Evaluation of the Antibacterial Properties of Iron Oxide, Polyethylene Glycol, and Gentamicin Conjugated Nanoparticles against Some Multidrug-Resistant Bacteria
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Preparation of Iron Oxide Nanoparticles (Fe3O4)
2.3. Coating PEG with Fe3O4 NPs
2.4. Loading of Antibiotic Gen
2.5. Preparation of the Gen-PEG-Fe3O4 NPs
2.6. Characterization of Fe3O4 NPs
2.7. Collection of Bacterial Isolates
2.8. Antibacterial Activity
2.9. Effect of Prepared Nanoparticles on Bacterial Biofilm by Tube Method
2.10. Determination of the MIC and MBC
2.11. Determination of Growth Curve
2.12. Statistical Analysis
3. Results and Discussion
3.1. Synthesis of Fe3O4 NPs
3.2. Characterization of Prepared Nanoparticles
3.2.1. FTIR Spectrometer
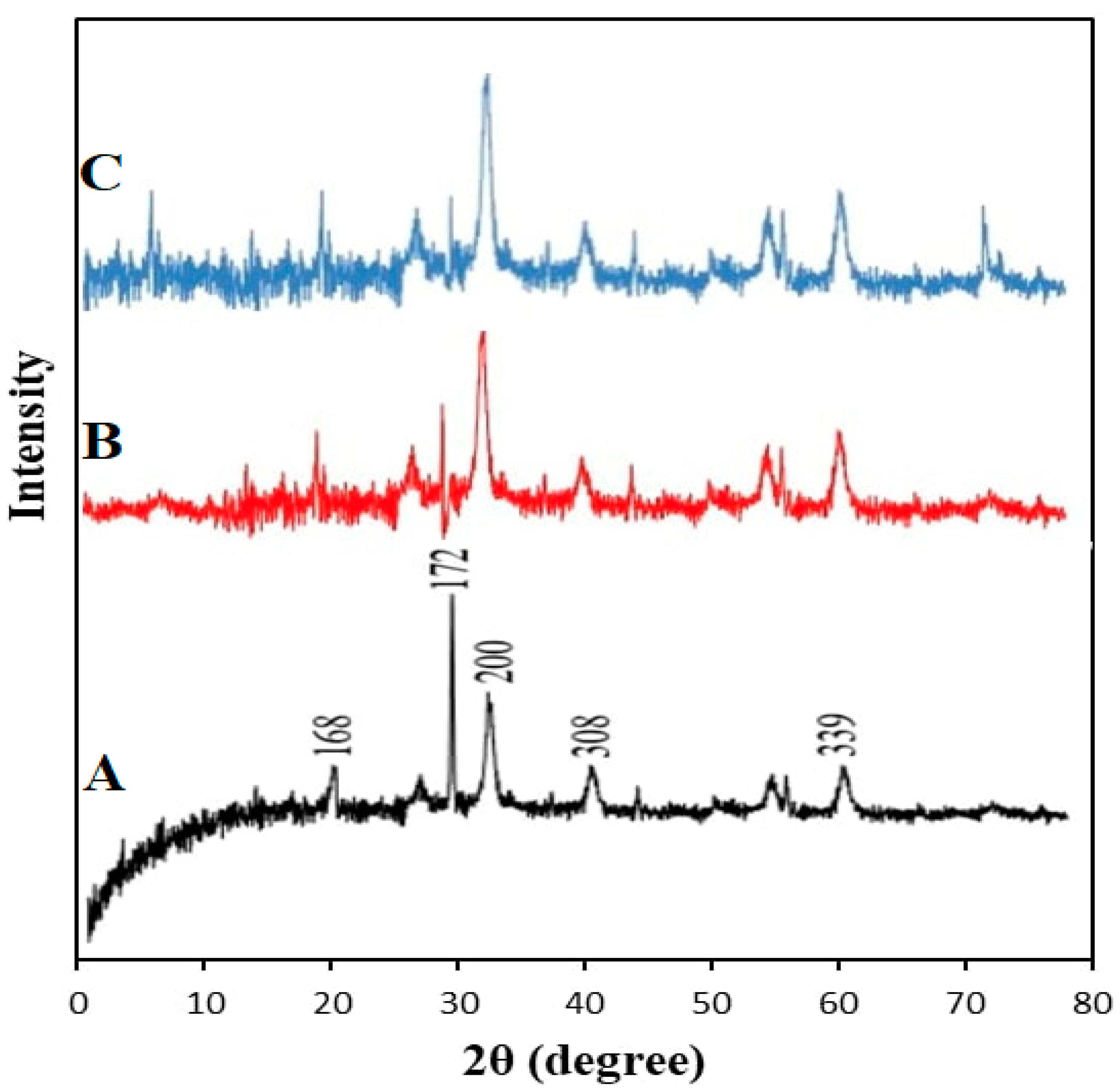
3.2.2. X-ray Diffraction
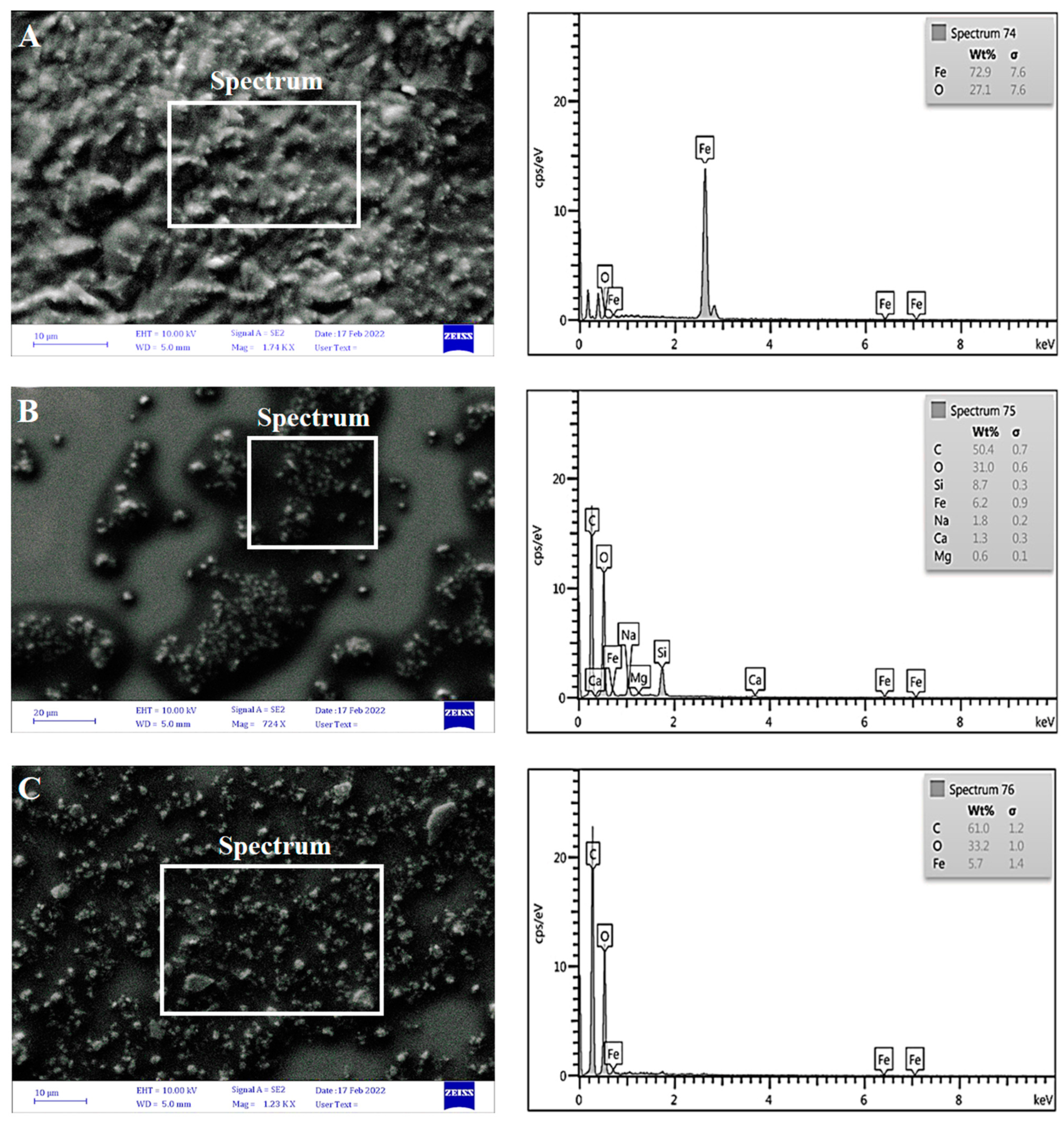
3.2.3. Scanning Electron Microscopy (SEM)
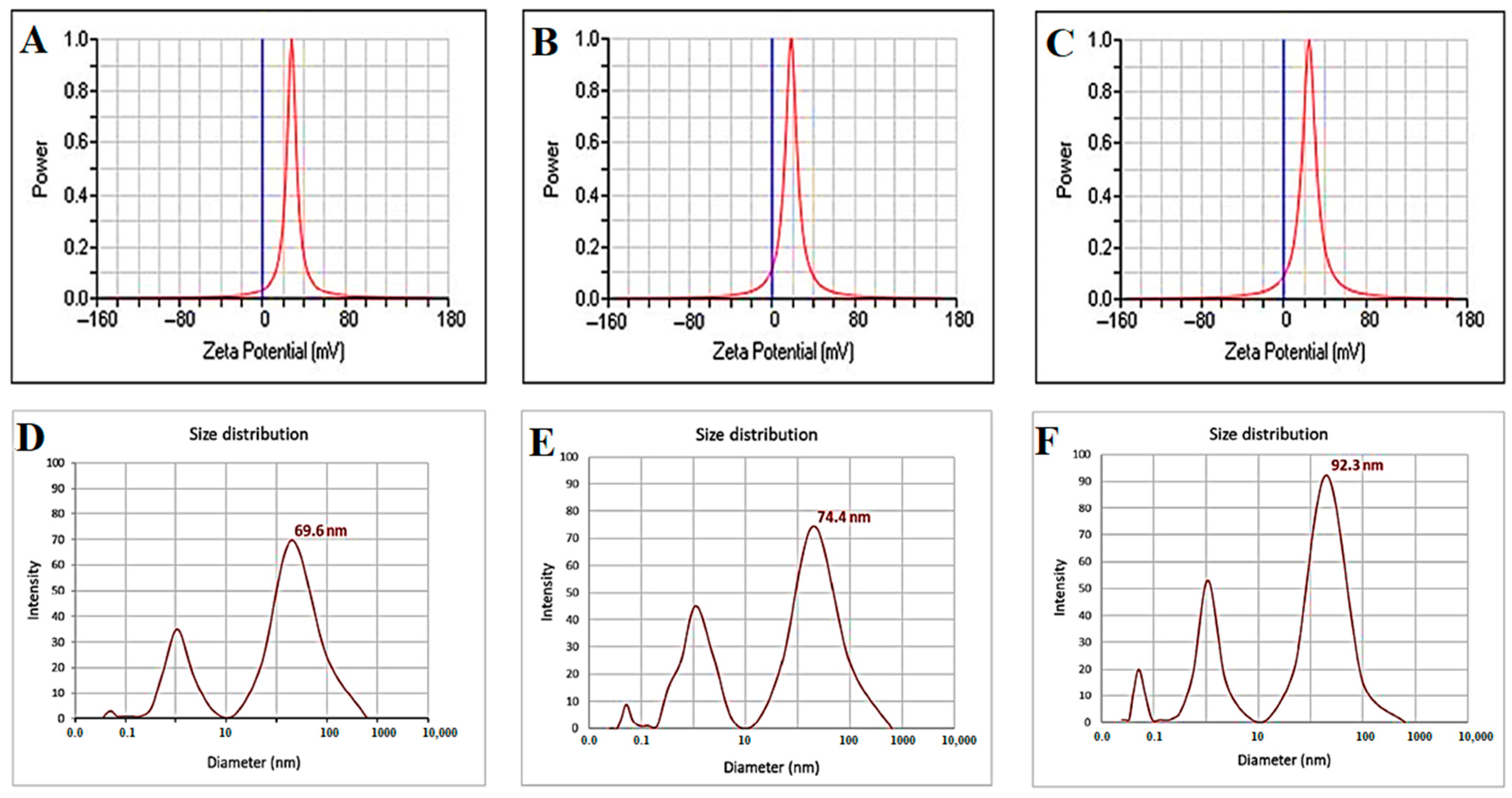
3.2.4. Zeta Potential Analysis and Average Size Distribution
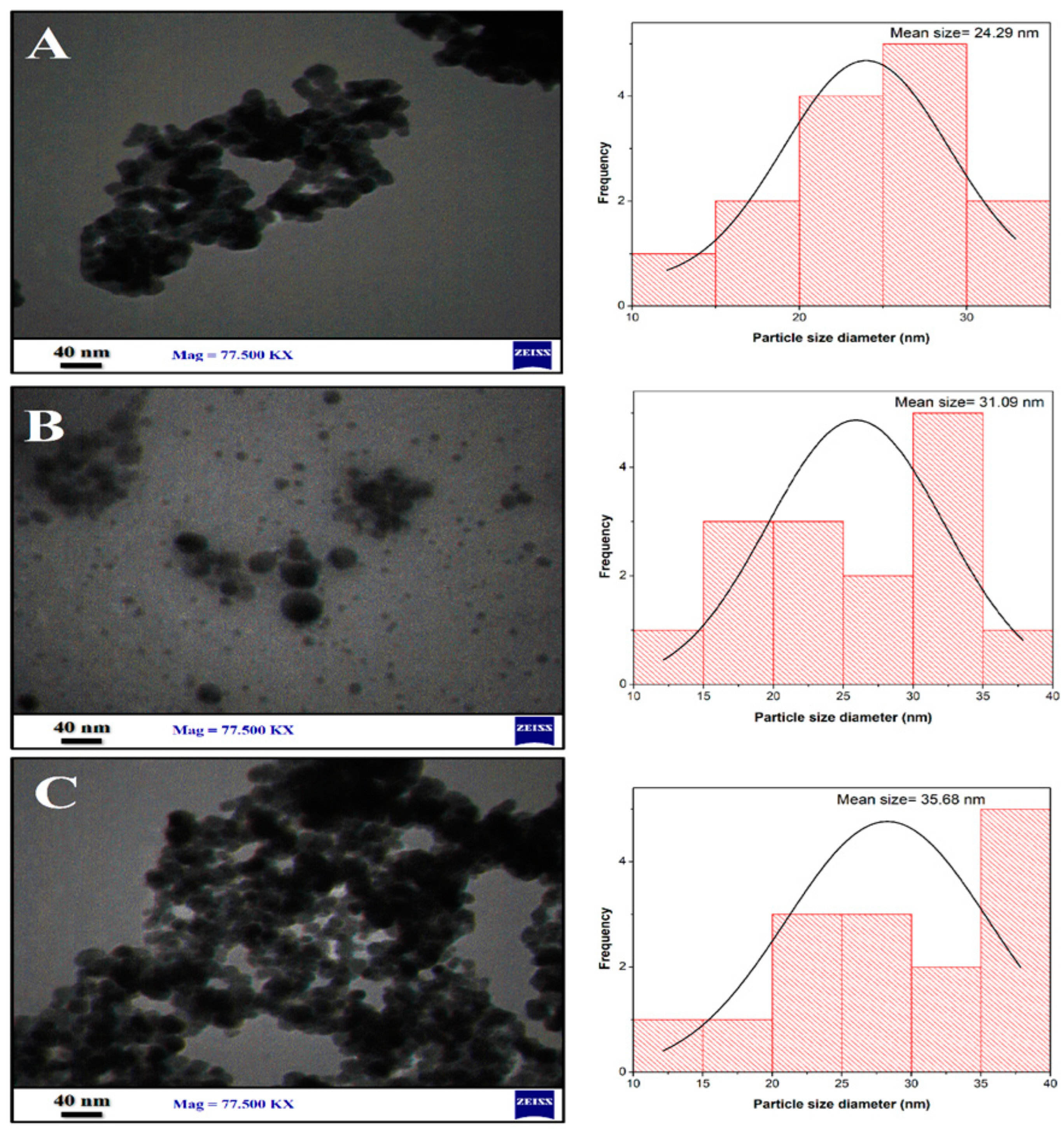
3.2.5. Transmission Electron Microscopy (TEM)
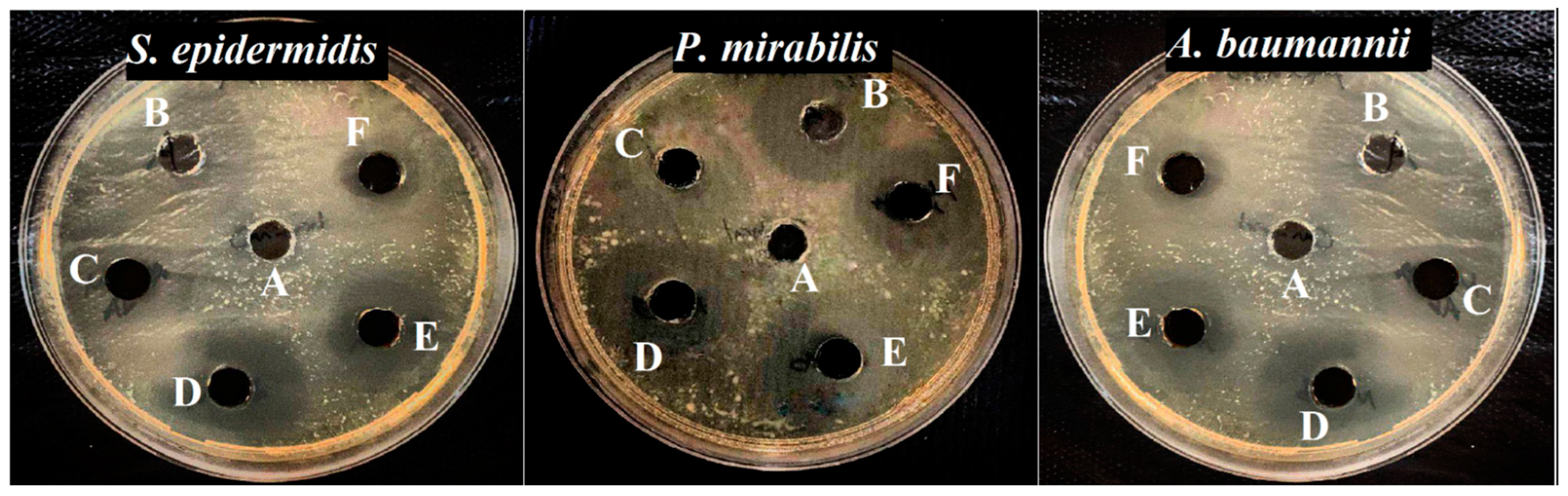
3.3. Antibacterial Activity
3.4. Effect of Fe3O4 NPs on Bacterial Biofilm by Tube Method
3.5. Determination of Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC) for Fe3O4 NPs
3.6. Determination of Growth Curve
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Weller, C.; Team, V. Interactive dressings and their role in moist wound management. In Advanced Textiles for Wound Care; Elsevier: Amsterdam, The Netherlands, 2019; pp. 105–134. [Google Scholar]
- Miguel, M.G.; Lourenço, J.P.; Faleiro, M.L. Superparamagnetic Iron Oxide Nanoparticles and Essential Oils: A New Tool for Biological Applications. Int. J. Mol. Sci. 2020, 28, 6633. [Google Scholar] [CrossRef] [PubMed]
- Dabiri, G.; Damstetter, E.; Phillips, T. Choosing a Wound Dressing Based on Common Wound Characteristics. Adv. Wound Care 2016, 5, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Presterl, E.; Schahawi, M.D.-E.; Lusignani, L.S.; Paula, H.; Reilly, J.S. Basic Principles and Introduction to Disinfectants and Antiseptics for Skin, Mucosa, and Wounds. In Basic Microbiology and Infection Control for Midwives; Springer: Berlin/Heidelberg, Germany, 2019; pp. 51–57. [Google Scholar] [CrossRef]
- Benítez, J.M.; Montáns, F.J. The mechanical behavior of skin: Structures and models for the finite element analysis. Comput. Struct. 2017, 190, 75–107. [Google Scholar] [CrossRef]
- Wang, F.; Fang, R.H.; Luk, B.T.; Hu, C.-M.J.; Thamphiwatana, S.; Dehaini, D.; Angsantikul, P.; Kroll, A.V.; Pang, Z.; Gao, W.; et al. Nanoparticle-Based Antivirulence Vaccine for the Management of Methicillin-Resistant Staphylococcus aureus Skin Infection. Adv. Funct. Mater. 2016, 26, 1628–1635. [Google Scholar] [CrossRef] [PubMed]
- Mihai, M.M.; Preda, M.; Lungu, I.; Gestal, M.C.; Popa, M.I.; Holban, A.M. Nanocoatings for Chronic Wound Repair—Modulation of Microbial Colonization and Biofilm Formation. Int. J. Mol. Sci. 2018, 19, 1179. [Google Scholar] [CrossRef]
- Mendoza, R.A.; Hsieh, J.-C.; Galiano, R.D. The Impact of Biofilm Formation on Wound Healing. Wound Heal. Curr. Perspect. 2019, 10, 235–250. [Google Scholar] [CrossRef]
- Aljghami, M.E.; Saboor, S.; Amini-Nik, S. Emerging Innovative Wound Dressings. Ann. Biomed. Eng. 2019, 47, 659–675. [Google Scholar] [CrossRef]
- Banin, E.; Hughes, D.; Kuipers, O.P. Editorial: Bacterial pathogens, antibiotics and antibiotic resistance. FEMS Microbiol. Rev. 2017, 41, 450–452. [Google Scholar] [CrossRef]
- Sorbiun, M.; Mehr, E.S.; Ramazani, A.; Malekzadeh, A.M. Biosynthesis of metallic nanoparticles using plant extracts and evaluation of their antibacterial properties. Nanochemistry Res. 2018, 1, 1–16. [Google Scholar] [CrossRef]
- Dinali, R.; Ebrahiminezhad, A.; Manley-Harris, M.; Ghasemi, Y.; Berenjian, A. Iron oxide nanoparticles in modern microbiology and biotechnology. Crit. Rev. Microbiol. 2017, 4, 493–507. [Google Scholar] [CrossRef]
- Jayanthi, S.A.; Sukanya, D.; Pragasam, A.J.; Sagayaraj, P. The influence of PEG 20,000 concentration on the size control and magnetic properties of functionalized bio-compatible magnetic nanoparticles. J. Med. Chem. Pharm. Chem. Pharm. Sci. 2013, 5, 90–102. [Google Scholar]
- Eskandari, M.J.; Hasanzadeh, I. Size-controlled synthesis of Fe3O4 magnetic nanoparticles via an alternating magnetic field and ultrasonic-assisted chemical co-precipitation. Mater. Sci. Eng. B 2021, 266, 115050. [Google Scholar] [CrossRef]
- Sundaresan, V.; Menon, J.U.; Rahimi, M.; Nguyen, K.T.; Wadajkar, A.S. Dual-responsive polymer-coated iron oxide nanoparticles for drug delivery and imaging applications. Int. J. Pharm. 2014, 466, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Gupta, M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 2005, 26, 3995–4021. [Google Scholar] [CrossRef] [PubMed]
- Sharifiaghdam, M.; Shaabani, E.; Asghari, F.; Faridi-Majidi, R. Chitosan coated metallic nanoparticles with stability, antioxidant, and antibacterial properties: Potential for wound healing application. J. Appl. Polym. Sci. 2021, 139, 51766. [Google Scholar] [CrossRef]
- Alven, S.; Buyana, B.; Feketshane, Z.; Aderibigbe, B.A. Electrospun Nanofibers/Nanofibrous Scaffolds Loaded with Silver Nanoparticles as Effective Antibacterial Wound Dressing Materials. Pharmaceutics 2021, 13, 964. [Google Scholar] [CrossRef]
- Yu, R.; Zhang, H.; Guo, B. Conductive Biomaterials as Bioactive Wound Dressing for Wound Healing and Skin Tissue Engineering. Nano-Micro Lett. 2022, 14, 1–46. [Google Scholar] [CrossRef]
- Chong, W.J.; Shen, S.; Li, Y.; Trinchi, A.; Pejak, D.; Kyratzis, I.L.; Sola, A.; Wen, C. Additive manufacturing of antibacterial PLA-ZnO nanocomposites: Benefits, limitations and open challenges. J. Mater. Sci. Technol. 2022, 111, 120–151. [Google Scholar] [CrossRef]
- Zhang, R.; Yu, B.; Tian, Y.; Pang, L.; Xu, T.; Cong, H.; Shen, Y. Diversified antibacterial modification and latest applications of polysaccharide-based hydrogels for wound healthcare. Appl. Mater. Today 2022, 26, 101396. [Google Scholar] [CrossRef]
- Yuan, Y.; Ding, L.; Chen, Y.; Chen, G.; Zhao, T.; Yu, Y. Nano-silver functionalized polysaccharides as a platform for wound dressings: A review. Int. J. Biol. Macromol. 2022, 194, 644–653. [Google Scholar] [CrossRef]
- Salvo, J.; Sandoval, C. Role of copper nanoparticles in wound healing for chronic wounds: Literature review. Burn. Trauma 2022, 10. [Google Scholar] [CrossRef] [PubMed]
- Kwiatkowska, A.; Drabik, M.; Lipko, A.; Grzeczkowicz, A.; Stachowiak, R.; Marszalik, A.; Granicka, L.H. Composite Membrane Dressings System with Metallic Nanoparticles as an Antibacterial Factor in Wound Healing. Membranes 2022, 12, 215. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.-S.; Kumar, S.R.; Ma, W.-T.; Shih, C.-M.; Teng, L.-W.; Yang, C.-C.; Lue, S.J. Gradiently distributed iron oxide@graphene oxide nanofillers in quaternized polyvinyl alcohol composite to enhance alkaline fuel cell power density. J. Membr. Sci. 2017, 543, 28–39. [Google Scholar] [CrossRef]
- Hussein, M.Z.; Al Ali, S.; Geilich, B.; El Zowalaty, M.; Webster, T.J. Synthesis, characterization, and antimicrobial activity of an ampicillin-conjugated magnetic nanoantibiotic for medical applications. Int. J. Nanomed. 2014, 9, 3801. [Google Scholar] [CrossRef]
- Mohsen, E.; El-Borady, O.M.; Mohamed, M.B.; Fahim, I.S. Synthesis and characterization of ciprofloxacin loaded silver nanoparticles and investigation of their antibacterial effect. J. Radiat. Res. Appl. Sci. 2020, 13, 416–425. [Google Scholar] [CrossRef]
- Mahmoodabadi, A.N.; Kompany, A.; Mashreghi, M. Characterization, antibacterial and cytotoxicity studies of graphene-Fe3O4 nanocomposites and Fe3O4 nanoparticles synthesized by a facile solvothermal method. Mater. Chem. Phys. 2018, 213, 285–294. [Google Scholar] [CrossRef]
- Pandya, S.R.; Singh, M. Preparation and characterization of magnetic nanoparticles and their impact on anticancer drug binding and release processes moderated through a 1st tier dendrimer. RSC Adv. 2016, 6, 37391–37402. [Google Scholar] [CrossRef]
- Anbarasu, M.; Anandan, M.; Chinnasamy, E.; Gopinath, V.; Balamurugan, K. Synthesis and characterization of polyethylene glycol (PEG) coated Fe3O4 nanoparticles by chemical co-precipitation method for biomedical applications. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 135, 536–539. [Google Scholar] [CrossRef]
- Bahjat, H.H.; Ismail, R.A.; Sulaiman, G.M.; Mohammed, H.A.; Al-Omar, M.; Mohammed, S.A.; Khan, R.A. Preparation of iron oxide and titania-based composite, core-shell populated, nanoparticulates material by two-step LASER ablation in aqueous media as antimicrobial and anticancer agents. Bioinorg. Chem. Appl. 2022, 2022, 1854473. [Google Scholar] [CrossRef]
- Ismail, R.A.; Sulaiman, G.M.; Abdulrahman, S.A.; Marzoog, T.R. Antibacterial activity of magnetic iron oxide nanoparticles synthesized by laser ablation in liquid. Mater. Sci. Eng. C 2015, 53, 286–297. [Google Scholar] [CrossRef]
- Álvarez, E.; Estévez, M.; Gallo-Cordova, A.; González, B.; Castillo, R.R.; Morales, M.D.P.; Colilla, M.; Izquierdo-Barba, I.; Vallet-Regí, M. Superparamagnetic Iron Oxide Nanoparticles Decorated Mesoporous Silica Nanosystem for Combined Antibiofilm Therapy. Pharmaceutics 2022, 14, 163. [Google Scholar] [CrossRef] [PubMed]
- Arias, L.S.; Pessan, J.P.; Vieira, A.P.M.; de Lima, T.M.T.; Delbem, A.C.B.; Monteiro, D.R. Iron Oxide Nanoparticles for Biomedical Applications: A Perspective on Synthesis, Drugs, Antimicrobial Activity, and Toxicity. Antibiotics 2018, 7, 46. [Google Scholar] [CrossRef] [PubMed]
- Ayanwale, A.P.; Reyes-López, S.Y. ZrO2–ZnO Nanoparticles as Antibacterial Agents. ACS Omega 2019, 4, 19216–19224. [Google Scholar] [CrossRef] [PubMed]
- El-Sigeny, S.M.; Abou Taleb, M.F. Synthesis, characterization, and application of dendrimer modified magnetite nanoparticles as antimicrobial agent. Life Sci. J. 2015, 12, 161–170. [Google Scholar]
- Sulaiman, G.M.; Tawfeeq, A.T.; Naji, A.S. Biosynthesis, characterization of magnetic iron oxide nanoparticles and evaluations of the cytotoxicity and DNA damage of human breast carcinoma cell lines. Artif. Cells Nanomed. Biotechnol. 2018, 46, 1215–1229. [Google Scholar] [CrossRef]
- Visan, A.I.; Popescu-Pelin, G.; Gherasim, O.; Grumezescu, V.; Socol, M.; Zgura, I.; Florica, C.; Popescu, R.C.; Savu, D.; Holban, A.M.; et al. Laser Processed Antimicrobial Nanocomposite Based on Polyaniline Grafted Lignin Loaded with Gentamicin-Functionalized Magnetite. Polymers 2019, 11, 283. [Google Scholar] [CrossRef]
- Chaki, S.H.; Malek, T.J.; Chaudhary, M.D.; Tailor, J.P.; Deshpande, M.P. Magnetite Fe3O4 nanoparticles synthesis by wet chemical reduction and their characterization. Adv. Nat. Sci. Nanosci. Nanotechnol. 2015, 6, 035009. [Google Scholar] [CrossRef]
- Miri, A.; Najafzadeh, H.; Darroudi, M.; Miri, M.J.; Kouhbanani, M.A.J.; Sarani, M. Iron Oxide Nanoparticles: Biosynthesis, Magnetic Behavior, Cytotoxic Effect. ChemistryOpen 2021, 3, 327–333. [Google Scholar] [CrossRef]
- Darwish, M.S. Effect of carriers on heating efficiency of oleic acid-stabilized magnetite nanoparticles. J. Mol. Liq. 2017, 231, 80–85. [Google Scholar] [CrossRef]
- Anuje, M.; Pawaskar, P.N.; Khot, V.; Sivan, A.; Jadhav, S.; Meshram, J.; Thombare, B. Synthesis, characterization, and cyto-toxicity evaluation of polyethylene glycol-coated iron oxide nanoparticles for radiotherapy application. J. Med. Physics 2021, 3, 154. [Google Scholar]
- Kędzierska, M.; Potemski, P.; Drabczyk, A.; Kudłacik-Kramarczyk, S.; Głąb, M.; Grabowska, B.; Mierzwiński, D.; Tyliszczak, B. The Synthesis Methodology of PEGylated Fe3O4@Ag Nanoparticles Supported by Their Physicochemical Evaluation. Molecules 2021, 26, 1744. [Google Scholar] [CrossRef] [PubMed]
- LaGrow, A.P.; Besenhard, M.O.; Hodzic, A.; Sergides, A.; Bogart, L.K.; Gavriilidis, A.; Thanh, N.T.K. Unravelling the growth mechanism of the co-precipitation of iron oxide nanoparticles with the aid of synchrotron X-Ray diffraction in solution. Nanoscale 2019, 14, 6620–6628. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karimzadeh, I.; Aghazadeh, M.; Doroudi, T.; Ganjali, M.R.; Kolivand, P.H. Superparamagnetic Iron Oxide (Fe3O4) Nanoparticles Coated with PEG/PEI for Biomedical Applications: A Facile and Scalable Preparation Route Based on the Cathodic Electrochemical Deposition Method. Adv. Phys. Chem. 2017, 2017, 1–7. [Google Scholar] [CrossRef]
- Predescu, A.M.; Matei, E.; Berbecaru, A.C.; Pantilimon, C.; Drăgan, C.; Vidu, R.; Predescu, C.; Kuncser, V. Synthesis and characterization of dextran-coated iron oxide nanoparticles. R. Soc. Open Sci. 2018, 3, 171525. [Google Scholar] [CrossRef]
- Minnici, K.; Kwon, Y.H.; Huie, M.M.; de Simon, M.V.; Zhang, B.; Bock, D.C.; Wang, J.; Wang, J.; Takeuchi, K.J.; Takeuchi, E.S.; et al. High capacity Li-ion battery anodes: Impact of crystallite size, surface chemistry and PEG-coating. Electrochim. Acta 2018, 260, 235–245. [Google Scholar] [CrossRef]
- Saqib, S.; Munis, M.F.H.; Zaman, W.; Ullah, F.; Shah, S.N.; Ayaz, A.; Farooq, M.; Bahadur, S. Synthesis, characterization and use of iron oxide nano particles for antibacterial activity. Microsc. Res. Tech. 2019, 82, 415–420. [Google Scholar] [CrossRef]
- Zein, I.; Jalil, Z. Identification of Magnetite Material (Fe3O4) Based on Natural Materials as Catalyst for Industrial Raw Material Application. J. Phys. Conf. Ser. 2019, 1, 012054. [Google Scholar] [CrossRef]
- Albukhaty, S.; Al-Musawi, S.; Mahdi, S.A.; Sulaiman, G.M.; Alwahibi, M.S.; Dewir, Y.H.; Soliman, D.; Rizwana, H. Investigation of Dextran-Coated Superparamagnetic Nanoparticles for Targeted Vinblastine Controlled Release, Delivery, Apoptosis Induction, and Gene Expression in Pancreatic Cancer Cells. Molecules 2020, 25, 4721. [Google Scholar] [CrossRef]
- Quevedo, I.R.; Olsson, A.L.; Clark, R.J.; Veinot, J.G.; Tufenkji, N. Interpreting Deposition Behavior of Polydisperse Surface-Modified Nanoparticles Using QCM-D and Sand-Packed Columns. Environ. Eng. Sci. 2014, 31, 326–337. [Google Scholar] [CrossRef]
- Ma, P.; Luo, Q.; Chen, J.; Gan, Y.; Du, J.; Ding, S.; Xi, Z.; Yang, X. Intraperitoneal injection of magnetic Fe3O4-nanoparticle induces hepatic and renal tissue injury via oxidative stress in mice. Int. J. Nanomed. 2012, 7, 4809–4818. [Google Scholar] [CrossRef]
- Nayek, C.; Manna, K.; Bhattacharjee, G.; Murugavel, P.; Obaidat, I. Investigating Size- and Temperature-Dependent Coercivity and Saturation Magnetization in PEG Coated Fe3O4 Nanoparticles. Magnetochemistry 2017, 3, 19. [Google Scholar] [CrossRef]
- Guo, L.; Chen, H.; He, N.; Deng, Y. Effects of surface modifications on the physicochemical properties of iron oxide nanoparticles and their performance as anticancer drug carriers. Chin. Chem. Lett. 2018, 12, 1829–1833. [Google Scholar] [CrossRef]
- Karimzadeh, I.; Dizaji, H.R.; Aghazadeh, M. Preparation, characterization and PEGylation of superparamagnetic Fe3O4nanoparticles from ethanol medium via cathodic electrochemical deposition (CED) method. Mater. Res. Express 2016, 3, 095022. [Google Scholar] [CrossRef]
- Harris, T.V.; Szilagyi, R.K. Iron–sulfur bond covalency from electronic structure calculations for classical iron–sulfur clusters. J. Comput. Chem. 2014, 35, 540–552. [Google Scholar] [CrossRef] [PubMed]
- Rufus, A.; Sreeju, N.; Philip, D. Synthesis of biogenic hematite (α-Fe2O3) nanoparticles for antibacterial and nanofluid applications. RSC Adv. 2016, 6, 94206–94217. [Google Scholar] [CrossRef]
- Li, W.; Wei, W.; Wu, X.; Zhao, Y.; Dai, H. The antibacterial and antibiofilm activities of mesoporous hollow Fe3O4 nanoparticles in an alternating magnetic field. Biomater. Sci. 2020, 8, 4492–4507. [Google Scholar] [CrossRef]
- Bhattacharya, P.; Neogi, S. Gentamicin coated iron oxide nanoparticles as novel antibacterial agents. Mater. Res. Express 2017, 4, 095005. [Google Scholar] [CrossRef]
- Safarkar, R.; Ebrahimzadeh Rajaei, G.; Khalili-Arjagi, S. The study of antibacterial properties of iron oxide nanoparticles synthesized using the extract of lichen Ramalina sinensis. Asian J. Nanosci. Mater. 2020, 3, 157–166. [Google Scholar]
- Wang, X.; Deng, A.; Cao, W.; Li, Q.; Wang, L.; Zhou, J.; Hu, B.; Xing, X. Synthesis of chitosan/poly (ethylene glycol)-modified magnetic nanoparticles for antibiotic delivery and their enhanced anti-biofilm activity in the presence of magnetic field. J. Mater. Sci. 2018, 9, 6433–6449. [Google Scholar] [CrossRef]
- Prabhu, Y.T.; Rao, K.V.; Kumari, B.S.; Kumar, V.S.S.; Pavani, T. Synthesis of Fe3O4 nanoparticles and its antibacterial application. Int. Nano Lett. 2015, 2, 85–92. [Google Scholar] [CrossRef]
- Gudkov, S.V.; Burmistrov, D.E.; Serov, D.A.; Rebezov, M.B.; Semenova, A.A.; Lisitsyn, A.B. Do Iron Oxide Nanoparticles Have Significant Antibacterial Properties? Antibiotics 2021, 7, 884. [Google Scholar] [CrossRef] [PubMed]
- Abdulla, A.A.; ALthahab, A.A.; Abed, T.A.; Mahdi, R.K.; Fadhil, S. Screening of virulence factors in Acintobacter baumannii isolated from clinical samples. Int. J. Curr. Res. Acad. Rev. 2015, 3, 128–134. [Google Scholar]
- Sharma, P.; Rana, S.; Barick, K.C.; Kumar, C.; Salunke, H.G.; Hassan, P.A. Biocompatible phosphate anchored Fe3O4 nanocarriers for drug delivery and hyperthermia. New J. Chem. 2014, 38, 5500–5508. [Google Scholar] [CrossRef]
- Shahzeidi, Z.S.; Amiri, G. Antibacterial activity of Fe. Int. J. Bio-Inorg. Hybrid Nanomater. 2015, 4, 135–140. [Google Scholar]
- Mahdavi, M.; Bin Ahmad, M.; Haron, M.J.; Namvar, F.; Nadi, B.; Ab Rahman, M.Z.; Amin, J. Synthesis, Surface Modification and Characterisation of Biocompatible Magnetic Iron Oxide Nanoparticles for Biomedical Applications. Molecules 2013, 18, 7533–7548. [Google Scholar] [CrossRef] [Green Version]
- Emamifar, A.; Kadivar, M.; Shahedi, M.; Soleimanian-Zad, S. Effect of nanocomposite packaging containing Ag and ZnO on inactivation of Lactobacillus plantarum in orange juice. Food Control 2011, 22, 408–413. [Google Scholar] [CrossRef]
- Sharaf, S.M.A.; Abbas, H.S.; Ismaeil, T.A.M. Characterization of spirugenic iron oxide nanoparticles and their antibacterial activity against multidrug-resistant Helicobacter pylori. Egypt. J. Phycol. 2019, 1, 1–28. [Google Scholar] [CrossRef]
- Kashmiri, Z.N.; Mankar, S.A. Free radicals and oxidative stress in bacteria. Int. J. Curr. Microbiol. Appl. Sci. 2014, 3, 34–40. [Google Scholar]


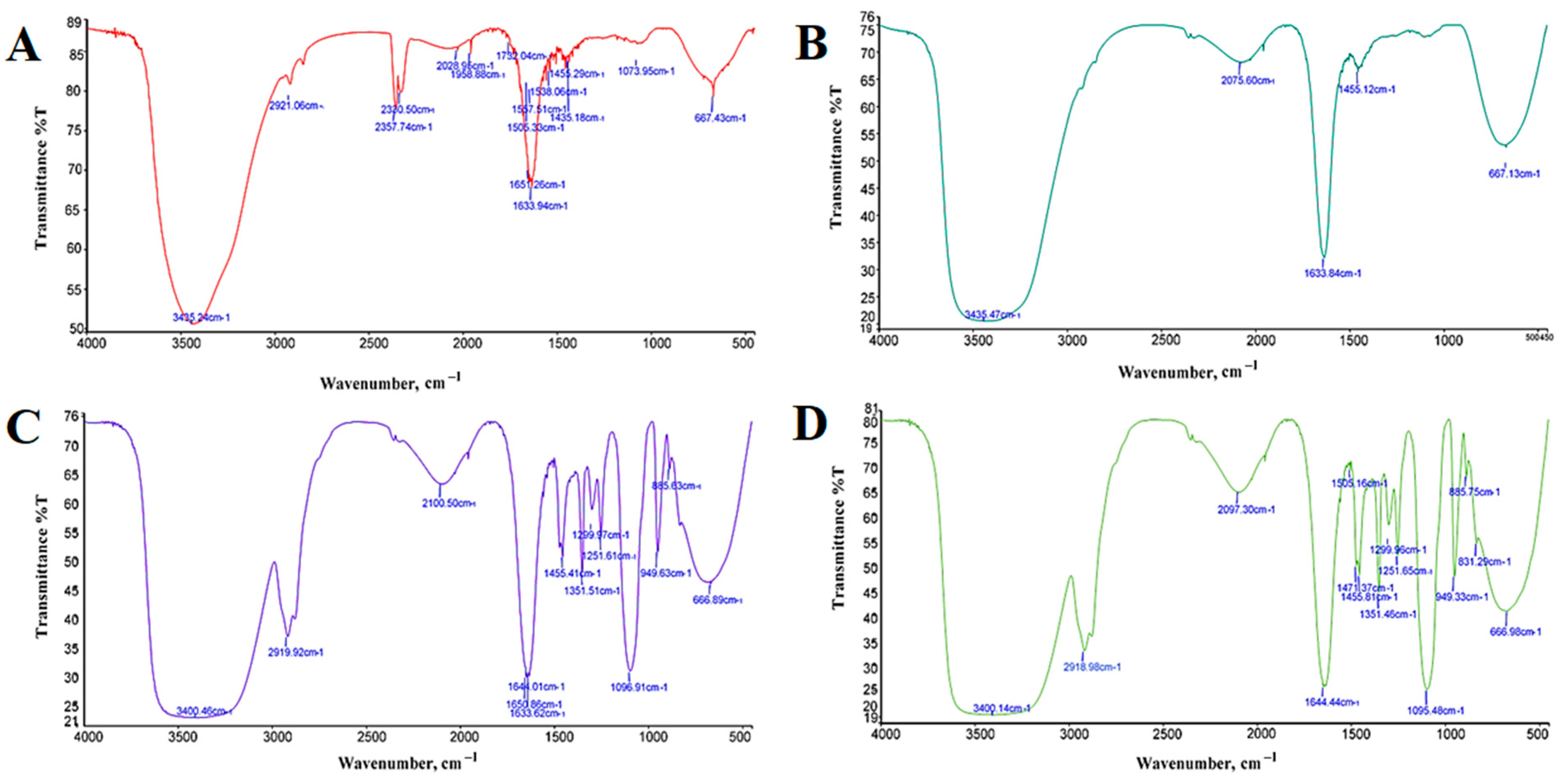
| Bond Type | Functional Group | Wavenumber (cm−1) | |||
|---|---|---|---|---|---|
| Fe3O4 NPs | Fe3O4 NPs+PEG | Fe3O4 NPs+Gen | Fe3O4 NPs+PEG+Gen | ||
| O-H | Hydroxyl | 3435.24 | 3400.46 | 3435.47 | 3400.14 |
| C-H | Stretching | - | 1455.41 | 1455.12 | 1471.37 |
| H-O-H | Bending | 1633.94 | 1650.86 | 1633.84 | 1644.44 |
| Fe-O | Ferrous Oxide | 667.43 | 666.89 | 667.13 | 666.98 |
| Bacterial Isolates | Inhibition Zone Diameter (mm) | ||||
|---|---|---|---|---|---|
| Gen | Fe3O4 NPs | Fe3O4 NPs+PEG | Fe3O4 NPs+Gen | Fe3O4 NPs+PEG+Gen | |
| S. epidermidis | 17.33 ± 0.57 | 17.66 ± 0.57 | 19.66 ± 0.57 | 21.33 ± 1.15 | 23.66 ± 0.57 |
| P. mirabilis | 17.51 ± 0.57 | 18.66 ± 0.57 | 21.66 ± 0.57 | 23.66 ± 0.57 | 25.66 ± 0.57 |
| A. baumannii | 17.66 ± 0.57 | 20.33 ± 0.57 | 22.00 ± 0.46 | 23.66 ± 0.57 | 26.33 ± 0.57 |
| Concentrations (µg mL−1) | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bacterial Isolates | Fe3O4 NPs | Fe3O4 NPs+PEG | Fe3O4 NPs+Gen | Fe3O4 NPs+PEG+Gen | ||||||||||||
| 100 | 50 | 25 | 12.5 | 100 | 50 | 25 | 12.5 | 100 | 50 | 25 | 12.5 | 100 | 50 | 25 | 12.5 | |
| S. epidermidis | − | + | + | + | − | + | + + | + + | − | − | + | + | − | − | − | − |
| P. mirabilis | − | + | + | + | − | + | + | + + | − | + | + | + | − | - | + | + |
| A. baumannii | − | + | + + | + + + | − | + | + + + | + + + | − | + | + | + + | − | + | + | + |
| Concentrations (µg mL−1) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Bacterial Isolates | A | B | C | D | ||||
| MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | |
| S. epidermidis | 25 | 50 | 25 | 50 | 50 | 100 | 50 | 100 |
| P. mirabilis | 50 | 100 | 50 | 100 | 25 | 50 | 25 | 50 |
| A. baumannii | 50 | 100 | 50 | 100 | 50 | 100 | 50 | 100 |
| Time (min) | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bacterial Isolates | Fe3O4 NPs | Fe3O4 NPs+PEG | Fe3O4 NPs+Gen | Fe3O4 NPs+PEG+Gen | ||||||||||||
| Zero | 30 | 60 | 90 | Zero | 30 | 60 | 90 | Zero | 30 | 60 | 90 | Zero | 30 | 60 | 90 | |
| S. epidermidis | + + + + + | + + + | + + | + | + + + + + | + + | + + + | + | + + + + + | + + + | + + | + | + + + + + | + + + | + + | + |
| P. mirabilis | + + + + + | + + + | + + | + | + + + + + | + + + | + + | + | + + + + + | + + + | + ++ | + | + + + + + | + + + | + + | + |
| A. baumannii | + + + + + | + + + | + + | + | + + + + + | + + + | + | + | + + + + + | + + + | + ++ | + | + + + + + | + + + | + + | + |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdulsada, F.M.; Hussein, N.N.; Sulaiman, G.M.; Al Ali, A.; Alhujaily, M. Evaluation of the Antibacterial Properties of Iron Oxide, Polyethylene Glycol, and Gentamicin Conjugated Nanoparticles against Some Multidrug-Resistant Bacteria. J. Funct. Biomater. 2022, 13, 138. https://doi.org/10.3390/jfb13030138
Abdulsada FM, Hussein NN, Sulaiman GM, Al Ali A, Alhujaily M. Evaluation of the Antibacterial Properties of Iron Oxide, Polyethylene Glycol, and Gentamicin Conjugated Nanoparticles against Some Multidrug-Resistant Bacteria. Journal of Functional Biomaterials. 2022; 13(3):138. https://doi.org/10.3390/jfb13030138
Chicago/Turabian StyleAbdulsada, Farah M., Nehia N. Hussein, Ghassan M. Sulaiman, Amer Al Ali, and Muhanad Alhujaily. 2022. "Evaluation of the Antibacterial Properties of Iron Oxide, Polyethylene Glycol, and Gentamicin Conjugated Nanoparticles against Some Multidrug-Resistant Bacteria" Journal of Functional Biomaterials 13, no. 3: 138. https://doi.org/10.3390/jfb13030138
APA StyleAbdulsada, F. M., Hussein, N. N., Sulaiman, G. M., Al Ali, A., & Alhujaily, M. (2022). Evaluation of the Antibacterial Properties of Iron Oxide, Polyethylene Glycol, and Gentamicin Conjugated Nanoparticles against Some Multidrug-Resistant Bacteria. Journal of Functional Biomaterials, 13(3), 138. https://doi.org/10.3390/jfb13030138








