Climate Change and Active Reef Restoration—Ways of Constructing the “Reefs of Tomorrow”
Abstract
:1. Introduction: Advocatus Diaboli (the Devil’s Advocate)
2. Active Reef Restoration—The “Gardening” Tenet

3. Active Reef Restoration—An Applied Tool for Mitigation of Global Change Impacts
4. The “Gardening” Approach as a Climate Change Impact Mitigator
4.1. Take-Home Lessons
| Date | Restoration Act | Environmental Catastrophe/Stressor | Major Monitoring Outcomes |
|---|---|---|---|
| August 2005 | Populating CN; >6800 fragments | High survivorship of fragments | |
| September | Monitoring CN | Normal coral growth in CN | |
| October | Monitoring CN | Normal coral growth in CN | |
| November | Monitoring CN | Normal coral growth in CN | |
| December | Monitoring CN | Normal coral growth in CN | |
| January 2006 | Monitoring CN | Normal coral growth in CN | |
| February | Monitoring CN | Normal coral growth in CN | |
| March | Monitoring CN | Normal coral growth in CN | |
| April | Monitoring CN | Normal coral growth in CN | |
| May | Monitoring CN | Category “4” typhoon (Caloy) | Increased mortality and colony detachments in nursery farmed corals |
| June | Monitoring CN | Post typhoon reduced growth in CN | |
| July | Preparation for CT | ||
| August | CT, about 1200 colonies | Heavy precipitation causing seepage of fresh-water from reef ground | Mass mortality (ca. 50%) of transplants near freshwater seepages |
| September | CN repopulated; >5400 fragments | Rough SWM season starts | |
| October | SWM season; super-typhoon Paeng | Extremely tempestuous season causing mass mortalities in CN and transplants | |
| November | Monitoring transplants | SWM season; super-typhoon Reming | Variable mortalities at specific site locations |
| Dec | Monitoring CN | Still impacts from the last typhoon | |
| January 2007 | |||
| February | Monitoring transplants | Survivors grow well, reduced mortality | |
| March | Monitoring CN | Normal coral growth in CN | |
| April | |||
| May | Monitoring transplants | Survivors grow well, reduced mortality | |
| June | Monitoring CN | Unusual elevated seawater temperature, extreme low tide and high radiation | Major bleaching event. Increased mortalities. Significant high partial mortalities in CN and transplants. Reduced growths |
| July | Monitoring CN and transplants | Many colonies recovered the bleaching. |
4.2. Genetics
4.3. Repository for Coral and Reef Species
4.4. Ecosystem Engineering
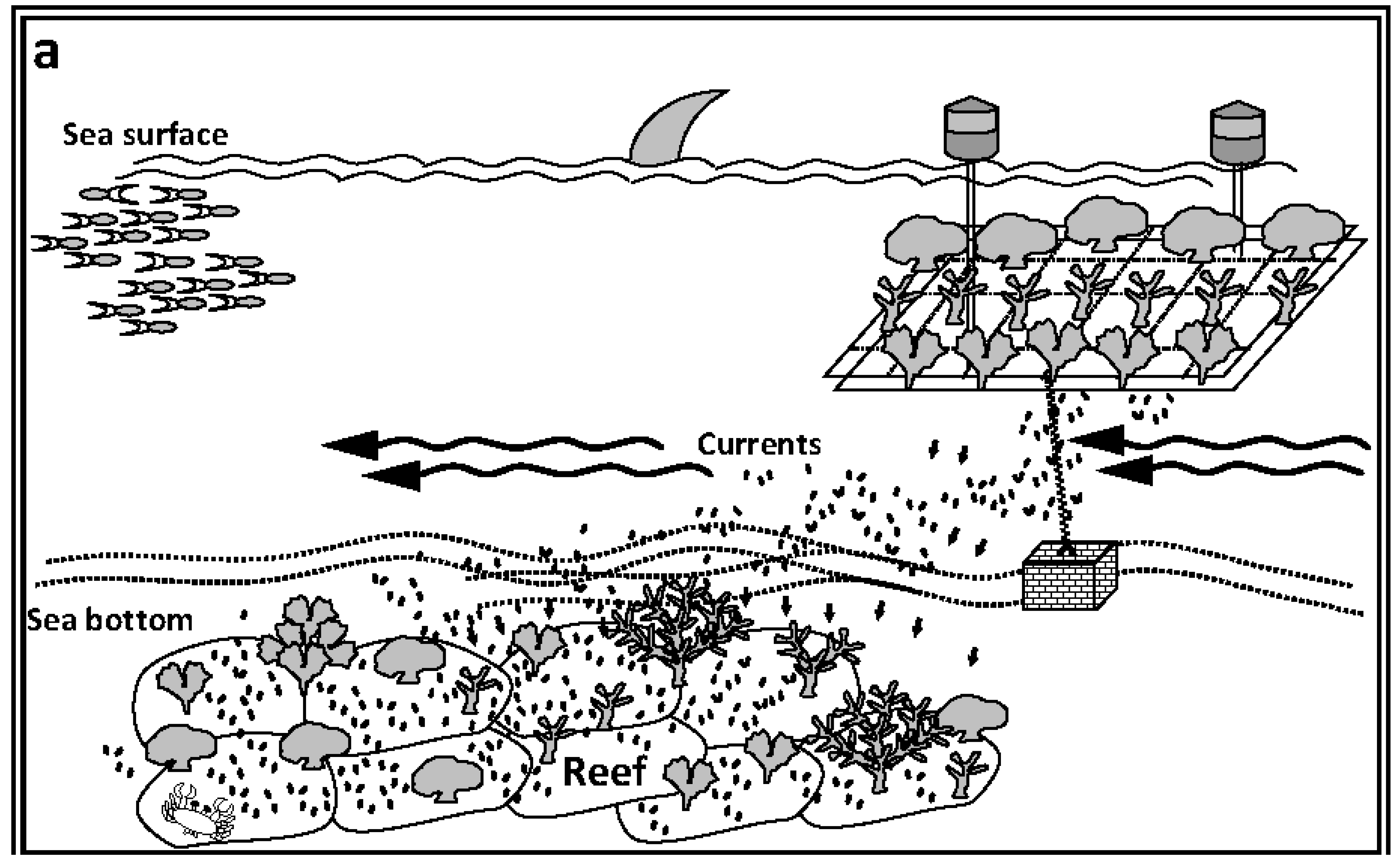
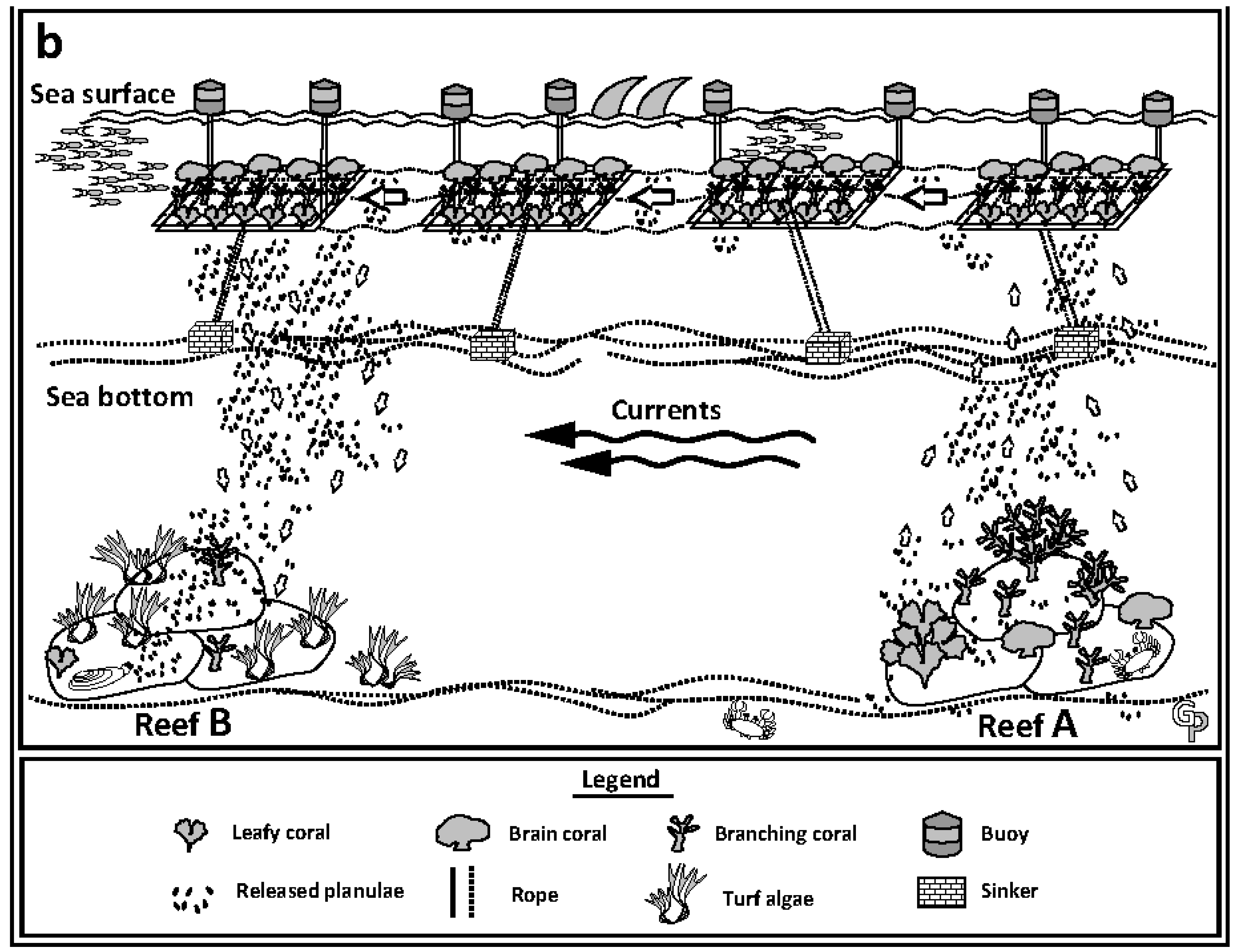
5. Closing Remarks
Acknowledgments
Conflicts of Interest
References
- Knutson, T.R.; McBride, J.L.; Chan, J.; Emanuel, K.; Holland, G.; Landsea, C.; Held, I.; Kossin, J.P.; Srivastava, A.K.; Sugi, M. Tropical cyclones and climate change. Nat. Geosci. 2010, 3, 157–163. [Google Scholar] [CrossRef]
- Mendelsohn, R.; Emanuel, K.; Chonabayashi, S.; Bakkensen, L. The impact of climate change on global tropical cyclone damage. Nat. Clim. Chang. 2012, 2, 205–209. [Google Scholar] [CrossRef]
- Perry, C.T.; Murphy, G.N.; Kench, P.S.; Edinger, E.N.; Smithers, S.G.; Steneck, R.S.; Mumby, P.J. Changing dynamics of Caribbean reef carbonate budgets: Emergence of reef bioeroders as critical controls on present and future reef growth potential. Proc. Biol. Sci. 2014, 281, 2014–2018. [Google Scholar] [CrossRef]
- Graham, N.A.; Cinner, J.E.; Norström, A.V.; Nyström, M. Coral reefs as novel ecosystems: Embracing new futures. Cur. Opin. Environ. Sustain. 2014, 7, 9–14. [Google Scholar] [CrossRef]
- Ateweberhan, M.; Feary, D.A.; Keshavmurthy, S.; Chen, A.; Schleyer, M.H.; Sheppard, C.R. Climate change impacts on coral reefs: Synergies with local effects, possibilities for acclimation, and management implications. Mar. Pollut. Bull. 2013, 74, 526–539. [Google Scholar] [CrossRef] [PubMed]
- Gardner, T.A.; Côté, I.M.; Gill, J.A.; Grant, A.; Watkinson, A.R. Long-term region-wide declines in Caribbean corals. Science 2003, 301, 958–960. [Google Scholar] [CrossRef]
- Côté, I.M.; Gill, J.A.; Gardner, T.A.; Watkinson, A.R. Measuring coral reef decline through meta-analyses. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2005, 360, 385–395. [Google Scholar] [CrossRef] [PubMed]
- De’ath, G.; Fabricius, K.E.; Sweatman, H.; Puotinen, M. The 27-year decline of coral cover on the Great Barrier Reef and its causes. Proc. Natl. Acad. Sci. 2012, 109, 17995–17999. [Google Scholar] [CrossRef] [PubMed]
- De’ath, G.; Lough, J.M.; Fabricius, K.E. Declining coral calcification on the Great Barrier Reef. Science 2009, 323, 116–119. [Google Scholar] [CrossRef] [PubMed]
- Cantin, N.E.; Cohen, A.L.; Karnauskas, K.B.; Tarrant, A.M.; McCorkle, D.C. Ocean warming slows coral growth in the central Red Sea. Science 2010, 329, 322–325. [Google Scholar] [CrossRef]
- Albright, R.; Mason, B.; Langdon, C. Effect of aragonite saturation state on settlement and post-settlement growth of Porites astreoides larvae. Coral Reefs 2008, 27, 485–490. [Google Scholar] [CrossRef]
- Yamano, H.; Sugihara, K.; Nomura, K. Rapid poleward range expansion of tropical reef corals in response to rising sea surface temperatures. Geophys. Res. Lett. 2011, 38, L04601. [Google Scholar] [CrossRef]
- Chen, I.C.; Hill, J.K.; Ohlemüller, R.; Roy, D.B.; Thomas, C.D. Rapid range shifts of species associated with high levels of climate warming. Science 2011, 333, 1024–1026. [Google Scholar] [CrossRef] [PubMed]
- Precht, E.F.; Aronson, R.B. Climate flickers and range shifts of reef corals. Front. Ecol. Environ. 2004, 2, 307–314. [Google Scholar] [CrossRef]
- Baird, A.H.; Sommer, B.; Madin, J.S. Pole-ward range expansion of Acropora spp. along the east coast of Australia. Coral Reefs 2012, 31, 1063. [Google Scholar] [CrossRef]
- Yamano, H.; Sugihara, K.; Goto, K.; Kazama, T.; Yokoyama, K.; Okuno, J. Ranges of obligate coral-dwelling crabs extend northward as their hosts move north. Coral Reefs 2012, 31, 663–663. [Google Scholar] [CrossRef]
- Figueira, W.F.; Booth, D.J. Increasing ocean temperatures allow tropical fishes to survive overwinter in temperate waters. Global Chang. Biol. 2010, 16, 506–516. [Google Scholar] [CrossRef]
- Hoegh-Guldberg, O. Climate change, coral bleaching and the future of the world’s coral reefs. Mar. Freshw. Res. 1999, 50, 839–866. [Google Scholar] [CrossRef]
- Albins, M.A.; Hixon, M.A. Worst case scenario: Potential long-term effects of invasive predatory lionfish (Pterois volitans) on Atlantic and Caribbean coral-reef communities. Environ. Biol. Fishes 2013, 96, 1151–1157. [Google Scholar] [CrossRef]
- Bruno, J.F.; Selig, E.R.; Casey, K.S.; Page, C.A.; Willis, B.L.; Harvell, C.D.; Sweatman, H.; Melendy, A.M. Thermal stress and coral cover as drivers of coral disease outbreaks. PLoS Biol. 2007, 5, 1220–1227. [Google Scholar] [CrossRef]
- Côté, I.M.; Precht, W.F.; Aronson, R.B.; Gardner, T.A. Is Jamaica a good model for understanding Caribbean coral reef dynamics? Mar. Pollut. Bull. 2013, 76, 28–31. [Google Scholar] [CrossRef] [PubMed]
- Rinkevich, B. Management of coral reefs: We have gone wrong when neglecting active reef restoration. Mar. Pollut. Bull. 2008, 56, 1821–1824. [Google Scholar] [CrossRef] [PubMed]
- Rinkevich, B. Rebuilding coral reefs: Does active reef restoration lead to sustainable reefs? Curr. Opin. Environ. Sustain. 2014, 7, 28–36. [Google Scholar] [CrossRef]
- Bruno, J.F.; Selig, E.R. Regional decline of coral cover in the Indo-Pacific: Timing, extent, and subregional comparisons. PLoS One 2007, 2, e711. [Google Scholar] [CrossRef] [PubMed]
- Miller, K.I.; Russ, G.R. Studies of no-take marine reserves: Methods for differentiating reserve and habitat effects. Ocean Coast. Manag. 2014, 96, 51–60. [Google Scholar] [CrossRef]
- Rinkevich, B. Restoration strategies for coral reefs damaged by recreational activities: The use of sexual and asexual recruits. Res. Ecol. 1995, 3, 241–251. [Google Scholar] [CrossRef]
- Rinkevich, B. Steps towards the evaluation of coral reef restoration by using small branch fragments. Mar. Biol. 2000, 136, 807–812. [Google Scholar] [CrossRef]
- Rinkevich, B. Conservation of coral reefs through active restoration measures: Recent approaches and last decade progress. Environ. Sci. Technol. 2005, 39, 4333–4342. [Google Scholar] [CrossRef] [PubMed]
- Rinkevich, B. The coral gardening concept and the use of underwater nurseries; lesson learned from silvics and silviculture. In Coral Reef Restoration Handbook; Precht, W.F., Ed.; CRC Press: Boca Raton, FL, USA, 2006; pp. 291–301. [Google Scholar]
- Levi, G.; Shaish, L.; Haim, A.; Rinkevich, B. Mid-water rope nursery—Testing design and performance of a novel reef restoration instrument. Ecol. Eng. 2010, 36, 560–569. [Google Scholar] [CrossRef]
- Shaish, L.; Levi, G.; Katzir, G.; Rinkevich, B. Coral reef restoration (Bolinao, the Philippines) in the face of frequent natural catastrophes. J. Soc. Ecol. Res. 2010, 18, 285–299. [Google Scholar] [CrossRef]
- Shaish, L.; Levi, G.; Katzir, G.; Rinkevich, B. Employing a highly fragmented, weedy coral species in reef restoration. Ecol. Eng. 2010, 36, 1424–1432. [Google Scholar] [CrossRef]
- Horoszowski-Fridman, Y.B.; Izhaki, I.; Rinkevich, B. Engineering of coral reef larval supply through transplantation of nursery-farmed gravid colonies. J. Exp. Mar. Biol. Ecol. 2011, 399, 162–166. [Google Scholar] [CrossRef]
- Linden, B.; Rinkevich, B. Creating stocks of young colonies from brooding-coral larvae, amenable to active reef restoration. J. Exp. Mar. Biol. Ecol. 2011, 398, 40–46. [Google Scholar] [CrossRef]
- Iversen, C.M. Digging deeper: Fine-root responses to rising atmospheric CO2 concentration in forested ecosystems. N. Phytol. 2010, 186, 346–357. [Google Scholar] [CrossRef]
- Loudermilk, E.L.; Scheller, R.M.; Weisberg, P.J.; Yang, J.; Dilts, T.E.; Karam, S.L.; Skinner, C. Carbon dynamics in the future forest: The importance of long-term successional legacy and climate—Fire interactions. Global Chang. Biol. 2013, 19, 3502–3515. [Google Scholar]
- Raymundo, L.J.; Maypa, A.P. Getting bigger faster: Mediation of size-specific mortality via fusion in juvenile coral transplants. Ecol. Appl. 2014, 14, 281–295. [Google Scholar] [CrossRef]
- Wild, C.; Hoegh-Guldberg, O.; Naumann, M.S.; Colombo-Pallotta, M.F.; Ateweberhan, M.; Fitt, W.K.; Iglesias-Prieto, R.; Palmer, C.; Bythell, J.C.; Ortiz, J.C.; et al. Climate change impedes scleractinian corals as primary reef ecosystem engineers. Mar. Freshw. Res. 2011, 62, 205–215. [Google Scholar] [CrossRef]
- Hughes, T.P.; Graham, N.A.J.; Jackson, J.B.C.; Mumby, P.J.; Steneck, R.S. Rising to the challenge of sustaining coral reef resilience. Trends Ecol. Evol. 2010, 25, 633–642. [Google Scholar] [CrossRef] [PubMed]
- Pandolfi, J.M.; Connolly, S.R.; Marshall, D.J.; Cohen, A.L. Projecting coral reef futures under global warming and ocean acidification. Science 2011, 333, 418–422. [Google Scholar] [CrossRef] [PubMed]
- Yakob, L.; Mumby, P.J. Climate change induces demographic resistance to disease in novel coral assemblages. Pro. Natl. Acad. Sci. 2011, 108, 1967–1969. [Google Scholar] [CrossRef]
- Tepe, T.L.; Meretsky, V.J. Forward-looking forest restoration under climate change—Are us nurseries ready? Rest. Ecol. 2011, 19, 295–298. [Google Scholar] [CrossRef]
- Gillson, L.; Dawson, T.P.; Jack, S.; McGeoch, M.A. Accommodating climate change contingencies in conservation strategy. Trends Ecol. Evol. 2013, 28, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Bay, R.A.; Palumbi, S.R. Multilocus adaptation associated with heat resistance in reef-building corals. Curr. Biol. 2014, 24, 2952–2956. [Google Scholar] [CrossRef] [PubMed]
- Cebrian, E.; Kipson, S.; Garrabou, J. Does thermal history influence the tolerance of temperate gorgonians to future warming? Mar. Environ. Res. 2013, 89, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Pistevos, J.C.A.; Calosi, P.; Widdicombe, S.; Bishop, J.D.D. Will variation among genetic individuals influence species responses to global climate change? Oikos 2011, 120, 675–689. [Google Scholar] [CrossRef]
- Evans, T.G.; Hoffman, G.E. Defining the limits of physiological plasticity: How gene expression can assess and predict the consequences of ocean change. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2012, 367, 1733–1745. [Google Scholar] [CrossRef] [PubMed]
- Fa, J.E.; Funk, S.M.; O’Connell, D.M. Zoo Conservation Biology; Cambridge University Press: Cambridge, UK, 2011. [Google Scholar]
- Schopmeyer, S.A.; Lirman, D.; Bartels, E.; Byrne, J.; Gilliam, D.S.; Hunt, J.; Johnson, M.E.; Larson, E.A.; Maxwell, K.; Nedimyer, K.; et al. In situ coral nurseries serve as genetic repositories for coral reef restoration after an extreme cold-water event. Restor. Ecol. 2011, 20, 696–703. [Google Scholar] [CrossRef]
- Shafir, S.; Rinkevich, B. Integrated long term mid-water coral nurseries: A management instrument evolving into a floating ecosystem. Maurit. Res. J. 2010, 16, 365–379. [Google Scholar]
- Sekercioglu, C.H.; Schneider, S.H.; Fay, J.P.; Loarie, S.R. Climate change, elevational range shifts, and bird extinctions. Conserv. Biol. 2008, 22, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Mouillot, D.; Bellwood, D.R.; Baraloto, C.; Chave, J.; Galzin, R.; Harmelin-Vivien, M.; Kulbicki, M.; Lavergne, S.; Lavorel, S.; Mouquet, N.; et al. Rare species support vulnerable functions in high-diversity ecosystems. PLoS Biol. 2013, 11, e1001569. [Google Scholar] [CrossRef] [PubMed]
- Gray, L.K.; Hamann, A. Strategies for reforestation under uncertain future climates: Guidelines for Alberta, Canada. PLoS One 2011, 6, e22977. [Google Scholar] [CrossRef]
- Shafir, S.; van Rijn, J.; Rinkevich, B. Steps in the construction of underwater coral nursery, an essential component in reef restoration acts. Mar. Biol. 2006, 149, 679–687. [Google Scholar] [CrossRef]
- Coles, S.L.; Riegl, B.M. Thermal tolerances of reef corals in the Gulf: A review of the potential for increasing coral survival and adaptation to climate change through assisted translocation. Mar. Pollut. Bull. 2012, 72, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Beger, M.; Sommer, B.; Harrison, P.L.; Smith, S.D.; Pandolfi, J.M. Conserving potential coral reef refuges at high latitudes. Divers. Distrib. 2014, 20, 245–257. [Google Scholar] [CrossRef]
- Bellwood, D.R.; Hughes, T.P.; Hoey, A.S. Sleeping functional group drives coral-reef recovery. Curr. Biol. 2006, 16, 2434–2439. [Google Scholar] [CrossRef] [PubMed]
- Ban, S.S.; Graham, N.A.; Connolly, S.R. Evidence for multiple stressor interactions and effects on coral reefs. Global Chang. Biol. 2014, 20, 681–697. [Google Scholar] [CrossRef]
- Amar, K.O.; Rinkevich, B. A floating mid-water coral nursery as larval dispersion hub: Testing an idea. Mar. Biol. 2007, 151, 713–718. [Google Scholar] [CrossRef]
- Guest, J.R.; Baria, M.V.; Gomez, E.D.; Heyward, A.J.; Edwards, A.J. Closing the circle: Is it feasible to rehabilitate reefs with sexually propagated corals? Coral Reefs 2014, 33, 45–55. [Google Scholar] [CrossRef]
- Munday, P.L.; Leis, J.M.; Lough, J.M.; Paris, C.B.; Kingsford, M.J.; Berumen, M.L.; Lambrechts, J. Climate change and coral reef connectivity. Coral Reefs 2009, 28, 379–395. [Google Scholar] [CrossRef]
- Sammarco, P.W.; Atchison, A.D.; Boland, G.S.; Sinclair, J.; Lirette, A. Geographic expansion of hermatypic and ahermatypic corals in the Gulf of Mexico, and implications for dispersal and recruitment. J. Exp. Mar. Biol. Ecol. 2012, 436, 36–49. [Google Scholar] [CrossRef]
- Hobbs, R.J.; Cramer, V.A. Restoration ecology: Interventionist approaches for restoring and maintaining ecosystem function in the face of rapid environmental change. Ann. Rev. Environ. Resour. 2008, 33, 39–61. [Google Scholar] [CrossRef]
- Nevo, E.; Fu, Y.B.; Pavlicek, T.; Khalifa, S.; Tavasi, M.; Beiles, A. Evolution of wild cereals during 28 years of global warming in Israel. Proc. Natl. Acad. Sci. USA 2012, 109, 3412–3415. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rinkevich, B. Climate Change and Active Reef Restoration—Ways of Constructing the “Reefs of Tomorrow”. J. Mar. Sci. Eng. 2015, 3, 111-127. https://doi.org/10.3390/jmse3010111
Rinkevich B. Climate Change and Active Reef Restoration—Ways of Constructing the “Reefs of Tomorrow”. Journal of Marine Science and Engineering. 2015; 3(1):111-127. https://doi.org/10.3390/jmse3010111
Chicago/Turabian StyleRinkevich, Baruch. 2015. "Climate Change and Active Reef Restoration—Ways of Constructing the “Reefs of Tomorrow”" Journal of Marine Science and Engineering 3, no. 1: 111-127. https://doi.org/10.3390/jmse3010111
APA StyleRinkevich, B. (2015). Climate Change and Active Reef Restoration—Ways of Constructing the “Reefs of Tomorrow”. Journal of Marine Science and Engineering, 3(1), 111-127. https://doi.org/10.3390/jmse3010111






