Fixed Prosthetic Restorations and Periodontal Health: A Narrative Review
Abstract
1. Introduction
2. Materials and Method
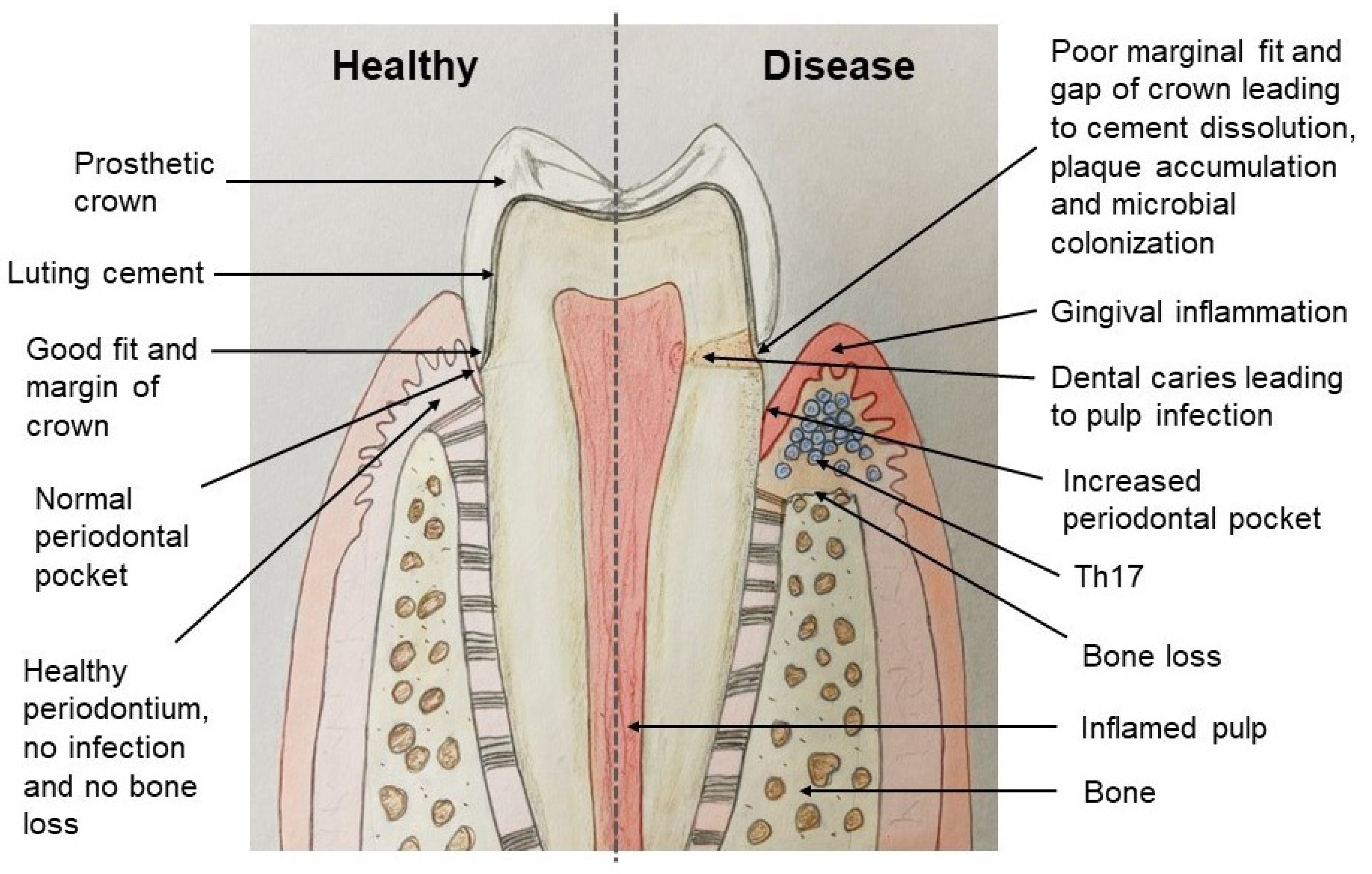
3. Marginal Fit and Internal Adaptation of Fixed Dental Prostheses
4. Biologic Width and Gingival Biotype Considerations in Fixed Prosthetic Restorations and Periodontal Health
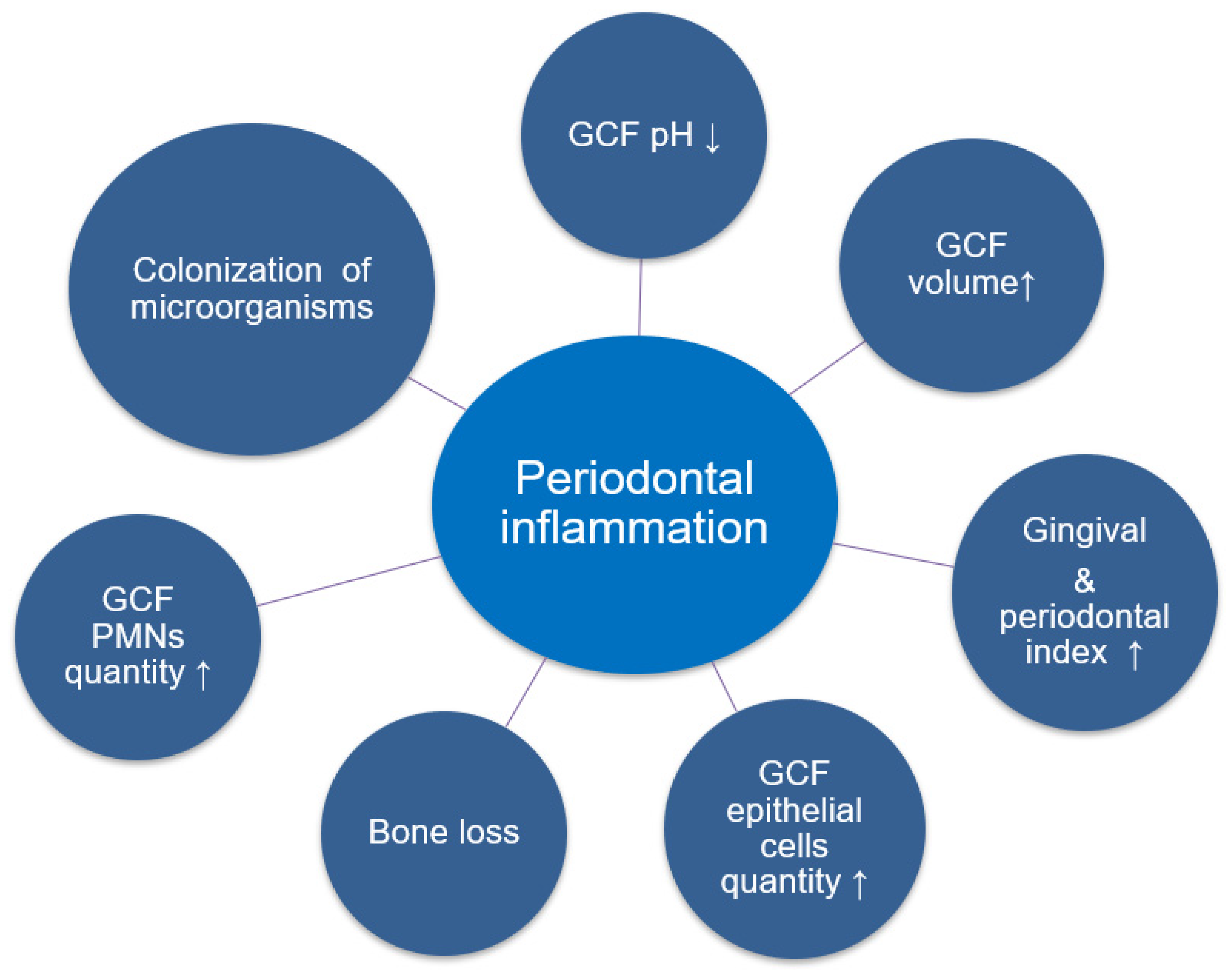
5. Gingival, Periodontal, Hygienic Indexes, and Clinical Manifestations
6. Cytomorphometric Analysis Following Fixed Dental Prosthesis
7. Salivary pH and GCF pH on Gingival and Periodontal Health
8. Bacteriological Evaluation Following Dental Prostheses
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Muddugangadhar, B.C.; Siddhi, T.; Suchismita, D. Prostho-perio-restorative interrelationship: A major junction. J. Adv. Oral Res. 2011, 2, 7–12. [Google Scholar] [CrossRef]
- Mojon, P.; Rentsch, A.; Budtz-Jørgensen, E. Relationship between prosthodontic status, caries, and periodontal disease in a geriatric population. Int. J. Prosthodont. 1995, 8, 564–571. [Google Scholar] [PubMed]
- Hsu, Y.-T.; Huang, N.; Wang, H.-L.; Kuo, Y.-w.; Chen, M.; Liu, T.-K.; Lin, H.-N.; Kuo, S.-L.; Juan, P.-K.; Liao, P.-B. Relationship between periodontics and prosthodontics: The two-way street. J. Prosthodont. Implantol. 2015, 4, 4–11. [Google Scholar]
- Avetisyan, A.; Markaryan, M.; Rokaya, D.; Tovani-Palone, M.R.; Zafar, M.S.; Khurshid, Z.; Vardanyan, A.; Heboyan, A. Characteristics of periodontal tissues in prosthetic treatment with fixed dental prostheses. Molecules 2021, 26, 1331. [Google Scholar] [CrossRef]
- Hao, Y.; Huang, X.; Zhou, X.; Li, M.; Ren, B.; Peng, X.; Cheng, L. Influence of dental prosthesis and restorative materials interface on oral biofilms. Int. J. Mol. Sci. 2018, 19, 3157. [Google Scholar] [CrossRef]
- Yin, X.J.; Wei, B.Y.; Ke, X.P.; Zhang, T.; Jiang, M.Y.; Luo, X.Y.; Sun, H.Q. Correlation between clinical parameters of crown and gingival morphology of anterior teeth and periodontal biotypes. BMC Oral Health 2020, 20, 59. [Google Scholar] [CrossRef]
- Kossioni, A.E.; Dontas, A.S. The stomatognathic system in the elderly. Useful information for the medical practitioner. Clin. Interv. Aging. 2007, 2, 591–597. [Google Scholar] [CrossRef]
- Pihlstrom, B.L.; Michalowicz, B.S.; Johnson, N.W. Periodontal diseases. Lancet 2005, 366, 1809–1820. [Google Scholar] [CrossRef]
- Nazir, M.A. Prevalence of periodontal disease, its association with systemic diseases and prevention. Int. J. Health Sci. 2017, 11, 72–80. [Google Scholar]
- Kornman, K.S.; Van Dyke, T.E. Bringing light to the heat: “Inflammation and periodontal diseases: A reappraisal”. J. Periodontol. 2008, 79, 1313. [Google Scholar] [CrossRef]
- Van Dyke, T.E.; Serhan, C.N. Resolution of inflammation: A new paradigm for the pathogenesis of periodontal diseases. J. Dent. Res. 2003, 82, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Vita, J.A. Endothelial function. Circulation 2011, 124, e906–e912. [Google Scholar] [CrossRef] [PubMed]
- Al-Odinee, N.M.; Al-Hamzi, M.; Al-Shami, I.Z.; Madfa, A.; Al-Kholani, A.I.; Al-Olofi, Y.M. Evaluation of the quality of fixed prosthesis impressions in private laboratories in a sample from yemen. BMC Oral Health 2020, 20, 304. [Google Scholar] [CrossRef]
- Knoernschild, K.L.; Campbell, S.D. Periodontal tissue responses after insertion of artificial crowns and fixed partial dentures. J. Prosthet. Dent. 2000, 84, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Heboyan, A. Marginal and internal fit of fixed prosthodontic constructions: A literature review. Int. J. Dent. Res. Rev. 2019, 2, 19. [Google Scholar] [CrossRef]
- Karataşli, O.; Kursoğlu, P.; Capa, N.; Kazazoğlu, E. Comparison of the marginal fit of different coping materials and designs produced by computer aided manufacturing systems. Dent. Mater. J. 2011, 30, 97–102. [Google Scholar] [CrossRef]
- Tan, P.L.; Gratton, D.G.; Diaz-Arnold, A.M.; Holmes, D.C. An in vitro comparison of vertical marginal gaps of cad/cam titanium and conventional cast restorations. J. Prosthodont. 2008, 17, 378–383. [Google Scholar] [CrossRef]
- Siadat, H.; Alikhasi, M.; Mirfazaelian, A.; Zade, M.M. Scanning electron microscope evaluation of vertical and horizontal discrepancy in cast copings for single-tooth implant-supported prostheses. Implant Dent. 2008, 17, 299–308. [Google Scholar] [CrossRef]
- Reich, S.; Gozdowski, S.; Trentzsch, L.; Frankenberger, R.; Lohbauer, U. Marginal fit of heat-pressed vs. Cad/cam processed all-ceramic onlays using a milling unit prototype. Oper. Dent. 2008, 33, 644–650. [Google Scholar] [CrossRef]
- Ushiwata, O.; de Moraes, J.V. Method for marginal measurements of restorations: Accessory device for toolmakers microscope. J. Prosthet. Dent. 2000, 83, 362–366. [Google Scholar] [CrossRef]
- Spagnuolo, G.; Desiderio, C.; Rivieccio, V.; Amato, M.; Rossetti, D.V.; D’Antò, V.; Schweikl, H.; Lupi, A.; Rengo, S.; Nocca, G. In vitro cellular detoxification of triethylene glycol dimethacrylate by adduct formation with N-acetylcysteine. Dent Mater. 2013, 29, e153-60. [Google Scholar] [CrossRef] [PubMed]
- Keshvad, A.; Hooshmand, T.; Asefzadeh, F.; Khalilinejad, F.; Alihemmati, M.; Van Noort, R. Marginal gap, internal fit, and fracture load of leucite-reinforced ceramic inlays fabricated by cerec inlab and hot-pressed techniques. J. Prosthodont. 2011, 20, 535–540. [Google Scholar] [CrossRef] [PubMed]
- Guess, P.C.; Vagkopoulou, T.; Zhang, Y.; Wolkewitz, M.; Strub, J.R. Marginal and internal fit of heat pressed versus cad/cam fabricated all-ceramic onlays after exposure to thermo-mechanical fatigue. J. Dent. 2014, 42, 199–209. [Google Scholar] [CrossRef] [PubMed]
- Riccitiello, F.; Amato, M.; Leone, R.; Spagnuolo, G.; Sorrentino, R. In vitro evaluation of the marginal fit and internal adaptation of zirconia and lithium disilicate single crowns: Micro-ct comparison between different manufacturing procedures. Open Dent. J. 2018, 12, 160–172. [Google Scholar] [CrossRef]
- Neves, F.D.; Prado, C.J.; Prudente, M.S.; Carneiro, T.A.; Zancopé, K.; Davi, L.R.; Mendonça, G.; Cooper, L.F.; Soares, C.J. Micro-computed tomography evaluation of marginal fit of lithium disilicate crowns fabricated by using chairside cad/cam systems or the heat-pressing technique. J. Prosthet. Dent. 2014, 112, 1134–1140. [Google Scholar] [CrossRef]
- Mously, H.A.; Finkelman, M.; Zandparsa, R.; Hirayama, H. Marginal and internal adaptation of ceramic crown restorations fabricated with cad/cam technology and the heat-press technique. J. Prosthet. Dent. 2014, 112, 249–256. [Google Scholar] [CrossRef]
- Freire, Y.; Gonzalo, E.; Lopez-Suarez, C.; Suarez, M.J. The marginal fit of cad/cam monolithic ceramic and metal-ceramic crowns. J. Prosthodont. 2019, 28, 299–304. [Google Scholar] [CrossRef]
- Ahrberg, D.; Lauer, H.C.; Ahrberg, M.; Weigl, P. Evaluation of fit and efficiency of cad/cam fabricated all-ceramic restorations based on direct and indirect digitalization: A double-blinded, randomized clinical trial. Clin. Oral Investig. 2016, 20, 291–300. [Google Scholar] [CrossRef]
- Abdullah, A.O.; Tsitrou, E.A.; Pollington, S. Comparative in vitro evaluation of cad/cam vs. conventional provisional crowns. J. Appl. Oral Sci. 2016, 24, 258–263. [Google Scholar] [CrossRef]
- Heboyan, A.G.; Movsisyan, N.M.; Khachatryan, V.A. Provisional restorations in restorative dentistry. World Sci. 2019, 3, 11–17. [Google Scholar]
- Sorrentino, R.; Navarra, C.O.; Di Lenarda, R.; Breschi, L.; Zarone, F.; Cadenaro, M.; Spagnuolo, G. Effects of finish line design and fatigue cyclic loading on phase transformation of zirconia dental ceramics: A qualitative micro-raman spectroscopic analysis. Materials 2019, 12, 863. [Google Scholar] [CrossRef] [PubMed]
- Heboyan, A.; Vardanyan, A.; Avetisyan, A. Cement selection in dental practice. World Sci. 2019, 2, 4–9. [Google Scholar]
- Diaz, P.I.; Chalmers, N.I.; Rickard, A.H.; Kong, C.; Milburn, C.L.; Palmer, R.J., Jr.; Kolenbrander, P.E. Molecular characterization of subject-specific oral microflora during initial colonization of enamel. Appl. Environ. Microbiol. 2006, 72, 2837–2848. [Google Scholar] [CrossRef] [PubMed]
- Pihlstrom, B.L. Periodontal risk assessment, diagnosis and treatment planning. Periodontol. 2000 2001, 25, 37–58. [Google Scholar] [CrossRef] [PubMed]
- Kazmi, S.M.R.; Iqbal, Z.; Muneer, M.U.; Riaz, S.; Zafar, M.S. Different pontic design for porcelain fused to metal fixed dental prosthesis: Contemporary guidelines and practice by general dental practitioners. Eur. J. Dent. 2018, 12, 375–379. [Google Scholar] [CrossRef] [PubMed]
- Rashid, H. How does fixed prosthodontics interface with periodontology? J. Res. Dent. 2015, 3, 96. [Google Scholar] [CrossRef]
- Goldberg, P.V.; Higginbottom, F.L.; Wilson, T.G. Periodontal considerations in restorative and implant therapy. Periodontol. 2000 2001, 25, 100–109. [Google Scholar] [CrossRef]
- Memari, Y.; Mohajerfar, M.; Armin, A.; Kamalian, F.; Rezayani, V.; Beyabanaki, E. Marginal adaptation of cad/cam all-ceramic crowns made by different impression methods: A literature review. J. Prosthodont. 2019, 28, e536–e544. [Google Scholar] [CrossRef]
- Tao, J.; Wu, Y.; Chen, J.; Su, J. A follow-up study of up to 5 years of metal-ceramic crowns in maxillary central incisors for different gingival biotypes. Int. J. Periodontics Restor. Dent. 2014, 34, e85–e92. [Google Scholar] [CrossRef]
- Yin, J.; Liu, D.; Huang, Y.; Wu, L.; Tang, X. Cad/cam techniques help in the rebuilding of ideal marginal gingiva contours of anterior maxillary teeth: A case report. J. Am. Dent. Assoc. 2017, 148, 834–839.e838. [Google Scholar] [CrossRef]
- Popa, D.; Bordea, I.R.; Burde, A.V.; Crisan, B.; Campian, R.S.; Constantiniuc, M. Surface modification of zirconia after laser irradiation. Optoelectron. Adv. Mater. Rapid Commun. 2016, 10, 785–788. [Google Scholar]
- Heboyan, A.; Manrikyan, M.; Markaryan, M.; Vardanyan, I. Changes in the parameters of gingival crevicular fluid in masticatory function restoration by various prosthodontic constructions. Int. J. Pharm. Sci. Res. 2020, 12, 2088–2093. [Google Scholar]
- Kois, J.C. The restorative-periodontal interface: Biological parameters. Periodontol. 2000 1996, 11, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Makigusa, K. Histologic comparison of biologic width around teeth versus implant: The effect on bone preservation. J. Implant. Reconstr. Dent. 2009, 1, 20–24. [Google Scholar]
- Nugala, B.; Kumar, B.S.; Sahitya, S.; Krishna, P.M. Biologic width and its importance in periodontal and restorative dentistry. J. Conserv. Dent. 2012, 15, 12–17. [Google Scholar] [CrossRef]
- Gargiulo, A.W.; Wentz, F.M.; Orban, B. Dimensions and relations of the dentogingival junction in humans. J. Periodontol. 1961, 32, 261–267. [Google Scholar] [CrossRef]
- Kina, J.R.; Dos Santos, P.H.; Kina, E.F.; Suzuki, T.Y.; Dos Santos, P.L. Periodontal and prosthetic biologic considerations to restore biological width in posterior teeth. J. Craniofac. Surg. 2011, 22, 1913–1916. [Google Scholar] [CrossRef]
- Doornewaard, R.; Bruyn, H.; Matthys, C.; Bronkhorst, E.; Vandeweghe, S.; Vervaeke, S. The long-term effect of adapting the vertical position of implants on peri-implant health: A 5-year intra-subject comparison in the edentulous mandible including oral health-related quality of life. J. Clin. Med. 2020, 9, 3320. [Google Scholar] [CrossRef]
- Nevins, M.; Skurow, H.M. The intracrevicular restorative margin, the biologic width, and the maintenance of the gingival margin. Int. J. Periodontics Restor. Dent. 1984, 4, 30–49. [Google Scholar]
- Reitemeier, B.; Hänsel, K.; Walter, M.H.; Kastner, C.; Toutenburg, H. Effect of posterior crown margin placement on gingival health. J. Prosthet. Dent. 2002, 87, 167–172. [Google Scholar] [CrossRef]
- Ercoli, C.; Caton, J.G. Dental prostheses and tooth-related factors. J. Periodontol. 2018, 89, S223–S236. [Google Scholar] [CrossRef] [PubMed]
- Olsson, M.; Lindhe, J. Periodontal characteristics in individuals with varying form of the upper central incisors. J. Clin. Periodontol. 1991, 18, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Dhir, S. The peri-implant esthetics: An unforgettable entity. J. Indian Soc. Periodontol. 2011, 15, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Kolte, R.; Kolte, A.; Mahajan, A. Assessment of gingival thickness with regards to age, gender and arch location. J. Indian Soc. Periodontol. 2014, 18, 478–481. [Google Scholar] [CrossRef]
- Zweers, J.; Thomas, R.Z.; Slot, D.E.; Weisgold, A.S.; Van der Weijden, F.G. Characteristics of periodontal biotype, its dimensions, associations and prevalence: A systematic review. J. Clin. Periodontol. 2014, 41, 958–971. [Google Scholar] [CrossRef]
- Nagaraj, K.R.; Savadi, R.C.; Savadi, A.R.; Prashanth Reddy, G.T.; Srilakshmi, J.; Dayalan, M.; John, J. Gingival biotype—Prosthodontic perspective. J. Indian Prosthodont. Soc. 2010, 10, 27–30. [Google Scholar] [CrossRef]
- Kan, J.Y.; Rungcharassaeng, K.; Morimoto, T.; Lozada, J. Facial gingival tissue stability after connective tissue graft with single immediate tooth replacement in the esthetic zone: Consecutive case report. J. Oral Maxillofac. Surg. 2009, 67, 40–48. [Google Scholar] [CrossRef]
- Shah, N.; Goyal, S. A study of neurocognitive and executive function of divers. J. Interdiscip. Dent. 2016, 6, 44–49. [Google Scholar] [CrossRef]
- Shah, D.; Duseja, S.; Vaishnav, K.; Shah, R. Adaptation of gingival biotype in response to prosthetic rehabilitation. Adv. Hum. Biol. 2017, 7, 85–88. [Google Scholar] [CrossRef]
- León-Martínez, R.; Montiel-Company, J.M.; Bellot-Arcís, C.; Solá-Ruíz, M.F.; Selva-Otaolaurruchi, E.; Agustín-Panadero, R. Periodontal behavior around teeth prepared with finishing line for restoration with fixed prostheses. A systematic review and meta-analysis. J. Clin. Med. 2020, 9, 249. [Google Scholar] [CrossRef]
- Agustín-Panadero, R.; Serra-Pastor, B.; Fons-Font, A.; Solá-Ruíz, M.F. Prospective clinical study of zirconia full-coverage restorations on teeth prepared with biologically oriented preparation technique on gingival health: Results after two-year follow-up. Oper. Dent. 2018, 43, 482–487. [Google Scholar] [CrossRef] [PubMed]
- Agustín-Panadero, R.; Solá-Ruíz, M.F. Vertical preparation for fixed prosthesis rehabilitation in the anterior sector. J. Prosthet. Dent. 2015, 114, 474–478. [Google Scholar] [CrossRef]
- Loi, I.; Di Felice, A. Biologically oriented preparation technique (bopt): A new approach for prosthetic restoration of periodontically healthy teeth. Eur. J. Esthet. Dent. Off. J. Eur. Acad. Esthet. Dent. 2013, 8, 10–23. [Google Scholar]
- Abduo, J.; Lyons, K.M. Interdisciplinary interface between fixed prosthodontics and periodontics. Periodontol. 2000 2017, 74, 40–62. [Google Scholar] [CrossRef] [PubMed]
- Cekici, A.; Kantarci, A.; Hasturk, H.; Van Dyke, T.E. Inflammatory and immune pathways in the pathogenesis of periodontal disease. Periodontol. 2000 2014, 64, 57–80. [Google Scholar] [CrossRef]
- Ercoli, C.; Caton, J.G. Dental prostheses and tooth-related factors. J. Clin. Periodontol. 2018, 45 (Suppl. S20), S207–S218. [Google Scholar] [CrossRef]
- Garlet, G.P. Destructive and protective roles of cytokines in periodontitis: A re-appraisal from host defense and tissue destruction viewpoints. J. Dent. Res. 2010, 89, 1349–1363. [Google Scholar] [CrossRef]
- Nakamura, K.; Kanno, T.; Milleding, P.; Ortengren, U. Zirconia as a dental implant abutment material: A systematic review. Int. J. Prosthodont. 2010, 23, 299–309. [Google Scholar]
- Zarone, F.; Di Mauro, M.I.; Spagnuolo, G.; Gherlone, E.; Sorrentino, R. Fourteen-year evaluation of posterior zirconia-based three-unit fixed dental prostheses: A prospective clinical study of all ceramic prosthesis. J. Dent. 2020, 101, 103419. [Google Scholar] [CrossRef]
- Xu, X.Y.; Zhang, Y.L.; Geng, F.H. Clinical efficacy and effects of cad/cam zirconia all-ceramic crown and metal-ceramic crown restoration on periodontal tissues. Shanghai Kou Qiang Yi Xue Shanghai J. Stomatol. 2017, 26, 331–335. [Google Scholar]
- Pabst, A.M.; Walter, C.; Grassmann, L.; Weyhrauch, M.; Brüllmann, D.D.; Ziebart, T.; Scheller, H.; Lehmann, K.M. Influence of cad/cam all-ceramic materials on cell viability, migration ability and adenylate kinase release of human gingival fibroblasts and oral keratinocytes. Clin. Oral Investig. 2014, 18, 1111–1118. [Google Scholar] [CrossRef] [PubMed]
- Wataha, J.C. Biocompatibility of dental casting alloys: A review. J. Prosthet. Dent. 2000, 83, 223–234. [Google Scholar] [CrossRef]
- Kc Basnyat, S.; Sapkota, B.; Shrestha, S. Oral hygiene and gingival health in patients with fixed prosthodontic appliances—A six month follow-up. Kathmandu Univ. Med. J. (KUMJ) 2015, 13, 328–332. [Google Scholar]
- McClanahan, S.F.; Bartizek, R.D.; Biesbrock, A.R. Identification and consequences of distinct löe-silness gingival index examiner styles for the clinical assessment of gingivitis. J. Periodontol. 2001, 72, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Bluma, E.; Vidzis, A.; Zigurs, G. The influence of fixed prostheses on periodontal health. Stomatologija 2016, 18, 112–121. [Google Scholar]
- Al-Sinaidi, A.; Preethanath, R.S. The effect of fixed partial dentures on periodontal status of abutment teeth. Saudi J. Dent. Res. 2014, 5, 104–108. [Google Scholar] [CrossRef]
- Heboyan, A.; Syed, A.U.Y.; Rokaya, D.; Cooper, P.R.; Manrikyan, M.; Markaryan, M. Cytomorphometric analysis of inflammation dynamics in the periodontium following the use of fixed dental prostheses. Molecules 2020, 25, 4650. [Google Scholar] [CrossRef]
- Rokaya, D.; Mahat, Y.; Sapkota, B.; Kc Basnyat, S. Full coverage crowns and resin-bonded bridge combination for missing mandibular anterior teeth. Kathmandu Univ. Med. J. (KUMJ) 2018, 16, 97–99. [Google Scholar]
- Cortelli, S.C.; Costa, F.O.; Rode, S.M.; Haas, A.N.; Andrade, A.K.P.; Pannuti, C.M.; Escobar, E.C.; Almeida, E.R.; Cortelli, J.R.; Pedrazzi, V. Mouthrinse recommendation for prosthodontic patients. Braz. Oral Res. 2014, 28, 1–9. [Google Scholar] [CrossRef]
- Gunsolley, J.C. Clinical efficacy of antimicrobial mouthrinses. J. Dent. 2010, 38 (Suppl. S1), S6–S10. [Google Scholar] [CrossRef]
- Vyas, T.; Bhatt, G.; Gaur, A.; Sharma, C.; Sharma, A.; Nagi, R. Chemical plaque control—A brief review. J. Family Med. Prim. Care. 2021, 10, 1562–1568. [Google Scholar] [CrossRef] [PubMed]
- Cecilia, E.C.; Myriam, A.K.; María, E.L. Cytological analysis of the periodontal pocket in patients with aggressive periodontitis and chronic periodontitis. Contemp. Clin. Dent. 2014, 5, 495–500. [Google Scholar] [CrossRef] [PubMed]
- Armitage, G.C. Analysis of gingival crevice fluid and risk of progression of periodontitis. Periodontol. 2000 2004, 34, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.; Zhang, Y.; Jagannathan, M.; Hasturk, H.; Kantarci, A.; Liu, H.; Van Dyke, T.E.; Ganley-Leal, L.M.; Nikolajczyk, B.S. B cells from periodontal disease patients express surface toll-like receptor 4. J. Leukoc. Biol. 2009, 85, 648–655. [Google Scholar] [CrossRef]
- Sigusch, B.; Eick, S.; Pfister, W.; Klinger, G.; Glockmann, E. Altered chemotactic behavior of crevicular pmns in different forms of periodontitis. J. Clin. Periodontol. 2001, 28, 162–167. [Google Scholar] [CrossRef]
- Hou, G.-L.; Hou, L.-T.; Weisgold, A. Survival rate of teeth with periodontally hopeless prognosis after therapies with intentional replantation and perioprosthetic procedures—A study of case series for 5–12 years. Clin. Exp. Dent. 2016, 2, 85–95. [Google Scholar] [CrossRef][Green Version]
- Moimaz, S.; Saliba, N.; Saliba, O.; Zina, L.; Bolonhez, M. Association between dental prosthesis and periodontal disease in a rural brazilian community. Braz. J. Oral Sci. 2016, 5, 1226–1231. [Google Scholar]
- Geurtsen, W. Biocompatibility of dental casting alloys. Crit. Rev. Oral Biol. Med. 2002, 13, 71–84. [Google Scholar] [CrossRef]
- Elshahawy, W.; Watanabe, I. Biocompatibility of dental alloys used in dental fixed prosthodontics. Tanta Dent. J. 2014, 11, 150–159. [Google Scholar] [CrossRef]
- Eliaz, N. Corrosion of metallic biomaterials: A review. Materials 2019, 12, 407. [Google Scholar] [CrossRef]
- Srimaneepong, V.; Rokaya, D.; Thunyakitpisal, P.; Qin, J.; Saengkiettiyut, K. Corrosion resistance of graphene oxide/silver coatings on ni-ti alloy and expression of il-6 and il-8 in human oral fibroblasts. Sci. Rep. 2020, 10, 3247. [Google Scholar] [CrossRef]
- Rokaya, D.; Srimaneepong, V.; Qin, J.; Thunyakitpisal, P.; Siraleartmukul, K. Surface adhesion properties and cytotoxicity of graphene oxide coatings and graphene oxide/silver nanocomposite coatings on biomedical niti alloy. Sci. Adv. Mater. 2019, 11, 1474–1487. [Google Scholar] [CrossRef]
- Rokaya, D.; Srimaneepong, V.; Qin, J.; Siraleartmukul, K.; Siriwongrungson, V. Graphene oxide/silver nanoparticle coating produced by electrophoretic deposition improved the mechanical and tribological properties of niti alloy for biomedical applications. J. Nanosci. Nanotechnol. 2019, 19, 3804–3810. [Google Scholar] [CrossRef]
- Gautam, C.; Joyner, J.; Gautam, A.; Rao, J.; Vajtai, R. Zirconia based dental ceramics: Structure, mechanical properties, biocompatibility and applications. Dalton Trans. 2016, 45, 19194–19215. [Google Scholar] [CrossRef]
- Mallineni, S.K.; Nuvvula, S.; Matinlinna, J.P.; Yiu, C.K.; King, N.M. Biocompatibility of various dental materials in contemporary dentistry: A narrative insight. J. Investig. Clin. Dent. 2013, 4, 9–19. [Google Scholar] [CrossRef]
- Shang, L.J.; Wu, Y.; Xu, Y.J. Effect of the cad/cam zirconia all-ceramic crown restoration on periodontal tissue. Chin. J. Tissue Eng. Res. 2014, 18, 4804–4809. [Google Scholar]
- Gupta, G. Gingival crevicular fluid as a periodontal diagnostic indicator- ii: Inflammatory mediators, host-response modifiers and chair side diagnostic aids. J. Med. Life 2013, 6, 7–13. [Google Scholar]
- Hasiuk, P.; Hasiuk, N.; Kindiy, D.; Ivanchyshyn, V.; Kalashnikov, D.; Zubchenko, S. Characteristics of cellular composition of periodontal pockets. Interv. Med. Appl. Sci. 2016, 8, 172–177. [Google Scholar] [CrossRef]
- Kantarci, A.; Oyaizu, K.; Van Dyke, T.E. Neutrophil-mediated tissue injury in periodontal disease pathogenesis: Findings from localized aggressive periodontitis. J. Periodontol. 2003, 74, 66–75. [Google Scholar] [CrossRef]
- Rizo-Gorrita, M.; Luna-Oliva, I.; Serrera-Figallo, M.-Á.; Gutiérrez-Pérez, J.-L.; Torres-Lagares, D. Comparison of cytomorphometry and early cell response of human gingival fibroblast (hgfs) between zirconium and new zirconia-reinforced lithium silicate ceramics (zls). Int. J. Mol. Sci. 2018, 19, 2718. [Google Scholar] [CrossRef]
- Bretz, W.A.; Weyant, R.J.; Corby, P.M.; Ren, D.; Weissfeld, L.; Kritchevsky, S.B.; Harris, T.; Kurella, M.; Satterfield, S.; Visser, M.; et al. Systemic inflammatory markers, periodontal diseases, and periodontal infections in an elderly population. J. Am. Geriatr. Soc. 2005, 53, 1532–1537. [Google Scholar] [CrossRef]
- Nair, S.; Faizuddin, M.; Dharmapalan, J. Role of autoimmune responses in periodontal disease. Autoimmune. Dis. 2014, 2014, 596824. [Google Scholar] [CrossRef]
- Van Dyke, T.E.; Kornman, K.S. Inflammation and factors that may regulate inflammatory response. J. Periodontol. 2008, 79, 1503–1507. [Google Scholar] [CrossRef]
- Andrews, T.; Sullivan, K.E. Infections in patients with inherited defects in phagocytic function. Clin. Microbiol. Rev. 2003, 16, 597–621. [Google Scholar] [CrossRef]
- Bosshardt, D.D.; Lang, N.P. The junctional epithelium: From health to disease. J. Dent. Res. 2005, 84, 9–20. [Google Scholar] [CrossRef]
- Delima, A.J.; Van Dyke, T.E. Origin and function of the cellular components in gingival crevice fluid. Periodontol. 2000 2003, 31, 55–76. [Google Scholar] [CrossRef]
- Bykov, V.L. Human gingival immunocompetent cells in the norm and in inflammatory periodontal diseases. Arkh. Patol. 2005, 67, 51–55. [Google Scholar]
- Biselli, R.; Ferlini, C.; Di Murro, C.; Paolantonio, M.; Fattorossi, A. Flow cytometric approach to human polymorphonuclear leukocyte activation induced by gingival crevicular fluid in periodontal disease. Inflammation 1995, 19, 479–487. [Google Scholar] [CrossRef]
- Gupta, G. Gingival crevicular fluid as a periodontal diagnostic indicator--i: Host derived enzymes and tissue breakdown products. J. Med. Life 2012, 5, 390–397. [Google Scholar]
- Ebersole, J.L. Humoral immune responses in gingival crevice fluid: Local and systemic implications. Periodontol. 2000 2003, 31, 135–166. [Google Scholar] [CrossRef]
- Baliga, S.; Muglikar, S.; Kale, R. Salivary ph: A diagnostic biomarker. J. Indian Soc. Periodontol. 2013, 17, 461–465. [Google Scholar] [CrossRef]
- Lynge Pedersen, A.M.; Belstrøm, D. The role of natural salivary defences in maintaining a healthy oral microbiota. J. Dent. 2019, 80, S3–S12. [Google Scholar] [CrossRef]
- Bickel, M.; Munoz, J.L.; Giovannini, P. Acid-base properties of human gingival crevicular fluid. J. Dent. Res. 1985, 64, 1218–1220. [Google Scholar] [CrossRef]
- Galgut, P.N. The relevance of ph to gingivitis and periodontitis. J. Int. Acad. Periodontol. 2001, 3, 61–67. [Google Scholar]
- Takahashi, N.; Saito, K.; Schachtele, C.F.; Yamada, T. Acid tolerance and acid-neutralizing activity of porphyromonas gingivalis, prevotella intermedia and fusobacterium nucleatum. Oral Microbiol. Immunol. 1997, 12, 323–328. [Google Scholar] [CrossRef]
- Takahashi, N.; Schachtele, C.F. Effect of ph on the growth and proteolytic activity of porphyromonas gingivalis and bacteroides intermedius. J. Dent. Res. 1990, 69, 1266–1269. [Google Scholar] [CrossRef]
- Smith, Q.T.; Au, G.S.; Freese, P.L.; Osborn, J.B.; Stoltenberg, J.L. Five parameters of gingival crevicular fluid from eight surfaces in periodontal health and disease. J. Periodontal. Res. 1992, 27, 466–475. [Google Scholar] [CrossRef]
- Khurshid, Z.; Mali, M.; Naseem, M.; Najeeb, S.; Zafar, M.S. Human gingival crevicular fluids (gcf) proteomics: An overview. Dent. J. 2017, 5, 12. [Google Scholar] [CrossRef]
- Fatima, T.; Khurshid, Z.; Rehman, A.; Imran, E.; Srivastava, K.C.; Shrivastava, D. Gingival crevicular fluid (gcf): A diagnostic tool for the detection of periodontal health and diseases. Molecules 2021, 26, 1208. [Google Scholar]
- Khurshid, Z.; Warsi, I.; Moin, S.F.; Slowey, P.D.; Latif, M.; Zohaib, S.; Zafar, M.S. Biochemical analysis of oral fluids for disease detection. Adv. Clin. Chem. 2021, 100, 205–253. [Google Scholar]
- Costantini, E.; Sinjari, B.; Piscopo, F.; Porreca, A.; Reale, M.; Caputi, S.; Murmura, G. Evaluation of salivary cytokines and vitamin d levels in periodontopathic patients. Int. J. Mol. Sci. 2020, 21, 2669. [Google Scholar] [CrossRef]
- Embery, G.; Waddington, R. Gingival crevicular fluid: Biomarkers of periodontal tissue activity. Adv. Dent. Res. 1994, 8, 329–336. [Google Scholar] [CrossRef]
- Ercoli, C.; Tarnow, D.; Poggio, C.E.; Tsigarida, A.; Ferrari, M.; Caton, J.G.; Chochlidakis, K. The relationships between tooth-supported fixed dental prostheses and restorations and the periodontium. J. Prosthodont. 2021, 30, 305–317. [Google Scholar] [CrossRef]
- Heboyan, A.; Manrikyan, M.; Zafar, M.S.; Rokaya, D.; Nushikyan, R.; Vardanyan, I.; Vardanyan, A.; Khurshid, Z. Bacteriological evaluation of gingival crevicular fluid in teeth restored using fixed dental prostheses: An in vivo study. Int. J. Mol. Sci. 2021, 22, 5463. [Google Scholar] [CrossRef]
- Said, H.S.; Suda, W.; Nakagome, S.; Chinen, H.; Oshima, K.; Kim, S.; Kimura, R.; Iraha, A.; Ishida, H.; Fujita, J.; et al. Dysbiosis of salivary microbiota in inflammatory bowel disease and its association with oral immunological biomarkers. DNA Res. 2014, 21, 15–25. [Google Scholar] [CrossRef]
- Xie, H.; Cook, G.S.; Costerton, J.W.; Bruce, G.; Rose, T.M.; Lamont, R.J. Intergeneric communication in dental plaque biofilms. J. Bacteriol. 2000, 182, 7067–7069. [Google Scholar] [CrossRef]
- Periasamy, S.; Kolenbrander, P.E. Mutualistic biofilm communities develop with porphyromonas gingivalis and initial, early, and late colonizers of enamel. J. Bacteriol. 2009, 191, 6804–6811. [Google Scholar] [CrossRef]
- Elmanfi, S.; Zhou, J.; Sintim, H.O.; Könönen, E.; Gürsoy, M.; Gürsoy, U.K. Regulation of gingival epithelial cytokine response by bacterial cyclic dinucleotides. J. Oral Microbiol. 2019, 11, 1538927. [Google Scholar] [CrossRef]
- Holt, S.C.; Kesavalu, L.; Walker, S.; Genco, C.A. Virulence factors of porphyromonas gingivalis. Periodontol. 2000 1999, 20, 168–238. [Google Scholar] [CrossRef]
- Benakanakere, M.; Kinane, D.F. Innate cellular responses to the periodontal biofilm. Front. Oral Biol. 2012, 15, 41–55. [Google Scholar]
- Holt, S.C.; Ebersole, J.L. Porphyromonas gingivalis, treponema denticola, and tannerella forsythia: The “red complex”, a prototype polybacterial pathogenic consortium in periodontitis. Periodontol. 2000 2005, 38, 72–122. [Google Scholar] [CrossRef] [PubMed]
- Willis, J.R.; Gabaldón, T. The human oral microbiome in health and disease: From sequences to ecosystems. Microorganisms 2020, 8, 308. [Google Scholar] [CrossRef] [PubMed]
- Pei, J.; Li, F.; Xie, Y.; Liu, J.; Yu, T.; Feng, X. Microbial and metabolomic analysis of gingival crevicular fluid in general chronic periodontitis patients: Lessons for a predictive, preventive, and personalized medical approach. EPMA J. 2020, 11, 197–215. [Google Scholar] [CrossRef]
- Patini, R.; Gallenzi, P.; Spagnuolo, G.; Cordaro, M.; Cantiani, M.; Amalfitano, A.; Arcovito, A.; Callà, C.; Mingrone, G.; Nocca, G. Correlation between metabolic syndrome, periodontitis and reactive oxygen species production. A pilot study. Open Dent. J. 2017, 11, 621–627. [Google Scholar] [CrossRef] [PubMed]
- Cafiero, C.; Spagnuolo, G.; Marenzi, G.; Martuscelli, R.; Colamaio, M.; Leuci, S. Predictive periodontitis: The most promising salivary biomarkers for early diagnosis of periodontitis. J. Clin. Med. 2021, 10, 1488. [Google Scholar] [CrossRef]
- Del Giudice, C.; Vaia, E.; Liccardo, D.; Marzano, F.; Valletta, A.; Spagnuolo, G.; Ferrara, N.; Rengo, C.; Cannavo, A.; Rengo, G. Infective endocarditis: A focus on oral microbiota. Microorganisms 2021, 9, 1218. [Google Scholar] [CrossRef]
- Könönen, E.; Gursoy, M.; Gursoy, U.K. Periodontitis: A multifaceted disease of tooth-supporting tissues. J. Clin. Med. 2019, 8, 1135. [Google Scholar] [CrossRef]
- Socransky, S.S.; Haffajee, A.D.; Cugini, M.A.; Smith, C.; Kent, R.L., Jr. Microbial complexes in subgingival plaque. J. Clin. Periodontol. 1998, 25, 134–144. [Google Scholar] [CrossRef]
- Souza, J.C.; Mota, R.R.; Sordi, M.B.; Passoni, B.B.; Benfatti, C.A.; Magini, R.S. Biofilm formation on different materials used in oral rehabilitation. Braz. Dent. J. 2016, 27, 141–147. [Google Scholar] [CrossRef]
- Sinjari, B.; Murmura, G.; Caputi, S.; Ricci, L.; Varvara, G.; Scarano, A. Use of oral chroma™ in the assessment of volatile sulfur compounds in patients with fixed protheses. Int. J. Immunopathol. Pharmacol. 2013, 26, 691–697. [Google Scholar] [CrossRef]
- Alauddin, M.S.; Baharuddin, A.S.; Mohd Ghazali, M.I. The modern and digital transformation of oral health care: A mini review. Healthcare 2021, 9, 118. [Google Scholar] [CrossRef] [PubMed]
- Humagain, M.; Rokaya, D. Integrating digital technologies in dentistry to enhance the clinical success. Kathmandu Univ. Med. J. (KUMJ) 2019, 17, 256–257. [Google Scholar]
- Schwendicke, F. Digital dentistry: Advances and challenges. J. Clin. Med. 2020, 9, 4005. [Google Scholar] [CrossRef] [PubMed]
- Amornvit, P.; Rokaya, D.; Peampring, C.; Sanohkan, S. Confocal 3d optical intraoral scanners and comparison of image capturing accuracy. Comput. Mater. Contin. 2021, 66, 303–314. [Google Scholar] [CrossRef]
- Amornvit, P.; Rokaya, D.; Sanohkan, S. Comparison of accuracy of current ten intraoral scanners. BioMed Res. Int. 2021, 2021, 2673040. [Google Scholar] [CrossRef]
- Tian, Y.; Chen, C.; Xu, X.; Wang, J.; Hou, X.; Li, K.; Lu, X.; Shi, H.; Lee, E.-S.; Jiang, H.B. A review of 3d printing in dentistry: Technologies, affecting factors, and applications. Scanning 2021, 2021, 9950131. [Google Scholar] [CrossRef]
- Dawood, A.; Marti Marti, B.; Sauret-Jackson, V.; Darwood, A. 3d printing in dentistry. Br. Dent. J. 2015, 219, 521–529. [Google Scholar] [CrossRef]
- Schweiger, J.; Edelhoff, D.; Güth, J.F. 3d printing in digital prosthetic dentistry: An overview of recent developments in additive manufacturing. J. Clin. Med. 2021, 10, 2010. [Google Scholar] [CrossRef]
- Pillai, S.; Upadhyay, A.; Khayambashi, P.; Farooq, I.; Sabri, H.; Tarar, M.; Lee, K.T.; Harb, I.; Zhou, S.; Wang, Y.; et al. Dental 3d-printing: Transferring art from the laboratories to the clinics. Polymers 2021, 13, 157. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Srimaneepong, V.; Heboyan, A.; Zafar, M.S.; Khurshid, Z.; Marya, A.; Fernandes, G.V.O.; Rokaya, D. Fixed Prosthetic Restorations and Periodontal Health: A Narrative Review. J. Funct. Biomater. 2022, 13, 15. https://doi.org/10.3390/jfb13010015
Srimaneepong V, Heboyan A, Zafar MS, Khurshid Z, Marya A, Fernandes GVO, Rokaya D. Fixed Prosthetic Restorations and Periodontal Health: A Narrative Review. Journal of Functional Biomaterials. 2022; 13(1):15. https://doi.org/10.3390/jfb13010015
Chicago/Turabian StyleSrimaneepong, Viritpon, Artak Heboyan, Muhammad Sohail Zafar, Zohaib Khurshid, Anand Marya, Gustavo V. O. Fernandes, and Dinesh Rokaya. 2022. "Fixed Prosthetic Restorations and Periodontal Health: A Narrative Review" Journal of Functional Biomaterials 13, no. 1: 15. https://doi.org/10.3390/jfb13010015
APA StyleSrimaneepong, V., Heboyan, A., Zafar, M. S., Khurshid, Z., Marya, A., Fernandes, G. V. O., & Rokaya, D. (2022). Fixed Prosthetic Restorations and Periodontal Health: A Narrative Review. Journal of Functional Biomaterials, 13(1), 15. https://doi.org/10.3390/jfb13010015










