1. Introduction
In 2018, greenhouse gas (GHG) emissions from shipping accounted for approximately 3% of global emissions. The International Maritime Organization (IMO) has imposed environmental regulations to control GHG emissions from the shipping industry. The 2023 IMO GHG Strategy was adopted at the 80th Marine Environment Protection Committee (MEPC) meeting in July 2023, raising the existing goal of reducing total emissions by 50% compared to 2008 levels by 2050. The committee agreed to achieve at least a 20% reduction by 2030, at least 70% by 2040, and zero net emissions by 2050 [
1]. The 2023 IMO GHG Strategy includes an ambition to use at least 5% carbon-free fuels by 2030, with efforts to reach 10%.
To achieve these objectives, various methods have been proposed for reducing CO
2 emissions from ships, such as improvements in hull design, enhancements in propulsion efficiency, operational measures (like reducing speed), and the utilization of alternative energy [
2]. Moreover, nitrogen oxide (NO
X) emissions are being regulated based on engine speed, as depicted in
Table 1 [
3]. Currently, NO
X emissions from ships meet regulations by applying the SCR system using Urea. However, merely implementing measures to improve technological and operational efficiency is not sufficient to meet the CO
2 reduction targets for ships [
4]. Furthermore, the need for long-term strategies is urgent, such as the obligatory use of zero-carbon fuels and a more rigorous approach to reducing carbon dioxide emissions. Therefore, a shift to alternative energy sources is essential [
5].
Researchers are considering various alternative energies to achieve the 2050 goal of zero carbon emissions, but the two most common carbon-free energies are hydrogen (H
2) and ammonia (NH
3) [
6]. Unlike heavy fuel oil (HFO), liquefied natural gas (LNG), and liquefied petroleum gas (LPG), the primary energy source for ships, the carbon reduction rates of hydrogen and ammonia are 100% [
7]. Liquid hydrogen (LH
2) has a higher low heating value (LHV) than other energies but requires a very low storage temperature of −253 °C at 1 bar [
7,
8], which creates high facility costs for use on board ships. However, the volumetric energy density of liquid ammonia (LNH
3; 14,100 MJ/m
3) is higher than that of liquid H
2 (8500 MJ/m
3). Additionally, the storage temperature of LNH
3 is −33.4 °C at a vapor pressure of 1 bar [
7,
8]. This is similar to the boiling temperature of LPG (−42 °C at 1 bar). Because of these properties, ammonia has technological advantages in storage and handling [
8].
Several organizations have published outlook reports on ammonia as a marine fuel. For instance, the International Energy Agency (IEA) [
9] forecasts that ammonia’s proportion of the fuel market will increase to 8% by 2030 and 44% by 2050, and it is anticipated to secure a significant share of the shipping fuel market. The American Bureau of Shipping (ABS) noted that using ammonia is a quick way to meet GHG emission regulations [
10]. Lloyd’s Register (LR) noted that ammonia regulations for ships using low-flashpoint fuel would take effect in July 2023 [
11]. The company DNV-GL released a report on ammonia as a marine fuel, emphasizing its potential role in the decarbonization of maritime transport [
12]. Additionally, the Korean Register (KR) published a technical report on ammonia-fueled ships and investigated their characteristics, safety, technology, and research trends with the goal of establishing future regulatory directions for ammonia-fueled ships. We also analyzed important international requirements, such as IGC and IGF CODE [
8]. Based on the low-flashpoint-fuel ship rules, design guidelines describing the latest safety regulations and inspection standards for ammonia-fueled ships were published [
13].
Two approaches are typically employed to use ammonia as a marine fuel: fuel cells and combustion-based systems [
14]. Utilizing ammonia in a fuel cell involves converting it into high-purity hydrogen through an external reformer. This hydrogen is then supplied to a polymer electrolyte membrane (PEM) fuel cell [
15], characterized by a low operating temperature, or directly to a solid oxide fuel cell (SOFC), which operates at a high temperature [
16]. A fuel cell generates electricity through an electrochemical reaction, unlike the combustion reactions in internal combustion (IC) engines. This method offers the advantage of producing no nitrogen oxide (NO
X) emissions. However, it faces challenges related to its relatively low technological maturity compared to IC engines, including additional onboard facility costs and its limited capacity to respond to changes in load [
17].
The second approach, combustion-based systems, has been studied for ammonia-fueled IC engines and fuel supply systems [
18]. The engine maker MAN E&S expects about 27% of the fuel used by large merchant ships to be ammonia by 2050. MAN E&S is developing an ammonia engine based on LPG-fueled engines to provide a carbon-free ammonia-fueled propulsion system as a marine solution. To operate NH
3 engines, the concept of the fuel supply system is being developed for supplying liquid NH
3 to the engines. The two-stroke engine ME-LGIA is being studied with the goal of delivery in 2025, and MAN B&W recently reported that successful ammonia combustion results were obtained for its two-stroke 4T50ME-X engine [
19]. In addition, Wartsila noted that ammonia fuels are promising as carbon-free fuels in the marine industry that can satisfy IMO regulations [
20]. To this end, the existing Wartsila 25 engine was developed to use ammonia fuel in a four-stroke engine [
21]. However, despite these studies, solving the problem of NOx emissions continues to be a challenge due to the combustion of ammonia [
22].
Kim et al. [
23] conducted a study using ammonia as a potential marine fuel, taking environmental and operational impacts into account. They proposed four propulsion systems, each suitable for a specific ship type that uses ammonia as fuel. To comprehensively evaluate the viability of these systems, they built a model ship system using five combinations of these propulsion techniques. The evaluation criteria included the economic feasibility of each union, GHG emissions, and operational efficiency. Their approach provided the potential to reduce the environmental impact of ammonia and cost-effectiveness in maritime operations. At the same time, they evaluated the economic feasibility of the system through net present value (NPV) calculations.
Lee et al. [
24] conducted a techno-economic analysis of an NH
3 fuel supply and onboard re-liquefaction system for NH
3-fueled ships. Through thermodynamic and economic evaluations, including exergy destruction and Net Present Value (NPV) analysis, the research identifies conditions under which NH
3 fuel becomes economically feasible, particularly with respect to NH
3 pricing and carbon tax implications. The findings suggest that NH
3 is a promising alternative marine fuel.
Seo et al. [
25] analyzed the impact of installing ammonia fuel tanks on ships and proposed two methods: cargo tanks on ammonia carriers and installing independent cylindrical tanks. Their study evaluated the economic feasibility of both methods, including an analysis of sales and life cycle costs. As a result, the practicality and economy of the installation methods were analyzed. The sensitivity analysis demonstrated that profits were significantly affected by the prices of NH
3 fuel and transportation costs.
Akturk M et al. [
14] investigated the feasibility of using NH
3 fuels in medium LPG/NH
3 carriers. Their approach included a comprehensive review of the existing literature, comparing various eco-friendly fuels and focusing on their characteristics and suitability for marine use. They evaluated the potential of ammonia as a marine fuel and conducted a risk assessment of using NH
3 as a ship fuel using two methods: the What-If Technique (SWIFT) and a Hazard Identification Study (HAZID). These methodologies systematically identified and evaluated potential operational risks associated with ammonia use.
Lesmana H et al. [
26] conducted an analysis on the use of ammonia as an alternative to carbon-containing fuels in the ship’s combustion engines. The thermochemical properties of conventional fuels and hydrogen and ammonia alternative fuels were compared, and the basic combustion properties and properties were summarized. In addition, it provides a theoretical basis for the general fuel system and NH
3 storage and handling system for evaluating NH
3 combustion in IC engines. Additionally, the feasibility was verified through research on combustion performance through hydrogen. NH
3 has potential as a fuel but mentioned the importance of controlling NO
X, particularly related to emissions, in practical applications of combustion technology.
For the conversion of potential ship eco-friendly fuels, various aspects of ammonia fuel are continuously being studied on ships, including international regulations for the storage and transportation of ammonia, potential risks, and operational safety [
27]. However, there is a lack of feasibility and economic research and analysis of BOG treatment systems, which are important solutions for NH
3-fueled ships.
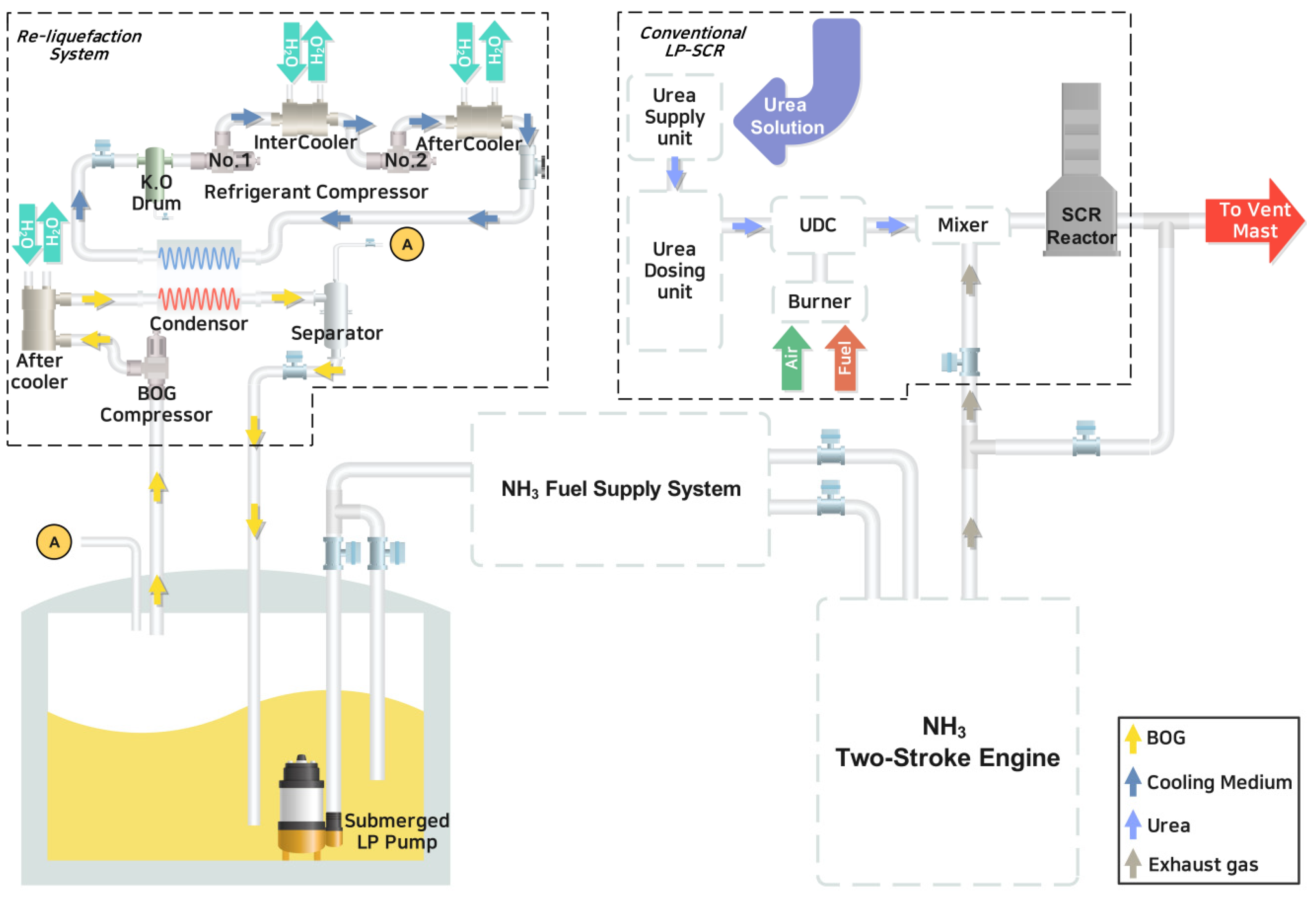
Therefore, in this study, a new preliminary study was conducted on BOG generated from ships using NH
3 as a sustainable fuel for ships. This presents a solution feasible with on-board re-liquefaction systems and LP-SCR to reduce economic loss from ships. Therefore, this study proposed the LP-SCR system, a novel approach for BOG processing that distinguishes it from existing methodologies in the literature concerning ammonia-fueled ships. This presents a new perspective on BOG processing systems for future NH
3-fueled ships. The direct LP-SCR system was quantitatively compared with the conventional LP-SCR system, considered a re-liquefaction system, from thermodynamic performance and economic perspectives. The rest of this paper is organized as follows:
Section 2 explains materials and methodologies for system design, analysis, and evaluation.
Section 3 introduces the results and discussion, and
Section 4 presents the conclusions of this study.
4. Conclusions
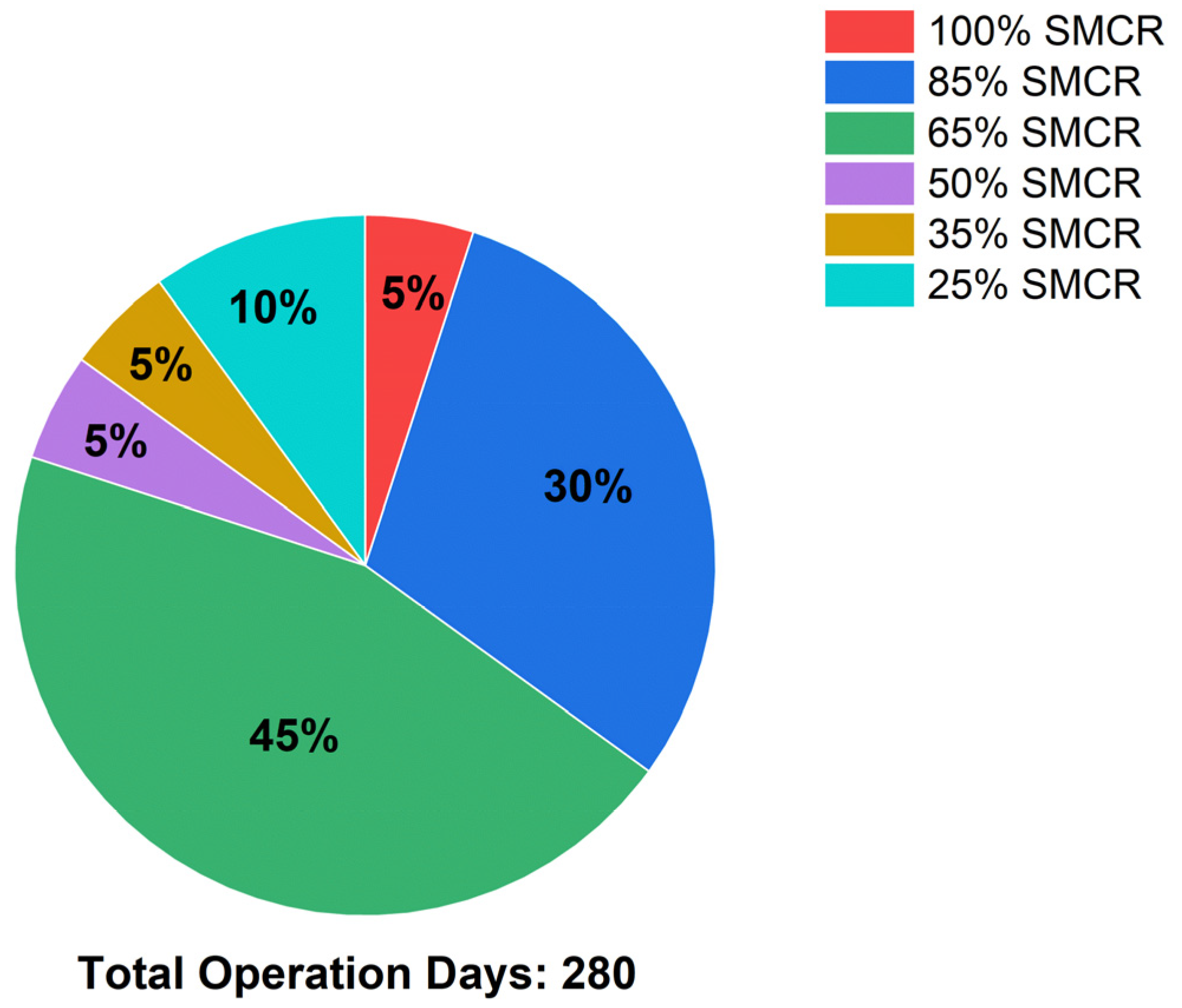
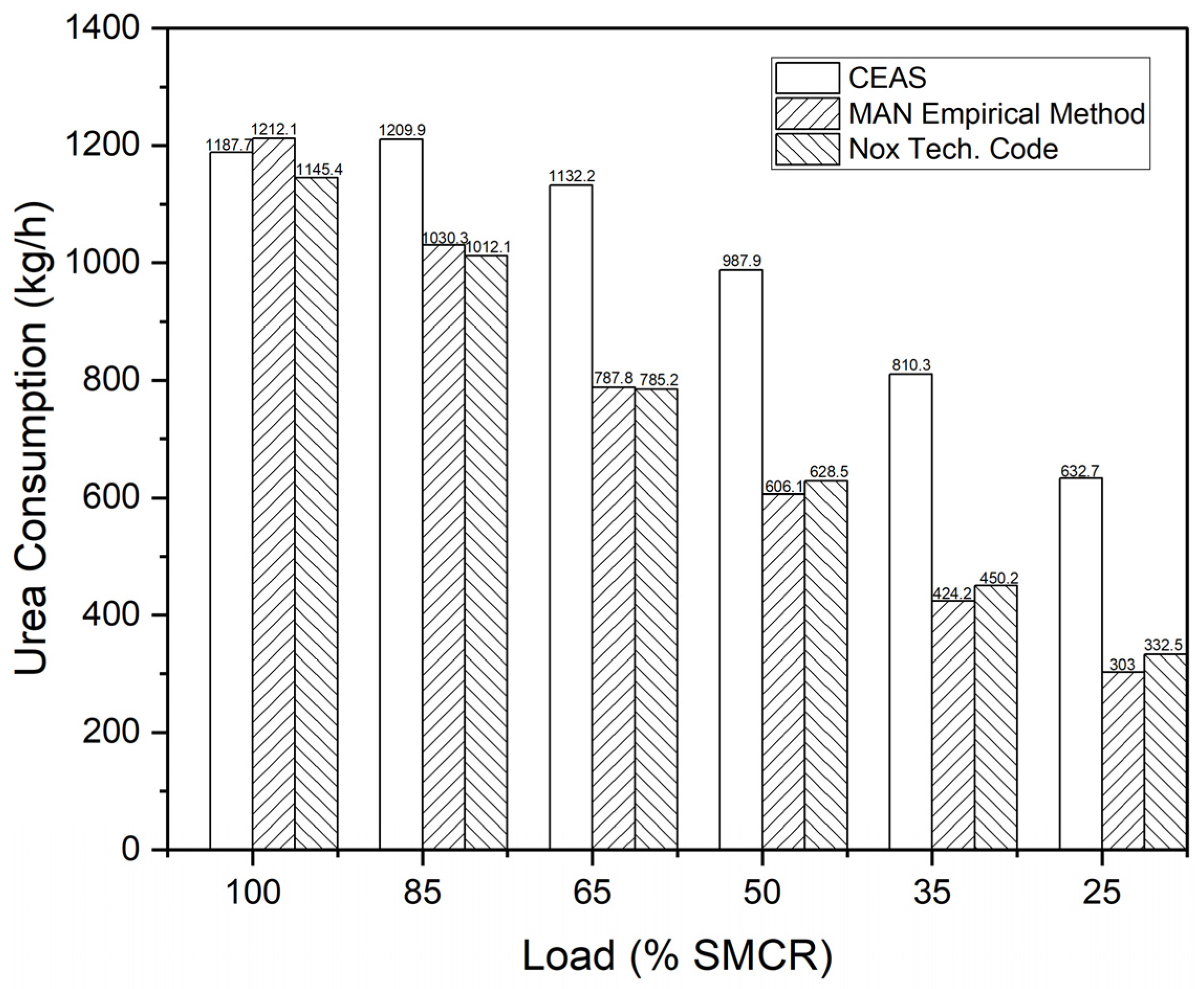
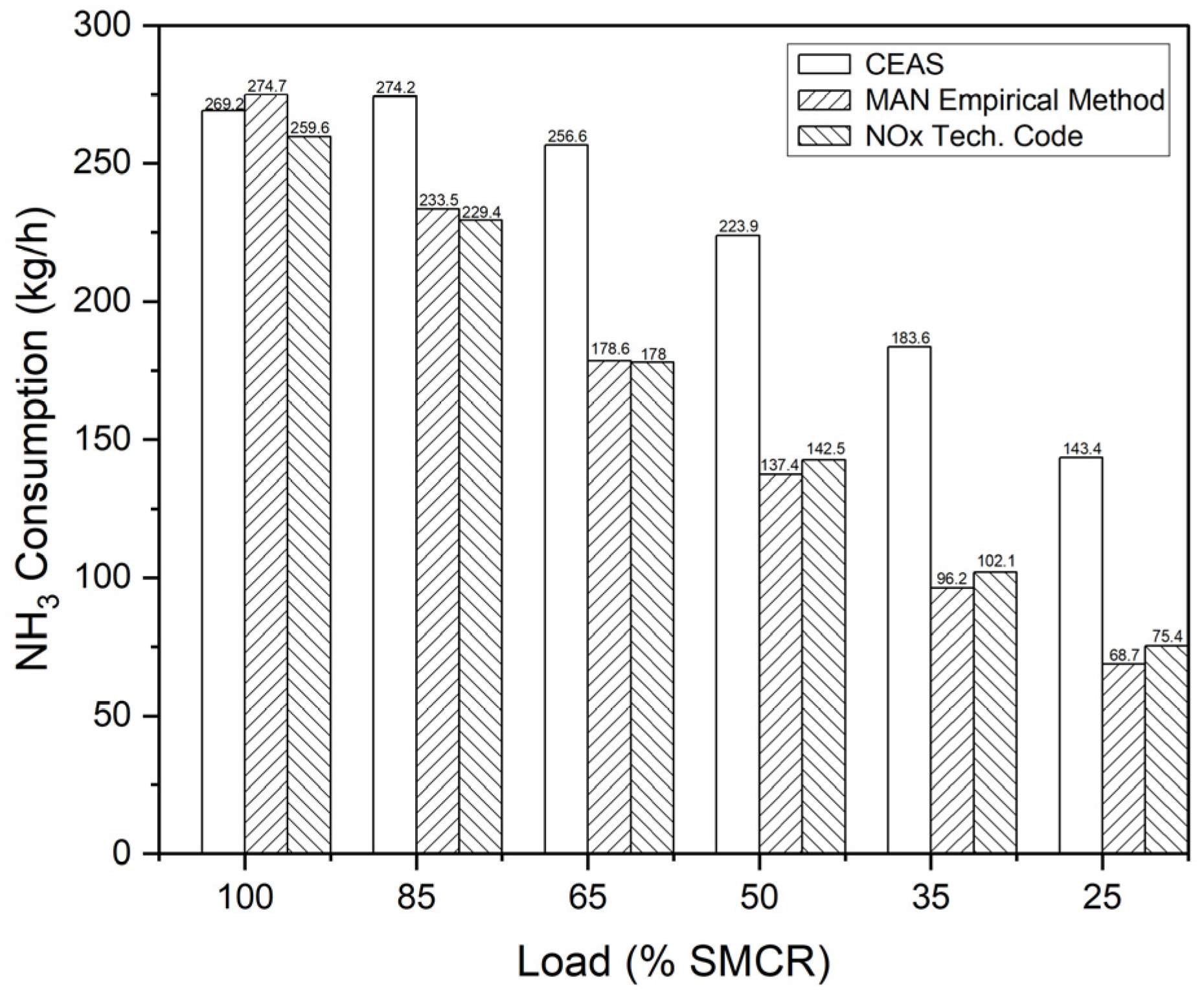
This study aims to address the treatment of BOG generated within the tanks of a ship utilizing NH3 as fuel. To this end, a re-liquefaction system, an existing SCR system, and an NH3 direct LP-SCR system were proposed and assessed for technological and economic feasibility. The re-liquefaction system operates on a steam-compression refrigeration cycle using NH3 as the refrigerant. The SEC of this system, 0.223 kWh/kg, was determined based on the power consumption of the three compressors to assess the energy utilized within the system. In addition, we conducted an economic analysis of the existing LP-SCR system integrated with the re-liquefaction system. Furthermore, the urea flow rate needed for NOX removal was computed using CEAS engine data, the MAN empirical method, and the NOX Technical Code, with costs estimated at USD 250 per ton. As the price of NH3 increases, the profitability of the re-liquefaction system also increases.
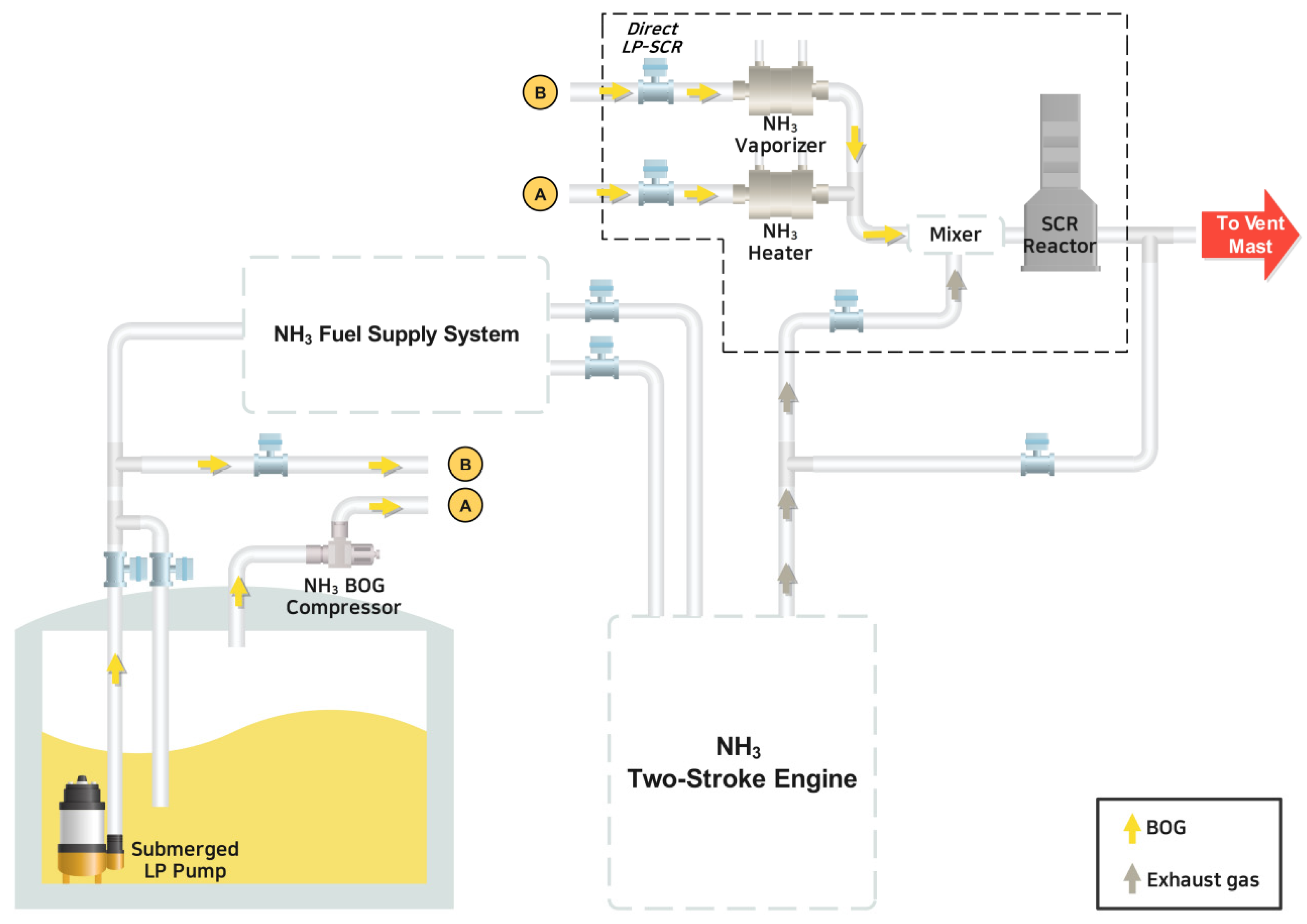
A direct LP-SCR system was proposed and analyzed for the treatment of BOG. In this system, NH3 is directly supplied as a reducing agent for NOX removal in the exhaust gas from the NH3 fuel tank. This is accomplished by boosting the BOG from 1.4 bar and −20 °C to 2 bar using a compressor. Alternatively, if the amount of NH3 is insufficient, liquid NH3 from the tank is supplied through a pump. The flow rate, adjusted according to the engine load, and the SEC were analyzed. According to MAN CEAS data, the most conservative method, the system consumes 1653 tons of NH3 annually. Additionally, system operation requires a compressor and a submerged pump, leading to an annual NH3 fuel consumption of 9.6 tons. Furthermore, through thermodynamic analysis, the costs of power generation and the reducing agent required by the system were evaluated in relation to the price of NH3. According to the LCC analysis, the direct LP-SCR system’s annual cost is more cost-effective when integrated with the re-liquefaction system.
As a result, the direct LP-SCR system offers relatively stable operational costs without any significant variability due to changes in NH3 prices for ships utilizing NH3 as fuel. Three methodologies were used to determine the necessary ammonia flow rate for the LP-SCR system to ensure reliable analysis results. Furthermore, in the NH3 direct LP-SCR system, since the need for core equipment is minimal, power consumption for supplying NH3, whether as a BOG or a liquid, is comparatively low. According to the LCC analysis, the NH3 direct LP-SCR system is more cost-effective than the combination of re-liquefaction and the existing SCR system. However, the initial capital cost for the existing LP-SCR system is higher when comparing CAPEX between the re-liquefaction system, the existing LP-SCR system, and the direct LP-SCR system. If only operating costs are considered, excluding initial capital costs, the re-liquefaction system is deemed economical when the price of NH3 exceeds USD 1500 per ton.
NH3 is emerging as a major energy source due to the strengthening environmental regulations. In the previous study, the study on NH3 BOG treatment proposed a BOG re-liquefaction system integrated with the fuel supply system in an ammonia propulsion ship. According to this study, the feasible solution is proposed for solving the problems about NOX emission and BOG treatment, which are main concerns of NH3-fueled ships. The direct LP-SCR system is an attractive solution for using NH3 fuel. It is applicable to the 14,000 TEU container ships considered in this study, as well as ships using internal combustion engines with NH3 fuel. However, further research is still needed to commercialize and optimize the BOG treatment system. We hope that the results of this study will be a useful reference for supporting the research and development of ships using NH3 as fuels.