A Sensitive FRET Biosensor Based on Carbon Dots-Modified Nanoporous Membrane for 8-hydroxy-2′-Deoxyguanosine (8-OHdG) Detection with Au@ZIF-8 Nanoparticles as Signal Quenchers
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of Carbon Dots (CDs)
2.3. Surface Functionalization of Nanoporous Alumina Membrane
2.4. Synthesis of Au@ZIF-8 Nanoparticles
2.5. Surface Biofunctionalization of Au@ZIF-8 Nanoparticles
2.6. Characterization
2.7. FRET Biosensor for 8-OHdG Detection
2.8. Detection of 8-OHdG in Urine Samples
3. Results
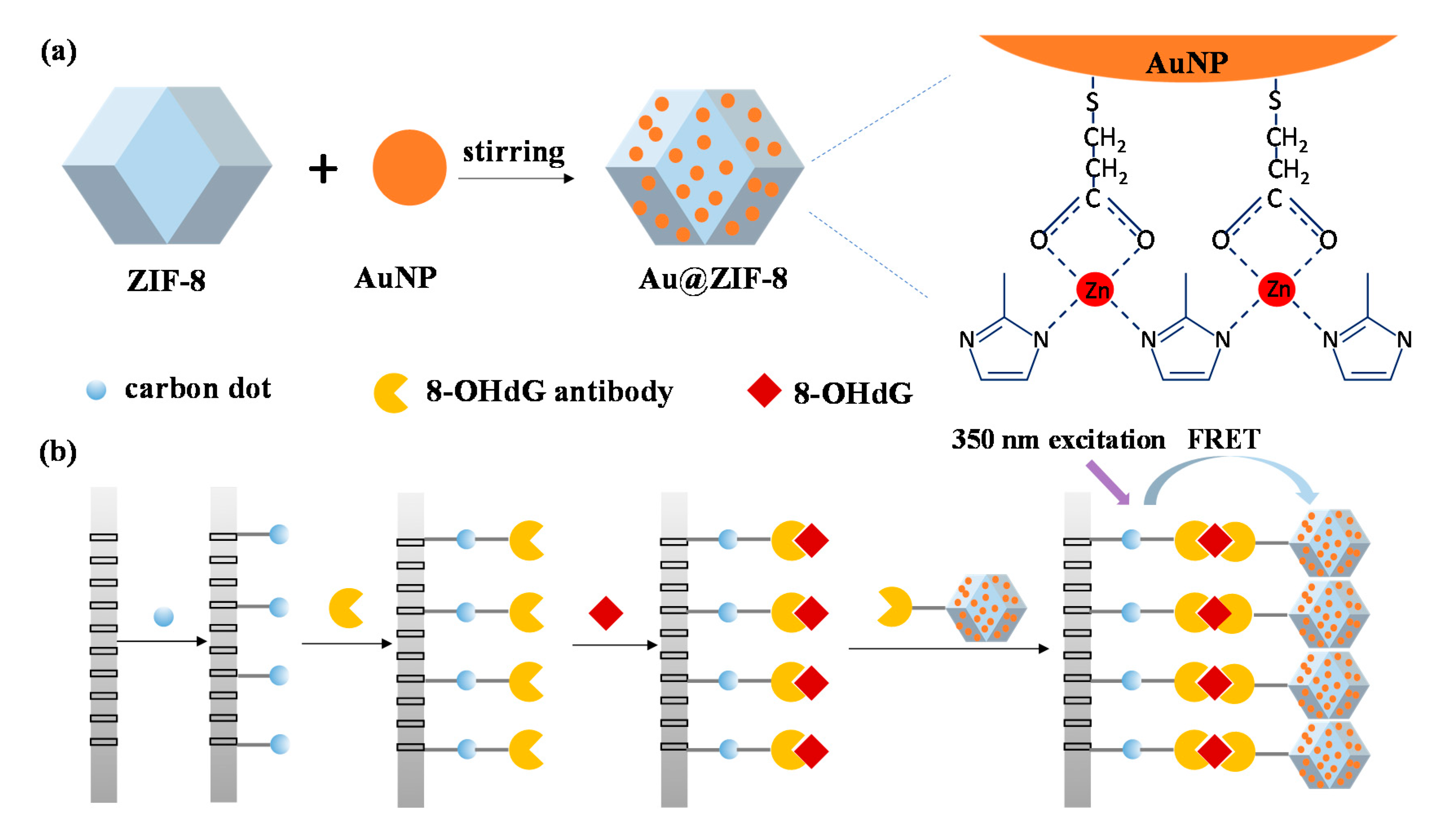
3.1. Principle of FRET Biosensor
3.2. Characterization of CDs and Au@ZIF-8 Nanoparticles
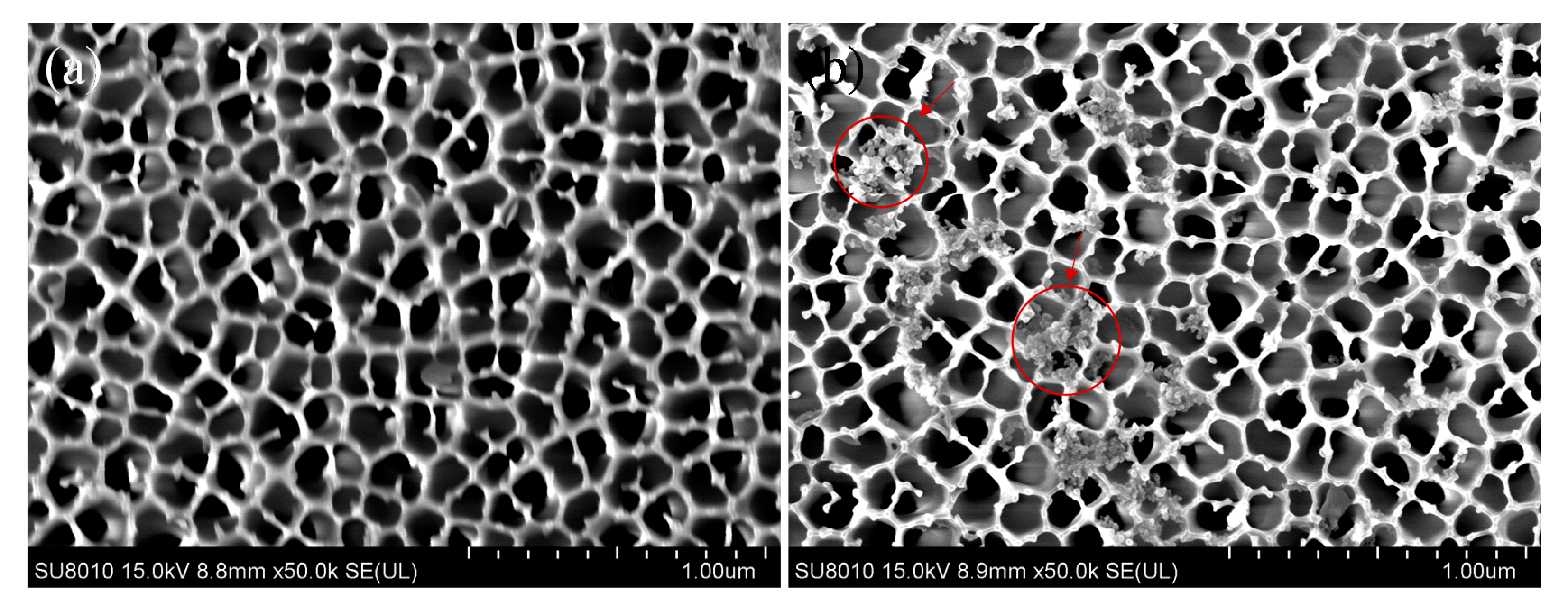
3.3. Morphologies of Sensing Surfaces
3.4. Optimization of the Experimental Conditions
3.5. Detection of 8-OHdG by the FRET Biosensor
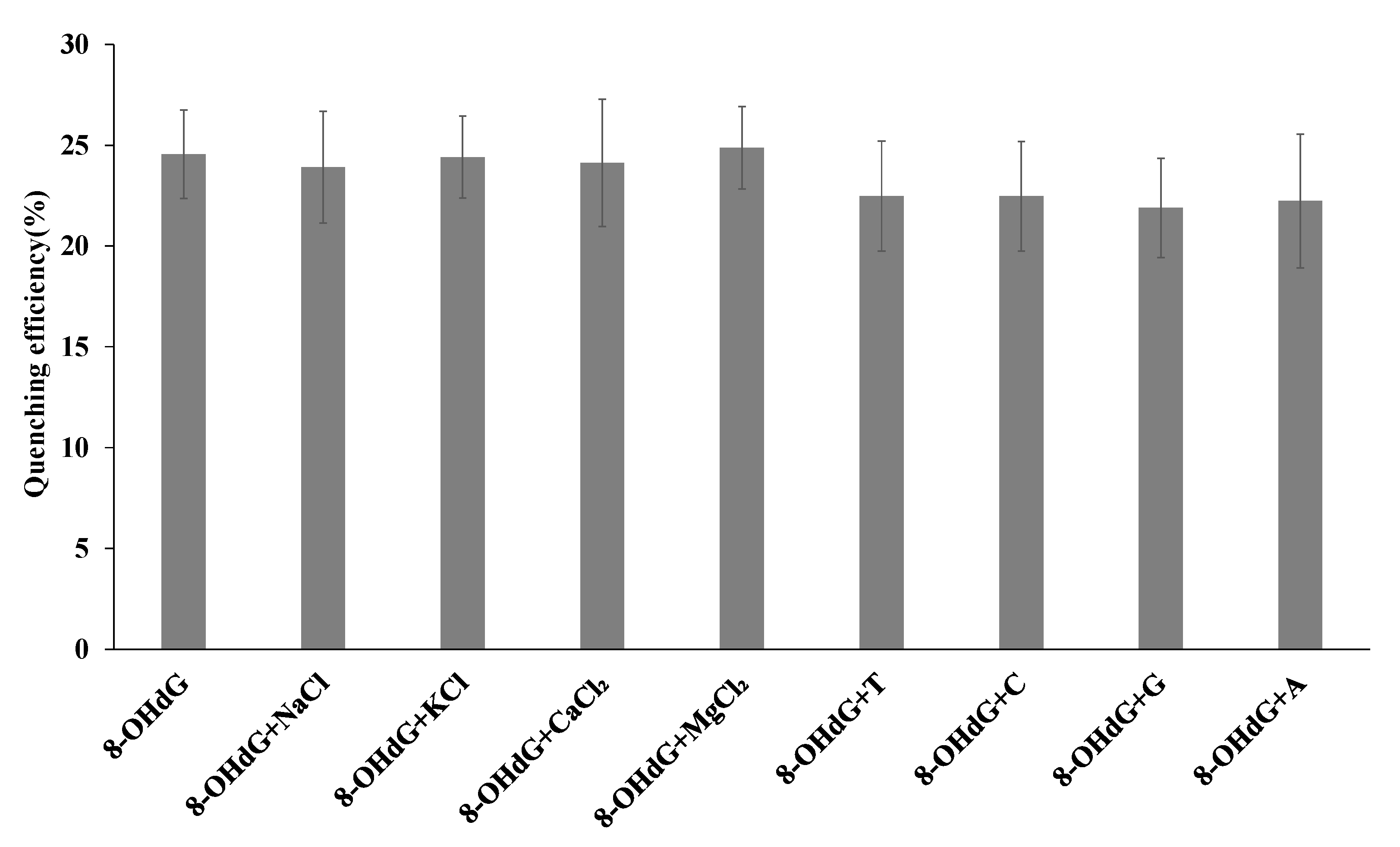
3.6. Specificity of FRET Biosensor
3.7. Analysis of Human Urine Samples
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wu, L.L.; Chiou, C.C.; Chang, P.Y.; Wu, J.T. Urinary 8-OHdG: A marker of oxidative stress to DNA and a risk factor for cancer, atherosclerosis and diabetics. Clin. Chim. Acta 2004, 339, 1–9. [Google Scholar] [CrossRef]
- Guo, Z.P.; Liu, X.H.; Liu, Y.L.; Wu, G.F.; Lu, X.Q. Constructing a novel 8-hydroxy-2′-deoxyguanosine electrochemical sensor and application in evaluating the oxidative damages of DNA and guanine. Biosens. Bioelectron. 2016, 86, 671–676. [Google Scholar] [CrossRef]
- Zhang, S.W.; Song, X.; Zhang, W.Y.; Luo, N.; Cai, L.S. Determination of low urinary 8-hydroxy-2-deoxyguanosine excretion with capillary electrophoresis and molecularly imprinted monolith solid phase microextraction. Sci. Total Environ. 2013, 450, 266–270. [Google Scholar] [CrossRef] [PubMed]
- Changenet-Barret, P.; Gustavsson, T.; Improta, R.; Markovitsi, D. Ultrafast excited-state deactivation of 8-hydroxy-2′-deoxyguanosine studied by femtosecond fluorescence spectroscopy and quantum-chemical calculations. J. Phys. Chem. A 2015, 119, 6131–6139. [Google Scholar] [CrossRef] [PubMed]
- Faria, A.M.; Peixoto, E.B.M.I.; Adamo, C.B.; Flacker, A.; Longo, E.; Mazon, T. Controlling parameters and characteristics of electrochemical biosensors for enhanced detection of 8-hydroxy-2’-deoxyguanosine. Sci. Rep. 2019, 9, 7411–7420. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Xue, K.; Yan, L. Resonance Rayleigh scattering spectral method for determination of urinary 8-hydroxy-2′-deoxyguanosine using gold nanoparticles as probe. Sensor. Actuators B Chem. 2012, 171–172, 1038–1045. [Google Scholar] [CrossRef]
- Mei, S.; Yao, Q.; Wu, C.; Xu, G. Determination of urinary 8-hydroxy-2′-deoxyguanosine by two approaches-capillary electrophoresis and GC/MS: An assay for in vivo oxidative DNA damage in cancer patients. J. Chromatogr. B. 2005, 827, 83–87. [Google Scholar] [CrossRef]
- Yano, T.; Shoji, F.; Baba, H.; Koga, T.; Shiraishi, T.; Orita, H.; Kohno, H. Significance of the urinary 8-OHdG level as an oxidative stress marker in lung cancer patients. Lung Cancer 2009, 63, 111–114. [Google Scholar] [CrossRef]
- Kasai, H. Analysis of a form of oxidative DNA damage, 8-hydroxy-2′-deoxyguanosine, as a marker of cellular oxidative stress during carcinogenesis. Mutat. Res. Rev. Mutat. 1997, 387, 147–163. [Google Scholar] [CrossRef]
- Lv, Y.Q.; Chen, S.Y.; Shen, Y.F.; Ji, J.J.; Zhou, Q.; Liu, S.Q.; Zhang, Y.J. Competitive multiple-mechanism-driven electrochemiluminescent detection of 8-hydroxy-2′-deoxyguanosine. J. Am. Chem. Soc. 2018, 140, 2801–2804. [Google Scholar] [CrossRef]
- Gupta, P.; Oyama, M.; Goyal, R.N. Electrochemical investigations of 8-hydroxydeoxyguanosine and its determination at an edge plane pyrolytic graphite electrode. RSC Adv. 2016, 6, 1722–1728. [Google Scholar] [CrossRef]
- Zhang, X.J.; Hu, Y.; Yang, X.T.; Tang, Y.Y.; Han, S.Y.; Kang, A.; Deng, H.S.; Chi, Y.M.; Zhu, D.; Lu, Y. FÖrster resonance energy transfer (FRET)-based biosensors for biological applications. Biosens. Bioelectron. 2019, 138, 111314. [Google Scholar] [CrossRef] [PubMed]
- Ammanath, G.; Yildiz, U.H.; Palaniappan, A.; Liedberg, B. Luminescent device for the detection of oxidative stress biomarkers in artificial urine. ACS Appl. Mater. Inter. 2018, 10, 7730–7736. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Wang, Y.S.; Tang, X.; Yang, H.X.; Chen, S.H.; Zhao, H.; Liu, S.D.; Zhu, Y.F.; Wang, X.F.; Huang, Y.Q. A novel fluorescence aptasensor for 8-hydroxy-2′-deoxyguanosine based on the conformational switching of K+-stabilized G-quadruplex. J. Pharmaceut. Biomed. 2016, 118, 177–182. [Google Scholar] [CrossRef]
- Rodenas, T.; Luz, I.; Prieto, G.; Seoane, B.; Miro, H.; Corma, A.; Kapteijn, F.; i Xamena, F.X.L.; Gascon, J. Metal-organic framework nanosheets in polymer composite materials for gas separation. Nat. Mater. 2015, 14, 48–55. [Google Scholar] [CrossRef]
- Lu, K.D.; Theint, A.; Guo, N.N.; Ralph, W.; Lin, W.B. Nanoscale Metal-organic frameworks for therapeutic, imaging, and sensing applications. Adv. Mater. 2018, 30, 1707634. [Google Scholar] [CrossRef]
- Cheng, C.M.; Zhang, R.L.; Wang, J.H.; Zhang, Y.; Xiong, S.S.; Huang, Y.; Yang, M. Porphyrinic metal-organic framework nanorod-based dual-modal nanoprobe for sensing and bioimaging of phosphate. ACS Appl. Mater. Interfaces 2020, 12, 26391–26398. [Google Scholar] [CrossRef]
- James, S.L. Metal-organic frameworks. Chem. Soc. Rev. 2003, 32, 276–288. [Google Scholar] [CrossRef]
- Yaghi, O.M.; O’Keeffe, M.; Ockwig, N.W.; Chae, H.K.; Eddaoudi, M.; Kim, J. Reticular synthesis and the design of new materials. Nature 2003, 423, 705–714. [Google Scholar] [CrossRef]
- Li, Z.; Zeng, H.C. Surface and bulk integrations of single-layered Au or Ag nanoparticles onto designated crystal planes {110} or {100} of ZIF-8. Chem. Mater. 2013, 25, 1761–1768. [Google Scholar] [CrossRef]
- Meng, F.C.; Zhang, S.Y.; Ma, L.; Zhang, W.N.; Li, M.; Wu, T.P.; Li, H.; Zhang, T.; Lu, X.H.; Huo, F.W.; et al. Construction of hierarchically porous nanoparticles@ metal–organic frameworks composites by inherent defects for the enhancement of catalytic efficiency. Adv. Mater. 2018, 30, 1803263. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.Y.; Peng, Y.; Wang, H.; Gu, Z.Z.; Duan, C.Y. Synthesis of Au@ ZIF-8 single-or multi-core–shell structures for photocatalysis. Chem. Commun. 2014, 50, 8651–8654. [Google Scholar] [CrossRef] [PubMed]
- Ye, W.W.; Guo, J.B.; Chen, S.; Yang, M. Nanoporous membrane based impedance sensors to detect the enzymatic activity of botulinum neurotoxin A. J. Mater. Chem. B 2013, 1, 6544–6550. [Google Scholar] [CrossRef] [PubMed]
- Ye, W.W.; Shi, J.Y.; Chan, C.Y.; Zhang, Y.; Yang, M. A nanoporous membrane based impedance sensing platform for DNA sensing with gold nanoparticle amplification. Sensor. Actuators B Chem. 2014, 193, 877–882. [Google Scholar] [CrossRef]
- Kumeria, T.; Yu, J.X.; Alsawat, M.; Kurkuri, M.D.; Santos, A.; Abell, A.D.; Losic, D. Photoswitchable membranes based on peptide-modified nanoporous anodic alumina: Toward smart membranes for on-demand molecular transport. Adv. Mater. 2015, 27, 3019–3024. [Google Scholar] [CrossRef]
- Tsang, M.K.; Ye, W.W.; Wang, G.J.; Li, J.M.; Yang, M.; Hao, J.H. Ultrasensitive detection of Ebola virus oligonucleotide based on upconversion nanoprobe/nanoporous membrane system. ACS Nano 2016, 10, 598–605. [Google Scholar] [CrossRef]
- Ye, W.W.; Chen, T.; Mao, Y.J.; Tian, F.; Sun, P.L.; Yang, M. The effect of pore size in an ultrasensitive DNA sandwich-hybridization assay for the Escherichia coli O157: H7 gene based on the use of a nanoporous alumina membrane. Microchim. Acta 2017, 184, 4835–4844. [Google Scholar] [CrossRef]
- Mao, Y.J.; Zhang, Y.; Hu, W.; Ye, W.W. Carbon dots-modified nanoporous membrane and Fe3O4@Au magnet nanocomposites-based FRET assay for ultrasensitive histamine detection. Molecules 2019, 24, 3039–3050. [Google Scholar] [CrossRef] [Green Version]
- Li, M.; Gou, H.L.; Al-Ogaidi, I.; Wu, N.Q. Nanostructured sensors for detection of heavy metals: A review. ACS Sustain. Chem. Eng. 2013, 1, 713–723. [Google Scholar] [CrossRef]
- Jiang, K.; Sun, S.; Zhang, L.; Lu, Y.; Wu, A.G.; Cai, C.Z.; Lin, H.W. Red, green, and blue luminescence by carbon dots: Full-color emission tuning and multicolor cellular imaging. Angew. Chem. Int. Edit. 2015, 127, 5360–5363. [Google Scholar] [CrossRef]
- Zhang, G.; Ji, Q.H.; Wu, Z.; Wang, G.C.; Liu, H.J.; Qu, J.H.; Li, J.H. Facile “spot-heating” synthesis of carbon dots/carbon nitride for solar hydrogen evolution synchronously with contaminant decomposition. Adv. Funct. Mater. 2018, 28, 1706462. [Google Scholar] [CrossRef]
- Li, L.B.; Yu, B.; You, T.Y. Nitrogen and sulfur co-doped carbon dots for highly selective and sensitive detection of Hg (Ⅱ) ions. Biosens. Bioelectron. 2015, 74, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.J.; Hu, D.H.; Wang, W.N.; Zhang, Q.H.; Meng, Z.; Jia, X.D.; Xi, K. Carbon dot cluster as an efficient “off–on” fluorescent probe to detect Au (III) and glutathione. Biosens. Bioelectron. 2015, 68, 27–33. [Google Scholar] [CrossRef]
- Divya, K.P.; Karthikeyan, R.; Sinduja, B.; Grace, A.A.; John, S.A.; Hahn, J.H.; Dharuman, V. Carbon dots stabilized silver–lipid nano hybrids for sensitive label free DNA detection. Biosens. Bioelectron. 2019, 133, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.X.; Liu, L.Q.; Liu, Z.D.; Wang, Y.; Zhao, X.J.; Huang, C.Z. Highly selective detection of phosphate in very complicated matrixes with an off–on fluorescent probe of europium-adjusted carbon dots. Chem. Commun. 2011, 47, 2604–2606. [Google Scholar] [CrossRef]
- Wang, B.; Liu, F.; Wu, Y.Y.; Chen, Y.F.; Weng, B.; Li, C.M. Synthesis of catalytically active multielement-doped carbon dots and application for colorimetric detection of glucose. Sensor. Actuators B Chem. 2018, 255, 2601–2607. [Google Scholar] [CrossRef]
- Dhenadhayalan, N.; Lin, K.C.; Saleh, T.A. Recent advances in functionalized carbon dots toward the design of efficient materials for sensing and catalysis applications. Small 2020, 16, 1905767. [Google Scholar] [CrossRef]
- Hu, W.; Chen, T.; Zhang, Y.; Ye, W.W. A carbon dot and gold nanoparticle-based fluorometric immunoassay for 8-hydroxy-2′-deoxyguanosine in oxidatively damaged DNA. Microchim. Acta 2019, 186, 303–311. [Google Scholar] [CrossRef]
- Ding, H.; Wei, J.S.; Xiong, H.M. Nitrogen and sulfur co-doped carbon dots with strong blue luminescence. Nanoscale 2014, 6, 13817–13823. [Google Scholar] [CrossRef]
- Schejn, A.; Balan, L.; Falk, V.; Aranda, L.; Medjahdi, G.; Schneider, R. Controlling ZIF-8 nano-and microcrystal formation and reactivity through zinc salt variations. CrystEngComm 2014, 16, 4493–4500. [Google Scholar] [CrossRef]
- Pan, Y.C.; Liu, Y.Y.; Zeng, G.F.; Zhao, L.; Lai, Z.P. Rapid synthesis of zeolitic imidazolate framework-8 (ZIF-8) nanocrystals in an aqueous system. Chem. Commun. 2011, 47, 2071–2073. [Google Scholar] [CrossRef] [PubMed]
- Ehtesabi, H.; Hallaji, Z.; Nobar, S.N.; Bagheri, Z. Carbon dots with pH-responsive fluorescence: A review on synthesis and cell biological applications. Microchim. Acta 2020, 187, 150–167. [Google Scholar] [CrossRef] [PubMed]
- Ersöz, A.; Diltemiz, S.E.; Özcan, A.A.; Denizli, A.; Say, R. 8-OHdG sensing with MIP based solid phase extraction and QCM technique. Sensor. Actuators B Chem. 2009, 137, 7–11. [Google Scholar] [CrossRef]
- Ersöz, A.; Diltemiz, S.E.; Özcan, A.A.; Denizli, A.; Say, R. Synergie between molecular imprinted polymer based on solid-phase extraction and quartz crystal microbalance technique for 8-OHdG sensing. Biosens. Bioelectron. 2008, 24, 742–747. [Google Scholar] [CrossRef]
- Hao, J.X.; Wu, K.B.; Wan, C.D.; Tang, Y. Reduced graphene oxide-ZnO nanocomposite based electrochemical sensor for sensitive and selective monitoring of 8-hydroxy-2′-deoxyguanosine. Talanta 2018, 185, 550–556. [Google Scholar] [CrossRef]
- Goyal, R.N. Determination of 8-Hydroxydeoxyguanosine: A potential biomarker of oxidative stress, using carbon-allotropic nanomaterials modified glassy carbon sensor. Talanta 2016, 161, 735–742. [Google Scholar] [CrossRef]
- Zhang, S.W.; Xing, J.; Cai, L.S.; Wu, C.Y. Molecularly imprinted monolith in-tube solid-phase microextraction coupled with HPLC/UV detection for determination of 8-hydroxy-2′-deoxyguanosine in urine. Anal. Bioanal. Chem. 2009, 395, 479–487. [Google Scholar] [CrossRef]
- Harri, M.; Kasai, H.; Mori, T.; Tornaeus, J.; Savela, K.; Peltonen, K. Analysis of 8-hydroxy-2’-deoxyguanosine in urine using high-performance liquid chromatography–electrospray tandem mass spectrometry. J. Chromatogr. B 2007, 853, 242–246. [Google Scholar] [CrossRef]


| Method Applied | Materials Used | Detection Range | LOD | References |
|---|---|---|---|---|
| QCM | Gold surface/MAAP-Fe(III) | 0.1–1 mM | 12.5 nM | [43] |
| QCM | MAH-Pt(II) | 0.1–1 μM | 8.3 nM | [44] |
| Electrochemical sensor | ZnO@rGO/GCE | 5.0–5000.0 nM | 1.25 nM | [45] |
| Voltammetric sensor | MWCNTs/ErGO | 3–75 μM | 35 nM | [46] |
| Fluorescence aptasensor | MNPs | 3.96–211 nM | 1.19 nM | [14] |
| FRET | Nanoporous alumina membrane/CDs/Au@ZIF-8 | 0.1–1750 nM | 0.31 nM | This work |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ye, W.; Zhang, Y.; Hu, W.; Wang, L.; Zhang, Y.; Wang, P. A Sensitive FRET Biosensor Based on Carbon Dots-Modified Nanoporous Membrane for 8-hydroxy-2′-Deoxyguanosine (8-OHdG) Detection with Au@ZIF-8 Nanoparticles as Signal Quenchers. Nanomaterials 2020, 10, 2044. https://doi.org/10.3390/nano10102044
Ye W, Zhang Y, Hu W, Wang L, Zhang Y, Wang P. A Sensitive FRET Biosensor Based on Carbon Dots-Modified Nanoporous Membrane for 8-hydroxy-2′-Deoxyguanosine (8-OHdG) Detection with Au@ZIF-8 Nanoparticles as Signal Quenchers. Nanomaterials. 2020; 10(10):2044. https://doi.org/10.3390/nano10102044
Chicago/Turabian StyleYe, Weiwei, Yu Zhang, Wei Hu, Liwen Wang, Yu Zhang, and Ping Wang. 2020. "A Sensitive FRET Biosensor Based on Carbon Dots-Modified Nanoporous Membrane for 8-hydroxy-2′-Deoxyguanosine (8-OHdG) Detection with Au@ZIF-8 Nanoparticles as Signal Quenchers" Nanomaterials 10, no. 10: 2044. https://doi.org/10.3390/nano10102044
APA StyleYe, W., Zhang, Y., Hu, W., Wang, L., Zhang, Y., & Wang, P. (2020). A Sensitive FRET Biosensor Based on Carbon Dots-Modified Nanoporous Membrane for 8-hydroxy-2′-Deoxyguanosine (8-OHdG) Detection with Au@ZIF-8 Nanoparticles as Signal Quenchers. Nanomaterials, 10(10), 2044. https://doi.org/10.3390/nano10102044





