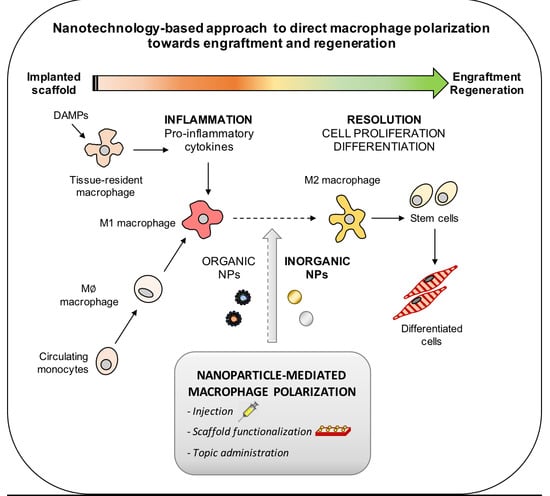
Harnessing Inorganic Nanoparticles to Direct Macrophage Polarization for Skeletal Muscle Regeneration
Abstract
:1. Introduction
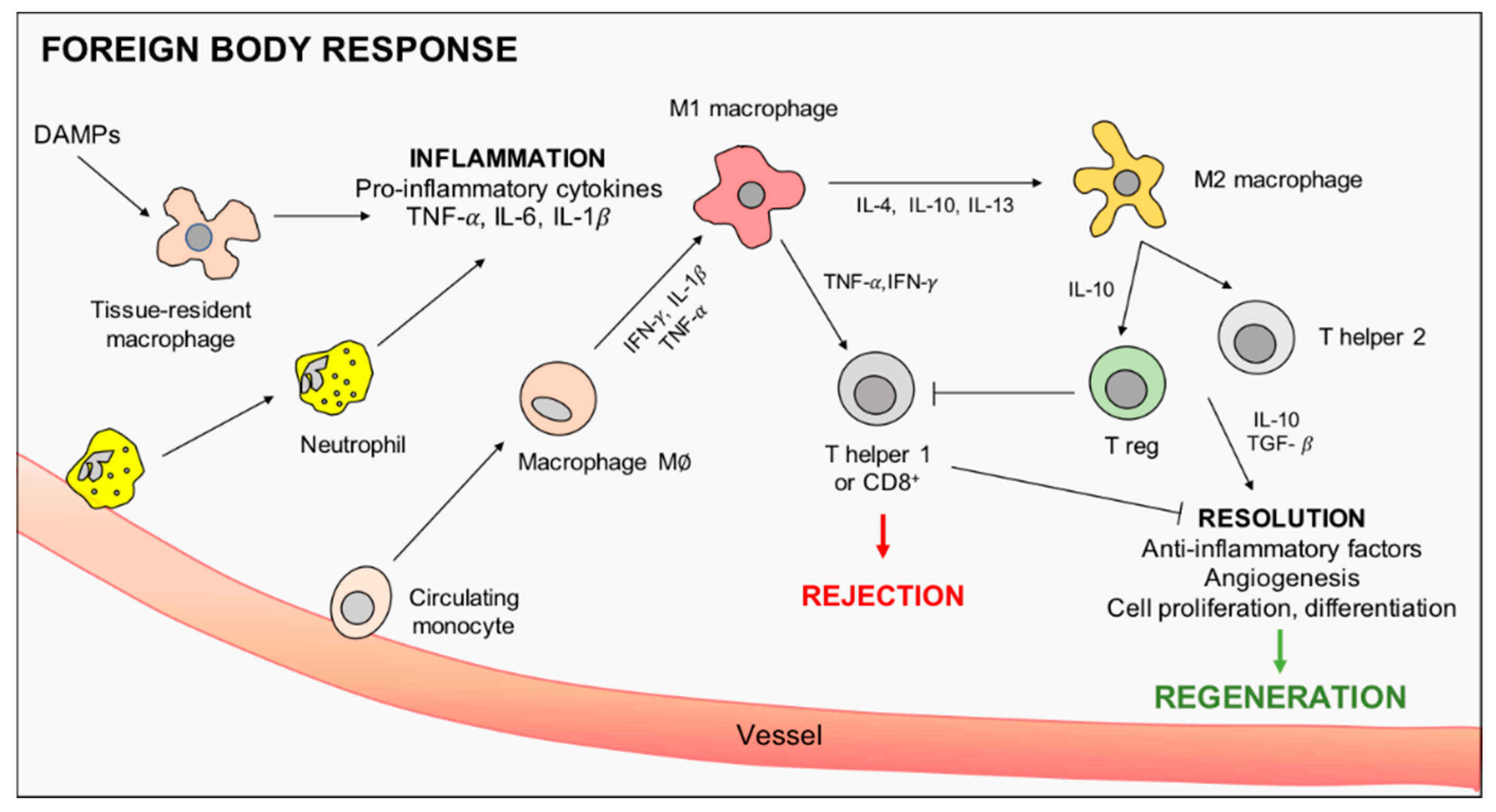
2. The Immune Response to Implanted Materials: The Driving Role of Macrophages
2.1. Macrophages Orchestrate Innate Immunity
2.2. Macrophage Role in Adaptive Immunity
3. Immunomodulation by Implanted Scaffolds
3.1. Native Scaffolds: Decellularized ECM
3.2. Synthetic Scaffolds
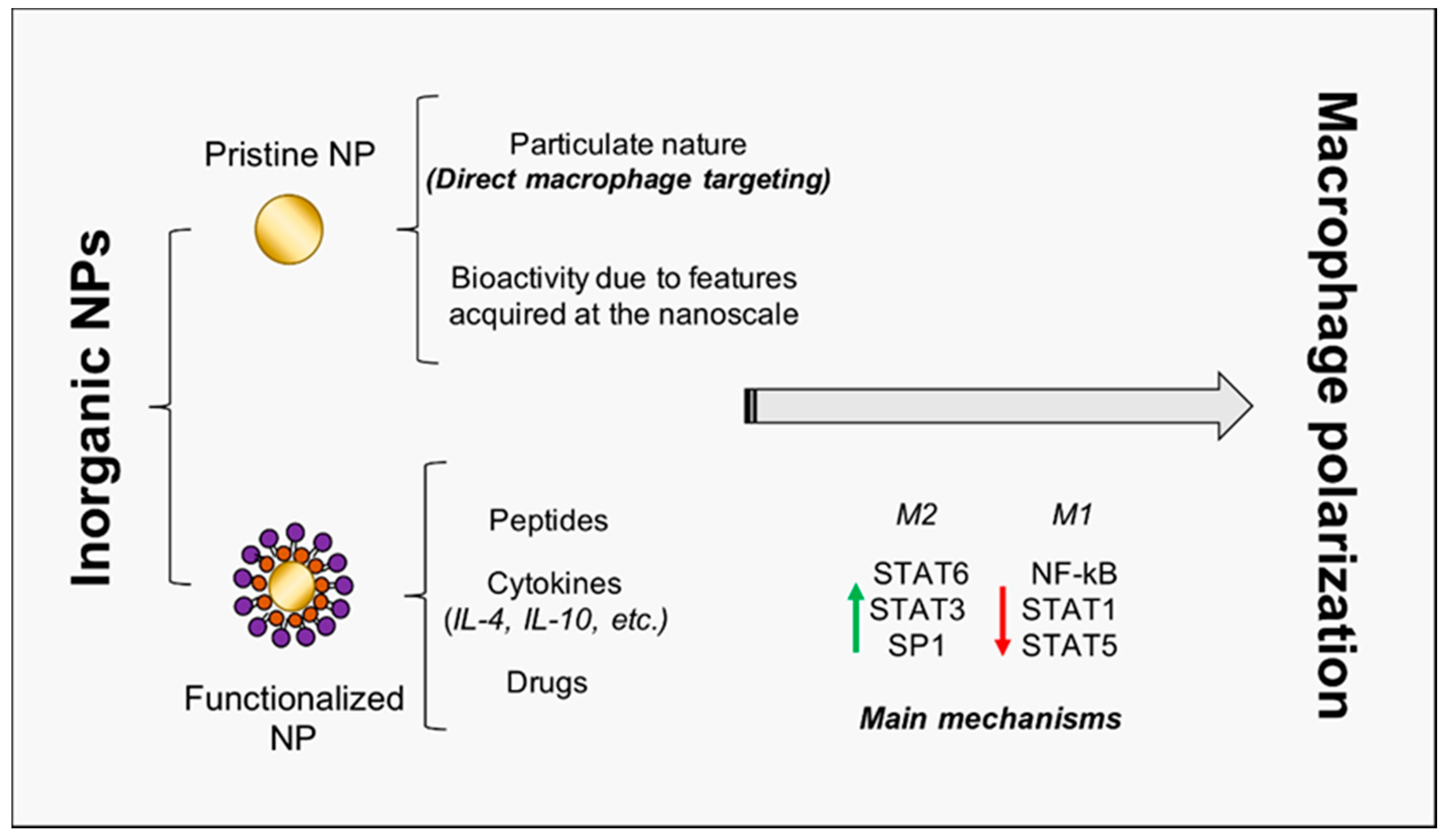
4. Nanoparticles to Direct Macrophage Polarization
4.1. Harnessing Gold Nanoparticles to Trigger Skeletal Muscle Regeneration
4.2. Titanium Oxide NPs: From Osteogenesis to Muscle Regeneration?
4.3. Cerium Oxide NPs: Not Just Antioxidants
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Atala, A. Engineering tissues, organs and cells. J. Tissue Eng. Regen. Med. 2007, 1, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, L.; Lako, M.; Buckley, N.; Lappin, T.; Murphy, M.; Nolta, J.; Pittenger, M.; Stojkovic, M. Editorial: Our top 10 developments in stem cell biology over the last 30 years. Stem Cells 2011, 30, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Watson, D.; Hunziker, R.; Wikswo, J. Fitting tissue chips and microphysiological systems into the grand scheme of medicine, biology, pharmacology, and toxicology. Exp. Biol. Med. 2017, 242, 1559–1572. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crupi, A.; Costa, A.; Tarnok, A.; Melzer, S.; Teodori, L. Inflammation in tissue engineering: The Janus between engraftment and rejection. Eur. J. Immunol. 2015, 45, 3222–3236. [Google Scholar] [CrossRef] [PubMed]
- Carotenuto, F.; Teodori, L.; Maccari, A.; Delbono, L.; Orlando, G.; Di Nardo, P. Turning regenerative technologies into treatment to repair myocardial injuries. J. Cell Mol. Med. 2019, 24, 2704–2716. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koh, T.; DiPietro, L. Inflammation and wound healing: The role of the macrophage. Expert Rev. Mol. Med. 2011, 13, e23. [Google Scholar] [CrossRef] [Green Version]
- Miao, X.; Leng, X.; Zhang, Q. The current state of nanoparticle-induced macrophage polarization and reprogramming research. Int. J. Mol. Sci. 2017, 18, 336. [Google Scholar] [CrossRef] [Green Version]
- Zanganeh, S.; Hutter, G.; Spitler, R.; Lenkov, O.; Mahmoudi, M.; Shaw, A.; Pajarinen, J.; Nejadnik, H.; Goodman, S.; Moseley, M.; et al. Iron oxide nanoparticles inhibit tumour growth by inducing pro-inflammatory macrophage polarization in tumour tissues. Nat. Nanotechnol. 2016, 11, 986–994. [Google Scholar] [CrossRef]
- Jain, S.; Tran, T.; Amiji, M. Macrophage repolarization with targeted alginate nanoparticles containing IL-10 plasmid DNA for the treatment of experimental arthritis. Biomaterials 2015, 61, 162–177. [Google Scholar] [CrossRef] [Green Version]
- Julier, Z.; Park, A.; Briquez, P.; Martino, M. Promoting tissue regeneration by modulating the immune system. Acta Biomater. 2017, 53, 13–28. [Google Scholar] [CrossRef]
- Kasama, T.; Kobayashi, K.; Yajima, N.; Shiozawa, F.; Yoda, Y.; Takeuchi, H.; Mori, Y.; Negishi, M.; Ide, H.; Adachi, M. Expression of vascular endothelial growth factor by synovial fluid neutrophils in rheumatoid arthritis (RA). Clin. Exp. Immunol. 2000, 121, 533–538. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Yang, Y.; Yang, Z.; Li, T.; Chen, F. Snapshot: Targeting macrophages as a candidate for tissue regeneration. Curr. Issues Mol. Biol 2018, 29, 37–48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martinez, F.O.; Sica, A.; Mantovani, A.; Locati, M. Macrophage activation and polarization. Front. Biosci. 2008, 13, 453–461. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martinez, F.O.; Gordon, S. The M1 and M2 paradigm of macrophage activation: Time for reassessment. F1000Prime Rep. 2014, 6, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xue, J.; Schmidt, S.V.; Sander, J.; Draffehn, A.; Krebs, W.; Quester, I.; Dominic De Nardo, D.; Gohel, T.D.; Emde, M.; Schmidleithner, L.; et al. Transcriptome-based network analysis reveals a spectrum model of human macrophage activation. Immunity 2014, 40, 274–288. [Google Scholar] [CrossRef] [Green Version]
- Chazaud, B.; Sonnet, C.; Lafuste, P.; Bassez, G.; Rimaniol, A.; Poron, F.; Authier, F.; Dreyfus, P.; Gherardi, R. Satellite cells attract monocytes and use macrophages as a support to escape apoptosis and enhance muscle growth. J. Cell Biol. 2003, 163, 1133–1143. [Google Scholar] [CrossRef] [Green Version]
- Teodori, L.; Costa, A.; Campanella, L.; Albertini, M. Skeletal muscle atrophy in simulated microgravity might be triggered by immune-related microRNAs. Front. Physiol. 2019, 9, 1926. [Google Scholar] [CrossRef]
- Carotenuto, F.; Albertini, M.; Coletti, D.; Vilmercati, A.; Campanella, L.; Darzynkiewicz, Z.; Teodori, L. How diet intervention via modulation of DNA damage response through microRNAs may have an effect on cancer prevention and aging, an in silico study. Int. J. Mol. Sci. 2016, 17, 752. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Jiang, T.; Li, M.; Zheng, X.; Zhao, G. Transcriptional regulation of macrophages polarization by microRNAs. Front. Immunol. 2018, 9, 1175. [Google Scholar] [CrossRef]
- Reinke, S.; Geissler, S.; Taylor, W.; Schmidt-Bleek, K.; Juelke, K.; Schwachmeyer, V.; Dahne, M.; Hartwig, T.; Akyuz, L.; Meisel, C.; et al. Terminally differentiated CD8+ T cells negatively affect bone regeneration in humans. Sci. Transl. Med. 2013, 5, 177ra36. [Google Scholar] [CrossRef]
- Lei, H.; Schmidt-Bleek, K.; Dienelt, A.; Reinke, P.; Volk, H. Regulatory T cell-mediated anti-inflammatory effects promote successful tissue repair in both indirect and direct manners. Front. Pharmacol. 2015, 6, 184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meng, X.; Yang, J.; Dong, M.; Zhang, K.; Tu, E.; Gao, Q.; Chen, W.; Zhang, C.; Zhang, Y. Regulatory T cells in cardiovascular diseases. Nat. Rev. Cardiol. 2015, 13, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Burzyn, D.; Kuswanto, W.; Kolodin, D.; Shadrach, J.; Cerletti, M.; Jang, Y.; Sefik, E.; Tan, T.; Wagers, A.; Benoist, C.; et al. A special population of regulatory T cells potentiates muscle repair. Cell 2013, 155, 1282–1295. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wynn, T.; Vannella, K. Macrophages in tissue repair, regeneration, and fibrosis. Immunity 2016, 44, 450–462. [Google Scholar] [CrossRef] [Green Version]
- Smith, T.; Nagalla, R.; Chen, E.; Liu, W. Harnessing macrophage plasticity for tissue regeneration. Adv. Drug Deliv. Rev. 2017, 114, 193–205. [Google Scholar] [CrossRef]
- Huleihel, L.; Bartolacci, J.; Dziki, J.; Vorobyov, T.; Arnold, B.; Scarritt, M.; Pineda Molina, C.; LoPresti, S.; Brown, B.; Naranjo, J.; et al. Matrix-bound nanovesicles recapitulate extracellular matrix effects on macrophage phenotype. Tissue Eng. Part A 2017, 23, 1283–1294. [Google Scholar] [CrossRef]
- Huleihel, L.; Dziki, J.; Bartolacci, J.; Rausch, T.; Scarritt, M.; Cramer, M.; Vorobyov, T.; LoPresti, S.; Swineheart, I.; White, L.; et al. Macrophage phenotype in response to ECM bioscaffolds. Semin. Immunol. 2017, 29, 2–13. [Google Scholar] [CrossRef]
- Fishman, J.; Lowdell, M.; Urbani, L.; Ansari, T.; Burns, A.; Turmaine, M.; North, J.; Sibbons, P.; Seifalian, A.; Wood, K.; et al. Immunomodulatory effect of a decellularized skeletal muscle scaffold in a discordant xenotransplantation model. Proc. Natl. Acad. Sci. USA 2013, 110, 14360–14365. [Google Scholar] [CrossRef] [Green Version]
- Badylak, S.; Gilbert, T. Immune response to biologic scaffold materials. Semin. Immunol. 2008, 20, 109–116. [Google Scholar] [CrossRef] [Green Version]
- Wu, R.; He, X.; Zhu, J.; Yin, Y.; Li, X.; Liu, X.; Chen, F. Modulating macrophage responses to promote tissue regeneration by changing the formulation of bone extracellular matrix from filler particles to gel bioscaffolds. Mater. Sci. Eng. C 2019, 101, 330–340. [Google Scholar] [CrossRef]
- Ciocci, M.; Mochi, F.; Carotenuto, F.; Di Giovanni, E.; Prosposito, P.; Francini, R.; De Matteis, F.; Reshetov, I.; Casalboni, M.; Melino, S.; et al. Scaffold-in-scaffold potential to induce growth and differentiation of cardiac progenitor cells. Stem Cells Dev. 2017, 26, 1438–1447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mehrban, N.; Pineda Molina, C.; Quijano, L.; Bowen, J.; Johnson, S.; Bartolacci, J.; Chang, J.; Scott, D.; Woolfson, D.; Birchall, M.; et al. Host macrophage response to injectable hydrogels derived from ECM and α-helical peptides. Acta Biomater. 2020, 111, 141–152. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.; Santos, S.; Oliveira, M.; Torres, A.; Barbosa, M. Chitosan drives anti-inflammatory macrophage polarisation and pro-inflammatory dendritic cell stimulation. Eur. Cells Mater. 2012, 24, 136–153. [Google Scholar] [CrossRef]
- Blakney, A.; Swartzlander, M.; Bryant, S. The effects of substrate stiffness on the in vitro activation of macrophages and in vivo host response to poly(ethylene glycol)-based hydrogels. J. Biomed. Mater. Res. A 2012, 100, 1375–1386. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tylek, T.; Blum, C.; Hrynevich, A.; Schlegelmilch, K.; Schilling, T.; Dalton, P.; Groll, J. Precisely defined fiber scaffolds with 40 μm porosity induce elongation driven M2-like polarization of human macrophages. Biofabrication 2020, 12, 025007. [Google Scholar] [CrossRef] [PubMed]
- Karazisis, D.; Petronis, S.; Agheli, H.; Emanuelsson, L.; Norlindh, B.; Johansson, A.; Rasmusson, L.; Thomsen, P.; Omar, O. The influence of controlled surface nanotopography on the early biological events of osseointegration. Acta Biomater. 2017, 53, 559–571. [Google Scholar] [CrossRef]
- Luu, T.U.; Gott, S.C.; Woo, B.W.; Rao, M.P.; Liu, W.F. Micro-and nanopatterned topographical cues for regulating macrophage cell shape and phenotype. ACS Appl. Mater. Interfaces 2015, 7, 28665–28672. [Google Scholar] [CrossRef] [Green Version]
- Sridharan, R.; Cavanagh, B.; Cameron, A.; Kelly, D.; O’Brien, F. Material stiffness influences the polarization state, function and migration mode of macrophages. Acta Biomater. 2019, 89, 47–59. [Google Scholar] [CrossRef]
- Sridharan, R.; Ryan, E.; Kearney, C.; Kelly, D.; O’Brien, F. Macrophage polarization in response to collagen scaffold stiffness is dependent on cross-linking agent used to modulate the stiffness. ACS Biomater. Sci. Eng. 2018, 5, 544–552. [Google Scholar] [CrossRef]
- Jia, Z.; Cheng, Y.; Jiang, X.; Zhang, C.; Wang, G.; Xu, J.; Li, Y.; Peng, Q.; Gao, Y. 3D culture system for liver tissue mimicking hepatic plates for improvement of human hepatocyte (C3A) function and polarity. BioMed Res. Int. 2020, 2020, 6354183. [Google Scholar] [CrossRef] [Green Version]
- Corsi, F.; Caputo, F.; Traversa, E.; Ghibelli, L. Not only redox: The multifaceted activity of cerium oxide nanoparticles in cancer prevention and therapy. Front. Oncol. 2018, 8, 309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hasan, A.; Morshed, M.; Memic, A.; Hassan, S.; Webster, T.; Marei, H. Nanoparticles in tissue engineering: Applications, challenges and prospects. Int. J. Nanomed. 2018, 13, 5637–5655. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, J.; Lee, S.; Christ, G.; Atala, A.; Yoo, J. The influence of electrospun aligned poly(ε-caprolactone)/collagen nanofiber meshes on the formation of self-aligned skeletal muscle myotubes. Biomaterials 2008, 29, 2899–2906. [Google Scholar] [CrossRef] [PubMed]
- Mehanna, M.M.; Mohyeldin, S.M.; Elgindy, N.A. Respirable nanocarriers as a promising strategy for antitubercular drug delivery. J. Control. Release 2014, 187, 183–197. [Google Scholar] [CrossRef] [PubMed]
- Caso, M.; Carotenuto, F.; Di Nardo, P.; Migliore, A.; Aguilera, A.; Lopez, C.; Venanzi, M.; Cavalieri, F.; Rinaldi, A. Nanoporous microsponge particles (NMP) of polysaccharides as universal carriers for biomolecules delivery. Nanomaterials 2020, 10, 1075. [Google Scholar] [CrossRef] [PubMed]
- Firouzi-Amandi, A.; Dadashpour, M.; Nouri, M.; Zarghami, N.; Serati-Nouri, H.; Jafari-Gharabaghlou, D.; Karzar, B.; Mellatyar, H.; Aghebati-Maleki, L.; Babaloo, Z.; et al. Chrysin-nanoencapsulated PLGA-PEG for macrophage repolarization: Possible application in tissue regeneration. Biomed. Pharmacother. 2018, 105, 773–780. [Google Scholar] [CrossRef] [PubMed]
- Bartneck, M.; Ritz, T.; Keul, H.; Wambach, M.; Bornemann, J.; Gbureck, U.; Ehling, J.; Lammers, T.; Heymann, F.; Gassler, N.; et al. Peptide-functionalized gold nanorods increase liver injury in hepatitis. ACS Nano 2012, 6, 8767–8777. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, H.; Sun, L.; Gao, W.; Xiong, Y.; Ma, A.; Liu, X.; Shen, L.; Li, Q.; Yang, H. Manipulation of macrophage polarization by peptide-coated gold nanoparticles and its protective effects on acute lung injury. J. Nanobiotechnol. 2020, 18, 38. [Google Scholar] [CrossRef] [Green Version]
- Raimondo, T.; Mooney, D. Functional muscle recovery with nanoparticle-directed M2 macrophage polarization in mice. Proc. Natl. Acad. Sci. USA 2018, 115, 10648–10653. [Google Scholar] [CrossRef] [Green Version]
- Shen, X.; Yu, Y.; Ma, P.; Luo, Z.; Hu, Y.; Li, M.; He, Y.; Zhang, Y.; Peng, Z.; Song, G.; et al. Titania nanotubes promote osteogenesis via mediating crosstalk between macrophages and MSCs under oxidative stress. Colloid Surf. B 2019, 180, 39–48. [Google Scholar] [CrossRef]
- Lee, C.; Kim, Y.; Jang, J.; Park, J. Modulating macrophage polarization with divalent cations in nanostructured titanium implant surfaces. Nanotechnology 2016, 27, 085101. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.C.; Dong, X.; Ding, J.L.; Liu, J.C.; Xu, J.J.; Tang, Y.H.; Yi, Y.P.; Lu, C.; Yang, W.; Yang, J.S.; et al. Nanotubular TiO2 regulates macrophage M2 polarization and increases macrophage secretion of VEGF to accelerate endothelialization via the ERK1/2 and PI3K/AKT pathways. Int J. Nanomed. 2019, 14, 441–455. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, K.; Shen, Q.; Xie, Y.; You, M.; Huang, L.; Zheng, X. Incorporation of cerium oxide into hydroxyapatite coating regulates osteogenic activity of mesenchymal stem cell and macrophage polarization. J. Biomater. Appl. 2016, 31, 1062–1076. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wen, J.; Li, B.; Li, W.; Qiao, W.; Shen, J.; Jin, W.; Jiang, X.; Yeung, K.; Chu, P. Valence state manipulation of cerium oxide nanoparticles on a titanium surface for modulating cell fate and bone formation. Adv. Sci. 2017, 5, 1700678. [Google Scholar] [CrossRef]
- Coletti, D.; Scaramuzzo, F.; Montemiglio, L.; Pristerà, A.; Teodori, L.; Adamo, S.; Barteri, M. Culture of skeletal muscle cells in unprecedented proximity to a gold surface. J. Biomed. Mater. Res. A 2009, 91, 370–377. [Google Scholar] [CrossRef]
- Ko, W.; Kim, S.; Heo, D.; Han, I.; Kim, S.; Kwon, I.; Sohn, S. Double layers of gold nanoparticles immobilized titanium implants improve the osseointegration in rabbit models. Nanomed. Nanotechnol. Biol. Med. 2020, 24, 102129. [Google Scholar] [CrossRef]
- Ni, C.; Zhou, J.; Kong, N.; Bian, T.; Zhang, Y.; Huang, X.; Xiao, Y.; Yang, W.; Yan, F. Gold nanoparticles modulate the crosstalk between macrophages and periodontal ligament cells for periodontitis treatment. Biomaterials 2019, 206, 115–132. [Google Scholar] [CrossRef]
- Liu, M.; Li, N.; He, Y.; Ge, Y.; Song, G. Dually emitting gold-silver nanoclusters as viable ratiometric fluorescent probes for cysteine and arginine. Microchim. Acta 2018, 185, 147. [Google Scholar] [CrossRef]
- Ishizaki, K.; Sugita, Y.; Iwasa, F.; Minamikawa, H.; Ueno, T.; Yamada, M.; Suzuki, T.; Ogawa, T. Nanometer-thin TiO2 enhances skeletal muscle cell phenotype and behavior. Int. J. Nanomed. 2011, 2011, 2191–2203. [Google Scholar]
- Lucarelli, M.; Gatti, A.M.; Savarino, G.; Quattroni, P.; Martinelli, L.; Monari, E.; Boraschi, D. Innate defence functions of macrophages can be biased by nano-sized ceramic and metallic particles. Eur. Cytokine Netw. 2004, 15, 339–346. [Google Scholar]
- Caputo, F.; Giovanetti, A.; Corsi, F.; Maresca, V.; Briganti, S.; Licoccia, S.; Traversa, E.; Ghibelli, L. Cerium oxide nanoparticles re-establish cell integrity checkpoints and apoptosis competence in irradiated HaCaT cells via novel redox-independent activity. Front. Pharmacol. 2018, 9, 1183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chigurupati, S.; Mughal, M.; Okun, E.; Das, S.; Kumar, A.; McCaffery, M.; Seal, S.; Mattson, M. Effects of cerium oxide nanoparticles on the growth of keratinocytes, fibroblasts and vascular endothelial cells in cutaneous wound healing. Biomaterials 2013, 34, 2194–2201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siposova, K.; Huntosova, V.; Shlapa, Y.; Lenkavska, L.; Macajova, M.; Belous, A.; Musatov, A. Advances in the study of cerium oxide nanoparticles: New insights into antiamyloidogenic activity. ACS Appl. Bio Mater. 2019, 2, 1884–1896. [Google Scholar] [CrossRef]
- Walczynska, M.; Jakubowski, W.; Wasiak, T.; Kadziola, K.; Bartoszek, N.; Kotarba, S.; Siatkowska, M.; Komorowski, P.; Walkowiak, B. Toxicity of silver nanoparticles, multiwalled carbon nanotubes, and dendrimers assessed with multicellular organism Caenorhabditis elegans. Toxicol. Mech. Methods 2018, 28, 432–439. [Google Scholar] [CrossRef] [PubMed]
| NPs | Shape | Size (nm) | Surface Modification | Initial Phenotype | Polarized Phenotype | Model | Used Markers | Clinical Aim | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Au | rods | ND | PEGylation + RGD | M∅ | M2 | Mouse | Arg-1, IL-4, TNF-α, Retnla | Acute hepatitis | [47] |
| spheres | 13 | hexapeptides | M2 | M2 | Mouse BMDMs | IL-12, IL-6, IL-10, iNOS, Arg-1, YM1 | Acute lung injury | [48] | |
| M1 | M2 | ||||||||
| M2 | ALI Mouse model | CD80, CD206 | |||||||
| spheres | 30 | PEGylation + IL-4 | M∅ | M2 | Human THP-1 cell line | CD206, CD163, IL-4 | Muscle recovery | [49] | |
| 30 | PEGylation + IL-4 | M∅ | M2 | Mouse | CD206, CD80 | ||||
| 100 | None | M∅ | M1 | Human THP-1 cell line | CD206, CD163, IL-4 | ||||
| TiO2 | tubes | 110 | None | M∅ | M1 | Mouse RAW 264.7 cell line | CCR7, IL-6, IL-10, VEGF, BMP2, TGF-β1 | Osteogenesis | [50] |
| M∅ | M2 | Mouse RAW 264.7 cell line + MSCs | |||||||
| disks | 10 * | Ca2+, Sr2+ | M∅ | M2 | Mouse RAW 264.7 cell line | Arg-1, CD163, CD86, iNOS | Osteogenesis | [51] | |
| tubes | 65 92 142 | None | M∅ | M2 | Human THP-1 cell line | Arg-1, CD206, IL-10, VEGF | Heart valve replacement | [52] | |
| CeO2 | thin layer | ND | None | M∅ | M2 | Mouse RAW 264.7 cell line | CD163, CD206, IL-6, TNF-α, IL-10, TGF-β1 | Osteogenesis | [53] |
| thin layer | ND | None | M∅ | M2 | Mouse RAW 264.7 cell line + BMSCs | CCR7, CD206, TNF-α, IL-10 | Osteogenesis | [54] | |
| ND | None | Rat |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corsi, F.; Carotenuto, F.; Di Nardo, P.; Teodori, L. Harnessing Inorganic Nanoparticles to Direct Macrophage Polarization for Skeletal Muscle Regeneration. Nanomaterials 2020, 10, 1963. https://doi.org/10.3390/nano10101963
Corsi F, Carotenuto F, Di Nardo P, Teodori L. Harnessing Inorganic Nanoparticles to Direct Macrophage Polarization for Skeletal Muscle Regeneration. Nanomaterials. 2020; 10(10):1963. https://doi.org/10.3390/nano10101963
Chicago/Turabian StyleCorsi, Francesca, Felicia Carotenuto, Paolo Di Nardo, and Laura Teodori. 2020. "Harnessing Inorganic Nanoparticles to Direct Macrophage Polarization for Skeletal Muscle Regeneration" Nanomaterials 10, no. 10: 1963. https://doi.org/10.3390/nano10101963
APA StyleCorsi, F., Carotenuto, F., Di Nardo, P., & Teodori, L. (2020). Harnessing Inorganic Nanoparticles to Direct Macrophage Polarization for Skeletal Muscle Regeneration. Nanomaterials, 10(10), 1963. https://doi.org/10.3390/nano10101963