Non-Indigenous Species on Artificial Coastal Environments: Experimental Comparison between Aquaculture Farms and Recreational Marinas
Abstract
:1. Introduction
2. Materials and Methods
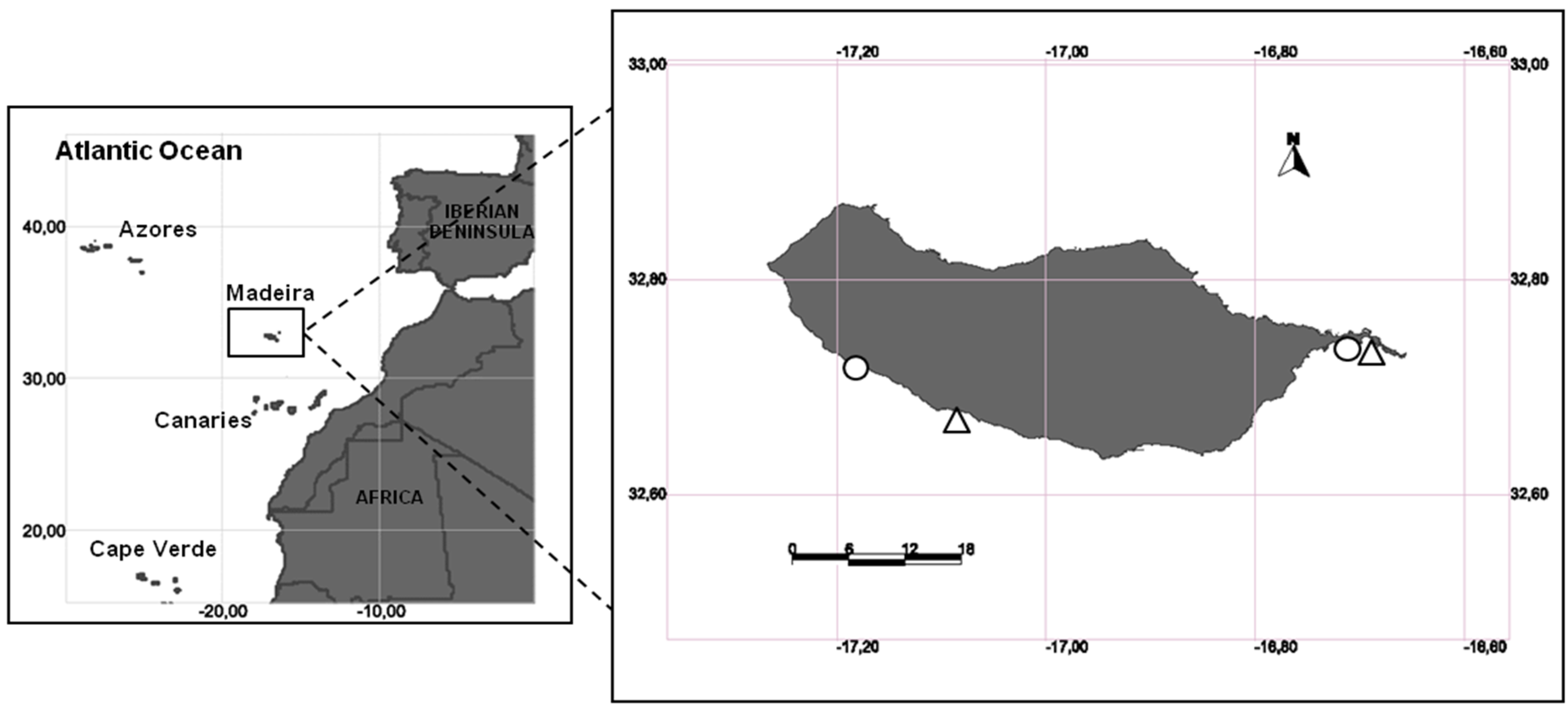
2.1. Study Area and Sampling Design
2.2. Statistical Analyses
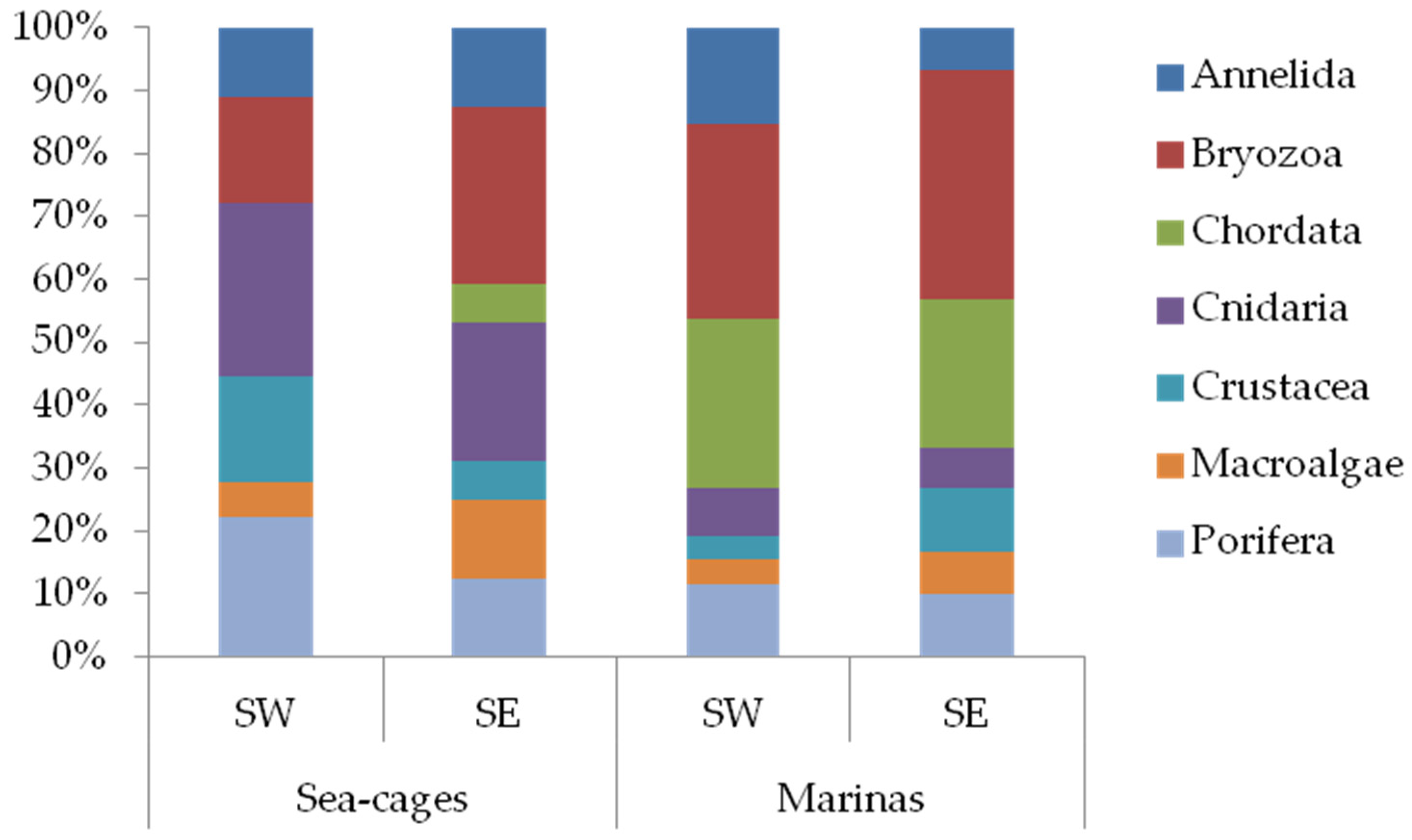
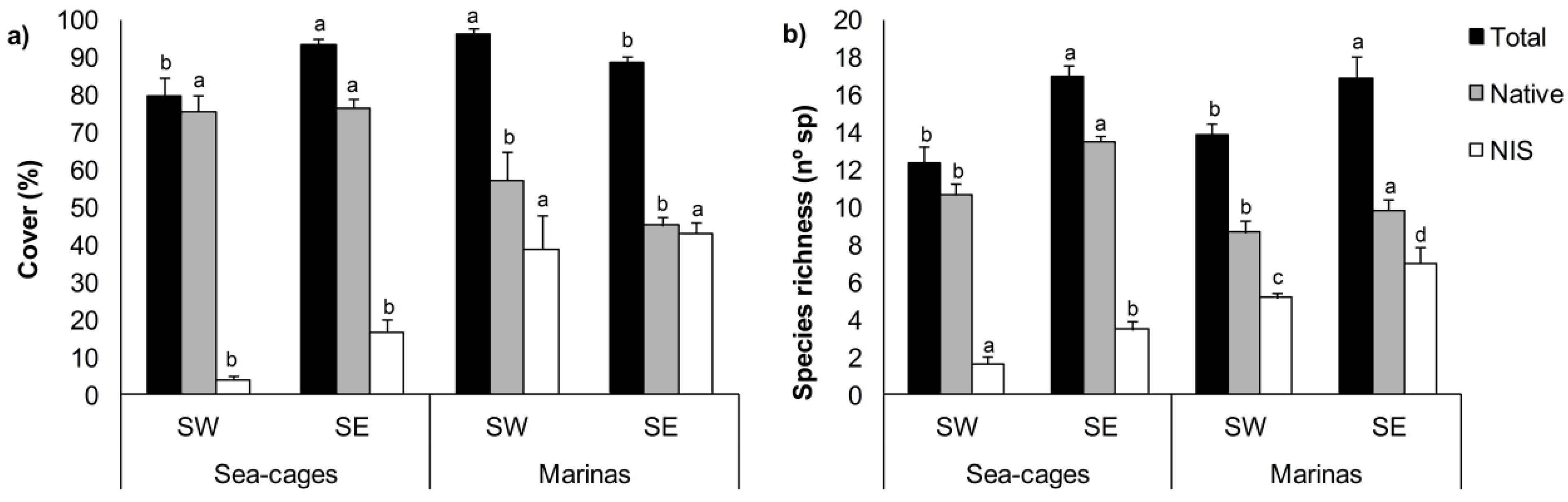
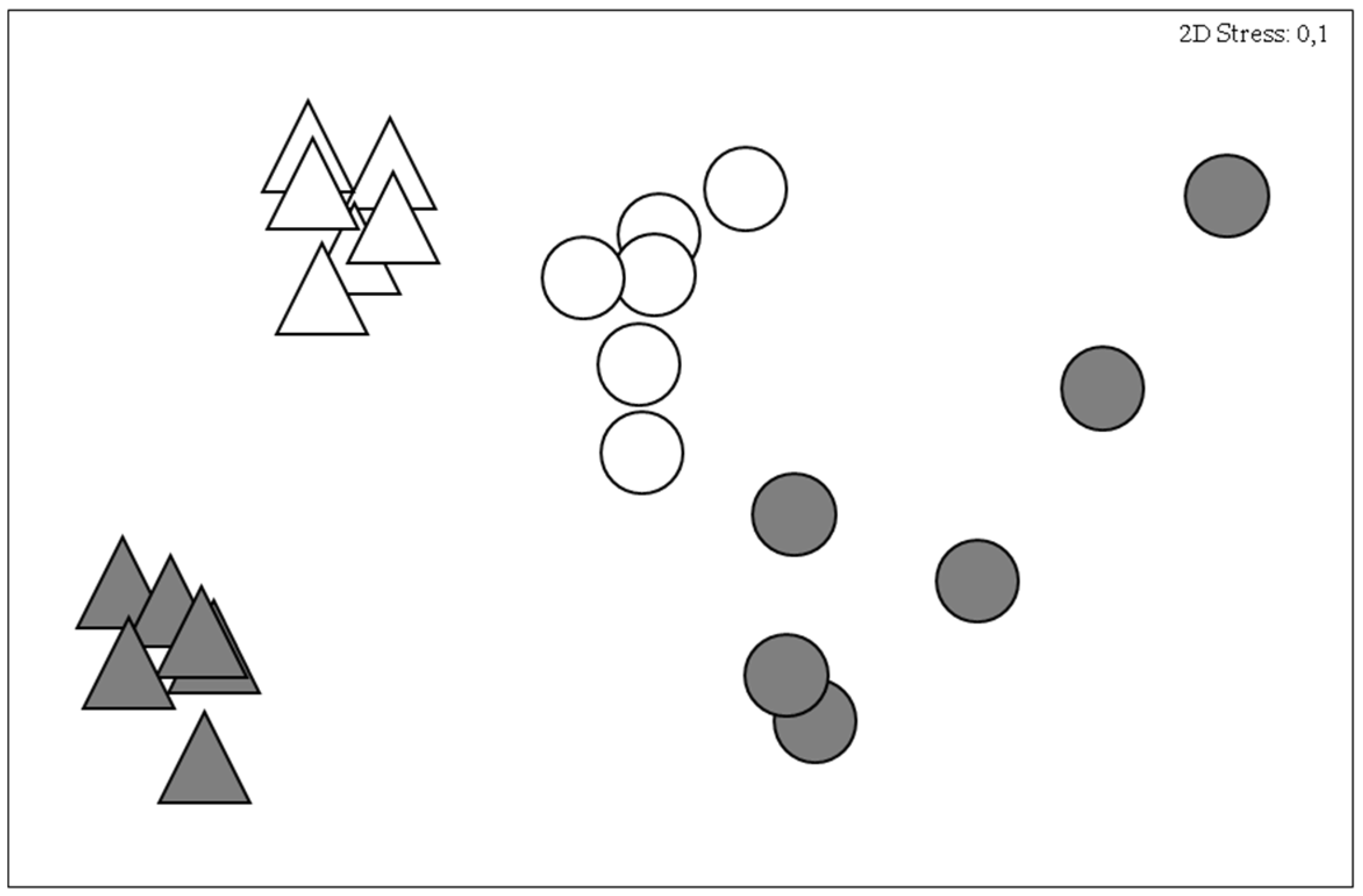
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Png-Gonzalez, L.; Andrade, C.; Abramic, A.; Nogueira, N. Analysis of the Aquaculture Industry in Macaronesia under MSFD; Technical Report for PLASMAR Project: 2019. Available online: http://hdl.handle.net/10553/55195 (accessed on 12 October 2021).
- Brito Hernández, A. Biogeografía y conservación de la biodiversidad marina en la Macaronesia. Rev. Acad. Canar. Cienc. 2010, XXII, 215–229. [Google Scholar]
- Madruga, L.; Wallenstein, F.; Azevedo, J.M.N. Regional Ecosystem Profile—Macaronesian Region; Technical Report for European Commission: 2016. Available online: https://ec.europa.eu/environment/nature/biodiversity/best/regions/index_en.htm (accessed on 12 October 2021).
- Direção Regional de Estatística da Madeira. Available online: https://estatistica.madeira.gov.pt (accessed on 27 July 2021).
- Fernandez-Gonzalez, V.; Sanchez-Jerez, P. Fouling assemblages associated with off-coast aquaculture facilities: An overall assessment of the Mediterranean Sea. Mediterr. Mar. Sci. 2017, 18, 87–96. [Google Scholar] [CrossRef]
- Dürr, S.; Watson, D.I. Biofouling and antifouling in aquaculture. In Biofouling; Dürr, S., Thomason, J.C., Eds.; Wiley-Blackwell: West Sussex, UK, 2009; pp. 267–287. [Google Scholar]
- Bannister, J.; Sievers, M.; Bush, F.; Bloecher, N. Biofouling in marine aquaculture: A review of recent research and developments. Biofouling 2019, 35, 631–648. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gansel, L.C.; Plew, D.R.; Endresen, P.C.; Olsen, A.I.; Misimi, E.; Guenther, J.; Jensen, Ø. Drag of clean and fouled net panels—Measurements and parameterization of fouling. PLoS ONE 2015, 10, e0131051. [Google Scholar] [CrossRef] [PubMed]
- Giangrande, A.; Lezzi, M.; Del Pasqua, M.; Pierri, C.; Longo, C.; Gravina, M.F. Two cases study of fouling colonization patterns in the Mediterranean Sea in the perspective of integrated aquaculture systems. Aquacult. Rep. 2020, 18, 1004555. [Google Scholar] [CrossRef]
- Fitridge, I.; Dempster, T.; Guenther, J.; de Nys, R. The impact and control of biofouling in marine aquaculture: A review. Biofouling 2012, 28, 649–669. [Google Scholar] [CrossRef] [PubMed]
- Glaropoulos, A.; Papadakis, V.M.; Papadakis, I.E.; Kentouri, M. Escape-related behavior and coping ability of sea bream due to food supply. Aquacult. Int. 2012, 20, 965–979. [Google Scholar] [CrossRef]
- Glaropoulos, A.; Papadakis, V.M.; Papadakis, I.E.; Georgara, A.; Kentouri, M. Sea bream interactions toward the aquaculture net due to the presence of micro-fouling. Aquacult. Int. 2014, 22, 1203–1214. [Google Scholar] [CrossRef]
- Nunes, A.L.; Katsanevakis, S.; Zenetos, A.; Cardoso, A.C. Gateways to alien invasions in the European seas. Aquat. Invasions 2014, 9, 133–144. [Google Scholar] [CrossRef]
- Campbell, M.L.; King, S.; Heppenstall, L.D.; van Gool, E.; Martin, R.; Hewitt, C.L. Aquaculture and urban marine structures facilitate native and non-indigenous species transfer through generation and accumulation of marine debris. Mar. Pollut. Bull. 2017, 123, 304–312. [Google Scholar] [CrossRef]
- Simberloff, D.; Martin, J.; Genovesi, P.; Maris, V.; Wardle, D.A.; Aronson, J.; Courchamp, F.; Galil, B.; García-Berthou, E.; Pascal, M.; et al. Impacts of biological invasions: What’s what and the way forward. Trends Ecol. Evol. 2013, 28, 58–66. [Google Scholar] [CrossRef] [Green Version]
- Mack, R.N.; Simberloff, D.; Lonsdale, W.M.; Evans, H.; Clout, M.; Bazzaz, F.A. Biotic invasions: Causes, epidemiology, global consequences, and control. Ecol. Appl. 2000, 10, 689–710. [Google Scholar] [CrossRef]
- Molnar, J.L.; Gamboa, R.L.; Revenga, C.; Spalding, M.D. Assessing the global threat of invasive species to marine biodiversity. Front. Ecol. Environ. 2008, 6, 485–492. [Google Scholar] [CrossRef]
- De Mesel, I.; Kerckhof, F.; Norro, A.; Rumes, B.; Degraer, S. Succession and seasonal dynamics of the epifauna community on offshore wind farm foundations and their role as stepping stones for non-indigenous species. Hydrobiologia 2015, 756, 37–50. [Google Scholar] [CrossRef]
- Rius, M.; Heasman, K.G.; McQuaid, C.D. Long-term coexistence of non-indigenous species in aquaculture facilities. Mar. Pollut. Bull. 2011, 62, 2395–2403. [Google Scholar] [CrossRef]
- Naylor, R.L.; Williams, S.L.; Strong, D.R. Aquaculture—A gateway for exotic species. Science 2001, 294, 1655–1656. [Google Scholar] [CrossRef] [Green Version]
- Mineur, F.; Cook, E.J.; Minchin, D.; Bohn, K.; MacLeod, A.; Maggs, C.A. Oceanography and Marine Biology: An Annual Review; Gibson, R.N., Atkinson, R.J.A., Gordon, J.D.M., Hughes, R.N., Eds.; CRC Press: Abingdon, UK, 2012; Volume 50, pp. 189–234. [Google Scholar]
- Simkanin, C.; Davidson, I.C.; Dower, J.F.; Jamieson, G.; Therriault, T.W. Anthropogenic structures and the infiltration of natural benthos by invasive ascidians. Mar. Ecol. 2012, 33, 499–511. [Google Scholar] [CrossRef]
- Fernandez-Gonzalez, V.; Sanchez-Jerez, P. First occurrence of Caprella scaura Templeton, 1836 (Crustacea: Amphipoda) on off-coast fish farm cages in the Mediterranean Sea. Helgol. Mar. Res. 2014, 68, 187–191. [Google Scholar] [CrossRef] [Green Version]
- Astudillo, J.C.; Bravo, M.; Dumont, C.P.; Thiel, M. Detached aquaculture buoys in the SE Pacific: Potential dispersal vehicles for associated organisms. Aquat. Biol. 2009, 5, 219–231. [Google Scholar] [CrossRef] [Green Version]
- Gansel, L.C.; Bloecher, N.; Floerl, O.; Guenther, J. Quantification of biofouling on nets: A comparison of wet weight measurements and optical (image analysis) methods. Aquacult. Int. 2017, 25, 679–692. [Google Scholar] [CrossRef]
- Ferrario, J.; Caronni, S.; Occhipinti-Ambrogi, A.; Marchini, A. Role of commercial harbours and recreational marinas in the spread of non-indigenous fouling species. Biofouling 2017, 33, 651–660. [Google Scholar] [CrossRef]
- Ulman, A.; Ferrario, J.; Occhipinti-Ambrogi, A.; Arvanitidis, C.; Bandi, A.; Bertolino, M.; Bogi, C.; Chatzigeorgiou, G.; Çiçek, B.A.; Deidun, A.; et al. A massive update of non-indigenous species records in Mediterranean marinas. PeerJ 2017, 5, e3954. [Google Scholar] [CrossRef] [PubMed]
- Tamburini, M.; Keppel, E.; Marchini, A.; Repetto, M.F.; Ruiz, G.M.; Ferrario, J.; Occhipinti-Ambrogi, A. Monitoring non-indigenous species in port habitats: First application of a standardized North American protocol in the Mediterranean Sea. Front. Mar. Sci. 2021, 8, 700730. [Google Scholar] [CrossRef]
- National Exotic Marine and Estuarine Species Information System. Available online: http://invasions.si.edu/nemesis/ (accessed on 27 July 2021).
- Ulman, A.; Ferrario, J.; Forcada, A.; Seebens, H.; Arvanitidis, C.; Occhipinti-Ambrogi, A.; Marchini, A. Alien species spreading via biofouling on recreational vessels in the Mediterranean Sea. J. Appl. Ecol. 2019, 56, 2620–2629. [Google Scholar] [CrossRef]
- Castro, N.; Ramalhosa, P.; Jiménez, J.; Lino, J. Exploring marine invasions connectivity in a NE Atlantic Island through the lens of historical maritime traffic patterns. Reg. Stud. Mar. Sci. 2020, 37, 101333. [Google Scholar] [CrossRef]
- Giangrande, A.; Arduini, D.; Borghese, J.; Del Pasqua, M.; Lezzi, M.; Petrocelli, A.; Cecere, E.; Longo, C.; Pierri, C. Macrobenthic success of non-indigenous species related to substrate features in the Mar Grande of Taranto, Italy (Mediterranean Sea). BioInvasions Rec. 2021, 10, 238–256. [Google Scholar] [CrossRef]
- Canning-Clode, J.; Fofonoff, P.; McCann, L.; Carlton, J.T.; Ruiz, G. Marine invasions on a subtropical island: Fouling studies and new records in a recent marina on Madeira Island (Eastern Atlantic Ocean). Aquat. Invasions 2013, 8, 1–10. [Google Scholar] [CrossRef]
- Ramalhosa, P.; Canning-Clode, J. The invasive caprellid Caprella scaura Templeton, 1836 (Crustacea: Amphipoda: Caprellidae) arrives to Madeira Island, Portugal. BioInvasions Rec. 2015, 4, 97–102. [Google Scholar] [CrossRef]
- Gestoso, I.; Ramalhosa, P.; Canning-Clode, J. Biotic effects during the settlement process of non-indigenous species in marine benthic communities. Aquat. Invasions 2018, 13, 247–259. [Google Scholar] [CrossRef]
- Ramalhosa, P.; Gestoso, I.; Duarte, B.; Caçador, I.; Canning-Clode, J. Metal pollution affects both native and non-indigenous biofouling recruitment in a subtropical island system. Mar. Pollut. Bull. 2019, 141, 373–386. [Google Scholar] [CrossRef]
- Ramalhosa, P.; Gestoso, I.; Rocha, R.; Lambert, G.; Canning-Clode, J. The Ascidian biodiversity of the Madeira Archipelago shallow waters: New records, spatial-temporal distribution, habitat type, and biogeographic notes. Reg. Stud. Mar. Sci. 2021, 43, 101672. [Google Scholar] [CrossRef]
- Marraffini, M.; Ashton, G.; Brown, C.; Chang, A.; Ruiz, G. Settlement plates as monitoring devices for non-indigenous species in marine fouling communities. Manag. Biol. Invasions 2017, 8, 559–566. [Google Scholar] [CrossRef] [Green Version]
- Lezzi, M.; Del Pasqua, M.; Pierri, C.; Giangrande, A. Seasonal non-indigenous species succession in a marine macrofouling invertebrate community. Biol. Invasions 2018, 20, 937–961. [Google Scholar] [CrossRef]
- Jimenez, H.; Keppel, E.; Chang, A.L.; Ruiz, G.M. Invasions in marine communities: Contrasting species richness and community composition across habitats and salinity. Estuar. Coast. 2018, 41, 484–494. [Google Scholar] [CrossRef]
- Canning-Clode, J.; Fofonoff, P.; Riedel, G.F.; Torchin, M.; Ruiz, G.M. The effects of copper pollution on fouling assemblage diversity: A tropical-temperate comparison. PLoS ONE 2011, 6, e18026. [Google Scholar] [CrossRef] [PubMed]
- Ramalhosa, P.; Camacho-Cruz, K.; Bastida-Zavala, R.; Canning-Clode, J. First record of Branchiomma bairdi McIntosh, 1885 (Annelida: Sabellidae) from Madeira Island, Portugal (northeastern Atlantic Ocean). BioInvasions Rec. 2014, 3, 235–239. [Google Scholar] [CrossRef]
- Carlton, J.T. Pattern, process, and prediction in marine invasion ecology. Biol. Conserv. 1996, 78, 97–106. [Google Scholar] [CrossRef]
- Gestoso, I.; Ramalhosa, P.; Oliveira, P.; Canning-Clode, J. Marine protected communities against biological invasions: A case study from an offshore island. Mar. Pollut. Bull. 2017, 119, 72–80. [Google Scholar] [CrossRef]
- Chainho, P.; Fernandes, A.; Amorim, A.; Ávila, S.P.; Canning-Clode, J.; Castro, J.J.; Costa, A.C.; Costa, J.L.; Cruz, T.; Gollasch, S.; et al. Non-indigenous species in Portuguese coastal areas, coastal lagoons, estuaries and islands. Estuar. Coast. Shelf Sci. 2015, 167, 199–211. [Google Scholar] [CrossRef]
- Kohler, K.E.; Gill, S.M. Coral Point Count with Excel extensions (CPCe): A Visual Basic programme for the determination of coral and substrate coverage using random point count methodology. Comput. Geosci. 2006, 32, 1259–1269. [Google Scholar] [CrossRef]
- Airoldi, L.; Turon, X.; Perkol-Finkel, S.; Rius, M. Corridors for aliens but not for natives: Effects of marine urban sprawl at a regional scale. Divers. Distrib. 2015, 21, 755–768. [Google Scholar] [CrossRef] [Green Version]
- Underwood, A.J. Experiments in Ecology: Their Logical Design and Interpretation Using Analysis of Variance; Cambridge University Press: Cambridge, UK, 1997. [Google Scholar]
- Clarke, K.R. Non-parametric multivariate analyses of changes in community. Aust. J. Ecol. 1993, 18, 117–143. [Google Scholar] [CrossRef]
- Clarke, K.R.; Gorley, R.N. PRIMER v6: User manual—Tutorial; Plymouth Marine Laboratory: Plymouth, UK, 2006. [Google Scholar]
- Anderson, M.J. A new method for non-parametric multivariate analysis of variance. Austral Ecol. 2008, 26, 32–46. [Google Scholar] [CrossRef]
- Núñez, J.C.; Talavera, J.A. Fauna of the polychaetous annelids from Madeira. Bol. Mus. Munic. Funchal 1995, 4, 511–530. [Google Scholar]
- Langerhans, P. Die Wurmfauna von Madeira. III. Z. Wiss. Zool. 1880, 34, 87–143. [Google Scholar]
- Norman, A.M. The Polyzoa of Madeira and neighbouring islands. Zool. J. Linnean Soc. 1909, 30, 275–314. [Google Scholar] [CrossRef]
- Wirtz, P.; Canning-Clode, J. The invasive bryozoan Zoobotryon verticillatum has arrived at Madeira Island. Aquat. Invasions 2009, 4, 669–670. [Google Scholar] [CrossRef]
- Ramalhosa, P.; Souto, J.; Canning-Clode, J. Diversity of Bugulidae (Bryozoa, Cheilostomata) colonising artificial substrates in the Madeira Archipelago (NE Atlantic Ocean). Helgol. Mar. Res. 2017, 71, 1. [Google Scholar] [CrossRef] [Green Version]
- Souto, J.; Ramalhosa, P.; Canning-Clode, J. Three non-indigenous species from Madeira harbours, including a new species of Parasmittina (Bryozoa). Mar. Biodivers. 2018, 48, 977–986. [Google Scholar] [CrossRef] [Green Version]
- Souto, J.; Kaufmann, M.J.; Canning-Clode, J. New species and new records of bryozoans from shallow waters of Madeira Island. Zootaxa 2015, 3925, 581–593. [Google Scholar] [CrossRef] [PubMed]
- Wirtz, P. On a collection of hydroids (Cnidaria, Hydrozoa) from the Madeira archipelago. Arquipelago 2007, 24, 11–16. [Google Scholar]
- Wirtz, P. Unterwasserführer Madeira, Kanaren, Azoren—Niedere Tiere; Edition Naglschmid: Stuttgart, Germany, 1995. [Google Scholar]
- Vervoort, W. Leptolida (Cnidaria: Hydrozoa) collected during the CANCAP and Mauritania-II expeditions of the National Museum of Natural History, Leiden, The Netherlands [Anthoathecata, various families of Leptothecata and addenda]. Zool. Med. Leiden 2006, 80, 181–318. [Google Scholar]
- Bianchi, C.N.; Morri, C.; Sartoni, G.; Wirtz, P. Sublittoral epibenthic communities around Funchal (Ilha da Madeira, NE Atlantic). Bol. Mus. Munic. Funchal 1998, 5, 59–80. [Google Scholar]
- Wirtz, P. Ten invertebrates new for the marine fauna of Madeira. Arquipelago 2006, 23A, 75–78. [Google Scholar]
- Ramalhosa, P.; Nebra, A.; Gestoso, I.; Canning-Clode, J. First record of the non-indigenous isopods Paracerceis sculpta (Holmes, 1904) and Sphaeroma walkeri Stebbing, 1905 (Isopoda, Sphaeromatidae) for Madeira Island. Crustaceana 2017, 90, 1747–1764. [Google Scholar] [CrossRef]
- Levring, T. The marine algae of the Archipelago of Madeira. Bol. Mus. Munic. Funchal 1974, 28, 5–111. [Google Scholar]
- Neto, A.I.; Cravo, D.C.; Haroun, R.T. Checklist of the Benthic Marine Plants of the Madeira Archipelago. Bot. Mar. 2001, 44, 391–414. [Google Scholar] [CrossRef] [Green Version]
- Cruz, T. Esponjas Marinas de Canarias; Gobierno de Canarias: Santa Cruz Tenerife, Spain, 2002. [Google Scholar]
- Madin, J.; Chong, V.C.; Hartstein, N.D. Effects of water flow velocity and fish culture on net biofouling in fish cages. Aquac. Res. 2010, 41, e602–e617. [Google Scholar] [CrossRef]
- Longo, C.; Mastrototaro, F.; Corriero, G. Occurrence of Paraleucilla magna (Porifera: Calcarea) in the Mediterranean Sea. J. Mar. Biol. Assoc. 2007, 87, 1749–1755. [Google Scholar] [CrossRef] [Green Version]
- Bachetarzi, R.; Dilmi, S.; Uriz, M.J.; Vázquez-Luis, M.; Deudero, S.; Rebzani-Zahaf, C. The non-indigenous and invasive species Paraleucilla magna Klautau, Monteiro & Borojevic, 2004 (Porifera: Calcarea) in the Algerian coast (Southwestern of Mediterranean Sea). Acta Adriat. 2019, 60, 41–46. [Google Scholar] [CrossRef]
- Fitridge, I.; Keough, M.J. Ruinous resident: The hydroid Ectopleura crocea negatively affects suspended culture of the mussel Mytilus galloprovincialis. Biofouling 2013, 29, 119–131. [Google Scholar] [CrossRef] [PubMed]
- Bosch-Belmar, M.; Azzurro, E.; Pulis, K.; Milisenda, G.; Fuentes, V.; Kéfi-Daly Yahia, O.; Micallef, A.; Deidun, A.; Piraino, S. Jellyfish blooms perception in Mediterranean finfish aquaculture. Mar. Policy 2017, 76, 1–7. [Google Scholar] [CrossRef]
- Bosch-Belmar, M.; Escurriola, A.; Milisenda, G.; Fuentes, V.L.; Piraino, S. Harmful Fouling Communities on Fish Farms in the SW Mediterranean Sea: Composition, Growth and Reproductive Periods. J. Mar. Sci. Eng. 2019, 7, 288. [Google Scholar] [CrossRef] [Green Version]
- Schuchert, P. The European athecate hydroids and their medusae (Hydrozoa, Cnidaria): Filifera Part 4. Rev. Suisse De Zool. 2008, 115, 677–757. [Google Scholar] [CrossRef]
- World Register of Marine Species. Available online: http://www.marinespecies.org/ (accessed on 19 September 2021).
- European Alien Species Information Network. Available online: https://easin.jrc.ec.europa.eu/ (accessed on 19 September 2021).
- Mangano, M.C.; Ape, F.; Mirto, S. The role of two non-indigenous serpulid tube worms in shaping artificial hard substrata communities: Case study of a fish farm in the central Mediterranean Sea. Aquacult. Environ. Interact. 2019, 11, 41–51. [Google Scholar] [CrossRef] [Green Version]
- Rumbold, C.E.; Ruiz Barlett, T.; Gavio, M.A.; Obenat, S.M. Population dynamics of two invasive amphipods in the Southwestern Atlantic: Monocorophium acherusicum and Ericthonius punctatus (Crustacea). Mar. Biol. Res. 2016, 12, 268–277. [Google Scholar] [CrossRef]
- Png-Gonzalez, L.; Vázquez-Luis, M.; Tuya, F. Comparison of epifaunal assemblages between Cymodocea nodosa and Caulerpa prolifera meadows in Gran Canaria (eastern Atlantic). J. Mar. Biolog. Assoc. UK 2014, 94, 241–253. [Google Scholar] [CrossRef] [Green Version]
- Bamber, R.G.; Robbins, R. The soft-sediment infauna off São Miguel, Azores, and a comparison with other Azorean invertebrate habitats. Açoreana 2009, 6, 201–210. [Google Scholar]
- Ros, M.; Navarro-Barranco, C.; González-Sánchez, M.; Ostalé-Valriberas, E.; Cervera-Currado, L.; Guerra-García, J.M. Starting the stowaway pathway: The role of dispersal behavior in the invasion success of low-mobile marine species. Biol. Invasions 2020, 22, 2797–2812. [Google Scholar] [CrossRef]
- Cronin, E.R.; Cheshire, A.C.; Clarke, S.M.; Melville, A.J. An investigation into the composition, biomass and oxygen budget of the fouling community on tuna aquaculture farm. Biofouling 1999, 13, 279–299. [Google Scholar] [CrossRef]
- Howes, S.; Herbinger, C.M.; Darnell, P.; Vercaemer, B. Spatial and temporal patterns of recruitment of the tunicate Ciona intestinalis on a mussel farm in Nova Scotia, Canada. J. Exp. Mar. Biol. Ecol. 2007, 342, 85–92. [Google Scholar] [CrossRef]
- Guenther, J.; Misimi, E.; Sunde, L.M. The development of biofouling, particularly the hydroid Ectopleura larynx, on commercial salmon cage nets in Mid-Norway. Aquaculture 2010, 300, 120–127. [Google Scholar] [CrossRef]
- Andrade, C.A.P. A fish farm pilot-project in Madeira Archipelago, Northeastern Atlantic-II. Environment impact assessment. In Proceedings of the International Conference: Open Ocean Aquaculture, Portland, OR, USA, 8–10 May 1996; Polk, M., Ed.; pp. 377–382. [Google Scholar]
- Galil, B.S. Alien species in the Mediterranean Sea—Which, when, where, why? Hydrobiologia 2008, 606, 105–116. [Google Scholar] [CrossRef]
- Alharbi, W.; Petrovskii, S. Effect of complex landscape geometry on the invasive species spread: Invasion with stepping stones. J. Theor. Biol. 2019, 464, 85–97. [Google Scholar] [CrossRef] [Green Version]
- Lezzi, M.; Giangrande, A. Seasonal and bathymetric effects on macrofouling invertebrates’ primary succession in a Mediterranean non-indigenous species hotspot area. Mediterr. Mar. Sci. 2018, 19, 572–588. [Google Scholar] [CrossRef] [Green Version]
- Pica, D.; Bloecher, N.; Dell’Anno, A.; Bellucci, A.; Pinto, T.; Pola, L.; Puce, S. Dynamics of a biofouling community in finfish aquaculture: A case study from the South Adriatic Sea. Biofouling 2019, 35, 696–709. [Google Scholar] [CrossRef]
| Taxa | Status | Sources | Sea-Cages | Marinas | ||
|---|---|---|---|---|---|---|
| SW | SE | SW | SE | |||
| Annelida (4) | ||||||
| Protula tubularia (Montagu, 1803) | C | [36,52] | ○ | ● | ● | ○ |
| Salmacina dysteri (Huxley, 1855) | N | [33,52,53] | ● | ● | ● | ● |
| Spirobranchus triqueter (Linnaeus, 1758) | N | [36,44] | ● | ● | ● | ● |
| Spirorbis sp. | U | ○ | ● | ● | ● | |
| Bryozoa (18) | ||||||
| Aetea sica (Couch, 1844) | C | [54] | ○ | ● | ○ | ○ |
| Amathia verticillata (delle Chiaje, 1822) | NIS | [36,46,55] | ○ | ○ | ● | ○ |
| Bugula sp. Oken, 1815 | U | ● | ○ | ○ | ○ | |
| Bugula neritina (Linnaeus, 1758) | NIS | [36,54,56] | ○ | ○ | ● | ● |
| Bugulina simplex (Hincks, 1886) | NIS | [36,56] | ○ | ○ | ● | ● |
| Celleporaria inaudita Tilbrook, Hayward and Gordon, 2001 | NIS | [33,36,57] | ○ | ○ | ○ | ● |
| Cradoscrupocellaria bertholletii (Audouin, 1826) | NIS | [33,36,46] | ○ | ● | ● | ● |
| Crisia sp. Lamouroux, 1812 | U | ● | ● | ● | ● | |
| Fenestrulina sp. Jullien, 1888 | U | ○ | ○ | ○ | ● | |
| Nolella gigantea (Busk, 1856) | C | [36] | ○ | ● | ○ | ○ |
| Parasmittina alba Ramalho, Muricy and Taylor, 2011 | NIS | [36,57] | ○ | ○ | ● | ● |
| Schizoporella errata (Waters, 1878) | C | [36] | ○ | ● | ● | ● |
| Scruparia sp. Oken, 1815 | U | ○ | ● | ○ | ○ | |
| Scrupocaberea maderensis (Busk, 1860) | N | [54,58] | ○ | ○ | ○ | ● |
| Scrupocellaria sp. Van Beneden, 1845 | U | ● | ○ | ○ | ○ | |
| Smittoidea sp. Osburn, 1952 | U | ○ | ● | ○ | ● | |
| Tricellaria sp. Fleming, 1828 | U | ○ | ● | ○ | ○ | |
| Watersipora subtorquata (d’Orbigny, 1852) | NIS | [33,36,46] | ○ | ● | ● | ● |
| Chordata (10) | ||||||
| Aplidium glabrum (Verrill, 1871) | NIS | [36,37] | ○ | ○ | ● | ○ |
| Botrylloides niger Herdman, 1886 | NIS | [36,37] | ○ | ○ | ● | ● |
| Botryllus schlosseri (Pallas, 1766) | NIS | [33,36,37] | ○ | ● | ● | ○ |
| Diplosoma listerianum (Milne Edwards, 1841) | C | [36,37,44] | ○ | ● | ○ | ● |
| Distaplia corolla Monniot F., 1974 | NIS | [33,36,37,44] | ○ | ○ | ● | ● |
| Ecteinascidia sp. Herdman, 1880 | U | [37] | ○ | ○ | ○ | ● |
| Perophora listeri Wiegman, 1835 | C | [36,37] | ○ | ○ | ● | ● |
| Symplegma rubra Monniot C., 1972 | C | [36,37] | ○ | ○ | ● | ○ |
| Symplegma brakenhielmi (Michaelsen, 1904) | C | [37,44] | ○ | ○ | ○ | ● |
| Trididemnum cereum (Giard, 1872) | C | [33,36,37] | ○ | ○ | ● | ● |
| Cnidaria (8) | ||||||
| Aiptasia diaphana (Rapp, 1829) | NIS | [33,37] | ○ | ● | ○ | ● |
| Clytia hemisphaerica (Linnaeus, 1767) | C | [59] | ○ | ● | ○ | ○ |
| Ectopleura crocea (Agassiz, 1862) | NIS | [46,59,60] | ● | ● | ○ | ○ |
| Eudendrium capillare Alder, 1856 | C | This study | ● | ○ | ○ | ○ |
| Kirchenpaueria halecioides (Alder, 1859) | C | [36,59] | ● | ● | ● | ● |
| Obelia dichotoma (Linnaeus, 1758) | C | [36,59] | ● | ● | ○ | ○ |
| Pennaria disticha Goldfuss, 1820 | C | [36,59] | ● | ● | ○ | ○ |
| Sertularella ellisii (Deshayes and Milne Edwards, 1836) | C | [59,61] | ○ | ● | ● | ○ |
| Crustacea (5) | ||||||
| Caprella scaura Templeton, 1836 | NIS | [34,36] | ○ | ○ | ○ | ● |
| Chthamalus stellatus (Poli, 1791) | N | [62] | ● | ○ | ○ | ○ |
| Ericthonius punctatus (Spence Bate, 1857) | C | This study | ● | ● | ● | ● |
| Megabalanus azoricus (Pilsbry, 1916) | N | [63] | ● | ● | ○ | ○ |
| Paracerceis sculpta (Holmes, 1904) | NIS | [64] | ○ | ○ | ○ | ● |
| Macroalgae (4) | ||||||
| Bryopsis sp. J.V. Lamouroux, 1809 | U | ○ | ● | ○ | ○ | |
| Champiaceae sp. Kützing, 1843 | U | ○ | ● | ○ | ○ | |
| Lithophyllum incrustans Philippi, 1837 | N | [33,36,65] | ● | ● | ● | ● |
| Neosiphonia sertularioides (Grateloup) K.W. Nam and P.J. Kang, 2012 | N | [33,36,66] | ○ | ● | ○ | ● |
| Porifera (5) | ||||||
| Clathrina clathrus (Schmidt, 1864) | N | [36] | ● | ● | ○ | ○ |
| Mycale (Carmia) senegalensis Lévi, 1952 | NIS | [33,36,44] | ● | ● | ● | ● |
| Paraleucilla magna Klautau, Monteiro and Borojevic, 2004 | NIS | [33,36,44] | ● | ● | ○ | ● |
| Prosuberites longispinus Topsent, 1893 | NIS | [36] | ○ | ○ | ● | ○ |
| Sycon ciliatum (Fabricius, 1780) | N | [33,36,67] | ● | ● | ● | ● |
| Total Species Richness | 54 | Total species per region | 18 | 32 | 26 | 31 |
| Percentage (%) | 33.3 | 59.3 | 48.1 | 57.4 | ||
| Source | df | Full Community | |||||||
| Composition | Cover (%) | Species Richness | |||||||
| MS | Pseudo-F | MS | Pseudo-F | MS | Pseudo-F | ||||
| Region (Reg) | 1 | 12380 | 11.45 *** | 58.16 | 1.05 | 100.04 | 20.313 *** | ||
| Environment (Env) | 1 | 16861 | 2.23 | 214.32 | 0.32 | 5.04 | 0.72 | ||
| Reg × Env | 1 | 7568.3 | 7.00 *** | 664.23 | 12.04 ** | 7.04 | 1.43 | ||
| Residual | 20 | 1081.2 | 55.17 | 4.93 | |||||
| Total | 23 | ||||||||
| Source | df | Native Species | NIS | ||||||
| Cover (%) | Species Richness | Cover (%) | Species Richness | ||||||
| MS | Pseudo-F | MS | Pseudo-F | MS | Pseudo-F | MS | Pseudo-F | ||
| Region (Reg) | 1 | 138.1 | 0.90 | 30.38 | 16.2 *** | 383.52 | 2.16 | 20.17 | 10.86 ** |
| Environment (Env) | 1 | 3075.6 | 19.96 *** | 35.04 | 4.98 | 4942.7 | 27.86 *** | 66.67 | 35.90 *** |
| Reg × Env | 1 | 154.09 | pooled | 7.04 | 3.76 | 177.38 | pooled | 1.86 | pooled |
| Residual | 20 | 1.88 | |||||||
| Total | 23 | ||||||||
| (a) | Area | Taxon | Sea-Cages | Marina | |||||||
| Av.Abund | Av.Abund | Av.Diss | Diss/SD | Contrib% | Cum% | ||||||
| SW | Scrupocellaria sp. | 20.03 | 0.00 | 11.40 | 5.90 | 12.80 | 12.80 | ||||
| Crisia sp. | 19.36 | 1.01 | 10.29 | 2.56 | 11.55 | 24.35 | |||||
| Botrylloides niger | 0.00 | 17.34 | 9.76 | 0.77 | 10.96 | 35.31 | |||||
| Symplegma rubra | 0.00 | 11.28 | 6.69 | 0.68 | 7.50 | 42.81 | |||||
| Eudendrium sp. | 11.78 | 0.00 | 6.66 | 3.07 | 7.48 | 50.29 | |||||
| Schizoporella errata | 0.00 | 10.27 | 5.85 | 1.23 | 6.57 | 56.86 | |||||
| Spirorbis sp. | 0.00 | 9.76 | 5.64 | 1.49 | 6.33 | 63.19 | |||||
| Salmacina dysteri | 0.34 | 8.59 | 4.79 | 1.82 | 5.38 | 68.57 | |||||
| Prosuberiteslongispinus | 0.00 | 7.24 | 4.06 | 0.73 | 4.55 | 73.12 | |||||
| Parasmittina alba | 0.00 | 5.56 | 3.19 | 1.00 | 3.58 | 76.70 | |||||
| Lithophyllum incrustans | 8.42 | 5.72 | 2.91 | 1.34 | 3.26 | 79.96 | |||||
| Ectopleura crocea | 3.70 | 0.00 | 2.07 | 1.44 | 2.32 | 82.28 | |||||
| Ericthonius punctatus | 1.85 | 4.71 | 1.84 | 1.28 | 2.07 | 84.35 | |||||
| SE | Ericthonius punctatus | 24.91 | 3.87 | 11.59 | 3.13 | 16.93 | 16.93 | ||||
| Parasmittina alba | 0.00 | 12.46 | 6.87 | 0.98 | 10.03 | 26.97 | |||||
| Botrylloides niger | 0.00 | 10.77 | 5.89 | 1.58 | 8.61 | 35.58 | |||||
| Spirorbis sp. | 0.17 | 9.60 | 5.22 | 1.63 | 7.62 | 43.20 | |||||
| Crisia sp. | 12.29 | 4.88 | 4.53 | 1.79 | 6.62 | 49.82 | |||||
| Cradoscrupocellaria bertholletii | 10.61 | 11.62 | 4.21 | 1.50 | 6.15 | 55.97 | |||||
| Scruparia sp. | 6.73 | 0.00 | 3.74 | 1.55 | 5.46 | 61.43 | |||||
| Spirobranchus triqueter | 10.27 | 11.45 | 3.25 | 1.39 | 4.74 | 66.17 | |||||
| Diplosoma listerianum | 4.71 | 3.87 | 2.87 | 1.23 | 4.20 | 70.37 | |||||
| Kirchenpaueria halecioides | 4.21 | 0.17 | 2.23 | 2.00 | 3.26 | 73.62 | |||||
| Paraleucilla magna | 3.87 | 0.51 | 1.87 | 1.65 | 2.73 | 76.35 | |||||
| Perophora listeri | 0.00 | 3.03 | 1.66 | 0.88 | 2.42 | 78.77 | |||||
| (b) | Area | Taxon | Sea-Cages | Taxon | Marinas | ||||||
| Av.Abund | Sim/SD | Contrib% | Cum% | Av.Abund | Sim/SD | Contrib% | Cum% | ||||
| SW | Scrupocellaria sp. | 20.03 | 6.56 | 31.93 | 31.93 | Salmacina dysteri | 8.59 | 1.44 | 15.42 | 15.42 | |
| Crisia sp. | 19.36 | 3.61 | 25.42 | 57.34 | Spirorbis sp. | 9.76 | 1.07 | 15.05 | 30.46 | ||
| Eudendrium sp. | 11.78 | 2.92 | 16.38 | 73.72 | Schizoporella errata | 10.27 | 0.91 | 12.93 | 43.39 | ||
| Lithophyllum incrustans | 8.42 | 1.37 | 10.34 | 84.06 | Botrylloides niger | 17.34 | 0.36 | 10.23 | 53.62 | ||
| Ectopleura crocea | 3.7 | 0.96 | 3.5 | 87.56 | Lithophyllum incrustans | 5.72 | 2.11 | 10.14 | 63.76 | ||
| Paraleucilla magna | 2.69 | 1.11 | 2.77 | 90.33 | Ericthonius punctatus | 4.71 | 1.38 | 7.91 | 71.67 | ||
| Ericthonius punctatus | 1.85 | 3.15 | 2.56 | 92.89 | Parasmittina alba | 5.56 | 0.78 | 5.88 | 77.55 | ||
| Sycon ciliatum | 1.85 | 3.54 | 2.19 | 95.08 | Symplegma rubra | 11.28 | 0.26 | 5.48 | 83.02 | ||
| Spirobranchus triqueter | 2.69 | 2.41 | 4.02 | 87.05 | |||||||
| Prosuberites longispinus | 7.24 | 0.36 | 3.71 | 90.76 | |||||||
| Mycale (Carmia) senegalensis | 2.02 | 0.65 | 2.36 | 93.12 | |||||||
| SE | Ericthonius punctatus | 24.91 | 2.98 | 34.99 | 34.99 | Cradoscrupocellaria bertholletii | 11.62 | 1.4 | 17.04 | 17.04 | |
| Crisia sp. | 12.29 | 1.97 | 15.19 | 50.18 | Spirobranchus triqueter | 11.45 | 1.88 | 16.8 | 33.84 | ||
| Spirobranchus triqueter | 10.27 | 3.06 | 12.65 | 62.83 | Botrylloides niger | 10.77 | 1 | 14.38 | 48.21 | ||
| Cradoscrupocellaria bertholletii | 10.61 | 1.54 | 9.92 | 72.75 | Spirorbis sp. | 9.6 | 1.89 | 13.59 | 61.81 | ||
| Scruparia sp. | 6.73 | 1.11 | 6.39 | 79.15 | Parasmittina alba | 12.46 | 0.68 | 10.04 | 71.85 | ||
| Kirchenpaueria halecioides | 4.21 | 1.83 | 4.72 | 83.87 | Crisia sp. | 4.88 | 1.67 | 6.08 | 77.93 | ||
| Paraleucilla magna | 3.87 | 1.93 | 4.12 | 87.99 | Ericthonius punctatus | 3.87 | 2.08 | 4.96 | 82.89 | ||
| Diplosoma listerianum | 3.87 | 0.63 | 3.33 | 86.22 | |||||||
| Salmacina dysteri | 1.18 | 37.18 | 2.3 | 88.52 | |||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Png-Gonzalez, L.; Ramalhosa, P.; Gestoso, I.; Álvarez, S.; Nogueira, N. Non-Indigenous Species on Artificial Coastal Environments: Experimental Comparison between Aquaculture Farms and Recreational Marinas. J. Mar. Sci. Eng. 2021, 9, 1121. https://doi.org/10.3390/jmse9101121
Png-Gonzalez L, Ramalhosa P, Gestoso I, Álvarez S, Nogueira N. Non-Indigenous Species on Artificial Coastal Environments: Experimental Comparison between Aquaculture Farms and Recreational Marinas. Journal of Marine Science and Engineering. 2021; 9(10):1121. https://doi.org/10.3390/jmse9101121
Chicago/Turabian StylePng-Gonzalez, Lydia, Patrício Ramalhosa, Ignacio Gestoso, Soledad Álvarez, and Natacha Nogueira. 2021. "Non-Indigenous Species on Artificial Coastal Environments: Experimental Comparison between Aquaculture Farms and Recreational Marinas" Journal of Marine Science and Engineering 9, no. 10: 1121. https://doi.org/10.3390/jmse9101121
APA StylePng-Gonzalez, L., Ramalhosa, P., Gestoso, I., Álvarez, S., & Nogueira, N. (2021). Non-Indigenous Species on Artificial Coastal Environments: Experimental Comparison between Aquaculture Farms and Recreational Marinas. Journal of Marine Science and Engineering, 9(10), 1121. https://doi.org/10.3390/jmse9101121






