DOM Biological Lability in an Estuarine System in Two Contrasting Periods
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples Collection and Incubation Experiment Setup
2.2. Analytical Procedures for DOM and Heterotrophic Prokaryotes Abundance
2.3. Statistics
3. Results and Discussion
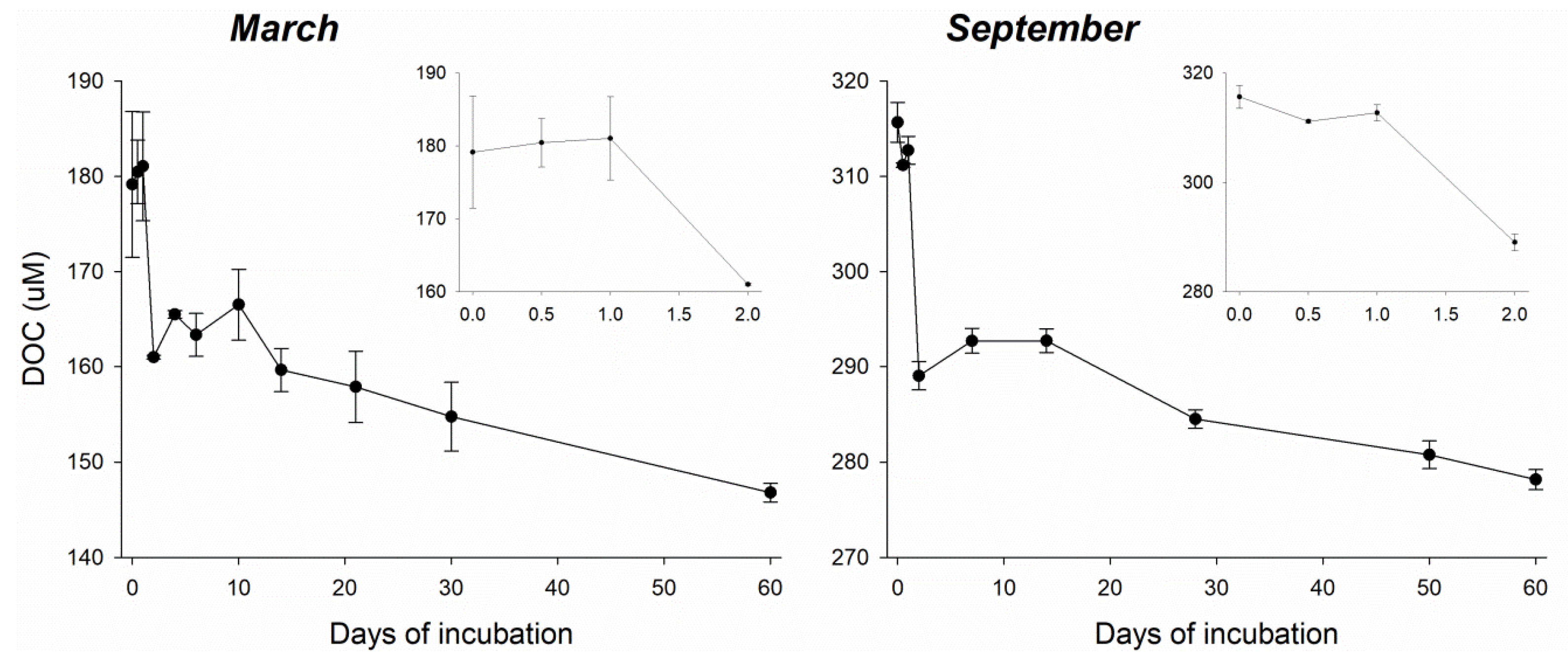
3.1. Biological Removal of DOC
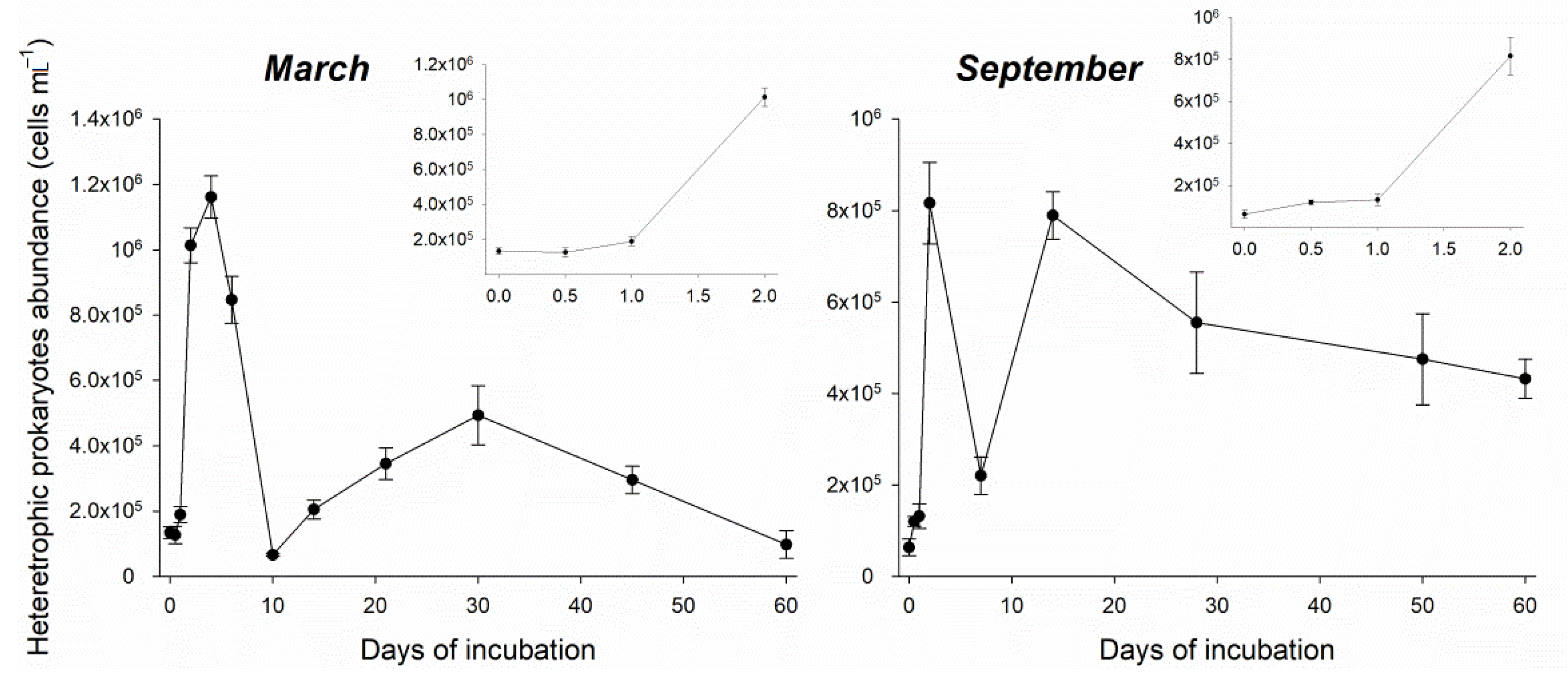
3.2. Heterotrophic Prokaryotes Growth Efficiency and CO2 Production Estimates
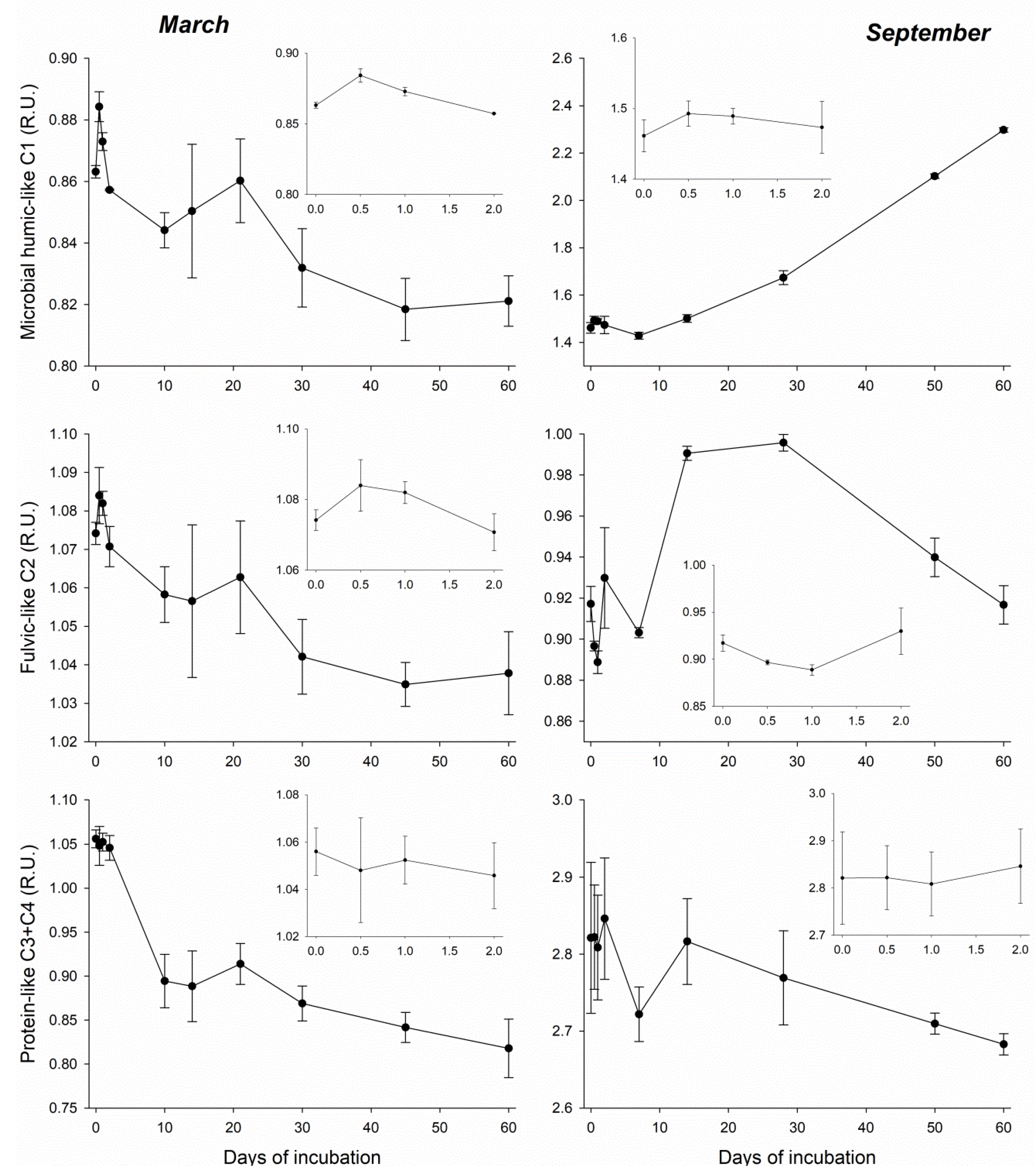
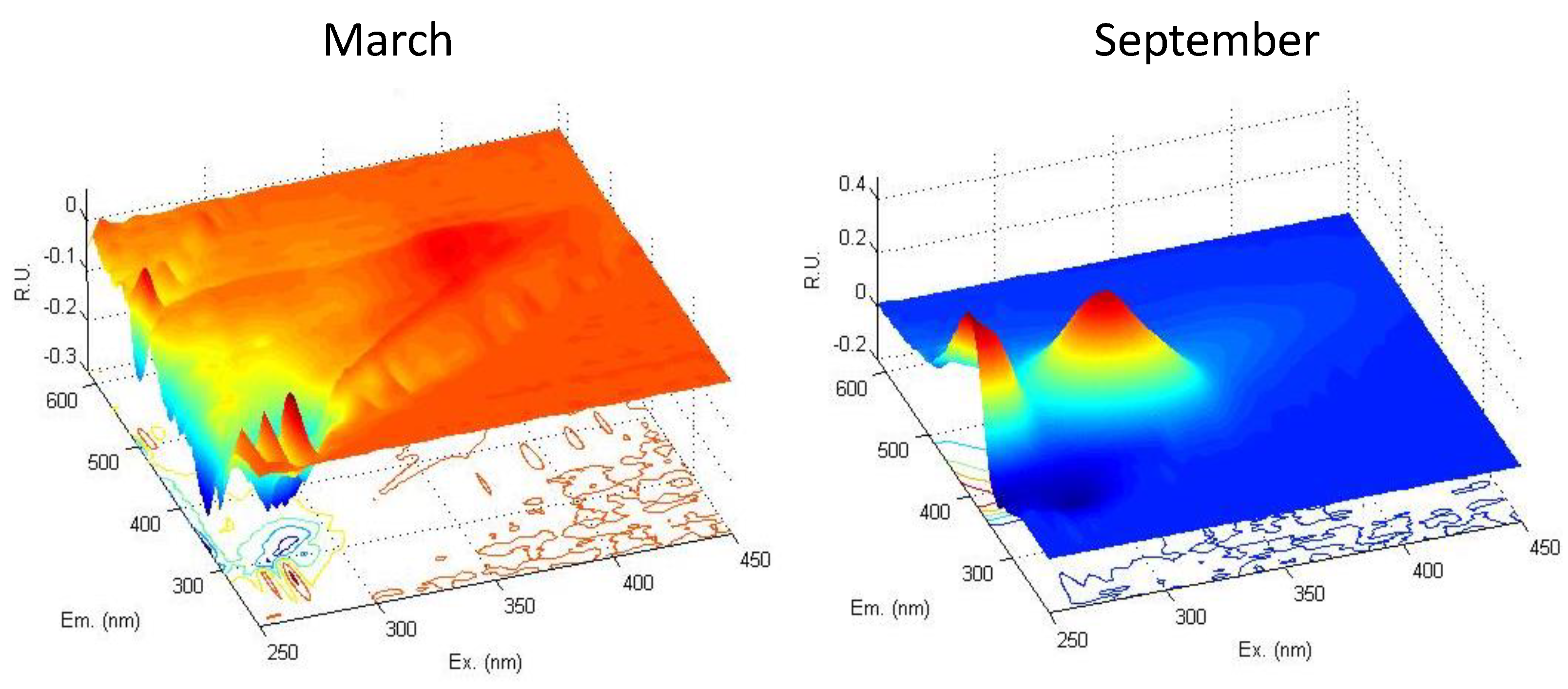
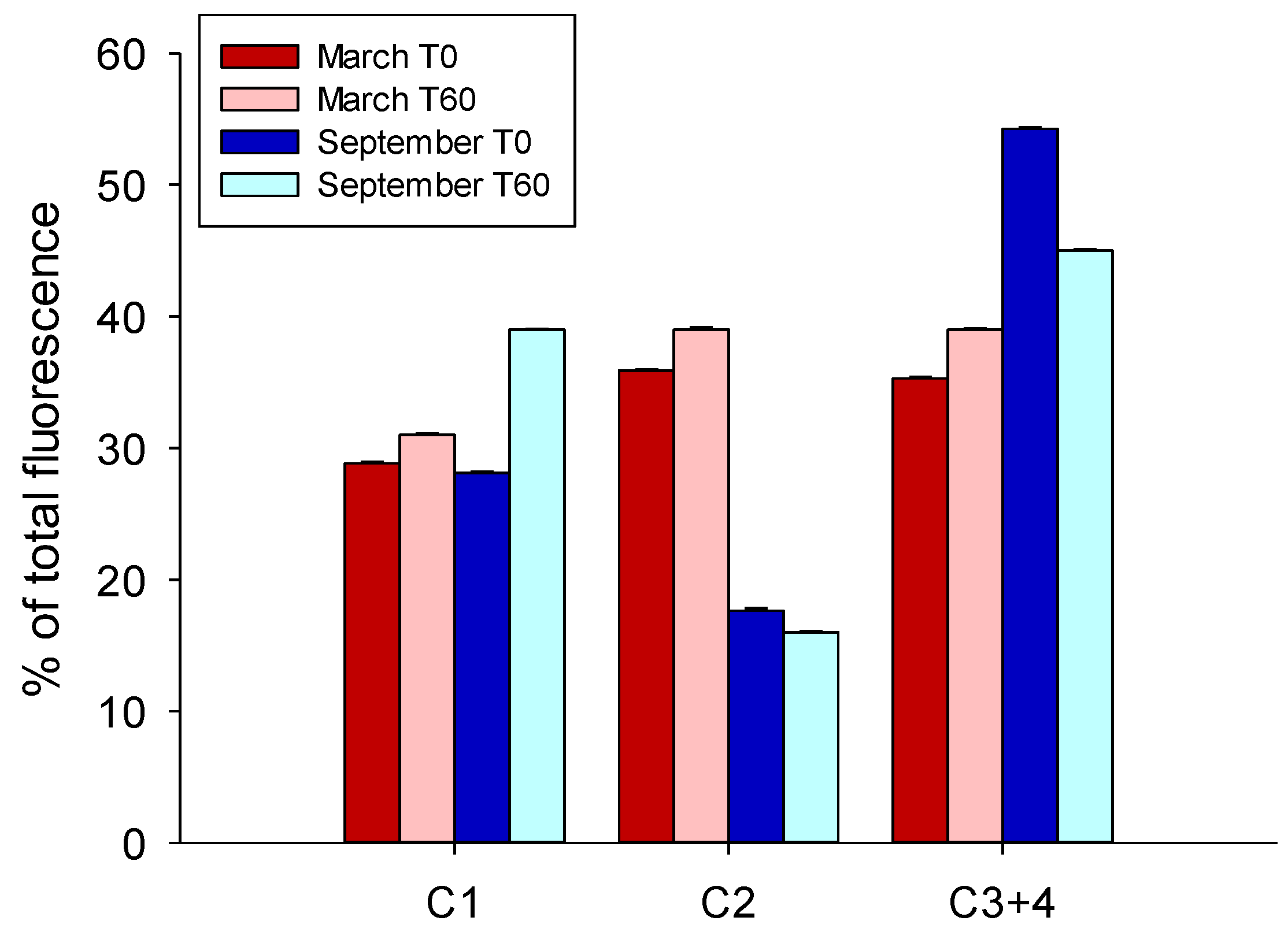
3.3. DOM Quality Affects Its Mineralization and Vice Versa
4. Summary and Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hedges, J.I.; Keil, R.G.; Benner, R. What happens to terrestrial organic matter in the ocean? Org. Geochem. 1997, 27, 195–212. [Google Scholar] [CrossRef]
- Raymond, P.A.; Spencer, R.G.M. Riverine DOM. In Biogeochemistry of Marine Dissolved Organic Matter, 2nd ed.; Academic Press: Cambridge, MA, USA, 2015; pp. 509–533. ISBN 9780124059405. [Google Scholar]
- Bauer, J.E.; Cai, W.-J.; Raymond, P.A.; Bianchi, T.S.; Hopkinson, C.S.; Regnier, P.A.G. The changing carbon cycle of the coastal ocean. Nature 2013, 504, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Benner, R. What happens to terrestrial organic matter in the ocean? Mar. Chem. 2004, 92, 307–310. [Google Scholar] [CrossRef]
- Richey, J.E. Pathways of Atmospheric CO2 through Fluvial Systems. In The Global Carbon Cycle: Integrating Humans, Climate and the Natural World; Island Press: Washington, DC, USA, 2004; pp. 329–340. [Google Scholar]
- Sabine, C.L.; Feely, R.A.; Gruber, N.; Key, R.M.; Lee, K.; Bullister, J.L.; Wanninkhof, R.; Wong, C.S.; Wallace, D.W.R.; Tilbrook, B.; et al. The oceanic sink for anthropogenic CO2. Science 2004, 305, 367–371. [Google Scholar] [CrossRef] [PubMed]
- Hernes, P.J.; Benner, R. Photochemical and microbial degradation of dissolved lignin phenols: Implications for the fate of terrigenous dissolved organic matter in marine environments. J. Geophys. Res. Ocean. 2003, 108, 1421. [Google Scholar] [CrossRef]
- Shank, G.C.; Zepp, R.G.; Whitehead, R.F.; Moran, M.A. Variations in the spectral properties of freshwater and estuarine CDOM caused by partitioning onto river and estuarine sediments. Estuar. Coast. Shelf Sci. 2005, 65, 289–301. [Google Scholar] [CrossRef]
- Uher, G.; Hughes, C.; Henry, G.; Upstill-Goddard, R.C. Non-conservative mixing behavior of colored dissolved organic matter in a humic-rich, turbid estuary. Geophys. Res. Lett. 2001, 28, 3309–3312. [Google Scholar] [CrossRef]
- Riedel, T.; Zark, M.; Vähätalo, A.V.; Niggemann, J.; Spencer, R.G.M.; Hernes, P.J.; Dittmar, T. Molecular signatures of biogeochemical transformations in dissolved organic matter from ten world rivers. Front. Earth Sci. 2016, 4, 85. [Google Scholar] [CrossRef]
- Paerl, H.W. Assessing and managing nutrient-enhanced eutrophication in estuarine and coastal waters: Interactive effects of human and climatic perturbations. Ecol. Eng. 2006, 26, 40–54. [Google Scholar] [CrossRef]
- Crump, B.C.; Hopkinson, C.S.; Sogin, M.L.; Hobbie, J.E. Microbial Biogeography along an Estuarine Salinity Gradient: Combined Influences of Bacterial Growth and Residence Time. Appl. Environ. Microbiol. 2004, 70, 1494–1505. [Google Scholar] [CrossRef] [PubMed]
- Lozupone, C.A.; Knight, R. Global patterns in bacterial diversity. Proc. Natl. Acad. Sci. USA 2007, 104, 11436–11440. [Google Scholar] [CrossRef]
- Troussellier, M.; Schäfer, H.; Batailler, N.; Bernard, L.; Courties, C.; Lebaron, P.; Muyzer, G.; Servais, P.; Vives-Rego, J. Bacterial activity and genetic richness along an estuarine gradient (Rhone River plume, France). Aquat. Microb. Ecol. 2002, 28, 028013. [Google Scholar] [CrossRef]
- Crump, B.C.; Armbrust, E.V.; Baross, J.A. Phylogenetic analysis of particle-attached and free-living bacterial communities in the Columbia River, its estuary, and the adjacent coastal ocean. Appl. Environ. Microbiol. 1999, 65, 3192–3204. [Google Scholar] [CrossRef]
- Goñi-Urriza, M.S.; Point, D.; Amouroux, D.; Guyoneaud, R.; Donard, O.F.X.; Caumette, P.; Duran, R. Bacterial community structure along the Adour estuary (French Atlantic coast): Influence of salinity gradient versus metal contamination. Aquat. Microb. Ecol. 2007, 49, 47–56. [Google Scholar] [CrossRef][Green Version]
- Apple, J.K.; del Giorgio, P.A.; Kemp, W.M. Temperature regulation of bacterial production, respiration, and growth efficiency in a temperate salt-marsh estuary. Aquat. Microb. Ecol. 2006, 43, 243–254. [Google Scholar] [CrossRef]
- Raymond, P.; Bauer, J. Bacterial consumption of DOC during transport through a temperate estuary. Aquat. Microb. Ecol. 2000, 22, 1–12. [Google Scholar] [CrossRef]
- Ram, A.S.P.; Nair, S.; Chandramohan, D. Bacterial growth efficiency in a tropical estuary: Seasonal variability subsidized by allochthonous carbon. Microb. Ecol. 2007, 53, 591–599. [Google Scholar] [CrossRef]
- Brogi, S.R.; Balestra, C.; Casotti, R.; Cossarini, G.; Galletti, Y.; Gonnelli, M.; Vestri, S.; Santinelli, C. Time resolved data unveils the complex DOM dynamics in a Mediterranean river. Sci. Total Environ. 2020, 733, 139212. [Google Scholar] [CrossRef] [PubMed]
- Sherr, E.; Sherr, B. Understanding Roles of Microbes in Marine Pelagic Food Webs: A Brief History. In Microbial Ecology of the Oceans, 2nd ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2008; ISBN 9780470043448. [Google Scholar]
- Gasol, J.M.; Zweifel, U.L.; Peters, F.; Fuhrman, J.A.; Hagström, Å. Significance of size and nucleic acid content heterogeneity as measured by flow cytometry in natural planktonic bacteria. Appl. Environ. Microbiol. 1999, 65, 4475–4483. [Google Scholar] [CrossRef]
- Grégori, G.; Citterio, S.; Ghiani, A.; Labra, M.; Sgorbati, S.; Brown, S.; Denis, M. Resolution of Viable and Membrane-Compromised Bacteria in Freshwater and Marine Waters Based on Analytical Flow Cytometry and Nucleic Acid Double Staining. Appl. Environ. Microbiol. 2001, 67, 4662–4670. [Google Scholar] [CrossRef] [PubMed]
- Santinelli, C.; Follett, C.; Brogi, S.R.; Xu, L.; Repeta, D. Carbon isotope measurements reveal unexpected cycling of dissolved organic matter in the deep Mediterranean Sea. Mar. Chem. 2015, 177, 267–277. [Google Scholar] [CrossRef]
- Hansell, D.A. Dissolved Organic Carbon Reference Material Program. Eos Trans. Am. Geophys. Union 2005, 86, 318. [Google Scholar] [CrossRef]
- Ducklow, H.W. Bacterial Production and Biomass in the Oceans; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2000; pp. 85–120. [Google Scholar]
- Brogi, S.R.; Charrière, B.; Gonnelli, M.; Vaultier, F.; Sempéré, R.; Vestri, S.; Santinelli, C. Effect of uv and visible radiation on optical properties of chromophoric dissolved organic matter released by emiliania huxleyi. J. Mar. Sci. Eng. 2020, 8, 888. [Google Scholar] [CrossRef]
- Murphy, K.R.; Stedmon, C.A.; Graeber, D.; Bro, R. Fluorescence spectroscopy and multi-way techniques. PARAFAC. Anal. Methods 2013, 5, 6557. [Google Scholar] [CrossRef]
- Sokal, R.R.; Rohlf, F.J. Biometry: The Principles and Practice of Statistics in Biological Research; W H Freeman & Company: New York, NY, USA, 1995; ISBN 0716724111. [Google Scholar]
- Retelletti Brogi, S.; Gonnelli, M.; Vestri, S.; Santinelli, C. Biophysical processes affecting DOM dynamics at the Arno river mouth (Tyrrhenian Sea). Biophys. Chem. 2015, 197, 1–9. [Google Scholar] [CrossRef]
- Avery, G.B.; Willey, J.D.; Kieber, R.J.; Shank, G.C.; Whitehead, R.F. Flux and bioavailability of Cape Fear River and rainwater dissolved organic carbon to Long Bay, southeastern United States. Glob. Biogeochem. Cycles 2003, 17, 2229. [Google Scholar] [CrossRef]
- Moran, M.A.; Sheldon, J.E. Biodegradation of Riverine Dissolved Estuaries of the Southeastern United Organic States Carbon in Five. Estuaries 1999, 22, 55–64. [Google Scholar] [CrossRef]
- Riemann, L.; Middelboe, M. Viral lysis of marine bacterioplankton: Implications for organic matter cycling and bacterial clonal composition. Ophelia 2002, 10409490. [Google Scholar] [CrossRef]
- Murphy, K.R.; Stedmon, C.A.; Wenig, P.; Bro, R. OpenFluor—An online spectral library of auto-fluorescence by organic compounds in the environment. Anal. Methods 2014, 6, 658–661. [Google Scholar] [CrossRef]
- Lambert, T.; Teodoru, C.R.; Nyoni, F.C.; Bouillon, S.; Darchambeau, F.F.; Massicotte, P.; Borges, A.V. Along-stream transport and transformation of dissolved organic matter in a large tropical river. Biogeosciences 2016, 13, 2727–2741. [Google Scholar] [CrossRef]
- Maie, N.; Sekiguchi, S.; Watanabe, A.; Tsutsuki, K.; Yamashita, Y.; Melling, L.; Cawley, K.M.; Shima, E.; Jaffé, R. Dissolved organic matter dynamics in the oligo/meso-haline zone of wetland-influenced coastal rivers. J. Sea Res. 2014, 91, 58–69. [Google Scholar] [CrossRef]
- Meng, F.; Huang, G.; Yang, X.; Li, Z.; Li, J.; Cao, J.; Wang, Z.; Sun, L. Identifying the sources and fate of anthropogenically impacted dissolved organic matter (DOM) in urbanized rivers. Water Res. 2013, 47, 5027–5039. [Google Scholar] [CrossRef] [PubMed]
- Lapierre, J.F.; del Giorgio, P.A. Partial coupling and differential regulation of biologically and photochemically labile dissolved organic carbon across boreal aquatic networks. Biogeosciences 2014, 11, 5969–5985. [Google Scholar] [CrossRef]
- Asmala, E.; Autio, R.; Kaartokallio, H.; Stedmon, C.A.; Thomas, D.N. Processing of humic-rich riverine dissolved organic matter by estuarine bacteria: Effects of predegradation and inorganic nutrients. Aquat. Sci. 2014, 76, 451–463. [Google Scholar] [CrossRef]
- Kinsey, J.D.; Corradino, G.; Ziervogel, K.; Schnetzer, A.; Osburn, C.L. Formation of chromophoric dissolved organic matter by bacterial degradation of phytoplankton-derived aggregates. Front. Mar. Sci. 2018, 4, 430. [Google Scholar] [CrossRef]
- Goto, S.; Tada, Y.; Suzuki, K.; Yamashita, Y. Evaluation of the Production of Dissolved Organic Matter by Three Marine Bacterial Strains. Front. Microbiol. 2020, 11, 2553. [Google Scholar] [CrossRef] [PubMed]
- Lønborg, C.; Álvarez-Salgado, X.A.; Davidson, K.; Miller, A.E.J. Production of bioavailable and refractory dissolved organic matter by coastal heterotrophic microbial populations. Estuar. Coast. Shelf Sci. 2009, 82, 682–688. [Google Scholar] [CrossRef]
- Arai, K.; Wada, S.; Shimotori, K.; Omori, Y.; Hama, T. Production and degradation of fluorescent dissolved organic matter derived from bacteria. J. Oceanogr. 2018, 74, 39–52. [Google Scholar] [CrossRef]
- Logue, J.B.; Stedmon, C.A.; Kellerman, A.M.; Nielsen, N.J.; Andersson, A.F.; Laudon, H.; Lindström, E.S.; Kritzberg, E.S. Experimental insights into the importance of aquatic bacterial community composition to the degradation of dissolved organic matter. ISME J. 2016, 10, 533–545. [Google Scholar] [CrossRef] [PubMed]
- Logue, J.B.; Lindström, E.S. Biogeography of Bacterioplankton in Inland Waters. Freshw. Rev. 2008, 1, 99–114. [Google Scholar] [CrossRef]
- Weiss, M.S.; Abele, U.; Weckesser, J.; Welte, W.; Schiltz, E.; Schulz, G.E. Molecular architecture and electrostatic properties of a bacterial porin. Science 1991, 254, 1627–1630. [Google Scholar] [CrossRef] [PubMed]
- Berlemont, R.; Martiny, A.C. Phylogenetic distribution of potential cellulases in bacteria. Appl. Environ. Microbiol. 2013, 79, 1545–1554. [Google Scholar] [CrossRef] [PubMed]
- Romera-Castillo, C.; Sarmento, H.; Alvarez-Salgado, X.A.; Gasol, J.M.; Marrasé, C. Net production and consumption of fluorescent colored dissolved organic matter by natural bacterial assemblages growing on marine phytoplankton exudates. Appl. Environ. Microbiol. 2011, 77, 7490–7498. [Google Scholar] [CrossRef]
- Stubbins, A.; Lapierre, J.-F.F.; Berggren, M.; Prairie, Y.T.; Dittmar, T.; del Giorgio, P.A. What’s in an EEM? Molecular signatures associated with dissolved organic fluorescence in boreal Canada. Environ. Sci. Technol. 2014, 48, 10598–10606. [Google Scholar] [CrossRef] [PubMed]
- Osterholz, H.; Kirchman, D.L.; Niggemann, J.; Dittmar, T. Diversity of bacterial communities and dissolved organic matter in a temperate estuary. FEMS Microbiol. Ecol. 2018, 94, fiy119. [Google Scholar] [CrossRef]


| March | September | |
|---|---|---|
| Temperature in situ (°C) | 11.5 | 22.2 |
| Salinity | 12.7 | 18.7 |
| DOC (µM) | 179 | 316 |
| HPA (cells mL−1) | 1.3 × 105 | 6.3 × 104 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Retelletti Brogi, S.; Casotti, R.; Misson, B.; Balestra, C.; Gonnelli, M.; Vestri, S.; Santinelli, C. DOM Biological Lability in an Estuarine System in Two Contrasting Periods. J. Mar. Sci. Eng. 2021, 9, 172. https://doi.org/10.3390/jmse9020172
Retelletti Brogi S, Casotti R, Misson B, Balestra C, Gonnelli M, Vestri S, Santinelli C. DOM Biological Lability in an Estuarine System in Two Contrasting Periods. Journal of Marine Science and Engineering. 2021; 9(2):172. https://doi.org/10.3390/jmse9020172
Chicago/Turabian StyleRetelletti Brogi, Simona, Raffaella Casotti, Benjamin Misson, Cecilia Balestra, Margherita Gonnelli, Stefano Vestri, and Chiara Santinelli. 2021. "DOM Biological Lability in an Estuarine System in Two Contrasting Periods" Journal of Marine Science and Engineering 9, no. 2: 172. https://doi.org/10.3390/jmse9020172
APA StyleRetelletti Brogi, S., Casotti, R., Misson, B., Balestra, C., Gonnelli, M., Vestri, S., & Santinelli, C. (2021). DOM Biological Lability in an Estuarine System in Two Contrasting Periods. Journal of Marine Science and Engineering, 9(2), 172. https://doi.org/10.3390/jmse9020172









