Nanocellulose Bio-Based Composites for Food Packaging
Abstract
1. Introduction
2. Food Packaging Requirements
3. Nanocellulose Applications in Food Packaging
3.1. Nanocellulose Sources
3.2. Nanocellulose Based Composites
3.3. Modification of Properties, Processability and Functionalization of Nanocellulose
3.3.1. Modifications to Improve the Compatibility between Nanocellulose and Plastic Matrixes
3.3.2. Modifications to Improve Moisture Resistance
3.3.3. Modifications to Provide Active and Intelligent Functionality
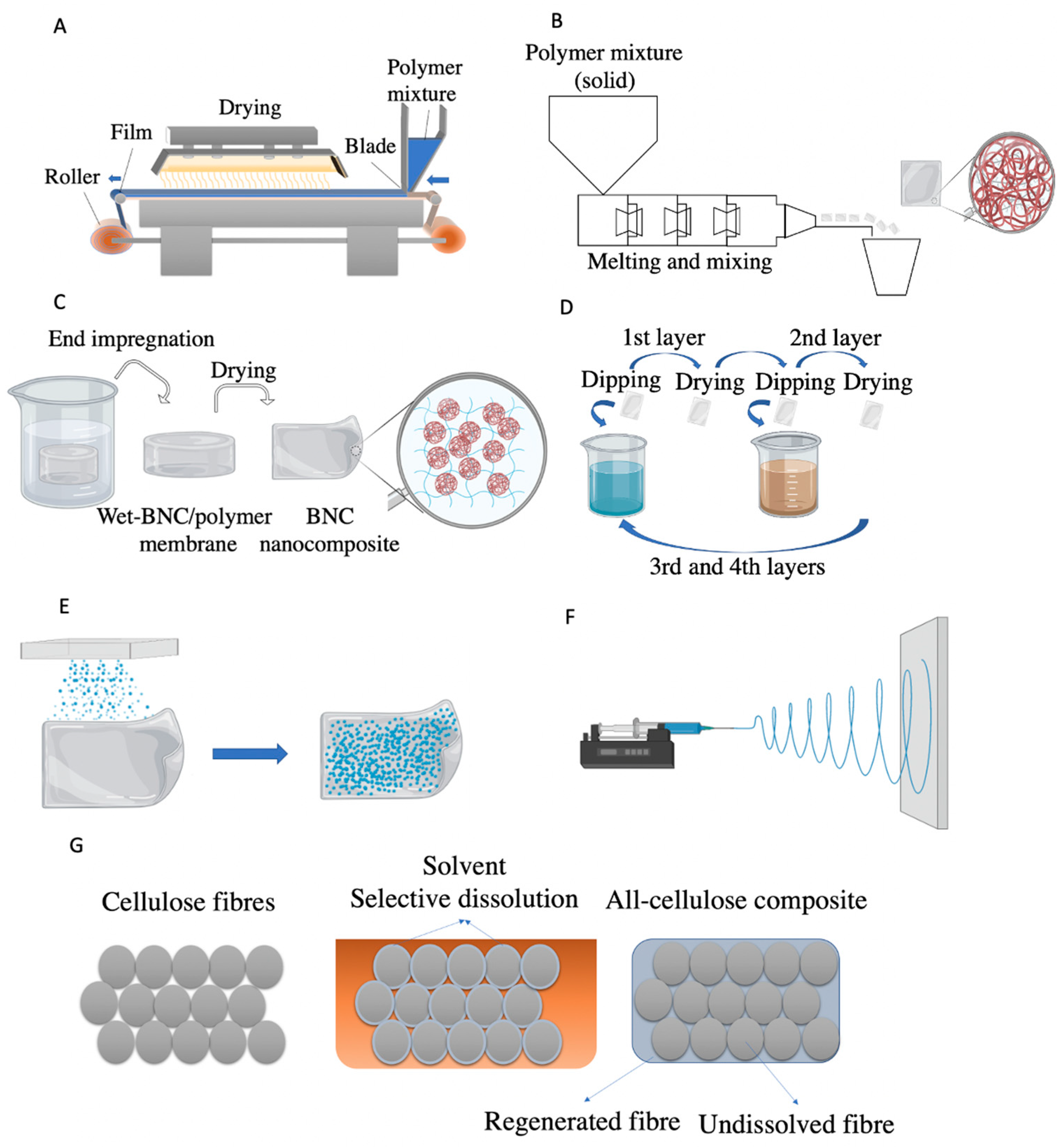
3.4. Nanocellulose Based Composites Processing
- -
- Solvent casting
- -
- Melt processing
- -
- Impregnation
- -
- Layer-by-layer assembly
- -
- Coating
- -
- All-cellulose composites
3.4.1. Solvent Casting
3.4.2. Melt Processing
3.4.3. In Situ and Impregnation Methods
3.4.4. Layer-by-Layer Assembly (LbL)
3.4.5. Coating
3.4.6. All-Cellulose Composites
- (i)
- Impregnation of previously fully dissolved cellulose into an undissolved cellulose matrix;
- (ii)
- Selective dissolution where a cellulose matrix is partially dissolved and subsequently regenerated in situ, to create a matrix around the non-dissolved portion (Figure 3G);
4. Safety of Nanocellulose Based Composites
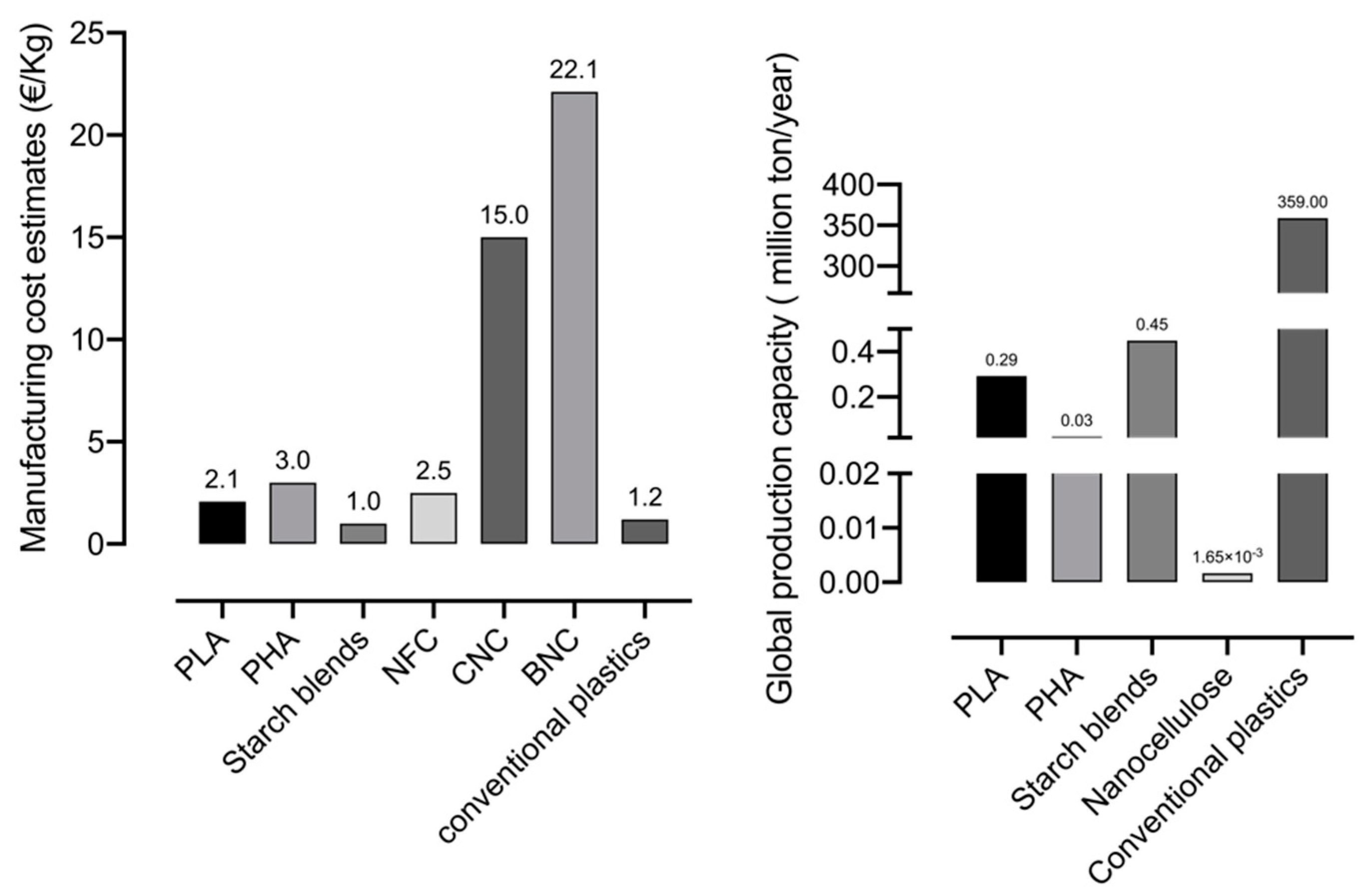
5. Technology Readiness Level of Nanocellulosic Composites
6. Final Considerations
Author Contributions
Funding
Conflicts of Interest
References
- Huang, J.; Dufresne, A.; Lin, N. Nanocellulose: From Fundamentals to Advanced Materials; John Wiley & Sons: Hoboken, NJ, USA, 2019. [Google Scholar]
- Sangroniz, A.; Zhu, J.B.; Tang, X.; Etxeberria, A.; Chen, E.Y.X.; Sardon, H. Packaging materials with desired mechanical and barrier properties and full chemical recyclability. Nat. Commun. 2019, 10, 1–7. [Google Scholar] [CrossRef]
- The European Parliament and the Council of the European Union. Directive (EU) 2019/904 of the European parliament and of council of 5 June 2019 on the reduction of the impact of certain plastic products on the environment. Off. J. Eur. Union 2019, L155, 19. [Google Scholar]
- The European Parliament and the Council of the European Union. Directive (EU) 2018/852 of the European parliament and of council of 30 May 2018 amending Directive 94/62/EC on packaging and packaging waste. Off. J. Eur. Union 2018, L150, 14. [Google Scholar]
- Eurostat. Available online: https://ec.europa.eu/eurostat/statistics-explained/index.php/Packaging_waste_statistics#Recycling_and_recovery_rates (accessed on 2 October 2020).
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, use, and fate of all plastics ever made. Sci. Adv. 2017, 3, 25–29. [Google Scholar] [CrossRef]
- De Souza Machado, A.A.; Kloas, W.; Zarfl, C.; Hempel, S.; Rillig, M.C. Microplastics as an emerging threat to terrestrial ecosystems. Global Change Biol. 2018, 24, 1405–1416. [Google Scholar] [CrossRef]
- Mondal, S. Review on nanocellulose polymer nanocomposites. Polym. Plast. Technol. Eng. 2018, 57, 1377–1391. [Google Scholar] [CrossRef]
- Khalil, H.P.S.A.; Davoudpour, Y.; Saurabh, C.K.; Hossain, M.S.; Adnan, A.S.; Dungani, R.; Paridah, M.T.; Sarker, M.Z.I.; Fazita, M.R.N.; Syakir, M.I.; et al. A review on nanocellulosic fibres as new material for sustainable packaging: Process and applications. Renew. Sustain. Energy Rev. 2016, 64, 823–836. [Google Scholar] [CrossRef]
- Lee, K.Y.; Aitomäki, Y.; Berglund, L.A.; Oksman, K.; Bismarck, A. On the use of nanocellulose as reinforcement in polymer matrix composites. Compos. Sci. Technol. 2014, 105, 15–27. [Google Scholar] [CrossRef]
- Fang, Z.; Hou, G.; Chen, C.; Hu, L. Nanocellulose-based films and their emerging applications. Curr. Opin. Solid State Mater. Sci. 2019, 23, 100764. [Google Scholar] [CrossRef]
- Khan, A.; Huq, T.; Khan, R.A.; Riedl, B.; Lacroix, M. Nanocellulose-Based Composites and Bioactive Agents for Food Packaging. Crit. Rev. Food Sci. Nutr. 2014, 54, 163–174. [Google Scholar] [CrossRef]
- Soykeabkaew, N.; Tawichai, N.; Thanomsilp, C.; Suwantong, O. Nanocellulose-reinforced ‘green’ composite materials. Walailak J. Sci. Technol. 2017, 14, 353–368. [Google Scholar]
- Hubbe, M.A.; Ferrer, A.; Tyagi, P.; Yin, Y.; Salas, C.; Pal, L.; Rojas, O.J. Nanocellulose in thin films, coatings, and plies for packaging applications: A review. BioResources 2017, 12, 2143–2233. [Google Scholar] [CrossRef]
- Kargarzadeh, H.; Mariano, M.; Huang, J.; Lin, N.; Ahmad, I.; Dufresne, A.; Thomas, S. Recent developments on nanocellulose reinforced polymer nanocomposites: A review. Polymer 2017, 132, 368–393. [Google Scholar] [CrossRef]
- Torres, F.G.; Arroyo, J.J.; Troncoso, O.P. Bacterial cellulose nanocomposites: An all-nano type of material. Mater. Sci. Eng. C 2019, 98, 1277–1293. [Google Scholar] [CrossRef]
- Lee, K.Y.; Ho, K.K.C.; Schlufter, K.; Bismarck, A. Hierarchical composites reinforced with robust short sisal fibre preforms utilising bacterial cellulose as binder. Compos. Sci. Technol. 2012, 72, 1479–1486. [Google Scholar] [CrossRef]
- Vilarinho, F.; Silva, A.S.; Vaz, M.F.; Farinha, J.P. Nanocellulose in green food packaging. Crit. Rev. Food Sci. Nutr. 2018, 58, 1526–1537. [Google Scholar] [CrossRef]
- Ferrer, A.; Pal, L.; Hubbe, M. Nanocellulose in packaging: Advances in barrier layer technologies. Ind. Crops Prod. 2017, 95, 574–582. [Google Scholar] [CrossRef]
- Bonwick, G.; Bradley, E.; Lock, I.; Romero, R. Bio-based materials for use in food contact applications. In Report to the Food Standards Agency; Fera Science Ltd.: York, UK, 2019. [Google Scholar]
- Fuertes, G.; Soto, I.; Carrasco, R.; Vargas, M.; Sabattin, J.; Lagos, C. Intelligent packaging systems: Sensors and nanosensors to monitor food quality and safety. J. Sens. 2016, 2016, 9. [Google Scholar] [CrossRef]
- Biji, K.B.; Ravishankar, C.N.; Mohan, C.O.; Srinivasa Gopal, T.K. Smart packaging systems for food applications: A review. J. Food Sci. Technol. 2015, 52, 6125–6135. [Google Scholar] [CrossRef]
- Vilela, C.; Moreirinha, C.; Domingues, E.M.; Figueiredo, F.M.L.; Almeida, A.; Freire, C.S.R. Antimicrobial and conductive nanocellulose-based films for active and intelligent food packaging. Nanomaterials 2019, 9, 980. [Google Scholar] [CrossRef]
- Wang, J.; Gardner, D.J.; Stark, N.M.; Bousfield, D.W.; Tajvidi, M.; Cai, Z. Moisture and oxygen barrier properties of cellulose nanomaterial-based films. acs sustain. Chem. Eng. 2018, 6, 49–70. [Google Scholar] [CrossRef]
- Langowski, H.-C. Shelf life & quality of packaged foods. In Food Packaging Materials-Testing & Quality Assurance; Singh, O., Wani, A.A., Langowski, H.-C., Eds.; CRC Press: Boca Raton, FL, USA, 2017; pp. 11–66. [Google Scholar]
- Paunonen, S. Strength and barrier enhancements of composites and packaging boards by nanocelluloses—A literature review. Nord. Pulp Pap. Res. J. 2013, 28, 165–181. [Google Scholar] [CrossRef]
- Sharma, A.; Thakur, M.; Bhattacharya, M.; Mandal, T.; Goswami, S. Commercial application of cellulose nano-composites—A review. Biotechnol. Rep. 2019, 21, e00316. [Google Scholar] [CrossRef] [PubMed]
- Garrison, T.F.; Murawski, A.; Quirino, R.L. Bio-based polymers with potential for biodegradability. Polymers 2016, 8, 262. [Google Scholar] [CrossRef] [PubMed]
- Fabra, M.J.; Lopez-Rubio, A.; Lagaron, J.M. Nanostructured interlayers of zein to improve the barrier properties of high barrier polyhydroxyalkanoates and other polyesters. J. Food Eng. 2014, 127, 1–9. [Google Scholar] [CrossRef]
- Pelissari, F.M.; Ferreira, D.C.; Louzada, L.B.; Santos, F.d.; Corrêa, A.C.; Moreira, F.K.V.; Mattoso, L.H. Starch-based edible films and coatings: An eco-friendly alternative for food packaging. In Starches for Food Application, 1st ed.; Clerici, M.T.P.S., Schmiele, M., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Mangaraj, S.; Goswami, T.K.; Mahajan, P.V. Applications of plastic films for modified atmosphere packaging of fruits and vegetables: A review. Food Eng. Rev. 2009, 1, 133–158. [Google Scholar] [CrossRef]
- Plackett, D. Biopolymers: New Materials for Sustainable Films and Coatings; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Teo, P.S.; Chow, W.S. Water vapour permeability of Poly(lactic acid)/Chitosan binary and ternary blends. KMUTNB Int. J. Appl. Sci. Technol. 2014, 7, 23–27. [Google Scholar]
- Espino-Pérez, E.; Bras, J.; Ducruet, V.; Guinault, A.; Dufresne, A.; Domenek, S. Influence of chemical surface modification of cellulose nanowhiskers on thermal, mechanical, and barrier properties of poly(lactide) based bionanocomposites. Eur. Polym. J. 2013, 49, 3144–3154. [Google Scholar] [CrossRef]
- Ruka, D.R.; Simon, G.P.; Dean, K.M. In situ modifications to bacterial cellulose with the water insoluble polymer poly-3-hydroxybutyrate. Carbohydr. Polym. 2013, 92, 1717–1723. [Google Scholar] [CrossRef]
- Lee, H.; You, J.; Jin, H.J.; Kwak, H.W. Chemical and physical reinforcement behavior of dialdehyde nanocellulose in PVA composite film: A comparison of nanofiber and nanocrystal. Carbohydr. Polym. 2020, 232, 115771. [Google Scholar] [CrossRef]
- Vartiainen, J.; Lahtinen, P.; Kaljunen, T.; Kunnari, V.; Peresin, M.S.; Tammelin, T. Comparison of properties between cellulose nanofibrils made from banana, sugar beet, hemp, softwood and hardwood pulps. O Pap. 2015, 76, 57–60. [Google Scholar]
- Piringer, O.G. Permeation of gases, water vapor and volatile organic compounds. In Plastic Packaging Material food barrier Function, Mass Transport, Quality Assurance and Legislation; Piringer, O.G., Baner, A.L., Eds.; Wiley-vch: Weinheim, Germany, 2000. [Google Scholar]
- Mohsen, A.H.; Ali, N.A. Mechanical, color and barrier, properties of biodegradable nanocomposites polylactic acid/nanoclay. J. Bioremediat. Biodegrad. 2018, 9, 6. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Díez-Vicente, A.L. Poly(3-hydroxybutyrate)/ZnO bionanocomposites with improved mechanical, barrier and antibacterial properties. Int. J. Mol. Sci. 2014, 15, 10950–10973. [Google Scholar] [CrossRef] [PubMed]
- Visanko, M.; Liimatainen, H.; Sirvio, J.A.; Mikkonen, K.S.; Tenkanen, M.; Sliz, R.; Hormi, O.; Niinimaki, J. Butylamino-functionalized cellulose nanocrystal films: Barrier properties and mechanical strength. RSC Adv. 2015, 5, 15140–15146. [Google Scholar] [CrossRef]
- Maes, C.; Luyten, W.; Herremans, G.; Peeters, R.; Carleer, R.; Buntinx, M. Recent Updates on the barrier properties of ethylene vinyl alcohol copolymer (EVOH): A review. Polym. Rev. 2018, 58, 209–246. [Google Scholar] [CrossRef]
- Bastarrachea, L.; Dhawan, S.; Sablani, S.S. Engineering properties of polymeric-based antimicrobial films for food packaging. Food Eng. Rev. 2011, 3, 79–93. [Google Scholar] [CrossRef]
- Nunes, T.F.G. Produção, Caracterização e Aplicação de Nanofibras de Celulose. Master’s Thesis, Engenharia Química da Faculdade de Ciências e Tecnologia, Universidade de Coimbra, Coimbra, Portugal, February 2014. [Google Scholar]
- Paunonen, S. Strength and barrier enhancements of cellophane and cellulose derivative films: A review. Bioesources 2013, 8, 3098–3121. [Google Scholar] [CrossRef]
- Jozala, A.F.; Lencastre-Novaes, L.C.; Lopes, A.M.; Santos-Ebinuma, V.C.; Mazzola, P.G.; Pessoa, A.; Grotto, D.; Gerenutti, M.; Chaud, M.V. Bacterial nanocellulose production and application: A 10-year overview. Appl. Microbiol. Biotechnol. 2016, 100, 2063–2072. [Google Scholar] [CrossRef]
- Bharimalla, A.K.; Deshmukh, S.P.; Vigneshwaran, N.; Patil, P.G.; Prasad, V. Nanocellulose-polymer composites for applications in food packaging: Current status, future prospects and challenges. Polym. Plast. Technol. Eng. 2017, 56, 805–823. [Google Scholar] [CrossRef]
- De Amorim, J.D.P.; de Souza, K.C.; Duarte, C.R.; Duarte, I.S.; Ribeiro, F.A.S.; Silva, G.S.; Farias, P.M.A.; Stingl, A.; Costa, A.F.S.; Vinhas, G.M.; et al. Plant and bacterial nanocellulose: Production, properties and applications in medicine, food, cosmetics, electronics and engineering. A review. Environ. Chem. Lett. 2020, 18, 851–869. [Google Scholar] [CrossRef]
- Gama, M.; Dourado, F. Bacterial Nanocellulose; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Kumar, V.; Elfving, A.; Koivula, H.; Bousfield, D.; Toivakka, M. Roll-to-Roll processed cellulose nanofiber coatings. Ind. Eng. Chem. Res. 2016, 55, 3603–3613. [Google Scholar] [CrossRef]
- Amini, E.; Azadfallah, M.; Layeghi, M.; Talaei-Hassanloui, R. Silver-nanoparticle-impregnated cellulose nanofiber coating for packaging paper. Cellulose 2016, 23, 557–570. [Google Scholar] [CrossRef]
- Mascheroni, E.; Rampazzo, R.; Ortenzi, M.A.; Piva, G.; Bonetti, S.; Piergiovanni, L. Comparison of cellulose nanocrystals obtained by sulfuric acid hydrolysis and ammonium persulfate, to be used as coating on flexible food-packaging materials. Cellulose 2016, 23, 779–793. [Google Scholar] [CrossRef]
- Bideau, B.; Loranger, E.; Daneault, C. Nanocellulose-polypyrrole-coated paperboard for food packaging application. Prog. Org. Coatings 2018, 123, 128–133. [Google Scholar] [CrossRef]
- Tyagi, P.; Lucia, L.A.; Hubbe, M.A.; Pal, L. Nanocellulose-based multilayer barrier coatings for gas, oil, and grease resistance. Carbohydr. Polym. 2019, 206, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Koppolu, R.; Lahti, J.; Abitbol, T.; Swerin, A.; Kuusipalo, J.; Toivakka, M. Continuous processing of nanocellulose and polylactic acid into multilayer barrier coatings. ACS Appl. Mater. Interfaces 2019, 11, 11920–11927. [Google Scholar] [CrossRef]
- Meriçer, Ç.; Minelli, M.; de Angelis, M.G.; Baschetti, M.G.; Stancampiano, A.; Laurita, R.; Gherardi, M.; Colombo, V.; Trifol, J.; Szabo, P.; et al. Atmospheric plasma assisted PLA/microfibrillated cellulose (MFC) multilayer biocomposite for sustainable barrier application. Ind. Crops Prod. 2016, 93, 235–243. [Google Scholar] [CrossRef]
- Vähä-Nissi, M.; Koivula, H.M.; Raisanen, H.M.; Vartiainen, J.; Ragni, P.; Kentta, E.; Kaljunen, T.; Malm, T.; Minkkinen, H.; Harlin, A. Cellulose nanofibrils in biobased multilayer films for food packaging. J. Appl. Polym. Sci. 2017, 134, 2–9. [Google Scholar] [CrossRef]
- Gitari, B.; Chang, P.; Misra, M.; Navabi, A.; Mohanty, A.K. A comparative study on the mechanical, thermal, and water barrier properties of PLA nanocomposite films prepared with bacterial nanocellulose and cellulose nanofibrils. BioResources 2019, 14, 1867–1889. [Google Scholar]
- Panaitescu, D.M.; Frone, A.N.; Chiulan, I.; Gabor, R.A.; Spataru, I.C.; Căşărică, A. Biocomposites from polylactic acid and bacterial cellulose nanofibers obtained by mechanical treatment. BioResources 2017, 12, 662–672. [Google Scholar] [CrossRef]
- Abdulkhani, A.; Hosseinzadeh, J.; Ashori, A.; Dadashi, S.; Takzare, Z. Preparation and characterization of modified cellulose nanofibers reinforced polylactic acid nanocomposite. Polym. Test. 2014, 35, 73–79. [Google Scholar] [CrossRef]
- Arrieta, M.P.; Fortunati, E.; Dominici, F.; Rayón, E.; López, J.; Kenny, J.M. Multifunctional PLA-PHB/cellulose nanocrystal films: Processing, structural and thermal properties. Carbohydr. Polym. 2014, 107, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Arrieta, M.P.; Fortunati, E.; Dominici, F.; López, J.; Kenny, J.M. Bionanocomposite films based on plasticized PLA-PHB/cellulose nanocrystal blends. Carbohydr. Polym. 2015, 121, 265–275. [Google Scholar] [CrossRef] [PubMed]
- Slavutsky, A.M.; Bertuzzi, M.A. Water barrier properties of starch films reinforced with cellulose nanocrystals obtained from sugarcane bagasse. Carbohydr. Polym. 2014, 110, 53–61. [Google Scholar] [CrossRef]
- Montero, B.; Rico, M.; Rodríguez-Llamazares, S.; Barral, L.; Bouza, R. Effect of nanocellulose as a filler on biodegradable thermoplastic starch films from tuber, cereal and legume. Carbohydr. Polym. 2017, 157, 1094–1104. [Google Scholar] [CrossRef]
- Martins, I.M.G.; Magina, S.P.; Oliveira, S.; Freire, C.S.R.; Silvestre, A.J.D.; Neto, C.P.; Gandini, A. New biocomposites based on thermoplastic starch and bacterial cellulose. Compos. Sci. Technol. 2009, 69, 2163–2168. [Google Scholar] [CrossRef]
- Wan, Y.Z.; Luo, H.; He, F.; Liang, H.; Huang, Y.; Li, X.L. Mechanical, moisture absorption, and biodegradation behaviours of bacterial cellulose fibre-reinforced starch biocomposites. Compos. Sci. Technol. 2009, 69, 1212–1217. [Google Scholar] [CrossRef]
- Savadekar, N.R.; Mhaske, S.T. Synthesis of nano cellulose fibers and effect on thermoplastics starch based films. Carbohydr. Polym. 2012, 89, 146–151. [Google Scholar] [CrossRef]
- Soykeabkaew, N.; Laosat, N.; Ngaokla, A.; Yodsuwan, N.; Tunkasiri, T. Reinforcing potential of micro- and nano-sized fibers in the starch-based biocomposites. Compos. Sci. Technol. 2012, 72, 845–852. [Google Scholar] [CrossRef]
- Hietala, M.; Mathew, A.P.; Oksman, K. Bionanocomposites of thermoplastic starch and cellulose nanofibers manufactured using twin-screw extrusion. Eur. Polym. J. 2013, 49, 950–956. [Google Scholar] [CrossRef]
- Martínez-Sanz, M.; Lopez-Rubio, A.; Villano, M.; Oliveira, C.S.S.; Majone, M.; Reis, M.; Lagarón, J.M. Production of bacterial nanobiocomposites of polyhydroxyalkanoates derived from waste and bacterial nanocellulose by the electrospinning enabling melt compounding method. J. Appl. Polym. Sci. 2016, 133, 42486. [Google Scholar] [CrossRef]
- Seoane, I.T.; Cerrutti, P.; Vazquez, A.; Manfredi, L.B.; Cyras, V.P. Polyhydroxybutyrate-based nanocomposites with cellulose nanocrystals and bacterial cellulose. J. Polym. Environ. 2017, 25, 586–598. [Google Scholar] [CrossRef]
- Espino-Pérez, E.; Bras, J.; Almeida, G.; Plessis, C.; Belgacem, N.; Perré, P.; Domenek, S. Designed cellulose nanocrystal surface properties for improving barrier properties in polylactide nanocomposites. Carbohydr. Polym. 2018, 183, 267–277. [Google Scholar] [CrossRef] [PubMed]
- Osterberg, M.; Vartiainen, J.; Lucenius, J.; Hippi, U.; Seppalä, J.; Serimaa, R.; Laine, J. A fast method to produce strong NFC films as a platform for barrier and functional materials. ACS Appl. Mater. Interfaces 2013, 5, 4640–4647. [Google Scholar] [CrossRef] [PubMed]
- Cherpinski, A.; Torres-Giner, S.; Vartiainen, J.; Peresin, M.S.; Lahtinen, P.; Lagaron, J.M. Improving the water resistance of nanocellulose-based films with polyhydroxyalkanoates processed by the electrospinning coating technique. Cellulose 2018, 25, 1291–1307. [Google Scholar] [CrossRef]
- Ramezani, H.; Behzad, T.; Bagheri, R. Synergistic effect of graphene oxide nanoplatelets and cellulose nanofibers on mechanical, thermal, and barrier properties of thermoplastic starch. Polym. Adv. Technol. 2020, 31, 553–565. [Google Scholar] [CrossRef]
- González, K.; Retegi, A.; González, A.; Eceiza, A.; Gabilondo, N. Starch and cellulose nanocrystals together into thermoplastic starch bionanocomposites. Carbohydr. Polym. 2015, 117, 83–90. [Google Scholar] [CrossRef]
- Jung, B.N.; Jung, H.W.; Kang, D.H.; Kim, G.H.; Lee, M.; Shim, J.K.; Hwang, S.W. The fabrication of flexible and oxygen barrier cellulose nanofiber/polylactic acid nanocomposites using cosolvent system. J. Appl. Polym. Sci. 2019, 137, e49536. [Google Scholar] [CrossRef]
- Ambrosio-Martín, J.; Fabra, M.J.; Lopez-Rubio, A.; Lagaron, J.M. Melt polycondensation to improve the dispersion of bacterial cellulose into polylactide via melt compounding: Enhanced barrier and mechanical properties. Cellulose 2015, 22, 1201–1226. [Google Scholar] [CrossRef]
- Dhar, P.; Bhardwaj, U.; Kumar, A.; Katiyar, V. Poly (3-hydroxybutyrate)/Cellulose Nanocrystal films for food packaging applications: Barrier and migration studies. Polym. Eng. Sci. 2015, 55, 2388–2395. [Google Scholar] [CrossRef]
- Grande, C.J.; Torres, F.G.; Gomez, C.M.; Troncoso, O.P.; Canet-Ferrer, J.; Martínez-Pastor, J. Development of self-assembled bacterial cellulose-starch nanocomposites. Mater. Sci. Eng. C 2009, 29, 1098–1104. [Google Scholar] [CrossRef]
- Aulin, C.; Salazar-Alvarez, G.; Lindström, T. High strength, flexible and transparent nanofibrillated cellulose-nanoclay biohybrid films with tunable oxygen and water vapor permeability. Nanoscale 2012, 4, 6622–6628. [Google Scholar] [CrossRef] [PubMed]
- Tomé, L.C.; Freire, M.G.; Rebelo, L.P.N.; Silvestre, A.J.; Neto, C.P.; Marrucho, I.M.; Freire, C.S. Surface hydrophobization of bacterial and vegetable cellulose fibers using ionic liquids as solvent media and catalysts. Green Chem. 2011, 13, 2464–2470. [Google Scholar] [CrossRef]
- Lee, K.Y.; Quero, F.; Blaker, J.J.; Hill, C.A.S.; Eichhorn, S.J.; Bismarck, A. Surface only modification of bacterial cellulose nanofibres with organic acids. Cellulose 2011, 18, 595–605. [Google Scholar] [CrossRef]
- Frone, A.N.; Panaitescu, D.M.; Chiulan, I.; Nicolae, C.A.; Casarica, A.; Gabor, A.R.; Trusca, R.; Damian, C.M.; Purcar, V.; Alexandrescu, E.; et al. Surface treatment of bacterial cellulose in mild, eco-friendly conditions. Coatings 2018, 8, 221. [Google Scholar] [CrossRef]
- Fernandes, S.C.M.; Sadocco, P.; Alonso-Varona, A.; Palomares, T.; Eceiza, A.; Silvestre, A.J.D.; Mondragon, I.; Freire, C.S.R. Bioinspired antimicrobial and biocompatible bacterial cellulose membranes obtained by surface functionalization with aminoalkyl groups. ACS Appl. Mater. Interfaces 2013, 5, 3290–3297. [Google Scholar] [CrossRef]
- Tsai, Y.H.; Yang, Y.N.; Ho, Y.C.; Tsai, M.L.; Mi, F.L. Drug release and antioxidant/antibacterial activities of silymarin-zein nanoparticle/bacterial cellulose nanofiber composite films. Carbohydr. Polym. 2018, 180, 286–296. [Google Scholar] [CrossRef]
- Shao, W.; Wu, J.; Liu, H.; Ye, S.; Jiang, L.; Liu, X. Novel bioactive surface functionalization of bacterial cellulose membrane. Carbohydr. Polym. 2017, 178, 270–276. [Google Scholar] [CrossRef]
- Bayazidi, P.; Almasi, H.; Asl, A.K. Immobilization of lysozyme on bacterial cellulose nanofibers: Characteristics, antimicrobial activity and morphological properties. Int. J. Biol. Macromol. 2018, 107, 2544–2551. [Google Scholar] [CrossRef]
- Chi, K.; Catchmark, J.M. Enhanced dispersion and interface compatibilization of crystalline nanocellulose in polylactide by surfactant adsorption. Cellulose 2017, 24, 4845–4860. [Google Scholar] [CrossRef]
- Aulin, C.; Gällstedt, M.; Lindström, T. Oxygen and oil barrier properties of microfibrillated cellulose films and coatings. Cellulose 2010, 17, 559–574. [Google Scholar] [CrossRef]
- George, J.; Sreekala, M.S.; Thomas, S. A review on interface modification and characterization of natural fiber reinforced plastic composites. Polym. Eng. Sci. 2001, 41, 1471–1485. [Google Scholar] [CrossRef]
- Espino-Pérez, E.; Gilbert, R.G.; Domenek, S.; Brochier-Salon, M.C.; Belgacem, M.N.; Bras, J. Nanocomposites with functionalised polysaccharide nanocrystals through aqueous free radical polymerisation promoted by ozonolysis. Carbohydr. Polym. 2016, 135, 256–266. [Google Scholar] [CrossRef] [PubMed]
- Ifuku, S.; Nogi, M.; Abe, K.; Handa, K.; Nakatsubo, F.; Yano, H. Surface modification of bacterial cellulose nanofibers for property enhancement of optically transparent composites: Dependence on acetyl-group DS. Biomacromolecules 2007, 8, 1973–1978. [Google Scholar] [CrossRef]
- Habibi, Y. Key advances in the chemical modification of nanocelluloses. Chem. Soc. Rev. 2014, 43, 1519–1542. [Google Scholar] [CrossRef]
- Syverud, K.; Xhanari, K.; Chinga-Carrasco, G.; Yu, Y.; Stenius, P. Films made of cellulose nanofibrils: Surface modification by adsorption of a cationic surfactant and characterization by computer-assisted electron microscopy. J. Nanopart. Res. 2011, 13, 773–782. [Google Scholar] [CrossRef]
- Xhanari, K.; Syverud, K.; Chinga-Carrasco, G.; Paso, K.; Stenius, P. Reduction of water wettability of nanofibrillated cellulose by adsorption of cationic surfactants. Cellulose 2011, 18, 257–270. [Google Scholar] [CrossRef]
- Moreirinha, C.; Vilela, C.; Silva, N.H.C.S.; Pinto, R.J.B.; Almeida, A.; Rocha, M.A.M.; Coelho, E.; Coimbra, M.A.; Silvestre, A.J.D.; Freire, C.S.R. Antioxidant and antimicrobial films based on brewers spent grain arabinoxylans, nanocellulose and feruloylated compounds for active packaging. Food Hydrocoll. 2020, 108, 105836. [Google Scholar] [CrossRef]
- Missio, A.L.; Mattos, B.D.; Ferreira, D.F.; Magalhães, W.L.E.; Bertuol, D.A.; Gatto, D.A.; Petutschnigg, A.; Tondi, G. Nanocellulose-tannin films: From trees to sustainable active packaging. J. Clean. Prod. 2018, 184, 143–151. [Google Scholar] [CrossRef]
- El-Wakil, N.A.; Hassan, E.A.; Abou-Zeid, R.E.; Dufresne, A. Development of wheat gluten/nanocellulose/titanium dioxide nanocomposites for active food packaging. Carbohydr. Polym. 2015, 124, 337–346. [Google Scholar] [CrossRef]
- Wang, W.; Yu, Z.; Alsammarraie, F.K.; Kong, F.; Lin, M.; Mustapha, A. Properties and antimicrobial activity of polyvinyl alcohol-modified bacterial nanocellulose packaging films incorporated with silver nanoparticles. Food Hydrocoll. 2020, 100, 105411. [Google Scholar] [CrossRef]
- Padrão, J.; Gonçalves, S.; Silva, J.P.; Sencadas, V.; Lanceros-Mendez, S.; Pinheiro, A.C.; Vicente, A.A.; Rodrigues, L.R.; Dourado, F. Bacterial cellulose-lactoferrin as an antimicrobial edible packaging. Food Hydrocoll. 2016, 58, 126–140. [Google Scholar] [CrossRef]
- Jipa, I.M.; Stoica-Guzun, A.; Stroescu, M. Controlled release of sorbic acid from bacterial cellulose based mono and multilayer antimicrobial films. LWT Food Sci. Technol. 2012, 47, 400–406. [Google Scholar] [CrossRef]
- Kuswandi, B.; Oktaviana, R.; Abdullah, A.; Heng, L.Y. A Novel on-package sticker sensor based on methyl red for real-time monitoring of broiler chicken cut freshness. Packag. Technol. Sci. 2013, 27, 69–81. [Google Scholar] [CrossRef]
- Lu, P.; Yang, Y.; Liu, R.; Liu, X.; Ma, J.; Wu, M.; Wang, S. Preparation of sugarcane bagasse nanocellulose hydrogel as a colourimetric freshness indicator for intelligent food packaging. Carbohydr. Polym. 2020, 249, 116831. [Google Scholar] [CrossRef]
- Bharimalla, A.K.; Patil, P.G.; Mukherjee, S.; Yadav, V.; Prasad, V. Nanocellulose-polymer composites:novel materials for food packaging applications. In Polymers for Agri-Food Applications; Gutiérrez, T.J., Ed.; Springer: Berlin, Germany, 2019. [Google Scholar]
- Oksman, K.; Aitomaki, Y.; Mathew, A.P.; Siqueira, G.; Zhou, Q.; Butylina, S.; Tanpichai, S.; Zhou, X.; Hooshmand, S. Review of the recent developments in cellulose nanocomposite processing. Compos. Part A Appl. Sci. Manuf. 2016, 83, 2–18. [Google Scholar] [CrossRef]
- Alves, J.S.; Reis, K.C.D.; Menezes, E.G.T.; Pereira, F.V.; Pereira, J. Effect of cellulose nanocrystals and gelatin in corn starch plasticized films. Carbohydr. Polym. 2015, 115, 215–222. [Google Scholar] [CrossRef]
- Gong, G.; Mathew, A.P.; Oksman, K. Toughening effect of cellulose nanowhiskers on polyvinyl acetate: Fracture toughness and viscoelastic analysis. Polym. Compos. 2011, 32, 1492–1498. [Google Scholar] [CrossRef]
- Bondeson, D.; Oksman, K. Polylactic acid/cellulose whisker nanocomposites modified by polyvinyl alcohol. Compos. Part A Appl. Sci. Manuf. 2007, 38, 2486–2492. [Google Scholar] [CrossRef]
- De Azeredo, H.M.C.; Rosa, M.F.; Figueiredo, M.C.B. Lignocellulosic-based nanostructures and their use in food packaging. In Nanomaterials Food Packaging; Elsevier: Amsterdam, The Netherlands, 2018; pp. 47–69. [Google Scholar]
- Maloney, T.C.; Paulapuro, H. Effect of drying conditions on the swelling and bonding properties of bleached kraft hardwood pulp. APPITA Annu. Conf. Proc. 2000, 1, 43–46. [Google Scholar]
- Gan, P.G.; Sam, S.T.; Abdullah, M.F.B.; Omar, M.F. Thermal properties of nanocellulose-reinforced composites: A review. J. Appl Polym Sci. 2020, 137, 48544. [Google Scholar] [CrossRef]
- Shah, N.; Ul-Islam, M.; Khattak, W.A.; Park, J.K. Overview of bacterial cellulose composites: A multipurpose advanced material. Carbohydr. Polym. 2013, 98, 1585–1598. [Google Scholar] [CrossRef] [PubMed]
- Pourjavaher, S.; Almasi, H.; Meshkini, S.; Pirsa, S.; Parandi, E. Development of a colorimetric pH indicator based on bacterial cellulose nanofibers and red cabbage (Brassica oleraceae) extract. Carbohydr. Polym. 2017, 156, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Azeredo, H.M.; Rosa, M.F.; Mattoso, L.H.C. Nanocellulose in bio-based food packaging applications. Ind. Crops Prod. 2017, 97, 664–671. [Google Scholar] [CrossRef]
- Decher, G.; Hong, J.-D. Buildup of ultrathin multilayer films by a self-assembly process, 1 consecutive adsorption of anionic and cationic bipolar amphiphiles on charged surfaces. Makromol. Chem. Macromol. Symp. 1991, 46, 321–327. [Google Scholar] [CrossRef]
- Martin, C.; Jean, B. Nanocellulose/polymer multilayered thin films: Tunable architectures towards tailored physical properties. Nord. Pulp Pap. Res. J. 2014, 29, 19–30. [Google Scholar] [CrossRef]
- Li, F.; Mascheroni, E.; Piergiovanni, L. The potential of nanocellulose in the packaging field: A review. Packag. Technol. Sci. 2015, 28, 475–508. [Google Scholar] [CrossRef]
- Hou, Q.; Wang, X.; Ragauskas, A.J. Preparation and characterization of nanocellulose–polyvinyl alcohol multilayer film by layer-by-layer method. Cellulose 2019, 26, 4787–4798. [Google Scholar] [CrossRef]
- Aulin, C.; Karabulut, E.; Tran, A.; Waìšgberg, L.; Lindström, T. Transparent nanocellulosic multilayer thin films on polylactic acid with tunable gas barrier properties. ACS Appl. Mater. Interfaces 2013, 5, 7352–7359. [Google Scholar] [CrossRef]
- Marais, A.; Utsel, S.; Gustafsson, E.; Wågberg, L. Towards a super-strainable paper using the Layer-by-Layer technique. Carbohydr. Polym. 2014, 100, 218–224. [Google Scholar] [CrossRef]
- Anukiruthika, T.; Sethupathy, P.; Wilson, A.; Kashampur, K.; Moses, J.A.; Anandharamakrishnan, C. Multilayer packaging: Advances in preparation techniques and emerging food applications. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1156–1186. [Google Scholar] [CrossRef]
- Moreira, J.B.; De Morais, M.G.; De Morais, E.G.; Vaz, S.; Costa, J.A.V. Electrospun polymeric nanofibers in food packaging. In Impact of Nanoscience in the Food Industry; Grumezescu, A.M., Holban, A.M., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2018; pp. 387–417. [Google Scholar]
- Kai, D.; Liow, S.S.; Loh, X.J. Biodegradable polymers for electrospinning: Towards biomedical applications. Mater. Sci. Eng. C 2014, 45, 659–670. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Duan, G.; Zhang, G.; Yang, H.; Jiang, S.; He, S. Electrospun functional materials toward food packaging applications: A review. Nanomaterials 2020, 10, 150. [Google Scholar] [CrossRef]
- De Dicastillo, C.L.; Garrido, L.; Alvarado, N.; Romero, J.; Palma, J.L.; Galotto, M.J. Improvement of polylactide properties through cellulose nanocrystals embedded in poly(vinyl alcohol) electrospun nanofibers. Nanomaterials 2017, 7, 106. [Google Scholar] [CrossRef]
- Nishino, T.; Matsuda, I.; Hirao, K. Cellulose self-reinforced composite. In Ecocomposites; University of London: London, UK, 2003. [Google Scholar]
- Guzman-Puyol, S.; Ceseracciu, L.; Tedeschi, G.; Marras, S.; Scarpellini, A.; Benítez, J.J.; Athanassiou, A.; Heredia-Guerrero, J.A. Transparent and robust all-cellulose nanocomposite packaging materials prepared in a mixture of trifluoroacetic acid and trifluoroacetic anhydride. Nanomaterials 2019, 9, 368. [Google Scholar] [CrossRef]
- Gindl, W.; Keckes, J. Tensile properties of cellulose acetate butyrate composites reinforced with bacterial cellulose. Compos. Sci. Technol. 2004, 64, 2407–2413. [Google Scholar] [CrossRef]
- Yousefi, H.; Faezipour, M.; Nishino, T.; Shakeri, A.; Ebrahimi, G. All-cellulose composite and nanocomposite made from partially dissolved micro-and nanofibers of canola straw. Polym. J. 2011, 43, 559–564. [Google Scholar] [CrossRef]
- Soykeabkaew, N.; Sian, C.; Gea, S.; Nishino, T.; Peijs, T. All-cellulose nanocomposites by surface selective dissolution of bacterial cellulose. Cellulose 2009, 16, 435–444. [Google Scholar] [CrossRef]
- Fink, H.P.; Weigel, P.; Purz, H.J.; Ganster, J. Structure formation of regenerated cellulose materials from NMMO-solutions. Prog. Polym. Sci. 2001, 26, 1473–1524. [Google Scholar] [CrossRef]
- Poças, F.; Franz, R. Overview on European regulatory issues, legislation, and EFSA evaluations of nanomaterials. In Nanomaterials for Food Packaging; Elsevier: Amsterdam, The Netherlands, 2018; pp. 277–300. [Google Scholar]
- Hardy, A.; Benford, D.; Halldorson, T.; Jeger, M.J.; Knutsen, H.K.; More, S.; Naegeli, H.; Noteborn, H.; Ockleford, C.; Ricci, A.; et al. Guidance on risk assessment of the application of nanoscience and nanotechnologies in the food and feed chain: Part 1, human and animal health. EFSA J. 2018, 16, e05327. [Google Scholar]
- Gómez, C.H.; Serpa, A.; Velásquez-Cock, J.; Ganán, P.; Castro, C.; Vélez, L.; Zuluaga, R. Vegetable nanocellulose in food science: A review. Food Hydrocoll. 2016, 57, 178–186. [Google Scholar] [CrossRef]
- Endes, C.; Camarero-Espinosa, S.; Mueller, S.; Foster, E.J.; Petri-Fink, A.; Rothen-Rutishauser, B.; Weder, C.; Clift, M.J.D. A critical review of the current knowledge regarding the biological impact of nanocellulose. J. Nanobiotechnol. 2016, 14, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Endes, C.; Schmid, O.; Kinnear, C.; Mueller, S.; Camarero-Espinosa, S.; Vanhecke, D.; Foster, E.J.; Petri-Fink, A.; Rutishauser, B.R.; Weder, C.; et al. An in vitro testing strategy towards mimicking the inhalation of high aspect ratio nanoparticles. Part. Fibre Toxicol. 2014, 11, 40. [Google Scholar] [CrossRef] [PubMed]
- Clift, M.J.; Foster, E.J.; Vanhecke, D.; Studer, D.; Wick, P.; Gehr, P.; Rutishauser, B.R.; Weder, C. Investigating the interaction of cellulose nanofibers derived from cotton with a sophisticated 3D human lung cell coculture. Biomacromolecules 2011, 12, 3666–3673. [Google Scholar] [CrossRef] [PubMed]
- Catalán, J.; Ilves, M.; Järventaus, H.; Hannukainen, K.S.; Kontturi, E.; Vanhala, E.; Alenius, H.; Savolainen, K.M.; Norppa, H. Genotoxic and immunotoxic effects of cellulose nanocrystals in vitro. Environ. Mol. Mutagen. 2015, 56, 171–182. [Google Scholar] [CrossRef]
- Farcas, M.T.; Kisin, E.R.; Menas, A.L.; Gutkin, D.W.; Star, A.; Reiner, R.S.; Yanamala, N.; Savolainen, K.; Shvedova, A.A. Pulmonary exposure to cellulose nanocrystals caused deleterious effects to reproductive system in male mice. J. Toxicol. Environ. Health Part A 2016, 79, 984–997. [Google Scholar] [CrossRef]
- Silva-Carvalho, R.; Silva, J.P.; Ferreirinha, P.; Leitão, A.F.; Andrade, F.K.; Da Costa, R.M.G.; Cristelo, C.; Rosa, F.M.; Vilanova, M.; Gama, F.M. Inhalation of bacterial cellulose nanofibrils triggers an inflammatory response and changes lung tissue morphology of mice. Toxicol. Res. 2019, 35, 45–63. [Google Scholar] [CrossRef]
- Lopes, V.R.; Strømme, M.; Ferraz, N. In vitro biological impact of nanocellulose fibers on human gut bacteria and gastrointestinal cells. Nanomaterials 2020, 10, 1159. [Google Scholar] [CrossRef]
- De Lima, A.; Cândido, P.; Fregonezi, N.F.; José, A.; Carvalho, F.; Trovatti, E. TEMPO-Oxidized cellulose nanofibers in vitro cyto-genotoxicity studies. BioNanoScience 2020, 10, 766–772. [Google Scholar] [CrossRef]
- Deloid, G.M.; Cao, X.; Molina, R.M.; Silva, D.I.; Bhattacharya, K.; Ng, K.W.; Loo, S.C.J.; Braina, J.D.; Demokritou, P. Toxicological effects of ingested nanocellulose in: In vitro intestinal epithelium and in vivo rat models. Environ. Sci. Nano 2019, 6, 2105–2115. [Google Scholar] [CrossRef]
- Zhang, H.; Yu, H.Y.; Wang, C.; Yao, J. Effect of silver contents in cellulose nanocrystal/silver nanohybrids on PHBV crystallization and property improvements. Carbohydr. Polym. 2017, 173, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Fortunati, E.; Peltzer, M.; Armentano, I.; Torre, L.; Jiménez, A.; Kenny, J.M. Effects of modified cellulose nanocrystals on the barrier and migration properties of PLA nano-biocomposites. Carbohydr. Polym. 2012, 90, 948–956. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.Y.; Yang, X.Y.; Lu, F.F.; Chen, G.Y.; Yao, J.M. Fabrication of multifunctional cellulose nanocrystals/poly(lactic acid) nanocomposites with silver nanoparticles by spraying method. Carbohydr. Polym. 2016, 140, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Vartiainen, J.; Laine, C.; Willberg-Keyriläinen, P.; Pitkänen, M.; Ohra-aho, T. Biobased mineral-oil barrier-coated food-packaging films. J. Appl. Polym. Sci. 2017, 134, 1–7. [Google Scholar] [CrossRef]
- Weidmann Fiber Technology. Available online: https://weidmannfibertechnology.com/ (accessed on 10 June 2020).
- Sappi. Available online: https://www.sappi.com/pt/nanocellulose (accessed on 18 June 2020).
- Exilva. Available online: https://www.exilva.com/blog/microfibrillated-cellulose-or-nanocellulose?ads_cmpid=1027096933&ads_adid=53266123591&ads_matchtype=b&ads_network=g&ads_creative=355152079425&utm_term=%2Bnanocellulose&ads_targetid=kwd-317912543023&utm_source=adwords&utm_medium (accessed on 18 June 2020).
- FiberLean Technologies. Available online: https://fiberlean.com/nanocellulose-threat-or-opportunity-2/ (accessed on 18 June 2020).
- The FiloCell Advantage. Available online: https://biomaterials.kruger.com/products/the-filocell-advantage/ (accessed on 18 June 2020).
- American Process. Available online: https://www.americanprocess.com/BiorefineryTechnologiesAndSolutions.aspx (accessed on 10 July 2020).
- US Forest Products Lab. Available online: https://www.fpl.fs.fed.us/research/facilities/nanocellulose_pilot-plant.php (accessed on 10 July 2020).
- Fabbri, P.; Viaggi, D.; Cavani, F.; Berlin, L.; Michetti, M.; Carneval, E.; Velasquez, J.O.; Martinez, G.A.; Espoti Degli, M.; Fischer, P.K.; et al. Top Emerging Bio-Based Products, Their Properties and Industrial Applications; Ecologic Institute: Berlin, Germany, 2018. [Google Scholar]
- University of Maine. Available online: https://umaine.edu/pdc/nanocellulose/ (accessed on 10 July 2020).
- Nippon Paper Group. Available online: https://www.nipponpapergroup.com/english/products/cnf/ (accessed on 10 July 2020).
- Oji Holdings. Available online: https://www.ojiholdings.co.jp/english/r_d/theme/cnf.html (accessed on 10 July 2020).
- Innventia. Available online: http://www.paperage.com/issues (accessed on 10 July 2020).
- Cellulose Lab. Available online: https://www.celluloselab.com/ (accessed on 18 June 2020).
- Melodea-Bio Based Solutions. Available online: https://melodea.eu/ (accessed on 10 July 2020).
- Innotech Alberta. Available online: https://innotechalberta.ca/research-facilities/cellulose-nanocrystals-cnc-pilot-plant/ (accessed on 10 July 2020).
- Blue Goose Biorefineries. Available online: https://bluegoosebiorefineries.com/ (accessed on 10 July 2020).
- Celluforce. Available online: https://www.celluforce.com/ (accessed on 18 June 2020).
- BOWIL Biotech. Available online: https://bowil.pl/en/ (accessed on 7 August 2020).
- JeNaCell. Available online: http://www.jenacell.com/de/ (accessed on 7 August 2020).
- HYLOMORPH. Available online: https://www.wysszurich.uzh.ch/ (accessed on 7 August 2020).
- Weyerhaeuser. Available online: https://www.weyerhaeuser.com/ (accessed on 7 August 2020).
- Xylos Corporation. Available online: https://www.sbir.gov/sbirsearch/detail/353438 (accessed on 7 August 2020).
- BioFill. Available online: https://www.dermafill.com/index.php?option=com_content&view=article&id=63:use-of-cellulose-film-biofill-as-biologic-dressing&catid=16:microbial-cellulose-medical&Itemid=27 (accessed on 7 August 2020).
- Klemm, D.; Cranston, E.D.; Fischer, D.; Gama, M.; Kedzior, S.A.; Kralisch, D.; Kramer, F.; Kondo, T.; Lindstrom, T.; Nietzsche, S.; et al. Nanocellulose as a natural source for groundbreaking applications in materials science: Today’s state. Mater. Today 2018, 21, 720–748. [Google Scholar] [CrossRef]
- Assis, C.A.; Houtman, C.; Phillips, R.; Bilek, E.M.T.; Rojas, O.J.; Pal, L.; Peresin, M.S.; Jameel, H.; Gonzalez, R. Conversion economics of forest biomaterials: Risk and financial analysis of CNC manufacturing. Biofuel. Bioprod. Biorefin. 2017, 6, 246–256. [Google Scholar]
- Dourado, F.; Fontão, A.; Leal, M.; Rodrigues, A.C. Process modeling and techno-economic evaluation of an industrial bacterial nanocellulose fermentation process. In Bacterial Nanocellulose; Gama, M., Dourado, F., Bielecki, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 199–214. [Google Scholar]
- Assis, C.A.; Iglesias, M.C.; Bilodeau, M.; Johnson, D.; Phillips, R.; Peresin, M.S.; Bilek, E.M.; Rojas, O.J.; Venditti, R.; Gonzalez, R. Cellulose micro- and nanofi brils (CMNF) manufacturing-financial and risk assessment. Biofuel Bioprod. Biorefin. 2017, 12, 251–264. [Google Scholar] [CrossRef]
- European Bioplastics. Bioplastics: Facts and Figures; European Bioplastics: Berlin, Germany, 2019. [Google Scholar]
- Cellulose Nanomaterials: An End-User Perspective. Available online: http://www.mktintell.com/files/Miller_Webinar_10_24_16.pdf (accessed on 30 August 2020).
- The Business Case for Commercial Production of Bioplastics in Maine; Maine Technology Institute: Brunswick, ME, USA, 2010.
- Koller, M.; Braunegg, G. Advanced approaches to produce polyhydroxyalkanoate (PHA) biopolyesters in a sustainable and economic fashion. EuroBiotech J. 2018, 2, 89–103. [Google Scholar] [CrossRef]
- Kolle, S.; Sell, D. Biopolymers on the Way up. Polyimers Biopolymers 2005. p. 26. Available online: https://www.founding-angels.com/Biopolymers_ECN_10-05.pdf (accessed on 18 June 2020).
- Dufresne, A. Crystalline starch-based nanoparticles. Curr. Opin. Colloid Interface Sci. 2014, 19, 397–408. [Google Scholar] [CrossRef]
- Kareline Natural Fibre Composites. Available online: https://plasthill.fi/en/products (accessed on 18 June 2020).
- Warnes, J. Wood Fibre Reinforced Composites: A Commercial Overview and Scions Woodforce; Scion Forest Products Innovation: Rotorua, New Zealand, 2019. [Google Scholar]
- FASAL. Available online: http://www.fasal.at/production/ (accessed on 18 June 2020).
- FuturaMat. Available online: https://futuramat.com/?lang=en (accessed on 18 June 2020).
- GreenGran. Available online: https://www.greengran.com/ (accessed on 18 June 2020).
- Terraprene_ FKUR. Available online: http://fkur-polymers.com/terraprene/ (accessed on 18 June 2020).
- Futamura. Available online: http://www.futamuragroup.com/en/divisions/cellulose-films/products/ (accessed on 18 June 2020).
- Bio4Pack. Available online: https://www.bio4pack.com/ (accessed on 18 June 2020).
- Cellucomp. Available online: https://www.cellucomp.com/applications (accessed on 30 August 2020).
- Storaenso. Available online: https://www.storaenso.com/ (accessed on 30 August 2020).
- Elopak. Available online: https://www.elopak.com/sustainable-forestry/ (accessed on 30 August 2020).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
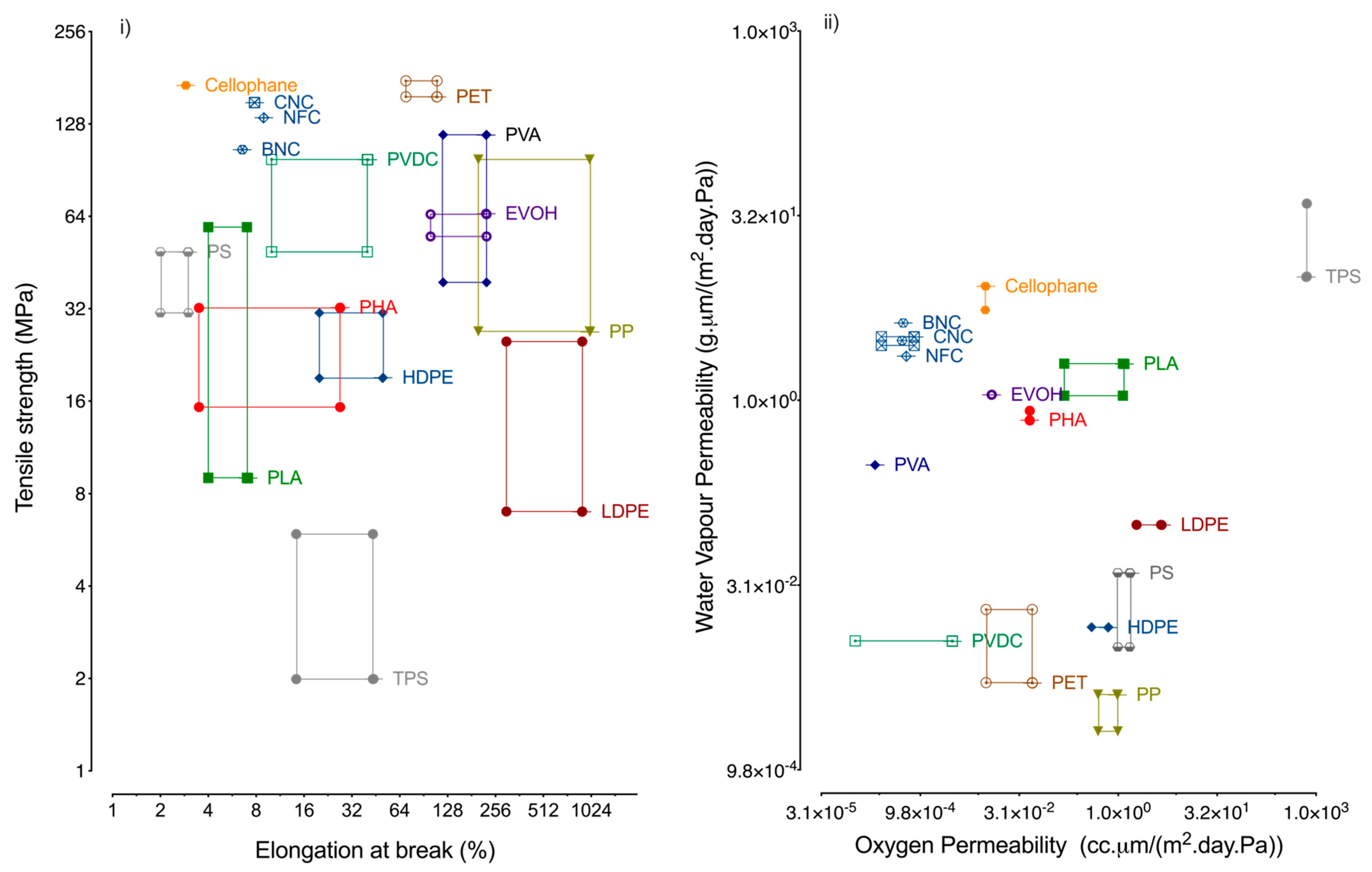
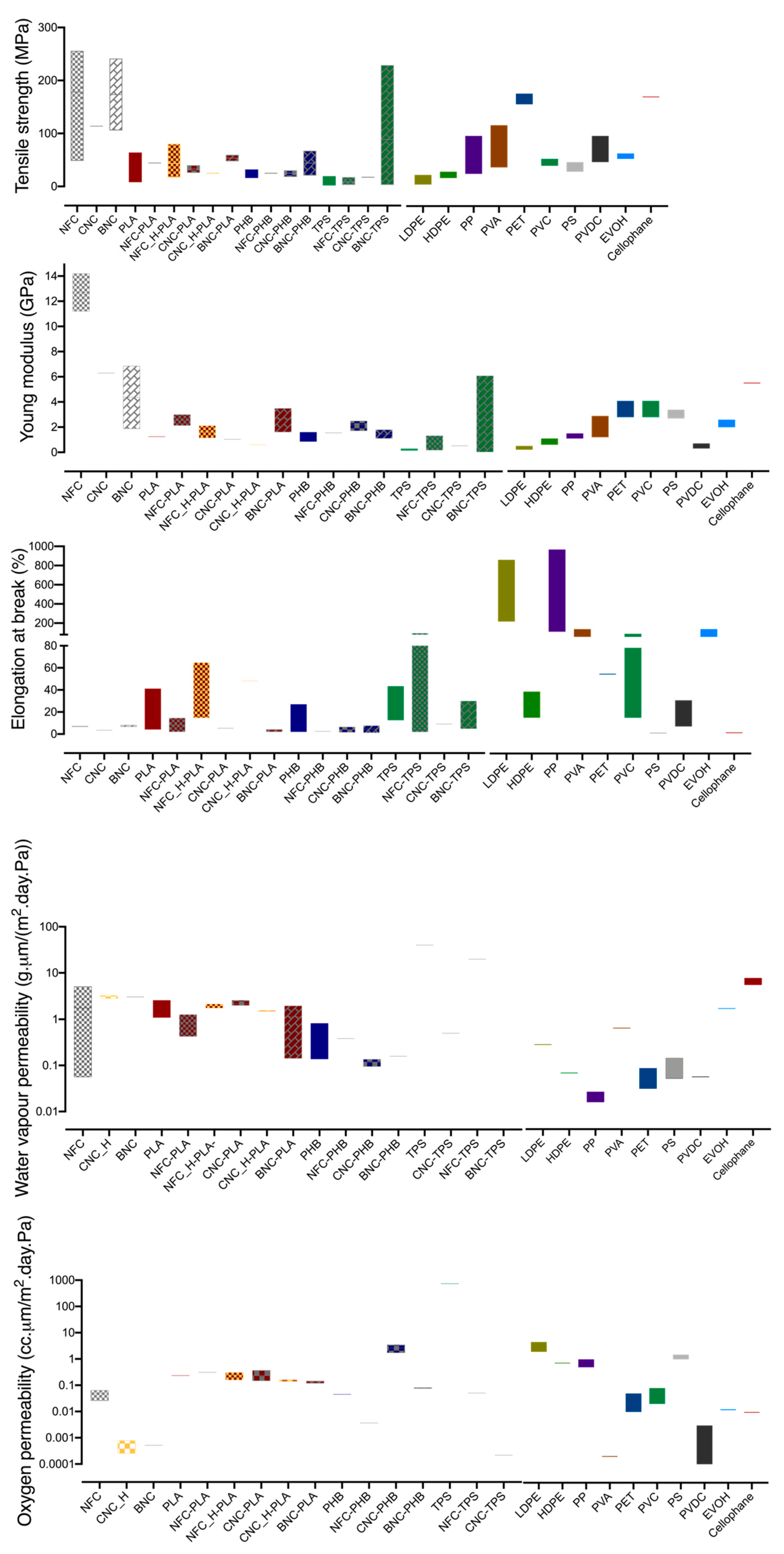
| Product | WVTR (g/m2.day) 23 °C | Oxygen Permeance (cm3STP/m2.day.Pa) 23 °C | Shelf-Life (Months) | Materials Typically Used |
|---|---|---|---|---|
| Low moisture foods, aw < 0.6 | ||||
| Nuts, snacks, chips | 0.093–3.0 | 1.6 × 10−6–9.6 × 10−5 | 3–12 | Metallised films, Laminates with EVOH, PP |
| Coffee | 0.61–1.1 | 8.7 × 10−6–1.3 × 10−5 | 12–18 | PP or PET metallised or AL foil laminates |
| Other dried foods | 0.14–1.7 | 6.8 × 10−7–8.2 × 10−6 | 12–24 | PP or PET metallised, Laminates with EVOH |
| Oils | <30 | 2.6 × 10−5–2.6 × 10−4 | >12 | PET, Glass |
| High moisture foods, aw > 0.9 | ||||
| Beer | 1.4–3.0 | 4.5 × 10−7–2 × 10−6 | 6–12 | Glass, PVDC- coated PET, Metal can |
| Wine | 1.0–1.4 | 1 × 10−6–9.5 × 10−6 | >12 | Glass, PET, Bag in box |
| Fruit juices, soft drinks | 0.47–12.2 | 6.1 × 10−6–6.14 × 10−4 | 1–18 | Glass, PET, Metal cans, bag in box, Aseptic multilayer |
| UHT milk | 2.7–5.3 | 3.5 × 10−6–5.6 × 10−5 | 2.5–5 | Aseptic multilayer |
| Hard cheese | 50 | 8.6 × 10−4–3.45 × 10−3 | 2 | PP, HDPE |
| Fats | 5.2–9.2 | 6.8 × 10−5–8.0 × 10−4 | 3 | Fat resistant paper, PP |
| Retorted food | 0.40–7.6 | 5.9 × 10−6–5.0 × 10−5 | 3–36 | Metal can, Glass jar, Laminates: PET or PP with EVOH or polyamide |
| Fresh foods | ||||
| Fruits, vegetables, fresh salads | 10–4000 | 1 × 10−1–2 | 0.25 | LDPE, PP |
| Meat and meat based products | 2–100 | 2 × 10−4–1 × 10−1 | 0.25–0.5 | PS and PET trays |
| Dairy products | 0.2–8 | 6 × 10−4–5 × 10−2 | 0.5 | HDPE, PP, PS |
| NC Type | Companies | Applications |
|---|---|---|
| CMF | Celova [149]; Sappi [150]; Exilva [151]; FiberLean tecnologies [152]; FiloCell (Kruger company) [153] | Paper; Packaging; coatings; Paints; Cosmetics; Food; Filtration; Environmental remediation; Art Conservation; Adhesives, Agricultural chemicals; HI&I cleaning; Engineered applications; Polymer composites; Cement; Cosmetics; Sealants; |
| NFC | Sappi [147]; American Process [154]; US Forest products Lab [155]; Paperlogic [156]; University of Maine [157]; Nippon Paper [158]; Oji Paper [159], Innventia [160]; Cellulose Lab [161]; FiloCell (Kruger company) [153] | High-tech spun fibres; Antimicrobial films; Water absorbent pads in medical applications; Electronic displays; Food packaging; Flavour carrier; Suspension stabiliser; Thickener in food; Polymer composites; Cement; Paper; Cosmetics; Paints; Coatings; Sealants; Adhesives |
| CNC | American Process [154]; Melodea [162]; Innotech Alberta [163]; US Forest products Lab [155]; Blue Goose Biorefineries [164]; Celluforce [165]; Cellulose Lab [161] | Packaging, Paints; Coatings; Oil and Gas; Adhesives; Paper and non-wovens; Cement; Plastics; Composites; Cosmetics; Health Care; Food and Beverages; Electronics; |
| BNC | Cellulose Lab [161]; Bowil Biotech [166]; JeNaCell GmbH [167]; HYLOMORPH [168]; Weyerhaeuser [169]; Xylos [170]; Biofill [171] | Cosmetics, Biomedical, |
| Company | Products | Applications | Reference | |
|---|---|---|---|---|
| Thermoplastics reinforced with natural fibres | Kareline | Natural fibres and plastics (injection moulding) | Tableware, appliances | [182] |
| Scion | Natural fibre reinforced plastics (mainly PP) (injection moulding) | Automotive sector | [183] | |
| Fasal | Natural fibre reinforcing maize, natural or synthetic resins (injection moulding) | Toys; | [184] | |
| FuturaMat | BioFibra- derived from renewable ressources (biopolymers, wood flour and additives of vegetable origin) PolyFibra- Made from vegetable fibres and partially biobased; | Toys; horticulture; Agriculture; Pieces of equipment; Furnitures; Construction; | [185] | |
| GreenGran BV | PLA and PHB reinforced with natural fibres | Tools | [186] | |
| FkuR | Bio-based TPE with wood fibres | Soft-touch handles, toys, tools or sports equipment | [187] | |
| Cellulose based products | Futamura | Cellophane Natureflex | Food Packaging | [188] |
| Bio4Pack | Paperwise | Food Packaging | [189] | |
| NC based products | Cellucomp | Curran® | Paints, coatings, Food and other packaging | [190] |
| Storaenso | Microfibrilated cellulose based materials | Paper, Food and other packaging, intelligent packaging | [191] | |
| Elopak | Naturally Pure-Pak® | Food packaging | [192] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, F.A.G.S.; Dourado, F.; Gama, M.; Poças, F. Nanocellulose Bio-Based Composites for Food Packaging. Nanomaterials 2020, 10, 2041. https://doi.org/10.3390/nano10102041
Silva FAGS, Dourado F, Gama M, Poças F. Nanocellulose Bio-Based Composites for Food Packaging. Nanomaterials. 2020; 10(10):2041. https://doi.org/10.3390/nano10102041
Chicago/Turabian StyleSilva, Francisco A. G. S., Fernando Dourado, Miguel Gama, and Fátima Poças. 2020. "Nanocellulose Bio-Based Composites for Food Packaging" Nanomaterials 10, no. 10: 2041. https://doi.org/10.3390/nano10102041
APA StyleSilva, F. A. G. S., Dourado, F., Gama, M., & Poças, F. (2020). Nanocellulose Bio-Based Composites for Food Packaging. Nanomaterials, 10(10), 2041. https://doi.org/10.3390/nano10102041








