Hydrides as High Capacity Anodes in Lithium Cells: An Italian “Futuro in Ricerca di Base FIRB-2010” Project
Abstract
:1. Introduction
2. The Futuro in Ricerca di Base FIRB-2010 Futuro in Ricerca Project
2.1. Basic Ideas
- The lack of experimental evidence of the electrochemical activity and HCR reversibility of almost all metallic and complex hydrides in lithium cells;
- The weak demonstration of an extended cycling ability of these materials in lithium cells;
- The almost negligible analysis of the chemical and electrochemical stability of the electrolyte/electrode interface in lithium cells.
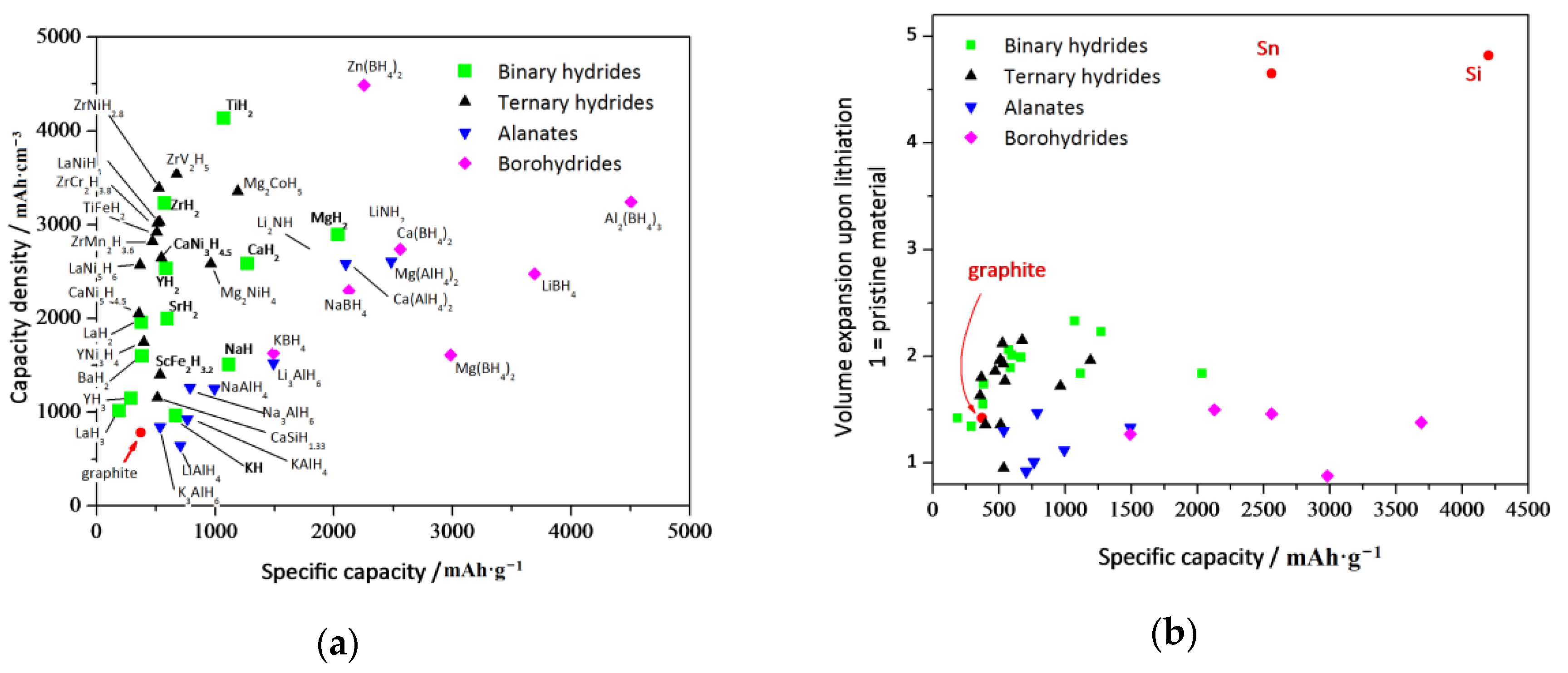
2.2. Hydride Selection
- Cost and environmental compatibility: Candidate hydrides are required to be constituted by atomic elements that are naturally abundant and thus not expensive. They must be environmentally friendly, non-toxic non-explosive, non-pyrophoric and possibly inert to water and oxygen. Among the light elements, the latter constraint necessarily implies discarding Be-based hydrides and to strongly limit V and Co.
- Estimated theoretical performances: Candidate hydrides must show theoretical capacities as high as possible, at least higher than that of graphite (370 mAh·g−1 or 550 mAh·cc−1). Furthermore, in order to assure both the safety and high energy in the final application, the estimated Nernst working potential in the corresponding HCR must range between 1 and 0.1 V vs. Li, far from the Li deposition potential.
- Safety constraint: Selected hydrides must be stable from room temperature up to 200–250 °C with a negligible H2 desorption pressure at room temperature; quantitatively, this parameter is given by the so-called hydride decomposition temperature, which accordingly, is required to be >200 °C.
2.3. Methodological Approach
3. Project Results and Achievements
3.1. Materials Screening
3.1.1. Simple Metal Hydrides
3.1.2. Borohydrides
3.1.3. Alanates
3.1.4. Other Complex Hydrides
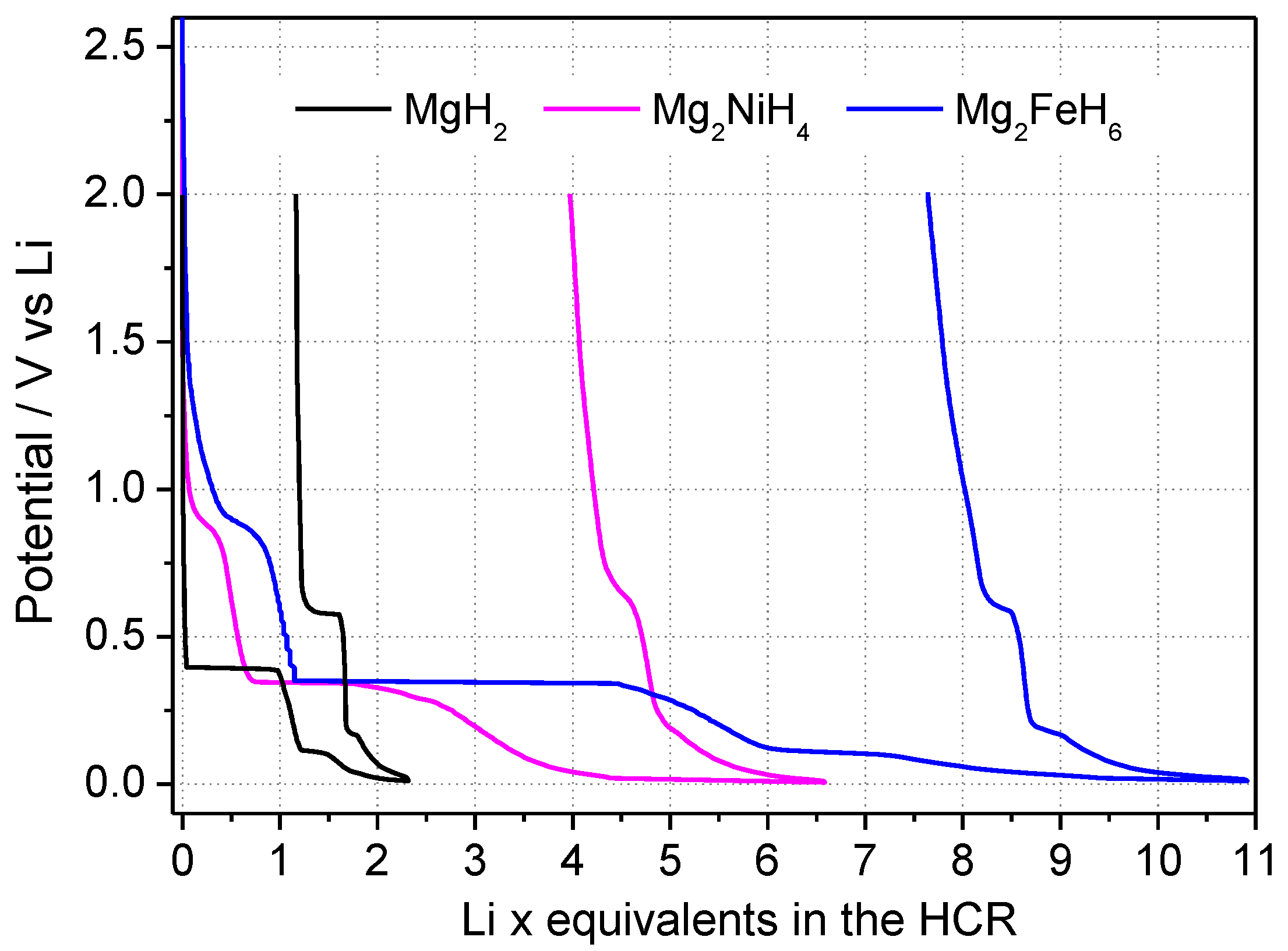
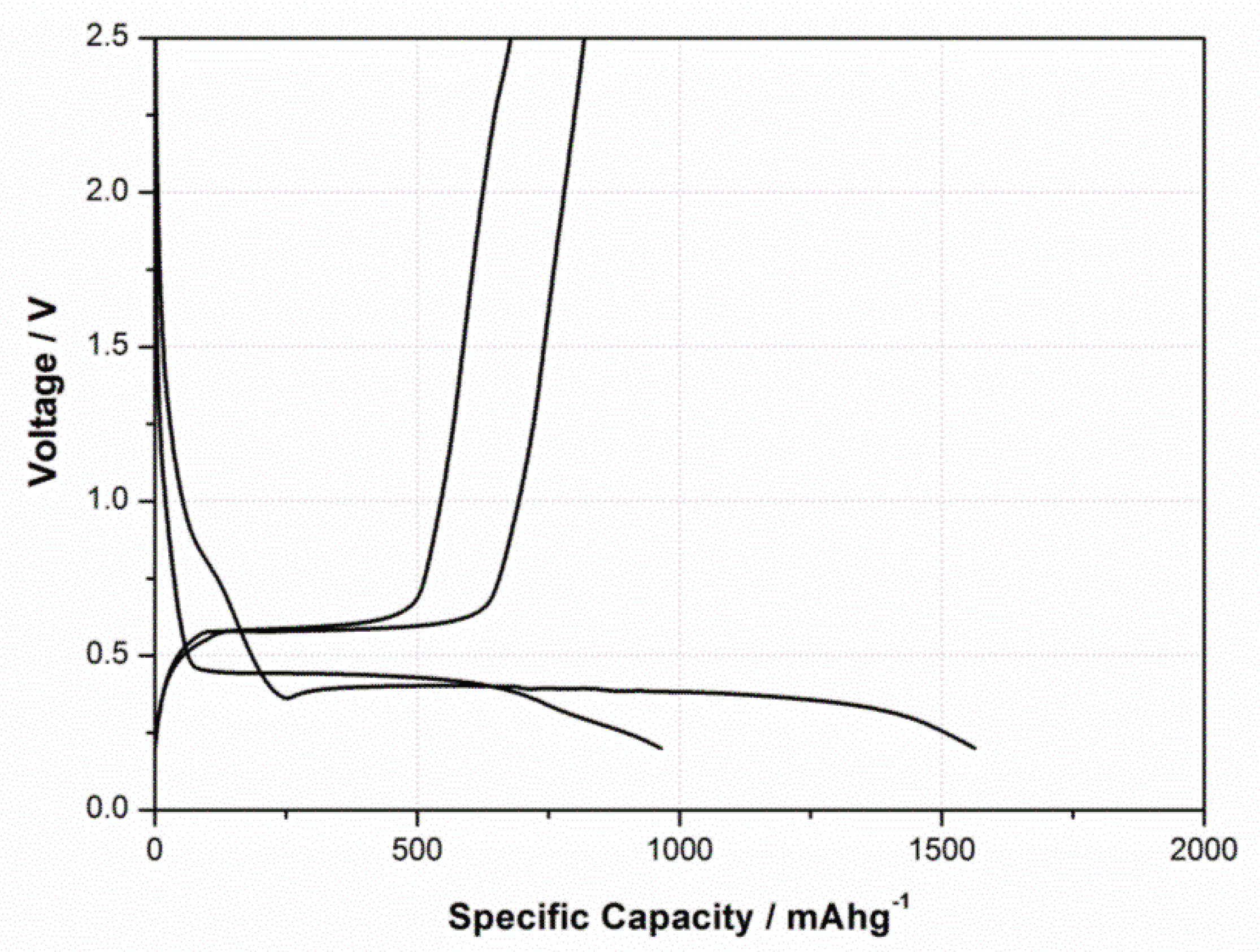
3.2. Mechanism Comprehension
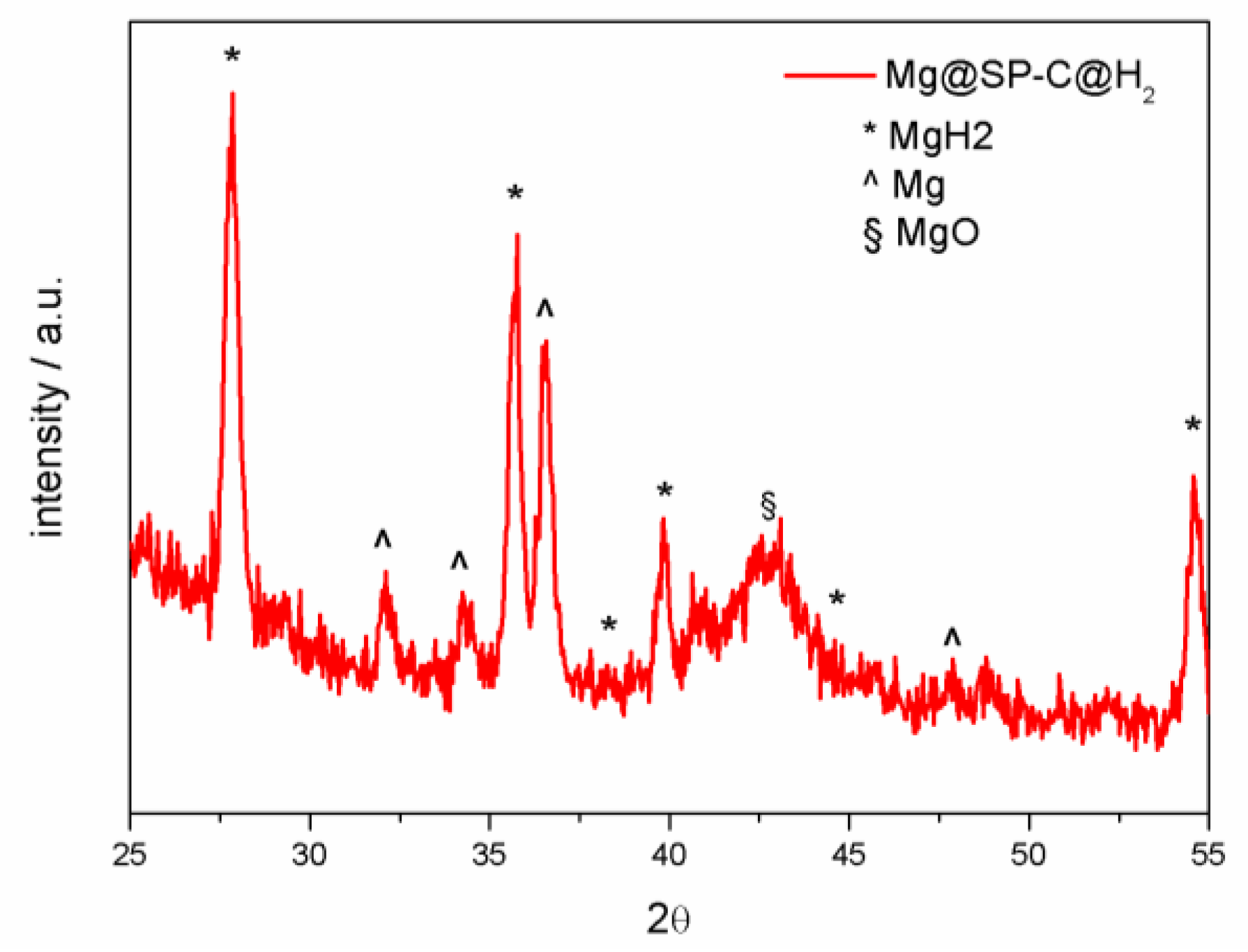
3.3. Materials Optimization Trials
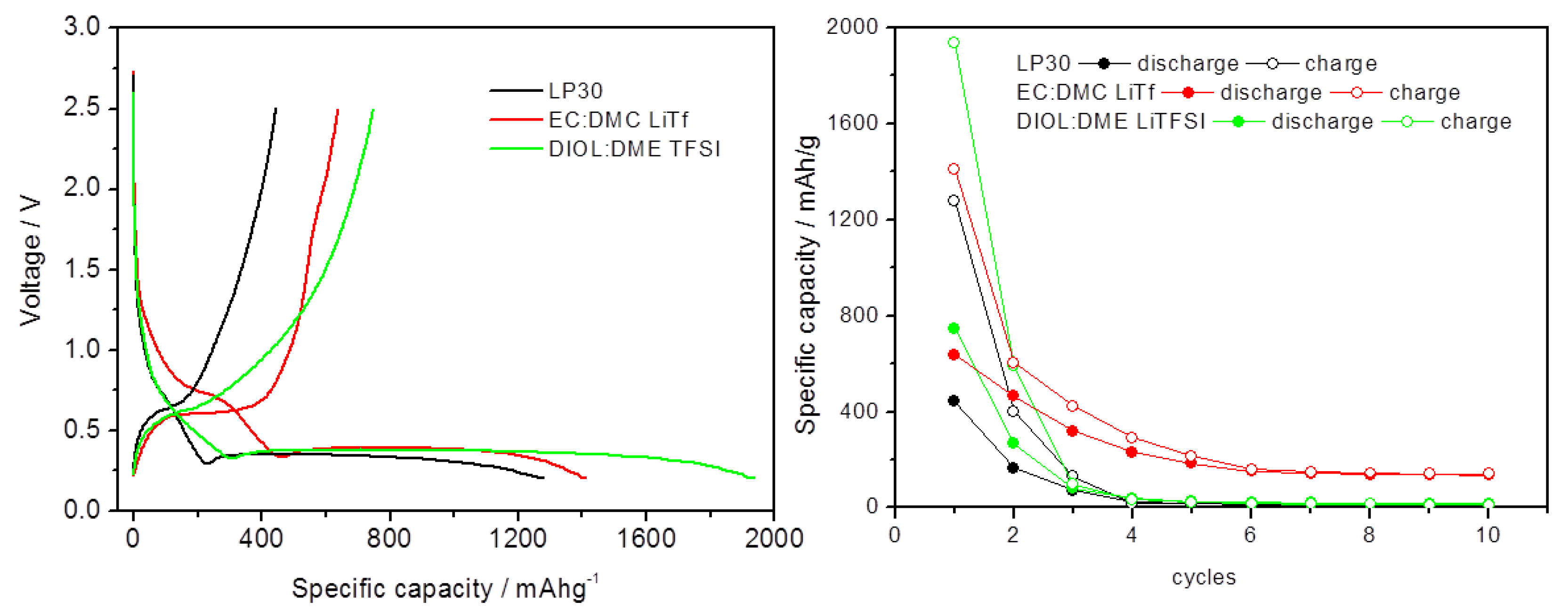
3.4. Electrochemical Cell Formulation Assessment
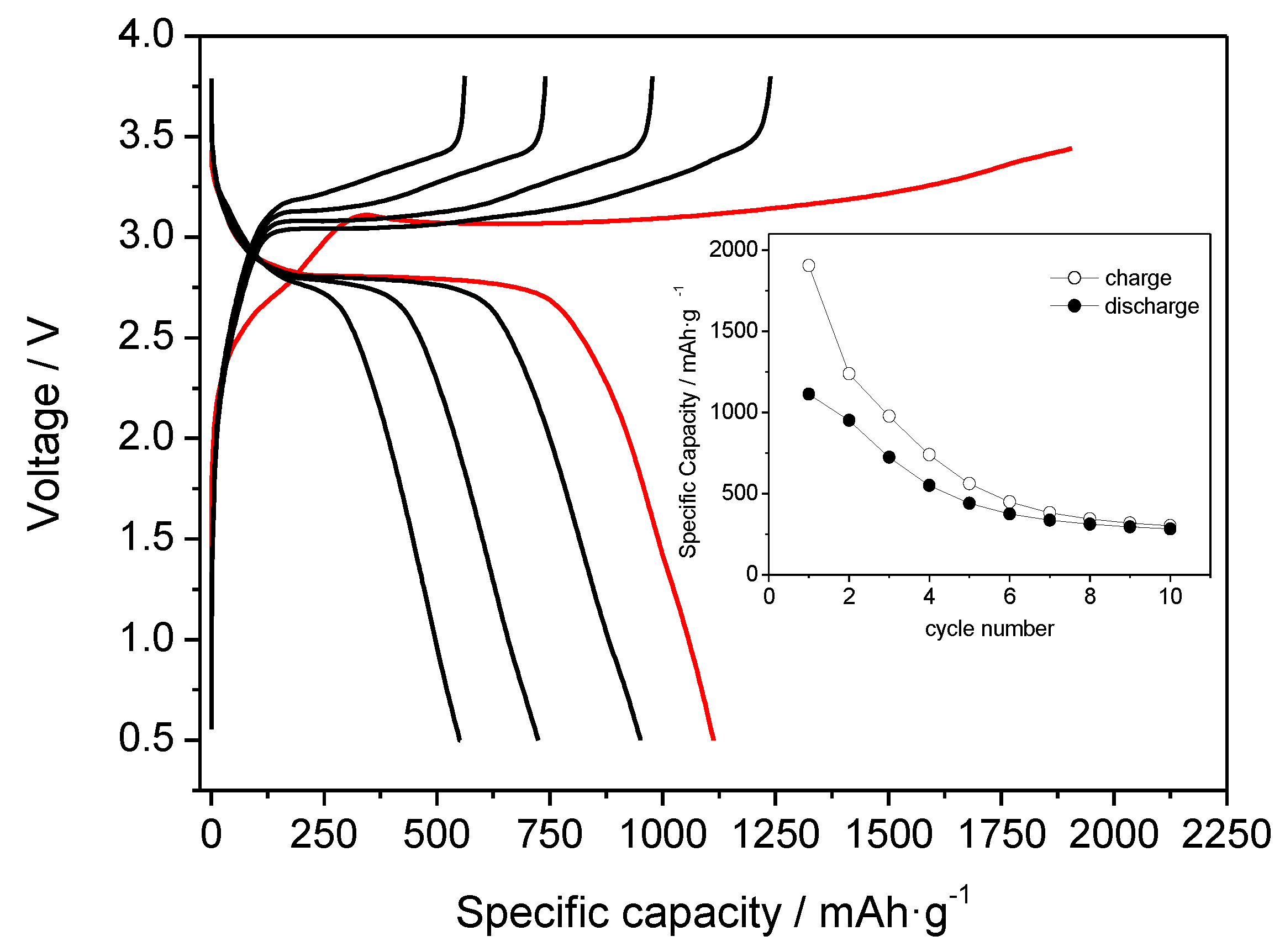
3.5. Li-Ion-Hydride Proof of Concept
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wagner, F.T.; Lakshmanan, B.; Mathias, M.F. Electrochemistry and the future of the automobile. J. Phys. Chem. Lett. 2010, 1, 2204–2219. [Google Scholar] [CrossRef]
- Tarascon, J.-M.; Armand, M. Issues and challenges facing rechargeable lithium batteries. Nature 2001, 414, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.G.; Cho, J. Reversible and high-capacity nanostructured electrode materials for Li-ion batteries. Adv. Funct. Mater. 2009, 19, 1497–1514. [Google Scholar] [CrossRef]
- Farrington, M.D. Safety of lithium batteries in transportation. J. Power Sources 2001, 96, 260–265. [Google Scholar] [CrossRef]
- Gnanaraj, J.S.; Zinigrad, E.; Asraf, L.; Gottlieb, H.E.; Sprecher, M.; Aurbach, D.; Schmidt, M. The use of accelerating rate calorimetry (ARC) for the study of the thermal reactions of Li-ion battery electrolyte solutions. J. Power Sources 2003, 119, 794–798. [Google Scholar] [CrossRef]
- Kitoh, K.; Nemoto, H. 100 Wh large size Li-ion batteries and safety tests. J. Power Sources 1999, 81, 887–890. [Google Scholar] [CrossRef]
- Ozawa, K. Lithium Ion Rechargeable Batteries; Wiley-VCH: Weinheim, Germany, 2009. [Google Scholar]
- Goals for Advanced Batteries for EVs. Available online: http://www.uscar.org/guest/publications.php (accessed on 25 January 2017).
- Bruce, P.G.; Scrosati, B.; Tarascon, J.-M. Nanomaterials for rechargeable lithium batteries. Angew. Chem. Int. Ed. 2008, 47, 2930–2946. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Wang, Z.; Chen, L.; Huang, X. Research on advanced materials for Li-ion batteries. Adv. Mater. 2009, 21, 4593–4607. [Google Scholar] [CrossRef]
- Tarascon, J.-M.; Poizot, P.; Laruelle, S.; Grugeon, S.; Dupont, L. Nano-sized transition-metal oxides as negative-electrode materials forlithium-ion batteries. Nature 2000, 407, 496–499. [Google Scholar] [CrossRef] [PubMed]
- Cabana, J.; Monconduit, L.; Larcher, D.; Palacín, M.R. Beyond intercalation-based Li-ion batteries: The state of the art and challenges of electrode materials reacting through conversion reactions. Adv. Mater. 2010, 22, 170–192. [Google Scholar] [CrossRef] [PubMed]
- Oumellal, Y.; Rougier, A.; Nazri, G.A.; Tarascon, J.-M.; Aymard, L. Metal hydrides for lithium-ion batteries. Nat. Mater. 2008, 7, 916–921. [Google Scholar] [CrossRef]
- Oumellal, Y.; Rougier, A.; Tarascon, J.M.; Aymard, L. 2LiH + M (M = Mg, Ti): New concept of negative electrode for rechargeable lithium-ion batteries. J. Power Sources 2009, 192, 698–702. [Google Scholar] [CrossRef]
- Zaidi, W.; Oumellal, Y.; Bonnet, J.P.; Zhang, J.; Cuevas, F.; Latroche, M.; Bobet, J.L.; Aymard, L. Carboxymethylcellulose and carboxymethylcellulose-formate as binders in MgH2-carbon composites negative electrode for lithium-ion batteries. J. Power Sources 2011, 196, 2854–2857. [Google Scholar] [CrossRef]
- Oumellal, Y.; Zaïdi, W.; Bonnet, J.P.; Cuevas, F.; Latroche, M.; Zhang, J.; Bobet, J.L.; Rougier, A.; Aymard, L. Reactivity of TiH2 hydride with lithium ion: Evidence for a new conversion mechanism. Int. J. Hydrogen Energy 2012, 37, 7831–7835. [Google Scholar] [CrossRef]
- Brutti, S.; Mulas, G.; Piciollo, E.; Panero, S.; Reale, P. Magnesium hydride as a high capacity negative electrode for lithium ion batteries. J. Mater. Chem. 2012, 22, 14531–14537. [Google Scholar] [CrossRef]
- Hirscher, H. Handobook of Hydrogen Storage; Wiley-VCH: Weinheim, Germany, 2010. [Google Scholar]
- Fichtner, M. Conversion materials for hydrogen storage and electrochemical applications—Concepts and similarities. J. Alloys Compd. 2011, 509, S529–S534. [Google Scholar] [CrossRef]
- IEA/DOE/SNL Hydride Databases. Available online: http://hydpark.ca.sandia.gov/DBFrame.html (accessed on 25 January 2017).
- Varin, R.; Czujko, T.; Wronski, Z. Nanomaterials for Solid State H2 Storage; Springer: Cleveland, OH, USA, 2008. [Google Scholar]
- Silvestri, L.; Farina, L.; Meggiolaro, D.; Panero, S.; Padella, F.; Brutti, S.; Reale, P. Reactivity of sodium alanates in lithium batteries. J. Phys. Chem. 2015, 119, 28766–28775. [Google Scholar] [CrossRef]
- Meggiolaro, D.; Farina, L.; Silvestri, L.; Panero, S.; Brutti, S.; Reale, P. Lightweight borohydrides electro-activity in lithium cells. Energies 2016, 9, 238. [Google Scholar] [CrossRef] [Green Version]
- Meggiolaro, D.; Gigli, G.; Paolone, A.; Vitucci, F.; Brutti, S. Incorporation of lithium by MgH2: An ab initio study. J. Phys. Chem. 2013, 117, 22467–22477. [Google Scholar] [CrossRef]
- Farina, L.; Munao, D.; Silvestri, L.; Panero, S.; Meggiolaro, D.; Brutti, S.; La Barbera, A.; Reale, P. Electrochemical activity of lightweight borohydrides in lithium cells. In Proceedings of the 2015 IEEE 15th International Conference on Environment and Electrical Engineering, EEEIC 2015, Rome, Italy, 10–13 June 2015; pp. 1827–1832.
- Silvestri, L.; Forgia, S.; Farina, L.; Meggiolaro, D.; Panero, S.; LaBarbera, A.; Brutti, S.; Reale, P. Lithium alanates as negative electrodes in lithium-ion batteries. Chem. Electro. Chem. 2015, 2, 877–886. [Google Scholar] [CrossRef]
- Riechel, T.L.; Wilkes, J.S. Reversible plating and stripping of sodium at inert electrodes in room temperature chloroaluminate molten salts. J. Electrochem. Soc. 1992, 139, 977. [Google Scholar] [CrossRef]
- Vitucci, F.M.; Paolone, A.; Brutti, S.; Munaò, D.; Silvestri, L.; Panero, S.; Reale, P. H2 thermal desorption and hydride conversion reactions in Li cells of TiH2/C amorphous nanocomposites. J. Alloys Compd. 2015, 645, S46–S50. [Google Scholar] [CrossRef]
- Farina, L.; Brutti, S.; Trequattrini, F.; Palumbo, O.; Gatto, S.; Reale, P.; Silvestri, L.; Panero, S.; Paolone, A. An extensive study of the Mg-Fe-H material obtained by reactive ball milling of MgH2 and Fe in a molar ration 3:1. Int. J. Hydrogen Energy 2017. submitted. [Google Scholar]
- Doeff, M.M.; Chen, G.; Cabana, J.; Richardson, T.J.; Mehta, A.; Shirpour, M.; Duncan, H.; Kim, C.; Kam, K.C.; Conry, T. Characterization of electrode materials for lithium ion and sodium ion batteries using synchrotron radiation techniques. J. Vis. Exp. 2013, 11, e50594. [Google Scholar] [CrossRef] [PubMed]
- Harks, P.P.R.M.L.; Mulder, F.M.; Notten, P.H. L. In situ methods for Li-ion battery research: A review of recent developments. J. Power Sources 2015, 288, 92–105. [Google Scholar] [CrossRef]
- Wimmer, E.; Christensen, M.; Eyert, V.; Wolf, W.; Reith, D.; Rozanska, X.; Freeman, C.; Saxe, P. Computational materials engineering: Recent applications of VASP in the MedeA® software environment. J. Korean Ceram. Soc. 2016, 53, 263–272. [Google Scholar] [CrossRef]
- Ben Yahia, M.; Lemoigno, F.; Rousse, G.; Boucher, F.; Tarascon, J.-M.; Doublet, M.-L. Origin of the 3.6 V to 3.9 V voltage increase in the LiFeSO4F cathodes for Li-ion batteries. Energy Environ. Sci. 2012, 5, 9584. [Google Scholar] [CrossRef]
- Arrouvel, C.; Parker, S.C.; Islam, M.S. Lithium insertion and transport in the TiO2-B anode material: A computational study. Chem. Mater. 2009, 21, 4778–4783. [Google Scholar] [CrossRef]
- Hautier, G.; Jain, A.; Ong, S.P.; Kang, B.; Moore, C.; Doe, R.; Ceder, G. Phosphates as lithium-ion battery cathodes: An evaluation based on high-throughput ab initio calculations. Chem. Mater. 2011, 23, 3495–3508. [Google Scholar] [CrossRef]
- Jónsson, E.; Johansson, P.; Xu, K. Electrochemical oxidation stability of anions for modern battery electrolytes: A CBS and DFT study. Phys. Chem. Chem. Phys. 2015, 17, 3697–3703. [Google Scholar] [CrossRef] [PubMed]
- Urban, A.; Matts, I.; Abdellahi, A.; Ceder, G. Computational design and preparation of cation-disordered oxides for high-energy-density li-ion batteries. Adv. Energy Mater. 2016, 6, 1600488. [Google Scholar] [CrossRef]
- Meggiolaro, D.; Gigli, G.; Paolone, A.; Reale, P.; Doublet, M.L.; Brutti, S. Origin of the voltage hysteresis of MgH2 electrodes in lithium batteries. J. Phys. Chem. 2015, 119, 17044–17052. [Google Scholar]
- Cirrincione, L.; Silvestri, L.; Mallia, C.; Stallworth, P.E.; Greenbaum, S.; Brutti, S.; Panero, S.; Reale, P. Investigation of the effects of mechanochemical treatment on NaAlH4 based anode materials for Li-ion batteries. J. Electrochem. Soc. 2016, 163, A2628–A2635. [Google Scholar] [CrossRef]
- Adelhelm, P.; de Jongh, P. The impact of carbon materials on the hydrogen storage propertie of lightmetal hydrides. J. Mater. Chem. 2011, 21, 2417. [Google Scholar] [CrossRef]
- Gao, J.; Adelhelm, P.; Verkuijlen, H.W.; Rongeat, C.; Herrich, M.; van Bentum, P.J.M.; Gutfleisch, O.; Kentgens, A.P.M.; de Jong, K.P.; de Jongh, P.E. Confinement of NaAlH4 in nanoporous carbon: impact on H2 release, reversibility and thermodynamics. J. Phys. Chem. 2010, 114, 4675. [Google Scholar]
- Oumellal, Y.; Zlotea, C.; Bastide, S.; Cachet-Vivier, C.; Léonel, E.; Sengmany, S.; Leroy, E.; Aymard, L.; Bonnet, J.-P.; Latroche, M. Bottom-up preparation of MgH₂ nanoparticles with enhanced cycle life stability during electrochemical conversion in Li-ion batteries. Nanoscale 2014, 6, 14459–14466. [Google Scholar] [CrossRef] [PubMed]
- Silvestri, L.; Paolone, A.; Cirrincione, L.; Stallworth, P.E.; Greenbaum, S.; Panero, S.; Brutti, S.; Reale, P. NaAlH4 nanoconfinement in mesoporous carbon for application in lithium ion batteries. J. Electrochem. Soc. 2017. submitted. [Google Scholar]





| Hydride | Symmetry Space Group | Formula Weight (g·mol−1) | Theoretical Capacity (mAh·g−1) |
|---|---|---|---|
| MgH2 | Pbcn | 26.3 | 2037 |
| NaH | Fm-3m | 24.0 | 1117 |
| TiH2 | I4/mmm | 49.9 | 1075 |
| CaH2 | Pnma | 42.1 | 1273 |
| LiBH4 | Pnma | 21.8 | 4992 |
| NaBH4 | P-421c | 37.8 | 2834 |
| KBH4 | P421c | 53.9 | 1987 |
| Mg(BH4)2 | P6122 | 54.0 | 3971 |
| Ca(BH4)2 | α Fddd | 69.8 | 3074 |
| β P42/m | |||
| LiAlH4 | P21/c | 37.9 | 2119 |
| Li3AlH6 | R-3 | 53.8 | 1493 |
| NaAlH4 | I41/a | 54.0 | 1985 |
| Na2LiAlH6 | Fm-3m | 85.9 | 1559 |
| Na3AlH6 | P21/n | 102.0 | 1578 |
| Mg2NiH4 | C2/c | 111.4 | 963 |
| Mg2FeH6 | Fm-3m | 110.5 | 1455 |
| Reaction | Theoretical Emf (V) |
|---|---|
| MgH2 + 2 Li = Mg + 2 LiH | 0.53 |
| NaH + Li = Na + LiH | 0.43 |
| TiH2 + 2 Li = Ti + 2 LiH | 0.15 |
| CaH2 + 2 Li = Ca + 2 LiH | −0.02 |
| Hydride | Theoretical Capacity (mAh·g−1) | Ball Milling Time (hours) | Ball Milling Time with Carbon (5:3 Ratio between Borohydride and Carbon) (hours) | First Discharge Capacity (mAh·g−1) | Li Equivalents Incorporated in PCGA Tests (First Discharge) |
|---|---|---|---|---|---|
| LiBH4 | 4992 | 15 | 5 | 52 | 0.04 |
| NaBH4 | 2834 | 1 | ½ | 74 | 0.10 |
| 15 | 5 | 211 | 0.30 | ||
| KBH4 | 1987 | 15 | 5 | 66 | 0.13 |
| Mg(BH4)2 | 3971 | 1 | ½ | 539 | 1.09 |
| 15 | 5 | 205 | 0.41 | ||
| Ca(BH4)2 | 3074 | 15 | 5 | 9 | 0.003 |
| Reaction | Theoretical Emf (V) |
|---|---|
| 2 NaAlH4 + 3 Li = LiNa2AlH6 + 2 LiH + Al | 0.73 |
| 3/2 LiNa2AlH6 + 3/2 Li = 3 LiH + 1/2 Al + Na3AlH6 | 0.66 |
| Na3AlH6 + 3 Li = 3 NaH + Al + 3 LiH | 0.61 |
| NaH + Li = Na + LiH | 0.43 |
| 3LiAlH4 + 6e + 6Li+ ⇄ Li3AlH6 + 2Al+6LiH | 0.86 |
| Li3AlH6 + 3e+3Li+ ⇄ 6LiH + Al | 0.74 |
| Al + Li ⇄ AlLi | 0.29 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brutti, S.; Panero, S.; Paolone, A.; Gatto, S.; Meggiolaro, D.; Vitucci, F.M.; Manzi, J.; Munaò, D.; Silvestri, L.; Farina, L.; et al. Hydrides as High Capacity Anodes in Lithium Cells: An Italian “Futuro in Ricerca di Base FIRB-2010” Project. Challenges 2017, 8, 8. https://doi.org/10.3390/challe8010008
Brutti S, Panero S, Paolone A, Gatto S, Meggiolaro D, Vitucci FM, Manzi J, Munaò D, Silvestri L, Farina L, et al. Hydrides as High Capacity Anodes in Lithium Cells: An Italian “Futuro in Ricerca di Base FIRB-2010” Project. Challenges. 2017; 8(1):8. https://doi.org/10.3390/challe8010008
Chicago/Turabian StyleBrutti, Sergio, Stefania Panero, Annalisa Paolone, Sara Gatto, Daniele Meggiolaro, Francesco M. Vitucci, Jessica Manzi, David Munaò, Laura Silvestri, Luca Farina, and et al. 2017. "Hydrides as High Capacity Anodes in Lithium Cells: An Italian “Futuro in Ricerca di Base FIRB-2010” Project" Challenges 8, no. 1: 8. https://doi.org/10.3390/challe8010008








