Evaluation of Osteoconduction of Biphasic Calcium Phosphate Ceramic in the Calvaria of Rats: Microscopic and Histometric Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design
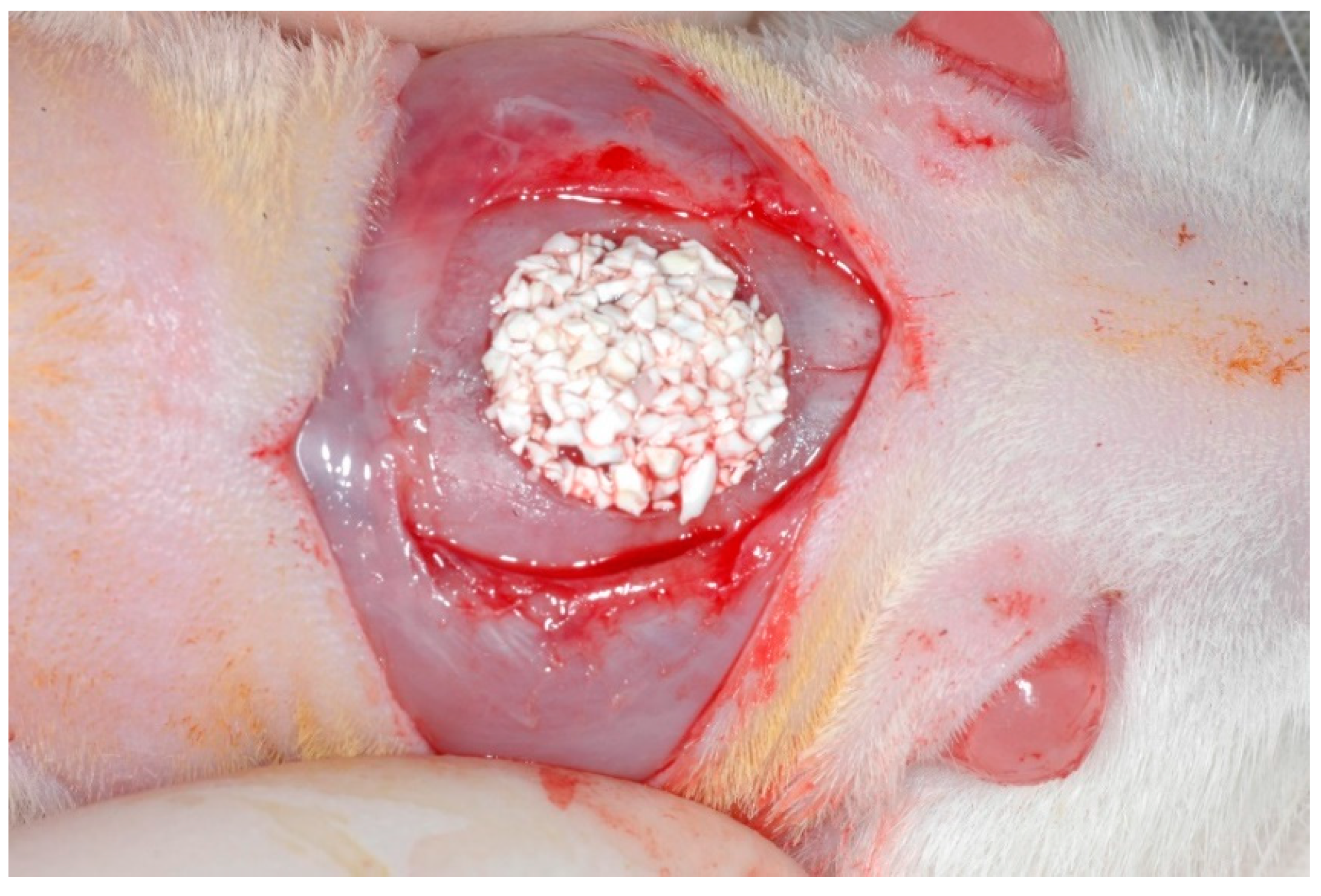
2.2. Surgical Procedures
2.3. Sample Preparation
2.4. Histological and Histometric Analysis
2.5. Statistical Analysis
3. Results
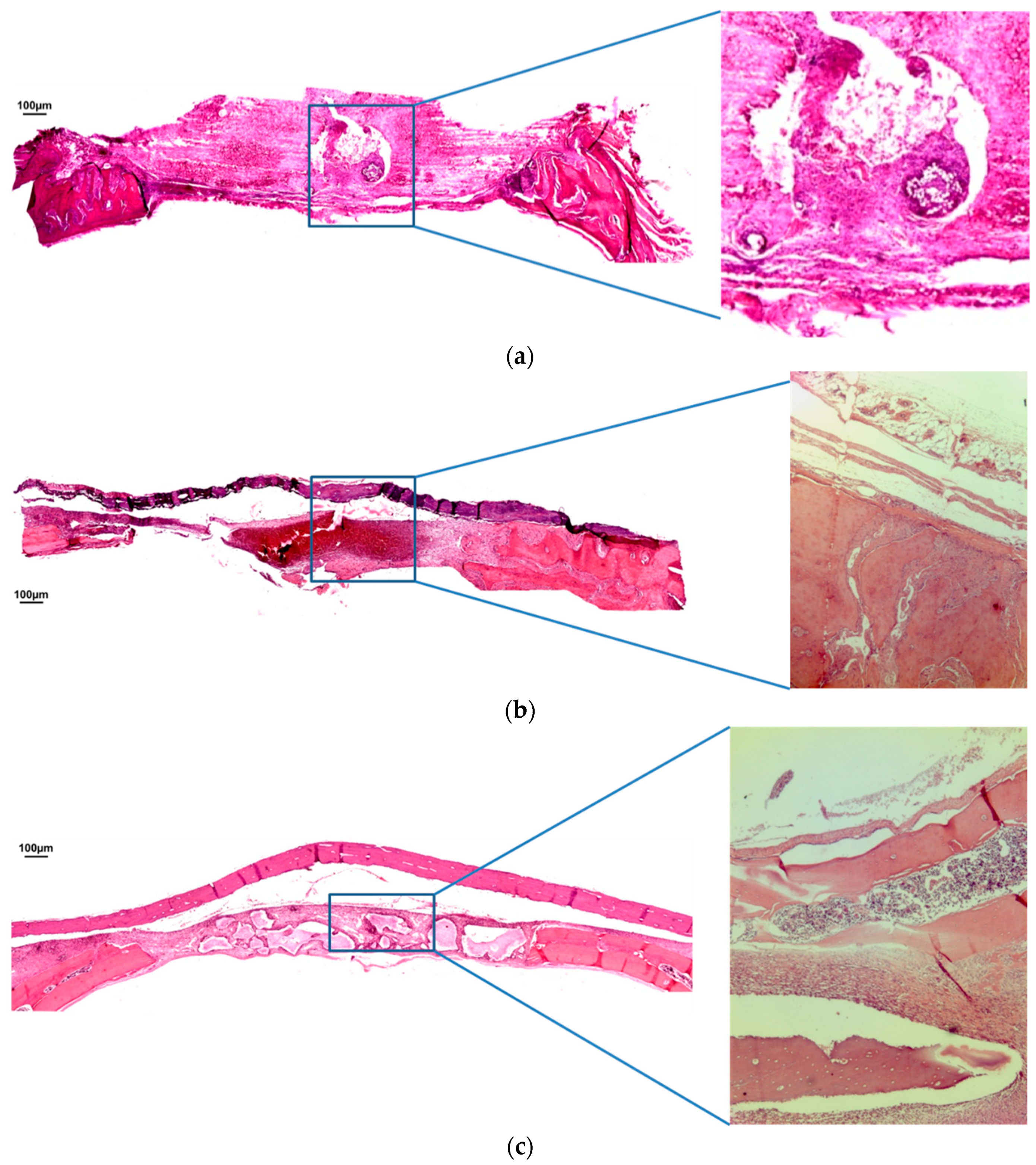
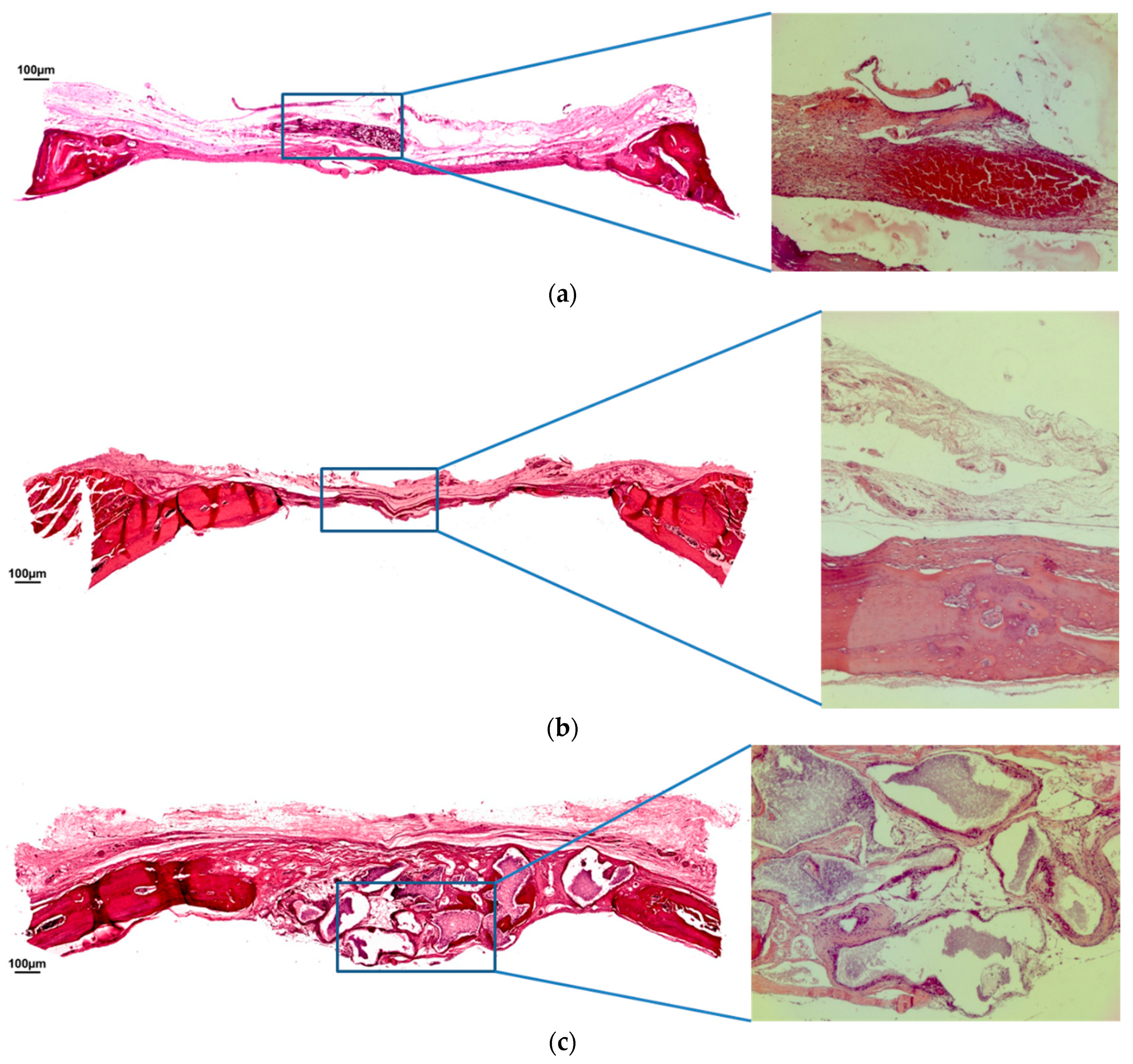
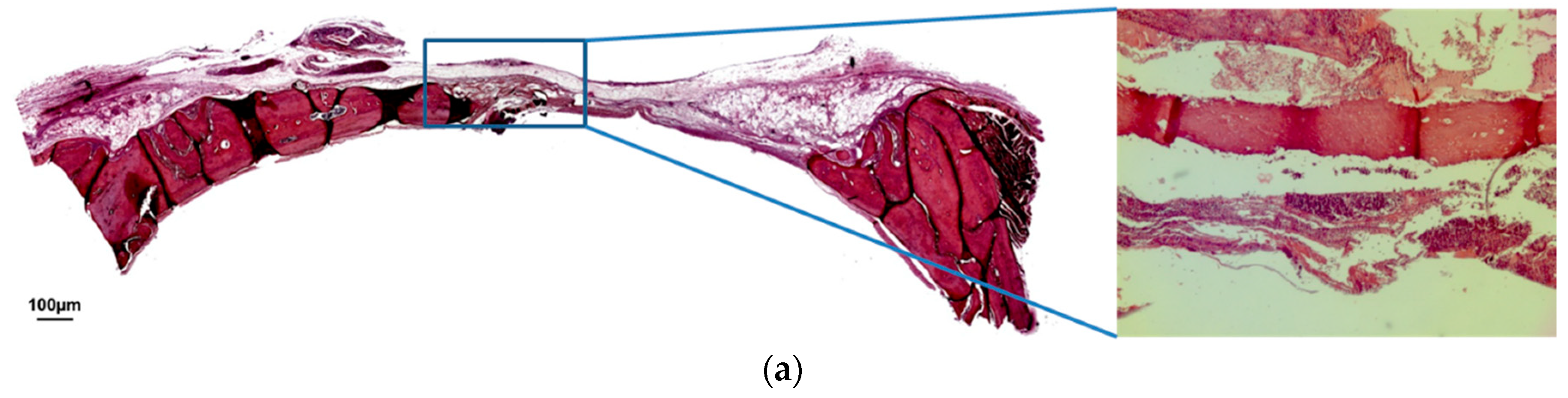
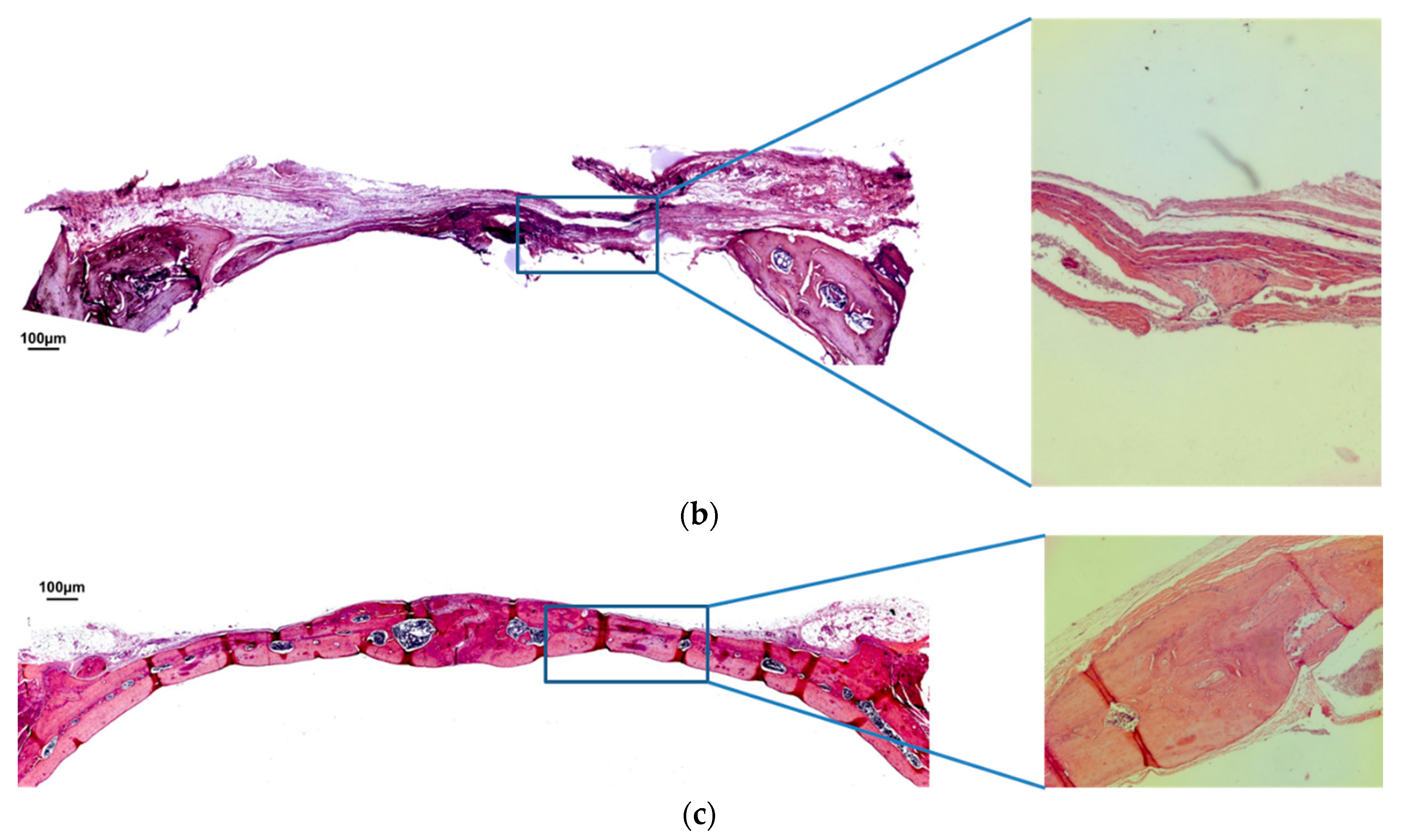
3.1. Qualitative Analysis
3.2. Quantitative Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
List of Abbreviations
| BCP | Biphasic calcium phosphate |
| BCG | Blood clot group |
| MBCG | Membrane blood clot group |
| BCPG | Biphasic calcium phosphate group |
| CaP | Calcium phosphate-based biomaterials |
| HA | Hydroxyapatite |
| β-TCP | Β-tricalcium phosphate |
| HA/β-TCP | Composite of hydroxyapatite and β-tricalcium phosphate |
| COBEA | Brazilian College of Animal Experimentation |
| FOA | Araçatuba Dental of School |
| Milli-Q water | Deionized water |
| μm | Micrometers |
| % | Percentage |
| R² | Coefficient of determination |
| β | Standardized coefficient |
References
- Horikawa, T.; Odatsu, T.; Itoh, T.; Soejima, Y.; Morinaga, H.; Abe, N.; Sawase, T. Retrospective cohort study of rough-surface titanium implants with at least 25 years’ function. Int. J. Implant Dent. 2017, 3, 42. [Google Scholar] [CrossRef] [PubMed]
- Lekholm, U.; Grondahl, K.; Jemt, T. Outcome of oral implant treatment in partially edentulous jaws followed 20 years in clinical function. Clin. Implant Dent. Relat. Res. 2006, 8, 178–186. [Google Scholar] [CrossRef] [PubMed]
- Astrand, P.; Ahlqvist, J.; Gunne, J.; Nilson, H. Implant treatment of patients with edentulous jaws: A 20-year follow-up. Clin. Implant Dent. Relat. Res. 2008, 10, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Knofler, W.; Barth, T.; Graul, R.; Krampe, D. Retrospective analysis of 10,000 implants from insertion up to 20 years-analysis of implantations using augmentative procedures. Int. J. Implant Dent. 2016, 2, 25. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, R.C.; Leles, C.R.; Lindh, C.; Ribeiro-Rota, R.F. Bone tissue microarchitectural characteristics at dental implant sites. Part 1: Identification of clinical-related parameters. Clin. Oral Implants Res. 2012, 23, 981–986. [Google Scholar] [CrossRef] [PubMed]
- Eriksen, E.F. Cellular mechanisms of bone remodeling. Rev. Endocr. Metab. Disord. 2010, 11, 219–227. [Google Scholar] [CrossRef] [PubMed]
- MacBeth, N.; Trullenque-Eriksson, A.; Donos, N.; Mardas, N. Hard and soft tissue changes following alveolar ridge preservation: A systematic review. Clin. Oral Implants Res. 2017, 28, 982–1004. [Google Scholar] [CrossRef] [PubMed]
- Troeltzsch, M.; Troeltzsch, M.; Kauffmann, P.; Gruber, R.; Brockmeyer, P.; Moser, N.; Schliephake, H. Clinical efficacy of grafting materials in alveolar ridge augmentation: A systematic review. J. Craniomaxillofac. Surg. 2016, 44, 1618–1629. [Google Scholar] [CrossRef]
- Sakkas, A.; Wilde, F.; Heufelder, M.; Winter, K.; Schramm, A. Autogenous bone grafts in oral implantology-is it still a ”gold standard“? A consecutive review of 279 patients with 456 clinical procedures. Int. J. Implant Dent. 2017, 3, 23. [Google Scholar] [CrossRef]
- Nkenke, E.; Neukam, F.W. Autogenous bone harvesting and grafting in advanced jaw resorption: Morbidity, resorption and implant survival. Eur. J. Oral Implantol. 2014, 7 (Suppl. 2), S203–S217. [Google Scholar]
- Thrivikraman, G.; Athirasala, A.; Twohig, C.; Boda, S.K.; Bertassoni, L.E. Biomaterials for Craniofacial Bone Regeneration. Dent. Clin. N. Am. 2017, 61, 835–856. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, M.; Botelho, M.G.; Dorozhkin, S.V. Biphasic calcium phosphates bioceramics (HA/TCP): Concept, physicochemical properties and the impact of standardization of study protocols in biomaterials research. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 71, 1293–1312. [Google Scholar] [CrossRef] [PubMed]
- Bouler, J.M.; Pilet, P.; Gauthier, O.; Verron, E. Biphasic calcium phosphate ceramics for bone reconstruction: A review of biological response. Acta Biomater. 2017, 53, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Danesh-Sani, S.A.; Wallace, S.S.; Movahed, A.; El Chaar, E.S.; Cho, S.C.; Khouly, I.; Testori, T. Maxillary Sinus Grafting With Biphasic Bone Ceramic or Autogenous Bone: Clinical, Histologic, and Histomorphometric Results From a Randomized Controlled Clinical Trial. Implant Dent. 2016, 25, 588–593. [Google Scholar] [CrossRef] [PubMed]
- de Lange, G.L.; Overman, J.R.; Farre-Guasch, E.; Klein-Nulend, J. A histomorphometric and micro-computed tomography study of bone regeneration in the maxillary sinus comparing biphasic calcium phosphate and deproteinized cancellous bovine bone in a human split-mouth model. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2014, 117, 8–22. [Google Scholar] [CrossRef] [PubMed]
- Park, J.C.; Bae, E.B.; Kim, S.E.; Kim, S.Y.; Choi, K.H.; Choi, J.W.; Huh, J.B. Effects of BMP-2 Delivery in Calcium Phosphate Bone Graft Materials with Different Compositions on Bone Regeneration. Materials 2016, 9, 954. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.U.; Lim, H.C.; Hong, J.Y.; Lee, J.S.; Jung, U.W.; Choi, S.H. Bone regenerative efficacy of biphasic calcium phosphate collagen composite as a carrier of rhBMP-2. Clin. Oral Implants Res. 2016, 27, e91–e99. [Google Scholar] [CrossRef]
- Dahlin, C.; Obrecht, M.; Dard, M.; Donos, N. Bone tissue modelling and remodeling following guided bone regeneration in combination with biphasic calcium phosphate materials presenting different microporosity. Clin. Oral Implants Res. 2015, 26, 814–822. [Google Scholar] [CrossRef]
- Lan Levengood, S.K.; Polak, S.J.; Poellmann, M.J.; Hoelzle, D.J.; Maki, A.J.; Clark, S.G.; Johnson, A.J.W. The effect of BMP-2 on micro- and macroscale osteointegration of biphasic calcium phosphate scaffolds with multiscale porosity. Acta Biomater. 2010, 6, 3283–3291. [Google Scholar] [CrossRef]
- Lim, H.C.; Hong, J.Y.; Lee, J.S.; Jung, U.W.; Choi, S.H. Late-term healing in an augmented sinus with different ratios of biphasic calcium phosphate: A pilot study using a rabbit sinus model. J. Periodontal Implant Sci. 2016, 46, 57–69. [Google Scholar] [CrossRef]
- Lim, H.C.; Zhang, M.L.; Lee, J.S.; Jung, U.W.; Choi, S.H. Effect of different hydroxyapatite:beta-tricalcium phosphate ratios on the osteoconductivity of biphasic calcium phosphate in the rabbit sinus model. Int. J. Oral Maxillofac. Implants 2015, 30, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.W.; Choi, K.H.; Yun, J.H.; Kim, C.S.; Choi, S.H.; Cho, K.S. Bone formation of block and particulated biphasic calcium phosphate lyophilized with Escherichia coli-derived recombinant human bone morphogenetic protein 2 in rat calvarial defects. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2011, 112, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Furlaneto, F.A.; Nagata, M.J.; Fucini, S.E.; Deliberador, T.M.; Okamoto, T.; Messora, M.R. Bone healing in critical-size defects treated with bioactive glass/calcium sulfate: A histologic and histometric study in rat calvaria. Clin. Oral Implants Res. 2007, 18, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Maciel, J.; Momesso, G.A.C.; Ramalho-Ferreira, G.; Consolaro, R.B.; de Carvalho, P.S.; Faverani, L.P.; Farnezi Bassi, A.P. Bone Healing Evaluation in Critical-Size Defects Treated With Xenogenous Bone Plus Porcine Collagen. Implant Dent. 2017, 26, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Yamada, S.; Heymann, D.; Bouler, J.M.; Daculsi, S. Osteoclastic resorption of calcium phosphate ceramics with different hydroxyapatite/beta-tricalcium phosphate ratios. Biomaterials 1997, 18, 1037–1041. [Google Scholar] [CrossRef]
- Rouvillain, J.L.; Lavalle, F.; Pascal-Mousselard, H.; Catonné, Y.; Dalcusi, G. Clinical, radiological and histological evaluation of biphasic calcium phosphate bioceramic wedges filling medial high tibial valgisation osteotomies. Knee 2009, 16, 392–397. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, K.; Li, X.; Wei, Q.; Chai, W.; Wang, S.; Zhang, B. 3D fabrication and characterization of phosphoric acid scaffold with a HA/beta-TCP weight ratio of 60:40 for bone tissue engineering applications. PLoS ONE 2017, 12, e0174870. [Google Scholar]
- Miramond, T.; Corre, P.; Borget, P.; Moreau, F.; Guicheux, J.; Daculsi, G.; Weiss, P. Osteoinduction of biphasic calcium phosphate scaffolds in a nude mouse model. J. Biomater. Appl. 2014, 29, 595–604. [Google Scholar] [CrossRef]
- Miron, R.J.; Zhang, Q.; Sculean, A.; Buser, D.; Pippenger, B.E.; Dard, M.; Zhang, Y. Osteoinductive potential of 4 commonly employed bone grafts. Clin. Oral Investig. 2016, 20, 2259–2265. [Google Scholar] [CrossRef]
- Yuan, H.; Fernandes, H.; Habibovic, P.; de Boer, J.; Barradas, A.M.; de Ruiter, A.; de Bruijn, J.D. Osteoinductive ceramics as a synthetic alternative to autologous bone grafting. Proc. Natl. Acad. Sci. USA 2010, 107, 13614–13619. [Google Scholar] [CrossRef]
- Santos, P.S.; Cestari, T.M.; Paulin, J.B.; Martins, R.; Rocha, C.A.; Arantes, R.V.N.; Taga, R. Osteoinductive porous biphasic calcium phosphate ceramic as an alternative to autogenous bone grafting in the treatment of mandibular bone critical-size defects. J. Biomed. Mater. Res. Part B Appl. Biomater. 2017, 106, 1546–1557. [Google Scholar] [CrossRef] [PubMed]
- AbdulQader, S.T.; Rahman, I.A.; Thirumulu, K.P.; Ismail, H.; Mahmood, Z. Effect of biphasic calcium phosphate scaffold porosities on odontogenic differentiation of human dental pulp cells. J. Biomater. Appl. 2016, 30, 1300–1311. [Google Scholar] [CrossRef] [PubMed]
- Rustom, L.E.; Boudou, T.; Lou, S.; Pignot-Paintrand, I.; Nemke, B.W.; Lu, Y.; Johnson, A.J.W. Micropore-induced capillarity enhances bone distribution in vivo in biphasic calcium phosphate scaffolds. Acta Biomater. 2016, 44, 144–154. [Google Scholar] [CrossRef] [PubMed]
- Klenke, F.M.; Liu, Y.; Yuan, H.; Hunziker, E.B.; Siebenrock, K.A.; Hofstetter, W. Impact of pore size on the vascularization and osseointegration of ceramic bone substitutes in vivo. J. Biomed. Mater. Res. A 2008, 85, 777–786. [Google Scholar] [CrossRef] [PubMed]
- Dahlin, C.; Linde, A.; Gottlow, J.; Nyman, S. Healing of bone defects by guided tissue regeneration. Plast. Reconstr. Surg. 1988, 81, 672–676. [Google Scholar] [CrossRef]
| Experimental Period | Control Group (BCG) | Blood Clot + Membrane (MBCG) | BCP + Membrane (BCPG) | p Value |
|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | ||
| 7 days | 0.76 ± 0.70 Aa | 1.73 ± 1.13 ABa | 4.52 ± 3.16 Ba | 0.013 * |
| 30 days | 4.45 ± 3.92 Aa | 6.04 ± 1.16 Aa | 8.22 ± 1.23 Aa | 0.065 |
| 60 days | 10.66 ± 5.57 Ab | 14.51 ± 7.00 Ab | 74.22 ± 25.39 Bb | <0.001 * |
| p value | 0.002 * | <0.001 * | <0.001 * |
| Groups | R² | β | p Value |
|---|---|---|---|
| Control group (BCG) | 0.55 | 0.18 | <0.001 * |
| Blood clot + membrane (MBCG) | 0.64 | 0.24 | <0.001 * |
| BCP + membrane (BCPG) | 0.72 | 1.35 | <0.001 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Puttini, I.d.O.; Poli, P.P.; Maiorana, C.; Vasconcelos, I.R.d.; Schmidt, L.E.; Colombo, L.T.; Hadad, H.; Santos, G.M.d.; Carvalho, P.S.P.d.; Souza, F.Á. Evaluation of Osteoconduction of Biphasic Calcium Phosphate Ceramic in the Calvaria of Rats: Microscopic and Histometric Analysis. J. Funct. Biomater. 2019, 10, 7. https://doi.org/10.3390/jfb10010007
Puttini IdO, Poli PP, Maiorana C, Vasconcelos IRd, Schmidt LE, Colombo LT, Hadad H, Santos GMd, Carvalho PSPd, Souza FÁ. Evaluation of Osteoconduction of Biphasic Calcium Phosphate Ceramic in the Calvaria of Rats: Microscopic and Histometric Analysis. Journal of Functional Biomaterials. 2019; 10(1):7. https://doi.org/10.3390/jfb10010007
Chicago/Turabian StylePuttini, Igor de Oliveira, Pier Paolo Poli, Carlo Maiorana, Igor Rodrigues de Vasconcelos, Luis Eduardo Schmidt, Luara Teixeira Colombo, Henrique Hadad, Gabriel Mulinari dos Santos, Paulo Sergio Perri de Carvalho, and Francisley Ávila Souza. 2019. "Evaluation of Osteoconduction of Biphasic Calcium Phosphate Ceramic in the Calvaria of Rats: Microscopic and Histometric Analysis" Journal of Functional Biomaterials 10, no. 1: 7. https://doi.org/10.3390/jfb10010007
APA StylePuttini, I. d. O., Poli, P. P., Maiorana, C., Vasconcelos, I. R. d., Schmidt, L. E., Colombo, L. T., Hadad, H., Santos, G. M. d., Carvalho, P. S. P. d., & Souza, F. Á. (2019). Evaluation of Osteoconduction of Biphasic Calcium Phosphate Ceramic in the Calvaria of Rats: Microscopic and Histometric Analysis. Journal of Functional Biomaterials, 10(1), 7. https://doi.org/10.3390/jfb10010007







