Cleaning Effectiveness of Three NiTi Rotary Instruments: A Focus on Biomaterial Properties
Abstract
:1. Introduction
2. Results and Discussion
| Group | Coronal | Middle | Apical | Overall |
|---|---|---|---|---|
| Mtwo | 1.33 a | 1.40 a | 1.53 a | 1.42 a |
| Revo-S | 1.33 a | 1.53 a | 1.60 a | 1.49 a |
| HyFlex CM | 2.60 b | 2.53 b | 2.40 b | 2.51 b |
| Group | Canal level | Score = 1 | Score = 2 | Score = 3 | Score = 4 | Score = 5 |
|---|---|---|---|---|---|---|
| Mtwo | Coronal | 11 | 3 | 1 | 0 | 0 |
| Middle | 10 | 4 | 1 | 0 | 0 | |
| Apical | 9 | 4 | 2 | 0 | 0 | |
| Revo-S | Coronal | 11 | 3 | 1 | 0 | 0 |
| Middle | 9 | 4 | 2 | 0 | 0 | |
| Apical | 9 | 3 | 3 | 0 | 0 | |
| HyFlex CM | Coronal | 1 | 4 | 10 | 0 | 0 |
| Middle | 2 | 3 | 10 | 0 | 0 | |
| Apical | 3 | 3 | 9 | 0 | 0 |
| Group | Coronal | Middle | Apical | Overall |
|---|---|---|---|---|
| Mtwo | 1.13 a | 1.33 a | 1.40 a | 1.29 a |
| Revo-S | 1.20 a | 1.27 a | 1.33 a | 1.27 a |
| HyFlex CM | 2.33 b | 2.60 b | 3.27 b | 2.73 b |
| Group | Canal level | Score = 1 | Score = 2 | Score = 3 | Score = 4 | Score = 5 |
|---|---|---|---|---|---|---|
| Mtwo | Coronal | 13 | 2 | 0 | 0 | 0 |
| Middle | 11 | 3 | 1 | 0 | 0 | |
| Apical | 11 | 2 | 2 | 0 | 0 | |
| Revo-S | Coronal | 12 | 3 | 0 | 0 | 0 |
| Middle | 12 | 2 | 1 | 0 | 0 | |
| Apical | 12 | 1 | 2 | 0 | 0 | |
| HyFlex CM | Coronal | 4 | 4 | 5 | 2 | 0 |
| Middle | 2 | 5 | 5 | 3 | 0 | |
| Apical | 0 | 3 | 7 | 3 | 2 |



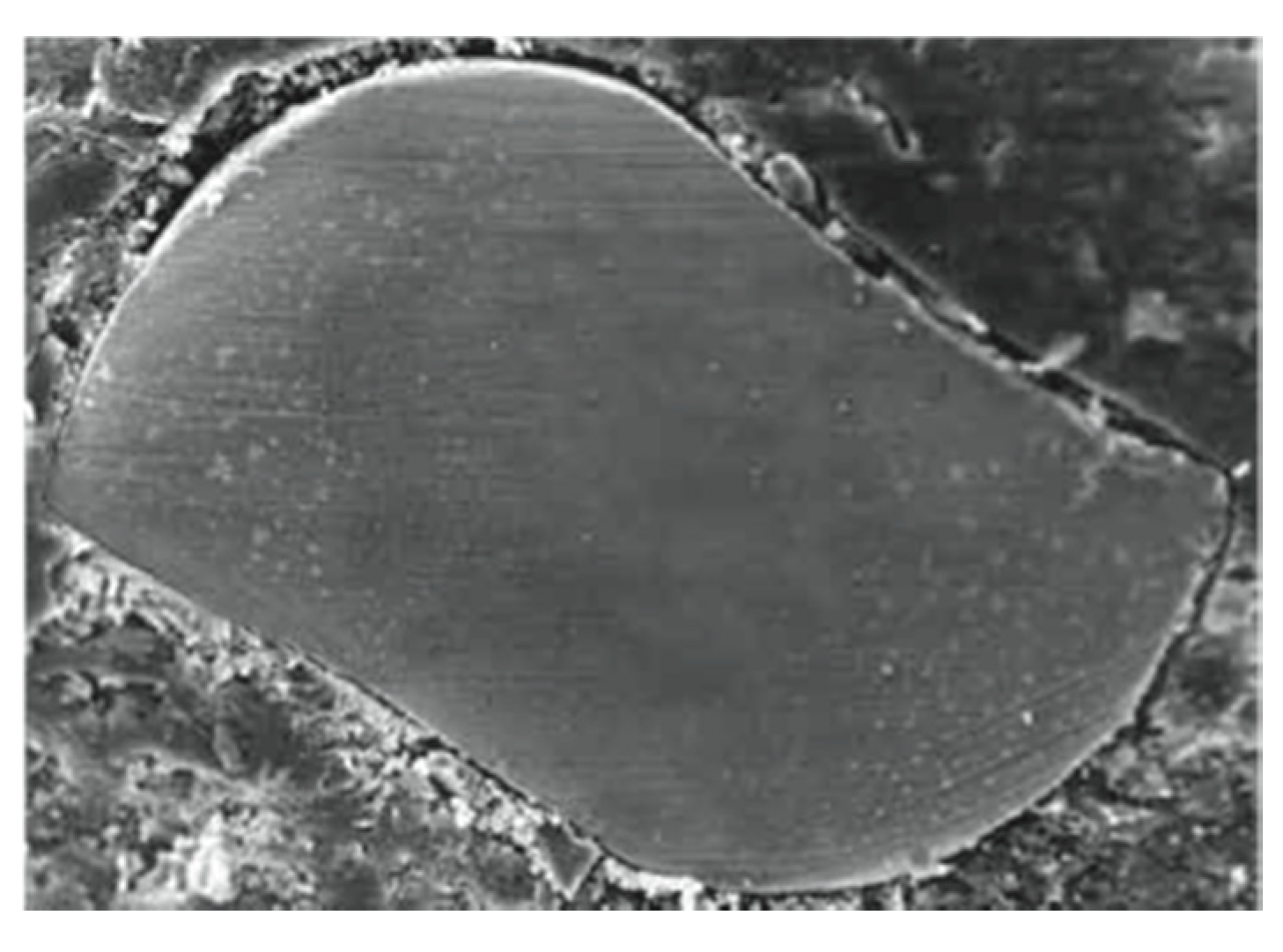
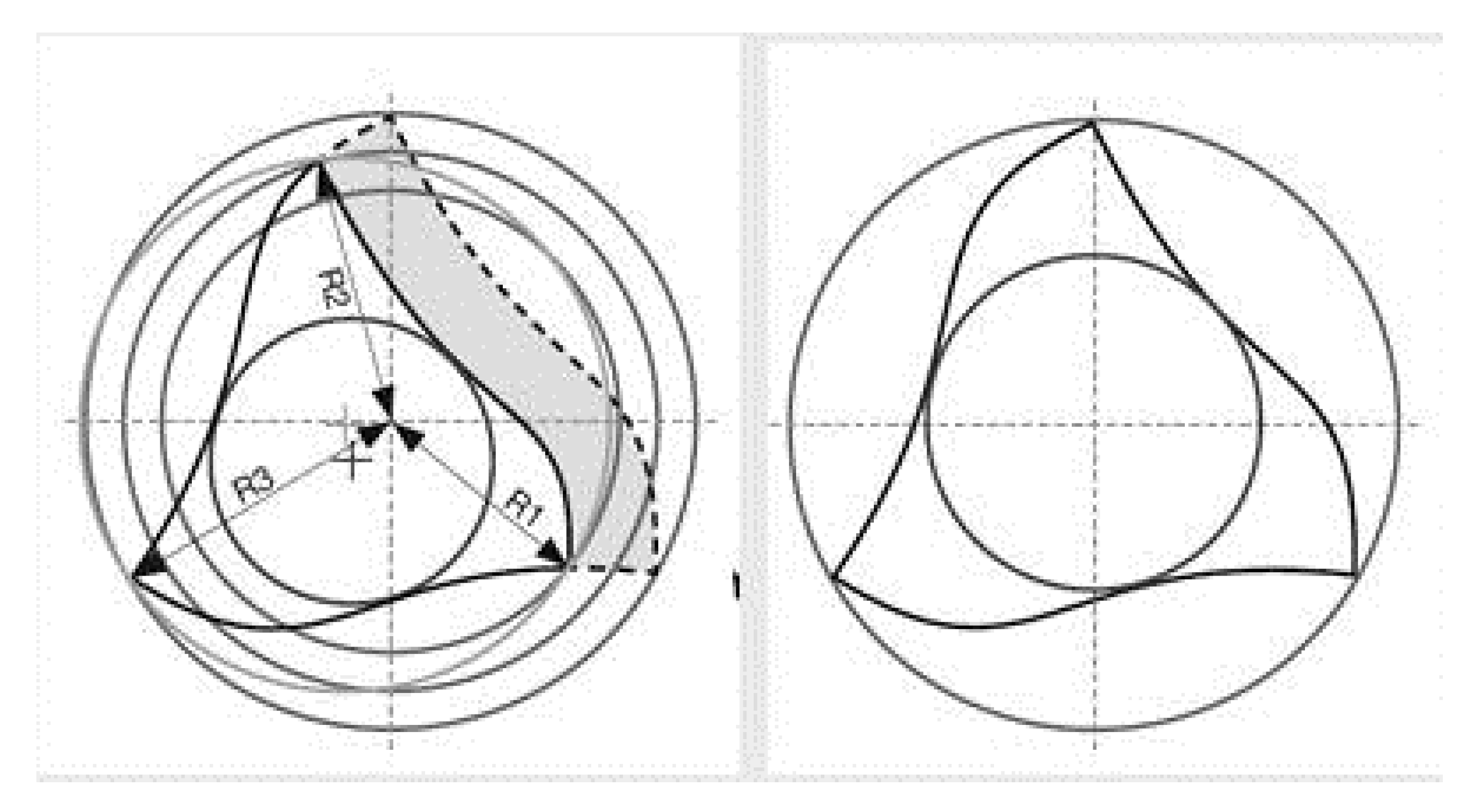
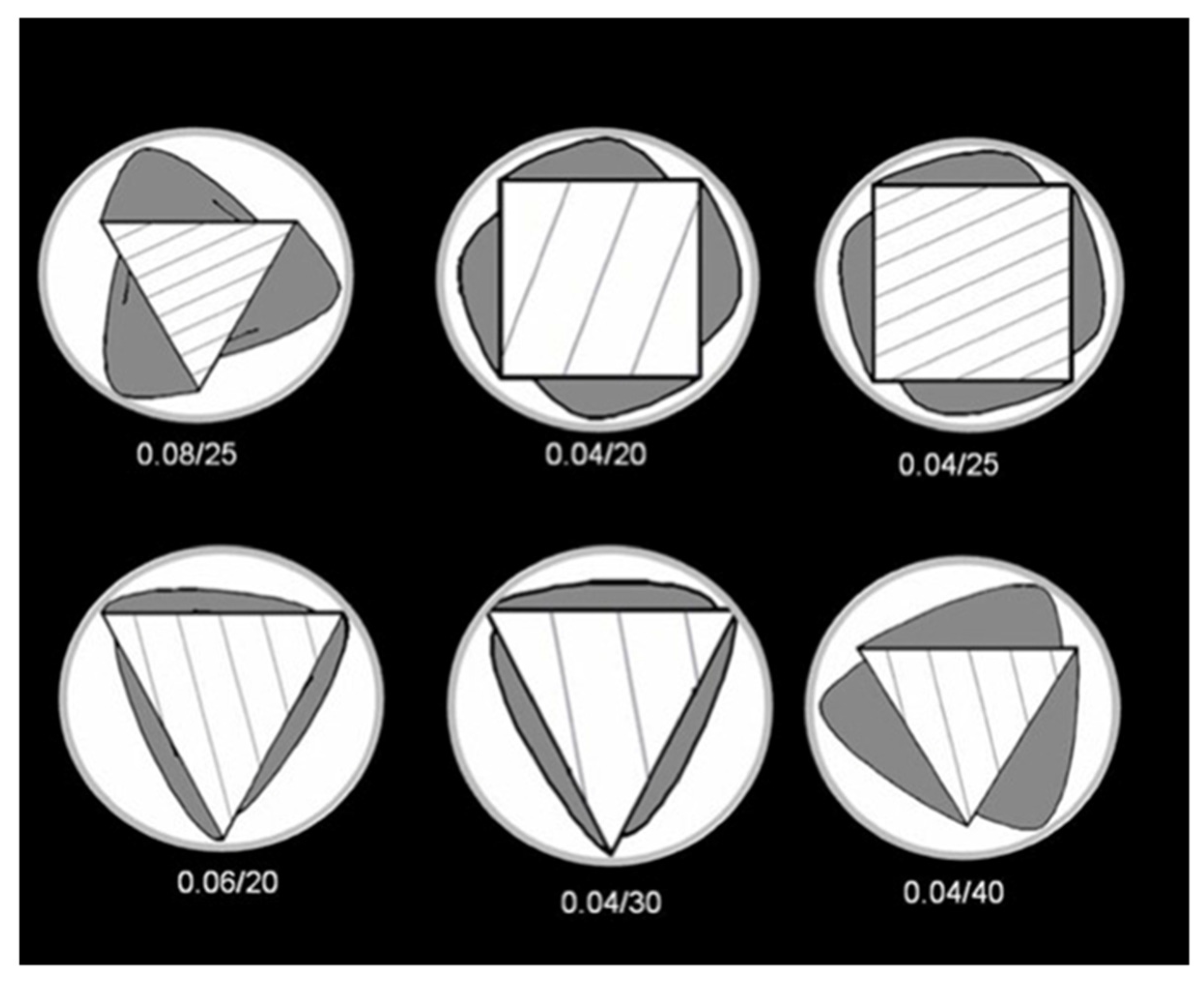
3. Experimental Section
3.1. Root Canal Instrumentation
- (Group A) Mtwo (Sweden Martina, Due Carrare, Padova, Italy);
- (Group B) Revo-S (MicroMega, Besancon, France);
- (Group C) HyFlex CM (Coltene Whaledent AG, Altstatten, Switzerland).
3.2. SEM Preparation and Examination
- Score 1: clean root canal wall, only few small debris particles;
- Score 2: few small agglomerations of debris;
- Score 3: many agglomeration of debris covering less than 50% of the root canal wall;
- Score 4: more than 50% of the root canal wall covered by debris;
- Score 5: complete or nearly complete root canal wall covered by debris.
- Score 1: no smear layer, orifices of dentinal tubules open;
- Score 2: small amount of smear layer, some dentinal tubules open;
- Score 3: homogenous smear layer covering the root canal wall, only few dentinal tubules open;
- Score 4: complete root canal wall covered by a homogenous smear layer, no open dentinal tubules;
- Score 5: heavy, homogenous smear layer covering the entire root canal wall.
3.3. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Torabinejad, M.; Walton, R.E. Endodontics: Principles and Practice, 4th ed.; Saunders Elsevier: St. Louis, MO, USA, 2009. [Google Scholar]
- Abou-Rass, M.; Piccinino, M.V. The effectiveness of four clinical irrigation methods on the removal of root canal debris. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 1982, 53, 524–526. [Google Scholar] [CrossRef]
- Briseño, B.M.; Wirth, R.; Hamm, G.; Standhartinger, W. Efficacy of different irrigation methods and concentrations of root canal irrigation solutions on bacteria in the root canal. Dent. Traumatol. 1992, 8, 6–11. [Google Scholar] [CrossRef]
- Kaplan, A.E.; Picca, M.; Bonzalez, M.I.; Macchi, R.L.; Molgatini, S.L. Antimicrobial effect of six endodontic sealers: An in vitro evaluation. Dent. Traumatol. 1999, 15, 42–45. [Google Scholar] [CrossRef]
- Mickel, A.K.; Nguyen, T.H.; Chogle, S. Antimicrobial activity of endodontic sealers on enterococcus faecalis. J. Endod. 2003, 29, 257–258. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.I.; Doran, J.E. An in vitro evaluation of the particle flotation capability of various irrigating solutions. J. Calif. Dent. Assoc. 1975, 3, 60–63. [Google Scholar] [PubMed]
- D’Arcangelo, C.; Varvara, G.; de Fazio, P. An evaluation of the action of different root canal irrigants on facoltative aerobic-anaerobic, obligate anaerobic, and microaerophilic bacteria. J. Endod. 1999, 25, 351–353. [Google Scholar] [CrossRef] [PubMed]
- Jeansonne, M.J.; White, R.R. A comparison of 2.0% chlorhexidine gluconate and 5.25% sodium hypochlorite as antimicrobial endodontic irrigants. J. Endod. 1994, 20, 276–278. [Google Scholar]
- Jeansonne, J., Jr.; Batista, M.; Fraga, R.; de Uzeda, M. Antibacterial effects of endodontic irrigants on black-pigmented gram-negative anaerobes and facultative bacteria. J. Endod. 1998, 24, 414–416. [Google Scholar] [CrossRef] [PubMed]
- Sundqvist, G.; Figdor, D.; Persson, S.; Sjogren, U. Microbiologic analysis of teeth with failed endodontic treatment and the outcome of conservative retreatment. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 1998, 85, 86–93. [Google Scholar] [CrossRef]
- Shabahang, S.; Pouresmail, M.; Torabinejad, M. In vitro antimicrobial efficacy of MTAD and sodium hypochlorite. J. Endod. 2003, 29, 450–452. [Google Scholar] [CrossRef] [PubMed]
- De-Deus, G.; Garcia-Filho, P. Influence of the NiTi rotary system on the debridement quality of the root canal space. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2009, 108, 71–76. [Google Scholar] [CrossRef]
- Rodig, T.; Hulsmann, M.; Kahlmeier, C. Comparison of root canal preparation with two rotary NiTi instruments: ProFile 0.04 and GT Rotary. Int. Endod. J. 2007, 40, 553–562. [Google Scholar] [CrossRef]
- Wadhwani, K.K.; Tikku, A.P.; Chandra, A.; Shakya, V.K. A comparative evaluation of smear layer removal with ethylenediaminetetraacetic acid in different states: A SEM study. Indian J. Dent. Res. 2011, 22, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Radcliffe, C.E.; Potouridou, L.; Qureshi, R.; Habahbeh, N.; Qualtrough, A.; Worthington, H.; Drucker, D.B. Antimicrobical activity of varying concentrations of sodium hypochlorite on the endodontic microorganisms Actinomyces israelii, A. naeslundii, Candida albicans and Enterococcus faecalis. Int. Endod. J. 2004, 37, 438–446. [Google Scholar] [CrossRef]
- Dagna, A.; Arciola, C.R.; Florindi, F.; Scribante, A.; Saino, E.; Visai, L.; Poggio, C. In vitro evaluation of antimicrobial efficacy of endodontic irrigants. Int. J. Artif. Organs 2011, 34, 914–919. [Google Scholar] [CrossRef] [PubMed]
- Shuping, G.B.; Orstavik, D.; Sigurdsson, A.; Trope, M. Reduction of intracanal bacteria using nickel-titanium rotary instrumentation and various medications. J. Endod. 2000, 26, 751–755. [Google Scholar] [CrossRef] [PubMed]
- Foschi, F.; Nucci, C.; Montebugnoli, L.; Breschi, L.; Malagnino, V.A.; Prati, C. SEM evaluation of canal wall dentine following use of Mtwo and ProTaper NiTi rotary instruments. Int. Endod. J. 2004, 37, 832–839. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Heredia, M.; Ferrer-Luque, C.M.; González-Rodríguez, M.P. The effectiveness of different acid irrigating solutions in root canal cleaning after hand and rotary instrumentation. J. Endod. 2006, 32, 993–997. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Wu, H.; Zheng, Y.; Zhang, H.; Li, H.; Zhou, X. Scanning electron microscopic evaluation of debris and smear layer remaining following use of ProTaper and Hero Shaper instruments in combination with NaOCl and EDTA irrigation. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2008, 106, 63–71. [Google Scholar] [CrossRef]
- Rollison, S.; Barnett, F.; Stevens, R.H. Efficacy of bacterial removal from instrumented root canals in vitro related to instrumentation technique and size. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2002, 94, 366–371. [Google Scholar] [CrossRef]
- De Lima Machado, M.E.; Bichels Sapia, L.A.; Cai, S.; Martins, G.H.; Nabeshima, C.K. Comparison of two rotary systems in root canal preparation regarding disinfection. J. Endod. 2010, 36, 1238–1240. [Google Scholar] [CrossRef] [PubMed]
- Dagna, A.; Arciola, C.R.; Visai, L.; Selan, L.; Colombo, M.; Bianchi, S.; Poggio, C. Antibacterial efficacy of conventional and single-use Ni-Ti endodontic instruments: An in vitro microbiological evaluation. Int. J. Artif. Organs 2012, 35, 826–831. [Google Scholar] [PubMed]
- Economides, N.; Liolios, E.; Kolokuris, I.; Beltes, P. Long-term evaluation of the influence of smear layer removal on the sealing ability of different sealers. J. Endod. 1999, 25, 123–125. [Google Scholar] [CrossRef] [PubMed]
- Karagöz-Küçükay, I.; Bayirli, G. An apical leakage study in the presence and absence of the smear layer. Int. Endod. J. 1994, 27, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Testarelli, L.; Plotino, G.; Al-Sudani, D.; Vincenzi, V.; Giansiracusa, A.; Grande, N.M.; Gambarini, G. Bending properties of a new nickel-titanium alloy with a lower percent by weight of nickel. J. Endod. 2011, 37, 1293–1295. [Google Scholar] [CrossRef] [PubMed]
- Peters, O.A.; Gluskin, A.K.; Weiss, R.A.; Han, J.T. An in vitro assessment of the physical propeties of novel Hyflex nickel-titanium rotary instruments. Int. Endod. J. 2012, 45, 1027–1034. [Google Scholar] [CrossRef] [PubMed]
- Hulsmann, M.; Rummelin, C.; Schafers, F. Root canal cleanliness after preparation with different endodontic handpieces and hand instruments: A comparative SEM investigation. J. Endod. 1997, 23, 301–306. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poggio, C.; Dagna, A.; Chiesa, M.; Beltrami, R.; Bianchi, S. Cleaning Effectiveness of Three NiTi Rotary Instruments: A Focus on Biomaterial Properties. J. Funct. Biomater. 2015, 6, 66-76. https://doi.org/10.3390/jfb6010066
Poggio C, Dagna A, Chiesa M, Beltrami R, Bianchi S. Cleaning Effectiveness of Three NiTi Rotary Instruments: A Focus on Biomaterial Properties. Journal of Functional Biomaterials. 2015; 6(1):66-76. https://doi.org/10.3390/jfb6010066
Chicago/Turabian StylePoggio, Claudio, Alberto Dagna, Marco Chiesa, Riccardo Beltrami, and Stefano Bianchi. 2015. "Cleaning Effectiveness of Three NiTi Rotary Instruments: A Focus on Biomaterial Properties" Journal of Functional Biomaterials 6, no. 1: 66-76. https://doi.org/10.3390/jfb6010066
APA StylePoggio, C., Dagna, A., Chiesa, M., Beltrami, R., & Bianchi, S. (2015). Cleaning Effectiveness of Three NiTi Rotary Instruments: A Focus on Biomaterial Properties. Journal of Functional Biomaterials, 6(1), 66-76. https://doi.org/10.3390/jfb6010066








