1. Introduction
Tissues such as nerves, cartilage and bone have a limited capacity for regeneration, especially when the damage is extensive [
1]. In order to address this challenge, tissue engineering (TE) combines scaffolds (i.e., materials), signalling molecules and cells to create constructs that support cell growth, replace damaged biological tissues and guide tissue regeneration [
2].
Electrospun fibres have been widely used as scaffolds for TE, as they are able to partially mimic the structure and spatial topographies of the natural extracellular matrix (ECM) [
3,
4], and therefore improve cell adhesion, proliferation and differentiation [
5], as well as reducing implant rejection [
6]. Moreover, the structures produced by electrospinning yield excellent mechanical properties [
7] and offer unique ones such as a high surface-area-to-volume ration and interfibrous porosity [
5,
6,
7].
One of the main advantages of electrospinning is the simplicity of the device which, coupled with the technique being easily accessible, highly effective, and an efficient method for fibre fabrication [
8], makes it a very attractive technique for scaffold fabrication. There are different ways to perform electrospinning, including needle-based (uses a hollow needle as the spinneret) and needleless (opts for the fabrication of fibres directly from an open liquid surface) [
9]. Typically, an apparatus comprises (1) a high-voltage power supply, (2) a tube with small diameter that is connected to a syringe pump (i.e., a dispenser) and (3) a grounded collector [
5,
10]. Needle-based electrospinning is easy to set up, can use a wide variety of materials to be easily processed, offers tightly controlled flow rate and minimises solution waste [
9].
Furthermore, electrospinning is a very versatile technique that can achieve, among other things, different fibre morphologies and orientation thanks to the range of spinnerets and collectors available [
7]. Spinnerets for needle-based electrospinning include porous hollow tubes, coaxial nozzles and multi-nozzles [
9]. In terms of the collectors available, rotating drums, rings, conveyors, parallel electrodes, and exterior frames [
9], to name a few, make it possible to produce aligned fibres; while patterned collectors are used for more complex geometries [
11].
Moreover, devices can be classified into bench-top and portable. This last category includes hand-held spinnerets, battery-powered, and generator-powered devices [
7]. While benchtop devices are able to produce high quality and reproducible samples, they lack flexibility. This limits their utility in many key applications, such as direct deposition of fibres and the creation of a range of fibre arrangements. Portable electrospinning devices aim to address these challenges. A brief comparison of benchtop and portable apparatus is presented in
Table 1.
Portable electrospinners are particularly useful for direct deposition of fibres onto the target site [
12]. In situ fibre deposition onto wound sites [
7], especially thermal, traumatic and chronic wounds [
13], allows it to be tailored to individual patients. This makes it possible to manage wound sites quickly, promoting healing [
14]. Moreover, the small sized, lightweight and battery/generator-powered devices allow for these to be operated in most locations, including emergency medical transport [
13], hospitals, clinics and patients’ homes, but also remote areas including humanitarian and low-resource settings.
The Oxford Portable Electrospinner (OPE) [
10] is a small, portable device that allows for a more flexible electrospinning (i.e., direction and type of target). Both the voltage (maximum voltage: 14 kV) and the polymer solution flow rate can be adjusted. The handheld apparatus has successfully created fibres from a range of polymers [
14], including poly(vinyl butyral) (PVB), poly ε-caprolactone (PCL), and poly-(lactic-co-glycolic acid) (PLGA). The OPE has not been employed to produce nanoparticle-embedded fibres until now.
Biomedical applications of electrospun fibres have focused, mainly, on fibrous scaffolds for tissue engineering, wound dressing, antibacterial studies, biosensors, enzyme immobilization for faster reaction rates in biological reactions and drug delivery [
4,
5]. However, nanofibers have a high fabrication complexity and moderate biomimicry [
15]. While the first issue is lessened with the use of a simple, portable electrospinning device, biomimicry can be increased with fibre modifications. Relevantly, a highly specialised ECM plays an instructive role in modulating cell behaviour, including the regulation of development, migration, function, and tissue repair [
16]. By providing mechanical support and preferential attachment sites, an electrospun scaffold can guide cell proliferation (i.e., achieving directional growth) [
2]. In brief, electrospun fibres can replicate some aspects of the native tissue and provide the relevant biochemical and physical cues at the appropriate times to create an optimal microenvironment and ensure regeneration.
Electrospun nano and microfibres may be natural, synthetic or hybrid (i.e., a combination of both) [
7]. In this research, we opt for a combination of polycaprolactone and gelatine to create the electrospun fibres. As addressed by Unal [
3], polycaprolactone is a synthetic polymer that offers advantages such as appropriate mechanical strength, biodegradability and non-toxic structural stability. Nevertheless, as a highly hydrophobic material, it is associated with decreased adhesion and reduced cell growth on its surface [
3]. As such, it is necessary to combine it with another material in order to improve its surface chemistry and, especially, mimic certain topographic features of the ECM [
16]. Gelatine, on the other hand, has excellent biocompatibility, is biodegradable, non-immunogenic and is an inexpensive material that may provide an additional 3D architecture for tissue engineering scaffolds [
3]. As such, the combination of polycaprolactone and gelatine results in a bioartificial polymeric material with good biocompatibility, with improved mechanical and physicochemical properties. The biocompatibility of PCL-Ge electrospun scaffolds has been validated in vitro and in vivo in the literature [
2,
17].
Enhancing fibres with additional factors, such as those naturally present in the ECM (e.g., glycosaminoglycans, proteoglycans and glycoproteins), can help create an environment that is more similar to the natural tissue. The enhancement of electrospun fibres with antibiotics, analgesics and other drugs [
9] and using them to create tissue engineering scaffolds is particularly attractive for site-specific delivery. Nevertheless, electrospinning drugs directly with the fibres might result in the loss of biological activity, low encapsulation efficiency, uneven drug distribution, compromised sterile environment, and burst release [
4]. Moreover, the addition of drugs to the polymer solution could affect the polymer’s properties (e.g., viscosity), therefore impacting fibre fabrication [
4].
Therefore, there is a need for methods that allow electrospun fibre functionalisation with, for instance, growth factors, antibiotics, antioxidants and drugs, without the limitations mentioned before. One of such methods encompasses nanoparticles, which can be loaded with several substances and then embedded into electrospun fibres. This strategy allows for a more versatile drug delivery system because both fibres and nanoparticles can be tailored to the specific needs of the tissue or patient. Drug-loaded nanoparticles and microspheres have been widely used for, among others, cancer [
18]. Beyond loading nanoparticles with drugs, electrospun fibres can be functionalised with a variety of substances and biomolecules, yielding the opportunity to enhance performance and achieve additional functions [
9].
Drug-loaded nanoparticle encapsulation, also considered to be a ”smart” drug delivery system [
4], offers target-specific and triggerable drug delivery. NPs are usually embedded into electrospun fibres by direct incorporation during the electrospinning process or as a post-treatment.
In the first case, NPs are either added into the polymer solution and electrospun jointly, or electrosprayed directly onto the fibre surface during electrospinning [
9]. In joint electrospinning, NPs are incorporated directly to the polymer solution before being loaded together into the electrospinning device. Alternatively, NPs can be incorporated before the electrospinning process by using a coaxial nozzle, where each of the two nozzles is loaded with either nanoparticles in a solution (usually, the core) or a polymer (commonly, the shell). This double layer overrides the sudden release associated with drugs electrospun jointly with the polymer solutions thanks to the barrier effect of the sheath structure [
4]. This allows for multicomponent loading of drugs and their controlled released, which derives from the degradation of the shell layer [
4].
On the other hand, post-electrospinning modifications protect bioactive agents from the electrospinning process itself, as well as permitting the addition of factors without the need to alter the electrospinning process or the polymer solutions. Nanoparticles can be generated on the surface of electrospun fibres by indirect fabrication through techniques such as (1) surface deposition, (2) in situ synthesis or (3) hydrothermal treatment [
9,
19]. The first method is the simplest one, and it comprises the immersion of the electrospun fibres into a colloidal suspension of NPs [
20] with the aim of capturing these through hydrogen bonding, chemical binding [
21] or electrostatic force [
19]. Special care must be taken to ensure that the fibres are not soluble in the colloidal suspension containing the nanoparticles. Moreover, it is possible to add multiple layers of charged nanoparticles by opting for a layer-by-layer approach [
19]. Nevertheless, a major drawback of this technique is that even distribution of NPs on the fibre’s surface is difficult to achieve.
In situ synthesis can improve distribution uniformity [
9] and is achieved through a number of processes, including liquid-phase deposition, mainly used for metal oxide nanoparticles [
22]; biomineralization, for calcium phosphate and apatite NPs [
23]; and reduction or annealing of the absorbed precursor, which has been employed for metal and metal oxide nanoparticles [
24]. Third, hydrothermal treatments have been employed to synthesise nanoparticles with different morphologies, determined by the hydrothermal conditions in place, like rods, plates, and spheres [
9].
Direct incorporation of nanoparticles into the polymer solution prior to electrospinning allows for the positioning of NPs within the fibre instead of on the surface only and helps maintain a sterile environment. However, the electrospinning process may have an impact on the nanoparticles or on their release profile [
9,
25]. It is important to bear in mind not only the impact of the electrospinning process itself (e.g., mechanical stress), but also from the exposure to the solvent used in the polymer solution, which might be harmful to the NPs.
Post-electrospinning addition of NPs does not intervene with the electrospinning process and both burst release and short-term release are mitigated [
25]. The two main limitations of this, however, are that achieving uniform distribution of nanoparticles along the fibres is challenging [
9,
25] and that an additional step after electrospinning is required. This could prevent direct fibre deposition onto, for instance, the wound site as enough time to ensure NP attachment to the fibre must be given before being able to place the scaffold in its final location.
For this research, chitosan (CS), a chitin-derived natural polysaccharide, and hyaluronic acid (HA), a non-sulphated glycosaminoglycan, were selected to create the nanoparticles. While hyaluronan is used in a variety of clinical applications [
26] due to its non-immunogenic, mucoadhesive, analgesic and biodegradable properties [
27,
28], chitosan is biocompatible and biodegradable, and has high stability and low immunogenicity [
26,
29]. These have been used to fabricate tissue engineering scaffolds, which promote cell attachment, proliferation and viability [
28]. Relevantly, chitosan can create nanostructures through electrostatic interactions, hydrophobic interactions, hydrogen bonds and van der Waals forces [
29], and hyaluronic acid has the ability to target specific cells by binding with receptors on the cell surface such as CD44 [
30,
31], which makes it suitable for drug delivery targeted at tumours [
29].
HA and CS nanoparticles have been synthesised by a number of methods, including complex coacervation [
26] and ionic gelation [
27,
32,
33]. Briefly, HA, a relatively high-molecular weight weak polyanion, and CS, a lower molecular weight weak polycation, create an asymmetric polyelectrolyte pair [
28,
34]. They bind together due to electrostatic interactions between the free amino groups in chitosan and the carboxyl group in hyaluronan [
28] (
Figure 1).
Ionic gelation (ion-induced gelation) is a simple process that requires the mixing of two aqueous phases at room temperature [
32]. It involves an opposite charge ionic polymer (e.g., sodium triphosphate pentabasic—Na
5O
10P
3) that initiates cross-linking. When dealing with polyanions or polycations, the electroneutrality principle cannot solely be accountable for the cross-linking; thus, additional elements (e.g., presence of other groups, 3D-structure) are considered to impact conjugation functionalities [
32,
33].
Controlled-sized nanostructures, such as HA-CS polyelectrolyte complex nanoparticles, are particularly useful for TE applications [
29]. These nanosystems interact well with cell surfaces, which are negatively charged [
28], and therefore offer prolonged residence time at the target site [
28,
35]. As reported by de la Fuente [
27], systems incorporating HA have been used to modify surfaces with the aim of improving their adhesive properties, and have excellent capacity to associate either hydrophilic or hydrophobic macromolecules [
29].
Nanoparticles offer high encapsulation efficiency and penetration ability, slow degradation rate, small mean size (10–1000 nm) and effective targetability [
36]; characteristics that are particularly useful for biomedical applications. Clinical applications of HA-CS NPs include non-viral vectors for gene delivery [
26], protein or drug delivery [
33], tumour-targeted magnetic resonance imaging (MRI) contrast agents and macromolecule micro/nanocarriers [
27] with controlled release, including heparin [
35], interleukin (IL)-1β [
37], DNA and RNA [
38].
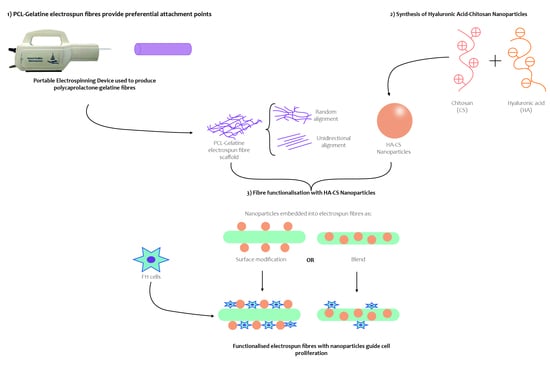
This research aims to develop a biocompatible scaffold to enhance neural cell attachment and viability, as well as favouring directional growth. Furthermore, the main objective of this paper is to demonstrate that hyaluronic acid-chitosan (HA-CS) nanoparticles can be electrospun together with polycaprolactone and gelatine fibres using a portable apparatus to create scaffolds for tissue repair.
4. Discussion
Developing scaffolds for tissue engineering that are both safe and able to guide cell proliferation is important for tissue repair. Electrospinning is a technique that has been employed for scaffold fabrication, particularly due to the benefits derived from the mechanical characteristics of the fibres it produces. Cells may fail to proliferate in a specific direction when a template is not provided, which is particularly common in in vitro testing and poses an additional challenge to successfully mimic the natural tissue. As such, an electrospun scaffold can guide cell proliferation by providing mechanical support and preferential attachment sites, as well as replicating to a certain extent some aspects of the native tissue. Cell proliferation guidance was achieved both in the unmodified fibres and the functionalised ones explored in this research.
A wide range of synthetic polymers, such as poly(vinyl butyral) (PVB), polydioxanone and polycaprolactone, can easily be electrospun into fibres by dissolving them in an organic solvent [
2]. While they exhibit excellent mechanical properties, they lack cell-binding sites [
3]. On the other hand, natural polymers, like gelatine and silk fibroin, have a good cell binding capacity and allow cell signalling and bioactivity [
3], but face rapid or uncontrolled degradation rate and poor mechanical strength [
7]. As such, combining these polymers into a hybrid, such as the case of the polycaprolactone-gelatine polymer explored in this research, seems to yield better results due to advantages like biocompatibility, maintaining differentiated function, provision of cell-binding sites and excellent mechanical properties [
2,
17], which are desirable elements for a tissue engineering scaffold.
In particular, the Oxford Portable Electrospinner has successfully electrospun a number of polymers, including the mixture of polycaprolactone-gelatine explored in this research. Fibres were successfully produced at different weight/volume concentrations (initial PCL or gelatine in solvent 8, 10, 12%) and PCL solution:gelatine solution ratios (85:15, 70:30, 50:50).
The fibre diameters from the resulting fibres suggests that increasing the initial PCL or gelatine in solvent weight/volume concentration (e.g., from 8% to 10% at 85:15 PCL:Ge solutions ratio), results in a higher fibre diameter (increase of 1.672 µm to 2.138 µm). This observation is expected as weight concentration determines the viscosity and surface tension of the solution. Also, this affects the required electric field to form fibres, which alters fibre morphology [
39]. Similarly, authors such as Chui [
14], also report that fibre diameters increase when increasing the weight/volume concentrations.
This research implements a factorial design approach—varying the PCL solution to gelatine solution ratio, as well as the initial PCL/Ge in solvent—and was used to look at cell viability and proliferation. From here, further work is required to determine the effect of these variations and, furthermore, the optimal conditions for cell viability and proliferation considering biocompatibility, biostability and electrospinnability. This could be achieved by, among others, applying design of experiment (DOE) methods, using physical or statistical models [
40]. DOE allows for a better understanding of the effects of different parameters, such as the ratio between PCL and gelatine, on the objective functions, including cell proliferation and viability. As such, it makes it possible to reduce the number of tests required, thus entailing minimal investment of time and resources, and is particularly used to optimise experimental formulations [
40]. Conventional statistical experimental design helps determine the optimal conditions based on measured values of the characteristic properties [
41], while other statistical design models, such as Taguchi, aim to identify optimal conditions by looking at the least variability [
40,
41]. In particular, formulation optimisation has been done using response surface methodology (RSM), where the mechanical and thermal properties of a polymer were optimised from different blend ratios [
42]. Similar strategies could be implemented in further research.
Portable electrospinning devices grant a higher degree of flexibility and, as a result, make it possible to craft fibres in different arrangements as well as in situ fibre deposition onto the target site. Portable devices have a practical use in personalised advanced wound care, particularly when incorporating drug delivery applications, and tissue regeneration. Not only would it be possible to customise the scaffold to a specific site and patient, it could also take advantage of characteristic properties found in the natural tissue and minimise scaffold damage resulting from the process of implantation. Furthermore, multi-layered scaffolds (either of the same or different materials) could be placed directly in the target site. These would allow catering for a single scaffold for different types of tissue or with varying functionalisation depending on the layer.
Particularly, handheld devices have the potential to be used in a wide range of locations, including emergency medical transports, emergency settings and operating rooms. As such, this portability could simplify and accelerate the process of implanting a scaffold in vivo or achieving direct deposition of fibres functionalised with, for instance, nanoparticles loaded with antibacterial and antifungal agents onto wound sites. Moreover, these nanoparticles could be crafted so that they release their load at a later time, thus allowing for timely supply of, for example, antibiotics in burn wounds. Nevertheless, factors such as the duration of the battery, the toxicity of the solvent, and the amount of polymer solution that can be loaded in a cartridge must be taken into account at all times.
The cell viability experiments conducted in this research show that fibres do not provoke adverse effects on cell viability in the short term (up to 72 h); therefore, suggesting that polycaprolactone, gelatine and TFE (used here as a solvent) do not have a toxic effect that could lead to apoptosis. This is expected as other authors have created scaffolds from this combination of materials [
17].
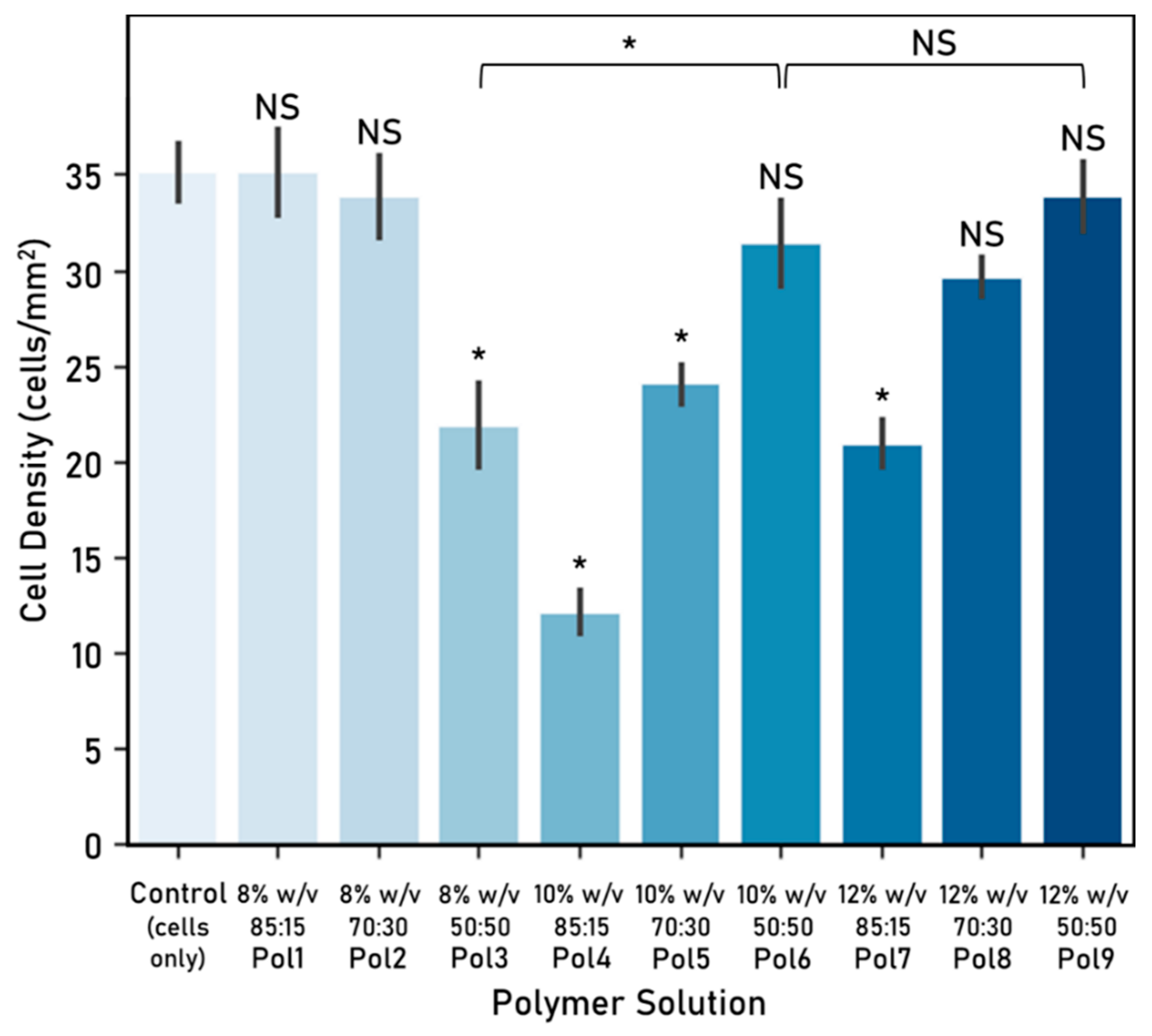
Also, cell viability does not seem to be significantly affected by the fibre arrangement. As such, scaffolds created with unidirectionally aligned or random fibres were associated with similar cell viability (e.g., 8% w/v initial PCL or Ge in solvent, 85:15 PCL:Ge solution ratio). In addition, unidirectional alignment offers the advantage of guiding cell proliferation in a uniaxial manner, which could be particularly interesting for nerve or muscle tissue repair. Although the unidirectional alignment (e.g., 84.26%, polymer: 8% w/v initial PCL or Ge in solvent 70:30 PCL:Ge solution ratio) yields a slightly smaller cell viability than the cells-only control (86.60%), the advantage of incorporating an electrospun scaffold is the capability to direct growth, which is particularly relevant for scaffolds intended to aid in injury repair and regeneration. Notably, when comparing different polymers (8% w/v initial PCL or Ge in solvent 85:15 PCL:Ge solution ratio, 8% w/v 70:30, and 10% w/v 50:50) within the same type of scaffold (unidirectional or random), there were significant differences based on cell viability. This suggests that polymer composition has a stronger influence on cell viability than fibre arrangements.
Some differences in cell viability can be identified when comparing the experimental work on cell–fibre interactions (
Figure 6) with the one on fibre arrangements (
Figure 9). These could potentially be attributed to the time cells were exposed to the electrospun fibres (48 h vs. 72 h).
The functionalisation of electrospun fibres is of particular interest to tissue engineering, as there is a need for nerve repair devices that mimic aspects of the native tissue and provide the appropriate biochemical and physical cues at the appropriate times. Replicating the natural environment more closely by, for instance, incorporating elements naturally present in the ECM can contribute to tissue repair and, if applicable, function restoration. Experimental work on modified fibres (i.e., coatings) with hyaluronic acid, chitosan, and NGF is included in the
Supplementary Materials.
The second part of this research focuses on the functionalisation of fibres with nanoparticles. Hyaluronic acid-chitosan nanoparticles were successfully embedded into polycaprolactone-gelatine electrospun fibres using a portable apparatus to create scaffolds for tissue repair. This was achieved as either a direct blend of the NPs with the polymer solutions (i.e., prior to electrospinning) or a post-electrospinning modification.
While coatings allow fibres to be electrospun first and then modified, possibly making escalation more feasible, blending requires less steps, which can reduce the total scaffold production time. Embedding NPs as a blend enables the distribution of these throughout the electrospun fibres rather than on the surface only, as it occurs with the coating. This allows for a more uniform distribution along the fibres, but it is important to consider the impact of putting the NPs in direct contact with the organic solvent and the mechanical stress that NPs endure while going through the electrospinning process.
Post-electrospinning addition of nanoparticles (i.e., coating) was achieved by immersing the electrospun fibres into a colloidal suspension of HA-CS NPs. Pointedly, nanoparticles bind to the fibre surface by adsorption. Incorporating NPs as a post-electrospinning modification of fibres can prevent bioactive agents from destabilisation and denaturalisation during the electrospinning process. Moreover, the polymer solution’s properties, such as viscosity, are not impacted when adding the NPs.
In this research, we looked at the differences in cell viability and proliferation when comparing fibre functionalisation methods. When exposing these functionalised scaffolds to cells, incorporating NPs into fibres as a blend delivered a higher viability than the unmodified fibres control, while opting for a NP coating yielded higher cell proliferation. It is possible that embedding nanoparticles as a blend is not as successful as attaching the NPs to the surface because the area that is in contact with the cells is greatly reduced.
As both nanoparticle functionalisation options performed similarly to unmodified fibres in terms of cell proliferation, this demonstrates that incorporating NPs into fibres does not alter negatively cell response. Moreover, enhancing electrospun fibres with nanoparticles can allow for a number of clinical applications, including drug delivery. The controlled release of drugs carried within the NPs is not demonstrated in this research.
Nanoparticles are particularly interesting for biomedical applications, mainly because they offer high encapsulation efficiency, are able to penetrate tissues, usually have a slow degradation rate, have a small mean size, and effective targetability [
36]. HA-CS nanoparticles have been used for a number of clinical applications, including protein and drug delivery [
26], contrast agents and macromolecule carriers [
27,
37,
38]. These nanoparticles are particularly useful for tissue engineering applications, as they interact well with cell surfaces and offer prolonged residence time at the target site [
35]. Due to these advantages, this research is interested in incorporating nanoparticles into the tissue engineering scaffolds with the long-term aim of achieving drug deliver.
In particular, HA-CS NPs have the advantage of good biocompatibility, biodegradability, non-toxicity and non-immunogenicity, which make them ideal carriers for the therapeutic drug delivery. Hyaluronic acid is able to target specific cells by binding with CD44, a cell surface receptor [
30,
31]. This is particularly useful for tumour-targeted drug delivery. Similarly, these NPs could be further enhanced by incorporating functional layers that can easily bind to specific receptors.
The hyaluronic acid-chitosan NPs synthesised in this research via ionic gelation have a smooth surface, are spherical, and their diameters are around 200–300 nm. Similar findings, as well as for particle size and particle yield, are reported by a number of authors, namely, Raik [
28], Pornpitchanarong [
33], Zhou [
26] and de la Fuente [
27]. Relevantly, a higher TPP concentration leads to a smaller particle size as a result of a more compacted nanostructure derived from strong electrostatic interactions [
27,
31]. Moreover, particle size in this research is slightly smaller than the mean particle size reported by de la Fuente [
27], on which the protocol used in this research was based on. This difference could potentially be attributed to the addition of a filtration step, which was added to obtain a more homogeneous population of nanoparticles by discarding, for instance, aggregated nanoparticles. This can be corroborated by the small polydispersity value, as small PDI figures are associated with highly monodisperse samples while larger figures obtained when there is a very broad size distribution. PDI values within the 0.1 to 0.4 range were considered acceptable, as they indicate that there is an adequate narrow size distribution of nanoparticles [
43]. The HA-CS nanoparticles synthesised in this research have a PDI that falls within this range, therefore suggesting that there is homogeneity in the particle population. The final composition of the nanoparticles, in terms of percentage of hyaluronic acid and chitosan, is not analysed in this paper.
While the PCL-gelatine electrospun scaffolds embedded with HA-CS nanoparticles are not expected to provoke any significant adverse effects, there are some concerns associated with the safety of nanoparticles, in general, for human health and the environment. It is possible to minimise or significantly reduce the risk of harm throughout the synthesis, functionalisation, use and disposal of nanoparticles by incorporating a safe by design (SbD) approach [
44]. This proactive approach aims to achieve this by integrating early a safety assessment of the materials and their interaction as early as possible in the development process. Strategies such as designing out the hazard in surface functionalisation, reducing release, and standardising production and characterisation methods have been used for this purpose [
44].
The use of low-hazard biomaterials significantly contributes to the SbD approach, as such, opting for biocompatible materials like polycaprolactone, gelatine, hyaluronic acid and chitosan can help to reduce the associated risk. Moreover, conducting safety assessment tests, such as the MTS assay used to evaluate the cytotoxicity of HA-CS nanoparticles on F11 cells, aid in the validation of safe nanoparticles. In here, the toxic dosages were eliminated and, therefore, exposure to a potentially hazardous amount is prevented.
In this research, the nanoparticle synthesis method is based on electrostatic interactions between HA, CS and the cross-linker agent TPP. As it occurs in aqueous media, it avoids organic solvents, high temperatures and shear rates, all of which carry a risk of generating safety concerns. Moreover, ionotropic gelation as a synthesis method for HA-CS nanoparticles has been proven to result in low-toxicity nanoparticles [
27].
UV light was used to sterilise the electrospun fibres before cell seeding. Exposure to UV light is a simple, low-cost and widely used method for fibre sterilisation. Relevantly, it does not influence fibre morphology or alignment, does not cause a critical effect in the physicochemical properties, and allows for cell adhesion and proliferation [
45].
This study has a number of limitations. First of all, the maximum voltage that could be applied was 13 kV, due to the physical limits of the device’s converter. As applied voltage has an impact on fibre morphology [
39], a wider range of voltage could have allowed us to achieve smaller fibre diameters. Another challenge derived from the use of the portable apparatus, which also affects fibre diameter is the solution feed rate [
39]. Fibre production is limited by the amount of polymer that can fit into the syringe cartridge. Thus, long electrospinning sessions or those requiring a high polymer solution feed rate would require frequent cartridge replacements, therefore posing a problem in terms of scalability. These could be addressed by incorporating continuous feed of the polymers into the apparatus.
Moreover, the plates in which the experiments were carried out, do not mimic the natural tissue in both physical and biological terms. While well plates are readily available and allow for specific data collection, key physiological interactions are missing. Finally, as the experimental work covered in this research is based on the short-term (up to 72 h) response of the cells to both the fibres and nanoparticles, longer term performance of the scaffold, including degradation testing, ought to be explored.
Further work on nanoparticle loading, characterisation of the release profile of loaded NPs and an assessment of controlled release of these would allow continued development of these scaffolds for clinical applications. Additional experimental work is required in order to obtain homogeneous distribution of HA-CS nanoparticles along the fibres, which could potentially be achieved by incorporating a coaxial nozzle into the portable apparatus or adapting the cartridge to allow for continuous mixing.
5. Conclusions
A significant challenge to the successful mimicking of the natural tissue derives from cells failing to proliferate in a specific direction when a template is not provided, particularly common in in vitro settings. This research aims to develop a biocompatible scaffold to enhance cell attachment and viability, as well as favouring directional growth. Electrospinning is a remarkably simple and versatile technique that has been widely used for tissue repair and regeneration. With it, it is possible to achieve the desired structure and properties (e.g., porosity, diameter, alignment and biodegradability) by modifying different parameters, thus allowing for widely customisable scaffold fabrication. Electrospinning also makes it possible to generate scaffolds that mimic the hierarchical structure of the ECM, which are critical for cell attachment and proliferation.
In tissue engineering particularly, the relevance of artificial scaffolds that mimic the natural structures and exhibit similar biological properties is key for tissue repair and regeneration. Moreover, the performance of these scaffolds also depends on the cytocompatibility and affinity to the tissue, on top of the durability of the scaffold itself. Therefore, the degradation rate of these polycaprolactone-gelatine scaffolds embedded with hyaluronic acid-chitosan nanoparticles must be explored.
In addition to the topographical cues provided by the electrospun fibres, these can be complemented with electrochemical and biochemical cues thanks to the addition of loaded NPs. Moreover, the release of the substances contained within them could be designed for controlled release, upon the interaction with a specific substance or exposure to stimuli, or even based on the degradation of the NP shell. Not only does this hold a great potential for drug delivery, but also to induce wound healing by releasing factors that promote cell migration to the injured site and the liberation of anti-infection and anti-inflammation by carrying antibiotics or other drugs with antibacterial or antifungal properties, thus promoting effective repair.
Biocompatible scaffolds in different arrangements and from a range of polymer solutions were successfully created with a portable device. Furthermore, fibre modifications and functionalisation were explored as an approach to biomimicry. Relevantly, PCL-gelatine electrospun fibres, including functionalised with nanoparticles, allow cell attachment and promote directional growth.
Finally, the OPE successfully electrospun fibres embedded with nanoparticles as a blend, a previously unexplored variation for this device. This is particularly interesting because portable electrospinning devices are particularly useful for direct fibre deposition onto the damaged site, which allows it to be tailored to individual patients. This could minimise any risks of contamination post electrospinning and avoid adverse effects of sterilisation after fabrication. Moreover, it has potential as a drug delivery agent, as macromolecules encapsulated within the NPs could be released when implanted in the body, at specific time points, or when exposed to external stimuli (e.g., electromagnetic field and environmental cues).