Carbon Dots Doped with Dysprosium: A Bimodal Nanoprobe for MRI and Fluorescence Imaging
Abstract
:1. Introduction
2. Experimental
2.1. Synthesis Process
2.2. Characterization
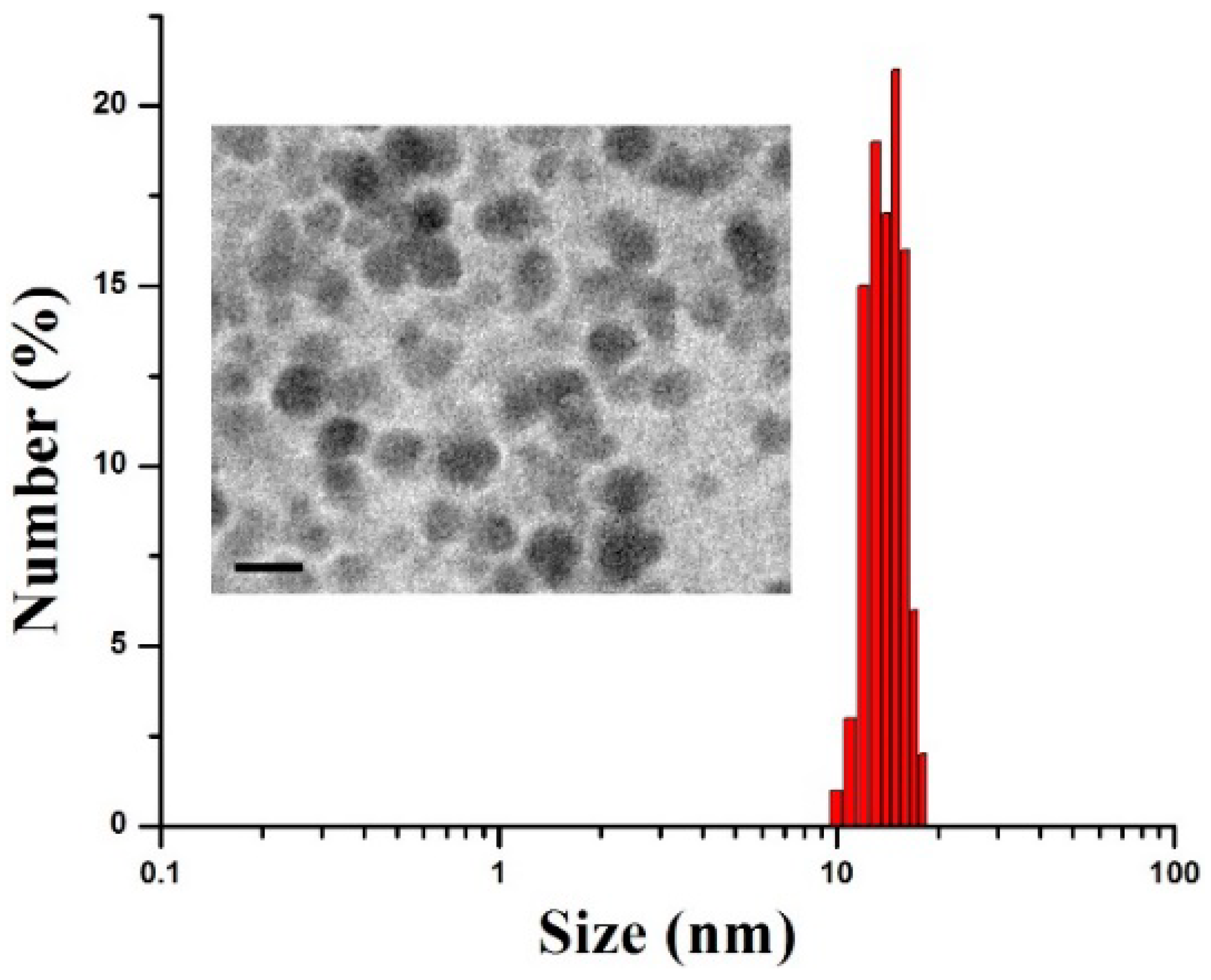
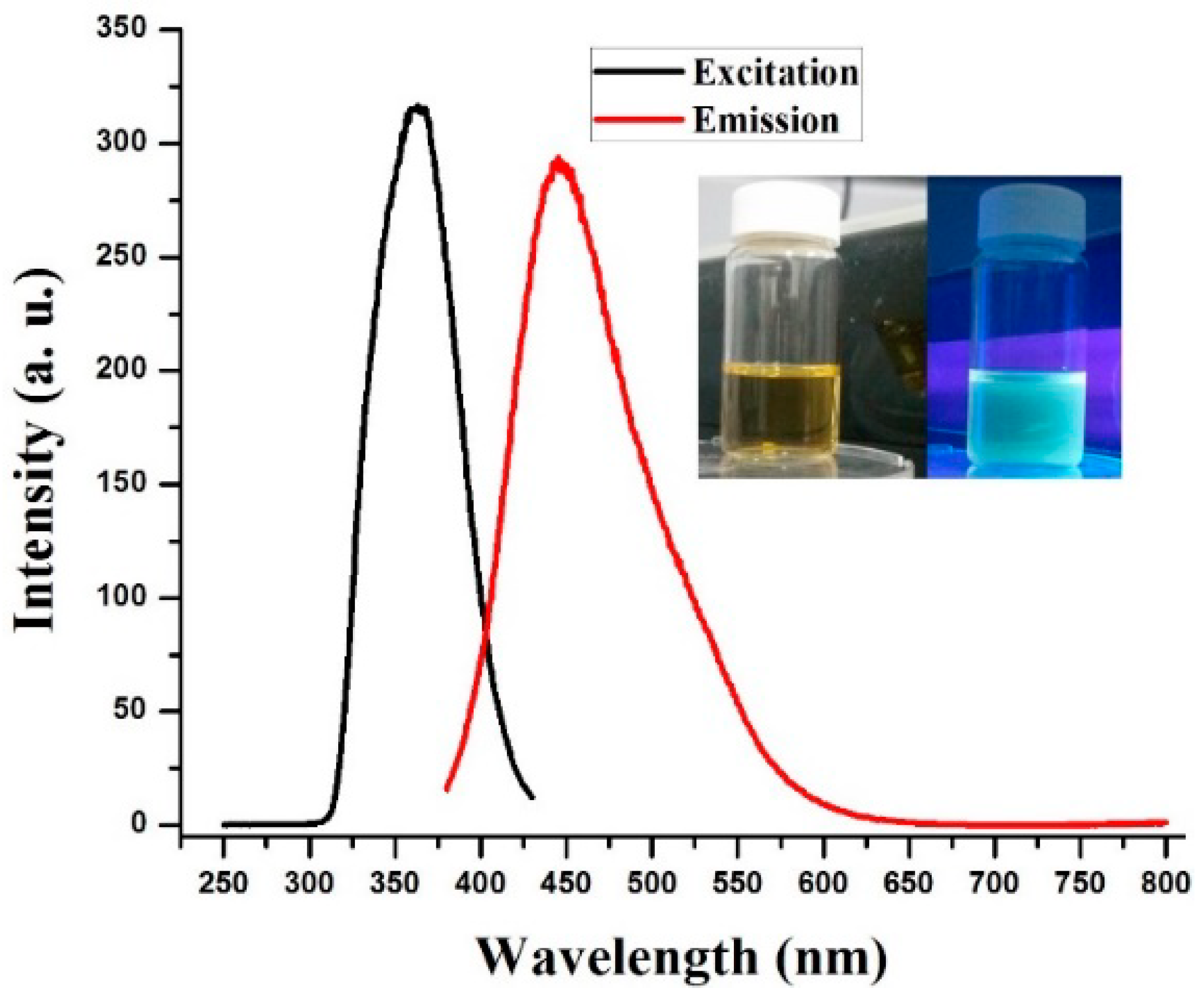
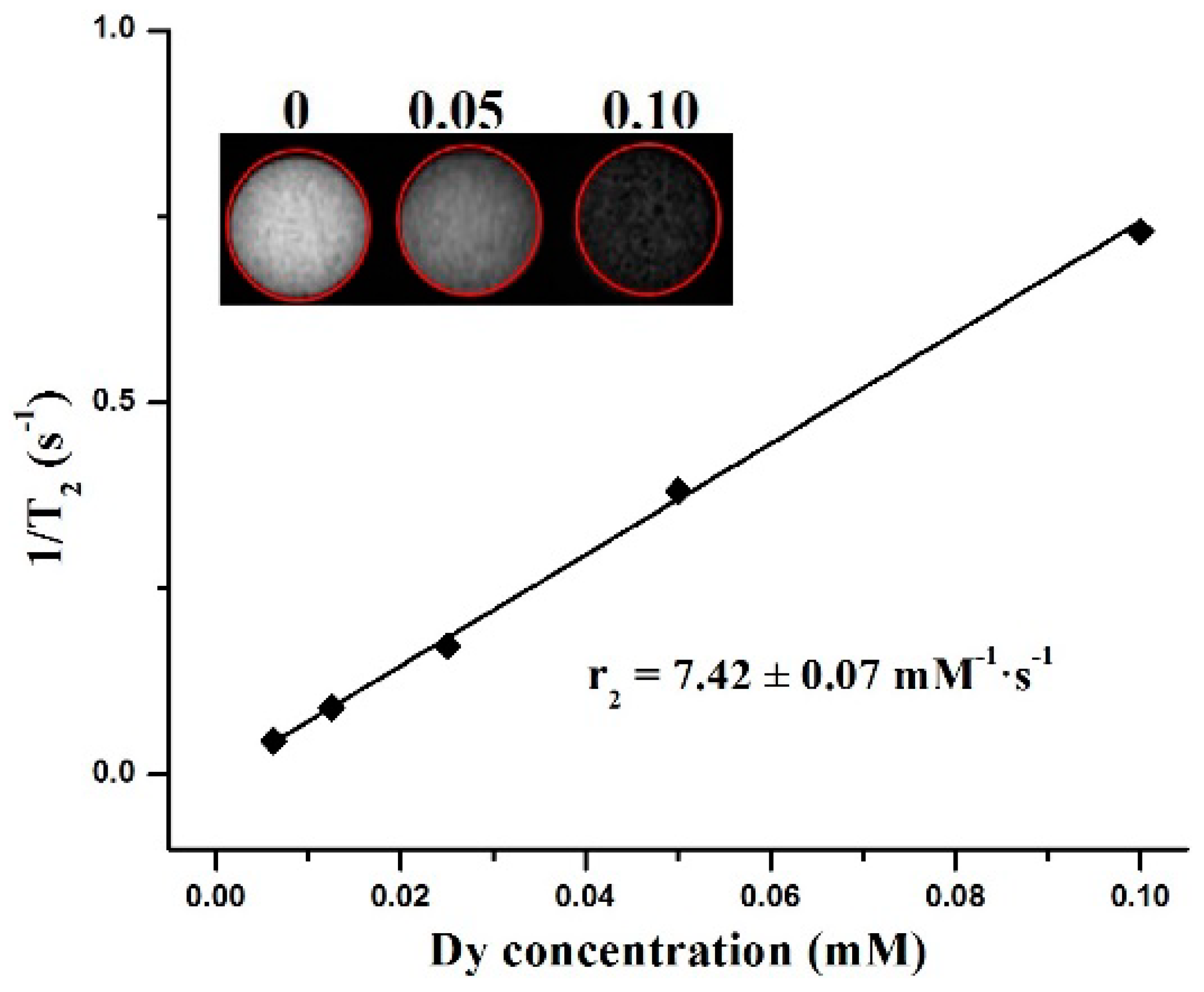
3. Results and Discussion
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Gu, J.; Zhang, W.; Yang, X. Preparation of a superparamagnetic MRI contrast agent with a tumor targeting function. Mater. Lett. 2013, 94, 8–10. [Google Scholar] [CrossRef]
- Atabaev, T.S. PEG-coated superparamagnetic dysprosium-doped Fe3O4 nanoparticles for potential MRI imaging. BioNanoScience 2018, 8, 299–303. [Google Scholar] [CrossRef]
- Chen, N.; Shao, C.; Qu, Y.; Li, S.; Gu, W.; Zheng, T.; Ye, L.; Yu, C. Folic acid-conjugated MnO nanoparticles as a T1 contrast agent for Magnetic Resonance imaging of tiny brain gliomas. ACS Appl. Mater. Interfaces 2014, 6, 19850–19857. [Google Scholar] [CrossRef] [PubMed]
- Norek, M.; Kampert, E.; Zeitler, U.; Peters, J.A. Tuning of the size of Dy2O3 nanoparticles for optimal performance as an MRI contrast agent. J. Am. Chem. Soc. 2008, 130, 5335–5340. [Google Scholar] [CrossRef] [PubMed]
- Atabaev, T.S.; Lee, J.H.; Han, D.-W.; Kim, H.-K.; Hwang, Y.-H. Ultrafine PEG-capped gadolinia nanoparticles: Cytotoxicity and potential biomedical applications for MRI and luminescent imaging. RSC Adv. 2014, 4, 34343–34349. [Google Scholar] [CrossRef]
- Atabaev, T.; Lee, J.; Shin, Y.; Han, D.-W.; Choo, K.; Jeon, U.; Hwang, J.; Yeom, J.; Kim, H.-K.; Hwang, Y.-H. Eu, Gd-Codoped Yttria Nanoprobes for Optical and T1-Weighted Magnetic Resonance Imaging. Nanomaterials 2017, 7, 35. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Kattel, K.; Park, J.Y.; Chang, Y.; Kim, T.J.; Lee, G.H. Paramagnetic nanoparticle T1 and T2 MRI contrast agents. Phys. Chem. Chem. Phys. 2012, 14, 12687. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Huang, S.; Yu, K.J.; Clyne, A.M. Dextran and polymer polyethylene glycol (PEG) coating reduce both 5 and 30 nm iron oxide nanoparticle cytotoxicity in 2D and 3D cell culture. Int. J. Mol. Sci. 2012, 13, 5554–5570. [Google Scholar] [CrossRef] [PubMed]
- Atabaev, T.S.; Lee, J.H.; Han, D.-W.; Kim, H.-K.; Hwang, Y.-H. Fabrication of carbon coated gadolinia particles for dual-mode magnetic resonance and fluorescence imaging. J. Adv. Ceram. 2015, 4, 118–122. [Google Scholar] [CrossRef]
- Atabaev, T.S.; Lee, J.H.; Han, D.W.; Choo, K.S.; Jeon, U.B.; Hwang, J.Y.; Yeom, J.A.; Kang, C.; Kim, H.K.; Hwang, Y.H. Multicolor nanoprobes based on silica-coated gadolinium oxide nanoparticles with highly reduced toxicity. RSC Adv. 2016, 6, 19758–19762. [Google Scholar] [CrossRef]
- Tesch, A.; Wenisch, C.; Herrmann, K.H.; Reichenbach, J.R.; Warncke, P.; Fischer, D.; Müller, F.A. Luminomagnetic Eu3+- and Dy3+-doped hydroxyapatite for multimodal imaging. Mater. Sci. Eng. C 2017, 81, 422–431. [Google Scholar] [CrossRef] [PubMed]
- Atabaev, T.S. Paramagnetic Gd-doped zirconia nanoparticles for potential T1-weighted MRI imaging. Nano Life 2017, 7, 1750007. [Google Scholar] [CrossRef]
- Ren, X.Y.; Yuan, X.X.; Wang, Y.P.; Liu, C.L.; Qin, Y.; Guo, L.P.; Liu, L.H. Facile preparation of Gd3+ doped carbon quantum dots: Photoluminescence materials with magnetic resonance response as magnetic resonance/fluorescence bimodal probes. Opt. Mater. 2016, 57, 56–62. [Google Scholar] [CrossRef]
- Yu, C.; Xuan, T.; Chen, Y.; Zhao, Z.; Liu, X.; Lian, G.; Li, H. Gadolinium-doped carbon dots with high quantum yield as an effective fluorescence and magnetic resonance bimodal imaging probe. J. Alloys Compd. 2016, 688, 611–619. [Google Scholar] [CrossRef]
- Briley-Saebo, K.; Bjørnerud, A.; Grant, D.; Ahlstrom, H.; Berg, T.; Kindberg, G.M. Hepatic cellular distribution and degradation of iron oxide nanoparticles following single intravenous injection in rats: Implications for magnetic resonance imaging. Cell Tissue Res. 2004, 316, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Atabaev, T.S.; Shin, Y.C.; Song, S.-J.; Han, D.-W.; Hong, N.H. Toxicity and T2-weighted magnetic resonance imaging potentials of holmium oxide nanoparticles. Nanomaterials 2017, 7, 216. [Google Scholar] [CrossRef] [PubMed]
- Liang, Q.; Ma, W.; Shi, Y.; Li, Z.; Yang, X. Easy synthesis of highly fluorescent carbon quantum dots from gelatin and their luminescent properties and applications. Carbon 2013, 60, 421–428. [Google Scholar] [CrossRef]
- Zhou, J.; Shan, X.; Ma, J.; Gu, Y.; Qian, Z.; Chen, J.; Feng, H. Facile synthesis of P-doped carbon quantum dots with highly efficient photoluminescence. RSC Adv. 2014, 4, 5465. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Atabaev, T.S.; Piao, Z.; Molkenova, A. Carbon Dots Doped with Dysprosium: A Bimodal Nanoprobe for MRI and Fluorescence Imaging. J. Funct. Biomater. 2018, 9, 35. https://doi.org/10.3390/jfb9020035
Atabaev TS, Piao Z, Molkenova A. Carbon Dots Doped with Dysprosium: A Bimodal Nanoprobe for MRI and Fluorescence Imaging. Journal of Functional Biomaterials. 2018; 9(2):35. https://doi.org/10.3390/jfb9020035
Chicago/Turabian StyleAtabaev, Timur Sh., Zhonglie Piao, and Anara Molkenova. 2018. "Carbon Dots Doped with Dysprosium: A Bimodal Nanoprobe for MRI and Fluorescence Imaging" Journal of Functional Biomaterials 9, no. 2: 35. https://doi.org/10.3390/jfb9020035
APA StyleAtabaev, T. S., Piao, Z., & Molkenova, A. (2018). Carbon Dots Doped with Dysprosium: A Bimodal Nanoprobe for MRI and Fluorescence Imaging. Journal of Functional Biomaterials, 9(2), 35. https://doi.org/10.3390/jfb9020035







