Mitochondrial Functions, Cognition, and the Evolution of Intelligence: Reply to Commentaries and Moving Forward
Abstract
1. Introduction
2. Relative Importance of Mitochondria
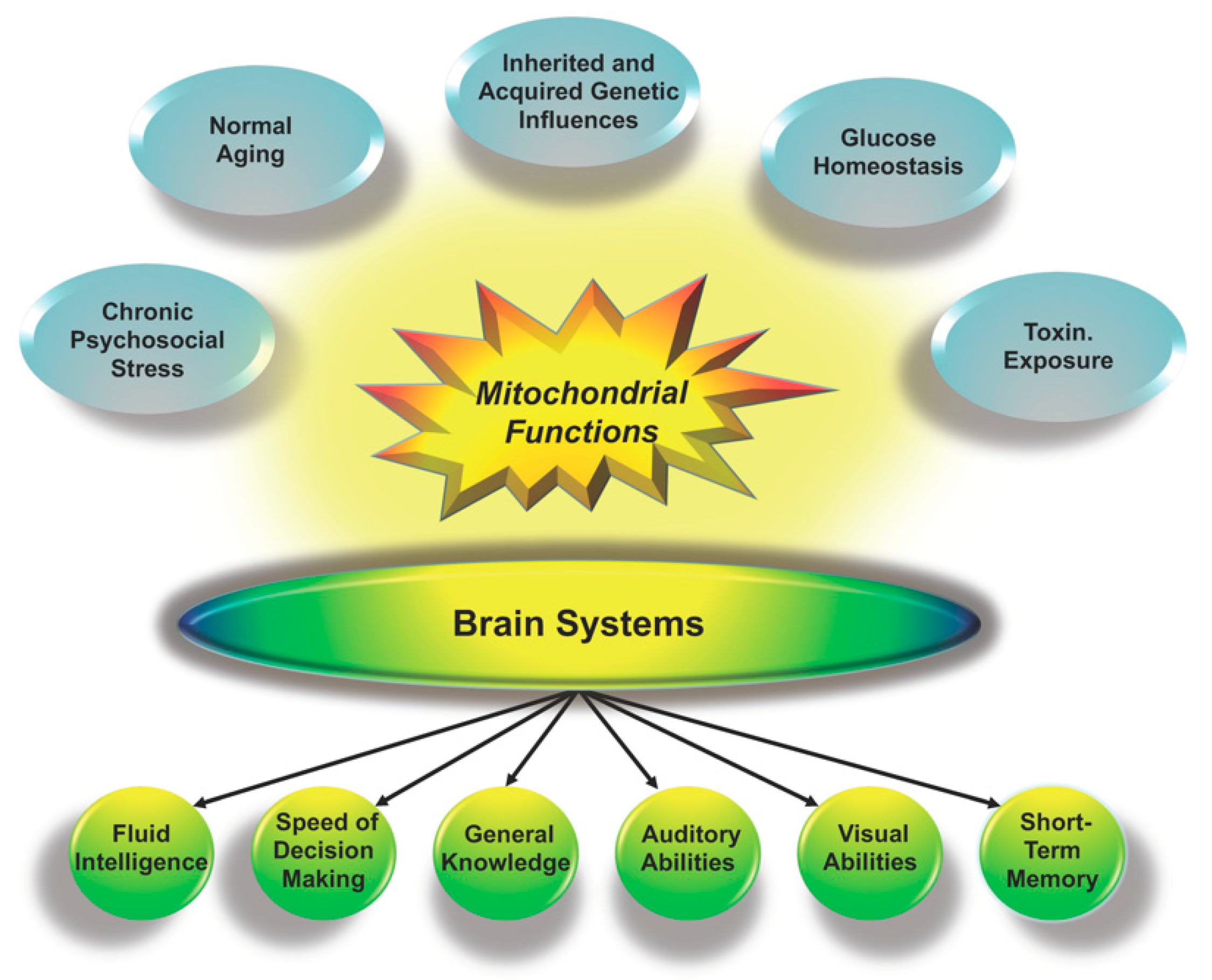
2.1. Cognition and Mitochondria
2.1.1. Mitochondrial Contributions as a Proportion of g
2.1.2. Mitochondria and Alternative Models of Human Cognition
2.1.3. Mitochondria, Environmental Conditions and the Flynn Effect
2.2. Genes and Mitochondria
2.2.1. Genome-Wide Association Studies, Mitochondrial Proteins, and g
2.2.2. Parental Genetic Influences on Mitochondrial Functions
3. Empirical Studies and Testing the Hypothesis
3.1. Mitochondrial Biomarkers
3.2. Mitochondrial-Related Disorders and Cognition
3.2.1. Mitochondrial Disorders
3.2.2. Obesity, Diabetes, and Inflammation
3.3. Mitochondrial Health and Cognition
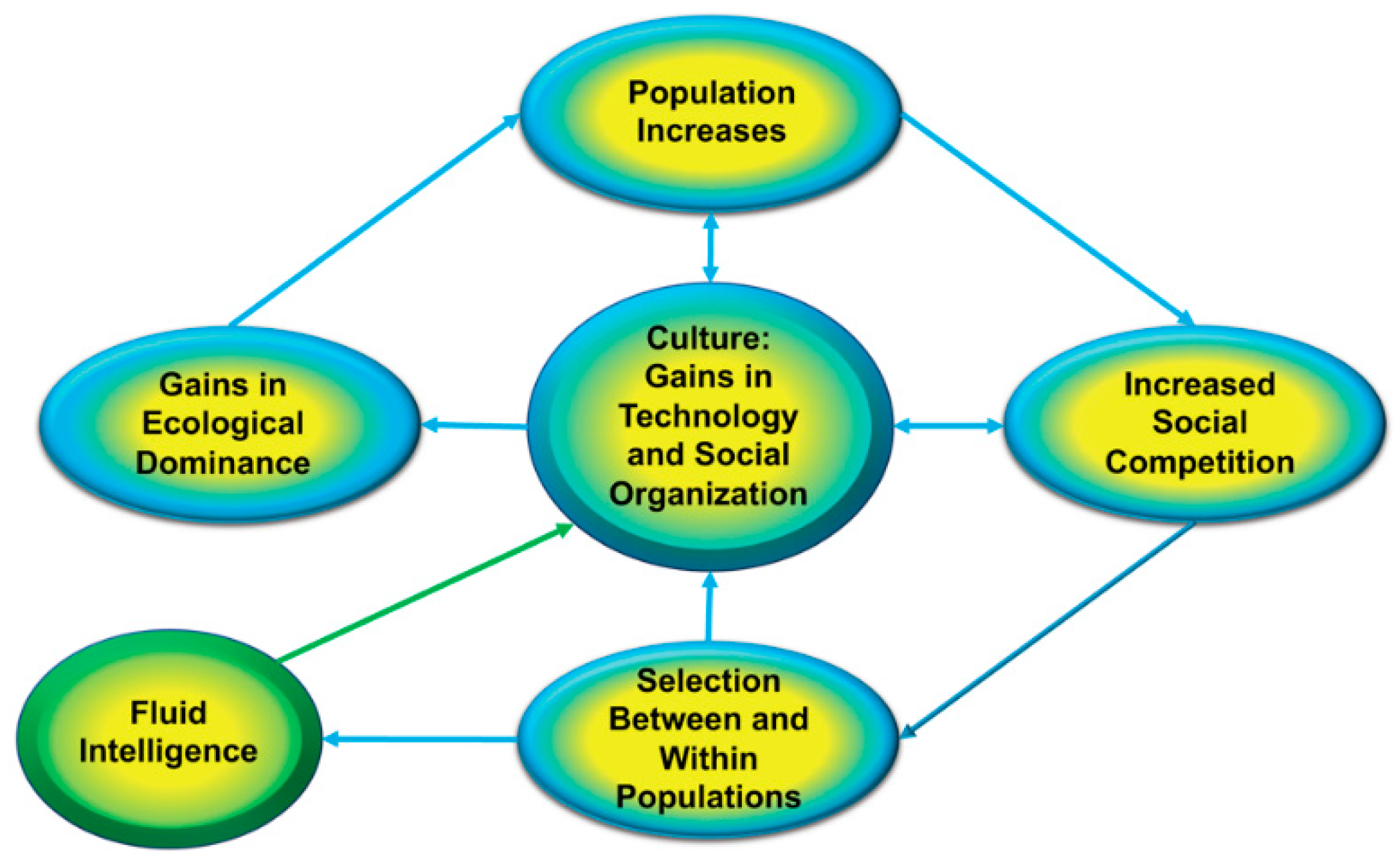
4. Adaptive Function of Intelligence
the ecological dominance of evolving humans diminished the effects of ‘extrinsic’ forces of natural selection such that within-species competition became the principle ‘hostile force of nature’ guiding the long-term evolution of behavioral capacities, traits, and tendencies.
5. Discussion
Funding
Conflicts of Interest
References
- Aiello, Leslie C., and Peter Wheeler. 1995. The expensive-tissue hypothesis: The brain and the digestive system in human and primate evolution. Current Anthropology 36: 199–221. [Google Scholar] [CrossRef]
- Alexander, Richard. D. 1989. Evolution of the human psyche. In The Human Revolution: Behavioural and Biological Perspectives on the Origins of Modern Humans. Edited by Paul Mellars and Chris Stringer. Princeton: Princeton University Press. [Google Scholar]
- Antón, Susan C., Richard Potts, and Leslie C. Aiello. 2014. Evolution of early Homo: An integrated biological perspective. Science 345: 1236828. [Google Scholar] [CrossRef] [PubMed]
- Ash, Jessica, and Gordan G. Gallup. 2007. Paleoclimatic variation and brain expansion during human evolution. Human Nature 18: 109–24. [Google Scholar] [CrossRef] [PubMed]
- Atti, Anna R., Stefano Valente, Antonia Iodice, Ilaria Caramella, Barbara Ferrari, Umberto Albert, Laura Mandelli, and Diana De Ronchi. 2019. Metabolic syndrome (MetS), mild cognitive impairment (MCI) and dementia: A meta-analysis of longitudinal studies. The American Journal of Geriatric Psychiatry 27: 625–37. [Google Scholar] [CrossRef] [PubMed]
- Avgerinos, Konstantinos I., Nikolaos Spyrou, Konstantinos I. Bougioukas, and Dimitrios Kapogiannis. 2018. Effects of creatine supplementation on cognitive function of healthy individuals: A systematic review of randomized controlled trials. Experimental Gerontology 108: 166–73. [Google Scholar] [CrossRef] [PubMed]
- Bailey, Drew H., and David C. Geary. 2009. Hominid brain evolution: Testing climatic, ecological, and social competition models. Human Nature 20: 67–79. [Google Scholar] [CrossRef]
- Baker, Laura D., Donna J. Cross, Satoshi Minoshima, Dana Belongia, G. Stennis Watson, and Suzanne Craft. 2011. Insulin resistance and Alzheimer-like reductions in regional cerebral glucose metabolism for cognitively normal adults with prediabetes or early type 2 diabetes. Archives of Neurology 68: 51–57. [Google Scholar] [CrossRef]
- Beekman, Madeleine, Damian K. Dowling, and Duur K. Aanen. 2014. The costs of being male: Are there sex-specific effects of uniparental mitochondrial inheritance? Philosophical Transactions of the Royal Society B: Biological Sciences 369: 20130440. [Google Scholar] [CrossRef] [PubMed]
- Bhatti, Jasvinder S., Gurjit K. Bhatti, and P. Hemachandra Reddy. 2017. Mitochondrial dysfunction and oxidative stress in metabolic disorders—A step towards mitochondria based therapeutic strategies. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease 1863: 1066–77. [Google Scholar] [CrossRef] [PubMed]
- Bijnens, Esmee M., Catherine Derom, Steven Weyers, Bram G. Janssen, Evert Thiery, and Tim S. Nawrot. 2019. Placental mitochondrial DNA content is associated with childhood intelligence. Journal of Translational Medicine 17: 361. [Google Scholar] [CrossRef] [PubMed]
- Bullmore, Ed, and Olaf Sporns. 2012. The economy of brain network organization. Nature Reviews Neuroscience 13: 336–49. [Google Scholar] [CrossRef] [PubMed]
- Burgoyne, Alexander P., and Randall W. Engle. 2020. Mitochondrial functioning and its relation to higher-order cognitive processes: Commentary on Geary (2018; 2019). Journal of Intelligence 8: 14. [Google Scholar] [CrossRef] [PubMed]
- Burkart, Judith M., Michele N. Schubiger, and Carel P. van Schaik. 2017. The evolution of general intelligence. Behavioral and Brain Sciences 40: e195. [Google Scholar] [CrossRef] [PubMed]
- Caito, Samuel W., and Michael Aschner. 2015. Mitochondrial redox dysfunction and environmental exposures. Antioxidants & Redox Signaling 23: 578–95. [Google Scholar] [CrossRef]
- Calvo, Sarah E., Karl R. Clauser, and Vamsi K. 2016. Mootha. MitoCarta2. 0: An updated inventory of mammalian mitochondrial proteins. Nucleic Acids Research 44: D1251–57. [Google Scholar] [CrossRef]
- Carroll, John B. 1993. Human Cognitive Abilities: A Survey of Factor-Analytic Studies. New York: Cambridge University Press. [Google Scholar]
- Cattell, Raymond B. 1963. Theory of fluid and crystallized intelligence: A critical experiment. Journal of Educational Psychology 54: 1–22. [Google Scholar] [CrossRef]
- Cheng, G., C. Huang, H. Deng, and H. Wang. 2012. Diabetes as a risk factor for dementia and mild cognitive impairment: A meta-analysis of longitudinal studies. Internal Medicine Journal 42: 484–91. [Google Scholar] [CrossRef]
- Chinnery, Patrick F., and Douglass M. Turnbull. 2001. Epidemiology and treatment of mitochondrial disorders. American Journal of Medical Genetics 106: 94–101. [Google Scholar] [CrossRef]
- Clark, Gregory. 2008. A Farewell to Alms: A Brief Economic History of the World. Princeton: Princeton University Press. [Google Scholar]
- Coleman, Jonathan R. I., Julien Bryois, Héléna A. Gaspar, Philip R. Jansen, Jeanne E. Savage, Nathan Skene, Robert Plomin, Ana B. Muñoz-Manchado, Sten Linnarsson, Greg Crawford, and et al. 2019. Biological annotation of genetic loci associated with intelligence in a meta-analysis of 87,740 individuals. Molecular Psychiatry 24: 182–97. [Google Scholar] [CrossRef]
- Cowan, Nelson. 2017. The many faces of working memory and short-term storage. Psychonomic Bulletin & Review 24: 1158–70. [Google Scholar] [CrossRef]
- Currie, Thomas E., Peter Turchin, Edward Turner, and Sergey Gavrilets. 2020. Duration of agriculture and distance from the steppe predict the evolution of large-scale human societies in Afro-Eurasia. Humanities and Social Sciences Communications 7: 34. [Google Scholar] [CrossRef]
- Darwin, Charles, and Alfred Wallace. 1858. On the tendency of species to form varieties, and on the perpetuation of varieties and species by natural means of selection. Journal of the Linnean Society of London, Zoology 3: 45–62. [Google Scholar] [CrossRef]
- Davies, Gail, Max Lam, Sarah E. Harris, Joey W. Trampush, Michelle Luciano, W. David Hill, Saskia P. Hagenaars, Stuart J. Ritchie, Riccardo E. Marioni, Chloe Fawns-Ritchie, and et al. 2018. Study of 300,486 individuals identifies 148 independent genetic loci influencing general cognitive function. Nature Communications 9: 1–16. [Google Scholar] [CrossRef] [PubMed]
- De Boeck, Paul, and Kristof Kovacs. 2020. The many faces of intelligence: A discussion of Geary’s mitochondrial functioning theory on general intelligence. Journal of Intelligence 8: 8. [Google Scholar] [CrossRef] [PubMed]
- de Mello, Aline H., Ana B. Costa, Jessica D. G. Engel, and Gislaine T. Rezin. 2018. Mitochondrial dysfunction in obesity. Life Sciences 192: 26–32. [Google Scholar] [CrossRef]
- Deary, Ian J., Alexander Weiss, and G. David Batty. 2010. Intelligence and personality as predictors of illness and death: How researchers in differential psychology and chronic disease epidemiology are collaborating to understand and address health inequalities. Psychological Science in the Public Interest 11: 53–79. [Google Scholar] [CrossRef]
- Debatin, Tobias. 2020. Neuroenergetics and “General Intelligence”: A systems biology perspective. Journal of Intelligence 8: 31. [Google Scholar] [CrossRef]
- Dicke, Ursula, and Gerhard Roth. 2016. Neuronal factors determining high intelligence. Philosophical Transactions of the Royal Society B: Biological Sciences 371: 20150180. [Google Scholar] [CrossRef]
- Dickens, William T., and James R. Flynn. 2001. Heritability estimates versus large environmental effects: The IQ paradox resolved. Psychological Review 108: 346–69. [Google Scholar] [CrossRef]
- Dye, Louise, Neil B. Boyle, Claire Champ, and Clare Lawton. 2017. The relationship between obesity and cognitive health and decline. Proceedings of the Nutrition Society 76: 443–54. [Google Scholar] [CrossRef]
- Edwards, Lindsay M., Andrew J. Murray, Cameron J. Holloway, Emma E. Carter, Graham J. Kemp, Ion Codreanu, Helen Brooker, Damian J. Tyler, Peter A. Robbins, and Kieran Clarke. 2011. Short-term consumption of a high-fat diet impairs whole-body efficiency and cognitive function in sedentary men. The FASEB Journal 25: 1088–96. [Google Scholar] [CrossRef] [PubMed]
- Elton, Sarah, Laura C. Bishop, and Bernard Wood. 2001. Comparative context of Plio-Pleistocene hominin brain evolution. Journal of Human Evolution 41: 1–27. [Google Scholar] [CrossRef] [PubMed]
- Engle, Randall W. 2002. Working memory capacity as executive attention. Current Directions in Psychological Science 11: 19–23. [Google Scholar] [CrossRef]
- Eppig, Christopher, Corey L. Fincher, and Randy Thornhill. 2010. Parasite prevalence and the worldwide distribution of cognitive ability. Proceedings of the Royal Society B: Biological Sciences 277: 3801–8. [Google Scholar] [CrossRef] [PubMed]
- Fanta, Vaclav, Miroslav Šálek, Jan Zouhar, Petr Sklenicka, and David Storch. 2018. Equilibrium dynamics of European pre-industrial populations: The evidence of carrying capacity in human agricultural societies. Proceedings of the Royal Society B: Biological Sciences 285: 20172500. [Google Scholar] [CrossRef] [PubMed]
- Finsterer, J. 2012. Cognitive dysfunction in mitochondrial disorders. Acta Neurologica Scandinavica 126: 1–11. [Google Scholar] [CrossRef]
- Flinn, Mark V., David C. Geary, and Carol V. Ward. 2005. Ecological dominance, social competition, and coalitionary arms races: Why humans evolved extraordinary intelligence. Evolution and Human Behavior 26: 10–46. [Google Scholar] [CrossRef]
- Flynn, James R. 1984. The mean IQ of Americans: Massive gains 1932 to 1978. Psychological Bulletin 95: 29–51. [Google Scholar] [CrossRef]
- Foley, Robert. 1987. Hominid species and stone-tool assemblages: How are they related. Antiquity 61: 380–92. [Google Scholar] [CrossRef]
- Foley, Robert, and Marta M. Lahr. 1997. Mode 3 technologies and the evolution of modern humans. Cambridge Archaeology Journal 7: 3–36. [Google Scholar] [CrossRef]
- Geary, David C. 2005. The Origin of Mind: Evolution of Brain, Cognition, and General Intelligence. Washington, DC: American Psychological Association. [Google Scholar]
- Geary, David C. 2015. Evolution of Vulnerability: Implications for sex Differences in Health and Development. San Diego: Elsevier Academic Press. [Google Scholar]
- Geary, David C. 2017. Evolution of human sex-specific cognitive vulnerabilities. The Quarterly Review of Biology 92: 361–410. [Google Scholar] [CrossRef]
- Geary, David C. 2018. Efficiency of mitochondrial functioning as the fundamental biological mechanism of general intelligence (g). Psychological Review 125: 1028–50. [Google Scholar] [CrossRef] [PubMed]
- Geary, David C. 2019a. Mitochondria as the linchpin of general intelligence and the link between g, health, and aging. Journal of Intelligence 7: 25. [Google Scholar] [CrossRef] [PubMed]
- Geary, David C. 2019b. The spark of life and the unification of intelligence, health, and aging. Current Directions in Psychological Science 28: 223–28. [Google Scholar] [CrossRef]
- Geary, David C. 2021. Male, Female: The Evolution of Human Sex Differences, 3rd ed. Washington, DC: American Psychological Association. [Google Scholar]
- Gignac, Gilles E., and Timothy C. Bates. 2017. Brain volume and intelligence: The moderating role of intelligence measurement quality. Intelligence 64: 18–29. [Google Scholar] [CrossRef]
- Gong, Yun, Jonathan Greenbaum, and Hong-Wen Deng. 2019. A statistical approach to fine-mapping for the identification of potential causal variants related to human intelligence. Journal of Human Genetics 64: 781–87. [Google Scholar] [CrossRef]
- Hamilton, Marcus J., and Robert S. Walker. 2018. A stochastic density-dependent model of long-term population dynamics in hunter-gatherer populations. Evolutionary Ecology Research 19: 85–102. [Google Scholar]
- Havird, Justin C., and Daniel B. Sloan. 2016. The roles of mutation, selection, and expression in determining relative rates of evolution in mitochondrial versus nuclear genomes. Molecular Biology and Evolution 33: 3042–53. [Google Scholar] [CrossRef]
- Hermann, Raphael, Andrea Dolfini, Rachel J. Crellin, Quanyu Wang, and Marion Uckelmann. 2020. Bronze age swordsmanship: New insights from experiments and wear analysis. Journal of Archaeological Method and Theory. [Google Scholar] [CrossRef]
- Hill, Geoffrey E. 2014. Cellular respiration: The nexus of stress, condition, and ornamentation. Integrative and Comparative Biology 54: 645–57. [Google Scholar] [CrossRef]
- Hirschfeld, Katherine. 2015. Gangster States: Organized Crime, Kleptocracy and Political Collapse. New York: Palgrave-MacMillan. [Google Scholar]
- Hulgan, Todd, Asha R. Kallianpur, Yan Guo, Jill S. Barnholtz-Sloan, Haley Gittleman, Todd T. Brown, Ronald Ellis, Scott Letendre, Robert K. Heaton, David C. Samuels, and et al. 2019. Peripheral blood mitochondrial DNA copy number obtained from genome-wide genotype data is associated with neurocognitive impairment in persons with chronic HIV infection. JAIDS Journal of Acquired Immune Deficiency Syndromes 80: e95–102. [Google Scholar] [CrossRef] [PubMed]
- Iozzo, Patricia, and Maria A. Guzzardi. 2019. Imaging of brain glucose uptake by PET in obesity and cognitive dysfunction: Life-course perspective. Endocrine Connections 8: R169–83. [Google Scholar] [CrossRef] [PubMed]
- Jensen, Arthur R. 1998. The g Factor: The Science of Mental Ability. Westport: Praeger. [Google Scholar]
- Johnson, Wendy, Andrew Carothers, and Ian J. Deary. 2008. Sex differences in variability in general intelligence: A new look at the old question. Perspectives on Psychological Science 3: 518–31. [Google Scholar] [CrossRef] [PubMed]
- Jung, Rex E., and Richard J. Haier. 2007. The Parieto-Frontal Integration Theory (P-FIT) of intelligence: Converging neuroimaging evidence. Behavioral and Brain Sciences 30: 135–54. [Google Scholar] [CrossRef] [PubMed]
- Kanazawa, Satoshi. 2004. General intelligence as a domain-specific adaptation. Psychological Review 111: 512–23. [Google Scholar] [CrossRef]
- Kanazawa, Satoshi. 2008. Temperature and evolutionary novelty as forces behind the evolution of general intelligence. Intelligence 36: 99–108. [Google Scholar] [CrossRef]
- Kaplan, Hillard, Kim Hill, Jane Lancaster, and A. Magdelana Hurtado. 2000. A theory of human life history evolution: Diet, intelligence, and longevity. Evolutionary Anthropology 9: 156–85. [Google Scholar] [CrossRef]
- Koch, Rebecca E., Chloe C. Josefson, and Geoffrey E. Hill. 2017. Mitochondrial function, ornamentation, and immunocompetence. Biological Reviews 92: 1459–74. [Google Scholar] [CrossRef]
- Kuczynski, B., W. Jagust, H. C. Chui, and B. Reed. 2009. An inverse association of cardiovascular risk and frontal lobe glucose metabolism. Neurology 72: 738–43. [Google Scholar] [CrossRef]
- Lee, James J., Matt McGue, William G. Iacono, Andrew M. Michael, and Christopher F. Chabris. 2019. The causal influence of brain size on human intelligence: Evidence from within-family phenotypic associations and GWAS modeling. Intelligence 75: 48–58. [Google Scholar] [CrossRef]
- Lee, Ji-Won, Ki D. Park, Jee-Aee Im, Moo Y. Kim, and Duk-Chul Lee. 2010. Mitochondrial DNA copy number in peripheral blood is associated with cognitive function in apparently healthy elderly women. Clinica Chimica Acta 411: 592–96. [Google Scholar] [CrossRef] [PubMed]
- Leonard, William R., and Marcia L. Robertson. 1994. Evolutionary perspectives on human nutrition: The influence of brain and body size on diet and metabolism. American Journal of Human Biology 6: 77–88. [Google Scholar] [CrossRef] [PubMed]
- Levine, Morgan E., Amal Harrati, and Eileen M. Crimmins. 2018. Predictors and implications of accelerated cognitive aging. Biodemography and Social Biology 64: 83–101. [Google Scholar] [CrossRef] [PubMed]
- Longchamps, Ryan J., Christina A. Castellani, Stephanie Y. Yang, Charles E. Newcomb, Jason A. Sumpter, John Lane, Megan L. Grove, Eliseo Guallar, Nathan Pankratz, Kent D. Taylor, and et al. 2020. Evaluation of mitochondrial DNA copy number estimation techniques. PLoS ONE 15: e0228166. [Google Scholar] [CrossRef]
- Mac Arthur, Robert H., and Edward O. Wilson. 1967. The Theory of Island Biogeography. Princeton: Princeton University Press. [Google Scholar]
- Malthus, T. Robert. 1798. An Essay on the Principle of Population as it Affects the Future Improvement of Society with Remarks on the Speculations of Mr. Godwin, M. Condorcet, and Other Writers. London: Printed for J. Johnson, in St. Paul’s church-yard. [Google Scholar]
- Martin, Paul S. 1967. Prehistoric overkill. In The Search for a Cause. Edited by Paul S. Martin and H. E. Wright. New Haven: Yale University Press. [Google Scholar]
- Martin, Paul S. 1973. The discovery of America: The first Americans may have swept the Western Hemisphere and decimated its fauna in 1000 years. Science 179: 969–74. [Google Scholar] [CrossRef]
- Mattson, Mark P. 2012. Energy intake and exercise as determinants of brain health and vulnerability to injury and disease. Cell Metabolism 16: 706–22. [Google Scholar] [CrossRef]
- Matzel, Louis D., Dylan W. Crawford, and Bruno Sauce. 2020. Déjà vu all over again: A unitary biological mechanism for intelligence is (probably) untenable. Journal of Intelligence 8: 24. [Google Scholar] [CrossRef]
- McHenry, Henry M. 1994. Tempo and mode in human evolution. Proceedings of the National Academy of Sciences of the United States of America 91: 6780–86. [Google Scholar] [CrossRef]
- Mengel-From, Jonas, Mikael Thinggaard, Christine Dalgård, Kirsten O. Kyvik, Kaare Christensen, and Lene Christiansen. 2014. Mitochondrial DNA copy number in peripheral blood cells declines with age and is associated with general health among elderly. Human Genetics 133: 1149–59. [Google Scholar] [CrossRef]
- Montier, Laura L. C., Janice J. Deng, and Yidong Bai. 2009. Number matters: Control of mammalian mitochondrial DNA copy number. Journal of Genetics and Genomics 36: 125–31. [Google Scholar] [CrossRef]
- Moore, H. L., A. P. Blain, D. M. Turnbull, and G. S. Gorman. 2020. Systematic review of cognitive deficits in adult mitochondrial disease. European Journal of Neurology 27: 3–17. [Google Scholar] [CrossRef] [PubMed]
- Moore, Heather L., Thomas Kelly, Alexandra Bright, Robert H. Field, Andrew M. Schaefer, Alasdair P. Blain, Robert W. Taylor, Robert McFarland, Doug M. Turnbull, and Gráinne S. Gorman. 2019. Cognitive deficits in adult m. 3243A> G-and m. 8344A> G-related mitochondrial disease: Importance of correcting for baseline intellectual ability. Annals of Clinical and Translational Neurology 6: 826–36. [Google Scholar] [CrossRef] [PubMed]
- Napoli, Nicola, Krupa Shah, Debra L. Waters, David R. Sinacore, Clifford Qualls, and Dennis T. Villareal. 2014. Effect of weight loss, exercise, or both on cognition and quality of life in obese older adults. The American Journal of Clinical Nutrition 100: 189–98. [Google Scholar] [CrossRef] [PubMed]
- Ngandu, Tiia, Jenni Lehtisalo, Alina Solomon, Esko Levälahti, Satu Ahtiluoto, Riitta Antikainen, Lars Bäckman, Tuomo Hänninen, Antti Jula, Tiina Laatikainen, and et al. 2015. A 2 year multidomain intervention of diet, exercise, cognitive training, and vascular risk monitoring versus control to prevent cognitive decline in at-risk elderly people (FINGER): A randomised controlled trial. The Lancet 385: 2255–63. [Google Scholar] [CrossRef]
- O’Brien, Phillipe D., Lucy M. Hinder, Brian C. Callaghan, and Eva L. Feldman. 2017. Neurological consequences of obesity. The Lancet Neurology 16: 465–77. [Google Scholar] [CrossRef]
- Pallier, Gerry, Richard D. Roberts, and Lazar Stankov. 2000. Biological versus psychometric intelligence: Halstead’s (1947) distinction revisited. Archives of Clinical Neuropsychology 15: 205–26. [Google Scholar] [CrossRef]
- Pellerin, Luc, and J. Pierre. 1994. Magistretti. Glutamate uptake into astrocytes stimulates aerobic glycolysis: A mechanism coupling neuronal activity to glucose utilization. Proceedings of the National Academy of Sciences of the United States of America 91: 10625–29. [Google Scholar] [CrossRef]
- Picard, Martin, and Doug M. Turnbull. 2013. Linking the metabolic state and mitochondrial DNA in chronic disease, health, and aging. Diabetes 62: 672–78. [Google Scholar] [CrossRef]
- Picard, Martin, Caroline Trumpff, and Yan Burelle. 2019. Mitochondrial psychobiology: Foundations and applications. Current Opinion in Behavioral Sciences 28: 142–51. [Google Scholar] [CrossRef]
- Picard, Martin, Robert-Paul Juster, and Bruce S. McEwen. 2014. Mitochondrial allostatic load puts The ‘gluc’ back in glucocorticoids. Nature Reviews Endocrinology 10: 303–10. [Google Scholar] [CrossRef]
- Pietschnig, Jakob, and Martin Voracek. 2015. One century of global IQ gains: A formal meta-analysis of the Flynn effect (1909–2013). Perspectives on Psychological Science 10: 282–306. [Google Scholar] [CrossRef] [PubMed]
- Potts, Richard. 1998. Variability selection in hominid evolution. Evolutionary Anthropology 7: 81–96. [Google Scholar] [CrossRef]
- Protzko, John. 2017. Raising IQ among school-aged children: Five meta-analyses and a review of randomized controlled trials. Developmental Review 46: 81–101. [Google Scholar] [CrossRef]
- Pugazhenthi, Subbiah, Limei Qin, and P. Hemachandra Reddy. 2017. Common neurodegenerative pathways in obesity, diabetes, and Alzheimer’s disease. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease 1863: 1037–45. [Google Scholar] [CrossRef] [PubMed]
- Rae, Caroline, Alison L. Digney, Sally R. McEwan, and Timothy C. Bates. 2003. Oral creatine monohydrate supplementation improves brain performance: A double–blind, placebo–controlled, cross–over trial. Proceedings of the Royal Society of London. Series B: Biological Sciences 270: 2147–50. [Google Scholar] [CrossRef]
- Richerson, Peter J., Robert Boyd, and Robert L. Bettinger. 2001. Was agriculture impossible during the Pleistocene but mandatory during the Holocene? A climate change hypothesis. American Antiquity 66: 387–411. [Google Scholar] [CrossRef]
- Sauce, Bruno, and Louis D. Matzel. 2018. The paradox of intelligence: Heritability and malleability coexist in hidden gene-environment interplay. Psychological Bulletin 144: 26–47. [Google Scholar] [CrossRef]
- Savi, Alexander O., Han L. J. van der Maas, Gunter K. Maris, and Maarten Marsman. 2020. Mitochondrial Functioning ≠ general intelligence. Journal of Intelligence 8: 20. [Google Scholar] [CrossRef]
- Schmidt, Reinhold, Helena Schmidt, J. David Curb, Kamal Masaki, Lon R. White, and Lenore J. Launer. 2002. Early inflammation and dementia: A 25-year follow-up of the Honolulu-Asia Aging Study. Annals of Neurology 52: 168–74. [Google Scholar] [CrossRef]
- Schubert, Anna-Lena, and Dirk Hagermann. 2020. The evidence for Geary’s theory on the role of mitochondrial functioning in human intelligence is not entirely convincing. Journal of Intelligence 8: 29. [Google Scholar] [CrossRef]
- Set, Kallol K., Kuntal Sen, A. H. M. Huq, and Rajkumar Agarwal. 2019. Mitochondrial disorders of the nervous system: A review. Clinical Pediatrics 58: 381–94. [Google Scholar] [CrossRef] [PubMed]
- Shemesh, Elad, Assaf Rudich, Ilana Harman-Boehm, and Tali Cukierman-Yaffe. 2012. Effect of intranasal insulin on cognitive function: A systematic review. The Journal of Clinical Endocrinology & Metabolism 97: 366–76. [Google Scholar] [CrossRef]
- Shields, Grant S., Wesley G. Moons, and George M. Slavich. 2017. Inflammation, self-regulation, and health: An immunologic model of self-regulatory failure. Perspectives on Psychological Science 12: 588–612. [Google Scholar] [CrossRef]
- Silzer, Talisa, Robert Barber, Jie Sun, Gita Pathak, Leigh Johnson, Sid O’Bryant, and Nicole Phillips. 2019. Circulating mitochondrial DNA: New indices of type 2 diabetes-related cognitive impairment in Mexican Americans. PloS ONE 14: e0213527. [Google Scholar] [CrossRef] [PubMed]
- Singh-Manoux, Archana, Aline Dugravot, Eric Brunner, Meena Kumari, Martin Shipley, Alexis Elbaz, and Mika Kivimaki. 2014. Interleukin-6 and C-reactive protein as predictors of cognitive decline in late midlife. Neurology 83: 486–93. [Google Scholar] [CrossRef] [PubMed]
- Smith, Patrick J., James A. Blumenthal, Benson M. Hoffman, Harris Cooper, Timothy A. Strauman, Kathleen Welsh-Bohmer, Jeffrey N. Browndyke, and Andrew Sherwood. 2010. Aerobic exercise and neurocognitive performance: A meta-analytic review of randomized controlled trials. Psychosomatic Medicine 72: 239–52. [Google Scholar] [CrossRef]
- Spitznagel, Mary B., Misty Hawkins, Michael Alosco, Rachel Galioto, Sarah Garcia, Lindsay Miller, and John Gunstad. 2015. Neurocognitive effects of obesity and bariatric surgery. European Eating Disorders Review 23: 488–95. [Google Scholar] [CrossRef]
- Spyridaki, Eirini C., Panagiotis Simos, Pavlina D. Avgoustinaki, Eirini Dermitzaki, Maria Venihaki, Achilles N. Bardos, and Andrew N. Margioris. 2014. The association between obesity and fluid intelligence impairment is mediated by chronic low-grade inflammation. British Journal of Nutrition 112: 1724–34. [Google Scholar] [CrossRef]
- Spyridaki, Eirini C., Pavlina D. Avgoustinaki, and Andrew N. Margioris. 2016. Obesity, inflammation and cognition. Current Opinion in Behavioral Sciences 9: 169–75. [Google Scholar] [CrossRef]
- Sripetchwandee, Jirapas, Nipon Chattipakorn, and Siriporn C. Chattipakorn. 2018. Links between obesity-induced brain insulin resistance, brain mitochondrial dysfunction, and dementia. Frontiers in Endocrinology 9: 496. [Google Scholar] [CrossRef]
- Stankov, Lazar. 2020. “Turtles-all-the-way-down” from g to mitochondrial functioning. Journal of Intelligence 8: 23. [Google Scholar] [CrossRef] [PubMed]
- Sternberg, Robert J. 2020. How mighty are the mitochondria in causing individual differences in intelligence?—Some questions for David Geary. Journal of Intelligence 8: 13. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, Anna J., Daniel L. McCartney, Robert F. Hillary, Paul Redmond, Adele M. Taylor, Qian Zhang, and et al. 2019. Childhood intelligence attenuates the association between biological ageing and health outcomes in later life. Translational Psychiatry 9: 1–8. [Google Scholar] [CrossRef] [PubMed]
- Trumble, Benjamin C., and Caleb E. Finch. 2019. The exposome in human evolution: From dust to diesel. The Quarterly Review of Biology 94: 333–94. [Google Scholar] [CrossRef] [PubMed]
- Tucker-Drob, Elliot M., Andreas M. Brandmaier, and Ulman Lindenberger. 2019. Coupled cognitive changes in adulthood: A meta-analysis. Psychological Bulletin 145: 273–301. [Google Scholar] [CrossRef] [PubMed]
- Turchin, Peter. 2009. A theory for formation of large empires. Journal of Global History 4: 191–217. [Google Scholar] [CrossRef]
- Turchin, Peter, Thomas E. Currie, Edward A. Turner, and Sergey Gavrilets. 2013. War, space, and the evolution of Old World complex societies. Proceedings of the National Academy of Sciences of the United States of America 110: 16384–389. [Google Scholar] [CrossRef]
- Ujma, Péter Przemyslaw, and Kristof Kovacs. 2020. The mitochondrial theory of g is incompatible with genetic evidence and does not explain statistical phenomena. Journal of Intelligence 8: 27. [Google Scholar] [CrossRef]
- Veronese, Nicola, Silvia Facchini, Brendon Stubbs, Claudio Luchini, Marco Solmi, Enzo Manzato, Giuseppe Sergi, Stefania Maggi, Theodore Cosco, and Luigi Fontana. 2017. Weight loss is associated with improvements in cognitive function among overweight and obese people: A systematic review and meta-analysis. Neuroscience & Biobehavioral Reviews 72: 87–94. [Google Scholar] [CrossRef]
- Volkow, Nora D., Gene-Jack Wang, Frank Telang, Joanna S. Fowler, Rita Z. Goldstein, Nelly Alia-Klein, and et al. 2009. Inverse association between BMI and prefrontal metabolic activity in healthy adults. Obesity 17: 60–65. [Google Scholar] [CrossRef]
- Vrba, Elizabeth S. 1995. The fossil record of African antelopes (Mammalia, Bovidae) in relation to human evolution and paleoclimate. In Paleoclimate and Evolution, with Emphasis on Human Origins. Edited by Elizabeth S. Vrba, George H. Denton, Timothy C. Partridge and Lloyd H. Burckle. New Haven: Yale University Press. [Google Scholar]
- Wachsmuth, Manja, Alexander Huebner, Mingkun Li, Burkhard Madea, and Mark Stoneking. 2016. Age-related and heteroplasmy-related variation in human mtDNA copy number. PLoS Genetics 12: e1005939. [Google Scholar] [CrossRef] [PubMed]
- Warne, Russell T., and Cassidy Burningham. 2019. Spearman’s g found in 31 non-Western nations: Strong evidence that g is a universal phenomenon. Psychological Bulletin 145: 237–72. [Google Scholar] [CrossRef]
- Whitley, Elise, Catharine R. Gale, Ian J. Deary, Mika Kivimaki, and G. David Batty. 2011. Association of maternal and paternal IQ with offspring conduct, emotional, and attention problem scores: transgenerational evidence from the 1958 British birth cohort study. Archives of General Psychiatry 68: 1032–38. [Google Scholar] [CrossRef] [PubMed]
- Willette, Auriel A., Barbara B. Bendlin, Erika J. Starks, Alex C. Birdsill, Sterling C. Johnson, Bradley T. Christian, Ozioma C. Okonkwo, Asenath La Rue, Bruce P. Hermann, Rebecca L. Koscik, and et al. 2015. Association of insulin resistance with cerebral glucose uptake in late middle–aged adults at risk for Alzheimer disease. JAMA Neurology 72: 1013–20. [Google Scholar] [CrossRef]
- Wingo, Aliza P., Eric B. Dammer, Michael S. Breen, Benjamin A. Logsdon, Duc M. Duong, Juan C. Troncosco, Madhav Thambisetty, Thomas G. Beach, Geidy E. Serrano, Eric M. Reiman, and et al. 2019. Large-scale proteomic analysis of human brain identifies proteins associated with cognitive trajectory in advanced age. Nature Communications 10: 1–14. [Google Scholar] [CrossRef] [PubMed]
- Yaffe, Kristene, Alka Kanaya, Karla Lindquist, Eleanor M. Simonsick, Tamara Harris, Ronald I. Shorr, and et al. 2004. The metabolic syndrome, inflammation, and risk of cognitive decline. JAMA 292: 2237–42. [Google Scholar] [CrossRef]
- Yang, Yingkai, Grant S. Shields, Cheng Guo, and Yanling Liu. 2018. Executive function performance in obesity and overweight individuals: A meta-analysis and review. Neuroscience & Biobehavioral Reviews 84: 225–44. [Google Scholar] [CrossRef]
- Zeng, Tian C., Alan J. Aw, and Marcus W. Feldman. 2018. Cultural hitchhiking and competition between patrilineal kin groups explain the post-Neolithic Y-chromosome bottleneck. Nature Communications 9: 2077. [Google Scholar] [CrossRef]
- Zerjal, Tatiana, Yali Xue, Giorgio Bertorelle, R. Spencer Wells, Weidong Bao, Suling Zhu, Raheel Qamar, Qasim Ayub, Aisha Mohyuddin, Songbin Fu, and et al. 2003. The genetic legacy of the Mongols. American Journal of Human Genetics 72: 717–21. [Google Scholar] [CrossRef]
| Authors | Core Critiques | Key Reply Points |
|---|---|---|
| Burgoyne and Engle | a. How can the hypothesis be falsified? b. What is the effect size directly related to variation in mitochondrial (mt) functioning? | a1. Strategies to falsify the hypothesis are in Table 1 of Geary (2018, p. 1030). See also Section 2.2 and Section 3. a2. Unknown; some proportion of the variance associated with the g factor. |
| Debatin | a. Systems approach to neuroenergetics is more comprehensive. | a1. Agreed, but mitochondrial functioning is central to this system. This is now noted in the introduction. |
| Matzel et al. | a. Multiple lower-order systems influence brain and cognition. b. Variation in energy production is not sufficient to place constraints on brain and cognition. c. Not sufficient variation in mtDNA genes to create a bottleneck in energy production. d. Processes closer to (e.g., reaction time) mt should be more predictive of intelligence. e. Should not smarter people should run faster? f. Should not intelligence be more strongly correlated with mothers’ than fathers’ intelligence? | a1. Correct, but their functioning is dependent on cellular energy production. b1. Probably true for many young people in wealthy and healthy populations. b2. Probably not true with normal aging, various health conditions, nutritional deficits, parasite and toxin exposure, and myriad other stressors that are common outside of Western middle-class populations. c1. Most mitochondrial functions are dependent on nuclear not mitochondrial genes. d. The prediction is that the most complex processes will be the better predictor. e. No. Efficient energy production will only help if there is also sufficient muscle mass and mix of slow- and fast-twitch muscle fiber. f. No. Most mt genes are nuclear and inherited from both parents (Section 2.2). |
| Savi et al. | a. There are cyclical biological mechanisms other than mt. b. mt place a ceiling on brain functions but the ceiling might never be reached. c. This is verbal speculation. d. A dynamic, network approach to intelligence is preferable. | a. True but their operation is dependent on energy availability. b. True in many cases. However, the ceiling appears to drop with normal aging and disorders that effect mt functions or substrates and may be raised with some interventions. c. Section 3 outlines ways to test the hypothesis. d. This is not incompatible with mt; the more complex the network the more energy needed to build and maintain it. |
| Schubert and Hagemann | a. Many cognitive systems rely on energy consuming long-distance brain networks and remain stable or gain with aging, such as vocabulary. b. Are intervention studies of healthy adults supportive of the hypothesis? c. Should not intelligence be more strongly correlated with mothers’ than fathers’ intelligence? | a. True, but retrieving a vocabulary definition, for example, has lower prefrontal engagement than using vocabulary knowledge to solve analogies. The differences in resource demands should be differentially compromised, with normal aging; the latter more than the former. b. See Section 3. c. No. Most mt genes are nuclear and inherited from both parents (Section 2.2). |
| Stankov | a. Broad theories of intelligence are preferable to g, which is weaker than stated. b. It is important to identify diseases that directly involve mt and directly link these to cognition. c. Strong claims regarding mt and cognitive aging are premature. | a. Agreed that the study of cognition must include multiple abilities, but a substantial g factor remains important. b. Agreed. See Section 3. c. Agreed. This is a proposed mechanism, not a theoretical edict. |
| Sternberg | a. Correlational and not causal model. b. Are individual differences in intelligence related to mt for any given age? c. One’s living environments are important for cognition. d. What is the evolutionary advantage of intelligence? | a1. Agreed that much of the evidence is correlation. a2. Methods to assess a causal relation are described in Section 3. b. They could be, if people vary in factors that influence mt functioning; See Section 3. c. True, as described in Section 2.1.3. d. See Section 4. |
| Ujma and Kovacs | a. Genetic evidence does not strongly implicate mt functioning in intelligence. b. There is no straightforward link between complexity of cognitive tasks and mt functioning. | a1. These studies are focused on cognitive phenotypes and thus are biased toward identifying higher-level systems. a2. The bottom-up focus on mt gene-product proteins reveals a relation between mt and cognition; see Section 2.2.1. b. Correct. This is an hypothesis and some methods in Section 3 might be used to test it. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Geary, D.C. Mitochondrial Functions, Cognition, and the Evolution of Intelligence: Reply to Commentaries and Moving Forward. J. Intell. 2020, 8, 42. https://doi.org/10.3390/jintelligence8040042
Geary DC. Mitochondrial Functions, Cognition, and the Evolution of Intelligence: Reply to Commentaries and Moving Forward. Journal of Intelligence. 2020; 8(4):42. https://doi.org/10.3390/jintelligence8040042
Chicago/Turabian StyleGeary, David C. 2020. "Mitochondrial Functions, Cognition, and the Evolution of Intelligence: Reply to Commentaries and Moving Forward" Journal of Intelligence 8, no. 4: 42. https://doi.org/10.3390/jintelligence8040042
APA StyleGeary, D. C. (2020). Mitochondrial Functions, Cognition, and the Evolution of Intelligence: Reply to Commentaries and Moving Forward. Journal of Intelligence, 8(4), 42. https://doi.org/10.3390/jintelligence8040042





