Evaluation of Clinical Performance of TiNi-Based Implants Used in Chest Wall Repair after Resection for Malignant Tumors
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- David, E.A.; Marshall, M.B. Review of chest wall tumors: A diagnostic, therapeutic, and reconstructive challenge. Semin. Plast. Surg. 2011, 25, 16–24. [Google Scholar] [CrossRef]
- Zarqane, H.; Viala, P.; Dallaudiere, B.; Vernhet, H.; Cyteva, C.; Larbi, A. Tumors of the rib. Diagn. Interv. Imaging 2013, 94, 1095–1108. [Google Scholar] [CrossRef] [PubMed]
- Pfannschmidt, J.; Geisbusch, P.; Muley, T.; Hoffmann, H.; Dienemann, H. Surgical resection of secondary chest wall tumors. Thorac. Cardiovasc. Surg. 2005, 53, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Ferrigno, P.; Monaci, N.; Pangoni, A.; Comacchio, G.; Natale, G.; Faccioli, E.; Zuin, A.; Dell’Amore, A.; Rea, F. Extensive abdominal and chest wall resection and reconstruction for invasive squamous cell carcinoma of the skin. J. Thorac. Dis. 2020, 12, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Carter, B.; Gladish, G. MR imaging of chest wall tumors. Magn. Reson. Imaging Clin. N. Am. 2015, 23, 197–215. [Google Scholar] [CrossRef]
- Kress, R.; Dalwadi, S.; Irani, A. R0 resection and reconstruction for a large, rapidly progressive chest wall sarcoma. J. Cardiothorac. Surg. 2018, 13, 127. [Google Scholar] [CrossRef] [PubMed]
- Loi, M.; Mazzella, A.; Desideri, I.; Fournel, L.; Hamelin, E.; Icard, P.; Bobbio, A.; Alifano, M. Chest wall resection and reconstruction for lung cancer: Surgical techniques and example of integrated multimodality approach. J. Thorac. Dis. 2020, 12, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Merritt, R. Chest wall reconstruction without prosthetic material. Thorac. Surg. Clin. 2017, 27, 165–169. [Google Scholar] [CrossRef]
- Marulli, G.; De Iaco, G.; Ferrigno, P.; De Palma, A.; Quercia, R.; Brascia, D.; Schiavon, M.; Mammana, M.; Rea, F. Sternochondral replacement: Use of cadaveric allograft for the reconstruction of ante-rior chest wall. J. Thorac. Dis. 2020, 12, 3–9. [Google Scholar] [CrossRef]
- Sandri, A.; Donati, G.; Blanc, C.; Nigra, V.; Gagliasso, M.; Barmasse, R. Anterior chest wall resection and sternal body wedge for primary chest wall tumor: Recon-struction technique with biological meshes and titanium plates. J. Thorac. Dis. 2020, 12, 17–21. [Google Scholar] [CrossRef] [PubMed]
- Sanna, S.; Brandolini, J.; Pardolesi, A.; Argnani, D.; Mengozzi, M.; Dell’Amore, A.; Solli, P. Materials and techniques in chest wall reconstruction: A review. J. Vis. Surg. 2017, 3, 95. [Google Scholar] [CrossRef] [PubMed]
- Seder, C.; Rocco, G. Chest wall reconstruction after extended resection. J. Thorac. Dis. 2016, 8, S863–S871. [Google Scholar] [CrossRef] [PubMed]
- Gao, E.; Li, Y.; Zhao, T.; Guo, X.; He, W.; Wu, W.; Zhao, Y.; Yang, Y. Reconstruction of anterior chest wall: A clinical analysis. J. Cardiothorac. Surg. 2018, 13, 124. [Google Scholar] [CrossRef] [PubMed]
- Aranda, J.; Jimenez, M.; Rodriguez, M.; Varela, G. Tridimensional titanium-printed custom-made prosthesis for sternocostal re-construction. Eur. J. Cardiothorac. Surg. 2015, 48, e92–e94. [Google Scholar] [CrossRef]
- Dzian, A.; Zivcak, J.; Penciak, R.; Hudak, R. Implantation of a 3D-printed titanium sternum in a patient with a sternal tumor. World J. Surg. Oncol. 2018, 16, 1315–1318. [Google Scholar] [CrossRef]
- Wen, X.; Gao, S.; Feng, J.; Li, S.; Gao, R.; Zhang, G. Chest-wall reconstruction with a customized titanium-alloy prosthesis fabricated by 3D printing and rapid prototyping. J. Cardiothorac. Surg. 2018, 13, 4. [Google Scholar] [CrossRef]
- Zhang, H.; Zhao, J.; Li, X.; Huang, L.; Wang, L. Necessity of pleura repair in the chest wall reconstruction with three-dimensional printed tita-nium implant. J. Thorac. Dis. 2020, 12, 2713–2716. [Google Scholar] [CrossRef]
- Weyant, M.; Bains, M.; Venkatraman, E.; Downey, R.; Park, B.; Flores, R.; Rizk, N.; Rusch, V. Results of chest wall resection and reconstruction with and without rigid prosthesis. Ann. Thorac. Surg. 2006, 81, 279–285. [Google Scholar] [CrossRef]
- Bille, A.; Okiror, L.; Karenovics, W.; Routledge, T. Experience with titanium devices for rib fixation and coverage of chest wall defects. Interact. Cardiovasc. Thorac. Surg. 2012, 15, 588–595. [Google Scholar] [CrossRef]
- Berthet, J.; Canaud, L.; D’Annoville, T.; Alric, P.; Marty-Ane, C. Titanium plates and Dualmesh: A modern combination for reconstructing very large chest wall defects. Ann. Thorac. Surg. 2011, 91, 1709–1716. [Google Scholar] [CrossRef]
- Ong, K.; Ong, C.; Chua, Y.; Fazuludeen, A.; Ahmed, A. The painless combination of anatomically contoured titanium plates and porcine dermal collagen patch for chest wall reconstruction. J. Thorac. Dis. 2018, 10, 2890–2897. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Xiong, S.; Chen, Y.; Zhao, F.; Hu, Y.; Guo, Y.; Wu, B.; Huang, P.; Yang, B. Effects of statherin on the biological properties of titanium metals subjected to different surface modification. Colloids Surf. B Biointerfaces 2020, 188, 110783. [Google Scholar] [CrossRef] [PubMed]
- Van Oirschot, B.; Meijer, G.; Bronkhorst, E.; Narhi, T.; Jansen, J.; Van den Beucken, J. Comparison of different surface modifications for titanium implants installed into the goat iliac crest. Clin. Oral Implant. Res. 2014, 27, e57–e67. [Google Scholar] [CrossRef] [PubMed]
- Muhamedov, M.; Kulbakin, D.; Gunther, V.; Choynzonov, E.; Chekalkin, T.; Hodorenko, V. Sparing surgery with the use of TiNi-based endografts in larynx cancer pa-tients. J. Surg. Oncol. 2015, 111, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Shtin, V.; Novikov, V.; Chekalkin, T.; Gunther, V.; Marchenko, E.; Choynzonov, E.; Kang, S.B.; Chang, M.J.; Kang, J.H.; Obrosov, A. Repair of orbital post-traumatic wall defects by custom-made TiNi mesh endografts. J. Funct. Biomater. 2019, 10, 27. [Google Scholar] [CrossRef] [PubMed]
- Chernyshova, A.; Kolomiets, L.; Chekalkin, T.; Chernov, V.; Sinilkin, I.; Gunther, V.; Marchenko, E.; Baigonakova, G.; Kang, J.H. Fertility-sparing surgery using knitted TiNi mesh implants and sentinel lymph nodes: A 10-year experience. J. Investig. Surg. 2021, 34, 1110–1118. [Google Scholar] [CrossRef] [PubMed]
- Kulbakin, D.; Chekalkin, T.; Muhamedov, M.; Choynzonov, E.; Kang, J.H.; Kang, S.B.; Gunther, V. Sparing surgery for the successful treatment of thyroid papillary carcinoma invading the trachea: A case report. Case Rep. Oncol. 2016, 9, 772–780. [Google Scholar] [CrossRef]
- Gunther, V.; Radkevich, A.; Kang, S.B.; Chekalkin, T.; Marchenko, E.; Gunther, S.; Pulikov, A.; Sinuk, I.; Kaunietis, S.; Podgorniy, V.; et al. Study of the knitted TiNi mesh graft in a rabbit cranioplasty model. Biomed. Phys. Eng. Express 2019, 5, 027005. [Google Scholar] [CrossRef]
- Yasenchuk, Y.; Marchenko, E.; Gunther, V.; Radkevich, A.; Kokorev, O.; Gunther, S.; Baigonakova, G.; Hodorenko, V.; Chekalkin, T.; Kang, J.H.; et al. Biocompatibility and clinical application of porous TiNi alloys made by self-propagating high-temperature synthesis (SHS). Materials 2019, 12, 2405. [Google Scholar] [CrossRef]
- Yasenchuk, Y.; Marchenko, E.; Baigonakova, G.; Gunther, S.; Kokorev, O.; Gunter, V.; Chekalkin, T.; Topolnitskiy, E.; Obrosov, A.; Kang, J.H. Study on tensile, bending, fatigue, and in vivo behavior of porous SHS-TiNi alloy used as a bone substitute. Biomed. Mater. 2021, 16, 021001. [Google Scholar] [CrossRef]
- Gunther, V.; Yasenchuk, Y.; Chekalkin, T.; Marchenko, E.; Gunther, S.; Baigonakova, G.; Hodorenko, V.; Kang, J.H.; Weiss, S.; Obrosov, A. Formation of pores and amorphous-nanocrystalline phases in porous TiNi alloys made by self-propagating high-temperature synthesis (SHS). Adv. Powder Technol. 2019, 30, 673–680. [Google Scholar] [CrossRef]
- Yasenchuk, Y.; Gunther, V.; Marchenko, E.; Chekalkin, T.; Baigonakova, G.; Hodorenko, V.; Gunther, S.; Kang, J.H.; Weiss, S.; Obrosov, A. Formation of mineral phases in self-propagating high-temperature synthesis (SHS) of porous TiNi alloy. Mater. Res. Express 2019, 6, 056522. [Google Scholar] [CrossRef]
- Kokorev, O.; Hodorenko, V.; Chekalkin, T.; Gunther, V.; Kang, S.B.; Chang, M.J.; Kang, J.H. Evaluation of allogenic hepato-tissue engineered in porous TiNi-based scaffolds for liver regeneration in a CCl4-induced cirrhosis rat model. Biomed. Phys. Eng. Express 2019, 5, 025018. [Google Scholar] [CrossRef]
- Kokorev, O.; Chekalkin, T.; Marchenko, E.; Yasenchuk, Y.; Gunther, S.; Serebrov, V.; Chernyshova, A.; Obrosov, A.; Uludintceva, E.; Kang, J.H. Exploring the role of surface modifications of TiNi-based alloys in evaluating in vitro cytocompatibility: A comparative study. Surf. Topogr. Metrol. Prop. 2020, 8, 045015. [Google Scholar] [CrossRef]
- Aihara, H.; Zider, J.; Fanton, G.; Duerig, T. Combustion synthesis porous Nitinol for biomedical applications. Int. J. Biomater. 2019, 2019, 4307461. [Google Scholar] [CrossRef]
- Topolnitskiy, E.; Dambayev, G.; Gyunter, V. The replacement of postresectional defects of the thorax with the use of tissue im-plant of nanostructural nickelid-titan thread. Khirurgiia 2011, 10, 47–53. [Google Scholar]
- Zheravin, A.; Gyunter, V.; Anisenya, I.; Garbukov, E.; Zhamgaryan, G.; Bogoutdinova, A. Reconstruction of the chest wall using titanium-nickelid for cancer patients. Sib. J. Oncol. 2015, 3, 31–38. [Google Scholar]
- Kudrjavtsev, A.; Zheravin, A.; Anikeeva, O.; Polovnikov, E.; Yarmoshuk, S.; Drobyazgin, E. Treatment of a patient with sarcoma of sternum, ribs and invasion into peri-card and pleura. Patologiya Krovoobrashcheniya i Kardiokhirurgiya 2015, 1924, 124–129. [Google Scholar]
- Wada, T.; Kawai, A.; Ihara, K.; Sasaki, M.; Sonoda, T.; Imaeda, T.; Yamashita, T. Construct validity of the Enneking score for measuring function in patients with malignant or aggressive benign tumours of the upper limb. J. Bone Joint Surg. 2007, 89, 659–663. [Google Scholar] [CrossRef][Green Version]

| Morphological Type | No. of Patients | |
|---|---|---|
| Abs. | % | |
| * NSCLC with invasion into the chest wall | 8 | 53.3 |
| Metastasis of renal cancer | 2 | 13.3 |
| Metastasis of NSCLC after radical lower lobectomy | 1 | 6.7 |
| Breast cancer | 1 | 6.7 |
| Plasmacytoma | 1 | 6.7 |
| Fibrous dysplasia | 2 | 13.3 |
| Total | 15 | 100 |
| Concomitant Diseases | No. of Patients | |
|---|---|---|
| Abs. | % | |
| Chronic obstructive pulmonary disease | 9 | 64.3 |
| Chronic nonspecific lung disease | 1 | 7.1 |
| Coronary artery disease | 2 | 14.3 |
| Abnormal cardiac rhythm | 2 | 14.3 |
| Type 2 diabetes mellitus | 2 | 14.3 |
| Obesity | 2 | 14.3 |
| Gastric and duodenal ulcer disease | 1 | 7.1 |
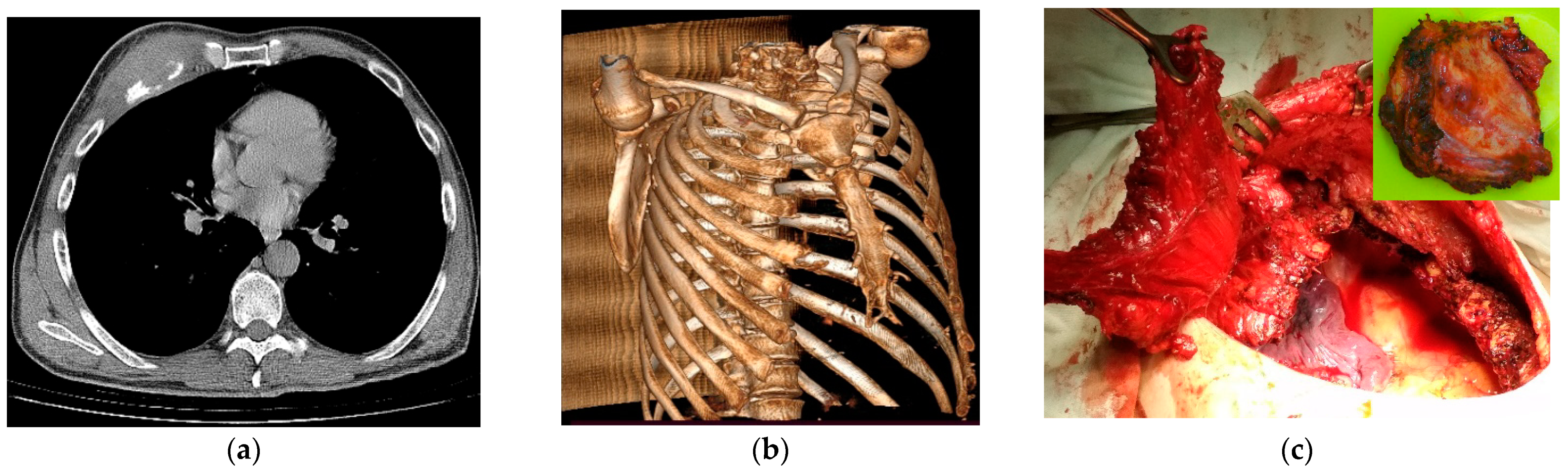
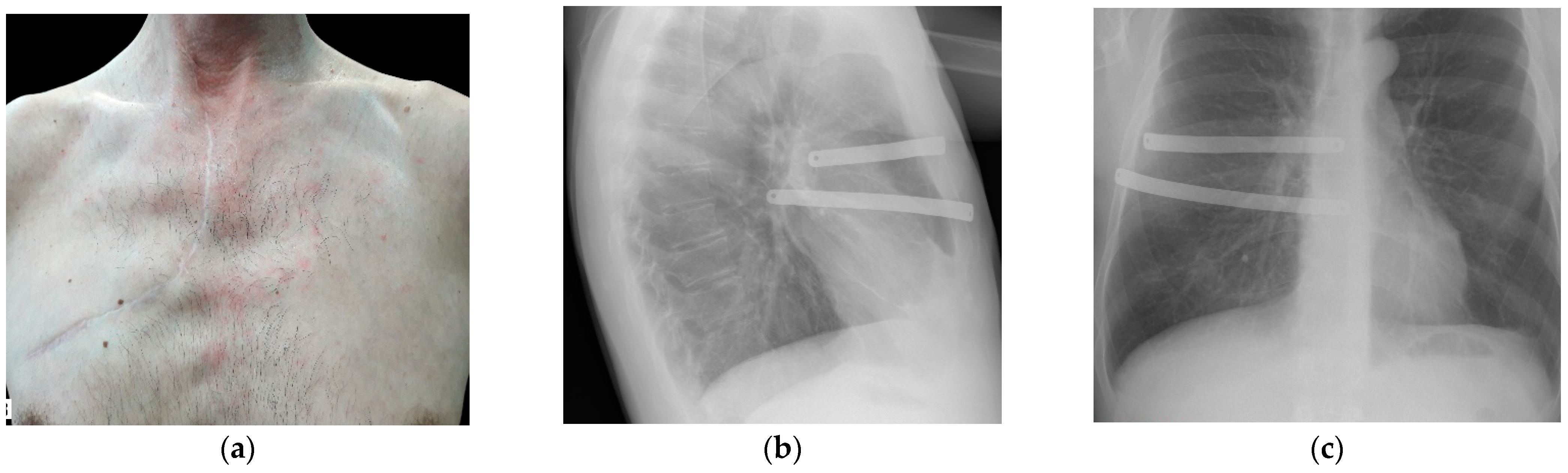
| No. | Defect Localization | No. of Excised Ribs | Lung Resection | Sternum Resection | Muscle Resection | Skin Resection | Others | Defect Area, cm2 | Operative Time, Min | ICU, Day | CTD, Day | PLoS, Day | Complications |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Lat | 2 | CP | ─ | ─ | ─ | Pericardial | 84 | 180 | 3 | 2 | 15 | |
| 2 | Ant/Lat | 2 | LUL | ─ | PM | ─ | ─ | 50 | 150 | 1 | 4 | 14 | |
| 3 | Post/Lat | 2 | ─ | ─ | LD, PV | Yes | ─ | 250 | 120 | 1 | 3 | 10 | |
| 4 | Ant/Lat | 4 | WR | ─ | PM, Pm | ─ | ─ | 198 | 185 | 1 | 5 | 14 | |
| 5 | Ant | 3 | RUL | ─ | PM, Pm | ─ | ─ | 78 | 140 | 2 | 4 | 12 | |
| 6 | Ant | 3 | RUL | ─ | PM, Pm | Yes | ─ | 98 | 240 | 5 | 8 | 18 | pAL |
| 7 | Ant/Lat | 1 | ─ | ─ | Sc | ─ | ─ | 36 | 80 | ─ | 1 | 7 | |
| 8 | Ant/Lat | 3 | LUL | ─ | SM | ─ | ─ | 98 | 210 | 1 | 5 | 15 | |
| 9 | Post/Lat | 2 | RUL | ─ | PV | VB | ─ | 84 | 180 | 5 | 4 | 21 | pAF |
| 10 | Ant/Lat | 4 | ─ | ─ | PM | Yes | ─ | 105 | 150 | 3 | 4 | 14 | |
| 11 | Ant/Lat | 1 | ─ | ─ | ─ | ─ | ─ | 70 | 85 | 0 | 1 | 5 | |
| 12 | Ant/Lat | 3 | LUL | ─ | SM | ─ | ─ | 92 | 130 | 2 | 6 | 12 | PE, S |
| 13 | Ant/Lat | 3 | RUL | ─ | SM | ─ | ─ | 98 | 110 | 2 | 5 | 12 | |
| 14 | Ant/Lat | 2 | RUL | ─ | ─ | ─ | ─ | 78 | 145 | 3 | 3 | 14 | |
| 15 | Ant | 8 | ─ | Subtotal | PM | Yes | ─ | 576 | 130 | 3 | 5 | 16 | |
| Mean (min–max) | ─ | 2.6 (1–8) | ─ | ─ | ─ | ─ | ─ | 133 (36–576) | 149 (80–240) | 2.1 (0–5) | 4 (1–8) | 13.3 (5–21) | ─ |
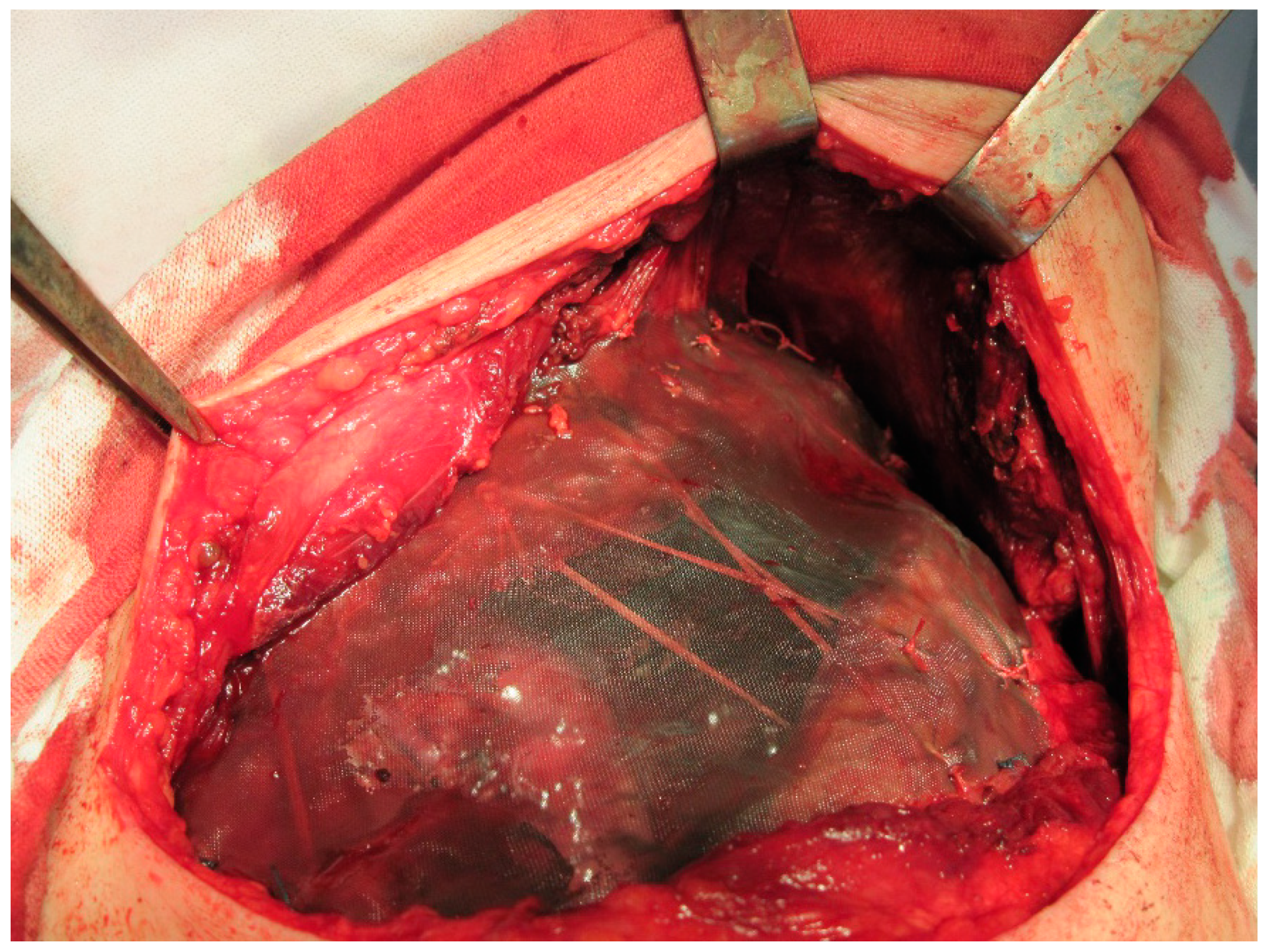
| No. | Mesh Type | Reinforcing Constituent Type | Soft Tissue Flap | Paradoxic Respiration |
|---|---|---|---|---|
| 1 | SL | PS | LT | ─ |
| 2 | SL | PS | Muscle | ─ |
| 3 | DL | PS | Muscle | ─ |
| 4 | DL | AR | Muscle | ─ |
| 5 | DL | 2 AR | LT | ─ |
| 6 | DL | 3 strips | Muscle | Yes |
| 7 | DL | ─ | Muscle | ─ |
| 8 | DL | AR | Muscle | ─ |
| 9 | DL | PS | LT | ─ |
| 10 | DL | 3 AR | Muscle | ─ |
| 11 | DL | ─ | LT | ─ |
| 12 | DL | 2 AR | LT | ─ |
| 13 | DL | 2 AR | LT | ─ |
| 14 | DL | 2 AR | LT | ─ |
| 15 | DL | 3 AR | Muscle | ─ |
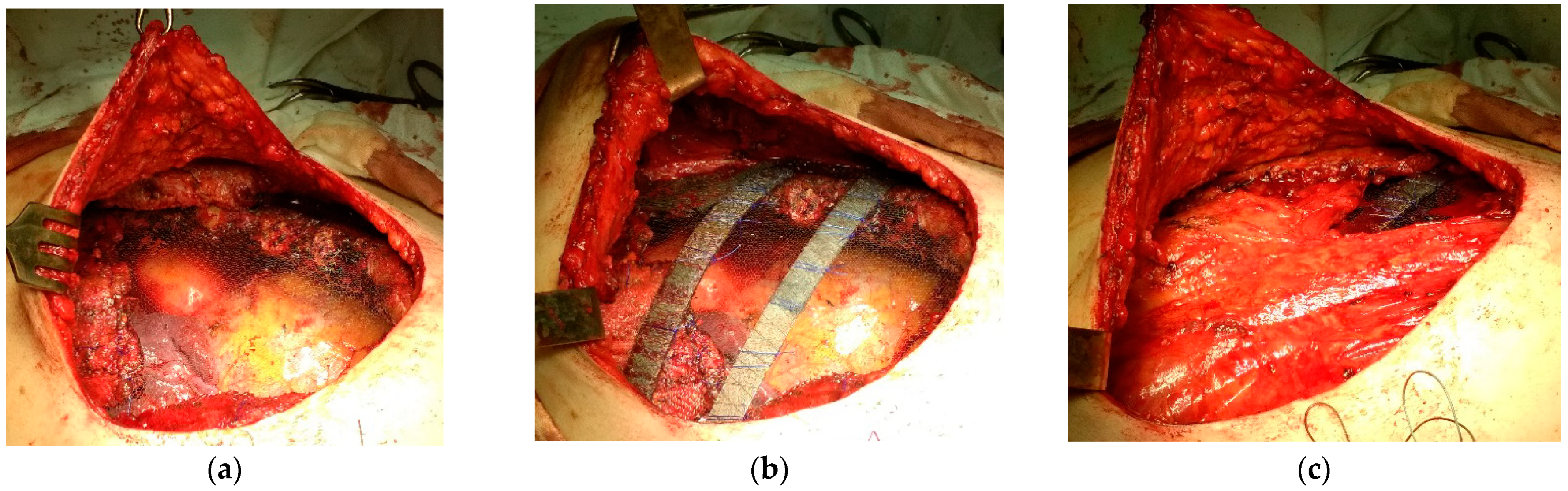
| Repair Option | No. of Patients | |
|---|---|---|
| Abs. | % | |
| Mesh + local tissues | 1 | 6.7 |
| Mesh + thoracodorsal flap | 1 | 6.7 |
| Double-layer mesh + local tissues | 2 | 13.3 |
| Double-layer mesh + pectoral flap | 2 | 13.3 |
| Double-layer mesh + strip (3 pcs) + pectoral flap | 1 | 6.7 |
| Double-layer mesh + rib prosthesis (1 pcs) + pectoral flap | 2 | 13.3 |
| Double-layer mesh + rib prosthesis (2 pcs) + local tissues | 4 | 26.7 |
| Double-layer mesh + rib prosthesis (3 pcs) + pectoral flap, external abdominal oblique muscle flap | 2 | 13.3 |
| Total | 15 | 100 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Topolnitskiy, E.; Chekalkin, T.; Marchenko, E.; Yasenchuk, Y.; Kang, S.-B.; Kang, J.-H.; Obrosov, A. Evaluation of Clinical Performance of TiNi-Based Implants Used in Chest Wall Repair after Resection for Malignant Tumors. J. Funct. Biomater. 2021, 12, 60. https://doi.org/10.3390/jfb12040060
Topolnitskiy E, Chekalkin T, Marchenko E, Yasenchuk Y, Kang S-B, Kang J-H, Obrosov A. Evaluation of Clinical Performance of TiNi-Based Implants Used in Chest Wall Repair after Resection for Malignant Tumors. Journal of Functional Biomaterials. 2021; 12(4):60. https://doi.org/10.3390/jfb12040060
Chicago/Turabian StyleTopolnitskiy, Evgeniy, Timofey Chekalkin, Ekaterina Marchenko, Yuri Yasenchuk, Seung-Baik Kang, Ji-Hoon Kang, and Aleksei Obrosov. 2021. "Evaluation of Clinical Performance of TiNi-Based Implants Used in Chest Wall Repair after Resection for Malignant Tumors" Journal of Functional Biomaterials 12, no. 4: 60. https://doi.org/10.3390/jfb12040060
APA StyleTopolnitskiy, E., Chekalkin, T., Marchenko, E., Yasenchuk, Y., Kang, S.-B., Kang, J.-H., & Obrosov, A. (2021). Evaluation of Clinical Performance of TiNi-Based Implants Used in Chest Wall Repair after Resection for Malignant Tumors. Journal of Functional Biomaterials, 12(4), 60. https://doi.org/10.3390/jfb12040060






