar] [CrossRef]
- Miyata, S. Physico-chemical Properties of Synthetic Hydrotalcites in Relation to Composition. Clays Clay Miner. 1980, 28, 50–56. [Google Scholar] [CrossRef]
- Parker, L.M.; Milestone, N.B.; Newman, R.H. The use of hydrotalcite as an anion absorbent. Ind. Eng. Chem. Res. 1995, 34, 1196–1202. [Google Scholar] [CrossRef]



| Sample | Reactor | Material | [NaOH] (mol L−1) | Down-Flow (mL min−1) | [MII + MIII] (mol L−1) | Up-Flow (mL min−1) |
|---|---|---|---|---|---|---|
| ZnO | 1 | ZnO | 0.050 | 20 | 0.05 | 10 |
| LDHinitial | 1 | Mg2Al-CO3 | 0.125 | 20 | 0.10 | 10 |
| † LDHmix | - | mixture of ZnO and LDHinitial | - | - | - | - |
| ZnO-LDH | 1 - 2 | ZnO - Mg2Al-CO3 | 0.05 - 0.15 | 40 | 0.05 - 0.05 | 20 |
| LDH-only experiments | ||||||
| LDH3 | 2 | Mg2Al-CO3 | 0.25 | 40 | 0.05 | 20 |
| * LDH4 | 2 | Mg2Al-CO3 | 0.15 | 40 | 0.05 | 20 |
| LDH5 | 2 | Mg2Al-CO3 | 0.15 | 40 | 0.05 | 20 |
| LDH6 | 2 | Mg2Al-CO3 | 0.25 | 40 | 0.05 | 20 |
| LDH7 | 2 | Mg2Al-CO3 | 0.15 | 40 | 0.05 | 20 |
| LDH8 | 2 | Mg2Al-CO3 | 0.25 | 40 | 0.05 | 20 |
| Sample | Miller Indices | a Lattice Parameter a (nm) | Miller Indices | a Lattice Parameter c (nm) |
|---|---|---|---|---|
| ZnO | 010 | 0.33 | 002 | 0.52 |
| LDHinitial | 110 | 0.30 | 003 | 2.32 |
| ZnO-LDH | 010 110 | 0.32 a 0.31 b | 002 003 | 0.52 a 2.28 b |
| Sample | XRD | TEM | |||
|---|---|---|---|---|---|
| CDL (012) (nm) | CDL (003) (nm) | CDL (110) (nm) | b Particle Size (nm) | b Particle Size (nm) | |
| ZnO | 25 | - | - | 22 ± 14 | - |
| LDHinitial | - | 8 | 25 | - | 34 ± 34 |
| ZnO-LDH | 42 | 12 | 27 | 53 ± 40 | 51 ± 22 |
| Sample | SBET (m2 g−1) | Pore Diameter (nm) | Pore Volume (cm−3 g−1) |
|---|---|---|---|
| ZnO | 53.0 ± 0.1 | 17.6 | 0.3 |
| LDHinitial | 58.2 ± 0.3 | 4.0 | 0.1 |
| LDHmix | 39.6 ± 0.1 | 7.2 | 0.1 |
| ZnO-LDH | 76.2 ± 0.1 | 17.0 | 0.7 |
| Sample | Band Gap (eV) |
|---|---|
| ZnO | 3.21 |
| LDHinitial | 5.24 |
| LDHmix | 3.22 |
| ZnO-LDH | 3.31 |
| Sample | Reactor | Flow Rate (mL min−1) | Re | CDL (003) (nm) | CDL (110) (nm) |
|---|---|---|---|---|---|
| LDHinitial | 1 | 30 | 235 | 8 | 25 |
| LDH3 | 2 | 60 | 297 | 7 | 30 |
| Sample | SBET (m2 g−1) | BJH (Barrett, Joyner, and Halenda) Pore Diameter (nm) | BJH Pore Volume (cm3 g−1) |
|---|---|---|---|
| LDHinitial | 58.2 ± 0.3 | 3.5 | 0.07 |
| LDH3 | 20.1 ± 0.1 | 4.2 | 0.04 |
| Sample | CDL (003)/nm | CDL (110)/nm |
|---|---|---|
| LDH4 | 6 | 25 |
| LDH5 | 6 | 26 |
| LDH6 | 7 | 25 |
| LDH7 | 5 | 27 |
| LDH8 | 8 | 27 |
| Sample | SBET (m2 g−1) | Pore Diameter (nm) | Pore Volume (cm3 g−1) |
|---|---|---|---|
| LDH4 | 266.8 ± 0.4 | 16.85 | 1.36 |
| LDH5 | 270.2 ± 0.5 | 15.87 | 1.29 |
| LDH6 | 22.6 ± 0.1 | 3.89 | 0.06 |
| LDH7 | 271.7 ± 0.5 | 14.55 | 1.20 |
| LDH8 | 26.1 ± 0.2 | 4.42 | 0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clark, I.; Smith, J.; Gomes, R.L.; Lester, E. Towards the Continuous Hydrothermal Synthesis of ZnO@Mg2Al-CO3 Core-Shell Composite Nanomaterials. Nanomaterials 2020, 10, 2052. https://doi.org/10.3390/nano10102052
Clark I, Smith J, Gomes RL, Lester E. Towards the Continuous Hydrothermal Synthesis of ZnO@Mg2Al-CO3 Core-Shell Composite Nanomaterials. Nanomaterials. 2020; 10(10):2052. https://doi.org/10.3390/nano10102052
Chicago/Turabian StyleClark, Ian, Jacob Smith, Rachel L. Gomes, and Edward Lester. 2020. "Towards the Continuous Hydrothermal Synthesis of ZnO@Mg2Al-CO3 Core-Shell Composite Nanomaterials" Nanomaterials 10, no. 10: 2052. https://doi.org/10.3390/nano10102052
APA StyleClark, I., Smith, J., Gomes, R. L., & Lester, E. (2020). Towards the Continuous Hydrothermal Synthesis of ZnO@Mg2Al-CO3 Core-Shell Composite Nanomaterials. Nanomaterials, 10(10), 2052. https://doi.org/10.3390/nano10102052






