Bisphenol Analogs in Aquatic Environments and Their Effects on Marine Species—A Review
Abstract
:1. Introduction
1.1. Bisphenol A Analogs Production and Usage
1.2. Biodegradation of Bisphenols
1.3. Occurrence of Bisphenols in Aquatic Environments
2. Effects of Bisphenols on Marine Species
2.1. Effects of Bisphenol A
2.2. Effects of Bisphenol A Analogs
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chen, D.; Kannan, K.; Tan, H.; Zheng, Z.; Feng, Y.L.; Wu, Y.; Widelka, M. Bisphenol analogues other than BPA: Environmental occurrence, human exposure, and toxicity—A review. Environ. Sci. Technol. 2016, 50, 5438–5453. [Google Scholar] [CrossRef] [PubMed]
- Rochester, J.R. Bisphenol A and human health: A review of the literature. Reprod. Toxicol. 2013, 42, 132–155. [Google Scholar] [CrossRef]
- Vandenberg, L.N.; Hauser, R.; Marcus, M.; Olea, N.; Welshons, W.V. Human exposure to bisphenol A (BPA). Reprod Toxicol. 2007, 24, 139–177. [Google Scholar] [CrossRef] [PubMed]
- Almeida, S.; Raposo, A.; Almeida-González, M.; Carrascosa, C. Bisphenol A: Food exposure and impact on human health. Compr. Rev. Food. Sci. Food. Saf. 2018, 17, 1503–1517. [Google Scholar] [CrossRef]
- Vandenberg, L.N.; Maffini, M.V.; Sonnenschein, C.; Rubin, B.S.; Soto, A.M. Bisphenol-A and the great divide: A review of controversies in the field of endocrine disruption. Endocr. Rev. 2009, 30, 75–95. [Google Scholar] [CrossRef]
- ECHA. Use of Bisphenol A and Its Alternatives in Thermal Paper in the EU—2018 Update; European Chemicals Agency: Helsinki, Finland, 2019. [Google Scholar]
- ECHA 2017A: ECHA (European Chemicals Agency). Inclusion of BPA as a Substance of Very High Concern (Reason for Inclusion: Toxic for Reproduction—Article 57c) in the Candidate List for Eventual Inclusion in Annex XIV. Decision of the European Chemicals Agency. 2017. Available online: https://echa.europa.eu/documents/10162/c11b5b68-67f4-8044-53a6-26759a106c80 (accessed on 11 August 2022).
- ECHA 2017 B: ECHA (European Chemicals Agency). Inclusion of BPA as a Substance of Very High Concern (Reason for Inclusion: Endocrine Disrupting Properties—Article 57f Human Health) in the Candidate List for Eventual Inclusion in Annex XIV. Decision of the European Chemicals Agency. 2017. Available online: https://echa.europa.eu/documents/10162/20a23653-34b1-bb48-4887-7ea77bedc637 (accessed on 11 August 2022).
- ECHA. Inclusion of BPA as a Substance of Very High Concern (Reason for Inclusion: Endocrine Disrupting Properties—Article 57f Environment) in the Candidate List for Eventual Inclusion in Annex XIV; Decision of the European Chemicals Agency; European Chemicals Agency: Helsinki, Finland, 2018. [Google Scholar]
- EU. Commission regulation 2016/2235 of 12 December 2016 amending annex XVII to regulation (EC) No 1907/2006 of the European parliament and of the council concerning the registration, evaluation, authorisation and restriction of chemicals (REACH) as regards bisphenol A. Off. J. Eur. Union 2016, L337, 3–5. [Google Scholar]
- ECHA. Assessment of Regulatory Needs. Version 1.0, 16 December 2021. Available online: https://echa.europa.eu/documents/10162/3448017/GMT_109_Bisphenols_Report_public_23502_en.pdf (accessed on 12 August 2022).
- Rochester, J.R.; Bolden, A.L. Bisphenol S and F: A systematic review and comparison of the hormonal activity of bisphenol A substitutes. Environ. Health Perspect. 2015, 123, 643–650. [Google Scholar] [CrossRef]
- Zhang, L.; Fang, P.; Yang, L.; Zhang, J.; Wang, X. Rapid method for the separation and recovery of endocrine-disrupting compound bisphenol AP from wastewater. Langmuir 2013, 29, 3968–3975. [Google Scholar] [CrossRef]
- Liu, A.F.; Qu, G.B.; Yu, M.; Liu, Y.W.; Shi, J.B.; Jiang, G.B. Tetrabromobisphenol-A/S and nine novel analogs in biological samples from the Chinese Bohai Sea: Implications for trophic transfer. Environ. Sci. Technol. 2016, 50, 4203–4211. [Google Scholar] [CrossRef]
- Usman, A.; Ikhlas, S.; Ahmad, M. Occurrence, toxicity and endocrine disrupting potential of Bisphenol-B and Bisphenol-F: A mini-review. Toxicol. Lett. 2019, 312, 222–227. [Google Scholar] [CrossRef]
- EFSA; FitzGerald, R.; Van Loveren, H.; Civitella, C.; Castoldi, A.F.; Giovanni Bernasconi, G. Assessment of New Information on Bisphenol S (BPS) Submitted in Response to the Decision under REACH Regulation (EC) No 1907/2006; EFSA Supporting Publication; European Food Safety Authority (EFSA): Parma, Italy, 2020; p. 39. [Google Scholar]
- Hu, Y.; Zhu, Q.; Yan, X.; Liao, C.; Jiang, G. Occurrence, fate and risk assessment of BPA and its substituents in wastewater treatment plant: A review. Environ. Res. 2019, 178, 108732. [Google Scholar] [CrossRef] [PubMed]
- ECHA. Bisphenol AF, Substance Infocard. Available online: https://echa.europa.eu/substance-information/-/substanceinfo/100.014.579 (accessed on 25 July 2022).
- Su, H.; Guan, G.; Ahmed, R.Z.; Lyu, L.; Li, Z.; Jin, X. TBBPA stimulated cell migration of endometrial cancer via the contribution of NOX-generated ROS in lieu of energy metabolism. J. Hazard. Mater. 2020, 400, 123204. [Google Scholar] [CrossRef] [PubMed]
- Russo, G.; Barbato, F.; Grumetto, L. Monitoring of bisphenol A and bisphenol S in thermal paper receipts from the Italian market and estimated transdermal human intake: A pilot study. Sci. Total Environ. 2017, 599, 68–75. [Google Scholar] [CrossRef]
- EU. Commission regulation (EC) No 1895/2005 of 18 November 2005 on the restriction of use of certain epoxy derivatives in materials and articles intended to come into contact with food. Off. J. Eur. Union 2005, L302, 28–32. [Google Scholar]
- Liu, X.; Shi, H.; Xie, B.; Dionysiou, D.D.; Zhao, Y. Microplastics as both a sink and a source of bisphenol A in the marine environment. Environ. Sci. Technol. 2019, 53, 10188–10196. [Google Scholar] [CrossRef]
- De Morais Farias, J.; Krepsky, N. Bacterial degradation of bisphenol A and its analogues: An overview. Res. Sq. 2022. [Google Scholar] [CrossRef]
- Ike, M.; Jin, C.S.; Fujita, M. Biodegradation of bisphenol A in the aquatic environment. Water Sci. Technol. 2000, 42, 31–38. [Google Scholar] [CrossRef]
- Zhang, W.; Yin, K.; Chen, L. Bacteria-Mediated bisphenol A degradation. Appl. Microbiol. Biotechnol. 2013, 97, 5681–5689. [Google Scholar] [CrossRef]
- Frankowski, R.; Zgoła-Grześkowiak, A.; Smułek, W.; Grześkowiak, T. Removal of bisphenol A and its potential substitutes by biodegradation. Appl. Biochem. Biotechnol. 2020, 191, 1100–1110. [Google Scholar] [CrossRef]
- Kang, J.H.; Kondo, F. Bisphenol A degradation in seawater is different from that in river water. Chemosphere 2005, 60, 1288–1292. [Google Scholar] [CrossRef]
- Ying, G.G.; Kookana, R.S. Degradation of five selected endocrine-disrupting chemicals in seawater and marine sediment. Envrion. Sci. Technol. 2003, 37, 1256–1260. [Google Scholar] [CrossRef]
- EU. European Union Updated Risk Assessment Report. Environment Addendum of April 2008 (To be Read in Conjunction with Published EU RAR of BPA, 2003) 4,4′-ISOPROPYLIDENEDIPHENOL (Bisphenol-A) Part 1 Environment; European Commission, Joint Research Centre, Institute for Health and Consumer Protection: Luxembourg, 2008. [Google Scholar]
- Zhao, X.; Qiu, W.; Zheng, Y.; Xiong, J.; Gao, C.; Hu, S. Occurrence, distribution, bioaccumulation, and ecological risk of bisphenol analogues, parabens and their metabolites in the Pearl River Estuary, South China. Ecotoxicol. Environ. Saf. 2019, 180, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Zhu, L. Occurrence and partitioning of bisphenol analogues in water and sediment from Liaohe River Basin and Taihu Lake, China. Water Res. 2016, 103, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Feng, Q.; Hu, G.; Gao, Z.; Zhu, X.; Epri, J.E. Simultaneous determination of seven bisphenol analogues in surface water by solid-phase extraction and ultra-performance liquid chromatography-tandem mass spectrometry. Microchem. J. 2022, 175, 107098. [Google Scholar] [CrossRef]
- Wang, Q.; Chen, M.; Shan, G.; Chen, P.; Cui, S.; Yi, S.; Zhu, L. Bioaccumulation and biomagnification of emerging bisphenol analogues in aquatic organisms from Taihu Lake, China. Sci. Total Environ. 2017, 598, 814–820. [Google Scholar] [CrossRef]
- Yamazaki, E.; Yamashita, N.; Taniyasu, S.; Lam, J.; Lam, P.K.; Moon, H.B.; Jeong, Y.; Kannan, P.; Achyuthan, H.; Munuswamy, N.; et al. Bisphenol A and other bisphenol analogues including BPS and BPF in surface water samples from Japan, China, Korea and India. Ecotoxicol. Environ. Saf. 2015, 122, 565–572. [Google Scholar] [CrossRef]
- Huang, C.; Wu, L.H.; Liu, G.Q.; Shi, L.; Guo, Y. Occurrence and ecological risk assessment of eight endocrine-disrupting chemicals in urban river water and sediments of South China. Arch. Environ. Contam. Toxicol. 2018, 75, 224–235. [Google Scholar] [CrossRef]
- Xie, J.; Zhao, N.; Zhang, Y.; Hu, H.; Zhao, M.; Jin, H. Occurrence and partitioning of bisphenol analogues, triclocarban, and triclosan in seawater and sediment from East China Sea. Chemosphere 2022, 287, 132218. [Google Scholar] [CrossRef]
- Zhao, N.; Hu, H.; Zhao, M.; Liu, W.; Jin, H. Occurrence of free-form and conjugated bisphenol analogues in marine organisms. Environ. Sci. Technol. 2021, 55, 4914–4922. [Google Scholar] [CrossRef]
- Guo, Y.; Yu, R.Q.; Zhang, L.; Liang, Y.; Liu, Z.; Sun, X.; Wu, Y. Cross-Generational Impacts of Diet Shift on Bisphenol Analogue Loads in Indo-Pacific Humpback Dolphins (Sousa chinensis). Environ. Sci. Technol. 2022, 56, 10764–10774. [Google Scholar] [CrossRef]
- Wang, H.; Liu, Z.H.; Zhang, J.; Huang, R.P.; Yin, H.; Dang, Z.; Wu, P.; Liu, Y. Insights into removal mechanisms of bisphenol A and its analogues in municipal wastewater treatment plants. Sci. Total Environ. 2019, 692, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Peteffi, G.P.; Fleck, J.D.; Kael, I.M.; Rosa, D.C.; Antunes, M.V.; Linden, R. Ecotoxicological risk assessment due to the presence of bisphenol A and caffeine in surface waters in the Sinos River Basin—Rio Grande do Sul—Brazil. Braz. J. Biol. 2019, 79, 712–721. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhang, Y.; Li, J.; Yang, M. Occurrence and exposure assessment of bisphenol analogues in source water and drinking water in China. Sci. Total Environ. 2019, 655, 607–613. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Liu, Y.; Yan, K.; Wu, S.; Han, Z.; Guo, R.; Chen, M.; Yang, Q.; Zhang, S.; Chen, J. Bisphenol analogues in surface water and sediment from the shallow Chinese freshwater lakes: Occurrence, distribution, source apportionment, and ecological and human health risk. Chemosphere 2017, 184, 318–328. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zhang, Y.; Feng, Q.; Hu, G.; Gao, Z.; Meng, Q.; Zhu, X. Occurrence, distribution, and risk assessment of bisphenol analogues in Luoma Lake and its inflow rivers in Jiangsu Province, China. Environ. Sci. Pollut. Res. 2022, 29, 1430–1445. [Google Scholar] [CrossRef]
- Ozhan, K.; Kocaman, E. Temporal and spatial distributions of bisphenol A in marine and freshwaters in Turkey. Arch. Environ. Contam. Toxicol. 2019, 76, 246–254. [Google Scholar] [CrossRef]
- Pojana, G.; Gomiero, A.; Jonkers, N.; Marcomini, A. Natural and synthetic endocrine disrupting compounds (EDCs) in water, sediment and biota of a coastal lagoon. Environ. Int. 2007, 33, 929–936. [Google Scholar] [CrossRef]
- Arditsoglou, A.; Voutsa, D. Occurrence and partitioning of endocrine-disrupting compounds in the marine environment of Thermaikos Gulf, Northern Aegean Sea, Greece. Mar. Pollut. Bull. 2012, 64, 2443–2452. [Google Scholar] [CrossRef]
- Basheer, C.; Lee, H.K.; Tan, K.S. Endocrine disrupting alkylphenols and bisphenol-A in coastal waters and supermarket seafood from Singapore. Mar. Pollut. Bull. 2004, 48, 1161–1167. [Google Scholar] [CrossRef]
- Bayen, S.; Zhang, H.; Desai, M.M.; Ooi, S.K.; Kelly, B.C. Occurrence and distribution of pharmaceutically active and endocrine disrupting compounds in Singapore’s marine environment: Influence of hydrodynamics and physical–chemical properties. Environ. Pollut. 2013, 182, 1–8. [Google Scholar] [CrossRef]
- Bayen, S.; Estrada, E.S.; Juhel, G.; Kit, L.W.; Kelly, B.C. Pharmaceutically active compounds and endocrine disrupting chemicals in water, sediments and mollusks in mangrove ecosystems from Singapore. Mar. Pollut. Bull. 2016, 109, 716–722. [Google Scholar] [CrossRef] [PubMed]
- Beck, I.C.; Bruhn, R.; Gandrass, J.; Ruck, W. Liquid chromatography–tandem mass spectrometry analysis of estrogenic compounds in coastal surface water of the Baltic Sea. J. Chromatogr. A 2005, 1090, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Tang, S.; Zhou, X.; Gao, R.; Liu, Z.; Song, X.; Zeng, F. Urinary concentrations of bisphenol analogues in the south of China population and their contribution to the per capital mass loads in wastewater. Environ. Res. 2022, 204, 112398. [Google Scholar] [CrossRef] [PubMed]
- Česen, M.; Ahel, M.; Terzić, S.; Heath, D.J.; Heath, E. The occurrence of contaminants of emerging concern in Slovenian and Croatian wastewaters and receiving Sava river. Sci. Total Environ. 2019, 650, 2446–2453. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Zhao, J.L.; Yang, Y.Y.; Jia, Y.W.; Zhang, Q.Q.; Chen, C.E.; Liu, Y.S.; Yang, B.; Xie, L.; Ying, G.G. Occurrence, mass loads and risks of bisphenol analogues in the Pearl River Delta region, South China: Urban rainfall runoff as a potential source for receiving rivers. Environ. Pollut. 2020, 263, 114361. [Google Scholar] [CrossRef]
- Kiejza, D.; Kotowska, U.; Polinska, W.; Karpinska, J. USAEME-GC/MS Method for Easy and Sensitive Determination of Nine Bisphenol Analogues in Water and Wastewater. Molecules 2022, 27, 4977. [Google Scholar] [CrossRef]
- Yin, J.; Meng, Z.; Zhu, Y.; Song, M.; Wang, H. Dummy molecularly imprinted polymer for selective screening of trace bisphenols in river water. Anal. Methods 2011, 3, 173–180. [Google Scholar] [CrossRef]
- Lalwani, D.; Ruan, Y.; Taniyasu, S.; Yamazaki, E.; Kumar, N.J.; Lam, P.K.; Wang, X.; Yamashita, N. Nationwide distribution and potential risk of bisphenol analogues in Indian waters. Ecotoxicol. Environ. Saf. 2020, 200, 110718. [Google Scholar] [CrossRef]
- Caban, M.; Stepnowski, P. The quantification of bisphenols and their analogues in wastewaters and surface water by an improved solid-phase extraction gas chromatography/mass spectrometry method. Environ. Sci. Pollut. Res. 2020, 27, 28829–28839. [Google Scholar] [CrossRef]
- Chiriac, F.L.; Paun, I.; Pirvu, F.; Pascu, L.F.; Galaon, T. Occurrence and Fate of Bisphenol A and its Congeners in Two Wastewater Treatment Plants and Receiving Surface Waters in Romania. Environ. Toxicol. Chem. 2021, 40, 435–446. [Google Scholar] [CrossRef]
- Wilkinson, J.L.; Hooda, P.S.; Swinden, J.; Barker, J.; Barton, S. Spatial distribution of organic contaminants in three rivers of Southern England bound to suspended particulate material and dissolved in water. Sci. Total Environ. 2017, 593, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Wang, S.; Liu, H.; Yan, Z. Tetrabromobisphenol A: Tissue distribution in fish, and seasonal variation in water and sediment of Chaohu Lake, China. Environ. Sci. Pollut. Res. 2012, 19, 4090–4096. [Google Scholar] [CrossRef] [PubMed]
- Harrad, S.; Abdallah, M.A.E.; Rose, N.L.; Turner, S.D.; Davidson, T.A. Current-Use brominated flame retardants in water, sediment, and fish from English lakes. Environ. Sci. Technol. 2009, 43, 9077–9083. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Liu, H.; Wu, J.; Yuan, L.; Wang, Y.; Du, X.; Wang, R.; Marwa, P.W.; Petlulu, P.; Chen, X.; et al. The adverse health effects of bisphenol A and related toxicity mechanisms. Environ. Res. 2019, 176, 108575. [Google Scholar]
- Aarab, N.; Lemaire-Gony, S.; Unruh, E.; Hansen, P.D.; Larsen, B.K.; Andersen, O.K.; Narbonne, J.F. Preliminary study of responses in mussel (Mytilus edilus) exposed to bisphenol A, diallyl phthalate and tetrabromodiphenyl ether. Aquat. Toxicol. 2006, 78, S86–S92. [Google Scholar] [CrossRef]
- Park, K.; Jo, H.; Kim, D.K.; Kwak, I.S. Environmental pollutants impair transcriptional regulation of the vitellogenin gene in the burrowing mud crab (Macrophthalmus japonicus). Appl. Sci. 2019, 9, 1401. [Google Scholar] [CrossRef]
- Park, K.; Kim, W.S.; Kwak, I.S. Characterization and transcriptional response of ecdysone receptor gene in the mud crab Macrophthalmus japonicus: Effects of osmotic stress and endocrine disrupting chemicals. Ocean. Sci. J. 2019, 54, 611–620. [Google Scholar] [CrossRef]
- Han, Y.; Shi, W.; Tang, Y.; Zhou, W.; Sun, H.; Zhang, J.; Yan, M.; Hu, L.; Liu, G. Microplastics and bisphenol A hamper gonadal development of whiteleg shrimp (Litopenaeus vannamei) by interfering with metabolism and disrupting hormone regulation. Sci. Total Environ. 2022, 810, 152354. [Google Scholar] [CrossRef]
- Kiyomoto, M.; Kikuchi, A.; Unuma, T.; Yokota, Y. Effects of ethynylestradiol and bisphenol A on the development of sea urchin embryos and juveniles. Mar. Biol. 2006, 149, 57–63. [Google Scholar] [CrossRef]
- Özlem, Ç.A.; Hatice, P. Effects of bisphenol A on the embryonic development of sea urchin (Paracentrotus lividus). Environ. Toxicol. 2008, 23, 387–392. [Google Scholar] [CrossRef]
- Mercurio, S.; Messinetti, S.; Barzaghi, B.; Pennati, R. Comparing the sensitivity of two cogeneric ascidian species to two plastic additives: Bisphenol A and the flame retardant tris (chloro-propyl) phosphate. Eur. Zool. J. 2022, 89, 437–445. [Google Scholar]
- Miglioli, A.; Balbi, T.; Besnardeau, L.; Dumollard, R.; Canesi, L. Bisphenol A interferes with first shell formation and development of the serotoninergic system in early larval stages of Mytilus galloprovincialis. Sci. Total Environ. 2021, 758, 144003. [Google Scholar] [CrossRef] [PubMed]
- Balbi, T.; Franzellitti, S.; Fabbri, R.; Montagna, M.; Fabbri, E.; Canesi, L. Impact of bisphenol A (BPA) on early embryo development in the marine mussel Mytilus galloprovincialis: Effects on gene transcription. Environ. Pollut. 2016, 218, 996–1004. [Google Scholar] [PubMed]
- Zhou, J.; Zhu, X.S.; Cai, Z.H. The impacts of bisphenol A (BPA) on abalone (Haliotis diversicolor supertexta) embryonic development. Chemosphere 2011, 82, 443–450. [Google Scholar]
- Arslan, O.C.; Parlak, H. Effects of bisphenol-A on the embryological development of the sea urchin Arbacia Lixula (Linnaeus, 1758). Fresenius Environ. Bull. 2008, 17, 127. [Google Scholar]
- George, O.; Bryant, B.K.; Chinnasamy, R.; Corona, C.; Arterburn, J.B.; Shuster, C.B. Bisphenol A directly targets tubulin to disrupt spindle organization in embryonic and somatic cells. ACS Chem. Biol. 2008, 3, 167–179. [Google Scholar]
- Canesi, L.; Betti, M.; Lorusso, L.C.; Ciacci, C.; Gallo, G. ‘In vivo’ effects of Bisphenol A in Mytilus hemocytes: Modulation of kinase-mediated signalling pathways. Aquat. Toxicol. 2005, 71, 73–84. [Google Scholar]
- Tang, Y.; Zhou, W.; Sun, S.; Du, X.; Han, Y.; Shi, W.; Liu, G. Immunotoxicity and neurotoxicity of bisphenol A and microplastics alone or in combination to a bivalve species, Tegillarca granosa. Environ. Pollut. 2020, 265, 115115. [Google Scholar]
- Minier, C.; Forget-Leray, J.; Bjørnstad, A.; Camus, L. Multixenobiotic resistance, acetyl-choline esterase activity and total oxyradical scavenging capacity of the Arctic spider crab, Hyas araneus, following exposure to bisphenol A, tetra bromo diphenyl ether and diallyl phthalate. Mar. Pollut. Bull. 2008, 56, 1410–1415. [Google Scholar]
- Peng, Y.Q.; Wang, M.J.; Chen, H.G.; Chen, J.H.; Gao, H.; Huang, H.H. Immunological responses in haemolymph and histologic changes in the hepatopancreas of Charybdis japonica (A. Milne-Edwards, 1861) (Decapoda: Brachyura: Portunidae) exposed to bisphenol A. J. Crustac. Biol. 2018, 38, 489–496. [Google Scholar] [CrossRef] [Green Version]
- Park, K.; Kwak, I.S. Characterize and gene expression of heat shock protein 90 in marine crab Charybdis japonica following bisphenol A and 4-nonylphenol exposures. Environ. Health Toxicol. 2014, 29, e2014002. [Google Scholar] [PubMed]
- Canesi, L.; Borghi, C.; Ciacci, C.; Fabbri, R.; Vergani, L.; Gallo, G. Bisphenol-A alters gene expression and functional parameters in molluscan hepatopancreas. Mol. Cell. Endocrinol. 2007, 276, 36–44. [Google Scholar] [PubMed]
- Juhel, G.; Bayen, S.; Goh, C.; Lee, W.K.; Kelly, B.C. Use of a suite of biomarkers to assess the effects of carbamazepine, bisphenol A, atrazine, and their mixtures on green mussels, Perna viridis. Environ. Toxicol. Chem. 2017, 36, 429–441. [Google Scholar] [PubMed]
- Ruberto, S.; Buono, D.; Santovito, A. Polychaetes as bioindicators of environmental Pollution: Impact of bisphenol A on the reproduction rate of Ophryotrocha diadema (Åkesson, 1976) (eunicida: Dorvilleidae). Zool. Ecol. 2021, 31, 61–65. [Google Scholar]
- Zhao, H.; Zhou, Y.; Li, Y.; Li, S.; Yang, D. Molecular cloning and expression of the gene for G protein alpha subunit induced by bisphenol A in marine polychaete Perinereis aibuhitensis. Environ. Toxicol. Pharmacol. 2014, 37, 521–528. [Google Scholar] [PubMed]
- Okuda, K.; Takiguchi, M.; Yoshihara, S. In vivo estrogenic potential of 4- methyl-2,4-bis(4-hydroxyphenyl)pent-1-ene, an active metabolite of bisphenol A, in uterus of ovariectomized rat. Toxicol. Lett. 2010, 197, 7–11. [Google Scholar]
- Yoshihara, S.; Mizutare, T.; Makishima, M.; Suzuki, N.; Fujimoto, N.; Igarashi, K.; Ohta, S. Potent estrogenic metabolites of bisphenol A and bisphenol B formed by rat liver S9 fraction: Their structures and estrogenic potency. Toxicol. Sci. 2004, 78, 50–59. [Google Scholar] [CrossRef]
- Park, J.C.; Lee, M.C.; Yoon, D.S.; Han, J.; Kim, M.; Hwang, U.K.; Jung, J.H.; Lee, J.S. Effects of bisphenol A and its analogs bisphenol F and S on life parameters, antioxidant system, and response of defensome in the marine rotifer Brachionus koreanus. Aquat. Toxicol. 2018, 199, 21–29. [Google Scholar]
- In, S.; Yoon, H.W.; Yoo, J.W.; Cho, H.; Kim, R.O.; Lee, Y.M. Acute toxicity of bisphenol A and its structural analogues and transcriptional modulation of the ecdysone-mediated pathway in the brackish water flea Diaphanosoma celebensis. Ecotoxicol. Environ. Saf. 2019, 179, 310–317. [Google Scholar]
- In, S.; Cho, H.; Lee, Y.M. Identification of ecdysteroid pathway-related genes and their transcriptional modulation in the brackish water flea Diaphanosoma celebensis exposed to bisphenol analogs. Toxicol. Environ. Health. Sci. 2021, 13, 261–268. [Google Scholar]
- In, S.; Cho, H.; Lee, K.W.; Won, E.J.; Lee, Y.M. Cloning and molecular characterization of estrogen-related receptor (ERR) and vitellogenin genes in the brackish water flea Diaphanosoma celebensis exposed to bisphenol A and its structural analogues. Mar. Pollut. Bull. 2020, 154, 111063. [Google Scholar]
- Raj, A.; Nair, S.N.; Abdulvahab, R.; Ittoop, G. In-Silico Modelling of Interaction Between Environmental Xenoestrogens and Estrogen Receptor of Pacific Oyster (Magallana gigas [Thunberg, 1793]) Using AutoDock. Inform. Stud. 2022, 9. [Google Scholar]
- Omedes, S.; Andrade, M.; Escolar, O.; Villanueva, R.; Freitas, R.; Solé, M. B-Esterases characterisation in the digestive tract of the common octopus and the European cuttlefish and their in vitro responses to contaminants of environmental concern. Environ. Res. 2022, 210, 112961. [Google Scholar] [PubMed]
- Sole, M.; Bassols, A.; Labrada-Martagón, V. Plasmatic B-esterases as potential biomarkers of exposure to marine plastics in loggerhead turtles. Environ. Res. 2022, 213, 113639. [Google Scholar] [PubMed]
- Cosentino, S.; Aureli, F.; Iannilli, V. Bisphenols A and its analogues induce genotoxic damage in marine and freshwater amphipods. Environ. Adv. 2022, 7, 100183. [Google Scholar]
- Frenzilli, G.; Martorell-Ribera, J.; Bernardeschi, M.; Scarcelli, V.; Jönsson, E.; Diano, N.; Moggio, M.; Guidi, P.; Sturve, J.; Asker, N. Bisphenol A and bisphenol S induce endocrine and chromosomal alterations in brown trout. Front. Endocrinol. 2021, 12, 645519. [Google Scholar]
- Kaptaner, B.; Yılmaz, C.; Aykut, H.; Doğan, E.; Fidan, C.; Bostancı, M.; Yıldız, F. Bisphenol S leads to cytotoxicity-induced antioxidant responses and oxidative stress in isolated rainbow trout (Oncorhyncus mykiss) hepatocytes. Mol. Biol. Rep. 2021, 48, 7657–7666. [Google Scholar] [PubMed]
- Aykut, H.; Kaptaner, B. In vitro effects of bisphenol F on antioxidant system indicators in the isolated hepatocytes of rainbow trout (Oncorhyncus mykiss). Mol. Biol. Rep. 2021, 48, 2591–2599. [Google Scholar]
- Jung, J.H.; Moon, Y.S.; Kim, B.M.; Lee, Y.M.; Kim, M.; Rhee, J.S. Comparative analysis of distinctive transcriptome profiles with biochemical evidence in bisphenol S-and benzo [a] pyrene-exposed liver tissues of the olive flounder Paralichthys olivaceus. PLoS ONE 2018, 13, e0196425. [Google Scholar]
- Li, X.; Liu, Y.; Chen, Y.; Song, X.; Chen, X.; Zhang, N.; Li, H.; Guo, Y.; Wang, Z.; Dong, Z. Long-Term exposure to bisphenol A and its analogues alters the behavior of marine medaka (Oryzias melastigma) and causes hepatic injury. Sci. Total Environ. 2022, 841, 156590. [Google Scholar]
- Balbi, T.; Vezzulli, L.; Lasa, A.; Pallavicini, A.; Canesi, L. Insight into the microbial communities associated with first larval stages of Mytilus galloprovincialis: Possible interference by estrogenic compounds. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2020, 237, 108833. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, Y.; Li, N.; He, Y.; Xiao, H.; Fang, D.; Chen, C. Toxic Effects of Bisphenol A and Bisphenol S on Chlorella pyrenoidosa under Single and Combined Action. Int. J. Environ. Res. Public Health 2022, 19, 4245. [Google Scholar] [CrossRef] [PubMed]
| Name (Abbreviation) | CAS Number | Structural Formula | Log Kow | Molecular Weight (g/mol) |
|---|---|---|---|---|
| Bisphenol A (BPA) | 80-05-7 | 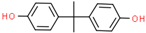 | 3.32 | 228.29 |
| Bisphenol AF (BPAF) | 1478-61-1 | 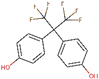 | 3.69 | 336.23 |
| Bisphenol AP (BPAP) | 1571-75-1 | 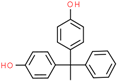 | 4.38 | 290.36 |
| Bisphenol B (BPB) | 77-40-7 | 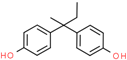 | 3.95 | 242.31 |
| Bisphenol BP (BPBP) | 1844-01-5 | 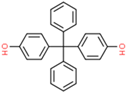 | 4.96 | 352.43 |
| Bisphenol C (BPC) | 79-97-0 | 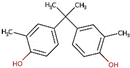 | 4.32 | 256.34 |
| Bisphenol E (BPE) | 2081-08-5 | 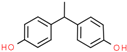 | 3.12 | 214.26 |
| Bisphenol F (BPF) | 620-92-8 | 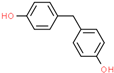 | 2.91 | 200.23 |
| Bisphenol FL (BPFL) | 3236-71-3 | 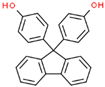 | 4.90 | 350.42 |
| Bisphenol G (BPG) | 127-54-8 | 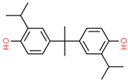 | 6.04 | 312.45 |
| Bisphenol M (BPM) | 13595-25-0 |  | 6.10 | 346.46 |
| Bisphenol P (BPP) | 2167-51-3 |  | 6.10 | 346.46 |
| Bisphenol PH (BPPH) | 24038-68-4 | 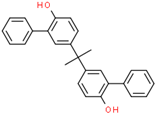 | 6.59 | 380.48 |
| Bisphenol S (BPS) | 80-09-1 | 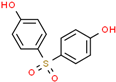 | 1.29 | 250.27 |
| Bisphenol TMC (BPTMC) | 129188-99-4 | 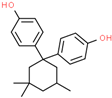 | 5.87 | 310.43 |
| Bisphenol Z (BPZ) | 843-55-0 | 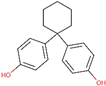 | 4.44 | 268.35 |
| Compound | Body Water, (Country) | Min–Max Concentration (Mean) (ng/L) | Reference |
|---|---|---|---|
| BPA | WWTP influent (China) | 3–62,010 (mean = 2031) | [39] |
| Surface water (Japan) | 3.1–120 | [34] | |
| Surface water (Korea) | 1.0–272 | [34] | |
| Surface water (China) | ND–98 | [34] | |
| Surface water (Brazil) | ND–517 | [40] | |
| Surface water (India) | ND–1950 | [34] | |
| Surface water (China) | 22.9–3360 | [33] | |
| Surface water (China) | ND–34.9 (mean = 12.8) | [41] | |
| Surface water (China) | 4.2–141 | [31] | |
| Surface water (China) | 28–560 | [42] | |
| Surface water (China) | 78.9–310 | [43] | |
| Surface water (Turkey) | 4620–29,920 | [44] | |
| Surface water (China) | 75.6–7480 (mean = 922) | [35] | |
| Seawater (Italy) | ND–145 | [45] | |
| Seawater (China) | 9.48–173 | [30] | |
| Seawater (Turkey) | 4160–16,920 | [44] | |
| Seawater (East China Sea) | 2.3–49 (mean = 18) | [37] | |
| Seawater (Tokyo Bay) | ND–431 (mean = 325) | [34] | |
| Seawater (Greece) | 10.6–52.3 (mean = 25) | [46] | |
| Seawater (Singapore) | ND–2470 | [47] | |
| Seawater (Singapore) | ND–694 | [48] | |
| Seawater (Singapore) | 6–1493 | [49] | |
| Seawater (Baltic Sea) | ND–5.7 | [50] | |
| Seawater (East China Sea) | 2.7–52 (mean = 23) | [36] | |
| BPAF | WWTP influent (China) | ND–9 (mean = 2) | [39] |
| WWTP (China) | 6.6–160 (mean = 17) | [51] | |
| WWTP (Slovenia–Croatia) | 0.0367–3.4 (mean = 1.47) | [52] | |
| WWTP (China) | ND–18.5 | [53] | |
| Surface water (China) | ND–2.58 (mean = 1.01) | [53] | |
| Surface water (China) | mean=140 ng/L | [33] | |
| Surface water (China) | ND–10.8 (mean = 3) | [41] | |
| Surface water (China) | 0.13–11 | [31] | |
| Surface water (China) | 0.7–84 | [42] | |
| Seawater (South China) | 0.40–3.59 | [30] | |
| Seawater (East China Sea) | 0.12–0.91 (mean = 0.21) | [36] | |
| Seawater (East China Sea) | ND–0.57 (mean = 0.24) | [37] | |
| BPAP | WWTP influent (China) | 1.1–75 (mean = 26) | [39] |
| WWTP (China) | ND–21 | [51] | |
| Surface water (Slovenia–Croatia) | 0.54–0.903 (mean = 0.704) | [52] | |
| Surface water (China) | ND–0.39 | [31] | |
| Surface water (China) | 1–56 | [42] | |
| BPB | WWTP (Slovenia–Croatia) | 27.1 | [52] |
| WWTP influent (China) | 1–8 (mean = 4) | [39] | |
| WWTP (China) | ND–8 (mean = 2.2) | [51] | |
| WWTP (Poland) | 29.29–62.49 | [54] | |
| Surface water (China) | ND–14.3 (mean = 1.0) | [41] | |
| Surface water (China) | ND–28 | [42] | |
| Surface water (China) | ND–7.9 | [43] | |
| Surface water (China) | ND–5.7 | [32] | |
| Seawater (South China) | 0.17–13.1 | [30] | |
| BPBP | WWTP (China) | ND–0.21 | [53] |
| Surface water (China) | ND–0.43 | [53] | |
| BPC | WWTP (Poland) | ND–7.57 | [54] |
| WWTP influent (China) | 6 | [39] | |
| WWTP (China) | ND–360 (mean = 68) | [51] | |
| WWTP (China) | ND–0.38 | [53] | |
| BPE | WWTP (Slovenia–Croatia) | 476 | [52] |
| WWTP influent (China) | 2–84 (mean = 16) | [39] | |
| WWTP (Poland) | 25.16–58.71 | [54] | |
| WWTP (China) | ND–31 (mean = 16) | [51] | |
| WWTP (China) | ND–7.71 | [53] | |
| Surface water (China) | ND–2.69 | [53] | |
| Surface water (China) | ND–6.18 (mean = 0.98) | [41] | |
| Surface water (China) | ND–20.3 | [55] | |
| BPF | WWTP (Slovenia–Croatia) | 2.54–117 (mean = 44.3) | [52] |
| WWTP influent (China) | 3–90 (mean = 39) | [39] | |
| Wastewater (India) | ND–333 | [56] | |
| WWTP (China) | ND–180 (mean = 26) | [51] | |
| WWTP (China) | 0.52–271 | [53] | |
| Surface water (Japan) | 76–2850 | [34] | |
| Surface water (China) | 0.24–34.4 | [53] | |
| Surface water (Korea) | ND–1300 | [34] | |
| Surface water (China) | ND–1110 | [34] | |
| Surface water (India) | ND–289 | [34] | |
| Surface water (India) | ND–209 | [56] | |
| Surface water (China) | 21.3–230 | [43] | |
| Surface water (China) | ND–12.56 (mean = 2.18) | [41] | |
| Surface water (China) | ND–474 (mean = 82.8) | [35] | |
| Surface water (China) | ND–5.6 (mean = 0.83) | [31] | |
| Surface water (China) | ND–1600 | [42] | |
| Seawater (East China Sea) | ND–0.65 (mean = 0.31) | [37] | |
| Seawater (South China) | 2.37–282 ng/L | [30] | |
| Seawater (East China Sea) | ND–0.91 | [36] | |
| Seawater (Tokyo Bay) | ND–1470 (mean = 373) | [34] | |
| BPFL | Surface water (China) | ND–0.069 | [31] |
| Surface water (China) | ND–2.21 | [30] | |
| BPG | WWTP (Poland) | ND–33.08 | [54] |
| WWTP (China) | ND–1.76 | [53] | |
| Surface water (China) | ND–2.47 | [53] | |
| BPM | Seawater (East China Sea) | ND–0.74 | [36] |
| BPP | WWTP influent (China) | 1.5–27 (mean = 8) | [39] |
| WWTP (China) | 2.7–300 (mean = 17) | [51] | |
| Surface water (China) | ND–1.93 | [53] | |
| Surface water (Slovenia–Croatia) | 6.45 | [52] | |
| Surface water (China) | 0.27–1.53 | [30] | |
| BPPH | WWTP (China) | ND–0.38 | [53] |
| Surface water (China) | ND–0.68 | [53] | |
| BPTMC | WWTP (China) | 0.09–5.3 | [53] |
| Surface water (China) | ND–101 (mean=8.8) | [53] | |
| BPS | WWTP (Slovenia–Croatia) | 108–435 (mean = 316) | [52] |
| WWTP (India) | ND–438 | [56] | |
| WWTP influent (China) | 7–318 (mean = 54) | [39] | |
| WWTP (China) | 90–1100 (mean = 290) | [51] | |
| WWTP (China) | 0.10–932 | [53] | |
| Surface water (Japan) | ND–8.7 | [34] | |
| Surface water (China) | 0.07–133 (mean = 12.7) | [53] | |
| Surface water (Korea) | ND–42 | [34] | |
| Surface water (China) | ND–135 | [34] | |
| Surface water (China) | 19.9–65,600 (mean = 3720) | [35] | |
| Surface water (India) | ND–7200 | [34] | |
| Surface water (Slovenia–Croatia) | 1.68–35.2 (mean = 9) | [52] | |
| Surface water (China) | mean= 27.6 ng/L | [33] | |
| Surface water (India) | ND–341 | [56] | |
| Surface water (Poland) | ND–1584 | [57] | |
| Surface water (Romania) | 6.15–8.23 | [58] | |
| Surface water (England) | ND–306 | [59] | |
| Surface water (China) | ND–5.2 (mean = 1.1) | [41] | |
| Surface water (China) | 0.22–67 | [31] | |
| Surface water (China) | ND–1600 | [42] | |
| Surface water (China) | 3.2–7.8 | [32] | |
| Seawater (East China Sea) | 0.15–12 (mean = 2.2) | [36] | |
| Seawater (South China) | 1.6–59.8 | [30] | |
| Seawater (East China Sea) | 0.12–11 (mean = 3.7) | [37] | |
| Seawater (Tokyo Bay) | ND–15 (mean = 8.5) | [34] | |
| BPZ | WWTP (China) | ND–540 (mean=7) | [51] |
| WWTP (Poland) | 24.64–66.62 | [54] | |
| WWTP influent (China) | 3–151 (mean = 77) | [39] | |
| WWTP (China) | ND–1.15 | [53] | |
| Surface water (Slovenia–Croatia) | 0.25–9.11 (mean = 4.68) | [52] | |
| Surface water (China) | ND–1.09 | [53] | |
| Surface water (China) | ND–0.70 (mean = 0.054) | [31] | |
| Surface water (China) | ND–45 | [42] | |
| Surface water (China) | ND–2.8 | [32] | |
| TBBPA | Surface water (China) | 23.9–224 | [55] |
| Surface water (China) | ND–4870 | [60] | |
| Surface water (England) | 0.14–3.2 | [61] | |
| Seawater (East China Sea) | 0.25–25 (mean = 2.3) | [36] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fabrello, J.; Matozzo, V. Bisphenol Analogs in Aquatic Environments and Their Effects on Marine Species—A Review. J. Mar. Sci. Eng. 2022, 10, 1271. https://doi.org/10.3390/jmse10091271
Fabrello J, Matozzo V. Bisphenol Analogs in Aquatic Environments and Their Effects on Marine Species—A Review. Journal of Marine Science and Engineering. 2022; 10(9):1271. https://doi.org/10.3390/jmse10091271
Chicago/Turabian StyleFabrello, Jacopo, and Valerio Matozzo. 2022. "Bisphenol Analogs in Aquatic Environments and Their Effects on Marine Species—A Review" Journal of Marine Science and Engineering 10, no. 9: 1271. https://doi.org/10.3390/jmse10091271
APA StyleFabrello, J., & Matozzo, V. (2022). Bisphenol Analogs in Aquatic Environments and Their Effects on Marine Species—A Review. Journal of Marine Science and Engineering, 10(9), 1271. https://doi.org/10.3390/jmse10091271







