Tracking Trends in Emissions of Developmental Toxicants and Potential Associations with Congenital Heart Disease in Alberta, Canada
Abstract
1. Introduction
2. Results
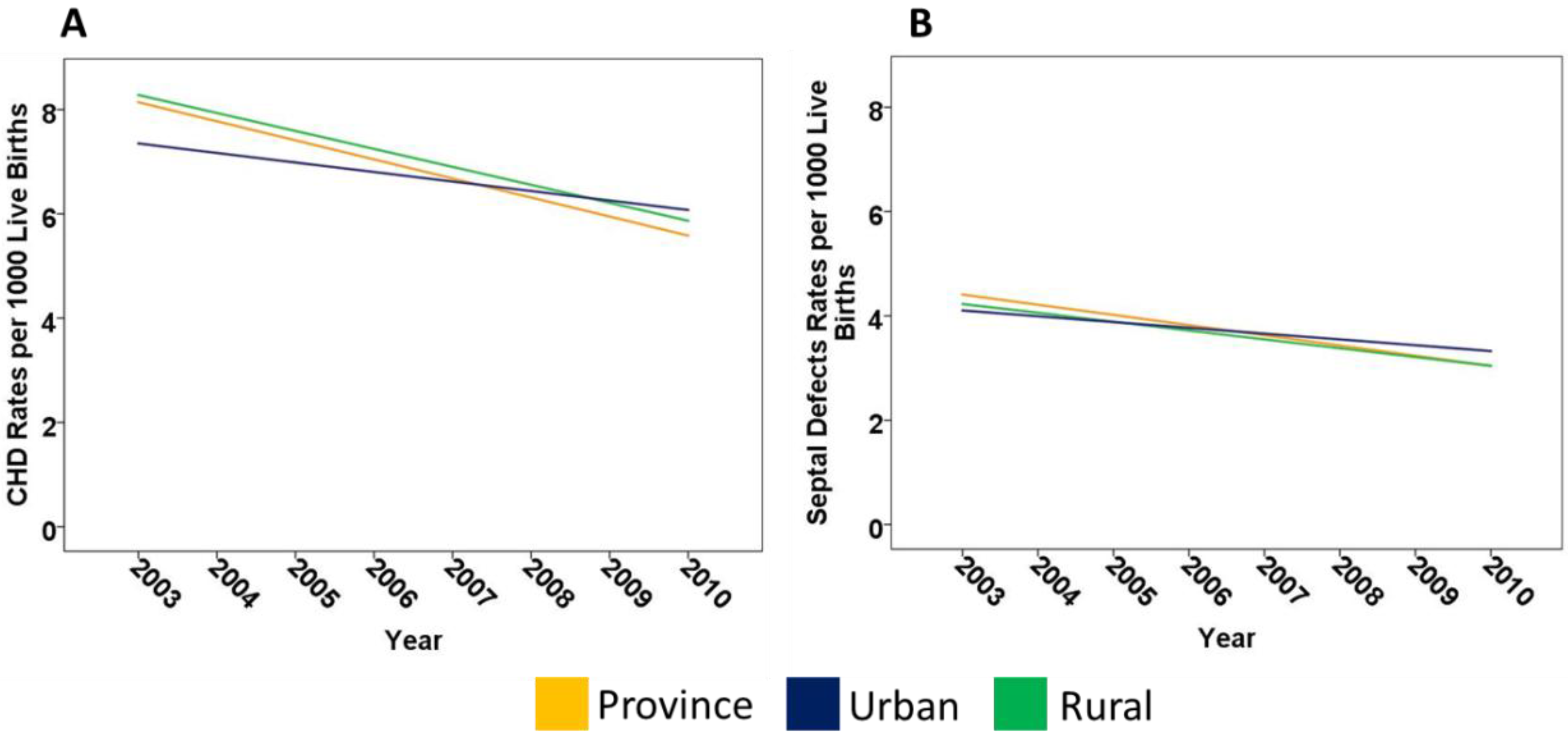
2.1. CHD in Alberta
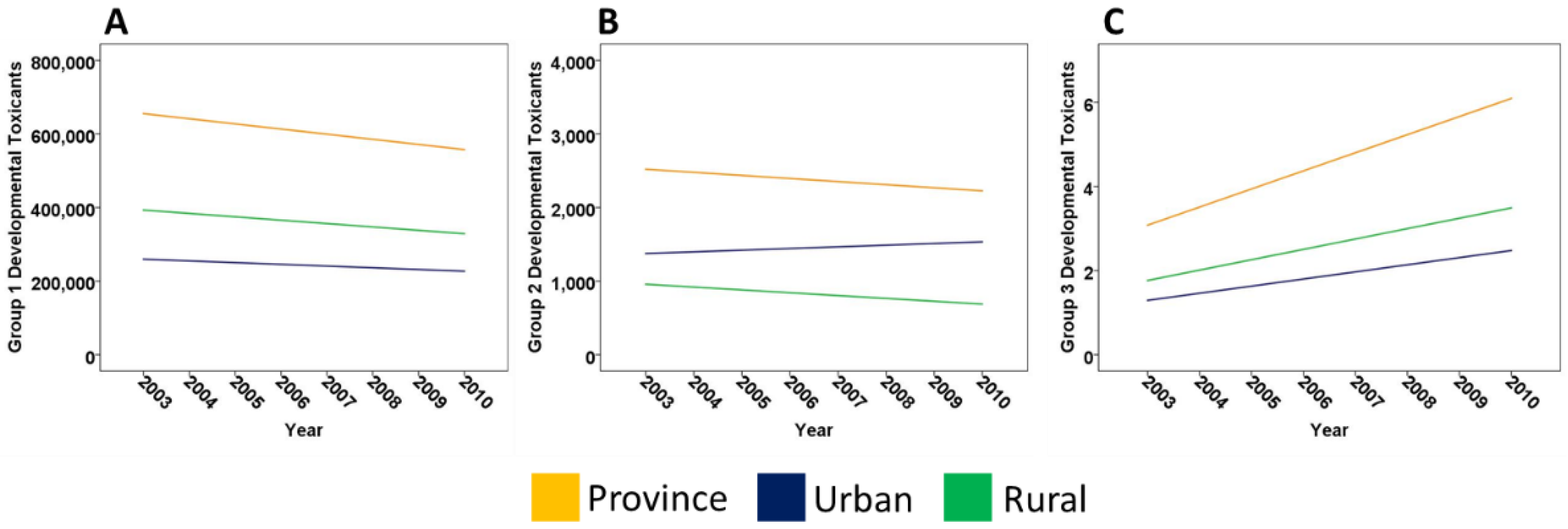
2.2. Developmental Toxicants in Alberta
2.3. Associations between Groups of DTs and CHD Rates in Alberta
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. Pollution Data
4.3. Spatio-Temporal Aggregation of DTs
4.4. Statistical Methods
4.5. Study Limitations
5. Conclusions and Recommendations
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Ethics Approval
Availability of Data Materials
References
- Van der Linde, D.; Konings, E.E.M.; Slager, M.A.; Witsenburg, M.; Helbing, W.A.; Takkenberg, J.J.M.; Roos-Hesselink, J.W. Birth Prevalence of Congenital Heart Disease Worldwide. J. Am. Cardiol. 2011, 58, 2241–2247. [Google Scholar] [CrossRef] [PubMed]
- Khairy, P.; Ionescu-Ittu, R.; Mackie, A.S.; Abrahamowicz, M.; Pilote, L.; Marelli, A.J. Changing mortality in congenital heart disease. J. Am. Coll. Cardiol. 2010, 56, 1149–1157. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Joseph, K.S.; Lisonkova, S.; Rouleau, J.; Van den Hof, M.; Sauve, R.; Kramer, M.S. Association between maternal chronic conditions and congenital heart defects: A population-based cohort study. Circulation 2013, 128, 583–589. [Google Scholar] [CrossRef] [PubMed]
- Pierpont, M.E.; Basson, C.T.; Benson, D.W.; Gelb, B.D.; Giglia, T.M.; Goldmuntz, E.; McGee, G.; Sable, C.A.; Srivastava, D.; et al. Genetic basis for congenital heart defects: Current knowledge. A scientific Statement from the American Heart Association Congenital Cardiac defects Committee, Council on Cardiovascular Disease in the Young. Circulation 2007, 115, 3015–3038. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.D.; Loffredo, C.A.; Correa-Villasenor, A.; Ferencz, C. Attributable Fraction for Cardiac Malformations. Am. J. Epidemiol. 1998, 148, 414–423. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, K.J.; Correa, A.; Feinstein, J.A.; Botto, L.; Britt, A.E.; Daniels, S.R.; Elixson, M.; Warnes, C.A.; Webb, C.L. Noninherited risk factors and congenital cardiovascular defects: Current knowledge. A scientific statement from the American Heart Asociation Council on Cardiovascular Disease in the Young. Circulation 2007, 115, 2995–3014. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Kartiko, S.; Finnell, R.H. Importance of gene-environment interactions in the etiology of selected birth defects. Clin. Genet. 2009, 75, 409–423. [Google Scholar] [CrossRef] [PubMed]
- Ferencz, C.; Loffredo, C.A.; Correa-Villasenor, A.; Wilson, P.D. Risk factor analysis: A synthesis. In Genetic and Environmental Risk Factors of Major Cardiovascular Malformations: The Baltimore-Washington Infant Study: 1981–1989; Ferencz, C., Loffredo, C.A., Correa-Villasenor, A., Wilson, P.D., Eds.; Futura Publishing Company Inc.: Armonk, NY, USA, 1997; Volume 5, pp. 359–382. [Google Scholar]
- Vrijheid, M.; Martinez, D.; Manzanares, S.; Dadvand, P.; Schembari, A.; Rankin, J.; Nieuwenhuijsen, M. Ambient air pollution and risk of congenital anomalies: A systematic review and meta-analysis. Environ. Health Perspect. 2011, 119, 598–606. [Google Scholar] [CrossRef] [PubMed]
- McKenzie, L.M.; Guo, R.; Witter, R.Z.; Savitz, D.; Newman, L.; Adgate, J. Birth outcomes and maternal residential proximity to natural gas development in rural Colorado. Environ. Health Perspect. 2014, 122, 412–417. [Google Scholar] [CrossRef] [PubMed]
- Stingone, J.A.; Luben, T.J.; Daniels, J.L.; Fuentes, M.; Richardson, D.B.; Aylsworth, A.S.; Herring, A.H.; Anderka, M.; Botto, L.; Correa, A.; et al. Maternal Exposure to Criteria Air Pollutants and Congenital Heart Defects in Offspring: Results from the National Birth Defects Prevention Study. Environ. Health Perspect. 2014, 122, 863–872. [Google Scholar] [CrossRef] [PubMed]
- National Pollutant Release Inventory: Environment Canada. 2014. Available online: https://www.ec.gc.ca/inrp-npri/default.asp?lang=en&n=0EC58C98 (accessed on 11 July 2018).
- Office of Environmental Health Hazard Assessment Proposition 65 California: State of California Environmental Protection Agency. Available online: https://oehha.ca.gov/proposition-65 (accessed on 11 July 2018).
- Statistics Canada. Population, Urban and Rural by Province and Territory 2011. Available online: http://www.statcan.gc.ca/tables-tableaux/sum-som/l01/cst01/demo62a-eng.htm (accessed on 11 July 2018).
- Economic Trends June 2013. Available online: https://open.alberta.ca/publications/5874657 (accessed on 11 July 2018).
- Alberta’s Rural Communities. Their Economic Contribution to Alberta and Canada: The Conference Board of Canada. Available online: http://www1.agric.gov.ab.ca/$Department/deptdocs.nsf/all/csi14195/$FILE/alberta-rural-communities-report.pdf (accessed on 11 July 2018).
- Beck, E.M.; Smits, J.E.G.; St Clair, C.C. Evidence of low toxicity of oil sands pocess affected water to birds invites re-evaluation of avian protection strategies. Conserv. Physiol. 2015, 3, cov038. [Google Scholar] [CrossRef] [PubMed]
- Canada Public Health Agency. Congenital Anomalies in Canada 2013: A Perinatal Health Surveillance Report; Canada Public Health Agency: Ottawa, ON, Canada, 2013. [Google Scholar]
- Wine, O.; Hackett, C.; Campbell, S.; Cabrera-Rivera, O.; Buka, I.; Zaiane, O.; DeVito, S.C.; Osornio-Vargas, A. Using pollutant release and transfer register data in human health research: A scoping review. Environ. Rev. 2014, 22, 51–65. [Google Scholar] [CrossRef]
- Mauderly, J.L.; Burnett, R.T.; Castillejos, M.; Özkaynak, H.; Samet, J.M.; Stieb, D.M.; Vedal, S.; Wyzga, R.E. Is the air pollution health research community prepared to support a multipollutant air quality management framework? Inhalation Toxicol. 2010, 22, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Air Pollution: Drivers and Impacts. Available online: http://www.ec.gc.ca/indicateurs-indicators/default.asp?lang=En&n=D189C09D-1 (accessed on 11 July 2018).
- Do Environmental Regulations Really Work? Available online: https://www.theregreview.org/2016/11/24/williamson-do-environmental-regulations-really-work/ (accessed on 11 July 2018).
- Cutter, S.L.; Scott, M.S.; Hill, A.A. Spatial Variability in Toxicity Indicators Used to Rank Chemical Risks. Am. J. Public Health 2002, 92, 420–422. [Google Scholar] [CrossRef] [PubMed]
- Ngwezi, D.P.; Hornberger, L.K.; Serrano-Lomelin, J.; Fruitman, D.; Osornio-Vargas, A. Industrial Developmental Toxicants and Congenital Heart Disease in Urban and Rural Alberta, Canada. Challenges 2018, 9, 26. [Google Scholar] [CrossRef]
- Wigle, D.T.; Arbuckle, T.E.; Turner, M.C.; Bérubé, A.; Yang, Q.; Liu, S.; Krewski, D. Epidemiologic evidence of relationships between reproductive and child health outcomes and environmental chemical contaminants. J. Toxicol. Environ. Health B 2008, 11, 373–517. [Google Scholar] [CrossRef] [PubMed]
- Holson, J.F.; Desesso, J.M.; Jacobson, C.F.; Farr, C.H. Appropriate use of animal models in the assessment of risk during prenatal development: An illustration using inorganic arsenic. Teratology 2000, 62, 51–71. [Google Scholar] [CrossRef]
- Dawson, B.V.; Johnson, P.D.; Goldberg, S.J.; Ulreich, J.B. Cardiac teratogenesis of trichloroethylene and dichloroethylene in a mammalian model. J. Am. Coll. Cardiol. 1990, 16, 1304–1309. [Google Scholar] [CrossRef]
- Hansen, J.M. Oxidative stress as a mechanism of teratogenesis. Birth Defects Res. Part C Embryo Today 2006, 78, 293–307. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Li, Y.; Tollefsbol, T.O. Gene-Environment Interactions and Epigenetic Basis of Human Diseases. Curr. Issues Mol. Biol. 2008, 10, 25–36. [Google Scholar] [PubMed]
- Rosenquist, T.H. Folate, homocysteine and the cardiac neural crest. Dev. Dyn. 2013, 242, 201–218. [Google Scholar] [CrossRef] [PubMed]
- Huhta, J.; Linask, K.K. Environmental Origins of congenital heart disease: The heart-placenta connection. Semin. Fetal Neonatal Med. 2013, 18, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Sanders, A.P.; Desrosiers, T.A.; Warren, J.L.; Herring, A.H.; Enright, D.; Olshan, A.F.; Meyer, R.E.; Fry, R.C. Association between arsenic, cadmium, manganese, and lead levels in private wells and birth defects prevalence in North Carolina: A semi-ecologic study. BMC Public Health 2014, 14, 955. [Google Scholar] [CrossRef] [PubMed]
- Botto, L.D.; Lin, A.E.; Riehle-Colarusso, T.; Malik, S.; Correa, A. National Birth Defects Prevention Study. Seeking causes: Classifying and evaluating congenital heart defects in etiologic studies. Birth Defects Res. Part A Clin. Mol. Teratol. 2007, 79, 714–727. [Google Scholar] [CrossRef] [PubMed]
- Commission for Environmental Cooperation. Toxic Chemicals and Children’s Health in North America: A Call for Efforts to Determine Sources, Levels of Exposure, and Risks that Industrial Chemicals Pose to Children’s Health; Commission for Environmental Cooperation: Montreal, QC, Canada, 2006. [Google Scholar]
- Scorecard: Goodguide. Available online: http://scorecard.goodguide.com/health-effects/ (accessed on 11 July 2018).
- Legendre, P.; Legendre, L. Numerical Ecology, 3rd ed.; Elsevier: New York, NY, USA, 2012. [Google Scholar]
- Statistics Canada North American Industrial Classification System. Available online: http://www.statcan.gc.ca/subjects-sujets/standard-norme/naics-scian/2012/introduction-eng.htm (accessed on 11 July 2018).
- Bender, R.; Lange, S. Adjustment for multiple testing—When and how? J. Clin. Epidemiol. 2001, 54, 343–349. [Google Scholar] [CrossRef]
- Colapinto, C.K.; O’Connor, D.L.; Tremblay, M.S. Folate status of the population in the Canadian Health Measures Survey. CMAJ 2011, 182, E100–E106. [Google Scholar] [CrossRef] [PubMed]
- Trines, J.; Fruitman, D.; Zuo, K.J.; Smallhorn, J.F.; Hornberger, L.K.; Mackie, A.S. Effectiveness of Prenatal Screening for Congenital Heart Disease: Assessment in a Jurisdiction with Universal Access to Health Care. Can. J. Cardiol. 2013, 29, 879–885. [Google Scholar] [CrossRef] [PubMed]
- Bedard, T.; Lowry, R.B.; Sibbald, B.; Harder, J.R.; Trevenen, C.; Horobec, V.; Dyck, J.D. Congenital heart defect case ascertainment by the Alberta Congenital Anomalies Surveillance System. Birth Defects Res. Part A Clin. Mol. Teratol. 2012, 94, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Incardona, J.P.; Carls, M.G.; Holland, L.; Linbo, T.L.; Baldwin, D.H.; Myers, M.S.; Peck, K.A.; Tagal, M.; Rice, S.D.; Scholz, N.L. Very low embryonic crude oil exposures cause lasting cardiac defects in salmon and herring. Sci. Rep. 2015, 5, 13499. [Google Scholar] [CrossRef] [PubMed]
- DeWitt, J.C.; Millsap, D.S.; Yeager, R.L.; Heise, S.S.; Sparks, D.W.; Henshel, D.S. External heart deformities in passerine birds exposed to environmental mixtures of polychlorinated biphenyls during development. Environ. Toxicol. Chem. 2006, 25, 541–551. [Google Scholar] [CrossRef] [PubMed]
| Embryological Group | Count (n = 2415) | Percentage (%) | Prevalence (per 1000 Live Births) |
|---|---|---|---|
| Septal | 1320 | 54.7 | 3.67 |
| LHO | 360 | 14.9 | 1.00 |
| Conotruncus | 263 | 10.9 | 0.73 |
| RHO | 220 | 9.1 | 0.61 |
| AVSD | 109 | 4.5 | 0.30 |
| PDA | 48 | 1.9 | 0.13 |
| Heterotaxy | 34 | 1.4 | 0.09 |
| APVR | 34 | 1.4 | 0.09 |
| Complex/SV | 21 | 0.9 | 0.06 |
| Other | 6 | 0.2 | 0.002 |
| Variables | Region | Regression Coefficient | 95% CI | * p Value |
|---|---|---|---|---|
| CHD Rates | Province | −0.4 | −0.6; −0.2 | 0.005 * |
| Rural | −0.3 | −0.5; −0.2 | 0.003 * | |
| Urban | −0.2 | −0.4; 0.1 | 0.133 | |
| Septal Defect Rates | Province | −0.2 | −0.3; −0.1 | 0.012 * |
| Rural | −0.2 | −0.3; −0.02 | 0.025 * | |
| Urban | −0.1 | −0.3; 0.03 | 0.105 |
| Developmental Toxicants | Principal Components | ||
|---|---|---|---|
| Group 1 | Group 2 | Group 3 | |
| Benzene | 0.98 | 0.11 | 0.01 |
| Carbon Disulfide | 0.95 | −0.09 | −0.04 |
| Carbon Monoxide | 0.95 | 0.21 | 0.12 |
| Sulphur Dioxide | 0.86 | −0.11 | 0.47 |
| Toluene | 0.86 | −0.04 | 0.04 |
| 1,3-Butadiene | 0.64 | 0.66 | 0.01 |
| Chloroform | 0.02 | 0.85 | 0.04 |
| Ethylene Oxide | 0.17 | 0.96 | 0.06 |
| Methanol | 0.03 | 0.86 | 0.06 |
| Methyl-isobutyl-ketone | 0.11 | 0.90 | 0.05 |
| Trichloroethylene | 0.11 | 0.79 | 0.05 |
| Arsenic | 0.16 | 0.11 | 0.95 |
| Cadmium | 0.36 | 0.06 | 0.60 |
| Hexachlorobenzene | −0.15 | −0.05 | 0.91 |
| Lead | 0.29 | 0.29 | 0.72 |
| Mercury | −0.52 | −0.08 | 0.97 |
| 2-Ethoxyethanol | −0.05 | −0.04 | −0.08 |
| N-Methyl-2-Pyrrolidone | −0.03 | 0.04 | 0.01 |
| Region | Groups | Amount | % | Risk Score | % |
|---|---|---|---|---|---|
| Province | Group 1 | 4,834,586 | 99.6 | 9,773,565 | 15.2 |
| Group 2 | 18,220 | 0.4 | 3623 | 0.01 | |
| Group 3 | 36 | 0.00 | 54,578,189 | 84.8 | |
| Total | 4,852,844 | 64,355,377 | |||
| (AV ± SD) | 95,153 ± 145,728 | 2,681,474 ± 2,986,681 | |||
| Urban | Group 1 | 1,946,446 | 99.4 | 4,274,371 | 23.3 |
| Group 2 | 11,637 | 0.6 | 3031 | 0.02 | |
| Group 3 | 15 | 0.00 | 14,059,472 | 76.7 | |
| Total | 1,958,101 | 40.3 | 18,336,874 | 28.5 | |
| (AV ± SD) | 92,984 ± 141,235 | 6,112,291 ± 5,883,839 | |||
| Rural | Group 1 | 2,888,139 | 99.7 | 5,499,194 | 11.9 |
| Group 2 | 6583 | 0.3 | 592 | 0.00 | |
| Group 3 | 21 | 0.00 | 40,518,716 | 88 | |
| Total | 2,894,743 | 59.7 | 46,018,502 | 71.5 | |
| (AV ± SD) | 98,669 ± 147,335 | 15,339,501 ± 17,945,349 |
| Variables | Region | Regression Coefficient | 95% CI | * p Value |
|---|---|---|---|---|
| DT Amounts | ||||
| Overall | Province | −14,003 | −21,446; −6539 | 0.004 * |
| Rural | −9226 | −16,016; −2436 | 0.016 | |
| Urban | −4634 | −9337; 69 | 0.053 | |
| Group 1 | Province | −13,962 | −21,427; −6497 | 0.004 * |
| Rural | −9188 | −15,970; −2405 | 0.016 | |
| Urban | −4658 | −9372; 58 | 0.052 | |
| Group 2 | Province | −42 | −87; 3 | 0.060 |
| Rural | −39 | −67; −10 | 0.016 | |
| Urban | 23 | −18; 63 | 0.221 | |
| Group 3 | Province | 0.4 | 0.3; 0.6 | <0.001 * |
| Rural | 0.5 | 0.05; 0.4 | 0.021 | |
| Urban | 0.2 | 0.04; 0.3 | 0.016 | |
| DT Risk Scores | ||||
| Overall | Province | 128,140 | −44,647; 300,928 | 0.120 |
| Rural | −68,809 | −338,413; 200,794 | 0.555 | |
| Urban | 196,950 | 72,289; 321,611 | 0.008 | |
| Group 1 | Province | −33,772 | −45,486; −22,057 | <0.001 * |
| Rural | −29,410 | −36,996; −21,824 | <0.001 * | |
| Urban | −4361 | −17,512; 8789 | 0.448 | |
| Group 2 | Province | −65 | −107; −24 | 0.008 |
| Rural | −4 | −6; −1 | 0.016 * | |
| Urban | −62 | −102; −22 | 0.009 | |
| Group 3 | Province | 161,977 | −7609; 331,563 | 0.058 |
| Rural | −39,396 | −305,239; 226,447 | 0.729 | |
| Urban | 201,373 | 71,479; 331,268 | 0.009 | |
| Region | Variable | Spearman’s Rho (95% CI) | * p Value | Spearman’s Rho (95% CI) | * p Value |
|---|---|---|---|---|---|
| DT Amounts | |||||
| CHD | Septal | ||||
| Province | Group 1 | 0.86 (0.39, 0.97) | 0.007 | 0.89 (0.48, 0.98) | 0.003 * |
| Group 2 | 0.50 (−0.32, 0.89) | 0.207 | 0.79 (0.16, 0.96) | 0.023 | |
| Group 3 | −0.74 (−0.95, −0.07) | 0.037 | −0.76 (−0.95, −0.12) | 0.031 | |
| Rural | Group 1 | 0.62 (−0.15, 0.92) | 0.102 | 0.60 (−0.19, 0.92) | 0.120 |
| Group 2 | 0.79 (0.18, 0.96) | 0.021 | 0.91 (0.55, 0.98) | 0.002 * | |
| Group 3 | −0.64 (−0.93, 0.11) | 0.086 | −0.81 (−0.96, −0.24) | 0.015 | |
| Urban | Group 1 | 0.71 (0.02, 0.94) | 0.047 | 0.74 (0.07, 0.95) | 0.037 |
| Group 2 | −0.07 (−0.74, 0.67) | 0.867 | −0.02 (−0.72, 0.69) | 0.955 | |
| Group 3 | −0.88 (−0.98, −0.47) | 0.004 * | −0.83 (−0.97, −0.31) | 0.010 | |
| DT Risk Scores | |||||
| CHD | Septal | ||||
| Province | Group 1 | 0.88 (0.47, 0.98) | 0.004 * | 0.85 (0.36, 0.97) | 0.007 |
| Group 2 | 0.86 (0.39, 0.97) | 0.007 | 0.97 (0.84, 0.99) | <0.001 * | |
| Group 3 | −0.41 (−0.86, 0.42) | 0.320 | −0.50 (−0.89, 0.31) | 0.204 | |
| Rural | Group 1 | 0.88 (0.47, 0.98) | 0.004 * | 0.76 (0.12, 0.95) | 0.028 |
| Group 2 | 0.79 (0.18, 0.96) | 0.021 | 0.91 (0.55, 0.98) | 0.002 * | |
| Group 3 | −0.02 (−0.72, 0.69) | 0.955 | −0.12 (−0.76, 0.64) | 0.779 | |
| Urban | Group 1 | 0.69 (−0.03, 0.94) | 0.058 | 0.64 (−0.11, 0.93) | 0.086 |
| Group 2 | 0.69 (−0.03, 0.94) | 0.058 | 0.86 (0.39, 0.97) | 0.007 | |
| Group 3 | −0.79 (−0.96, −0.18) | 0.021 | −0.81 (−0.96, −0.24) | 0.015 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ngwezi, D.P.; Hornberger, L.K.; Cabeza-Gonzalez, J.L.; Chandra, S.; Fruitman, D.; Osornio-Vargas, A. Tracking Trends in Emissions of Developmental Toxicants and Potential Associations with Congenital Heart Disease in Alberta, Canada. Challenges 2018, 9, 28. https://doi.org/10.3390/challe9020028
Ngwezi DP, Hornberger LK, Cabeza-Gonzalez JL, Chandra S, Fruitman D, Osornio-Vargas A. Tracking Trends in Emissions of Developmental Toxicants and Potential Associations with Congenital Heart Disease in Alberta, Canada. Challenges. 2018; 9(2):28. https://doi.org/10.3390/challe9020028
Chicago/Turabian StyleNgwezi, Deliwe Precious, Lisa K. Hornberger, Jose Luis Cabeza-Gonzalez, Sujata Chandra, Deborah Fruitman, and Alvaro Osornio-Vargas. 2018. "Tracking Trends in Emissions of Developmental Toxicants and Potential Associations with Congenital Heart Disease in Alberta, Canada" Challenges 9, no. 2: 28. https://doi.org/10.3390/challe9020028
APA StyleNgwezi, D. P., Hornberger, L. K., Cabeza-Gonzalez, J. L., Chandra, S., Fruitman, D., & Osornio-Vargas, A. (2018). Tracking Trends in Emissions of Developmental Toxicants and Potential Associations with Congenital Heart Disease in Alberta, Canada. Challenges, 9(2), 28. https://doi.org/10.3390/challe9020028





