RETRACTED: Transcatheter Decellularized Tissue-Engineered Heart Valve (dTEHV) Grown on Polyglycolic Acid (PGA) Scaffold Coated with P4HB Shows Improved Functionality over 52 Weeks due to Polyether-Ether-Ketone (PEEK) Insert
Abstract
:1. Introduction
2. Results
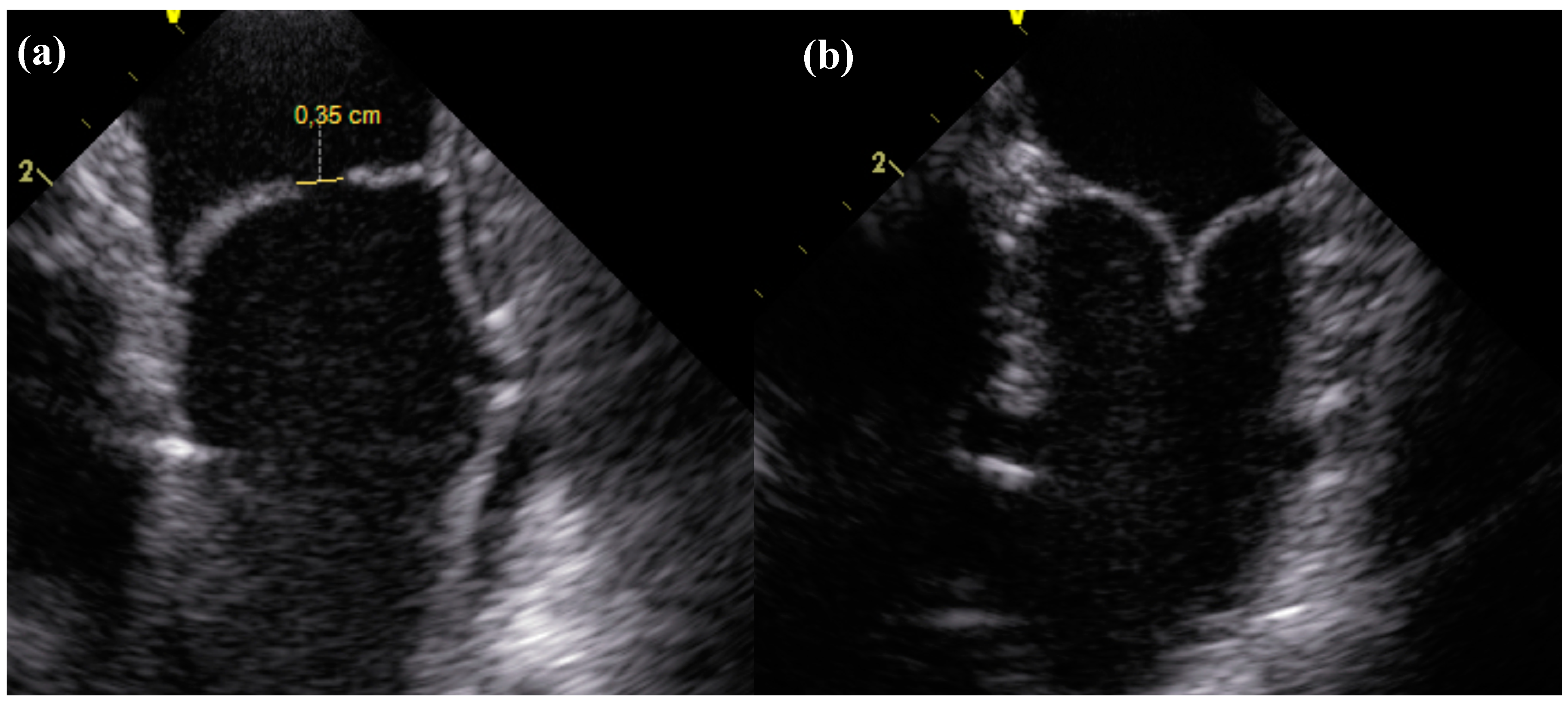
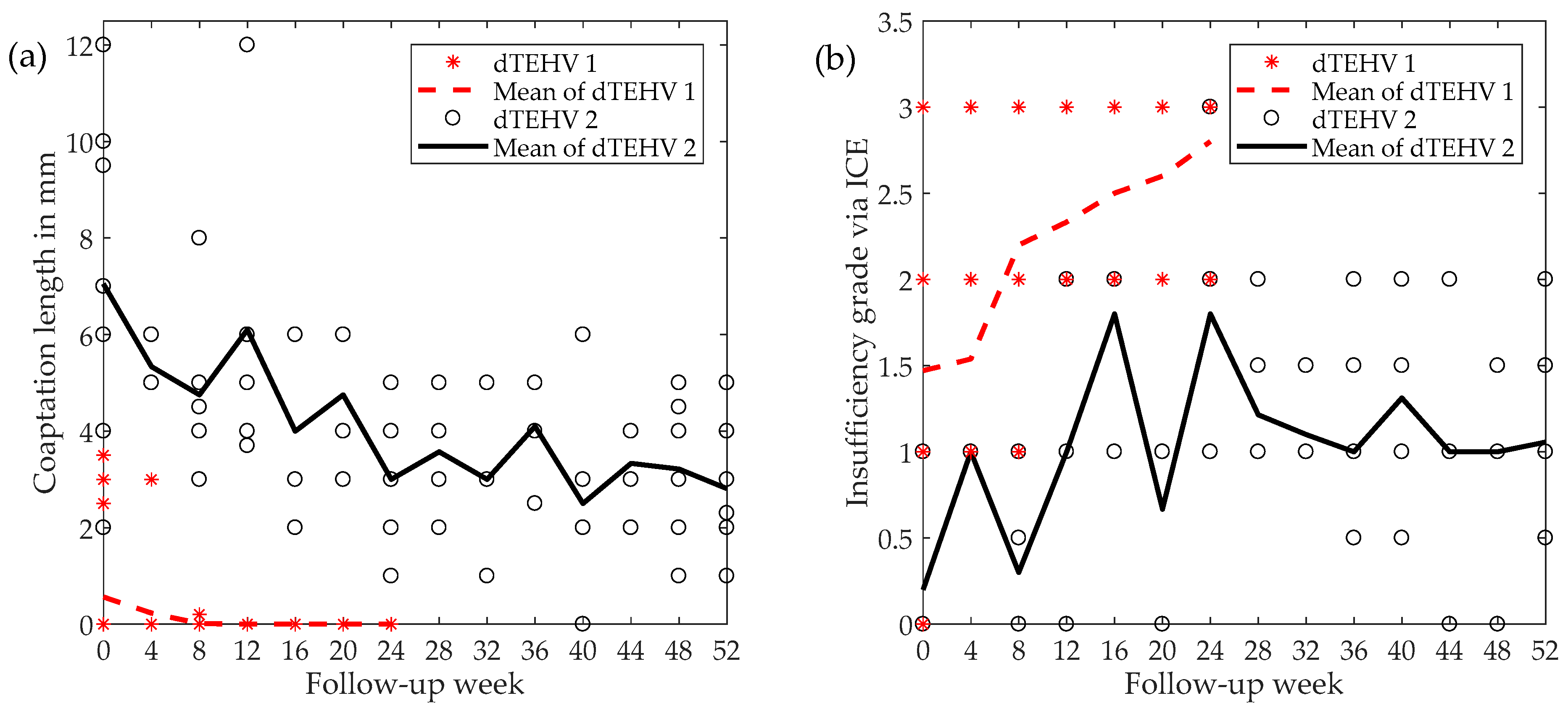
2.1. ICE
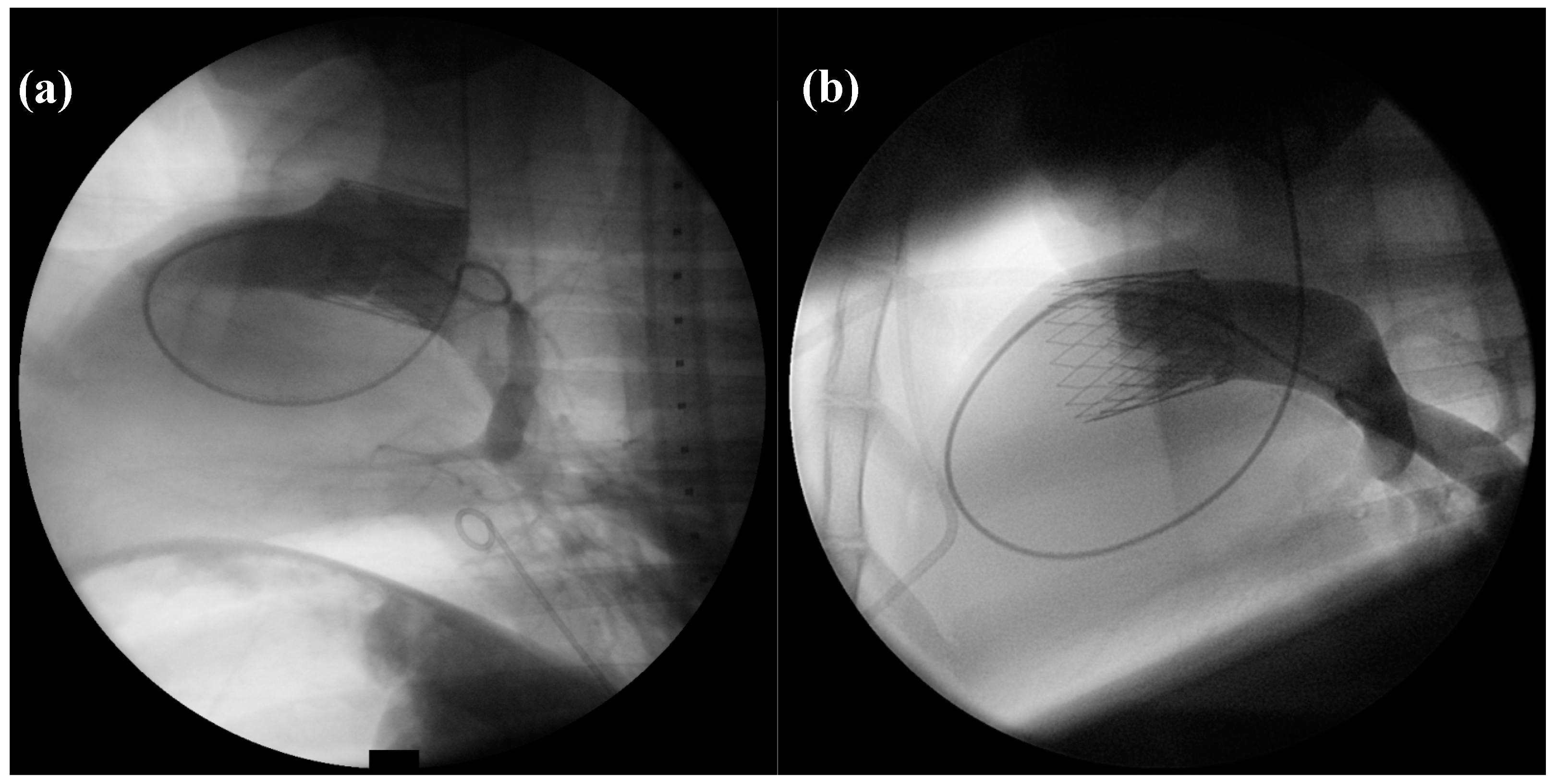
2.2. Angiography
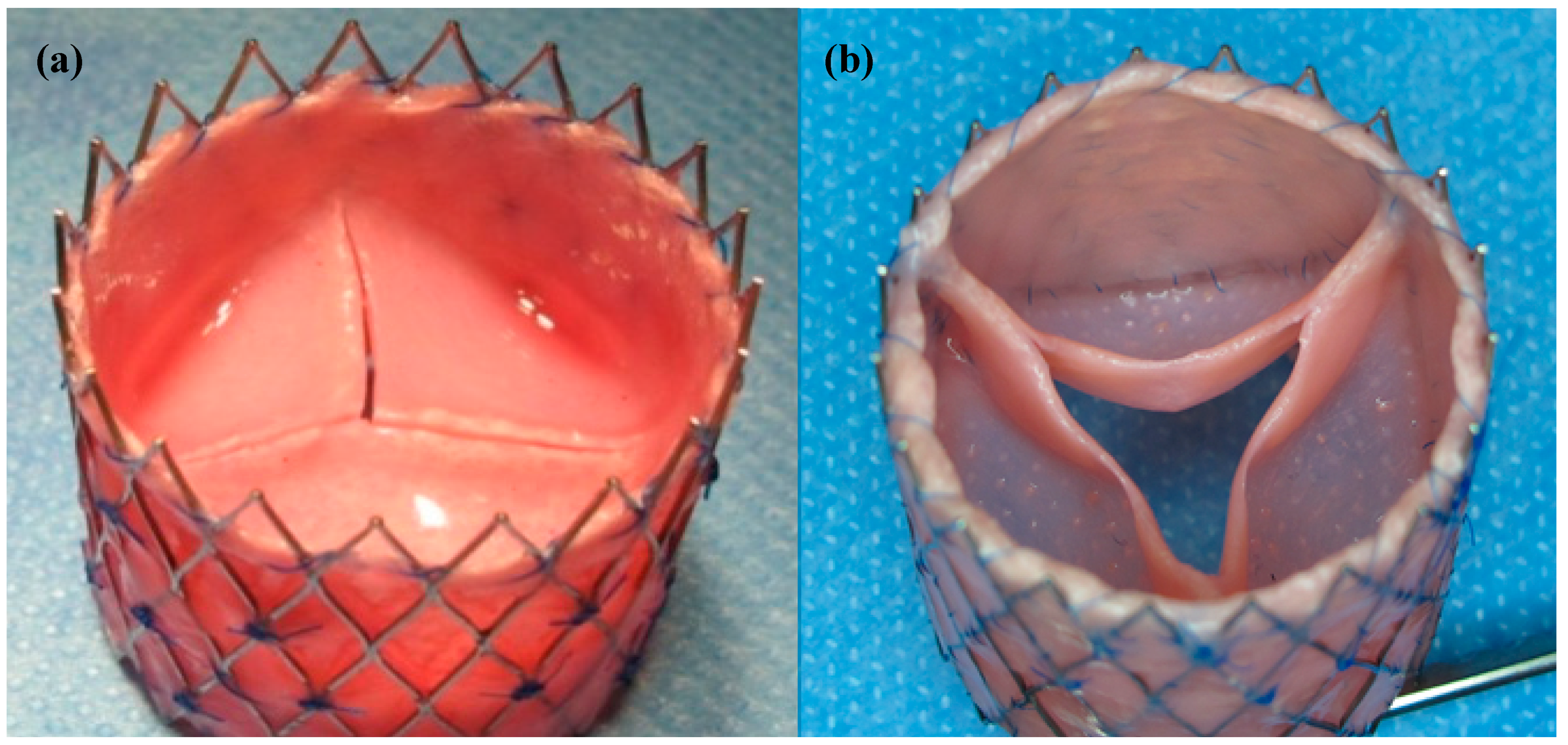
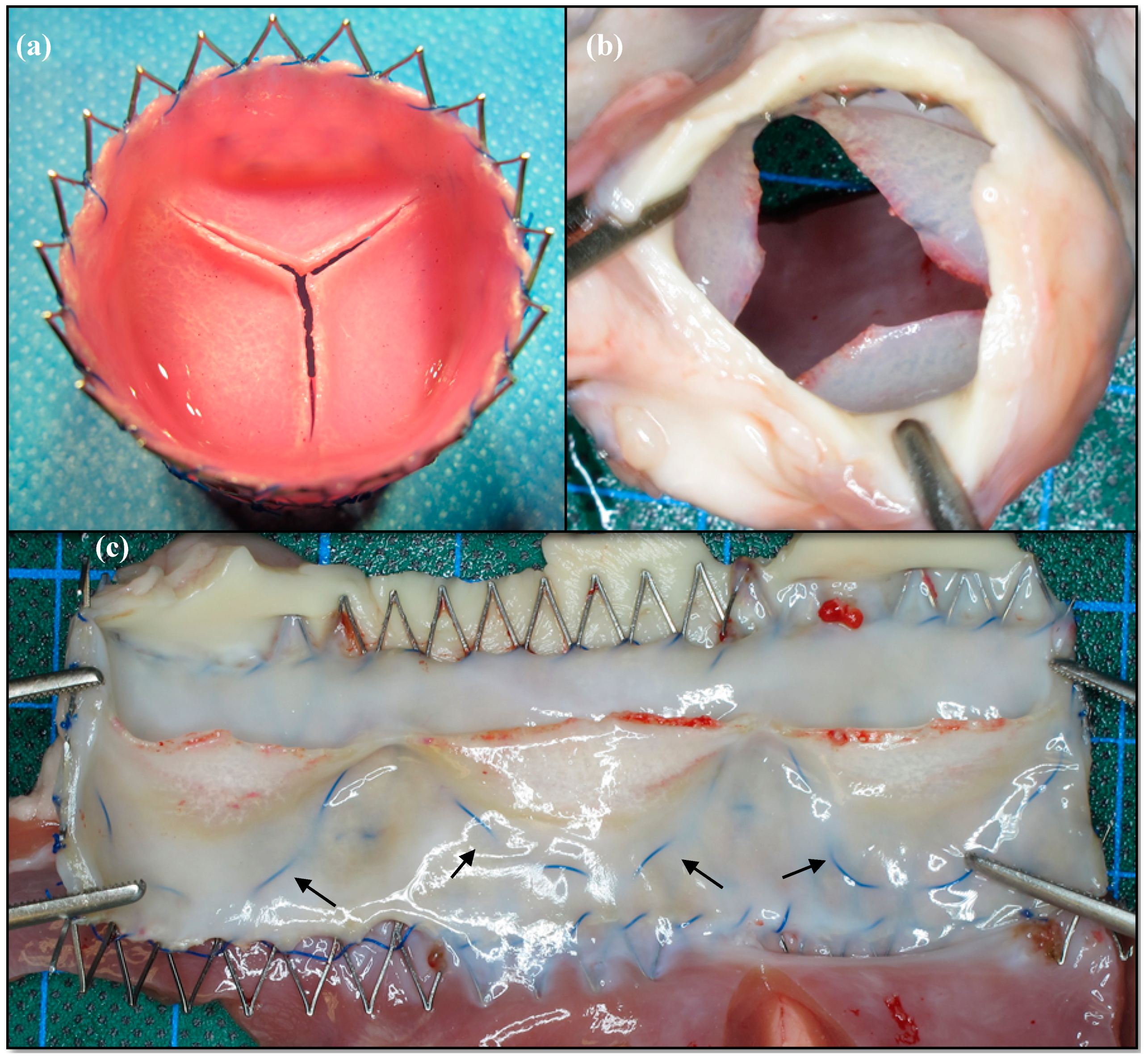
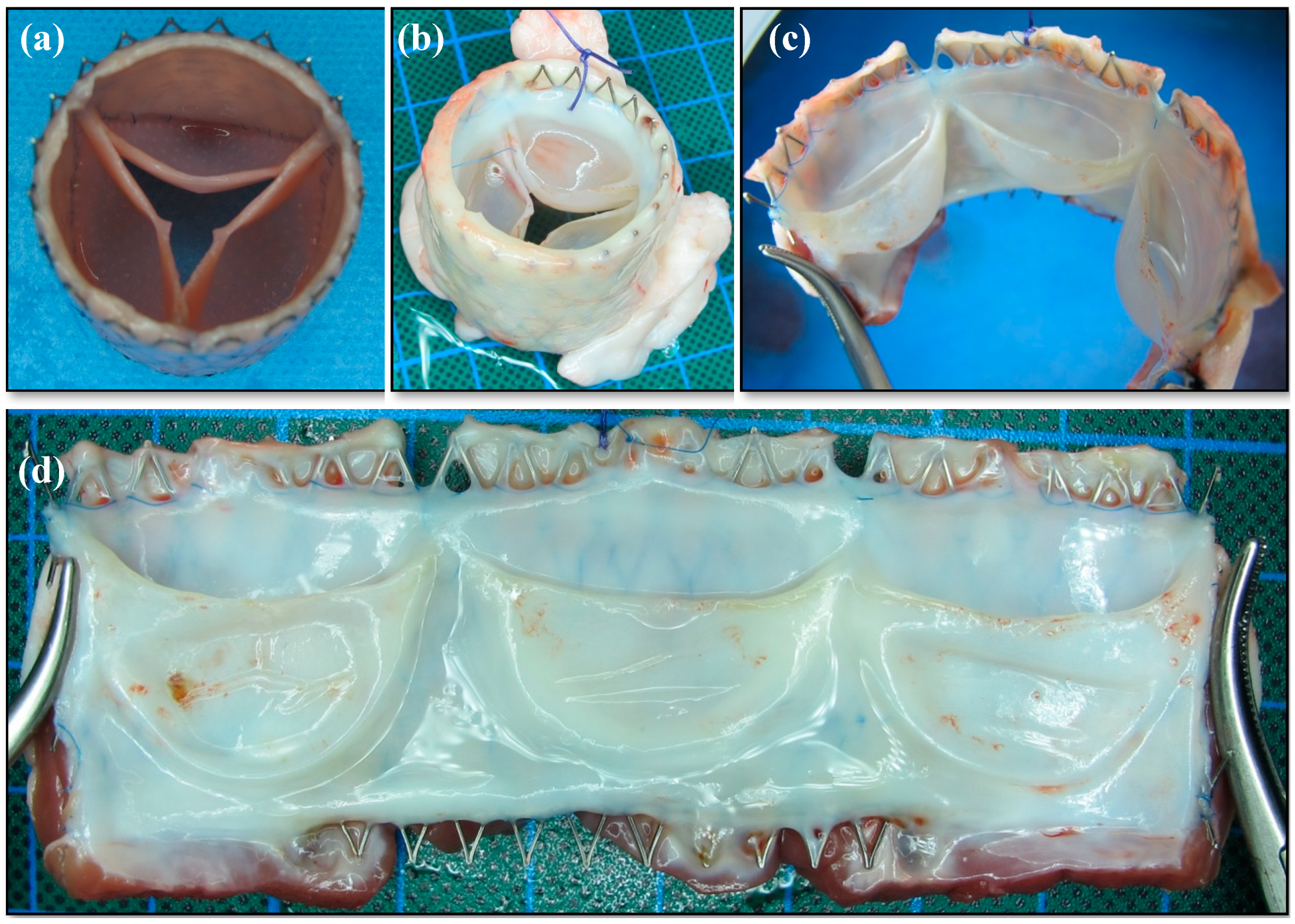
2.3. Macroscopic Analysis
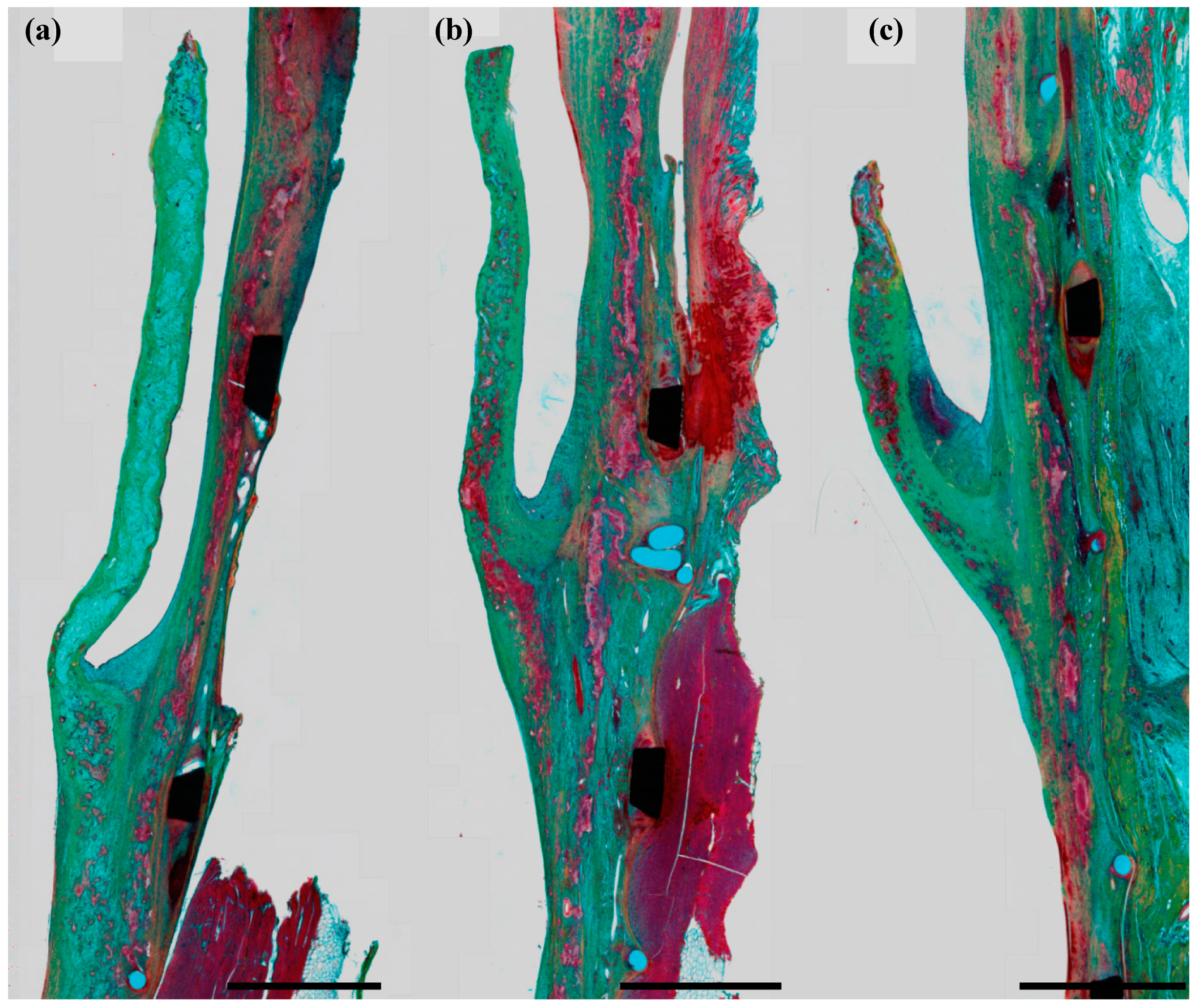
2.4. Histology
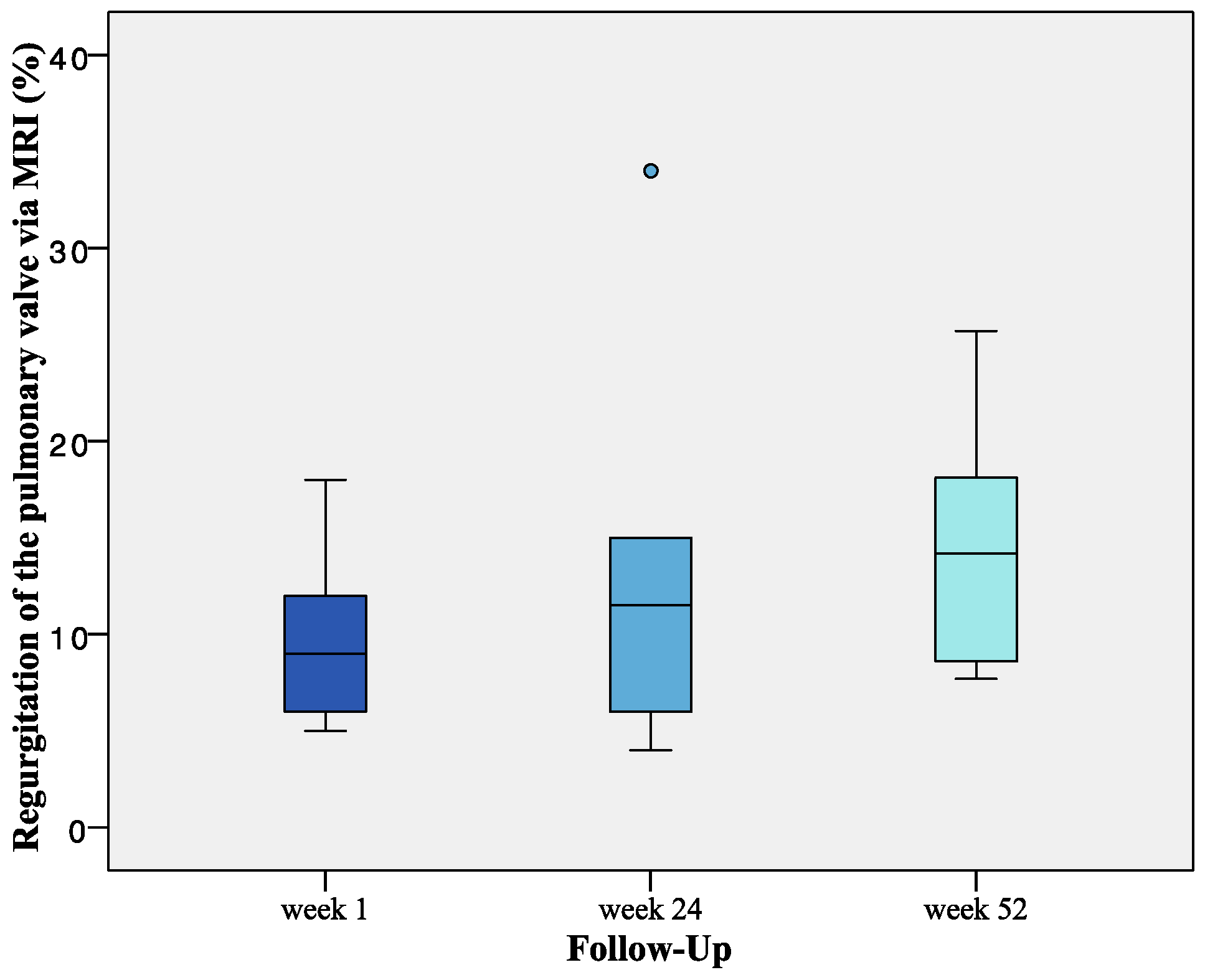
2.5. MRI
3. Discussion
4. Materials and Methods
4.1. Valve Manufacturing
4.2. Delivery System
4.3. Study Protocoll
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hoffman, J.I.E.; Kaplan, S. The incidence of congenital heart disease. J. Am. Coll. Cardiol. 2002, 39, 1890–1900. [Google Scholar] [CrossRef]
- Van der Linde, D.; Konings, E.E.; Slager, M.A.; Witsenburg, M.; Helbing, W.A.; Takkenberg, J.J.; Roos-Hesselink, J.W. Birth prevalence of congenital heart disease worldwide: A systematic review and meta-analysis. J. Am. Coll. Cardiol. 2011, 58, 2241–2247. [Google Scholar] [CrossRef] [PubMed]
- Reimer, J.; Syedain, Z.; Haynie, B.; Lahti, M.; Berry, J.; Tranquillo, R. Implantation of a Tissue-Engineered Tubular Heart Valve in Growing Lambs. Ann. Biomed. Eng. 2017, 45, 439–451. [Google Scholar] [CrossRef] [PubMed]
- Henaine, R.; Roubertie, F.; Vergnat, M.; Ninet, J. Valve replacement in children: A challenge for a whole life. Arch. Cardiovasc. Dis. 2012, 105, 517–528. [Google Scholar] [CrossRef] [PubMed]
- Cannegieter, S.C.; Rosendaal, F.R.; Briet, E. Thromboembolic and Bleeding Complications in Patients with Mechanical Heart-Valve Prostheses. Circulation 1994, 89, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Head, S.J.; Celik, M.; Kappetein, A.P. Mechanical versus bioprosthetic aortic valve replacement. Eur. Heart J. 2017, 38, 2183–2191. [Google Scholar] [CrossRef] [PubMed]
- Rabkin-Aikawa, E.; Mayer, J.E., Jr.; Schoen, F.J. Heart valve regeneration. Adv. Biochem. Eng. Biotechnol. 2005, 94, 141–179. [Google Scholar] [PubMed]
- Schoen, F.J.; Gotlieb, A.I. Heart valve health, disease, replacement, and repair: A 25-year cardiovascular pathology perspective. Cardiovasc. Pathol. 2016, 25, 341–352. [Google Scholar] [CrossRef] [PubMed]
- Fioretta, E.S.; Dijkman, P.E.; Emmert, M.Y.; Hoerstrup, S.P. The future of heart valve replacement: Recent developments and translational challenges for heart valve tissue engineering. J. Tissue Eng. Regen. Med. 2018, 12, e323–e335. [Google Scholar] [CrossRef] [PubMed]
- Ksiazek, A.A.; Mitchell, K.J.; Cesarovic, N.; Schwarzwald, C.C.; Hoerstrup, S.P.; Weber, B. PGA (polyglycolic acid)-P4HB (poly-4-hydroxybutyrate)-Based Bioengineered Valves in the Rat Aortic Circulation. J. Heart Valve Dis. 2016, 25, 380–388. [Google Scholar] [PubMed]
- Dijkman, P.E.; Driessen-Mol, A.; Frese, L.; Hoerstrup, S.P.; Baaijens, F.P.T. Decellularized homologous tissue-engineered heart valves as off-the-shelf alternatives to xeno- and homografts. Biomaterials 2012, 33, 4545–4554. [Google Scholar] [CrossRef] [PubMed]
- Generali, M.; Kehl, D.; Capulli, A.K.; Parker, K.K.; Hoerstrup, S.P.; Weber, B. Comparative analysis of poly-glycolic acid-based hybrid polymer starter matrices for in vitro tissue engineering. Colloids Surf. B Biointerfaces 2017, 158, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Kluin, J.; Talacua, H.; Smits, A.I.; Emmert, M.Y.; Brugmans, M.C.; Fioretta, E.S.; Dijkman, P.E.; Sontjens, S.H.; Duijvelshoff, R.; Dekker, S.; et al. In situ heart valve tissue engineering using a bioresorbable elastomeric implant—From material design to 12 months follow-up in sheep. Biomaterials 2017, 125, 101–117. [Google Scholar] [CrossRef] [PubMed]
- Sanders, B.; Driessen-Mol, A.; Bouten, C.V.C.; Baaijens, F.P.T. The Effects of Scaffold Remnants in Decellularized Tissue-Engineered Cardiovascular Constructs on the Recruitment of Blood Cells. Tissue Eng. Part A 2017, 23, 1142–1151. [Google Scholar] [CrossRef] [PubMed]
- Spriestersbach, H.; Prudlo, A.; Bartosch, M.; Sanders, B.; Radtke, T.; Baaijens, F.P.T.; Hoerstrup, S.P.; Berger, F.; Schmitt, B. First percutaneous implantation of a completely tissue-engineered self-expanding pulmonary heart valve prosthesis using a newly developed delivery system: A feasibility study in sheep. Cardiovasc. Interv. Ther. 2017, 32, 36–47. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, B.; Spriestersbach, H.; Radtke, T.; Bartosch, M.; Peters, H.; Sigler, M.; Frese, L.; Dijkman, P.E.; Baaijens, F.P.; Hoerstrup, S.P.; et al. Percutaneous pulmonary valve replacement using completely tissue-engineered off-the-shelf heart valves: Six-month in vivo functionality and matrix remodelling in sheep. EuroIntervention 2016, 12, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Driessen-Mol, A.; Emmert, M.Y.; Dijkman, P.E.; Frese, L.; Sanders, B.; Weber, B.; Cesarovic, N.; Sidler, M.; Leenders, J.; Jenni, R.; et al. Transcatheter Implantation of Homologous “Off-the-Shelf” Tissue-Engineered Heart Valves With Self-Repair Capacity. J. Am. Coll. Cardiol. 2014, 63, 1320–1329. [Google Scholar] [CrossRef] [PubMed]
- Sanders, B.; Loerakker, S.; Fioretta, E.S.; Bax, D.J.; Driessen-Mol, A.; Hoerstrup, S.P.; Baaijens, F.P. Improved Geometry of Decellularized Tissue Engineered Heart Valves to Prevent Leaflet Retraction. Ann. Biomed. Eng. 2016, 44, 1061–1071. [Google Scholar] [CrossRef] [PubMed]
- Lancellotti, P.; Tribouilloy, C.; Hagendorff, A.; Popescu, B.A.; Edvardsen, T.; Pierard, L.A.; Badano, L.; Zamorano, J.L.; Scientific Document Committee of the European Association of Cardiovascular. Recommendations for the echocardiographic assessment of native valvular regurgitation: An executive summary from the European Association of Cardiovascular Imaging. Eur. Heart J. Cardiovasc. Imaging 2013, 14, 611–644. [Google Scholar] [CrossRef] [PubMed]
- Emmert, M.Y.; Schmitt, B.A.; Loerakker, S.; Sanders, B.; Spriestersbach, H.; Fioretta, E.S.; Bruder, L.; Brakmann, K.; Motta, S.E.; Lintas, V.; et al. Computational modeling guides tissue-engineered heart valve design for long-term in vivo performance in a translational sheep model. Sci. Transl. Med. 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Aikawa, E.; Whittaker, P.; Farber, M.; Mendelson, K.; Padera, R.F.; Aikawa, M.; Schoen, F.J. Human semilunar cardiac valve remodeling by activated cells from fetus to adult: implications for postnatal adaptation, pathology, and tissue engineering. Circulation 2006, 113, 1344–1352. [Google Scholar] [CrossRef] [PubMed]
- Loerakker, S.; Argento, G.; Oomens, C.W.; Baaijens, F.P. Effects of valve geometry and tissue anisotropy on the radial stretch and coaptation area of tissue-engineered heart valves. J. Biomech. 2013, 46, 1792–1800. [Google Scholar] [CrossRef] [PubMed]
- Bartosch, M.; Peters, H.; Spriestersbach, H.; Darach, O.H.I.; Berger, F.; Schmitt, B. A Universal Delivery System for Percutaneous Heart Valve Implantation. Ann. Biomed. Eng. 2016, 44, 2683–2694. [Google Scholar] [CrossRef] [PubMed]


| Time Point and Measurement Variable | dTEVH 1 | dTEHV 2 | p-Value (Wilcoxon Test) | |
|---|---|---|---|---|
| Week 0 | Insufficiency grade | 1.47 ± 0.26 | 0.2 ± 0.13 | 0.003 * |
| Coaptation length | 0.56 ± 0.30 mm | 7.05 ± 0.92 mm | 0.000 * | |
| Week 24 | Insufficiency grade | 2.80 ± 0.2 | 1.80 ± 0.37 | 0.095 |
| Coaptation length in mm | 0.00 ± 0.00 mm | 3.00 ± 0.71 mm | 0.005 * | |
| Week 52 | Insufficiency grade | n.a. | 1.06 ± 0.16 | n.a. |
| Coaptation length in mm | n.a. | 2.81 ± 0.39 mm | n.a. | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bruder, L.; Spriestersbach, H.; Brakmann, K.; Stegner, V.; Sigler, M.; Berger, F.; Schmitt, B. RETRACTED: Transcatheter Decellularized Tissue-Engineered Heart Valve (dTEHV) Grown on Polyglycolic Acid (PGA) Scaffold Coated with P4HB Shows Improved Functionality over 52 Weeks due to Polyether-Ether-Ketone (PEEK) Insert. J. Funct. Biomater. 2018, 9, 64. https://doi.org/10.3390/jfb9040064
Bruder L, Spriestersbach H, Brakmann K, Stegner V, Sigler M, Berger F, Schmitt B. RETRACTED: Transcatheter Decellularized Tissue-Engineered Heart Valve (dTEHV) Grown on Polyglycolic Acid (PGA) Scaffold Coated with P4HB Shows Improved Functionality over 52 Weeks due to Polyether-Ether-Ketone (PEEK) Insert. Journal of Functional Biomaterials. 2018; 9(4):64. https://doi.org/10.3390/jfb9040064
Chicago/Turabian StyleBruder, Leon, Hendrik Spriestersbach, Kerstin Brakmann, Valentin Stegner, Matthias Sigler, Felix Berger, and Boris Schmitt. 2018. "RETRACTED: Transcatheter Decellularized Tissue-Engineered Heart Valve (dTEHV) Grown on Polyglycolic Acid (PGA) Scaffold Coated with P4HB Shows Improved Functionality over 52 Weeks due to Polyether-Ether-Ketone (PEEK) Insert" Journal of Functional Biomaterials 9, no. 4: 64. https://doi.org/10.3390/jfb9040064
APA StyleBruder, L., Spriestersbach, H., Brakmann, K., Stegner, V., Sigler, M., Berger, F., & Schmitt, B. (2018). RETRACTED: Transcatheter Decellularized Tissue-Engineered Heart Valve (dTEHV) Grown on Polyglycolic Acid (PGA) Scaffold Coated with P4HB Shows Improved Functionality over 52 Weeks due to Polyether-Ether-Ketone (PEEK) Insert. Journal of Functional Biomaterials, 9(4), 64. https://doi.org/10.3390/jfb9040064





