Ex Vivo Osteogenesis Induced by Calcium Silicate-Based Cement Extracts
Abstract
1. Introduction
2. Materials and Methods
2.1. Calcium Silicate-Based Cements and Preparation of the Extracts
2.2. Testing the Cement Extracts in the Ex Vivo Embryonic Chick Femurs Model
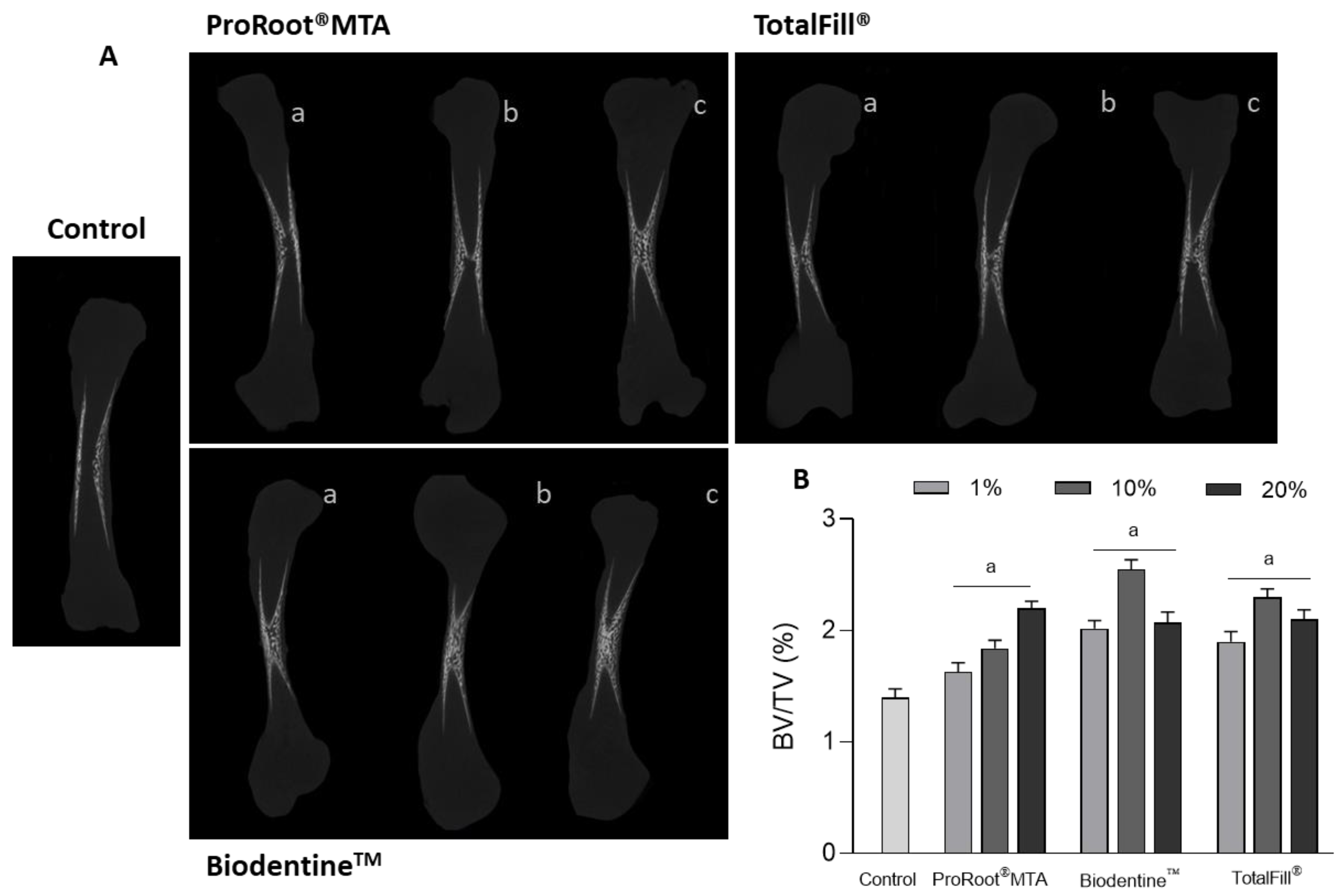
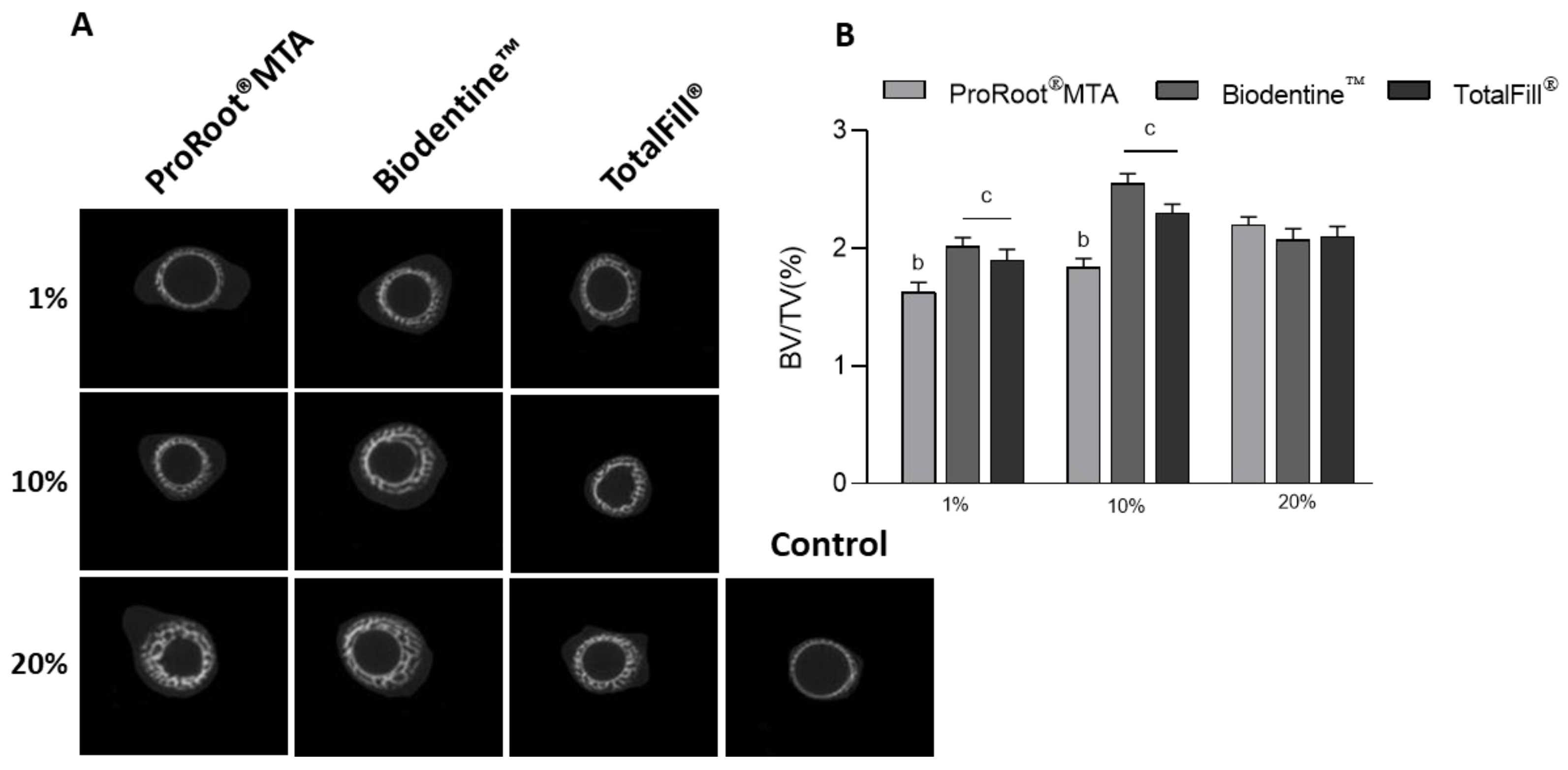
2.3. Microtomographic Evaluation
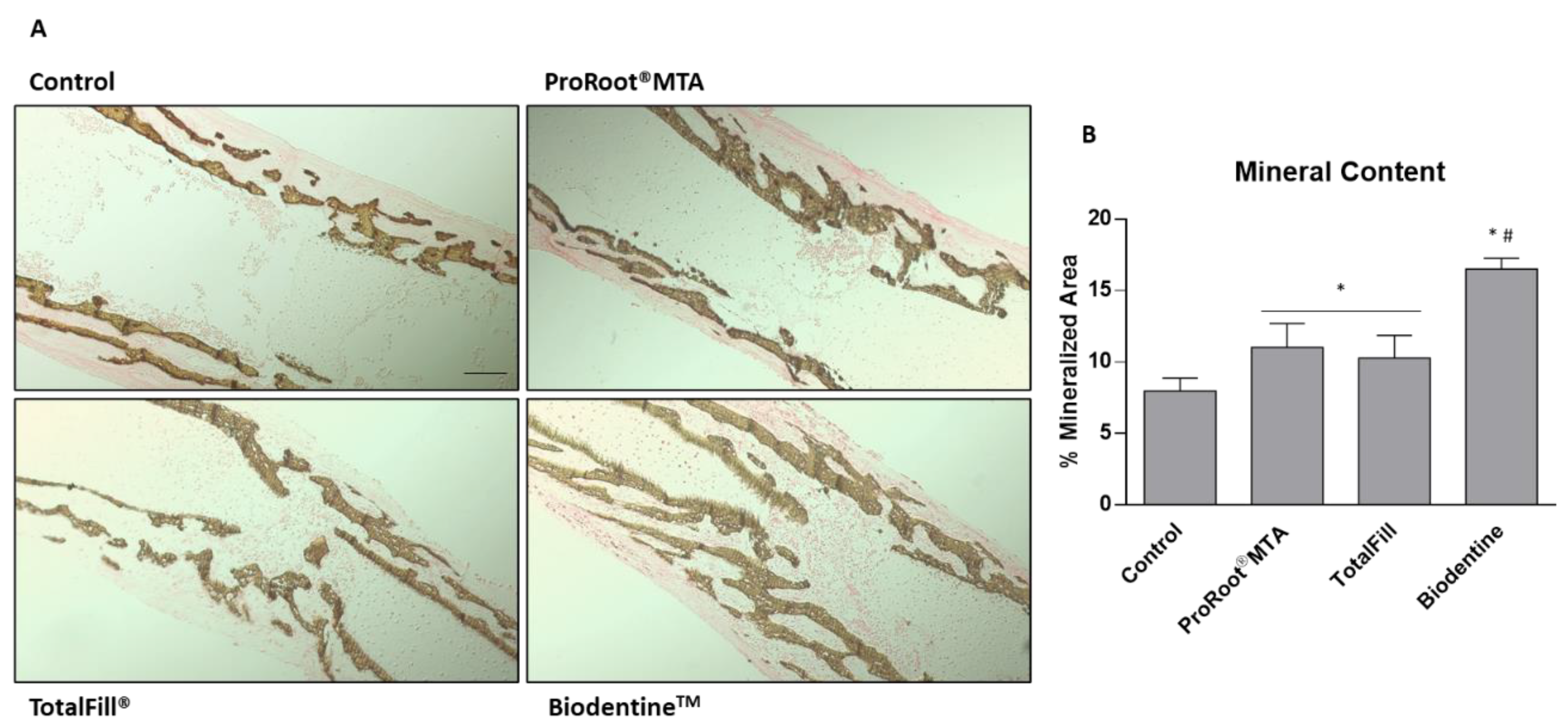
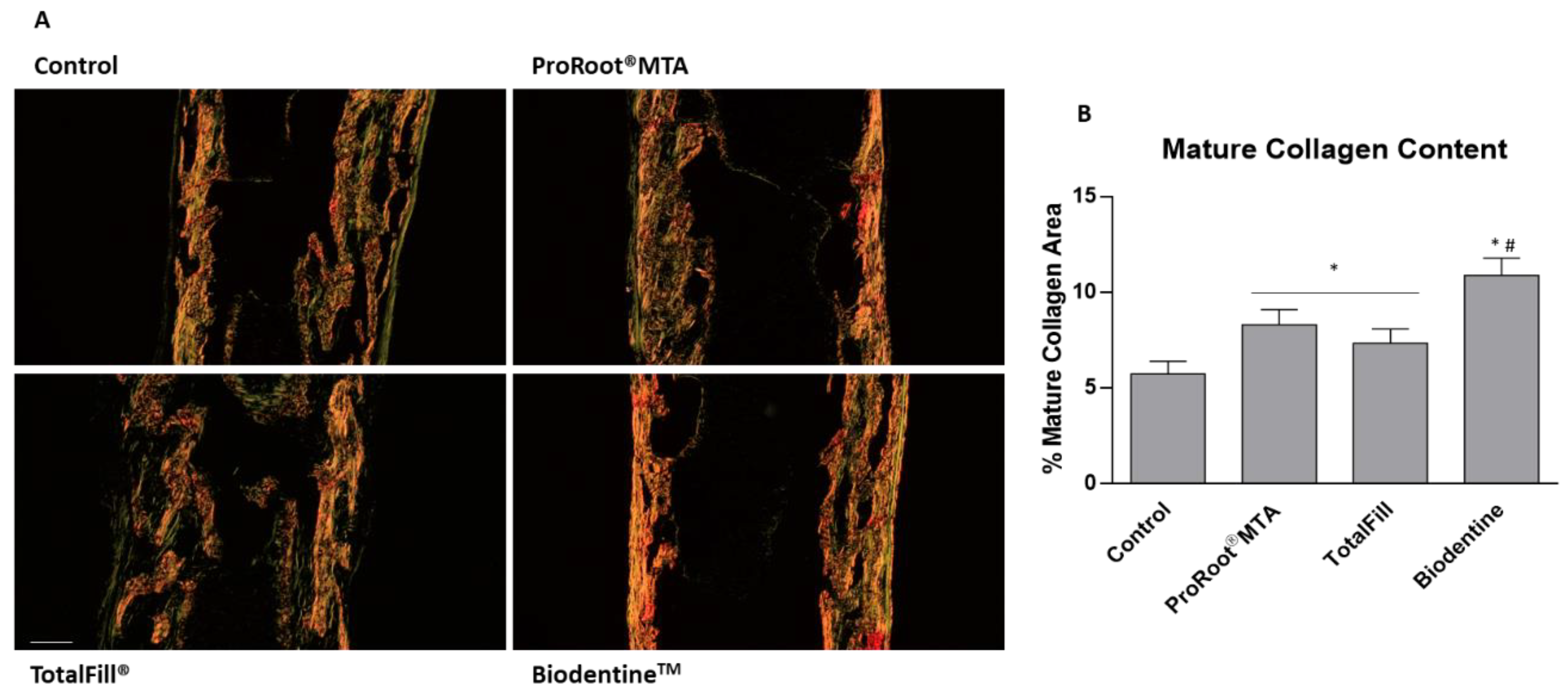
2.4. Histological Processing, Histochemical Staining, and Histomorphometric Analysis
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yin, H.; Yang, X.; Peng, L.; Xia, C.; Zhang, D.; Cui, F.; Huang, H.; Li, Z. Trends of calcium silicate biomaterials in medical research and applications: A bibliometric analysis from 1990 to 2020. Front. Pharmacol. 2022, 13, 991377. [Google Scholar] [CrossRef] [PubMed]
- Torabinejad, M.; Parirokh, M.; Dummer, P.M.H. Mineral trioxide aggregate and other bioactive endodontic cements: An updated overview—Part II: Other clinical applications and complications. Int. Endod. J. 2018, 51, 284–317. [Google Scholar] [CrossRef] [PubMed]
- Camilleri, J. Classification of Hydraulic Cements Used in Dentistry. Front. Dent. Med. 2020, 1, 9. [Google Scholar] [CrossRef]
- Mestry, S.U.; Kalmegh, S.; Mhaske, S.T. Mineral Trioxide Aggregates (MTA) in Dentistry: A Review on Chemistry, Synthesis Methods, and Critical Properties. Silicon 2023, 15, 2231–2249. [Google Scholar] [CrossRef]
- Shahi, S.; Fakhri, E.; Yavari, H.; Maleki Dizaj, S.; Salatin, S.; Khezri, K. Portland Cement: An Overview as a Root Repair Material. Biomed. Res. Int. 2022, 2022, 3314912. [Google Scholar] [CrossRef]
- Eskandari, F.; Razavian, A.; Hamidi, R.; Yousefi, K.; Borzou, S. An Updated Review on Properties and Indications of Calcium Silicate-Based Cements in Endodontic Therapy. Int. J. Dent. 2022, 2022, 6858088. [Google Scholar] [CrossRef]
- Prati, C.; Gandolfi, M.G. Calcium silicate bioactive cements: Biological perspectives and clinical applications. Dent. Mater. Off. Publ. Acad. Dent. Mater. 2015, 31, 351–370. [Google Scholar] [CrossRef]
- An, S.; Gao, Y.; Huang, Y.; Jiang, X.; Ma, K.; Ling, J. Short-term effects of calcium ions on the apoptosis and onset of mineralization of human dental pulp cells in vitro and in vivo. Int. J. Mol. Med. 2015, 36, 215–221. [Google Scholar] [CrossRef]
- Götz, W.; Tobiasch, E.; Witzleben, S.; Schulze, M. Effects of Silicon Compounds on Biomineralization, Osteogenesis, and Hard Tissue Formation. Pharmaceutics 2019, 11, 117. [Google Scholar] [CrossRef]
- Han, P.; Wu, C.; Xiao, Y. The effect of silicate ions on proliferation, osteogenic differentiation and cell signalling pathways (WNT and SHH) of bone marrow stromal cells. Biomater. Sci. 2013, 1, 379–392. [Google Scholar] [CrossRef]
- Ber, B.S.; Hatton, J.F.; Stewart, G.P. Chemical modification of proroot mta to improve handling characteristics and decrease setting time. J. Endod. 2007, 33, 1231–1234. [Google Scholar] [CrossRef] [PubMed]
- Abu Hasna, A.; de Paula Ramos, L.; Campos, T.; de Castro Lopes, S.; Rachi, M.; de Oliveira, L.; Carvalho, C. Biological and chemical properties of five mineral oxides and of mineral trioxide aggregate repair high plasticity: An in vitro study. Sci. Rep. 2022, 12, 13123. [Google Scholar] [CrossRef] [PubMed]
- Malkondu, Ö.; Karapinar Kazandağ, M.; Kazazoğlu, E. A review on biodentine, a contemporary dentine replacement and repair material. Biomed. Res. Int. 2014, 2014, 160951. [Google Scholar] [CrossRef] [PubMed]
- Camilleri, J.; Atmeh, A.; Li, X.; Meschi, N. Present status and future directions: Hydraulic materials for endodontic use. Int. Endod. J. 2022, 55, 710–777. [Google Scholar] [CrossRef]
- Septodont. Biodentine Active Biosilicate Technologyz—Scientific File; Septodont: Paris, France, 2010. [Google Scholar]
- Debelian, G.; Trope, M. The use of premixed bioceramic materials in endodontics. G. Ital. Di Endod. 2016, 30, 70–80. [Google Scholar] [CrossRef]
- Zamparini, F.; Siboni, F.; Prati, C.; Taddei, P.; Gandolfi, M.G. Properties of calcium silicate-monobasic calcium phosphate materials for endodontics containing tantalum pentoxide and zirconium oxide. Clin. Oral. Investig. 2019, 23, 445–457. [Google Scholar] [CrossRef]
- Safi, C.; Kohli, M.R.; Kratchman, S.I.; Setzer, F.C.; Karabucak, B. Outcome of Endodontic Microsurgery Using Mineral Trioxide Aggregate or Root Repair Material as Root-end Filling Material: A Randomized Controlled Trial with Cone-beam Computed Tomographic Evaluation. J. Endod. 2019, 45, 831–839. [Google Scholar] [CrossRef]
- Kim, D.; Lee, H.; Chung, M.; Kim, S.; Song, M.; Kim, E. Effects of fast- and slow-setting calcium silicate-based root-end filling materials on the outcome of endodontic microsurgery: A retrospective study up to 6 years. Clin. Oral. Investig. 2020, 24, 247–255. [Google Scholar] [CrossRef]
- Silva Almeida, L.H.; Moraes, R.R.; Morgental, R.D.; Pappen, F.G. Are Premixed Calcium Silicate-based Endodontic Sealers Comparable to Conventional Materials? A Systematic Review of In Vitro Studies. J. Endod. 2017, 43, 527–535. [Google Scholar] [CrossRef]
- Ashofteh Yazdi, K.; Ghabraei, S.; Bolhari, B.; Kafili, M.; Meraji, N.; Nekoofar, M.H.; Dummer, P.M.H. Microstructure and chemical analysis of four calcium silicate-based cements in different environmental conditions. Clin. Oral. Investig. 2019, 23, 43–52. [Google Scholar] [CrossRef]
- Schembri Wismayer, P.; Lung, C.Y.; Rappa, F.; Cappello, F.; Camilleri, J. Assessment of the interaction of Portland cement-based materials with blood and tissue fluids using an animal model. Sci. Rep. 2016, 6, 34547. [Google Scholar] [CrossRef] [PubMed]
- Nekoofar, M.H.; Stone, D.F.; Dummer, P.M. The effect of blood contamination on the compressive strength and surface microstructure of mineral trioxide aggregate. Int. Endod. J. 2010, 43, 782–791. [Google Scholar] [CrossRef] [PubMed]
- Nekoofar, M.H.; Oloomi, K.; Sheykhrezae, M.S.; Tabor, R.; Stone, D.F.; Dummer, P.M. An evaluation of the effect of blood and human serum on the surface microhardness and surface microstructure of mineral trioxide aggregate. Int. Endod. J. 2010, 43, 849–858. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Kim, S.; Shin, Y.S.; Jung, I.Y.; Lee, S.J. Failure of setting of mineral trioxide aggregate in the presence of fetal bovine serum and its prevention. J. Endod. 2012, 38, 536–540. [Google Scholar] [CrossRef]
- Song, M.; Yue, W.; Kim, S.; Kim, W.; Kim, Y.; Kim, J.W.; Kim, E. The effect of human blood on the setting and surface micro-hardness of calcium silicate cements. Clin. Oral. Investig. 2016, 20, 1997–2005. [Google Scholar] [CrossRef]
- Song, W.; Li, S.; Tang, Q.; Chen, L.; Yuan, Z. In vitro biocompatibility and bioactivity of calcium silicate-based bioceramics in endodontics (Review). Int. J. Mol. Med. 2021, 48, 1–22. [Google Scholar] [CrossRef]
- Koutroulis, A.; Kuehne, S.A.; Cooper, P.R.; Camilleri, J. The role of calcium ion release on biocompatibility and antimicrobial properties of hydraulic cements. Sci. Rep. 2019, 9, 19019. [Google Scholar] [CrossRef]
- Kim, J.M.; Choi, S.; Kwack, K.H.; Kim, S.Y.; Lee, H.W.; Park, K. G protein-coupled calcium-sensing receptor is a crucial mediator of MTA-induced biological activities. Biomaterials 2017, 127, 107–116. [Google Scholar] [CrossRef]
- Yamamoto, S.; Han, L.; Noiri, Y.; Okiji, T. Evaluation of the Ca ion release, pH and surface apatite formation of a prototype tricalcium silicate cement. Int. Endod. J. 2017, 50 (Suppl. S2), e73–e82. [Google Scholar] [CrossRef]
- Bagur, R.; Hajnóczky, G. Intracellular Ca(2+) Sensing: Its Role in Calcium Homeostasis and Signaling. Mol. Cell. 2017, 66, 780–788. [Google Scholar] [CrossRef]
- Bellido, T.; Delgado-Calle, J. Ex Vivo Organ Cultures as Models to Study Bone Biology. JBMR Plus 2020, 4. [Google Scholar] [CrossRef]
- Verderio, P.; Lecchi, M.; Ciniselli, C.M.; Shishmani, B.; Apolone, G.; Manenti, G. 3Rs Principle and Legislative Decrees to Achieve High Standard of Animal Research. Animals 2023, 13, 277. [Google Scholar] [CrossRef]
- Hubrecht, R.C.; Carter, E. The 3Rs and Humane Experimental Technique: Implementing Change. Animals 2019, 9, 754. [Google Scholar] [CrossRef]
- Graham, M.L.; Prescott, M.J. The multifactorial role of the 3Rs in shifting the harm-benefit analysis in animal models of disease. Eur. J. Pharmacol. 2015, 759, 19–29. [Google Scholar] [CrossRef]
- Staines, K.A.; Brown, G.; Farquharson, C. The Ex Vivo Organ Culture of Bone. Methods Mol. Biol. 2019, 1914, 199–215. [Google Scholar] [CrossRef] [PubMed]
- Gomes, P.S.; Pinheiro, B.; Colaço, B.; Fernandes, M.H. The Osteogenic Assessment of Mineral Trioxide Aggregate-based Endodontic Sealers in an Organotypic Ex Vivo Bone Development Model. J. Endod. 2021, 47, 1461–1466. [Google Scholar] [CrossRef] [PubMed]
- Kanczler, J.M.; Smith, E.L.; Roberts, C.A.; Oreffo, R.O. A novel approach for studying the temporal modulation of embryonic skeletal development using organotypic bone cultures and microcomputed tomography. Tissue Eng. Part. C. Methods 2012, 18, 747–760. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.L.; Kanczler, J.M.; Oreffo, R.O. A new take on an old story: Chick limb organ culture for skeletal niche development and regenerative medicine evaluation. Eur. Cell. Mater. 2013, 26, 91–106; discussion 106. [Google Scholar] [CrossRef]
- Garbieri, T.F.; Martin, V.; Santos, C.F.; Gomes, P.S.; Fernandes, M.H. The Embryonic Chick Femur Organotypic Model as a Tool to Analyze the Angiotensin II Axis on Bone Tissue. Pharmaceuticals 2021, 14, 469. [Google Scholar] [CrossRef] [PubMed]
- Francisco, I.; Vale, F.; Martin, V.; Fernandes, M.H.; Gomes, P.S. From Blood to Bone-The Osteogenic Activity of L-PRF Membranes on the Ex Vivo Embryonic Chick Femur Development Model. Materials 2021, 14, 7830. [Google Scholar] [CrossRef] [PubMed]
- Araújo, R.; Martin, V.; Ferreira, R.; Fernandes, M.H.; Gomes, P.S. A new ex vivo model of the bone tissue response to the hyperglycemic environment—The embryonic chicken femur organotypic culture in high glucose conditions. Bone 2022, 158, 116355. [Google Scholar] [CrossRef]
- Kato, G.; Gomes, P.S.; Neppelenbroek, K.H.; Rodrigues, C.; Fernandes, M.H.; Grenho, L. Fast-Setting Calcium Silicate-Based Pulp Capping Cements—Integrated Antibacterial, Irritation and Cytocompatibility Assessment. Materials 2023, 16, 450. [Google Scholar] [CrossRef]
- Setbon, H.M.; Devaux, J.; Iserentant, A.; Leloup, G.; Leprince, J.G. Influence of composition on setting kinetics of new injectable and/or fast setting tricalcium silicate cements. Dent. Mater. 2014, 30, 1291–1303. [Google Scholar] [CrossRef]
- Chaudhari, P.S.; Chandak, M.G.; Jaiswal, A.A.; Mankar, N.P.; Paul, P. A Breakthrough in the Era of Calcium Silicate-Based Cements: A Critical Review. Cureus 2022, 14, e28562. [Google Scholar] [CrossRef]
- Kim, Y.; Lee, D.; Kye, M.; Ha, Y.J.; Kim, S.Y. Biocompatible Properties and Mineralization Potential of Premixed Calcium Silicate-Based Cements and Fast-Set Calcium Silicate-Based Cements on Human Bone Marrow-Derived Mesenchymal Stem Cells. Materials 2022, 15, 7595. [Google Scholar] [CrossRef]
- Lee, D.; Park, J.B.; Song, D.; Kim, H.M.; Kim, S.Y. Cytotoxicity and Mineralization Potential of Four Calcium Silicate-Based Cements on Human Gingiva-Derived Stem Cells. Coatings 2020, 10, 279. [Google Scholar] [CrossRef]
- Mestieri, L.B.; Gomes-Cornélio, A.L.; Rodrigues, E.M.; Salles, L.P.; Bosso-Martelo, R.; Guerreiro-Tanomaru, J.M.; Tanomaru-Filho, M. Biocompatibility and bioactivity of calcium silicate-based endodontic sealers in human dental pulp cells. J. Appl. Oral. Sci. 2015, 23, 467–471. [Google Scholar] [CrossRef] [PubMed]
- Cramer, E.E.A.; Ito, K.; Hofmann, S. Ex vivo Bone Models and Their Potential in Preclinical Evaluation. Curr. Osteoporos. Rep. 2021, 19, 75–87. [Google Scholar] [CrossRef]
- Kanczler, J.; Smith, E.; Roberts, C.; Oreffo, R. Temporal analysis of embryonic bone development using an innovative organotypic bone culture model-application of micro-computed tomography. In Proceedings of the Frontiers Endocrinology Conference Abstract: 2011 Joint Meeting of the Bone Research Society & the British Orthopaedic Research Society, Cambridge, UK, 29 June 2011. [Google Scholar]
- Smith, E.L.; Kanczler, J.M.; Gothard, D.; Roberts, C.A.; Wells, J.A.; White, L.J.; Qutachi, O.; Sawkins, M.J.; Peto, H.; Rashidi, H.; et al. Evaluation of skeletal tissue repair, Part 2: Enhancement of skeletal tissue repair through dual-growth-factor-releasing hydrogels within an ex vivo chick femur defect model. Acta Biomaterialia 2014, 10, 4197–4205. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.L.; Rashidi, H.; Kanczler, J.M.; Shakesheff, K.M.; Oreffo, R.O. The effects of 1α, 25-dihydroxyvitamin D3 and transforming growth factor-β3 on bone development in an ex vivo organotypic culture system of embryonic chick femora. PLoS ONE 2015, 10, e0121653. [Google Scholar] [CrossRef] [PubMed]
- Kot, K.; Kucharski, Ł.; Marek, E.; Safranow, K.; Lipski, M. Alkalizing Properties of Six Calcium-Silicate Endodontic Biomaterials. Materials 2022, 15, 6482. [Google Scholar] [CrossRef] [PubMed]
- Kahler, B.; Chugal, N.; Lin, L.M. Alkaline Materials and Regenerative Endodontics: A Review. Materials 2017, 10, 1389. [Google Scholar] [CrossRef] [PubMed]
- Arora, M. Cell culture media: A review. Mater. Methods 2013, 3, 24. [Google Scholar] [CrossRef]
- Vis, M.A.; Ito, K.; Hofmann, S. Impact of culture medium on cellular interactions in in vitro co-culture systems. Front. Bioeng. Biotechnol. 2020, 8, 911. [Google Scholar] [CrossRef] [PubMed]
- Talabani, R.M.; Garib, B.T.; Masaeli, R. Bioactivity and Physicochemical Properties of Three Calcium Silicate-Based Cements: An In Vitro Study. Biomed. Res. Int. 2020, 2020, 9576930. [Google Scholar] [CrossRef]
- Rathinam, E.; Govindarajan, S.; Rajasekharan, S.; Declercq, H.; Elewaut, D.; De Coster, P.; Martens, L.; Leybaert, L. The calcium dynamics of human dental pulp stem cells stimulated with tricalcium silicate-based cements determine their differentiation and mineralization outcome. Sci. Rep. 2021, 11, 645. [Google Scholar] [CrossRef]
- Kang, S. Mineralization-inducing potentials of calcium silicate-based pulp capping materials in human dental pulp cells. Yeungnam Univ. J. Med. 2020, 37, 217–225. [Google Scholar] [CrossRef]
- Zeid, S.; Alothmani, O.S.; Yousef, M.K. Biodentine and mineral trioxide aggregate: An analysis of solubility, pH changes and leaching elements. Life Sci. J. 2015, 12, 18–23. [Google Scholar]
- Hakki, S.S.; Bozkurt, B.S.; Ozcopur, B.; Gandolfi, M.G.; Prati, C.; Belli, S. The response of cementoblasts to calcium phosphate resin-based and calcium silicate-based commercial sealers. Int. Endod. J. 2013, 46, 242–252. [Google Scholar] [CrossRef]
- Gandolfi, M.G.; Siboni, F.; Primus, C.M.; Prati, C. Ion release, porosity, solubility, and bioactivity of MTA Plus tricalcium silicate. J. Endod. 2014, 40, 1632–1637. [Google Scholar] [CrossRef]
- Freedman, G. Chapter 8—Adhesion. In Contemporary Esthetic Dentistry; Freedman, G., Ed.; Mosby: Saint Louis, MO, USA, 2012; pp. 168–213. [Google Scholar]
- Braga, R.R.; About, I. How far do calcium release measurements properly reflect its multiple roles in dental tissue mineralization? Clin. Oral. Investig. 2019, 23, 501. [Google Scholar] [CrossRef] [PubMed]
- Marciano, M.A.; Garcia, R.B.; Cavenago, B.C.; Minotti, P.G.; Midena, R.Z.; Guimarães, B.M.; Ordinola-Zapata, R.; Duarte, M.A.H. Influence of bismuth oxide concentration on the pH level and biocompatibility of white Portland cement. J. Appl. Oral. Sci. 2014, 22, 268–273. [Google Scholar] [CrossRef] [PubMed]
- Gandolfi, M.G.; Spagnuolo, G.; Siboni, F.; Procino, A.; Rivieccio, V.; Pelliccioni, G.A.; Prati, C.; Rengo, S. Calcium silicate/calcium phosphate biphasic cements for vital pulp therapy: Chemical-physical properties and human pulp cells response. Clin. Oral. Investig. 2015, 19, 2075–2089. [Google Scholar] [CrossRef] [PubMed]
- Sanz, J.L.; Guerrero-Gironés, J.; Pecci-Lloret, M.P.; Pecci-Lloret, M.R.; Melo, M. Biological interactions between calcium silicate-based endodontic biomaterials and periodontal ligament stem cells: A systematic review of in vitro studies. Int. Endod. J. 2021, 54, 2025–2043. [Google Scholar] [CrossRef] [PubMed]
- Santiago, M.C.; Gomes-Cornélio, A.L.; de Oliveira, L.A.; Tanomaru-Filho, M.; Salles, L.P. Calcium silicate-based cements cause environmental stiffness and show diverse potential to induce osteogenesis in human osteoblastic cells. Sci. Rep. 2021, 11, 16784. [Google Scholar] [CrossRef]
- Taha, N.A.; Aboyounes, F.B.; Tamimi, Z.Z. Root-end microsurgery using a premixed tricalcium silicate putty as root-end filling material: A prospective study. Clin. Oral. Investig. 2021, 25, 311–317. [Google Scholar] [CrossRef]
- Chan, S.; Glickman, G.N.; Woodmansey, K.F.; He, J. Retrospective Analysis of Root-end Microsurgery Outcomes in a Postgraduate Program in Endodontics Using Calcium Silicate-based Cements as Root-end Filling Materials. J. Endod. 2020, 46, 345–351. [Google Scholar] [CrossRef]
- Sfeir, G.; Zogheib, C.; Patel, S.; Giraud, T.; Nagendrababu, V.; Bukiet, F. Calcium Silicate-Based Root Canal Sealers: A Narrative Review and Clinical Perspectives. Materials 2021, 14, 3965. [Google Scholar] [CrossRef]
- Primus, C.M.; Tay, F.R.; Niu, L.N. Bioactive tri/dicalcium silicate cements for treatment of pulpal and periapical tissues. Acta Biomater. 2019, 96, 35–54. [Google Scholar] [CrossRef]

| Material | Composition | Manufacturer | Batch No. |
|---|---|---|---|
| ProRoot® MTA | Tricalcium silicate, dicalcium silicate, tricalcium aluminate, and calcium sulfate. | Dentsply DeTrey GmbH, Germany | 0000301574 |
| BiodentineTM | Powder: tricalcium silicate, dicalcium silicate, calcium carbonate, iron oxide, zirconium oxide. Liquid: water with calcium chloride and soluble polymer (polycarboxylate). | Septodont, France | B27532 |
| TotalFill® BC RRMTM Fast Putty | Zirconium oxide, tantalum oxide, calcium silicate, calcium phosphate monobasic and fillers. | FKG Dentaire SA, Switzerland | 2100004308 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kato, G.; Araújo, R.; Rodrigues, C.; Gomes, P.S.; Grenho, L.; Fernandes, M.H. Ex Vivo Osteogenesis Induced by Calcium Silicate-Based Cement Extracts. J. Funct. Biomater. 2023, 14, 314. https://doi.org/10.3390/jfb14060314
Kato G, Araújo R, Rodrigues C, Gomes PS, Grenho L, Fernandes MH. Ex Vivo Osteogenesis Induced by Calcium Silicate-Based Cement Extracts. Journal of Functional Biomaterials. 2023; 14(6):314. https://doi.org/10.3390/jfb14060314
Chicago/Turabian StyleKato, Gabriel, Rita Araújo, Cláudia Rodrigues, Pedro Sousa Gomes, Liliana Grenho, and Maria Helena Fernandes. 2023. "Ex Vivo Osteogenesis Induced by Calcium Silicate-Based Cement Extracts" Journal of Functional Biomaterials 14, no. 6: 314. https://doi.org/10.3390/jfb14060314
APA StyleKato, G., Araújo, R., Rodrigues, C., Gomes, P. S., Grenho, L., & Fernandes, M. H. (2023). Ex Vivo Osteogenesis Induced by Calcium Silicate-Based Cement Extracts. Journal of Functional Biomaterials, 14(6), 314. https://doi.org/10.3390/jfb14060314









