Effect of Monomer Mixture Composition on TiCl4-Al(i-C4H9)3 Catalytic System Activity in Butadiene–Isoprene Copolymerization: A Theoretical Study
Abstract
1. Introduction
- Under the influence of hydrodynamic factors (turbulent velocity pulsations in the reaction system, which cause significant shear stresses within it [24]) and chemical factors (propagation of polymer chains in the pores of catalyst particles, which wedges these particles apart [25]), the particles of the catalytic system undergo dispersion. Dispersion of the catalytic system particles during polymerization exposes previously hidden active surfaces [26,27].
- Changes in temperature and reaction time shift the equilibrium between active sites of different types [26].
- Introduction of comonomers accelerates activation by creating additional, more reactive sites [28].
- Complex mechanism of active sites formation. For example, a two-step mechanism for active sites formation has been described, whereby initially a limited number of active sites are formed, and then, as the catalytic system particles disperse and their surface reorganizes, active sites of new types appear [28,29].
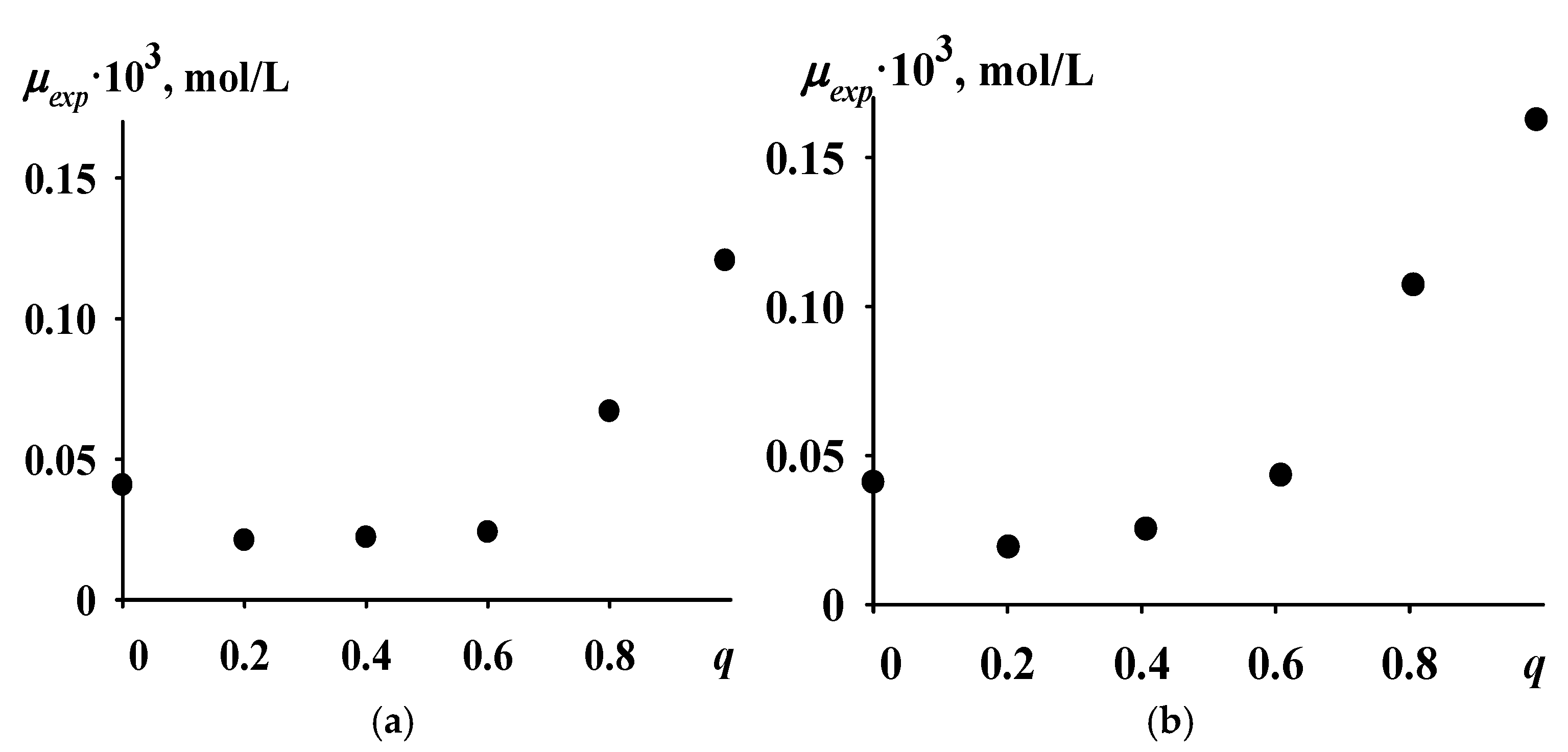
- The concentrations of active sites (μ) of the TiCl4-Al(i-C4H9)3 catalytic system during the butadiene–isoprene copolymerization were calculated at various monomer mixture compositions (q) based on experimental data on the copolymerization rate taken from [5]. Here, q = [M1]/([M1] + [M2]), [M1] is the concentration of butadiene in the monomer mixture, [M2] is the concentration of isoprene in the monomer mixture, q = 1 corresponds to the homopolymerization of butadiene, and q = 0 corresponds to the homopolymerization of isoprene (here and below, […]—denotes concentration) These calculated concentrations are hereafter referred to as experimental and denoted as μexp.
- Based on the Langmuir monomolecular adsorption theory [33] and the mass-action law, a kinetic model for the formation of active sites in the catalytic system was developed. As a result of the analytical solution of the system of kinetic equations, an equation was obtained which established a direct functional relationship between the concentration of active sites μ and the composition of the monomer mixture q. The concentration values calculated using this equation are hereafter referred to as calculated concentrations and denoted as μcalc.
- An analysis of the obtained equation was performed to determine the ranges of kinetic parameters of the active sites’ formation process for which the dependence of μcalc on q matches, in shape, the dependence of μexp on q.
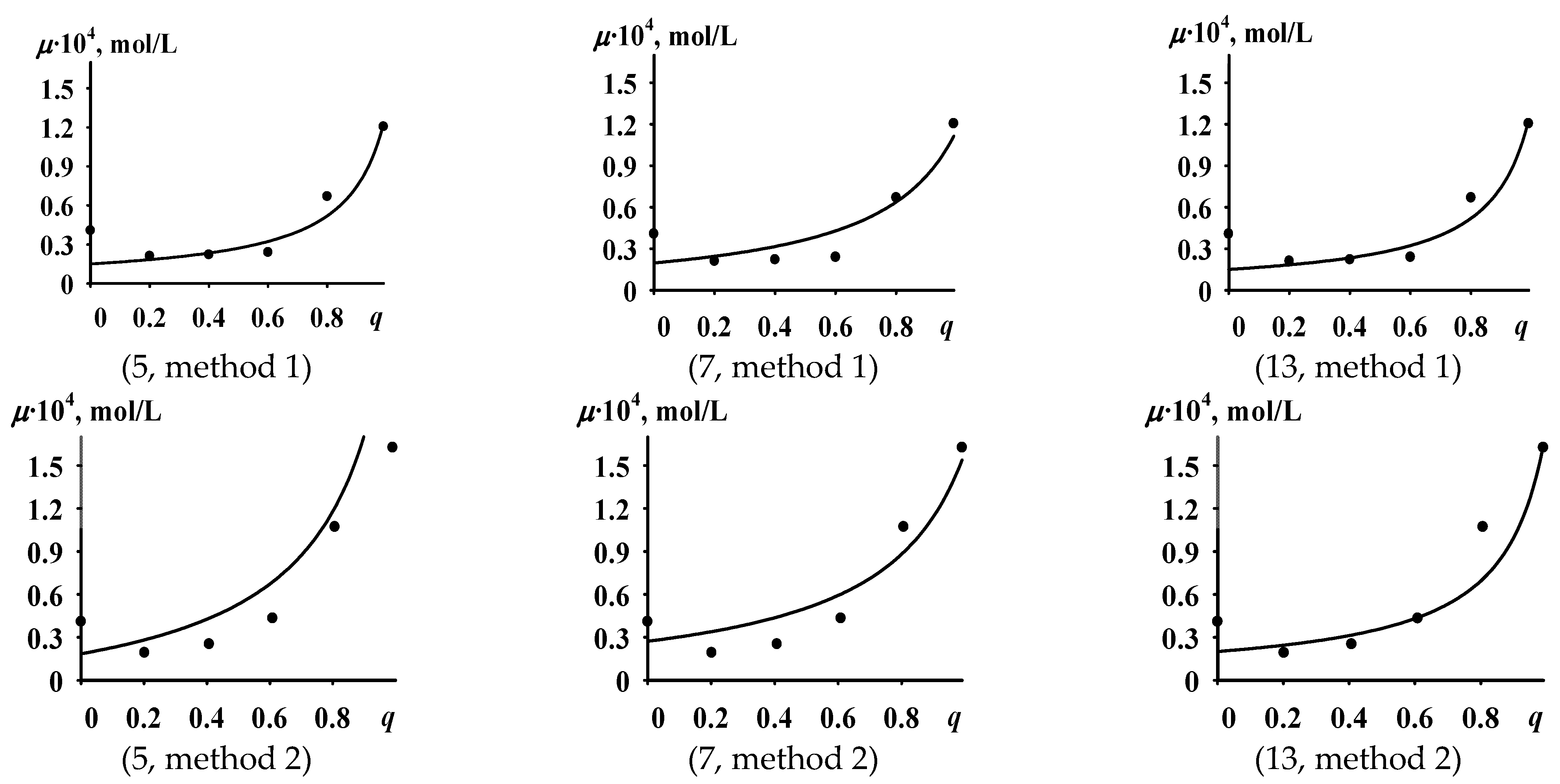
- Based on the results of solving the third task, specific quantitative values of the kinetic parameters of the active sites’ formation process were determined within the allowable parameter ranges, for which the dependence of μcalc on q quantitatively coincides with the dependence of μexp on q.
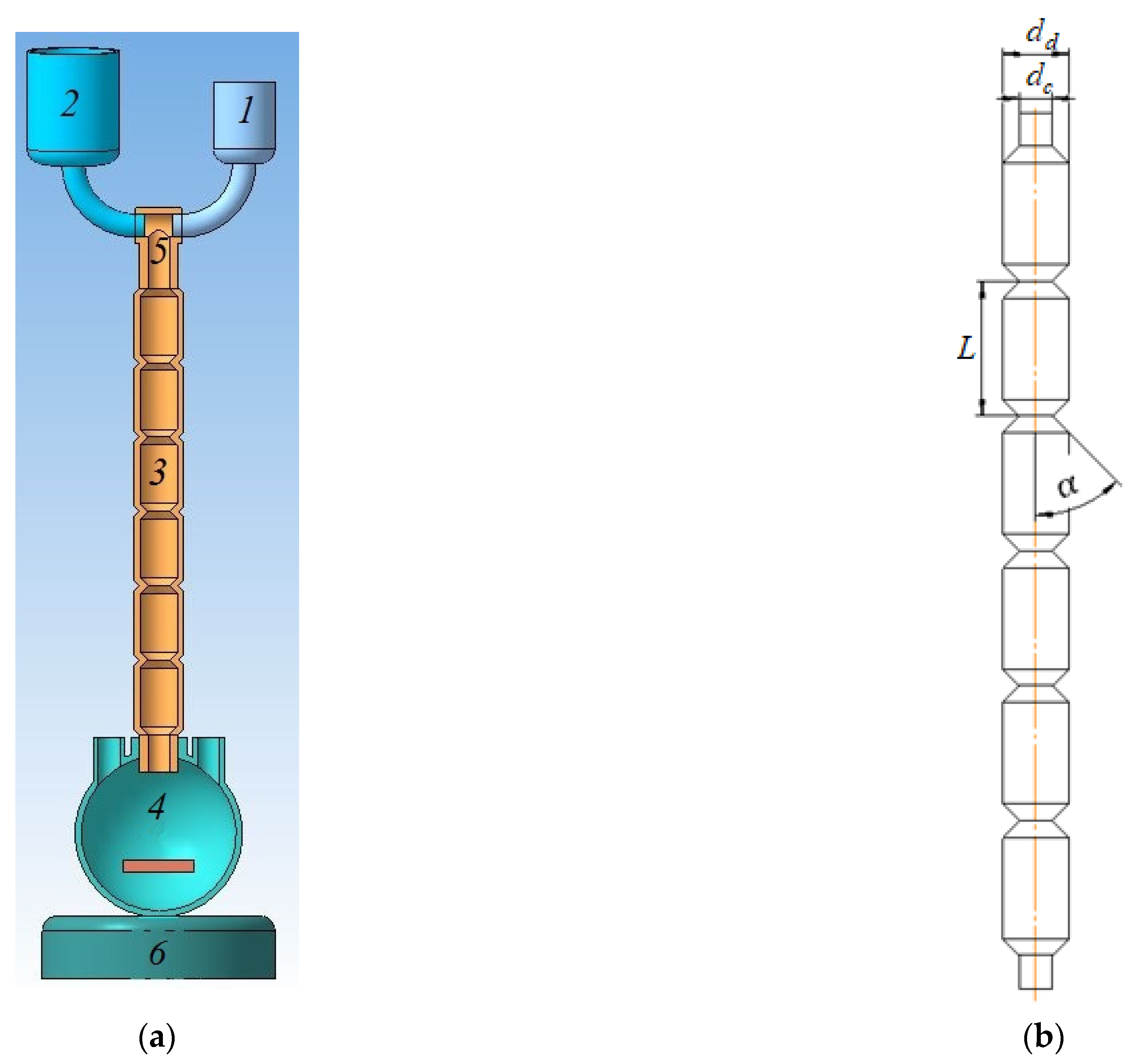
2. Materials and Methods
3. Results
3.1. Calculation of the Concentration of Active Sites of the TiCl4-Al(i-C4H9)3 Catalytic System
3.2. Development of a Kinetic Model for the Formation of Active Sites in the TiCl4-Al(i-C4H9)3 Catalytic System
- Adsorption occurs at adsorption sites on the surface of the adsorbent. In this case, the adsorbent consists of particles of the TiCl4-Al(i-C4H9)3 catalytic system. The adsorbate comprises molecules of butadiene or isoprene. Adsorption is competitive, meaning that butadiene and isoprene molecules adsorb at the same adsorption sites. An active site is formed after the adsorption of a monomer molecule onto the surface of the catalytic system particles and the formation of a Ti-C bond, where C is the carbon atom of the butadiene or isoprene molecule.
- Each adsorption site can adsorb only one adsorbate molecule.
- The adsorption process is reversible and at equilibrium, with its rate determined by the rate of reaching equilibrium. The formation of the Ti-C bond is also assumed to be reversible.
- Interaction between adsorbate molecules in the adsorbed state is absent.
3.3. Determination of the Kinetic Parameter Range for the Active Sites Formation Process Based on the Shape of the Dependence of Active Sites’ Concentration on Monomer Mixture Composition q
- 1.
- —the adsorption/desorption equilibrium of butadiene is shifted toward adsorption, i.e., butadiene is adsorbed by adsorption sites with high efficiency.
- 2.
- —the adsorption/desorption equilibrium of butadiene is shifted toward desorption, i.e., butadiene is adsorbed by adsorption sites with low efficiency.
- 3.
- —the adsorption/desorption equilibrium of isoprene is shifted toward adsorption, i.e., isoprene is adsorbed by adsorption sites with high efficiency.
- 4.
- —the adsorption/desorption equilibrium of isoprene is shifted toward desorption, i.e., isoprene is adsorbed by adsorption sites with low efficiency.
- 5.
- —the equilibrium of formation/breaking of the bond between the Ti atom of the catalytic system and the C atom of butadiene is shifted toward bond formation, i.e., butadiene forms stable active sites.
- 6.
- —the equilibrium of formation/breaking of the bond between the Ti atom of the catalytic system and the C atom of butadiene is shifted toward bond breaking, i.e., butadiene forms unstable active sites.
- 7.
- —the equilibrium of formation/breaking of the bond between the Ti atom of the catalytic system and the C atom of isoprene is shifted toward bond formation, i.e., isoprene forms stable active sites.
- 8.
- —the equilibrium of formation/breaking of the bond between the Ti atom of the catalytic system and the C atom of isoprene is shifted toward bond breaking, i.e., isoprene forms unstable active sites.
3.4. Determination of Specific Quantitative Values of Kinetic Parameters of the Active Sites Formation Process, at Which the Dependence of μcalc on q Quantitatively Coincides with the Dependence of μexp on q
- No solutions were found in subsets 1, 8, 15, and 16 that provided quantitatively exact agreement between the dependencies μexp on q and μcalc on q (specifically, none were found because they might exist if other initial approximations within these subsets were used);
- Local minima were found in subsets 5 and 13 with values (F = 2.81 × 10−8 and F = 1.54 × 10−8 respectively);
- No local minimum was found in subset 7 (!), despite the fact that the global minimum found in Variant 1 minimization lies in subset 7. This is probably due to the fact that during the optimization algorithm implementation, when searching for the global minimum, the values of Xf1, Xf2, X1, X2 initially located in subset 7 leave it and then re-enter it; this supports the idea that solutions in subsets 1, 8, 15, and 16 may exist but were not found with the chosen initial approximations.
- 1.
- Monomer molecules are easily adsorbed on the surface of the TiCl4-Al(i-C4H9)3 catalytic system particles; active sites formed by butadiene molecules are stable (i.e., the bond between the Ti atom of the catalytic system and the C atom of butadiene is stable), while active sites s formed by isoprene molecules are unstable (i.e., the bond between the Ti atom of the catalytic system and the C atom of isoprene is unstable). This description corresponds to subset 5 ( ).
- 2.
- Isoprene molecules are easily adsorbed, while butadiene molecules are adsorbed with difficulty; active sites formed by butadiene are stable, and active sites formed by isoprene are unstable. This description corresponds to subset 7 ( ).
- 3.
- Monomer molecules are easily adsorbed but form unstable active sites; the stability of active sites formed by butadiene should be higher than that of active sites formed by isoprene. This description corresponds to subset 13 ( ).
4. Conclusions
- 1.
- Based on experimental values of the copolymerization rate from study [5] and known values of chain propagation rate constants [35,36] and copolymerization constants from study [5], the concentrations of active sites of the TiCl4-Al(i-C4H9)3 catalytic system were calculated at various values of q. The experimental dependence of μ on q has the following features: 1. The minimum concentration of active sites μ corresponds to some value of q in the range q = 0.2–0.6, and the concentration μ predominantly increases as the butadiene concentration in the monomer mixture increases. 2. The dependence of μ on q is nonlinear, with ().
- 2.
- The kinetic model equations for the formation of active sites in the TiCl4-Al(i-C4H9)3 catalytic system were written. These kinetic equations were formulated based on the mass-action law and the Langmuir monomolecular adsorption theory for two variants of the kinetic scheme: one-stage and two-stage (physical stage—adsorption; chemical stage—formation of the Ti-C bond). The adsorption of butadiene and isoprene molecules on the catalytic system TiCl4-Al(i-C4H9)3 surface was considered competitive. The equation expressing the theoretical dependence of μ on q was obtained by analytically solving the system of kinetic model equations under the assumption of equilibrium in all stages of the formation of active sites.
- 3.
- The obtained equation was analyzed. It was found that the kinetic model based on the one-stage kinetic scheme cannot even qualitatively describe the experimental dependence of μ on q with the described features. The analogous kinetic model based on the two-stage kinetic scheme satisfactorily describes this experimental dependence (except for the existence of the minimum concentration of active sites μ).
- 4.
- The domains of kinetic parameter values for active sites formation were established, at which the theoretical dependence of μ on q reproduces the corresponding experimental dependence both qualitatively and quantitatively. Qualitatively, this occurs under the condition that isoprene adsorbs better than butadiene, but butadiene forms more stable active sites than isoprene. Quantitatively, this is ensured by any of the three sets of equilibrium rate constants for the stages of active sites formation found in this work.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bhatia, S.; Goel, A. Rubber Technology; Woodhead Publishing India: New Delhi, India, 2019; p. 666. [Google Scholar]
- Kotnees, D.; Bhomick, A. Rubber to Rubber Adhesion; Scrivener Publishing: Austin, TX, USA, 2021; p. 429. [Google Scholar]
- Wang, F.; Zhang, M.; Liu, H.; Hu, Y.; Zhang, X. Randomly Coordinative Chain Transfer Copolymerization of 1,3-Butadiene and Isoprene: A Highly Atom Economic Way for Accessing Butadiene/Isoprene Rubber. Ind. Eng. Chem. Res. 2020, 59, 10754–10762. [Google Scholar] [CrossRef]
- Taibulatov, P.A.; Mingaleev, V.Z.; Zakharov, V.P.; Ionova, I.A.; Monakov, Y.B. Kinetic Inhomogeneity of Copolymerization Sites of Butadiene and Isoprene on Titanium Catalyst. Pol. Sci. Ser. B 2010, 52, 450–458. [Google Scholar] [CrossRef][Green Version]
- Zakharov, V.P.; Ulitin, N.V.; Tereshchenko, K.A.; Zakharova, E.M. Turbulent Technologies in the Synthesis of Polydienes Using Ziegler-Natta Catalytic Components; Bashkir Encyclopedia: Ufa, Russia, 2018; p. 280. (In Russian) [Google Scholar]
- Liu, L.; Meira, D.M.; Arenal, R.; Concepción, P.; Puga, A.V.; Corma, A. Determination of the Evolution of Heterogeneous Single Metal Atoms and Nanoclusters under Reaction Conditions: Which Are the Working Catalytic Sites? ACS Catal. 2019, 9, 10626–10639. [Google Scholar] [CrossRef]
- Lu, Y.; Kuo, C.-T.; Kovarik, L.; Hoffman, A.S.; Boubnov, A.; Driscoll, D.M.; Morris, J.R.; Bare, S.R.; Karim, A.M. A Versatile Approach for Quantification of Surface Site Fractions using Reaction Kinetics: The Case of CO Oxidation on Supported Ir single Atoms and Nanoparticles. J. Catal. 2019, 378, 121–130. [Google Scholar] [CrossRef]
- Rana, R.; Hong, J.; Hoffman, A.S.; Werghi, B.; Bare, S.R.; Kulkarni, A.R. Quantifying the Site Heterogeneities of Non-Uniform Catalysts Using QuantEXAFS. Chem.–Meth. 2024, 5, e202400020. [Google Scholar] [CrossRef]
- DeRita, L.; Dai, S.; Lopez-Zepeda, K.; Pham, N.; Graham, G.W.; Pan, X.; Christopher, P. Catalyst Architecture for Stable Single Atom Dispersion Enables Site-Specific Spectroscopic and Reactivity Measurements of CO Adsorbed to Pt Atoms, Oxidized Pt Clusters, and Metallic Pt Clusters on TiO2. J. Am. Chem. Soc. 2017, 139, 14150–14165. [Google Scholar] [CrossRef]
- Bates, J.S.; Martinez, J.J.; Hall, M.N.; Al-Omari, A.A.; Murphy, E.; Zeng, Y.; Luo, F.; Primbs, M.; Menga, D.; Bibent, N.; et al. Chemical Kinetic Method for Active-Site Quantification in Fe-N-C Catalysts and Correlation with Molecular Probe and Spectroscopic Site-Couning Methods. J. Am. Chem. Soc. 2023, 145, 26222–26237. [Google Scholar] [CrossRef]
- Sahraie, N.R.; Kramm, U.I.; Steinberg, J.; Zhang, Y.; Thomas, A.; Reier, T.; Paraknowitsch, J.P.; Strasser, P. Quantifying the density and utilization of active sites in non-precious metal oxygen electroreduction catalysts. Nat. Commun. 2015, 6, 8618. [Google Scholar] [CrossRef] [PubMed]
- Jeong, B.; Abbas, H.G.; Klein, B.P.; Bae, G.; Velmurugan, A.R.; Choi, C.H.; Kim, G.; Kim, D.; Kim, K.-J.; Cha, B.J.; et al. CO Cryo-Sorption as a Surface-Sensitive Spectroscopic Probe of the Active Site Density of Single-Atom Catalysts. Angew. Chem. Int. Ed. 2025, 64, e202420673. [Google Scholar] [CrossRef]
- Boyes, E.D.; LaGrow, A.P.; Ward, M.R.; Mitchell, R.W.; Gai, P.L. Single Atom Dynamics in Chemical Reactions. Acc. Chem. Res. 2020, 53, 390–399. [Google Scholar] [CrossRef]
- Afrin, S.; Bollini, P. Beyond Upper Bound Estimates of Active Site Densities in Heterogeneous Catalysis: A Note on the Critical Role of Titrant Pressure. J. Catal. 2022, 413, 76–80. [Google Scholar] [CrossRef]
- Credendino, R.; Liguori, D.; Fan, Z.; Morini, G.; Cavallo, L. Toward a Unified Model Explaining Heterogeneous Ziegler−Natta Catalysis. ACS Catal. 2015, 5, 5431–5435. [Google Scholar] [CrossRef]
- Bahri-Laleha, N.; Hanifpour, A.; Mirmohammadi, S.A.; Poater, A.; Nekoomanesh-Haghighi, M.; Talarico, G.; Cavallo, L. Computational Modeling of Heterogeneous Ziegler-Natta Catalysts for Olefins Polymerization. Prog. Polym. Sci. 2018, 84, 89–114. [Google Scholar] [CrossRef]
- Kissin, Y.V. Active Centers in Ziegler–Natta Catalysts: Formation Kinetics and Structure. J. Catal. 2012, 292, 188–200. [Google Scholar] [CrossRef]
- Shetty, S. Synergistic, Reconstruction and Bonding Effects During the Adsorption of Internal Electron Donors and TiCl4 on MgCl2 Surface: A Periodic-DFT Investigation. Surf. Sci. 2016, 653, 55–65. [Google Scholar] [CrossRef]
- Bazvand, R.; Bahri-Laleh, N.; Abedini, H.; Nekoomanesh, M.; Poater, A. Chemical dealcoholation of MgCl2·EtOH Adduct by Al Compounds and Its Effect on the Performance of Ziegler–Natta Catalysts. Appl. Organomet. Chem. 2024, 38, e7300. [Google Scholar] [CrossRef]
- Mehdizadeh, M.; Karkhaneh, F.; Nekoomanesh, M.; Sadjadi, S.; Emami, M.; Teimoury, H.; Salimi, M.; Solà, M.; Poater, A.; Bahri-Laleh, N.; et al. Influence of the Ethanol Content of Adduct on the Comonomer Incorporation of Related Ziegler–Natta Catalysts in Propylene (Co)polymerizations. Polymers 2023, 15, 4476. [Google Scholar] [CrossRef] [PubMed]
- Vittoria, A.; Meppelder, A.; Friederichs, N.; Busico, V.; Cipullo, R. Demystifying Ziegler-Natta Catalysts: The Origin of Stereoselectivity. ACS Catal. 2017, 7, 4509–4518. [Google Scholar] [CrossRef]
- Rahmatiyan, S.; Bahri-Laleh, N.; Hanifpour, A.; Nekoomanesh-Haghighi, M. Different Behaviors of Metallocene and Ziegler–Natta Catalysts in Ethylene/1,5-hexadiene Copolymerization. Polym. Int. 2018, 68, 94–101. [Google Scholar] [CrossRef]
- Masoori, M.; Nekoomanesh, M.; Posada-Pérez, S.; Rashedi, R.; Bahri-Laleh, N. Exploring Cocatalyst Type Effect on the Ziegler–Natta Catalyzed Ethylene Polymerizations: Experimental and DFT Studies. J. Polym. Res. 2022, 29, 197. [Google Scholar] [CrossRef]
- Tereshchenko, K.A.; Shiyan, D.A.; Ziganshina, A.S.; Ganiev, G.M.; Ulitin, N.V.; Zakharov, V.P. Control of Molar Mass Characteristics of Polybutadiene—A Component of Sticky Glue—By Physical Modification of the Catalytic System in Turbulent Flows. Polym. Sci. Ser. D 2020, 13, 250–257. [Google Scholar] [CrossRef]
- Koen, W.B.; Laurens, D.B.M.; Nikolaos, N.; Yuanshuai, L.; Marcus, R.; de Peinder, P.; Bas, J.P.T.; Felix, W.; Joren, M.D.; Thomas, H.; et al. A Ziegler-type Spherical Cap Model Reveals Early Stage Ethylene Polymerization Growth versus Catalyst Fragmentation Relationships. Nat. Commun. 2022, 13, 4954. [Google Scholar] [CrossRef]
- Wei-Ping, Z.; Ya-Ping, M.; Da-Lin, D.; Ai-Hua, H.; Hua-Feng, S.; Chen-Guang, L. Polymerization Kinetics of Propylene with the MgCl2-Supported Ziegler-Natta Catalysts—Active Centers with Different Tacticity and Fragmentation of the Catalyst. Chin. J. Polym. Sci. 2020, 39, 70–80. [Google Scholar] [CrossRef]
- Maximilian, J.W.; Florian, M.; Bert, M.W. Visualizing the Structure, Composition and Activity of Single Catalyst Particles for Olefin Polymerization and Polyolefin Decomposition. Angew. Chem. Int. Ed. 2024, 63, e202306033. [Google Scholar] [CrossRef]
- Khan, A.; Guo, Y.; Zhang, Z.; Ali, A.; Fu, Z.; Fan, Z. Kinetics of Short-Duration Ethylene-Propylene Copolymerization with MgCl2-supported Ziegler–Natta catalyst: Differentiation of Active Centers on the External and Internal Surfaces of the Catalyst Particles. J. Appl. Polym. Sci. 2018, 135, 46030. [Google Scholar] [CrossRef]
- Shiri, M.; Parvazinia, M.; Yousefi, A.A.; Bahri-Laleh, N.; Poater, A. A Novel Method for Dynamic Molecular Weight Distribution Determination in Organometallic Catalyzed Olefin Polymerizations. Catalysts 2022, 12, 1130. [Google Scholar] [CrossRef]
- Kissin, Y.V.; Marin, V.P.; Nelson, P.J. Propylene Polymerization Reactions with Supported Ziegler–Natta Catalysts: Observing Polymer Material Produced by a Single Active Center. J. Polym. Sci. Part A Polym. Chem. 2017, 55, 3832–3841. [Google Scholar] [CrossRef]
- Zhang, B.; Qian, Q.; Yang, P.; Jiang, B.; Fu, Z.; Fan, Z. Responses of a Supported Ziegler–Natta Catalyst to Comonomer Feed Ratios in Ethylene–Propylene Copolymerization: Differentiation of Active Centers with Different Catalytic Features. Ind. Eng. Chem. Res. 2021, 60, 4575–4588. [Google Scholar] [CrossRef]
- Maity, B.; Cao, Z.; Kumawat, J.; Gupta, V.; Cavallo, L. A Multivariate Linear Regression Approach to Predict Ethene/1-Olefin Copolymerization Statistics Promoted by Group 4 Catalysts. ACS Catal. 2021, 11, 4061–4070. [Google Scholar] [CrossRef]
- Murzin, D.; Salmi, T. Chapter 2—Catalysis. In Catalytic Kinetics Chemistry and Engineering, 2nd ed.; Elsevier B.V.: Amsterdam, The Netherlands, 2016; pp. 35–100. [Google Scholar] [CrossRef]
- Monakov, Y.; Sigaeva, N.; Urazbaev, N. Active Sites of Polymerization: Multiplicity: Stereospecific and Kinetic Heterogeneity; Brill Academic Publishers: Leiden, The Netherlands, 2005; p. 397. [Google Scholar]
- Tereshchenko, K.A.; Ziganshina, A.S.; Zakharov, V.P.; Ulitin, N.V. Modeling of the Physicochemical Hydrodynamics of the Synthesis of Butadiene Rubber on the TiCl4−Al(i-C4H9)3 Catalytic System Modified in Turbulizing Flows. Russ. J. Phys. Chem. B 2017, 11, 504–512. [Google Scholar] [CrossRef]
- Ganiev, G.M.; Tereshchenko, K.A.; Shiyan, D.A.; Ziganshina, A.S.; Zakharov, V.P.; Ulitin, N.V. Relationship of Molecular-Mass Characteristics of Polyisoprene, Component of Vulcanized Sealant, with Particle Sizes of Catalytic System TiCl4-Al(i-C4H9)3 in Isoprene Polymerization. Polym. Sci., Ser. D 2021, 14, 392–395. [Google Scholar] [CrossRef]
- Wu, Y.; Ding, M.; Wang, J.; Zhao, B.; Wu, Z.; Zhao, P.; Tian, D.; Ding, Y.; Hu, A. Controlled Step-Growth Polymerization. CCS Chem. 2020, 2, 64–70. [Google Scholar] [CrossRef]
- Fazakas-Anca, I.S.; Modrea, A.; Vlase, S. Determination of Reactivity Ratios from Binary Copolymerization Using the k-Nearest Neighbor Non-Parametric Regression. Polymers 2021, 13, 3811. [Google Scholar] [CrossRef]
- Zakharov, V.P.; Zakharova, E.M.; Nasyrov, I.S.; Zhavoronkov, D.A. Use of a Turbulent Prereactor for Affecting the Site Multiplicity of a Titanium Catalyst for (Co)polymerization of Butadiene and Isoprene. Russ. J. Appl. Chem. 2014, 87, 613–618. [Google Scholar] [CrossRef]
- Vigdorowitsch, M.; Pchelintsev, A.; Tsygankova, L.; Tanygina, E. Freundlich Isotherm: An Adsorption Model Complete Framework. Appl. Sci. 2021, 11, 8078. [Google Scholar] [CrossRef]
- Al-Ghouti, M.A.; Da’ana, D.A. Guidelines for the Use and Interpretation of Adsorption Isotherm Models: A Review. J. Hazard. Mater. 2020, 393, 122383. [Google Scholar] [CrossRef] [PubMed]
- Sips, R. On the Structure of a Catalyst Surface. J. Chem. Phys. 1948, 16, 490–495. [Google Scholar] [CrossRef]
- Sips, R. On the structure of catalyst surface. II. J. Chem. Phys. 1950, 18, 1024–1026. [Google Scholar] [CrossRef]
- Van Tassel, P.R.; Davis, H.T.; Mccormick, A.V. New Lattice Model for Adsorption of Small molecules in Zeolite Micropores. AIChE J. 1994, 40, 925–934. [Google Scholar] [CrossRef]
- Pazzona, F.G.; Demontis, P.; Suffritti, G.B. Thermodynamics of the One-dimensional Parallel Kawasaki Model: Exact Solution and Mean-field Approximations. Phys. Rev. E 2014, 90, 022118. [Google Scholar] [CrossRef]
- Fowler, R.; Guggenheim, E. Stat. Thermodyn. Macmillan Inc.; Cambridge University Press: Cambridge, UK, 1956; p. 701. [Google Scholar]
- Barbero, G.; Evangelista, L.R.; Lelidis, I. Effective Adsorption Energy and Generalization of the Frumkin-Fowler-Guggenheim isotherm. J. Mol. Liq. 2020, 327, 114795. [Google Scholar] [CrossRef]
- Cabrera, M.I.; Grau, R.J. Methyl Oleate Isomerization and Hydrogenation over Ni/α-Al2O3: A Kinetic Study Recognizing Differences in the Molecular Size of Hydrogen and Organic Species. J. Mol. Catal. A Chem. 2008, 287, 24–32. [Google Scholar] [CrossRef]
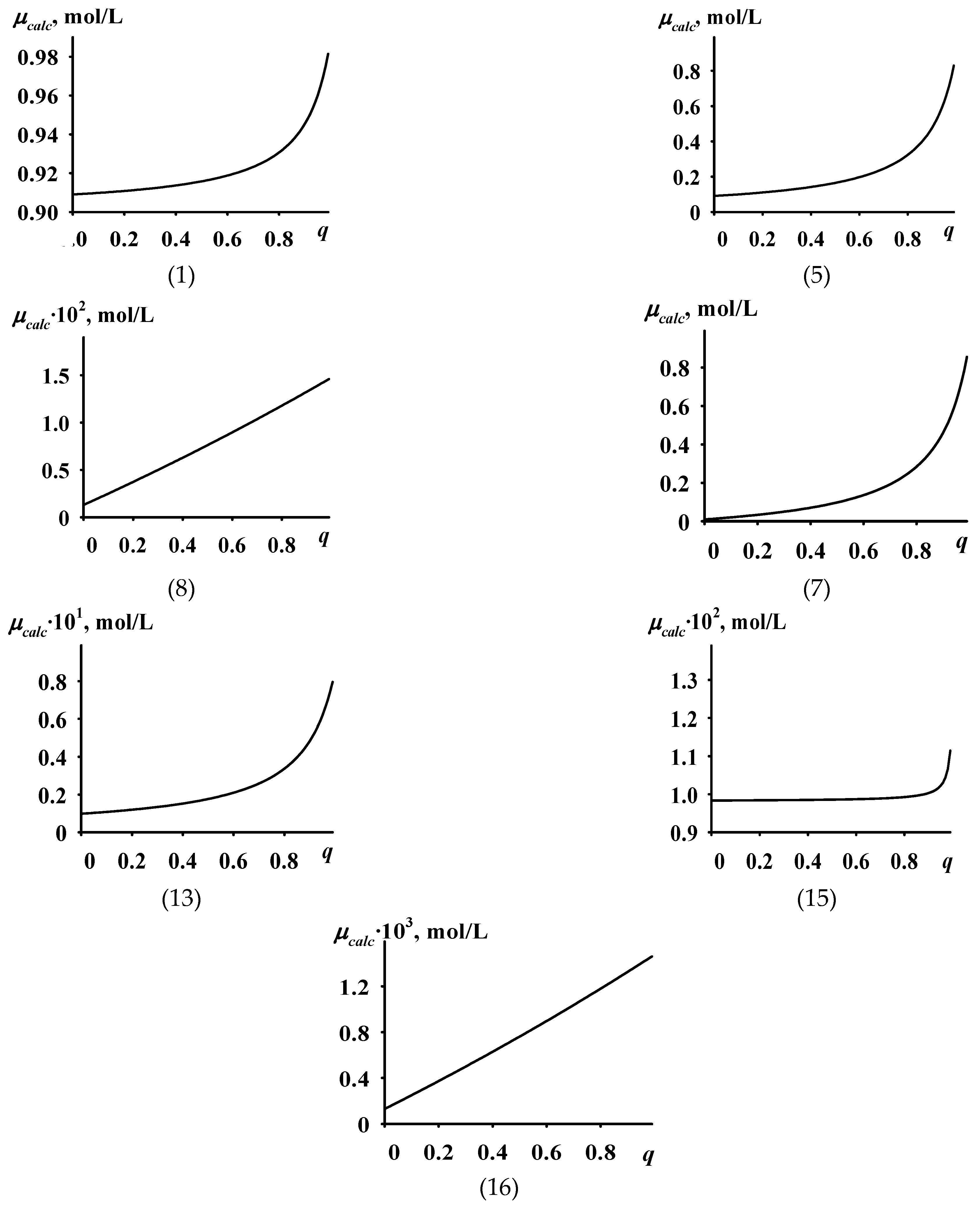
| Boundary of the Subset (Stage 1) | Kf1[M] >> 1, Kf2[M] >> 1 | Kf1[M] >> 1, Kf2[M] << 1 | Kf1[M] << 1, Kf2[M] >> 1 | Kf1[M] << 1, Kf2[M] << 1 | |
|---|---|---|---|---|---|
| Boundary of the Subset (Stage 2) | |||||
| K1>> 1, K2 >> 1 | subset 1 K1/K2 > 1, K2/K1 > Kf1/Kf2 | subset 2 K1/K2 > Kf2[M], K2/K1 > Kf1/Kf2 | subset 3 K1/K2 > 1/(Kf1[M]), K2/K1 > Kf1/Kf2 | subset 4 K1/K2 > Kf2/Kf1, K2/K1 > Kf1/Kf2 | |
| K1>> 1, K2 << 1 | subset 5 K1/K2 > 1, 1/K1 > Kf1/Kf2 | subset 6 K1/K2 > Kf2[M], 1/K1 > Kf1/Kf2 | subset 7 K1/K2 > 1/(Kf1[M]), 1/K1 > Kf1/Kf2 | subset 8 K1/K2 > Kf2/Kf1, 1/K1 > Kf1/Kf2 | |
| K1 << 1, K2 >> 1 | subset 9 K1/K2 > 1, K2 > Kf1/Kf2 | subset 10 K1/K2 > Kf2[M], K2 > Kf1/Kf2 | subset 11 K1/K2 > 1/(Kf1[M]), K2 > Kf1/Kf2 | subset 12 K1/K2 > Kf2/Kf1, K2 > Kf1/Kf2 | |
| K1<< 1, K2 << 1 | subset 13 K1/K2 > 1, 1 > Kf1/Kf2 | subset 14 K1/K2 > Kf2[M], 1 > Kf1/Kf2 | subset 15 K1/K2 > 1/(Kf1[M]), 1 > Kf1/Kf2 | subset 16 K1/K2 > Kf2/Kf1, 1 > Kf1/Kf2 | |
| Boundaries of Parameter Subsets | Kf1[M] >> 1, Kf2[M] >> 1 | Kf1[M] >> 1, Kf2[M] << 1 | Kf1[M] << 1, Kf2[M] >> 1 | Kf1[M] << 1, Kf2[M] << 1 |
|---|---|---|---|---|
| K1>> 1, K2 >> 1 | No solutions | No solutions | No solutions | |
| K1>> 1, K2 << 1 | No solutions | |||
| K1 << 1, K2 >> 1 | No solutions | No solutions | No solutions | No solutions |
| K1<< 1, K2 << 1 | No solutions |
| Boundaries of Subsets of Parameter Values | ||||
|---|---|---|---|---|
| Qualitative solution exists; no exact quantitative solution found | No solutions | No solutions | No solutions | |
(method 1 *) (method 2) , local minimum F = 2.81 × 10−8 | No solutions | (method 1) (method 2) , global minimum F = 4.42 × 10−9 | Qualitative solution exists; no exact quantitative solution found | |
| No solutions | No solutions | No solutions | No solutions | |
(method 1) (method 2) local minimum F = 1.54 × 10−8 | No solutions | Qualitative solution exists, no exact quantitative solution found | Qualitative solution exists, no exact quantitative solution found |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tereshchenko, K.A.; Ismagilov, R.T.; Ulitin, N.V.; Lyulinskaya, Y.L.; Novikov, A.S. Effect of Monomer Mixture Composition on TiCl4-Al(i-C4H9)3 Catalytic System Activity in Butadiene–Isoprene Copolymerization: A Theoretical Study. Computation 2025, 13, 184. https://doi.org/10.3390/computation13080184
Tereshchenko KA, Ismagilov RT, Ulitin NV, Lyulinskaya YL, Novikov AS. Effect of Monomer Mixture Composition on TiCl4-Al(i-C4H9)3 Catalytic System Activity in Butadiene–Isoprene Copolymerization: A Theoretical Study. Computation. 2025; 13(8):184. https://doi.org/10.3390/computation13080184
Chicago/Turabian StyleTereshchenko, Konstantin A., Rustem T. Ismagilov, Nikolai V. Ulitin, Yana L. Lyulinskaya, and Alexander S. Novikov. 2025. "Effect of Monomer Mixture Composition on TiCl4-Al(i-C4H9)3 Catalytic System Activity in Butadiene–Isoprene Copolymerization: A Theoretical Study" Computation 13, no. 8: 184. https://doi.org/10.3390/computation13080184
APA StyleTereshchenko, K. A., Ismagilov, R. T., Ulitin, N. V., Lyulinskaya, Y. L., & Novikov, A. S. (2025). Effect of Monomer Mixture Composition on TiCl4-Al(i-C4H9)3 Catalytic System Activity in Butadiene–Isoprene Copolymerization: A Theoretical Study. Computation, 13(8), 184. https://doi.org/10.3390/computation13080184







