Coral Reef Resilience in Taiwan: Lessons from Long-Term Ecological Research on the Coral Reefs of Kenting National Park (Taiwan)
Abstract
1. Introduction
1.1. Coral Reef Ecosystems and the Impacts of Environmental Change
1.2. Coral Reef Resilience under the Impacts of Environmental Change
1.3. Coral-Symbiodiniaceae Associations Play a Key Role in Coral Resilience to Thermal Stress
1.4. Coral Reefs in Taiwan, with a Focus on Kenting Coral Reefs
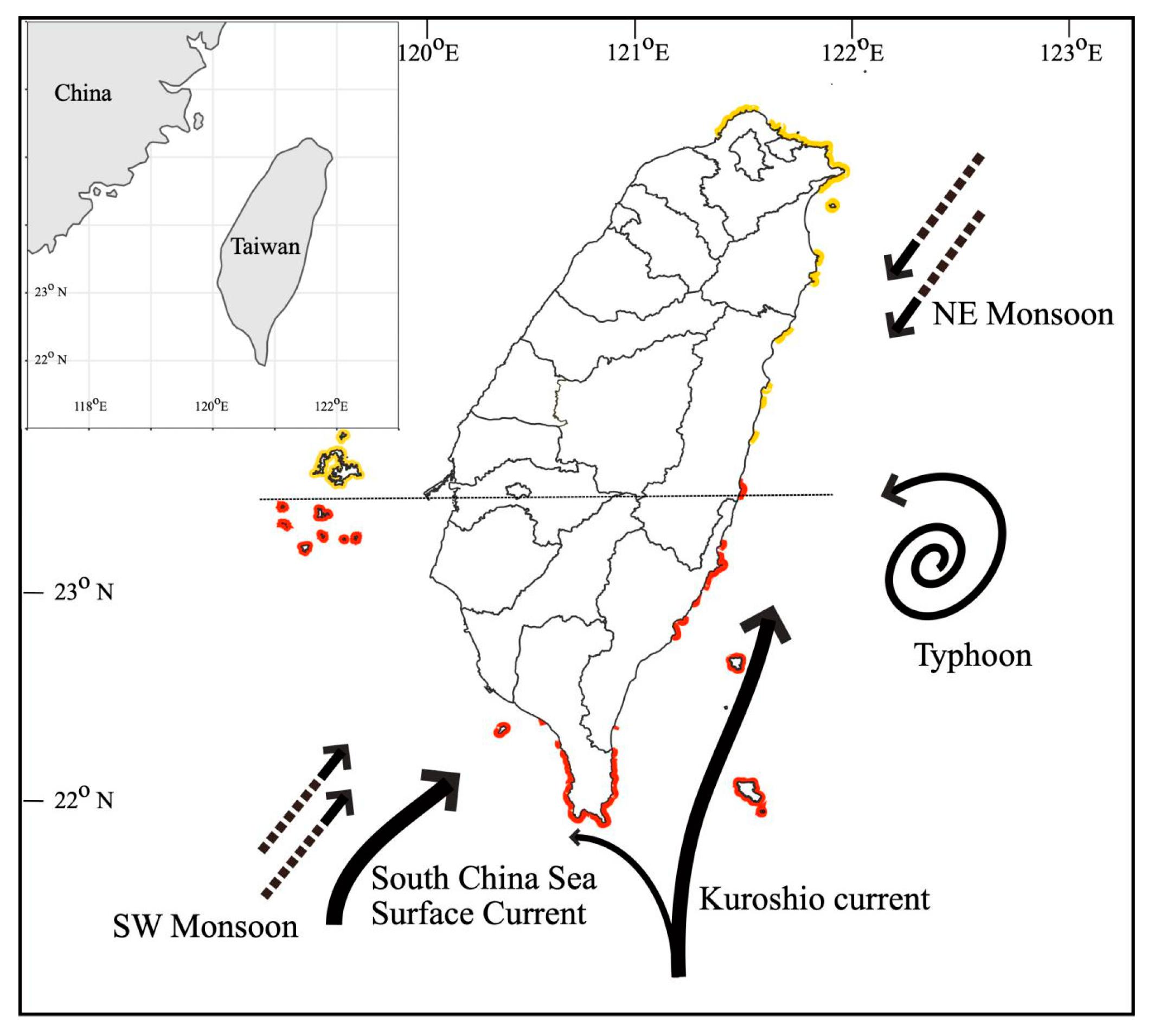
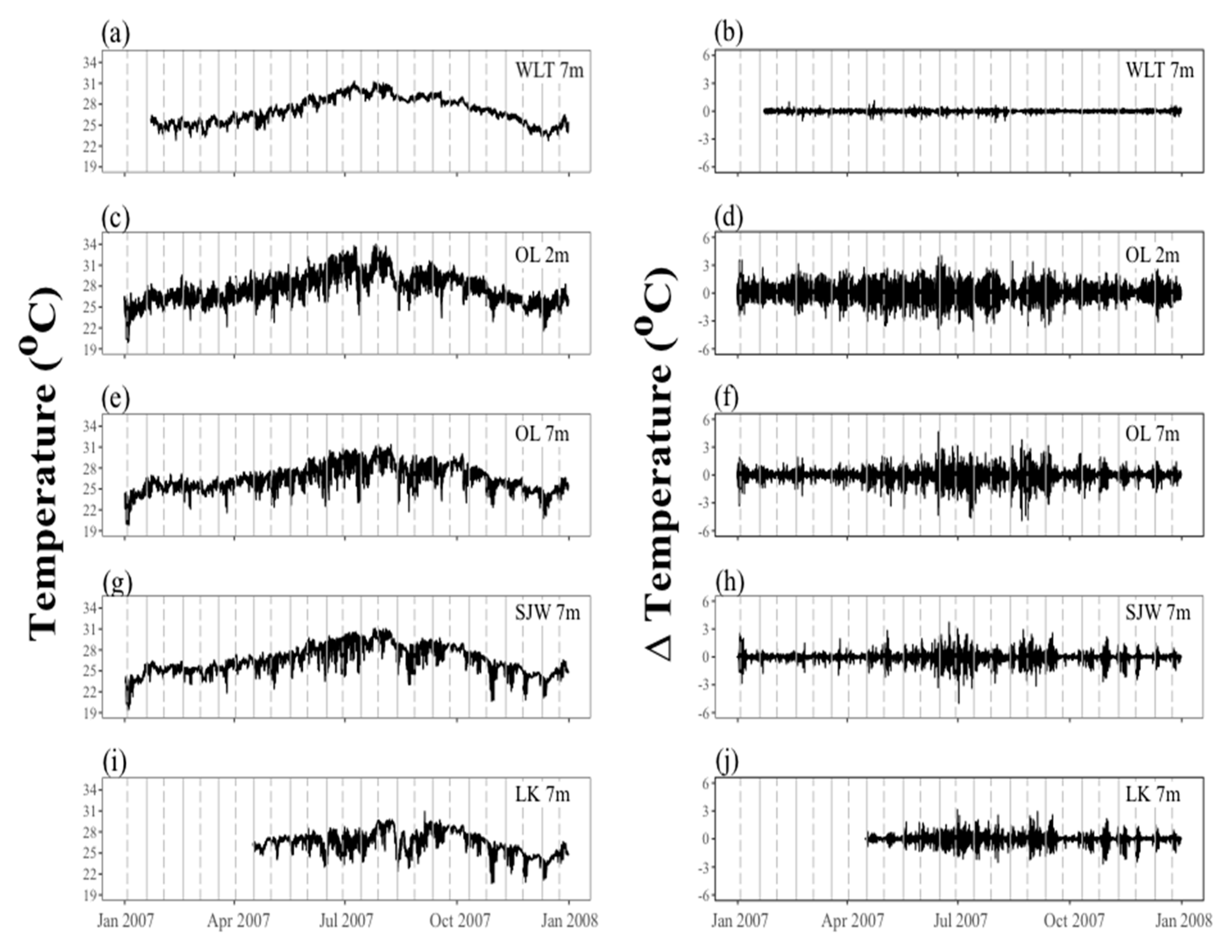
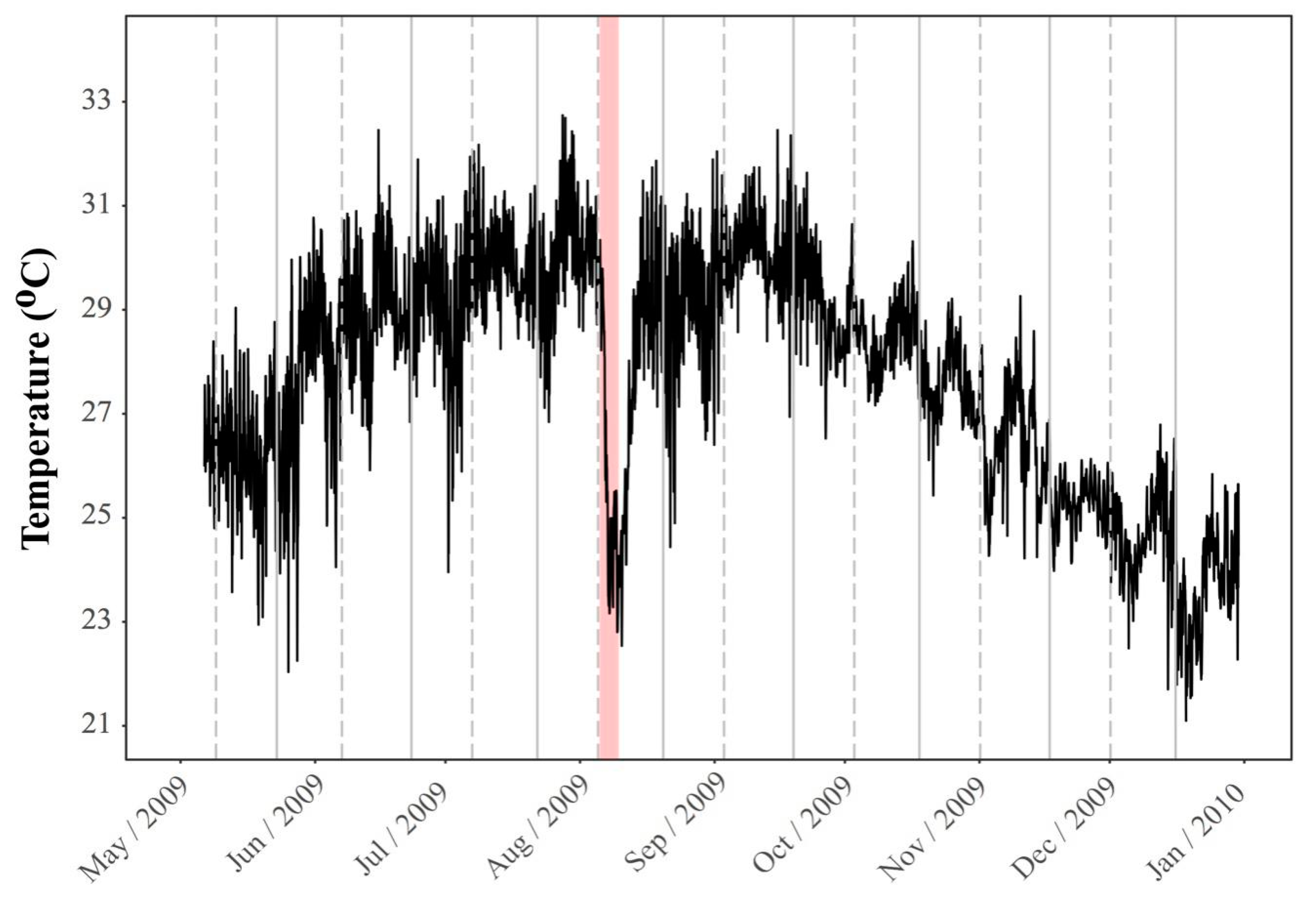
2. Environmental Settings of Coral Reef in KNP with Focus on Tide-Induced Upwelling
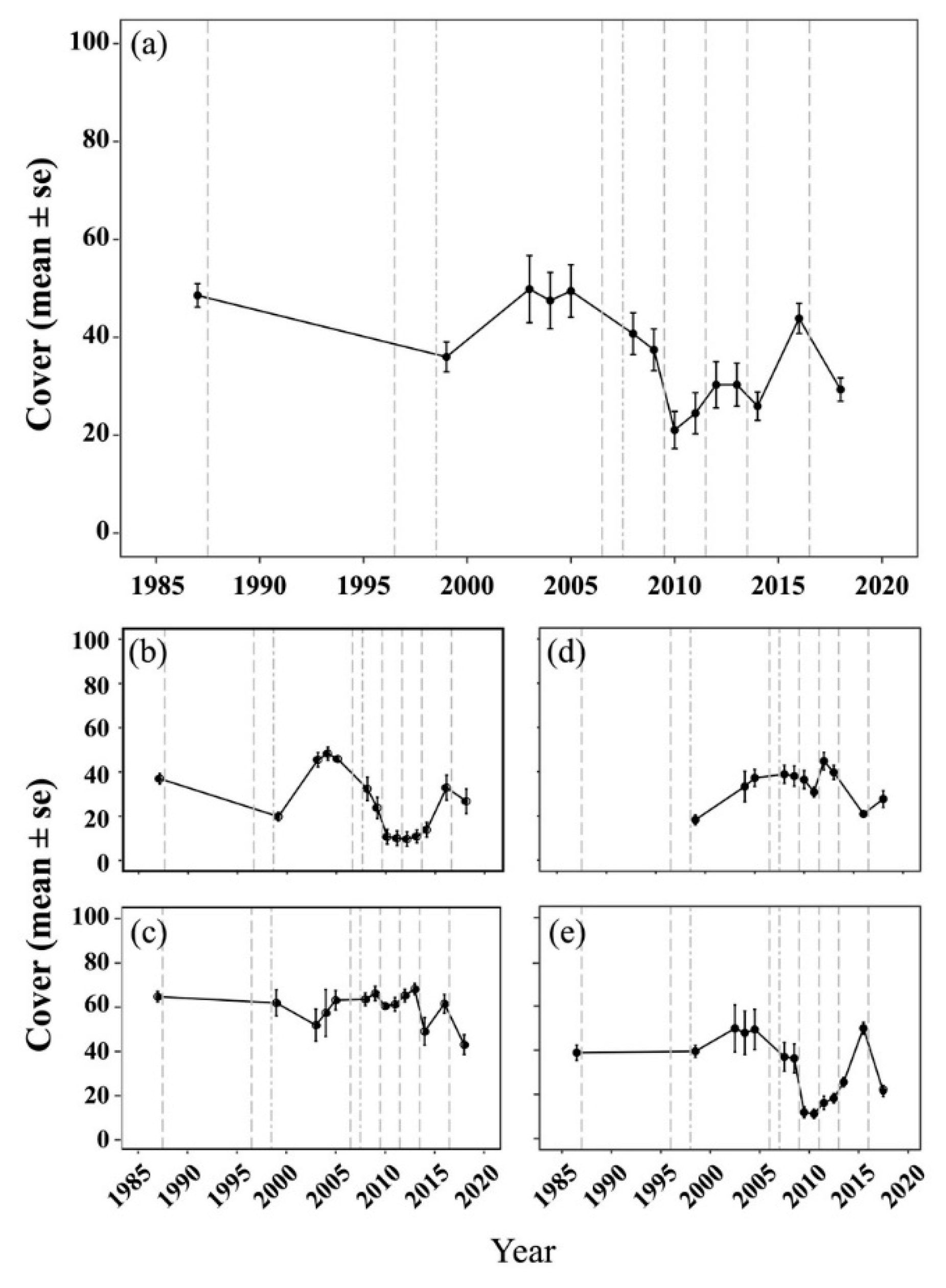
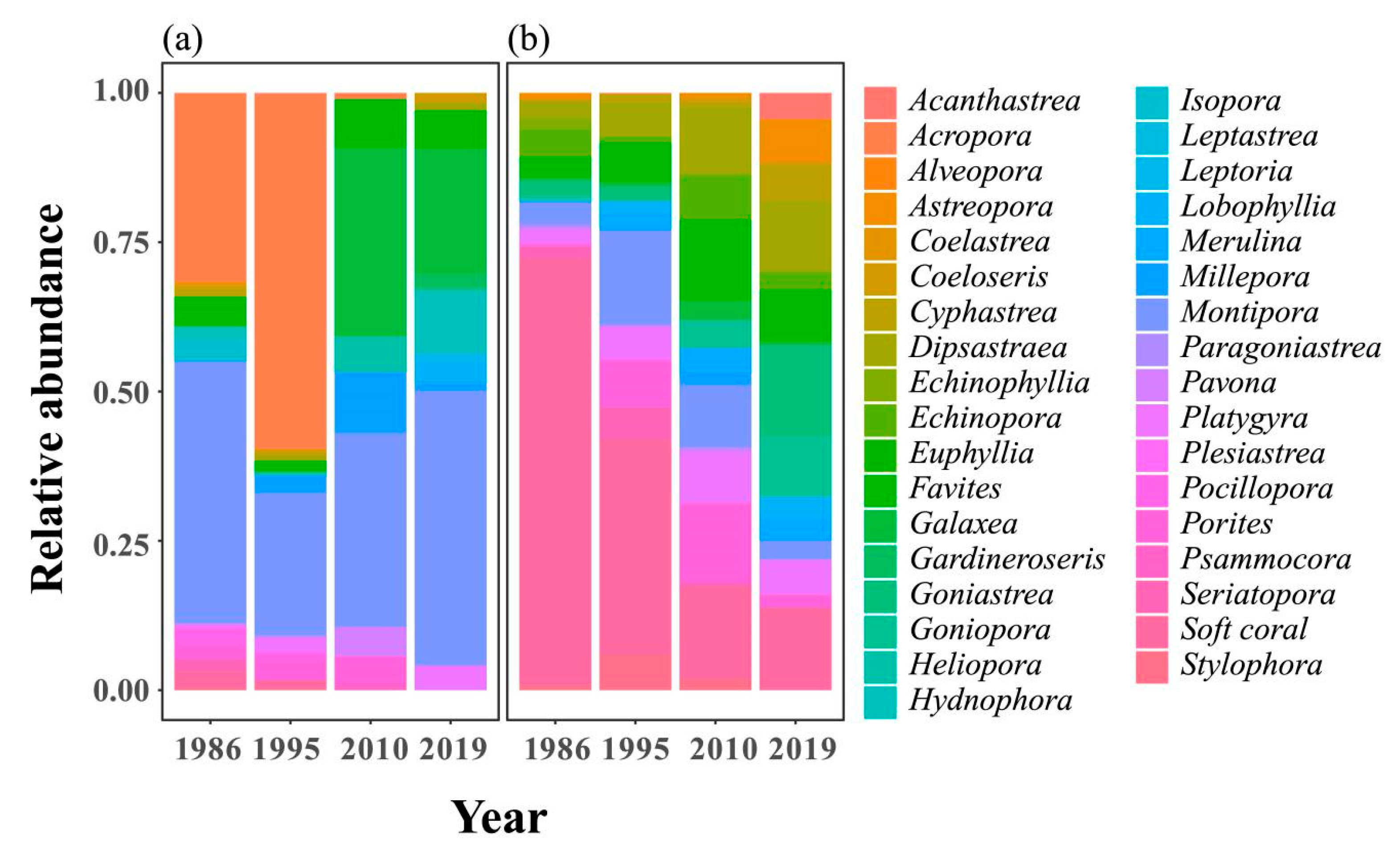
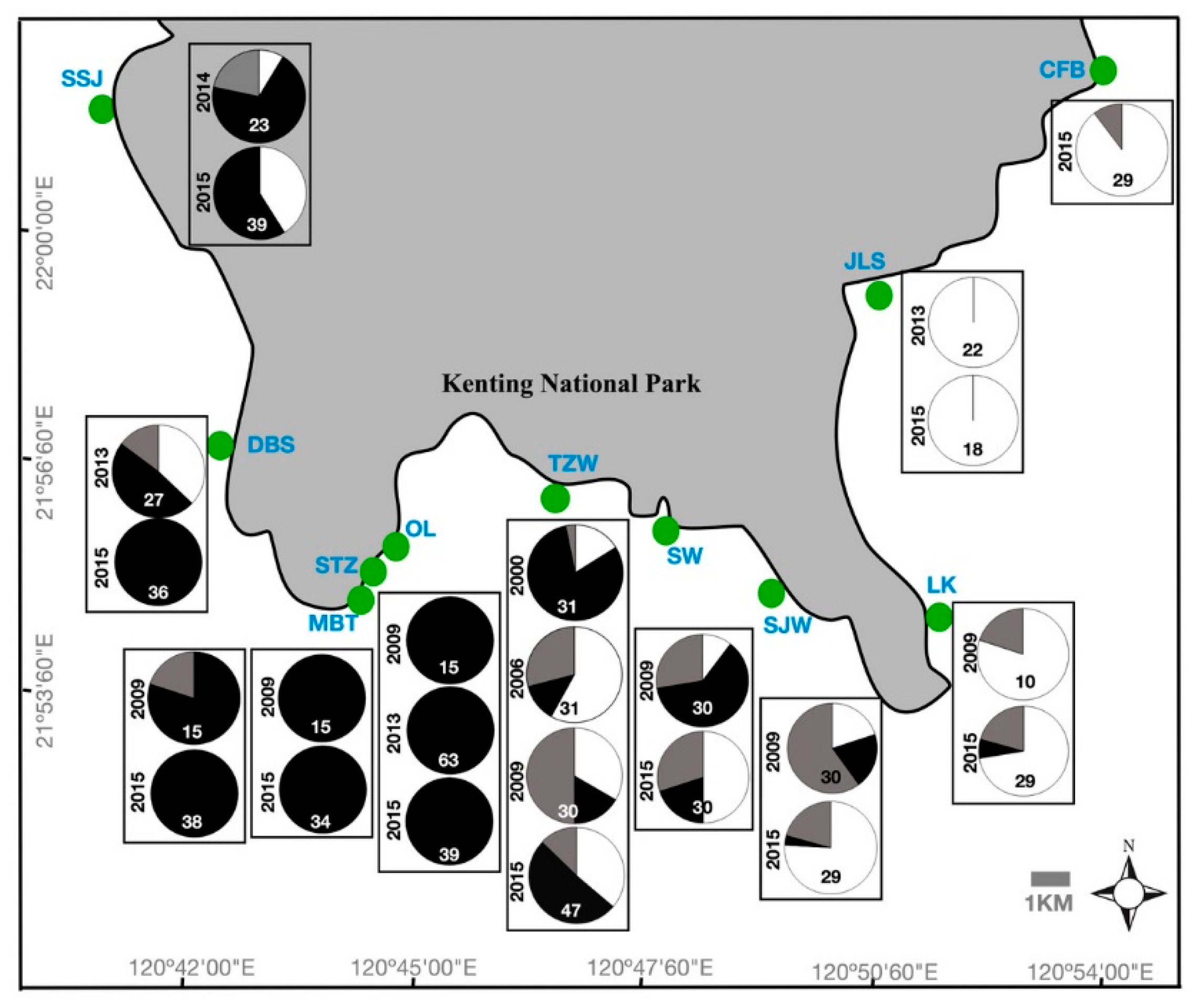
2.1. Spatial and Temporal Variability of Coral Communities in Responding to Large-Scale Disturbances
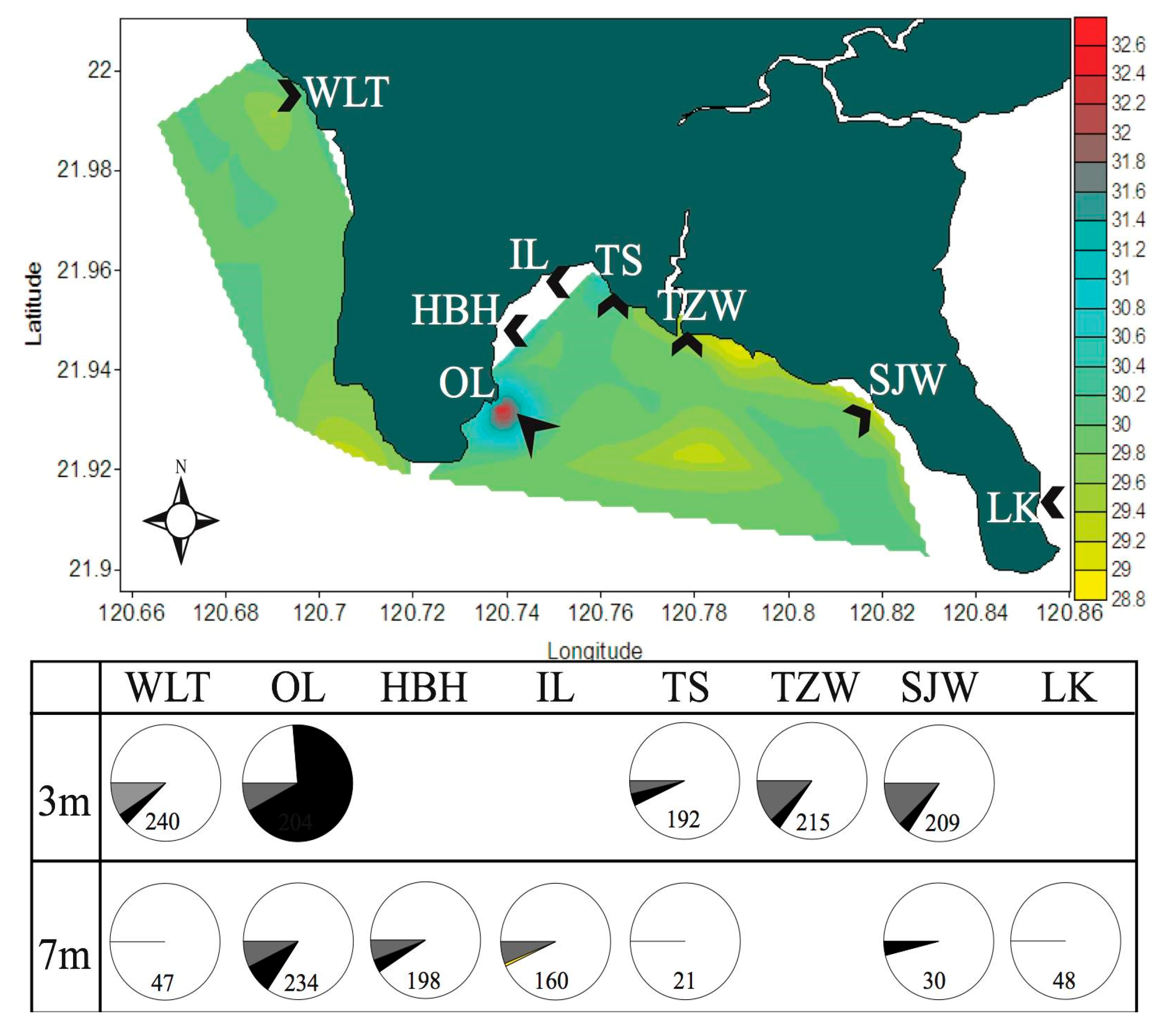
2.2. Symbiont Community Dynamics over Space and Time in KNP
3. Discussion and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ateweberhan, M.; Feary, D.A.; Keshavmurthy, S.; Chen, C.A.; Schleyer, M.H.; Sheppard, R.C. Climate change impacts on coral reefs: Synergies with local effects, possibilities for acclimation, and management implications. Mar. Pollut. Bull. 2013, 74, 526–539. [Google Scholar] [CrossRef] [PubMed]
- Hughes, T.P.; Baird, A.H.; Bellwood, D.R.; Card, M.; Connolly, S.R.; Folke, C.; Grosberg, R.; Hoegh-Guldberg, O.; Jackson, J.B.; Kleypas, J.; et al. Climate Change, Human Impacts, and the Resilience of Coral Reefs. Science 2013, 301, 929–933. [Google Scholar] [CrossRef] [PubMed]
- Hoegh-Guldberg, O.; Mumby, P.J.; Hooten, A.J.; Steneck, R.S.; Greenfield, P.; Gomez, E.; Harvell, C.D.; Sale, P.F.; Edwards, A.J.; Caldeira, K.; et al. Coral reefs under rapid climate change and ocean acidification. Science 2007, 318, 1737–1742. [Google Scholar] [CrossRef] [PubMed]
- National Academies of Sciences, Engineering, and Medicine. A Research Review of Interventions to Increase the Persistence and Resilience of Coral Reefs; The National Academies Press: Washington, DC, USA, 2018; 258p. [Google Scholar]
- Brown, B.E. Coral bleaching: Causes and consequences. Coral Reefs 1997, 16, S129–S138. [Google Scholar] [CrossRef]
- Hoegh-Guldberg, O. Climate change, coral bleaching and the future of the world’s coral reefs. Mar. Freshw. Res. 1999, 50, 839–866. [Google Scholar] [CrossRef]
- Hughes, T.P.; Kerry, J.T.; Álvarez-Noriega, M.; Álvarez-Romero, J.G.; Anderson, K.D.; Baird, A.H.; Babcock, R.C.; Beger, M.; Bellwood, D.R.; Berkelmans, R.; et al. Global warming and recurrent mass bleaching of corals. Nature 2017, 543, 373–377. [Google Scholar] [CrossRef]
- Hughes, T.P.; Anderson, K.D.; Connolly, S.R.; Heron, S.F.; Kerry, J.T.; Lough, J.M.; Baird, A.H.; Baum, J.K.; Berumen, M.L.; Bridge, T.C.; et al. Spatial and temporal patterns of mass bleaching of corals in the Anthropocene. Science 2018, 359, 80–83. [Google Scholar] [CrossRef]
- Gilmour, J.P.; Cook, K.L.; Ryan, N.M.; Puotinen, M.L.; Green, R.H.; Shedrawi, G.; Hobbs, J.-P.A.; Thomson, D.P.; Babcock, R.C.; Buckee, J.; et al. The state of Western Australia’s coral Reefs. Coral Reefs 2019, 38, 651–667. [Google Scholar] [CrossRef]
- Goyen, S.; Camp, E.F.; Fujise, L.; Lloyd, A.; Nitschke, M.R.; LaJeunensse, T.; Kahlke, T.; Ralph, P.J.; Suggett, D. Mass coral bleaching of P. verispora in Sydney Harbour driven by the 2015–2016 heatwave. Coral Reefs 2019, 38, 815–830. [Google Scholar] [CrossRef]
- Head, C.E.I.; Bayley, D.T.I.; Rowlands, G.; Roche, R.C.; Tickler, D.M.; Rogers, A.D.; Koldewey, H.; Turner, J.R.; Andradi-Brown, D.A. Coral bleaching impacts from back-to-back 2015–2016 thermal anomalies in the remote central Indian Ocean. Coral Reefs 2019, 38, 605–618. [Google Scholar] [CrossRef]
- Montefalcone, M.; Morri, C.; Bianchi, C.N. Long-term change in bioconstruction potential of Maldivian coral reefs following extreme climatic anomalies. Glob. Chang. Biol. 2018, 24, 5629–5641. [Google Scholar] [CrossRef] [PubMed]
- Fox, M.D.; Carter, A.L.; Edwards, C.B.; Takeshita, Y.; Johnson, M.D.; Petrovic, V.; Amir, C.G.; Sala, E.; Sandin, S.A.; Smith, J.E. Limited coral mortality following acute thermal stress and widespread bleaching on Palmyra Atoll, central Pacific. Coral Reefs 2019, 38, 701–712. [Google Scholar] [CrossRef]
- Raymundo, L.J.; Burdick, D.; Hoot, W.C.; Miller, R.M.; Brown, V.; Reynolds, T.; Gault, J.; Idechong, J.; Fifer, J.; Williams, A. Successive bleaching events cause mass coral mortality in Guam, Micronesia. Coral Reefs 2019, 38, 677–700. [Google Scholar] [CrossRef]
- Vargas-Ángel, B.; Huntington, B.; Brainard, R.E.; Venegas, R.; Oliver, T.; Barkley, H.; Cohen, A. El Nino-associated catastrophic coral mortality at Jarvis Island, central Equatorial Pacific. Coral Reefs 2019, 38, 731–741. [Google Scholar] [CrossRef]
- Smith, K.M.; Payton, T.G.; Sims, R.J.; Stroud, C.S.; Jeanes, R.C.; Hyatt, T.B.; Childress, M.J. Impacts of consecutive bleaching events and local algal abundance on transplanted coral colonies in the Florida Keys. Coral Reefs 2019, 38, 851–861. [Google Scholar] [CrossRef]
- Teixeira, C.D.; Leitão, R.L.L.; Ribeiro, F.V.; Moraes, F.C.; Neves, L.M.; Bastos, A.C.; Pereira-Filho, G.H.; Kampel, M.; Salomon, P.S.; Sá, J.A.; et al. Sustained mass coral bleaching (2016–2017) in Brazilian turbid-zone reefs: Taxonomic, cross-shelf and habitat-related trends. Coral Reefs 2019, 38, 801–813. [Google Scholar] [CrossRef]
- Eakin, M.C.; Sweatman, H.P.A.; Brainard, R.E. The 2014–2017 global-scale coral bleaching event: Insights and impacts. Coral Reefs 2019, 38, 539–545. [Google Scholar] [CrossRef]
- Masson-Delmotte, V.; Zhai, P.; Pörtner, H.O.; Roberts, D.; Skea, J.; Shukla, P.R.; Pirani, A.; Moufouma-Okia, W.; Péan, C.; Pidcock, R.; et al. (Eds.) IPCC, 2018: Summary for Policymakers. In Global Warming of 1.5 °C. An IPCC Special Report on the Impacts of Global Warming of 1.5 °C above Pre-Industrial Levels and Related Global Greenhouse Gas Emission Pathways, in the Context of Strengthening the Global Response to the Threat of Climate Change, Sustainable Development, and Efforts to Eradicate Poverty; World Meteorological Organization: Geneva, Switzerland, 2018; 32p. [Google Scholar]
- Riegl, B.M.; Purkis, S.J.; Al-Cibahy, A.S.; Abdel-Moati, M.A.; Hoegh-Guldberg, O. Present limits to heat-adaptability in corals and population-level responses to climate extremes. PLoS ONE 2011, 6, e24802. [Google Scholar] [CrossRef]
- Pandolfi, J.M.; Connolly, S.R.; Marshall, D.J.; Cohen, A.L. Projecting Coral Reef Futures Under Global Warming and Ocean Acidification. Science 2011, 333, 418–422. [Google Scholar] [CrossRef]
- Kao, K.-W.; Keshavmurthy, S.; Tsao, C.-H.; Wang, J.-T.; Chen, C.A. Repeated and Prolonged Temperature Anomalies Negate Symbiodiniaceae Genera Shuffling in the Coral Platygyra verweyi (Scleractinia; Merulinidae). Zool. Stud. 2018, 57. [Google Scholar] [CrossRef]
- Emanuel, K.A. Downscaling CMIP5 climate models shows increased tropical cyclone activity over the 21st century. Proc. Natl. Acad. Sci. USA 2013, 110, 12219–12224. [Google Scholar] [CrossRef] [PubMed]
- Tsuboki, K.; Yoshioka, M.K.; Shinoda, T.; Kato, M.; Kanada, S.; Kitoh, A. Future increase of super typhoon intensity acclimated with climate change. Geophys. Res. Lett. 2015, 42, 646–652. [Google Scholar] [CrossRef]
- Knutson, T.R.; McBride, J.L.; Chan, J.; Emanuel, K.; Holland, G.; Landsea, C.; Held, I.; Kossin, J.P.; Srivastava, A.K.; Sugi, M. Tropical cyclones and climate change. Nat. Geosci. 2010, 3, 157–163. [Google Scholar] [CrossRef]
- Christensen, J.H.; Kanikicharla, K.K.; Marshall, G.; Turner, J. Climate phenomena and their relevance for future regional climate change. In Climate Change 2013: The Physical Science Basis; Stocker, T.F., Qin, D., Plattner, G.-K., Tignor, M., Allen, S.K., Boschung, J., Nauels, A., Xia, Y., Bex, V., Midgley, P.M., Eds.; Cambridge University Press: Cambridge, UK, 2013; pp. 1217–1308. [Google Scholar]
- Walsh, K.J.; McBride, J.L.; Klotzbach, P.J.; Balachandran, S.; Camargo, S.J.; Holland, G.; Knutson, T.R.; Kossin, J.P.; Lee, T.; Sobel, A.; et al. Tropical cyclones and climate change. WIREs Clim. Chang. 2016, 7, 65–89. [Google Scholar] [CrossRef]
- Dollar, S.J. Wave stress and coral community structure in Hawaii. Coral Reefs 1982, 1, 71–81. [Google Scholar] [CrossRef]
- Cheal, A.J.; MacNeil, M.A.; Emslie, M.J.; Sweatman, H. The threat to coral reefs from more intense cyclones under climate change. Glob. Chang. Biol. 2017, 23, 1511–1524. [Google Scholar] [CrossRef]
- Holling, C.S. Resilience and stability of ecological systems. Annl. Rev. Ecol. Syst. 1973, 4, 1–23. [Google Scholar] [CrossRef]
- Nyström, M.; Folke, C. Spatial resilience of coral reefs. Ecosystems 2001, 4, 406–417. [Google Scholar] [CrossRef]
- Nystrom, M. Redundancy and response diversity of functional groups: Implications for the resilience of coral reefs. AMBIO 2006, 35, 30–35. [Google Scholar] [CrossRef]
- Norström, A.; Nyström, M.; Lokrantz, J.; Folke, C. Alternative states of coral reefs: Beyond coral-macroalgal phase shifts. Mar. Ecol. Prog. Ser. 2009, 376, 295–306. [Google Scholar] [CrossRef]
- Nyström, M.; Norström, A.; Blenckner, T.; de la Torre-Castro, M.; Eklöf, J.S.; Folke, C.; Österblom, H.; Steneck, R.S.; Thyresson, M.; Troell, M. Confronting Feedbacks of Degraded Marine Ecosystems. Ecosystems 2012, 15, 695–710. [Google Scholar] [CrossRef]
- Hughes, T.P.; Graham, N.A.J.; Jackson, J.B.C.; Mumby, P.J.; Steneck, R.S. Rising to the challenge of sustaining coral reef resilience. Trends Ecol. Evolut. 2010, 25, 633–642. [Google Scholar] [CrossRef] [PubMed]
- Montefalcone, M.; Parravicini, V.; Bianchi, C.N. Quantification of cosstal ecosystem resilience. In Treatise on Estuarine and Coastal Science; Elsevier: Amsterdam, The Netherlands, 2011; Volume 10, pp. 49–70. [Google Scholar]
- Folke, C. Resilience: The emergence of a perspective for social–ecological systems analyses. Glob. Environ. Chang. 2006, 16, 253–267. [Google Scholar] [CrossRef]
- Nystrom, M.; Graham, A.J.; Lokrantz, J.; Norstorm, A.V. Capturing the cornerstones of coral reef resilience: Linking theory to practice. Coral Reefs 2008, 27, 795–809. [Google Scholar] [CrossRef]
- Anthony, K.R.N. Coral Reefs Under Climate Change and Ocean Acidification: Challenges and Opportunities for Management and Policy. Annu. Rev. Environ. Res. 2016, 41, 59–81. [Google Scholar] [CrossRef]
- Barneah, O.; Brickner, I. Multibiont Symbioses in the Coral Reef Ecosystem. In Symbioses and Stress. Cellular Origin, Life in Extreme Habitats and Astrobiology; Seckbach, J., Grube, M., Eds.; Springer: Dordrecht, The Netherlands, 2010; Volume 17. [Google Scholar]
- Baker, A.C. Flexibility and specificity in coral-algal symbiosis: Diversity, ecology, and biogeography of Symbiodinium. Annu. Rev. Ecol. Evol. Syst. 2003, 34, 661–689. [Google Scholar] [CrossRef]
- Lajeunesse, T.C.; Pettay, D.T.; Sampayo, E.E.; Phongsuwan, N.; Brown, B.E.; Obura, D.O.; Hoegh-Guldberg, O.; Fitt, W.K. Long-standing environmental conditions, geographic isolation and host-symbiont specificity influence the relative ecological dominance and genetic diversification of coral endosymbionts in the genus Symbiodinium. J. Biogeogr. 2010, 37, 785–800. [Google Scholar] [CrossRef]
- Silverstein, R.N.; Correa, A.M.S.; Baker, A.C. Specificity is rarely absolute in coral-algal symbiosis: Implications for coral response to climate change. Proc. R. Soc. B Biol. Sci. 2012, 279, 2609–2618. [Google Scholar] [CrossRef]
- Lajeunesse, T.C.; Parkinson, J.E.; Gabrielson, P.W.; Jeong, H.J.; Reimer, J.D.; Voolstra, C.R.; Santos, S.R. Systematic Revision of Symbiodiniaceae Highlights the Antiquity and Diversity of Coral Endosymbionts. Curr. Biol. 2018, 28, 2570–2580. [Google Scholar] [CrossRef]
- Weber, M.; Medina, M. The role of microalgal symbionts (Symbiodinium) in holobiont physiology. Adv. Bot. Res. 2012, 64, 119–140. [Google Scholar]
- Sampayo, E.E.; Ridgway, T.; Bongaerts, P.; Hoegh-Guldberg, O. Bleaching susceptibility and mortality of corals are determined by fine-scale differences in symbiont type. Proc. Natl. Acad. Sci. USA 2008, 105, 10444–10449. [Google Scholar] [CrossRef]
- Jones, A.M.; Berkelmans, R.; Van Oppen, M.J.H.; Mieog, J.C.; Sinclair, W. A community change in the algal endosymbionts of a scleractinian coral following a natural bleaching event: Field evidence of acclimatization. Proc. R. Soc. B Biol. Sci. 2008, 275, 1359–1365. [Google Scholar] [CrossRef] [PubMed]
- Ulstrup, K.E.; Van Oppen, M. Geographic and habitat partitioning of genetically distinct zooxanthellae (Symbiodinium) in Acropora corals on the Great Barrier Reef. Mol. Ecol. 2003, 12, 3477–3484. [Google Scholar] [CrossRef]
- Lien, Y.-T.; Nakano, Y.; Plathong, S.; Fukami, H.; Wang, J.-T.; Chen, C.A. Occurrence of the putatively heat-tolerant Symbiodinium phylotype D in high-latitudinal outlying coral communities. Coral Reefs 2007, 26, 35–44. [Google Scholar] [CrossRef]
- Ghavam Mostafavi, P.; Fatemi, S.M.R.; Shahhosseiny, M.H.; Hoegh-Guldberg, O.; Loh, W.K.W. Predominance of clade D Symbiodinium in shallow-water reef-building corals off Kish and Larak Islands (Persian Gulf, Iran). Mar. Biol. 2007, 153, 25–34. [Google Scholar] [CrossRef]
- Oliver, T.A.; Palumbi, S.R. Many corals host thermally resistant symbionts in high-temperature habitat. Coral Reefs 2011, 30, 241–250. [Google Scholar] [CrossRef]
- Chen, C.A. Analysis of Scleractinian Distribution in Taiwan Indicating a Pattern Congruent with Sea Surface Temperatures and Currents: Examples from Acropora and Faviidae Corals. Zool. Stud. 1999, 38, 119–129. [Google Scholar]
- Chen, C.A.; Keshavmurthy, S. Taiwan as a connective stepping-stone in the Kuroshio Traiangle and the conservation of coral ecosystems under the impacts of climate change. Kuroshio Sci. 2009, 3, 15–22. [Google Scholar]
- Dai, C.F. Coastal and shallower water ecosystem. In Chapter 7. Regional Oceanography of Taiwan Version II; Jan, S., Ed.; National Taiwan University Press: Taipei, Taiwan, 2018; pp. 263–299. [Google Scholar]
- Dai, C.F. Assessment of the present health of coral reefs in Taiwan. In Status of Coral Reefs in the Pacific; UNIHI Sea Grant CP-98-01; Grigg, R.W., Birkeland, C., Eds.; Sea Grant Program, University of Hawaii: Honolulu, HI, USA, 1997; pp. 123–131. [Google Scholar]
- Bruno, J.F.; Selig, E.R. Regional Decline of Coral Cover in the Indo-Pacific: Timing, Extent, and Subregional Comparisons. PLoS ONE 2007, 2, e711. [Google Scholar] [CrossRef]
- Jackson, J.B.C.; Donovan, M.K.; Cramer, K.L.; Lam, V.V. (Eds.) Status and Trends of Caribbean Coral Reefs: 1970–2012. In Global Coral Reef Monitoring Network; IUCN: Gland, Switzerland, 2014; 298p. [Google Scholar]
- Meng, P.-J.; Chen, J.-P.; Chung, J.-N.; Liu, M.-C.; Fan, T.-Y.; Chang, C.-M.; Tian, W.-M.; Chang, Y.-C.; Lin, H.-J.; Fang, L.-S.; et al. Long-term ecological studies in Kenting National Park neighboring marine areas, on monitoring the impact factors from anthropogenic activities to the marine ecosystem and a preliminary database of its marine ecosystem. J. Natl. Park 2004, 14, 43–69, (In Chinese with English abstract). [Google Scholar]
- Meng, P.-J.; Lee, H.-J.; Wang, J.-T.; Chen, C.-C.; Lin, H.-J.; Tew, K.S.; Hsieh, W.-J. A long-term survey on anthropogenic impacts to the water quality of coral reefs, southern Taiwan. Environ. Pollut. 2008, 156, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.-J.; Meng, P.-J.; Liu, L.-L.; Wang, J.-T.; Leu, M.-Y. Impacts of human activities on coral reef ecosystems of southern Taiwan: A long-term study. Mar. Pollut. Bull. 2012, 64, 1129–1135. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.A. Status of Coral Reefs in Taiwan. In Status of Coral Reefs in East Asian Seas Region; Kimura, T., Tun, K., Chou, L.M., Eds.; Ministry of Environment: Tokyo, Japan, 2014; pp. 69–78. [Google Scholar]
- Kuo, C.-Y.; Yuen, Y.-S.; Meng, P.-J.; Ho, P.-H.; Wang, J.T.; Liu, P.J.; Chang, Y.C.; Dai, C.F.; Fan, T.Y.; Lin, H.J.; et al. Recurrent disturbances and the degradation of hard coral communities in Taiwan. PLoS ONE 2012, 7, e44364. [Google Scholar] [CrossRef] [PubMed]
- Ribas-Deulofeu, L.; Denis, V.; De Palmas, S.; Kuo, C.-Y.; Hsieh, H.J.; Chen, C.A. Structure of Benthic Communities along the Taiwan Latitudinal Gradient. PLoS ONE 2016, 11, e0160601. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-J.; Chao, S.-Y.; Fan, K.-L.; Kuo, T.-Y. Tide-Induced Eddies and Upwelling in a Semi-enclosed Basin: Nan Wan. Estuar. Coast. Shelf Sci. 1999, 49, 775–787. [Google Scholar] [CrossRef]
- Lee, H.-J.; Chao, S.-Y.; Fan, K.-L. Flood-ebb disparity of tidally induced recirculation Eddies in a semi-enclosed basin: Nan Wan Bay. Cont. Shelf Res. 1999, 19, 871–890. [Google Scholar] [CrossRef]
- Sugiyama, T. Reef building corals of Japanese coast. Pap. Inst. Geol. Paleontol. Tohoku Imp. Univ. 1937, 26, l–60. [Google Scholar]
- Kawaguti, S. Coral fauna of the Taiwan waters. Kagaku No Taiwan 1942, 11, l–6. [Google Scholar]
- Kawaguti, S. Coral fauna of Garampi. Trans. Formos. Nat. Hist. Soc. 1943, 33, 258–259. [Google Scholar]
- Kawaquti, S. Coral fauna of the island of Botel Tobago, Formosa with a list of corals from the Formosan waters. Biol. J. Okoyama Univ. 1953, 1, 185–198. [Google Scholar]
- Ma, T.Y.H. The effect of warm and cold currents in the southern-western Pacific on the growth rate of reef corals. Oceanogr. Sin. 1957, 5, l–34. [Google Scholar]
- MA, T.Y.H. The relation of growth rate of reef corals to surface temperature of sea water as basis for study of causes of diastrophisms instigating evolution of life. In Research on the Past Climate and Continental Drift; World Book Co.: Taipei, Taiwan, 1958; Volume 14. [Google Scholar]
- Ma, T.Y.H. Effect of water temperature on growth rate of corals. Oceanogr. Sin. Specif. 1959, 1, 116. [Google Scholar]
- Jones, O.A.; Randall, R.H.; Cheng, Y.M.; Kami, H.T.; Mak, S.M. A Marine Biological Survey of Southern Taiwan with Emphasis on Corals and Fishes; Institute of Oceanography, National Taiwan University: Taipei, Taiwan, 1972; 93p. [Google Scholar]
- Yang, R.T.; Huang, C.C.; Wang, C.H.; Yeh, S.Z.; Jan, Y.F.; Liu, S.L.; Chen, C.H.; Chen, S.J.; Chang, L.F.; Sun, C.L.; et al. A Marine Biological Data Acquisition Program Pertaining to the Construction of a Power Plant in the Nan-Wan Bay Area. Phase, I. A Preliminary Reconnaissance Survey; Institute of Oceanography, National Taiwan University: Taipei, Taiwan, 1976; 134p. [Google Scholar]
- Yang, R.T.; Huang, C.C.; Wang, C.H.; Yeh, S.Z.; Jan, Y.F.; Liu, S.L.; Chen, C.H.; Chen, S.J.; Chang, L.F.; Sun, C.L.; et al. A Marine Biological Data Acquisition Program Pertaining to the Construction of a Power Plant in the Nan-Wan Bay Area. Phasw 11, Biological Data Acquisition; Institute of Oceanography, National Taiwan University: Taipei, Taiwan, 1977; pp. 1–34. [Google Scholar]
- Randall, R.H.; Cheng, Y.M. Recent corals of Taiwan. Part, I. Description of reefs and coral environment. Acta Geol. Taiwanica 1977, 19, 79–102. [Google Scholar]
- Randall, R.H.; Cheng, Y.M. Recent corals of Taiwan. Part II. Description of reefs and coral environment. Acta Geol. Taiwanica 1979, 20, 1–32. [Google Scholar]
- Yang, R.T. Coral communities in Nan-wan Bay (Taiwan). In Proceedings of the Fifth International Coral Reef Congress, Tahiti, French Polynesia, 27 May–1 June 1985; Volume 6, pp. 273–278. [Google Scholar]
- Dai, C.-F. Community Ecology of Corals on the Fringing Reefs of Southern Taiwan. Ph.D. Thesis, Yale University, New Haven, CT, USA, 1988; p. 315. [Google Scholar]
- Dai, C.-F. Coral communities of southern Taiwan. In Proceedings of the 6th International Coral Reef Symposium, Townsville, Australia, 8–12 August 1988; Volume 2, pp. 647–652. [Google Scholar]
- Dai, C.-F.; Chen, Y.-T.; Kuo, K.-M.; Chuang, C.-H. Changes of coral communities in Nanwan Bay, Kenting National Park: 1987–1997. J. Natl. Park 1998, 8, 79–99, (In Chinese with English abstract). [Google Scholar]
- Dai, C.-F.; Kuo, K.-M.; Chen, Y.-T.; Chuang, C.-H. Changes of coral communities on the east and west coast of the Kenting National Park. J. Natl. Park 1999, 9, 112–130, (In Chinese with English abstract). [Google Scholar]
- Chang, J.-S.; Dai, C.-F.; Chang, J. The gametangium-like structure as propagation buds in Codium edule Silva (Bryopsidales: Chlorophya). Bot. Mar. 2003, 46, 431–437. [Google Scholar] [CrossRef]
- Chen, C.A.; Dai, C.-F. Local phase shift from Acropora-dominant to Condylactis- dominant community in the Tiao-Shi Reef, Kenting National Park, southern Taiwan. Coral Reefs 2004, 23, 508. [Google Scholar] [CrossRef]
- Shao, K.-T.; Fang, L.-S.; Liang, N.-K.; Meng, P.-J.; Chung, K.-N.; Li, J.-J.; Han, C.-C. Long-Term Ecological Research (LTER) Program of the Coral Reefs in Kenting National Park, Taiwan; Kenting National Park Headquarters, Construction and Planning Agency, Minister of the Interior: Taipei, Taiwan, 2002; p. 176. (In Chinese)
- Fang, L.-S.; Shao, K.-T.; Tian, W.-M.; Chang, Y.-C.; Lin, H.-J.; Fan, T.-Y.; Chung, K.-N.; Chen, J.-P.; Liu, M.-Y.; Meng, P.-J. Long-Term Ecological Research (LTER) Program of the Coral Reefs in Kenting National Park, Taiwan; Kenting National Park Headquarters, Construction and Planning Agency, Minister of the Interior: Taipei, Taiwan, 2003; p. 249. (In Chinese)
- Fang, L.-S.; Shao, K.-T.; Tian, W.-M.; Chang, Y.-C.; Lin, H.-J.; Fan, T.-Y.; Chung, K.-N.; Chen, J.-P.; Chen, M.-H.; Liu, M.-Y.; et al. Long-Term Ecological Research (LTER) Program of the Coral Reefs in Kenting National Park, Taiwan; Kenting National Park Headquarters, Construction and Planning Agency, Minister of the Interior: Taipei, Taiwan, 2004; p. 262. (In Chinese)
- Fang, L.-S.; Shao, K.-T.; Meng, P.-J.; Chung, K.-N.; Chen, J.-P.; Chen, M.-H.; Liu, M.-Y.; Chang, Y.-C.; Fan, T.-Y.; Lin, H.-J. Long-Term Ecological Research (LTER) Program of the Coral Reefs in the Kenting National Park; Kenting National Park Headquarters, Construction and Planning Agency, Minister of the Interior: Taipei, Taiwan, 2005; p. 295. (In Chinese)
- Fang, L.-S.; Shao, K.-T.; Meng, P.-J.; Chung, K.-N.; Chen, J.-P.; Chen, M.-H.; Liu, M.-Y.; Chang, Y.-C.; Fan, T.-Y.; Lin, H.-J. Long-Term Ecological Research (LTER) Program of the Coral Reefs in Kenting National Park, Taiwan; Kenting National Park Headquarters, Construction and Planning Agency, Minister of the Interior: Taipei, Taiwan, 2006; p. 233. (In Chinese)
- Wang, W.-H.; Shao, K.-T.; Meng, P.-J.; Fan, T.-Y.; Chen, J.-P.; Chen, M.-H.; Liu, M.-Y.; Chang, Y.-C.; Lin, H.-J.; Ho, P.-H. Long-Term Ecological Research (LTER) Program of the Coral Reefs in Kenting National Park, Taiwan; Kenting National Park Headquarters, Construction and Planning Agency, Minister of the Interior: Taipei, Taiwan, 2007; p. 226. (In Chinese)
- Ho, M.-J.; Cheng, N.-Y.; Chen, Y.-C.; Kuo, C.-Y.; Wen, C.K.-C.; Cherh, K.-L.; Chen, C.A. Investigating current status of benthic ecology in the Kenting National Park marine zones, Taiwan. J. Natl. Park 2016, 26, 46–56, (In Chinese with English abstract). [Google Scholar]
- Ho, P.-H.; Chen, C.A.; Chen, H.-Y.; Chen, J.-P.; Chiu, Y.-W.; Ho, P.-H.; Lin, H.-J.; Chang, Y.-C. Long-Term Ecological Research (LTER) Program of the Coral Reefs in Kenting National Park, Taiwan; Kenting National Park Headquarters, Construction and Planning Agency, Minister of the Interior: Taipei, Taiwan, 2008; p. 270. (In Chinese)
- Ho, P.-H.; Chen, C.A.; Meng, P.-J.; Chen, J.-P.; Chiu, Y.-W.; Lin, H.-J.; Chang, Y.-C. Long-Term Ecological Research (LTER) Program of the Coral Reefs in Kenting National Park, Taiwan; Kenting National Park Headquarters, Construction and Planning Agency, Minister of the Interior: Taipei, Taiwan, 2009; p. 276. (In Chinese)
- Ho, P.-H.; Chen, C.A.; Meng, P.-J.; Chen, J.-P.; Chiu, Y.-W.; Lin, H.-J.; Chang, Y.-C.; Liu, P.-J. Long-Term Ecological Research (LTER) Program of the Coral Reefs in Kenting National Park, Taiwan; Kenting National Park Headquarters, Construction and Planning Agency, Minister of the Interior: Taipei, Taiwan, 2010; p. 300. (In Chinese)
- Ho, P.-H.; Chen, C.A.; Meng, P.-J.; Chen, J.-P.; Chiu, Y.-W.; Lin, H.-J.; Chang, Y.-C.; Liu, P.-J. Long-Term Ecological Research (LTER) Program of the Coral Reefs in Kenting National Park, Taiwan; Kenting National Park Headquarters, Construction and Planning Agency, Minister of the Interior: Taipei, Taiwan, 2011; p. 330. (In Chinese)
- Chen, C.A. Long-Term Ecological Research (LTER) Program of the Coral Reefs in Kenting National Park, Taiwan; Kenting National Park Headquarters, Construction and Planning Agency, Minister of the Interior: Taipei, Taiwan, 2012; p. 73. (In Chinese)
- Chen, C.A.; Meng, P.-J.; Chiu, Y.-W. Annual Monitoring of Benthic Community, Water Quality and Shellfish in Kenting National Park; Kenting National Park Headquarters, Construction and Planning Agency, Minister of the Interior: Taipei, Taiwan, 2013; p. 119. (In Chinese)
- Chen, C.A. Evaluating and Re-Design the Zoning System in Kenting National Park, Taiwan; Kenting National Park Headquarters, Construction and Planning Agency, Minister of the Interior: Taipei, Taiwan, 2014; p. 133. (In Chinese)
- Chen, C.A. Annual Survey of the Biodiversity of Coral Reef Ecosystem in Kenting National Park, Taiwan; Kenting National Park Headquarters, Construction and Planning Agency, Minister of the Interior: Taipei, Taiwan, 2016; p. 82. (In Chinese)
- Chen, C.A. Annual Survey of the Biodiversity of Coral Reef Ecosystem in Kenting National Park, Taiwan; Kenting National Park Headquarters, Construction and Planning Agency, Minister of the Interior: Taipei, Taiwan, 2018; p. 100. (In Chinese)
- Kuo, C.-Y. The Structure and Variation of Benthic Communities in Coral Reefs of Southern Taiwan. Master’s Thesis, National Sun Yat-sen University, Kaohsiung, Taiwan, 2007; p. 71, (In Chinese with English abstract). [Google Scholar]
- Kuo, C.-Y.; Meng, P.-J.; Ho, P.-H.; Wang, J.-T.; Chen, J.-P.; Chiu, Y.-W.; Lin, H.-J.; Chang, Y.-C.; Fan, T.-Y.; Chen, C.M. Damage to the reefs of Siangjiao Bay marine protected area in Kenting National Park, Taiwan during Typhoon Morakot. Zool. Stud. 2011, 50, 85. [Google Scholar]
- Kuo, C.-Y.; Ho, M.-J.; Chen, Y.-C.; Yang, S.-Y.; Huang, Y.-Y.; Hsieh, H.-J.; Jeng, M.-S.; Chen, C.A. Status of Coral Reefs in East Asian Seas Region: Taiwan; Ministry of the Environment of Japan; Japan Wildlife Research Center: Tokyo, Japan, 2018; pp. 33–40.
- Kohler, K.E.; Gill, S.M. Coral point count with Excel extensions (CPCe): A visual basic program for the determination of coral and substrate coverage using random point count methodology. Comput. Geosci. 2006, 32, 1259–1269. [Google Scholar] [CrossRef]
- Price, J.F. Upper ocean response to a hurricane. J. Phys. Oceanogr. 1981, 11, 153–175. [Google Scholar] [CrossRef]
- Wada, A.; Uehara, T.; Ishizaki, S. Typhoon-induced sea surface cooling during the 2011 and 2012 typhoon seasons: Observational evidence and numerical investigations of the sea surface cooling effect using typhoon simulations. Prog. Earth Planet. Sci. 2014, 64, 3562–3578. [Google Scholar] [CrossRef]
- Keshavmurthy, S.; Meng, P.-J.; Wang, J.-T.; Kuo, C.-Y.; Yang, S.-Y.; Hsu, C.-M.; Gan, C.-H.; Dai, C.-F.; Chen, C.A. Can resistant coral-Symbiodinium associations enable coral communities to survive climate change? A study of a site exposed to long-term hot water input. PeerJ 2014, 2, e327. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.-M.; Keshavmurthy, S.; Denis, V.; Kuo, C.-Y.; Wang, J.-T.; Meng, P.-J.; Chen, C.A. Temporal and Spatial Variations in Symbiont Communities of Catch Bowl Coral Isopora palifera (Scleractinia: Acroporidae) on Reefs in Kenting National Park, Taiwan. Zool. Stud. 2012, 51, 1343–1353. [Google Scholar]
- Keshavmurthy, S.; Hsu, C.-M.; Kuo, C.-Y.; Meng, P.-J.; Wang, J.-T.; Chen, C.A. Symbiont communities and host genetic structure of the brain coral Platygyra verweyi, at the outlet of a nuclear power plant and adjacent areas. Mol. Ecol. 2012, 21, 4393–4407. [Google Scholar] [CrossRef]
- Carballo-Bolaños, R.; Denis, V.; Huang, Y.-Y.; Keshavmurthy, S.; Chen, C.A. Temporal variation and photochemical efficiency of species in Symbiodinaceae associated with coral Leptoria phrygia (Scleractinia; Merulinidae) exposed to contrasting temperature regimes. PLoS ONE 2019, 14, e0218801. [Google Scholar] [CrossRef]
- Chiou, W.-D.; Cheng, L.-Z.; Ou, H.-C. Relationship between the dispersion of thermal effluent and the tidal current in the waters near the outlet of the third nuclear power plant in southern Taiwan. J. Fish. Soc. Taiwan 1993, 20, 207–220. [Google Scholar]
- Fan, K.-L. The thermal effluent problems of three nuclear power plants in Taiwan. In Oceanography of Asian Marginal Seas, 54; Takano, K., Ed.; Elsevier: Amsterdam, The Netherlands, 1991; pp. 393–403. [Google Scholar]
- Pier, J.-J. Power uprate effect on thermal effluent of nuclear power plants in Taiwan. In Nuclear Power—Operation, Safety and Environment; Tsvetkov, P., Ed.; InTech Publication: Rijeka, Croatia, 2011; 287p. [Google Scholar]
- Chen, C.A.; Yang, Y.W.; Wei, N.V.; Tsai, W.S.; Fang, L.S. Symbiont diversity in scleractinian corals from tropical reefs and subtropical non-reef communities in Taiwan. Coral Reefs 2005, 24, 11–22. [Google Scholar] [CrossRef]
- Huang, Y.-Y.; Carballo-Bolaños, R.; Kuo, C.-Y.; Keshavmurthy, S.; Chen, C.A. Seasonal dynamics of the Symbiodiniaceae in Leptoria phrygia (Scleractinia; Merulinidae). (submitted, under review).
- Fong, W.-L. Spatial and Temporal Variations of Host Genetic Structure and Symbiont Community in the Catch Bowl Coral, Isopora Palifera (Scleractinia; Acroporidae), in Kenting National Park, Taiwan. Master’s Thesis, Institute of Oceanography, National Taiwan University, Taipei, Taiwan, 2016. [Google Scholar]
- Baker, A.C.; Starger, C.J.; McClanahan, T.R.; Glynn, P.W. Coral reefs: Corals’ adaptive response to climate change. Nature 2004, 430, 741. [Google Scholar] [CrossRef]
- Berkelmans, R.; Van Oppen, M. The role of zooxanthellae in the thermal tolerance of corals: A ‘nugget of hope’ for coral reefs in an era of climate change. Proc. R. Soc. B Biol. Sci. 2006, 273, 2305–2312. [Google Scholar] [CrossRef] [PubMed]
- Silverstein, R.N.; Cunning, R.; Baker, A.C. Change in algal symbiont communities after bleaching, not prior heat exposure, increases heat tolerance of reef corals. Glob. Chang. Biol. 2015, 21, 236–249. [Google Scholar] [CrossRef] [PubMed]
- Cunning, R.; Silverstein, R.N.; Baker, A.C. Investigating the causes and consequences of symbiont shuffling in a multi-partner reef coral symbiosis under environmental change. Proc. R. Soc. B Biol. Sci. 2015, 282, 20141725. [Google Scholar] [CrossRef]
- Boulotte, N.M.; Dalton, S.J.; Carroll, A.G.; Harrison, P.L.; Putnam, H.M.; Peplow, L.M.; van Oppen, M.J. Exploring the Symbiodinium rare biosphere provides evidence for symbiont switching in reef-building corals. ISME J. 2016. [Google Scholar] [CrossRef] [PubMed]
- Thornhill, D.J.; Lajeunesse, T.C.; Kemp, D.W.; Fitt, W.K.; Schmidt, G.W. Multi-year, seasonal genotypic surveys of coral-algal symbioses reveal prevalent stability or post-bleaching reversion. Mar. Biol. 2006, 148, 711–722. [Google Scholar] [CrossRef]
- Keshavmurthy, S.; Tang, K.-H.; Hsu, C.-M.; Gan, C.-H.; Kuo, C.-Y.; Soong, K.; Chou, H.-N.; Chen, C.A. Symbiodinium spp. associated with scleractinian corals from Dongsha Atoll (Pratas), Taiwan, in the South China Sea. PeerJ 2017, 5, e2871. [Google Scholar] [CrossRef]
- Tong, H.; Cai, L.; Zhou, G.; Yuan, T.; Zhang, W.; Tian, R.; Huang, H.; Qian, P.-Y. Temperature shapes coral-algal symbiosis in the South China Sea. Sci. Rep. 2017, 7, 40118. [Google Scholar] [CrossRef] [PubMed]
- Gong, S.; Chai, G.; Xiao, Y.; Xu, L.; Yu, K.; Li, J.; Liu, F.; Cheng, H.; Zhang, F.; Liao, B.; et al. Flexible symbiotic associations of Symbiodinium with five typical coral species in tropical and subtropical reef regions of the northern South China Sea. Front. Microbiol. 2018, 9, 2485. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.A.; Wang, J.-T.; Fang, L.-S.; Yang, Y.-W. Fluctuating algal symbiont communities in Acropora palifera (Scleractinia: Acroporidae) from Taiwan. Mar. Ecol. Prog. Ser. 2005, 295, 113–121. [Google Scholar] [CrossRef]
- Thornhill, D.J.; Fitt, W.K.; Schmidt, G.W. Highly stable symbioses among western Atlantic brooding corals. Coral Reefs 2006, 25, 515–519. [Google Scholar] [CrossRef]
- Tonk, L.; Sampayo, E.E.; Lajeunesse, T.C.; Schrameyer, V.; Hoegh-Guldberg, O. Symbiodinium (Dinophyceae) diversity in reef-invertebrates along an offshore to inshore reef gradient near Lizard Island, Great Barrier Reef. J. Phycol. 2014, 50, 552–563. [Google Scholar] [CrossRef] [PubMed]
- Beyer, H.L.; Kenndey, E.V.; Beger, M.; Chen, C.A.; Cinner, J.E.; Darling, E.S.; Eakin, C.M.; Gates, R.D.; Heron, S.F.; Knowlton, N.; et al. Risk-sensitive planning for conserving coral reefs under rapid climate change. Conserv. Lett. 2018, 11, e12587. [Google Scholar] [CrossRef]
- Ablan, M.C.A.; McManus, J.W.; Chen, C.A.; Shao, K.T.; Bell, J.; Cabanban, A.S.; Tuan, V.S.; Arthana, I.W. Meso-scale transboundary units for the management of coral reefs in the South China Sea area. NAGA Worldfish Cent. Q. 2002, 25, 4–9. [Google Scholar]
- Chen, C.A.; Ablan, M.C.A.; McManus, J.W.; Bell, J.D.; Tuan, V.S.; Cabanban, A.S.; Shao, K.-T. Population structure and genetic variability of six-bar wrasse (Thallasoma hardwicki) in northern South China Sea revealed by mitochondrial control region sequences. Mar. Biotechnol. 2004, 6, 312–326. [Google Scholar] [CrossRef]
- Chou, L.M. Southeast Asian reefs-a status update: Cambodia, Indonesia, Malaysia, Philippines, Singapore, Thailand and Vietnam. In Status of the Coral Reefs of the World: 2000; Wilkinson, C., Ed.; Australian Institute of Marine Science: Townsville, Australia, 2000; pp. 117–129. [Google Scholar]
- West, J.M.; Slam, R.V. Resistance and resilience to coral bleaching: Implications for coral reef conservation and management. Conserv. Biol. 2003, 4, 956–967. [Google Scholar] [CrossRef]
- Riegl, B.; Piller, W.E. Possible refugia for reefs in times of environmental stress. Int. J. Earth Sci. 2003, 92, 520. [Google Scholar] [CrossRef]
- Grimsditch, G.D.; Salm, R.V. Coral Reef Resilience and Resistance to Bleaching; IUCN: Gland, Switzerland, 2005; 50p. [Google Scholar]
- Chollett, I.; Mumby, P.J.; Cortes, J. Upwelling areas to not guarantee refuge for corals in a warming ocean. Mar. Ecol. Prog. Ser. 2010, 416, 47–56. [Google Scholar] [CrossRef]
- Glynn, P.W.; D’croz, L. Experimental evidence for high temperature stress as the cause of El Nino-coincident coral mortality. Coral Reefs 1990, 8, 181–191. [Google Scholar] [CrossRef]
- Sánchez-Noguera, C.; Stuhldreier, I.; Cortés, J.; Jiménez, C.; Morales, Á.; Wild, C.; Rixen, T. Natural ocean acidification at Papagayo upwelling system (North Pacific Costa Rica): Implications for reef development. Biogeosciences 2018, 15, 2349–2360. [Google Scholar] [CrossRef]
- Wizemann, A.; Nandini, S.D.; Stuhldreier, I.; Sánchez-Noguera, C.; Wisshak, M.; Westphal, H.; Rixen, T.; Wild, C.; Reymond, C.E. Rapid bioerosion in a tropical upwelling coral reef. PLoS ONE 2018, 13, e0202887. [Google Scholar] [CrossRef]

| Year | Sites | Survey Method | Survey Area per Transect | Number of Replicates at Each Site | Survey Depth | Methods | Identification Level | Reference |
|---|---|---|---|---|---|---|---|---|
| 1986–1987, 1998–1999 | Wanlitung (WLT) Hungchai (HC) Leidashih (LDS) Houbihu (HBH) Tiaoshi (TS) Siangjiaowan (SJW) | Line intercept transect | 10 m | 25 | 3–23 | A transect tape was placed perpendicular to the coast and extended seaward from 3 m depth to the reef edge at 25 m depth. A 10 m metal chain was placed parallel to the transect at 15 m intervals. | Species for corals Total algae | Dai, C.-F. (1988); Dai, C.-F. etc. (1998, 1999) |
| 2003–2005 2008–2014 2016, 2018 | Howan (HW) Wanlitung (WLT) Hungchai (HC) Leidashih (LDS) Houbihu (HBH) NPP Inlet (IL) Tiaoshi (TS) Siangjiaowan (SJW) Longkeng (LK) Jialeshuei (JLS) | 30 m × 0.25 m belt transects except 20 m × 0.25 m in TS | 7.5 m2 and 5 m2 in TS | 3, except 9 in TS | 5–10 | Three or nine (at Tiaoshi) permanent belt transects were established along depth contours between 5 and 10 m depth at each site. Benthic organisms were quantified using 25 × 25 cm2 photo-quadrats.The percent cover of the benthic categories was determined using Coral Count with Excel Extensions software (Kohler, K.E. 2016), with 30 random points per quadrat. Surveys were conducted between March and May each year. | Species for corals (2003–2005, 2011, 2014); Genus for corals (2008–2010, 2012–2013); Morphology for scleractinian corals (2016); Genus and morphology for scleractinian corals (2018) Macroalgae Turf algae | Kuo, C.-Y. etc. (2012); Shao, K.-T. etc. (2002); Fang, L.-S. etc. (2003–2006); Wang, W.-H. etc. (2007); Ho, M.-J. etc. (2016); Ho, P.-H. etc. (2008–2011); Chen, C.A. etc. (2012–2014, 2016, 2018); Kuo, C.-Y. etc. (2007) |
| Subregions | Site | Depth (m) | Yearly Mean Temperature | Max SST | Min SST | Max. Variability within 2 Hours |
|---|---|---|---|---|---|---|
| West Hengchun Peninsula | Wanlitung | 7 | 27.090 | 31.33 (July) | 22.77 (Dec.) | 3.15 (July) |
| West Nawan | Outlet | 2 | 27.580 | 34.02 (July) | 19.91 (Jan.) | 4.08 (July) |
| West Nawan | Outlet | 7 | 26.620 | 31.36 (Aug.) | 18.70 (Jan.) | 4.90 (Aug.) |
| East Nawan | Siangjiaowan | 7 | 26.580 | 31.09 (July) | 17.82 (Jan.) | 4.99 (July) |
| East Hengchun Peninsula | Longkeng | 7 | 26.460 | 30.93 (Sep.) | 20.63 (Oct.) | 3.13 (June) |
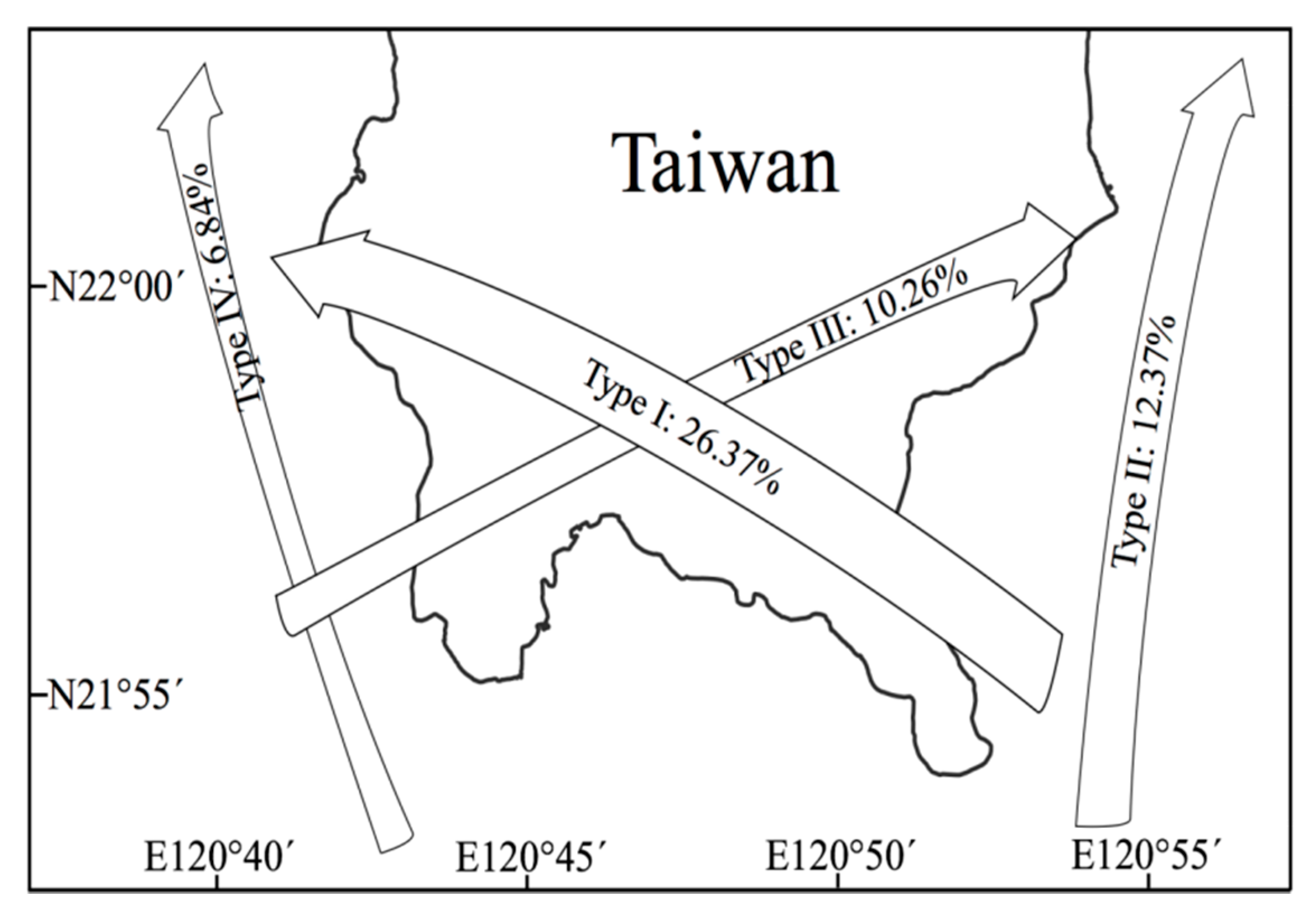
| Category | Name | Year | Note |
|---|---|---|---|
| Typhoon | Peggy (5) | 1986 | Type I |
| Typhoon | Gerald (4) | 1987 | Type I |
| Typhoon | Lynn (5) | 1987 | Type I |
| Typhoon | Herb (5) | 1996 | Type I |
| Typhoon | Chanchu (4) | 2006 | Type IV |
| Typhoon | Morakot (2) | 2009 | Type I |
| Typhoon | Nanmadol (5) | 2011 | Type I |
| Typhoon | Usagi (4) | 2013 | Type I |
| Typhoon | Soudelor (5) | 2015 | Type I |
| Typhoon | Meranti (5) | 2016 | Type I |
| Typhoon | Megi (4) | 2016 | Type I |
| Temperature anomaly | Bleaching | 1998 | Nearly all the colonies on the reefs shallower than 5m in depth in Outlet were bleached. |
| Temperature anomaly | Bleaching | 2002 | Minor, very small scale and local bleaching event were recorded in Wanlitung, Houbihu, and Siangjiaowan |
| Temperature anomaly | Bleaching | 2007 | 50% In Outlet and up to 25% on the West coast of Hengchun Peninsula and Nanwan |
| Temperature anomaly | Bleaching | 2010 | Minor scale on the hallow reef of the NPP OL |
| Temperature anomaly | Bleaching | 2014 | There were around 30% of the corals bleaching in KNP except in the Outlet that 50% and 20% of the corals were bleached on the shallow (shallower than 5m in depth) and deep (10m in depth) reefs. |
| Temperature anomaly | Bleaching | 2016 | Minor scale from Outlet to Nanwan beach |
| Temperature anomaly | Bleaching | 2017 | Minor scale on the West coast of Hengchun Peninsula |
| Ship grounding | Amorgos | 2001 | Limited on the East coast of Hengchun Penunsula, in particular Longkeng |
| Ship grounding | Colombo Queen | 2009 | East coast of Hengchun Peninsula |
| Ship grounding | WO-BUDMO | 2009 | West coast of Hengchun Peninsula |
| Biological outbreak | Sea anemone Condylactis sp. | Late 1996–2008 | Limited in the shallow area of Tiaoshi |
| Biological outbreak | Green alga Codium edule | Late 1996–2002 | Limited in the shallow area of Tiaoshi with significantly seasonal variation |
| Year | Host Species (Family/Genus) | Symbiodiniaceae Clade/Type/Genera/Species | Study Sites in KNP | Genetic Method for ID | Reference |
|---|---|---|---|---|---|
| 1997–2001 | Acropora | C3, C1, D1, D2, | srDNA- RFLP | Chen, C.A. etc. 2005 | |
| Montipora | C1 | ||||
| Pocilloporidae | C1, C2 | ||||
| Euphyllidae | C1, D1 | ||||
| Poritidae | C1 | ||||
| Siderastreidae | C1 | ||||
| Agariciidae | C1 | ||||
| Oculinidae | C3 | ||||
| Merulinidae | C1 | ||||
| Faviidae | C1 | ||||
| 2000–2001 | Isopora palifera | C, D | Tantzei Bay | srDNA- RFLP | Hsu, C.-M. etc. 2012 |
| 2006–2009 | Isopora palifera | C3, D1a | Tantzei Bay, Maobitou, | srDNA- RFLP | |
| Siatanzai, NPP-OL, | ITS2-DGGE | ||||
| Shiaowan, Siangjiaowan | |||||
| Longkeng | |||||
| 2009 | Platygyra verweyi | C3, D1a | Leidashih, Siatanzai | ITS2-DGGE | Keshavmurthy, S. etc. 2012 |
| Maobitou, NPP-OL | ITS1-qPCR | ||||
| Wanlitung, Hungchai | |||||
| NPP-IL, Tiaoshi, | |||||
| Tantzei Bay, Longkeng | |||||
| 2009–2010 | Acanthastrea | C1, D1a | Houbihu, NPP-OL | srDNA- RFLP | Keshavmurthy, S. etc. 2014 |
| Acropora | C21a, C3, D1a | Siangjiaowan, NPP-IL | ITS2-DGGE | ||
| Cyphastrea | C3, D1a | Wanlitung, Tiaoshi | ITS1-qPCR | ||
| Favia | C3, D1a | Tantzei Bay, Longkeng | |||
| Favites | C3, D1a | ||||
| Galaxea | C1, D1a | ||||
| Goniastrea | C1, D1a | ||||
| Isopora | C3, D1a | ||||
| Leptastrea | D1a | ||||
| Leptoria | C1, D1a | ||||
| Montastrea | C1, C3, D1a | ||||
| Montipora | C15, D1a | ||||
| Pavona | C1, D1a | ||||
| Platygyra | C3, D1a | ||||
| Pocillopora | C3, D1a | ||||
| Porites | C15, D1a | ||||
| Seriatopora | C1 | ||||
| Stylophora | C1 | ||||
| 2016–2017 | Leptoria phyrgia | Durusdinium glynii | Wanlitung | ITS2-DGGE | Carballo-Bolaños, R. etc. 2019 |
| Durusdinium trenchii | NPP-OL | ITS1-qPCR | |||
| Cladocopium C3w | |||||
| Cladocopium C21a | |||||
| Cladocopium sp. | |||||
| 2019 | Leptoria phyrgia | Durusdinium glynii | Wanlitung | ITS2 Amplicon | Huang, Y-Y. etc. 2019 |
| Durusdinium trenchii | NPP-OL | ||||
| Durusdinium D1.6, D17, D2, D5, D6 | |||||
| Cladocopicum C116, C15.7, C21a, C2r, C3.1 | |||||
| C3.8, C3b, C3d, C3e, C3s, C50 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Keshavmurthy, S.; Kuo, C.-Y.; Huang, Y.-Y.; Carballo-Bolaños, R.; Meng, P.-J.; Wang, J.-T.; Chen, C.A. Coral Reef Resilience in Taiwan: Lessons from Long-Term Ecological Research on the Coral Reefs of Kenting National Park (Taiwan). J. Mar. Sci. Eng. 2019, 7, 388. https://doi.org/10.3390/jmse7110388
Keshavmurthy S, Kuo C-Y, Huang Y-Y, Carballo-Bolaños R, Meng P-J, Wang J-T, Chen CA. Coral Reef Resilience in Taiwan: Lessons from Long-Term Ecological Research on the Coral Reefs of Kenting National Park (Taiwan). Journal of Marine Science and Engineering. 2019; 7(11):388. https://doi.org/10.3390/jmse7110388
Chicago/Turabian StyleKeshavmurthy, Shashank, Chao-Yang Kuo, Ya-Yi Huang, Rodrigo Carballo-Bolaños, Pei-Jei Meng, Jih-Terng Wang, and Chaolun Allen Chen. 2019. "Coral Reef Resilience in Taiwan: Lessons from Long-Term Ecological Research on the Coral Reefs of Kenting National Park (Taiwan)" Journal of Marine Science and Engineering 7, no. 11: 388. https://doi.org/10.3390/jmse7110388
APA StyleKeshavmurthy, S., Kuo, C.-Y., Huang, Y.-Y., Carballo-Bolaños, R., Meng, P.-J., Wang, J.-T., & Chen, C. A. (2019). Coral Reef Resilience in Taiwan: Lessons from Long-Term Ecological Research on the Coral Reefs of Kenting National Park (Taiwan). Journal of Marine Science and Engineering, 7(11), 388. https://doi.org/10.3390/jmse7110388





