Contact-Less Real-Time Monitoring of Cardiovascular Risk Using Video Imaging and Fuzzy Inference Rules
Abstract
1. Introduction
2. Materials and Methods
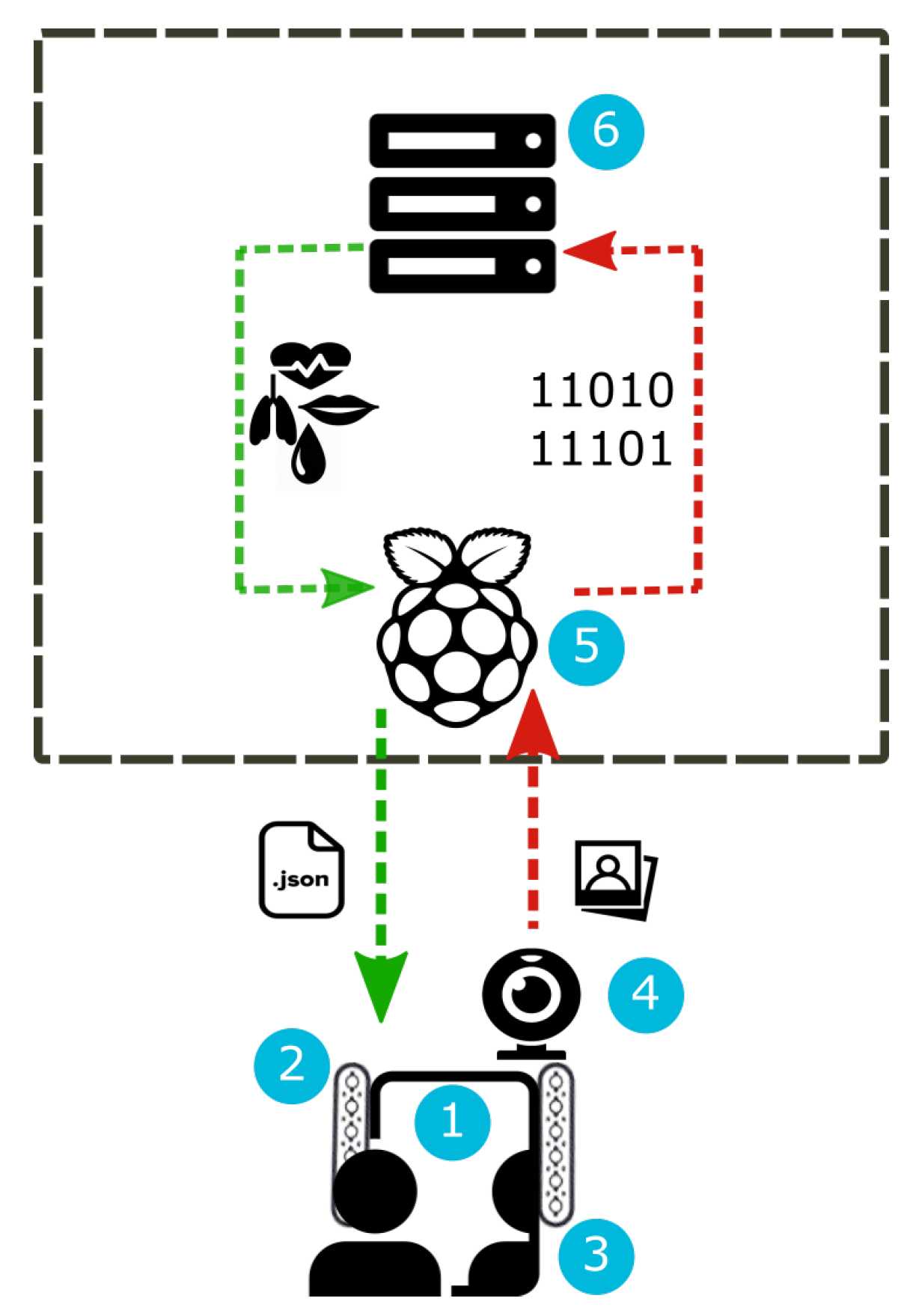
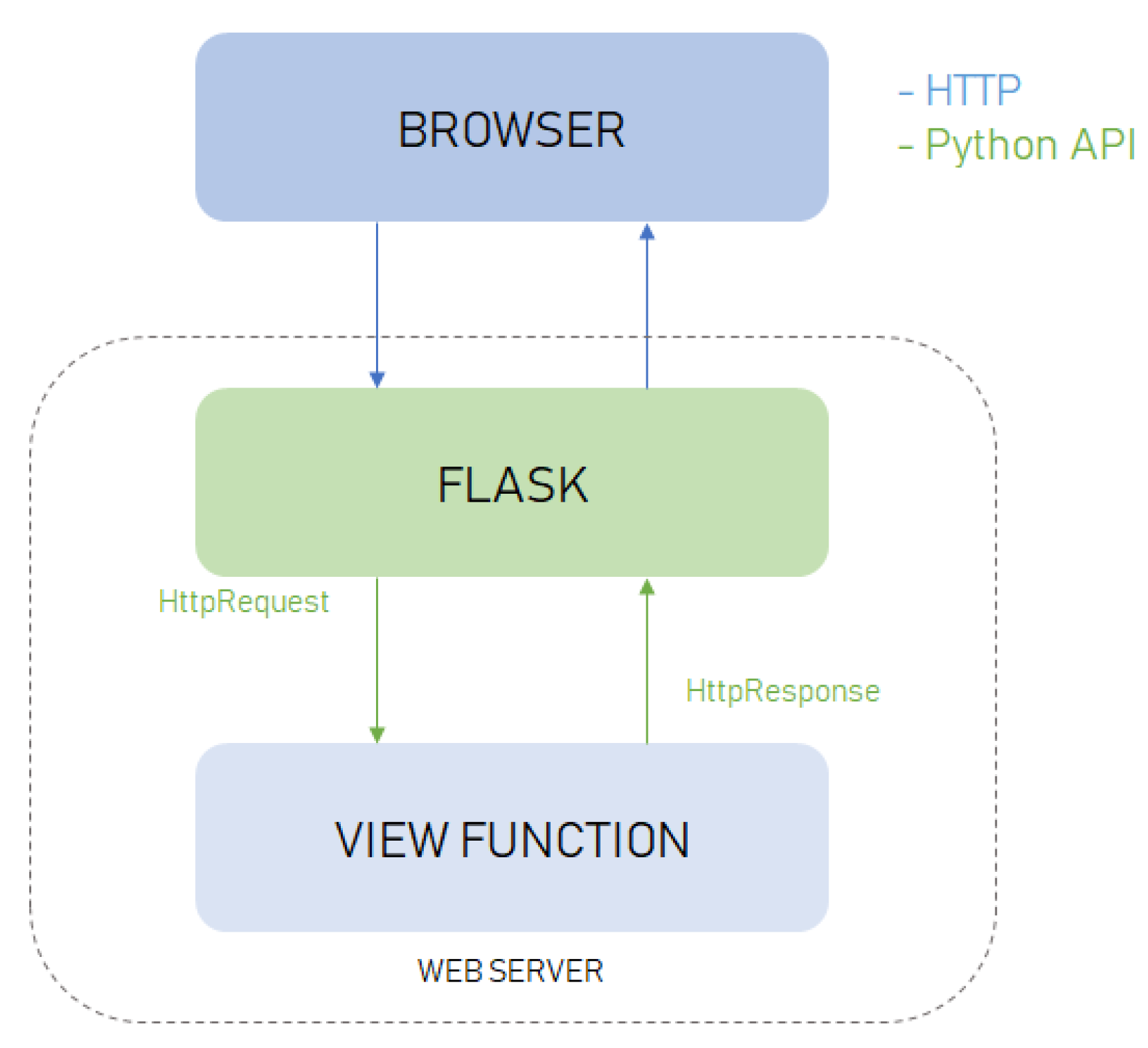
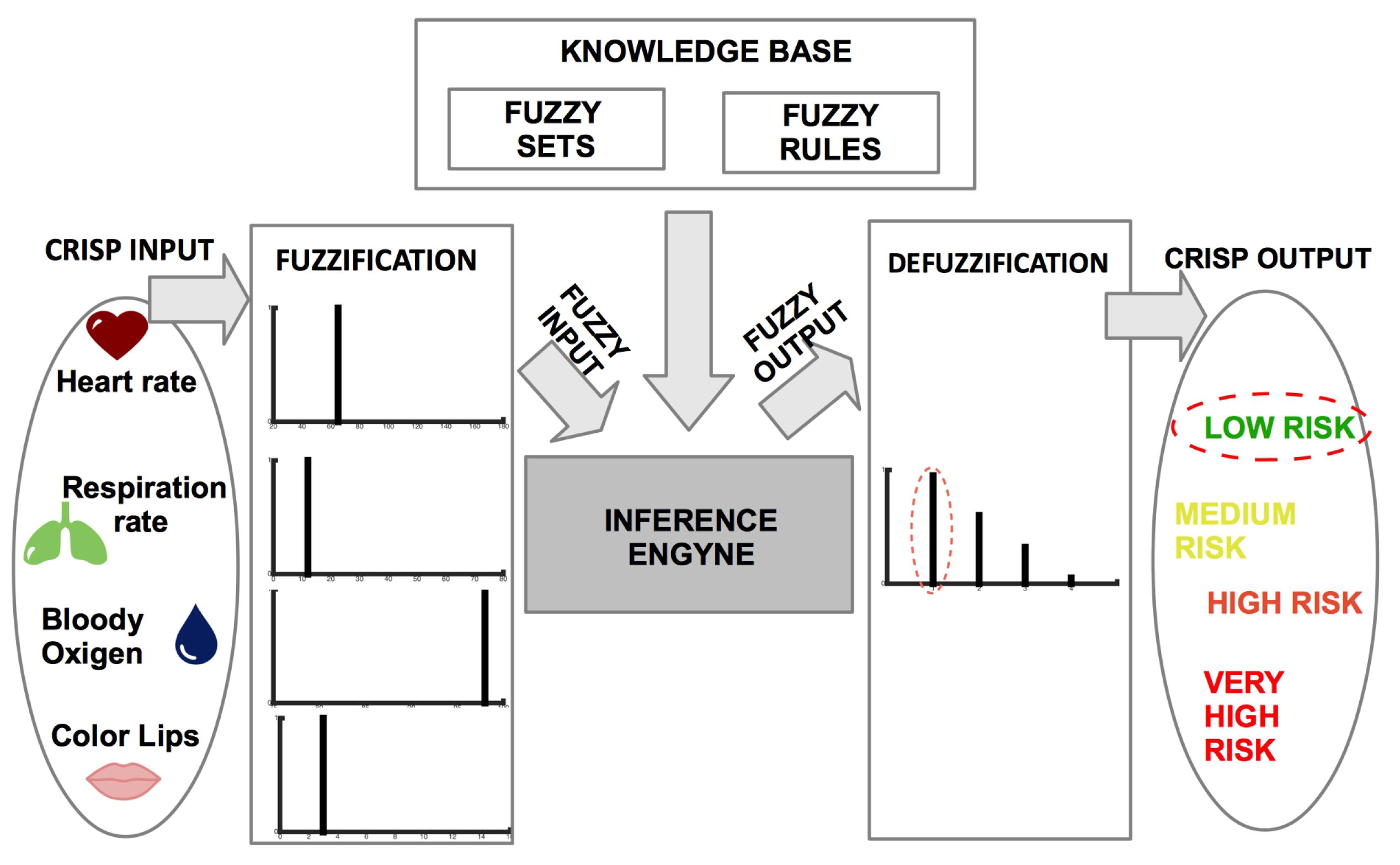
2.1. Architecture
2.2. Evaluation of Vital Parameters
- 1.
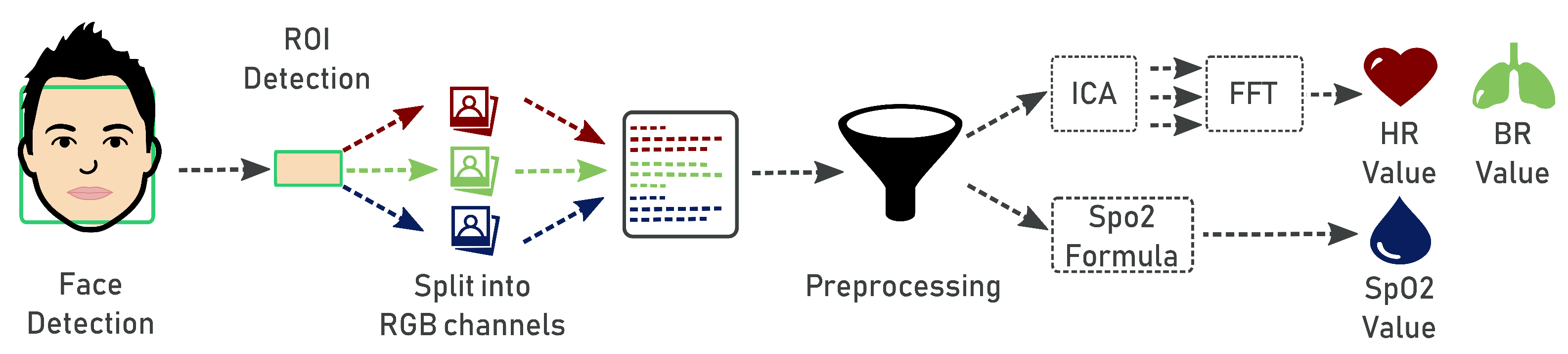
- The first step is face detection. The frames are acquired in real-time through the camera and represented in 24-bit RGB color with 3 channels × 8 bits/channel with resolution of pixel. At the start, we perform a preliminary acquisition cycle of 40 s. This is necessary to avoid the initial distortion due to the start of the camera. Afterwards, the camera acquires a video frame every 5 s. The frame acquisition phase was developed using the WebRTC (Web Real-Time Communications) API (https://webrtc.org/). WebRTC is a technology that allows applications and web pages to capture audio or video streams and to exchange data among different browsers, being a cross-platform technology. To detect the face within the video frames, we used the pre-trained frontal face detector available with the Python library Dlib (http://dlib.net/). This detector is based on a modified linear support vector machine (SVM) combined with histogram of oriented gradients (HOG) [18]. Given the detected face, we apply a predictive model that provides a set of 68 facial landmarks given in the form of coordinates. This model was obtained in [19] by training an ensemble of regression trees on images from the iBUG 300-W dataset (https://ibug.doc.ic.ac.uk/resources/300-W/).
- 2.
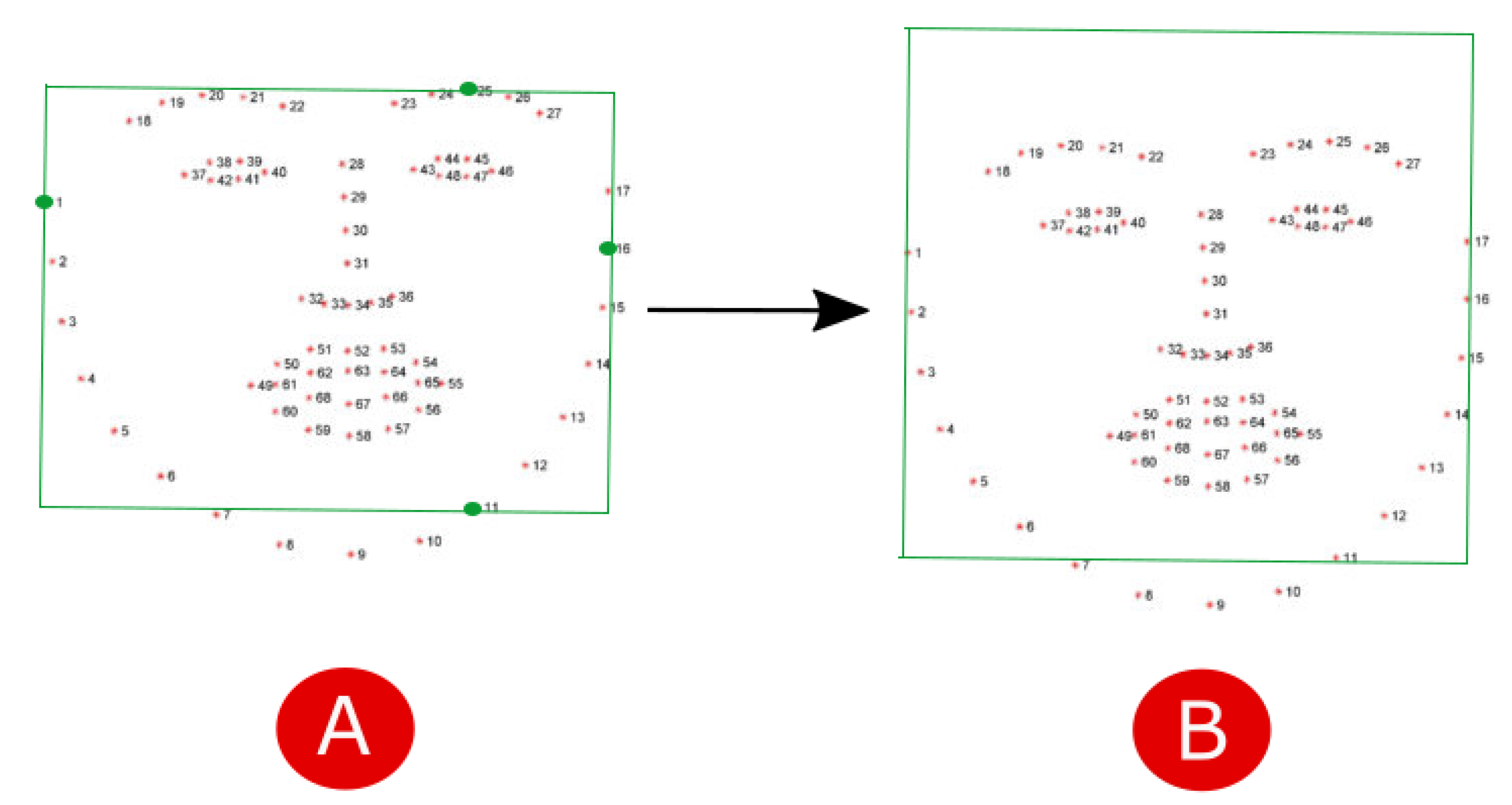
- In the second step, given the face region identified by landmarks, we localize the region of interest (ROI) corresponding to a region with a strong passage of blood modulation, so as to enable evaluation of heart rate, breathing and oxygen saturation by means of photoplethysmography. According to [20], the most suitable areas for the detection of heart rate are the forehead and cheeks. To simplify the computation we decided to consider only the forehead. A rectangle centered in the forehead of size pixels was considered. To locate the ROI, we firstly construct a bounding box including the face by considering the landmarks 1-11-16-25 as vertexes (Figure 4A). Then, in order to include the forehead, we build an enlarged rectangle by augmenting the height of the bounding box of 20 pixels (Figure 4B).
- 3.
- The next step is the analysis of the signals coming from the forehead ROI marked in each frame. The ROI is separated into the three RGB channels and spatially averaged over all pixels to yield a red, blue and green measurement value for each frame. The acquired average signals are composed of N data, being N the number of frames, hence we obtain a matrix with dimensions.
- 4.
- Since the values in the RGB components are easily disturbed by the motion and fluctuations in image lightness, they can not be directly used for the skin color analysis. Indeed, the signals coming from the detected ROI pick up a mixture of the reflected plethysmographic signal along with other sources of fluctuations in light due to artifacts such as motion and changes in ambient lighting conditions. Therefore, the noisy signals are improved through a preprocessing phase involving linear interpolation, detrend [21], normalization and band-pass filtering. Since high-frequency noise may occur in the image signals, we apply a filter based on the frequency characteristics of the heart rate to reduce the disturbance. The adopted frequency of the band-pass filter was fixed between 0.6 and 4 Hz (corresponding to heart rate [36, 240]/bpm) for heart rate estimation.
- 5.
- In order to evaluate the hearth rate and the breath rate, the independent source signals from each multivariate signal (i.e., color channel) should be uncovered. To do this, we apply Independent Component Analysis (ICA) [22] as a blind source separation technique. In conventional ICA, the number of recoverable sources cannot exceed the number of observations, thus we assume three underlying source signals [23]. We use the FastICA method available in the Scikit-learn Python library (http://scikit-learn.org/stable/). Then, the spectral analysis of the components is applied in order to represent the distribution of the power of the signal itself. To perform spectral analysis, we use the Fast Fourier Transform (FFT) implemented in the Python Numpy library (http://www.numpy.org/). In order to select the component containing the strongest signal, we apply the periodogram function that calculates the spectral density of each component. Given the selected component, we find the frequency corresponding to the maximum peak’s intensity in the same band of the filter , which corresponds to the range [51 to 210 bpm] of the heart rate. Likewise, we find the frequency corresponding to the maximum peak’s intensity in the same band of the filter , which corresponds to the range [9 to 30 bpm] of the breath rate. Finally, we obtain the value of the heart rate () and breath rate (), meant as average beats per minute, by multiplying the frequency values and by 60, namely and .
- 6.
- Blood saturation indicates the intensity of oxygen in blood and is defined as:where is the oxygenated hemoglobin, and is the deoxygenated hemoglobin. According to [24], an estimation of can be obtained by considering the cardiovascular pulse wave signal at two different wavelengths, hence, by comparing the red and blue bands, the blue band representative of the infrared wavelength being used in traditional pulse oximeter calculations. The standard deviations of the red and blue bands at each time point are used as the pulsatile alternating current () component, while the non-pulsatile direct current () component is computed as the red and blue band mean intensities at each time point. Oxygen saturation is then computed as follows:where we fix and according to the empirical evaluation made in [24].
2.3. Risk Assessment by Fuzzy Rules
3. Application and Results
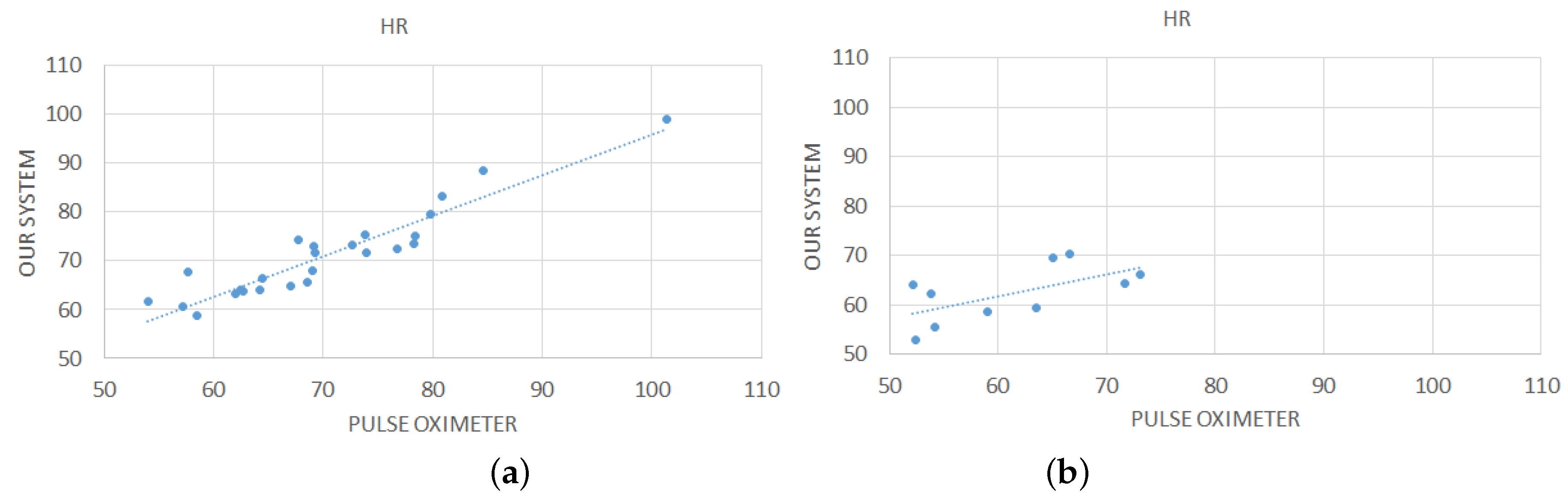
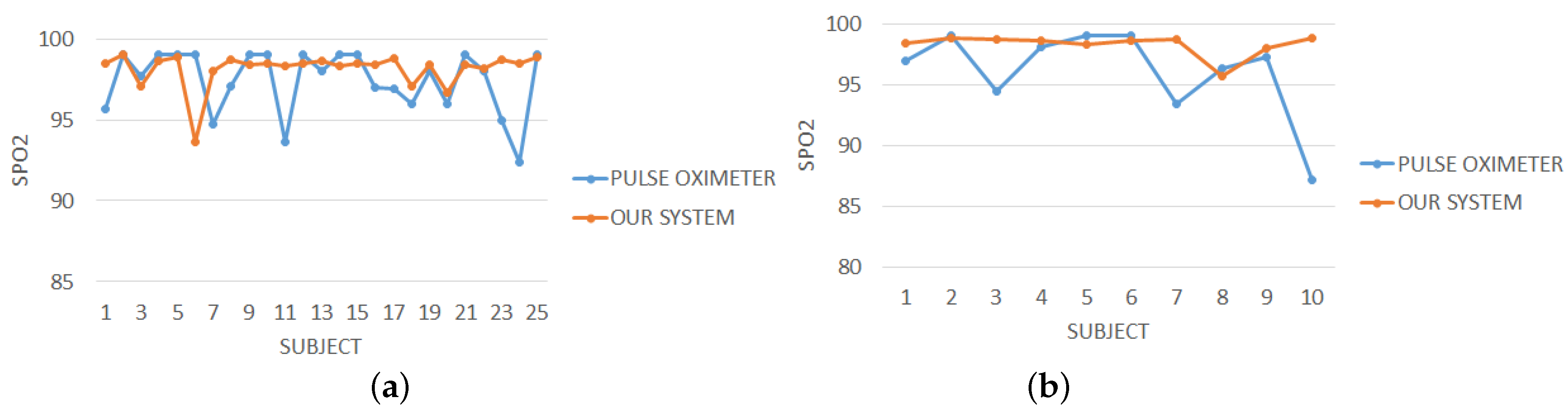
3.1. Evaluation of Vital Parameters
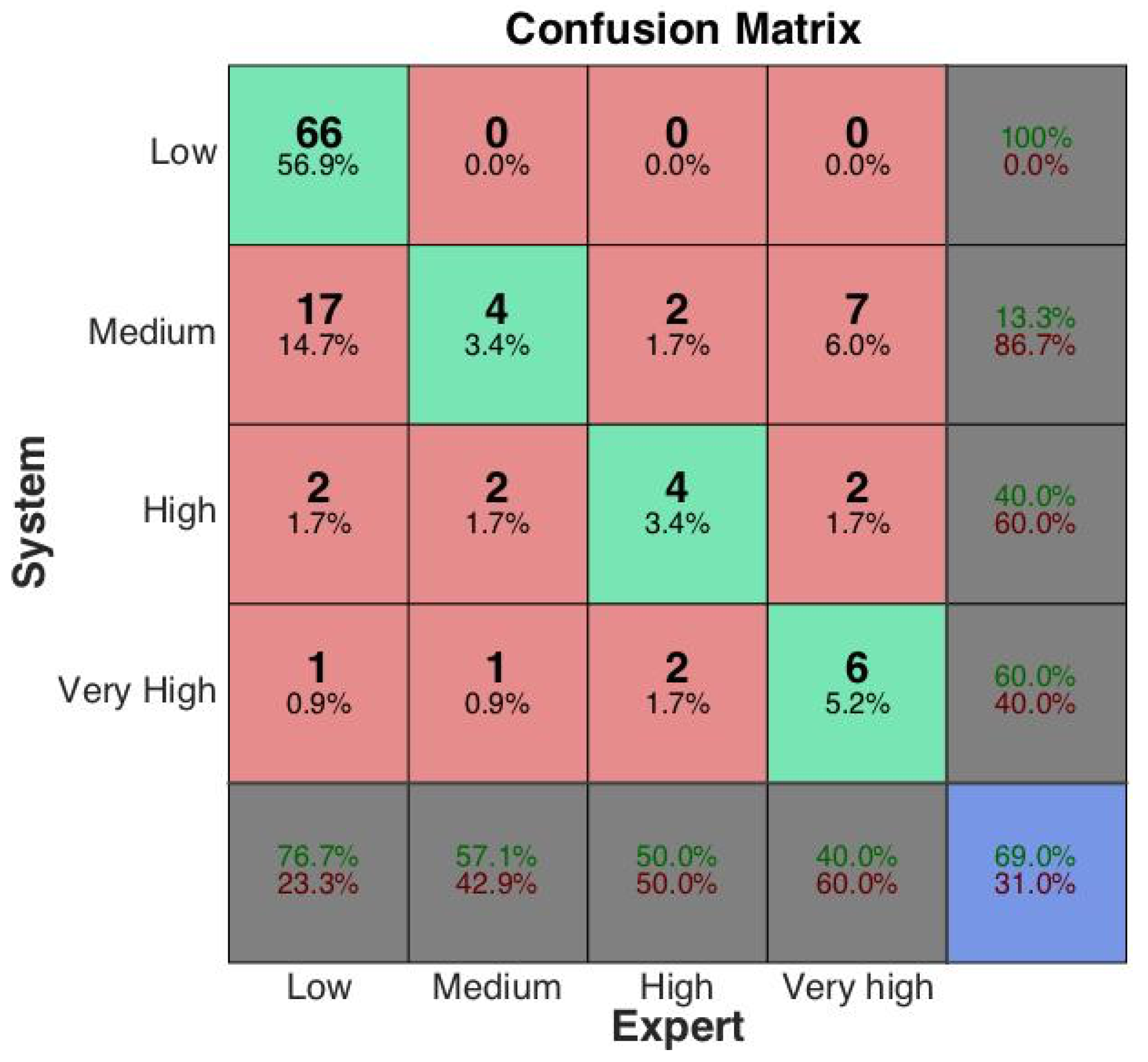
3.2. Risk Assessment by Fuzzy Rules
- Accuracy, i.e., the ratio of correct discriminations w.r.t. class c:
- Positive Predictive Value, i.e., the ratio of correctly classified samples w.r.t. those identified as pertaining to class c:
- Negative Predictive Value, i.e., the ratio of correctly classified samples w.r.t. those identified as not pertaining to class c:
- True Positive Rate, i.e., the ratio of samples correctly classified as belonging to class c w.r.t. those actually belonging to class c:
- True Negative Rate, i.e., the ratio of samples correctly classified as not belonging to class c w.r.t. those actually not belonging to class c:
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cook, S.; Togni, M.; Schaub, M.C.; Wenaweser, P.; Hess, O.M. High heart rate: A cardiovascular risk factor? Eur. Heart J. 2006, 27, 2387–2393. [Google Scholar] [CrossRef] [PubMed]
- Pavlidis, I.; Dowdall, J.; Sun, N.; Puri, C.; Fei, J.; Garbey, M. Interacting with human physiology. Comput. Vis. Image Understand. 2007, 108, 150–170. [Google Scholar] [CrossRef]
- Allen, J. Photoplethysmography and its application in clinical physiological measurement. Physiol. Meas. 2007, 28, R1. [Google Scholar] [CrossRef] [PubMed]
- Wieringa, F.P.; Mastik, F.; van der Steen, A.F. Contactless multiple wavelength photoplethysmographic imaging: A first step toward “SpO2 camera” technology. Ann. Biomed. Eng. 2005, 33, 1034–1041. [Google Scholar] [CrossRef] [PubMed]
- Humphreys, K.; Ward, T.; Markham, C. Noncontact simultaneous dual wavelength photoplethysmography: A further step toward noncontact pulse oximetry. Rev. Sci. Instrum. 2007, 78, 044304. [Google Scholar] [CrossRef] [PubMed]
- Takano, C.; Ohta, Y. Heart rate measurement based on a time-lapse image. Med. Eng. Phys. 2007, 29, 853–857. [Google Scholar] [CrossRef]
- Zheng, J.; Hu, S.; Chouliaras, V.; Summers, R. Feasibility of imaging photoplethysmography. In Proceedings of the International Conference on BioMedical Engineering and Informatics (BMEI 2008), Sanya, China, 27–30 May 2008; Volume 2, pp. 72–75. [Google Scholar]
- Verkruysse, W.; Svaasand, L.O.; Nelson, J.S. Remote plethysmographic imaging using ambient light. Opt. Express 2008, 16, 21434–21445. [Google Scholar] [CrossRef]
- Garbey, M.; Sun, N.; Merla, A.; Pavlidis, I. Contact-free measurement of cardiac pulse based on the analysis of thermal imagery. IEEE Trans. Biomed. Eng. 2007, 54, 1418–1426. [Google Scholar] [CrossRef]
- Rouast, P.V.; Adam, M.T.; Chiong, R.; Cornforth, D.; Lux, E. Remote heart rate measurement using low-cost RGB face video: A technical literature review. Front. Comput. Sci. 2016, 1–15. [Google Scholar] [CrossRef]
- Hassan, M.; Malik, A.S.; Fofi, D.; Saad, N.; Karasfi, B.; Ali, Y.S.; Meriaudeau, F. Heart rate estimation using facial video: A review. Biomed. Signal Process. Control 2017, 38, 346–360. [Google Scholar] [CrossRef]
- Poh, M.Z.; McDuff, D.J.; Picard, R.W. Non-contact, automated cardiac pulse measurements using video imaging and blind source separation. Opt. Express 2010, 18, 10762–10774. [Google Scholar] [CrossRef]
- Bosi, I.; Cogerino, C.; Bazzani, M. Real-time monitoring of heart rate by processing of Microsoft Kinect™ 2.0 generated streams. In Proceedings of the International Multidisciplinary Conference on Computer and Energy Science (SpliTech), Split, Croatia, 13–15 July 2016; pp. 1–6. [Google Scholar]
- Zhang, Q.; Wu, Q.; Zhou, Y.; Wu, X.; Ou, Y.; Zhou, H. Webcam-based, non-contact, real-time measurement for the physiological parameters of drivers. Measurement 2017, 100, 311–321. [Google Scholar] [CrossRef]
- Kwon, S.; Kim, H.; Park, K.S. Validation of heart rate extraction using video imaging on a built-in camera system of a smartphone. In Proceedings of the Annual International Conference of the IEEE on Engineering in Medicine and Biology Society (EMBC2012), San Diego, CA, USA, 28 August–1 September 2012; pp. 2174–2177. [Google Scholar]
- Lewandowska, M.; Rumiński, J.; Kocejko, T.; Nowak, J. Measuring pulse rate with a webcam—A non-contact method for evaluating cardiac activity. In Proceedings of the Federated Conference on Computer Science and Information Systems (FedCSIS2011), Szczecin, Poland, 18–21 September 2011; pp. 405–410. [Google Scholar]
- Scully, C.G.; Lee, J.; Meyer, J.; Gorbach, A.M.; Granquist-Fraser, D.; Mendelson, Y.; Chon, K.H. Physiological parameter monitoring from optical recordings with a mobile phone. IEEE Trans. Biomed. Eng. 2012, 59, 303–306. [Google Scholar] [CrossRef] [PubMed]
- Dalal, N.; Triggs, B. Histograms of oriented gradients for human detection. In Proceedings of the IEEE Computer Society Conference on Computer Vision and Pattern Recognition (CVPR 2005), San Diego, CA, USA, 20–25 June 2005; Volume 1, pp. 886–893. [Google Scholar]
- Kazemi, V.; Sullivan, J. One millisecond face alignment with an ensemble of regression trees. In Proceedings of the IEEE Conference on Computer Vision and Pattern Recognition, Columbus, OH, USA, 23–28 June 2014; pp. 1867–1874. [Google Scholar]
- Challoner, A. Photoelectric plethysmography for estimating cutaneous blood flow. Non-Invasive Physiol. Meas. 1979, 1, 125–151. [Google Scholar]
- Tarvainen, M.P.; Ranta-Aho, P.O.; Karjalainen, P.A. An advanced detrending method with application to HRV analysis. IEEE Trans. Biomed. Eng. 2002, 49, 172–175. [Google Scholar] [CrossRef] [PubMed]
- Casalino, G.; Del Buono, N.; Mencar, C. Subtractive Initialization of Nonnegative Matrix Factorizations for Document Clustering. In Lecture Notes in Computer Science, Proceedings of the Fuzzy Logic and Applications, Trani, Italy, 29–31 August 2011; Fanelli, A., Pedrycz, W., Petrosino, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; Volume 6857, pp. 188–195. [Google Scholar]
- Casalino, G.; Del Buono, N.; Mencar, C. Part-based data analysis with masked non-negative matrix factorization. In Lecture Notes in Computer Science, Proceedings of the Computational Science and Its Applications, Guimarães, Portugal, 30 June–3 July 2014; Springer: Cham, Switzerland, 2014; Volume 8584, pp. 440–454. [Google Scholar]
- Kong, L.; Zhao, Y.; Dong, L.; Jian, Y.; Jin, X.; Li, B.; Feng, Y.; Liu, M.; Liu, X.; Wu, H. Non-contact detection of oxygen saturation based on visible light imaging device using ambient light. Opt. Express 2013, 21, 17464–17471. [Google Scholar] [CrossRef]
- Singh, S.; Kumar, A.; Panneerselvam, K.; Vennila, J.J. Diagnosis of arthritis through fuzzy inference system. J. Med. Syst. 2012, 36, 1459–1468. [Google Scholar] [CrossRef]
- Efosa, I.; Akwukwuma, V. Knowledge-based fuzzy inference system for sepsis diagnosis. Int. J. Comput. Sci. Inf. Technol. IJCSITY 2013, 1, 1–7. [Google Scholar] [CrossRef]
- Sabri, N.; Aljunid, S.; Salim, M.; Badlishah, R.; Kamaruddin, R.; Malek, M.A. Fuzzy Inference System: Short Review and Design. Int. Rev. Autom. Control 2013, 6, 441–449. [Google Scholar]
- Phuong, N.H.; Kreinovich, V. Fuzzy logic and its applications in medicine. Int. J. Med. Inform. 2001, 62, 165–173. [Google Scholar] [CrossRef]
- Tyagi, P.K.; Gupta, N.K.; Gupta, S. Diagnosis of Pulmonary Tuberculosis using fuzzy Inference System. In Proceedings of the Second International Innovative Applications of Computational Intelligence on Power, Energy and Controls with their Impact on Humanity (CIPECH2016), Ghaziabad, India, 18–19 November 2016; pp. 3–7. [Google Scholar]
- Thakur, S.; Raw, S.N.; Sharma, R. Design of a fuzzy model for thalassemia disease diagnosis: Using mamdani type fuzzy inference system (FIS). Int. J. Pharm. Pharm. Sci. 2016, 8, 356–361. [Google Scholar]
- Paramita, A.; Mustika, F.A. Diagnosis Of the Diabetes Mellitus disease with Fuzzy Inference System Mamdani. J. Math. 2017, 1, 126–133. [Google Scholar]
- Rana, M.; Sedamkar, R. Design of Expert System for Medical Diagnosis Using Fuzzy Logic. Int. J. Sci. Eng. Res. 2013, 4, 2914–2921. [Google Scholar]
- Walia, N.; Singh, H.; Tiwari, S.K.; Sharma, A. A decision support system for tuberculosis diagnosability. Int. J. Soft Comput. 2015, 6. [Google Scholar] [CrossRef]
- Khanale, P.; Ambilwade, R. A fuzzy inference system for diagnosis of hypothyroidism. J. Artif. Intell. 2011, 4, 45–54. [Google Scholar] [CrossRef]
- De Medeiros, I.B.; Machado, M.A.S.; Damasceno, W.J.; Caldeira, A.M.; dos Santos, R.C.; da Silva Filho, J.B. A Fuzzy Inference System to Support Medical Diagnosis in Real Time. Procedia Comput. Sci. 2017, 122, 167–173. [Google Scholar] [CrossRef]
- Dagar, P.; Jatain, A.; Gaur, D. Medical diagnosis system using fuzzy logic toolbox. In Proceedings of the 2015 International Conference on Computing, Communication & Automation (ICCCA2015), Noida, India, 15–16 May 2015; pp. 193–197. [Google Scholar]
- Adlassnig, K.P. Fuzzy set theory in medical diagnosis. IEEE Trans. Syst. Man Cybern. 1986, 16, 260–265. [Google Scholar] [CrossRef]
- Alonso, J.; Castiello, C.; Lucarelli, M.; Mencar, C. Modeling interpretable fuzzy rule-based classifiers for medical decision support. In Data Mining: Concepts, Methodologies, Tools, and Applications; IGI Global: Hershey, PA, USA, 2013; pp. 1064–1081. [Google Scholar]
- Zadeh, L. Fuzzy logic and soft computing: Issues, contentions and perspectives. Proc. IIZUKA 1994, 94, 1–2. [Google Scholar]
- Zadeh, L.A. The concept of a linguistic variable and its application to approximate reasoning—I. Inf. Sci. 1975, 8, 199–249. [Google Scholar] [CrossRef]
- Zadeh, L.A. Fuzzy sets. Inf. Control 1965, 8, 338–353. [Google Scholar] [CrossRef]
- Castellano, G.; Castiello, C.; Pasquadibisceglie, V.; Zaza, G. FISDeT: Fuzzy Inference System Development Tool. Int. J. Comput. Intell. Syst. 2017, 10, 13–22. [Google Scholar] [CrossRef]
- Lam, A.; Kuno, Y. Robust heart rate measurement from video using select random patches. In Proceedings of the IEEE International Conference on Computer Vision, Santiago, Chile, 7–13 December 2015; pp. 3640–3648. [Google Scholar]
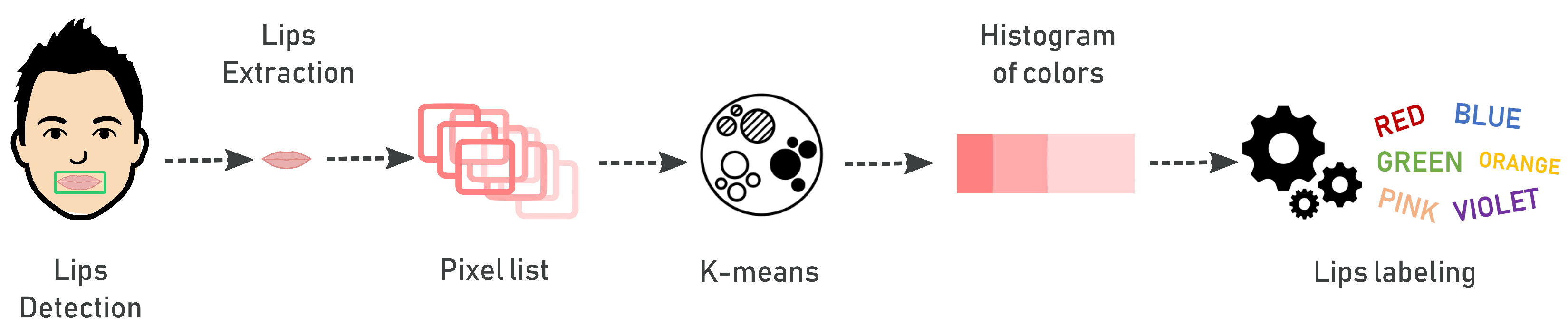
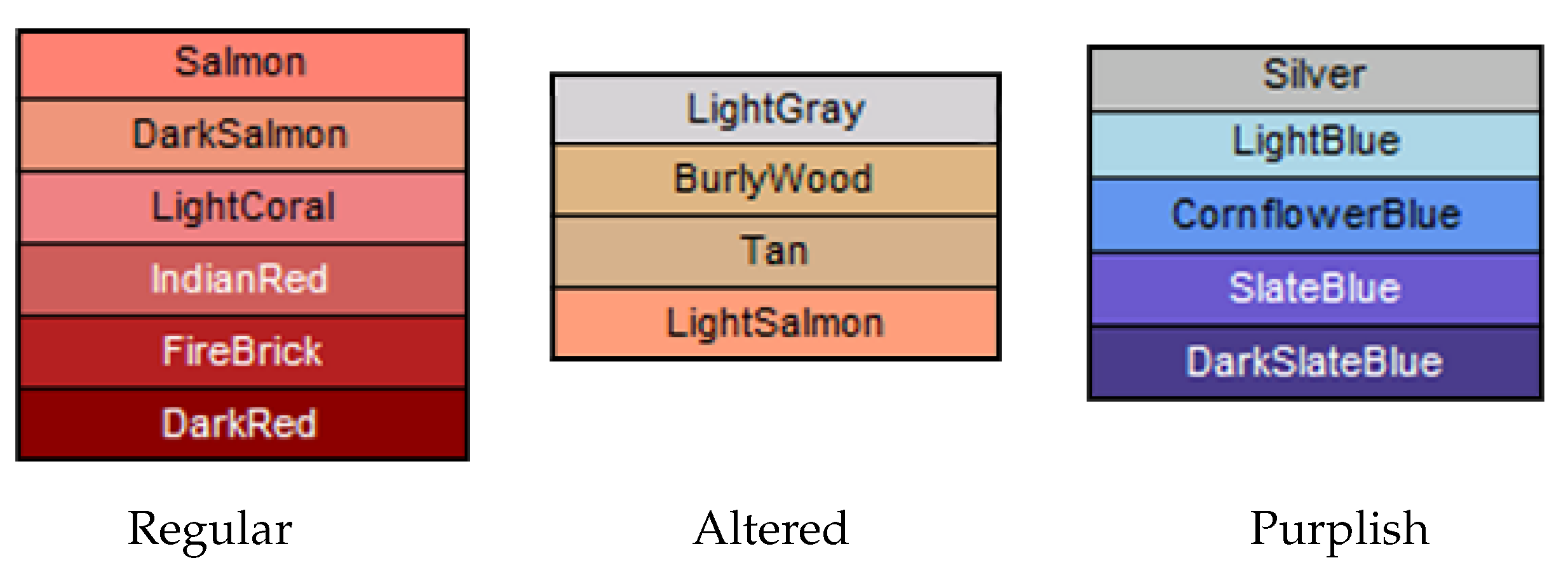
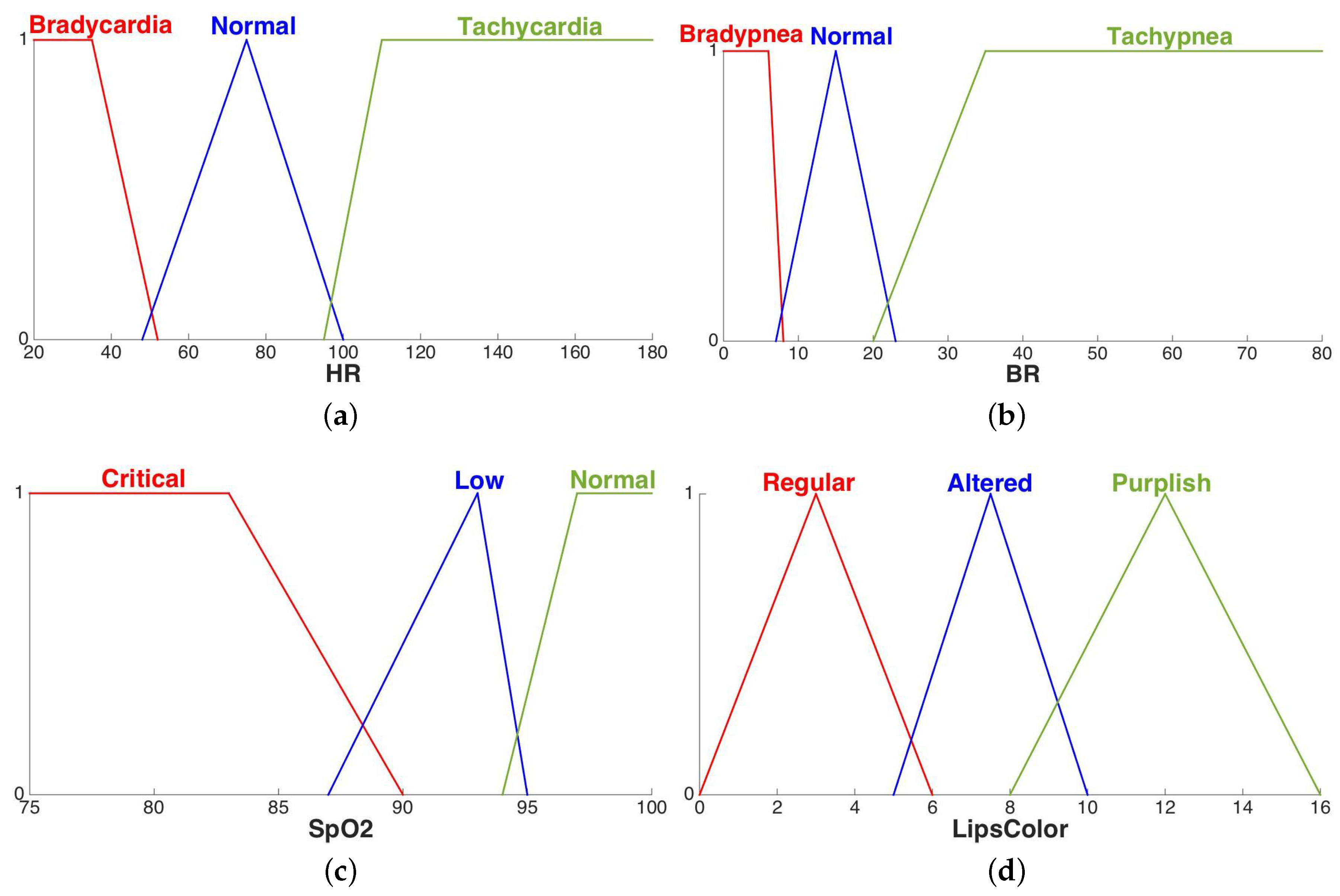

| Linguistic Variables | Range | Fuzzy Values |
|---|---|---|
| Bradycardia | ||
| Normal | ||
| Tachycardia | ||
| Bradypnea | ||
| Normal | ||
| Tachypnea | ||
| Critical | ||
| Low | ||
| Normal | ||
| Regular | ||
| Altered | ||
| Purplish |
| Rule No. | Antecedent | Consequent | |||
|---|---|---|---|---|---|
| 1 | Normal | Normal | Normal | Regular | Low |
| 2 | Normal | Bradypnea | Normal | Regular | Medium |
| 3 | Normal | Tachypnea | Critical | Regular | High |
| 4 | Tachycardia | Tachypnea | Critical | Regular | VeryHigh |
| … | |||||
| 81 | Tachycardia | Tachypnea | Low | Purplish | VeryHigh |
| All Subjects | Healthy Subjects | Unhealthy Subjects | |
|---|---|---|---|
| (a) Measurements of Vital Signs. | | | (b) Expert Diagnosis. | |||||
|---|---|---|---|---|---|---|---|
| | | Subject | ||||||
| S1 | 73.0 | 10.7 | 98.9 | 12 | | | S1 | Low |
| S2 | 98.3 | 9.4 | 98.4 | 12 | | | S2 | Medium |
| S3 | 136.6 | 9.0 | 94.0 | 12 | | | S3 | Very High |
| S4 | 79.1 | 10.8 | 93.6 | 1 | | | S4 | Medium |
| S5 | 88.4 | 21.2 | 98.0 | 1 | | | S4 | Medium |
| … | … | … | … | … | | | … | … |
| S116 | 70.8 | 31.4 | 92.0 | 1 | | | S116 | High |
| Low Risk | Medium Risk | High Risk | Very High Risk | |
|---|---|---|---|---|
| No. Subjects | 86 | 7 | 8 | 15 |
| (a) Evaluation Measures Derived for Each Risk Class. | | | (b) TP, TN, FP, FN for Each Risk Class. | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Class | ACC | TNR | TPR | PPV | NPV | | | Risk Class | TP | TN | FP | FN |
| Low | 0.83 | 1.00 | 0.77 | 1.00 | 0.60 | | | Low | 66 | 30 | 0 | 20 |
| Medium | 0.75 | 0.76 | 0.57 | 0.13 | 0.96 | | | Medium | 4 | 83 | 26 | 3 |
| High | 0.91 | 0.94 | 0.50 | 0.40 | 0.96 | | | High | 4 | 102 | 6 | 4 |
| Very High | 0.88 | 0.96 | 0.40 | 0.60 | 0.91 | | | Very High | 6 | 97 | 4 | 9 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Casalino, G.; Castellano, G.; Pasquadibisceglie, V.; Zaza, G. Contact-Less Real-Time Monitoring of Cardiovascular Risk Using Video Imaging and Fuzzy Inference Rules. Information 2019, 10, 9. https://doi.org/10.3390/info10010009
Casalino G, Castellano G, Pasquadibisceglie V, Zaza G. Contact-Less Real-Time Monitoring of Cardiovascular Risk Using Video Imaging and Fuzzy Inference Rules. Information. 2019; 10(1):9. https://doi.org/10.3390/info10010009
Chicago/Turabian StyleCasalino, Gabriella, Giovanna Castellano, Vincenzo Pasquadibisceglie, and Gianluca Zaza. 2019. "Contact-Less Real-Time Monitoring of Cardiovascular Risk Using Video Imaging and Fuzzy Inference Rules" Information 10, no. 1: 9. https://doi.org/10.3390/info10010009
APA StyleCasalino, G., Castellano, G., Pasquadibisceglie, V., & Zaza, G. (2019). Contact-Less Real-Time Monitoring of Cardiovascular Risk Using Video Imaging and Fuzzy Inference Rules. Information, 10(1), 9. https://doi.org/10.3390/info10010009






