Tomato Yield, Quality, Mineral Composition and Antioxidants as Affected by Beneficial Microorganisms Under Soil Salinity Induced by Balanced Nutrient Solutions
Abstract
1. Introduction
2. Materials and Methods
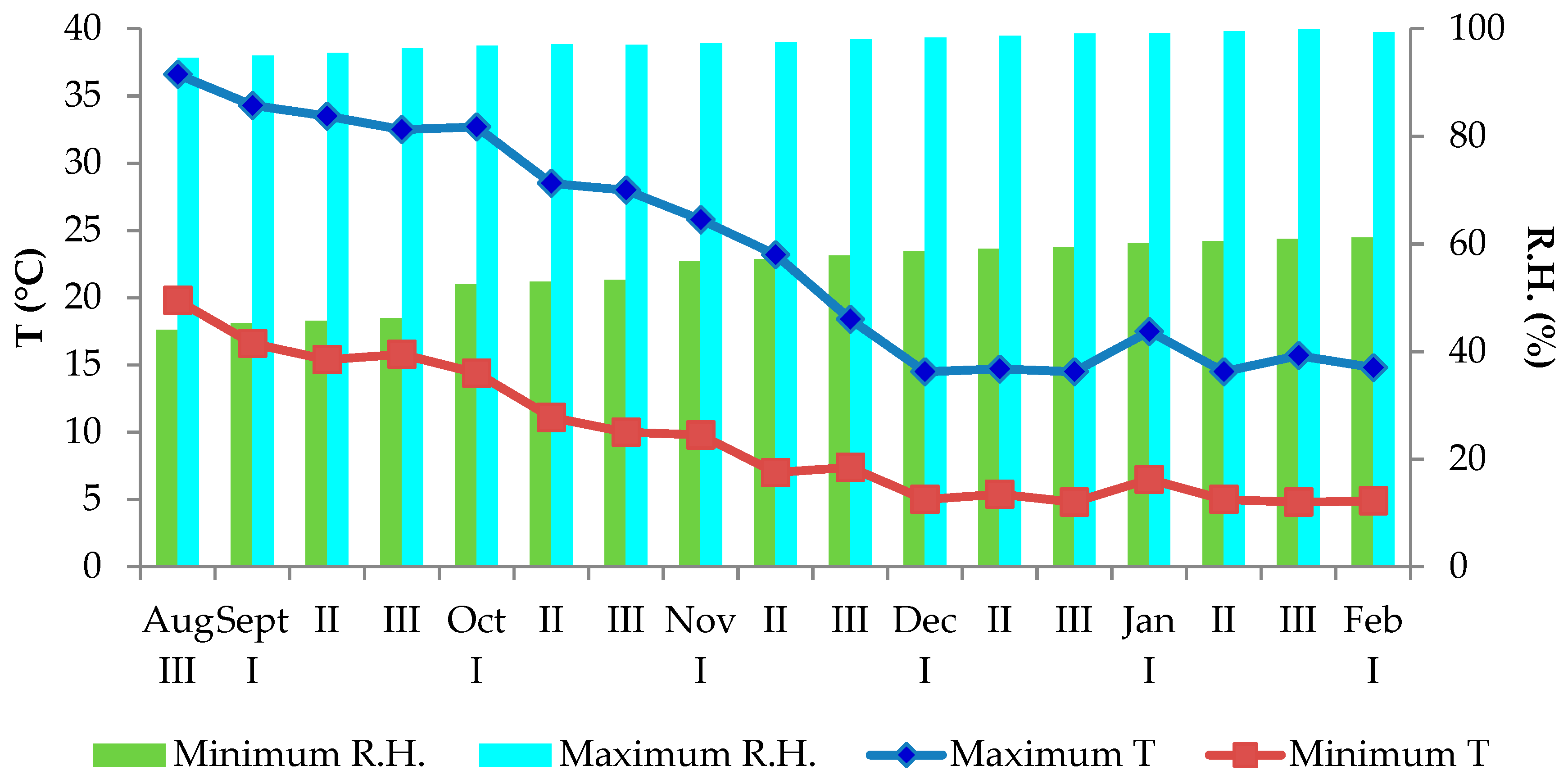
2.1. Experimental Protocol and Growing Conditions
2.2. Determinations of Root Mycorrhizal Colonization, Plant Growth Indices and Yield Components
2.3. Determinations of Fruit Quality, Mineral Composition and Antioxidant Compounds and Activity
2.4. Statistical Processing
3. Results and Discussion
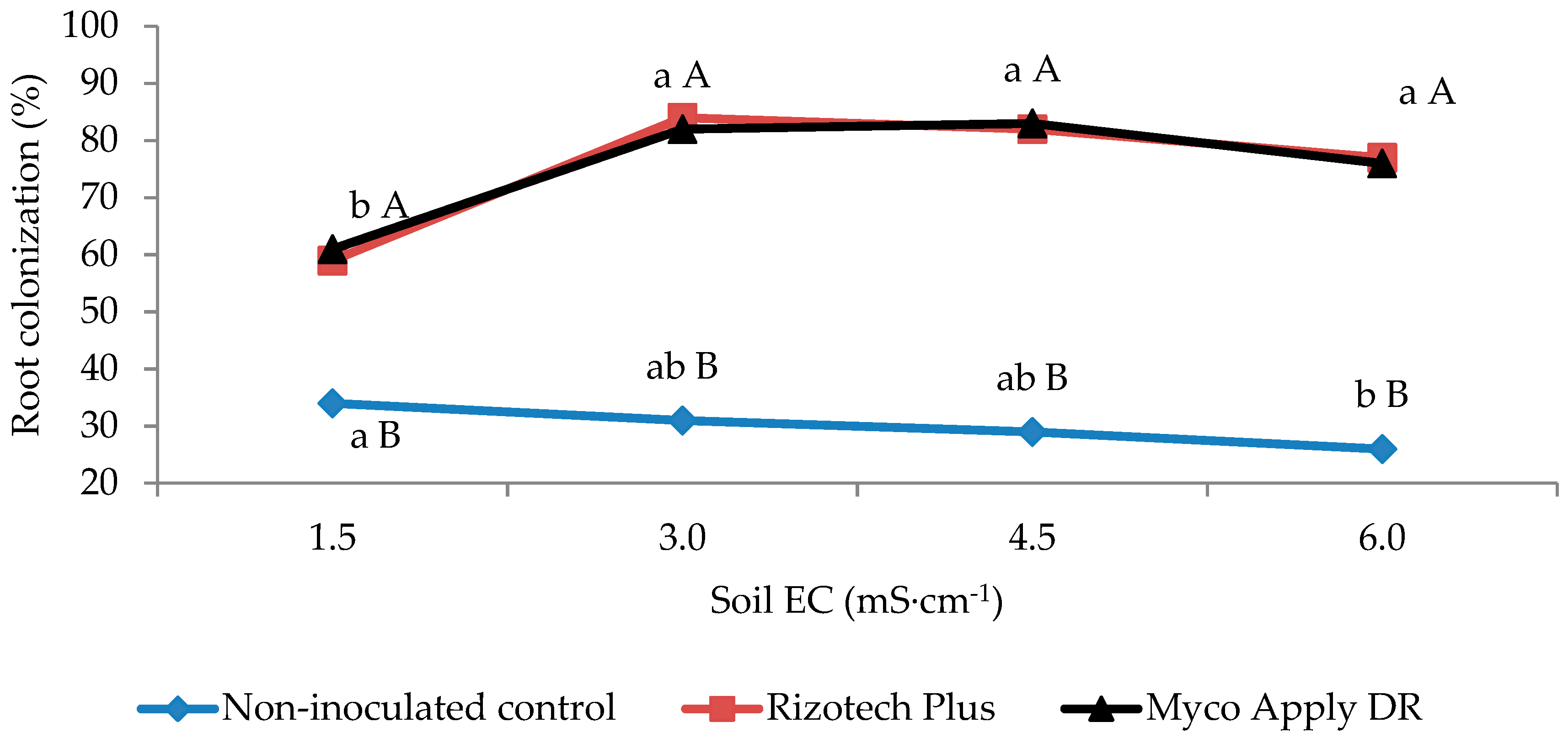
3.1. Root Mycorrhizal Colonization
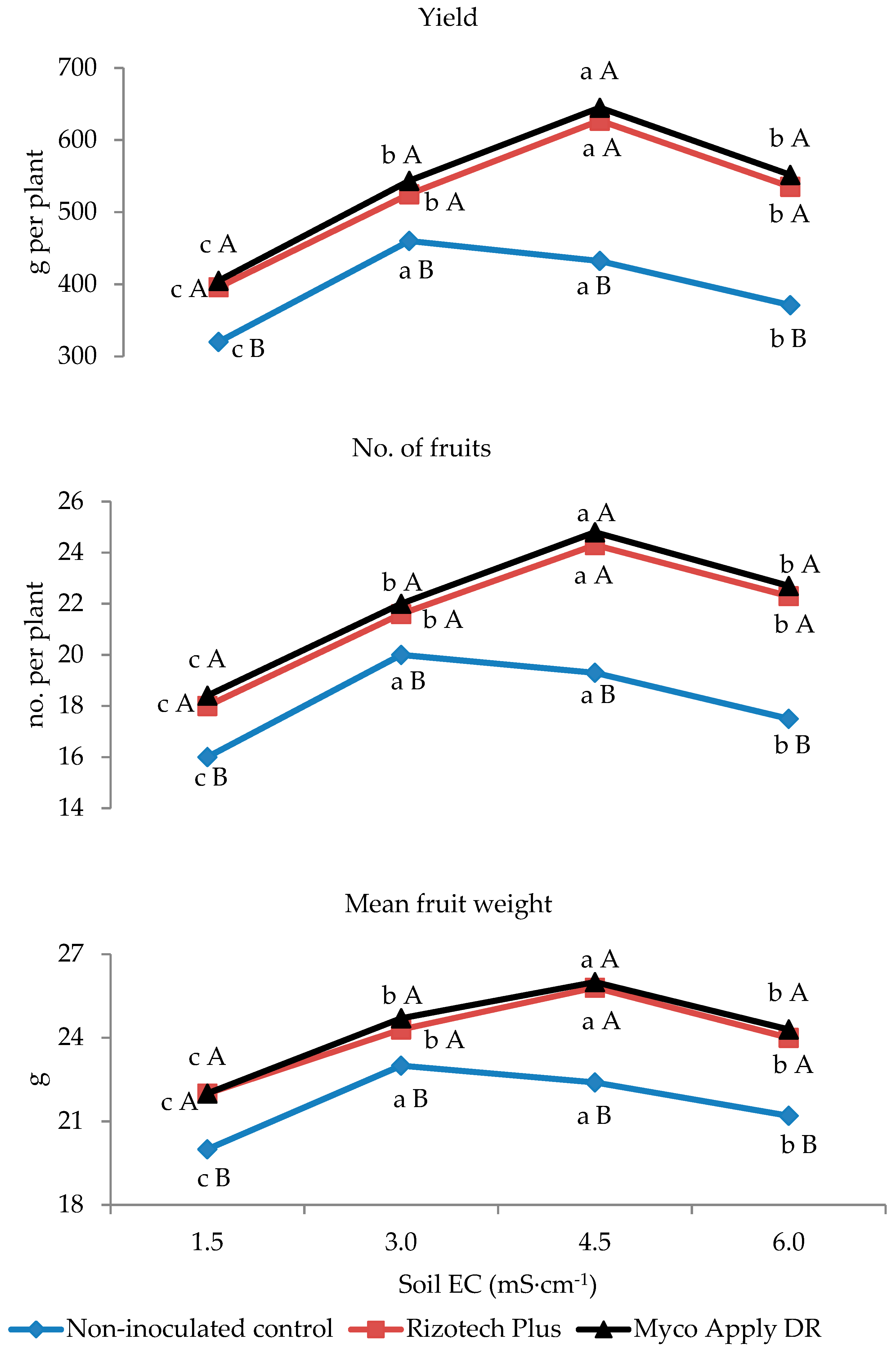
3.2. Plant Growth and Yield
3.3. Fruit Quality, Mineral Composition and Antioxidant Compounds and Activity
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- FAOSTAT. 2014. Available online: http://faostat3.fao.org/browse/Q/QC/E (accessed on 26 April 2019).
- Dorais, M.; Ehret, D.L.; Papadopoulos, A.P. Tomato (Solanum lycopersicum) health components: From the seed to the consumer. Phytochem. Rev. 2008, 7, 231–250. [Google Scholar] [CrossRef]
- Stefan, M.; Munteanu, N.; Stoleru, V.; Mihasan, M.; Hritcu, L. Seed inoculation with plant growth promoting rhizobacteria enhances photosynthesis and yield of runner bean (Phaseolus coccineus L.). Sci. Hortic. 2013, 151, 22–29. [Google Scholar] [CrossRef]
- Pereira, J.A.P.; Vieira, I.J.C.; Freitas, M.S.M.; Prins, C.L.; Martins, M.A.; Rodrigues, R. Effects of arbuscular mycorrhizal fungi on Capsicum spp. J. Agric. Sci. 2016, 154, 828–849. [Google Scholar] [CrossRef]
- Rouphael, Y.; Franken, Ph.; Schneider, C.; Schwarz, D.; Giovannetti, M.; Agnolucci, M.; De Pascale, S.; Bonini, P.; Colla, G. Arbuscular mycorrhizal fungi act as biostimulants in horticultural crops. Sci. Hortic. 2015, 196, 91–108. [Google Scholar] [CrossRef]
- Smith, S.E.; Read, D.J. Mycorrhizal Symbiosis, 3rd ed.; Academic Press: London, UK, 2008. [Google Scholar]
- Zahran, H.H. Diversity, adaptation and activity of the bacterial flora in saline environments. Biol. Fertil. Soils 1997, 25, 211–223. [Google Scholar] [CrossRef]
- Colla, G.; Rouphael, Y.; Di Mattia, E.; El-Nakhel, C.; Cardarelli, M. Co-inoculation of Glomus intraradices and Trichoderma atroviride acts as a biostimulant to promote growth, yield and nutrient uptake of vegetable crops. J. Sci. Food Agric. 2015, 95, 1706–1715. [Google Scholar] [CrossRef] [PubMed]
- Kowalska, I.; Konieczny, A.; Gastol, M.; Sady, W.; Hanus-Fajerska, E. Effect of mycorrhiza and phosphorus content in nutrient solution on the yield and nutritional status of tomato plants grown on rockwool or coconut coir. Agric. Food Sci. 2015, 24, 39–51. [Google Scholar] [CrossRef]
- Hart, M.; Ehret, D.L.; Krumbein, A.; Leung, C.; Murch, S.; Turi, C.; Franken, P. Inoculation with arbuscular mycorrhizal fungi improves the nutritional value of tomatoes. Mycorrhiza 2015, 25, 359–376. [Google Scholar] [CrossRef]
- Tawaraya, K.; Tokairin, K.; Wagatsuma, T. Dependence of Allium fistulosum cultivars on the arbuscular mycorrhizal fungus, Glomus fasciculatum. Appl. Soil Ecol. 2001, 17, 119–124. [Google Scholar] [CrossRef]
- Chen, K.; Liu, W.X.; Guo, S.X.; Liu, R.J.; Li, M. Diversity of arbuscular mycorrhizal fungi in continuous cropping soils used for pepper production. Afr. J. Microbiol. Res. 2012, 6, 2469–2474. [Google Scholar]
- Khalil, S.; Loynachan, T.E.; Tabatabai, M.A. Plant determinants of mycorrhizal dependency in soybean. Agron. J. 1999, 91, 135–141. [Google Scholar] [CrossRef]
- Kormanik, P.P.; Bryan, W.C.; Schultz, R.C. Influence of endomycorrhizae on growth of sweetgum seedlings from eight mother trees. For. Sci. 1977, 23, 500–505. [Google Scholar]
- Ortas, I.; Sari, N.; Akpinar, C. Effect of mycorrhizal inoculation and soil fumigation on the yield and nutrient uptake of some Solanaceae crops (tomato, eggplant and pepper) under field conditions. Agric. Mediterr. 2003, 133, 249–258. [Google Scholar]
- Ortas, I. Effect of mycorrhiza application on plant growth and nutrient uptake in cucumber production under field conditions. Span. J. Agric. Res. 2010, 8, S116–S122. [Google Scholar] [CrossRef]
- Walder, F.; van der Heijden, M.G.A. Regulation of resource exchange in the arbuscular mycorrhizal symbiosis. Nat. Plants 2015, 1, 15159. [Google Scholar] [CrossRef]
- Bona, E.; Cantamessa, S.; Massa, N.; Manassero, P.; Marsano, F.; Copetta, A.; Lingua, G.; D’Agostino, G.; Gamalero, E.; Berta, G. Arbuscular mycorrhizal fungi and plant growth-promoting pseudomonads improve yield, quality and nutritional value of tomato: A field study. Mycorrhiza 2017. [Google Scholar] [CrossRef]
- Juniper, S.; Abbott, L.K. Soil salinity delays germination and limits growth of hyphae from propagules of arbuscular mycorrhizal fungi. Mycorrhiza 2006, 16, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Porcel, R.; Aroca, R.; Ruíz-Lozano, J.M. Salinity stress alleviation using arbuscular mycorrhizal fungi. A review. Agron. Sustain. Dev. 2012, 32, 181–200. [Google Scholar] [CrossRef]
- Chandrasekaran, M.; Kim, K.; Krishnamoorthy, R.; Walitang, D.; Sundaram, S.; Joe, M.M.; Selvakumar, G.; Hu, S.; Oh, S.-H.; Sa, T. Mycorrhizal symbiotic efficiency on C3 and C4 plants under salinity stress—A meta-analysis. Front. Microbiol. 2016, 7, 1246. [Google Scholar] [CrossRef] [PubMed]
- Porras-Soriano, A.; Soriano-Martín, M.L.; Porras-Piedra, A.; Azcón, R. Arbuscular mycorrhizal fungi increased growth, nutrient uptake and tolerance to salinity in olive trees under nursery conditions. J. Plant Physiol. 2009, 166, 1350–1359. [Google Scholar] [CrossRef] [PubMed]
- Navarro, J.M.; Pérez-Tornero, O.; Morte, A. Alleviation of salt stress in citrus seedlings inoculated with arbuscular mycorrhizal fungi depends on the rootstock salt tolerance. J. Plant Physiol. 2014, 171, 76–85. [Google Scholar] [CrossRef]
- Yang, S.-J.; Zhang, Z.-L.; Xue, Y.-X.; Zhang, Z.-F.; Shi, S.-Y. Arbuscular mycorrhizal fungi increase salt tolerance of apple seedlings. Bot. Stud. 2014, 55, 70. [Google Scholar] [CrossRef]
- Pollastri, S.; Savvides, A.; Pesando, M.; Lumini, E.; Volpe, M.G.; Ozudogru, E.A.; Faccio, A.; De Cunzo, F.; Michelozzi, M.; Lambardi, M.; et al. Impact of two arbuscular mycorrhizal fungi on Arundo donax L. response to salt stress. Planta 2018, 247, 573–585. [Google Scholar] [CrossRef]
- Sánchez, E.; Scordia, D.; Lino, G.; Arias, C.; Cosentino, S.L.; Nogués, S. Salinity and water stress effects on biomass production in different Arundo donax L. clones. Bioenerg. Res. 2015, 8, 1461. [Google Scholar] [CrossRef]
- Chitarra, W.; Pagliarani, C.; Maserti, B.; Lumini, E.; Siciliano, I.; Cascone, P.; Schubert, A.; Gambino, G.; Balestrini, R.; Guerrieri, E. Insights on the impact of arbuscular mycorrhizal symbiosis on tomato tolerance to water stress. Plant Physiol. 2016, 171, 1–15. [Google Scholar] [CrossRef]
- Augé, R.M.; Toler, H.D.; Saxton, A.M. Arbuscular mycorrhizal symbiosis alters stomatal conductance of host plants more under drought than under amply watered conditions: A meta-analysis. Mycorrhiza 2015, 25, 13–24. [Google Scholar] [CrossRef]
- Chialva, M.; Fangel, J.U.; Novero, M.; Zouari, I.; Salvioli di Fossalunga, A.; Willats, W.G.T.; Bonfante, P.; Balestrini, R. Understanding changes in tomato cell walls in roots and fruits: The contribution of arbuscular mycorrhizal colonization. Int. J. Mol. Sci. 2019, 20, 415. [Google Scholar] [CrossRef]
- Chitarra, W.; Maserti, B.; Gambino, G.; Guerrieri, E.; Balestrini, R. Arbuscular mycorrhizal symbiosis-mediated tomato tolerance to drought. Plant Signal. Behav. 2016, 11, e1197468. [Google Scholar] [CrossRef]
- Brilli, F.; Pollastri, S.; Raio, A.; Baraldi, R.; Neri, L.; Bartolini, P.; Podda, A.; Loreto, F.; Maserti, B.E.; Balestrini, R. Root colonization by Pseudomonas chlororaphis primes tomato (Lycopersicum esculentum) plants for enhanced tolerance to water stress. J. Plant Physiol. 2019, 232, 82–93. [Google Scholar] [CrossRef]
- Volpe, V.; Chitarra, W.; Cascone, P.; Volpe, M.G.; Bartolini, P.; Moneti, G.; Pieraccini, G.; Di Serio, C.; Maserti, B.; Guerrieri, E.; et al. Arbuscular mycorrhizal fungi differently affects water stress tolerance in tomato. Front. Plant Sci. 2018, 9, 1480. [Google Scholar] [CrossRef]
- Trouvelot, A.; Kough, J.; Gianinazzi-Pearson, V. Mesure du taux de mycorrhization VA d’un système radiculaire. Recherche de méthodes d’estimation ayant une signification functionnelle. In Physiological and Genetical Aspects of Mycorrhizae; Gianninazzi-Pearson, V., Gianinazzi, S., Eds.; INRA: Paris, France, 1986; pp. 217–221. (In French) [Google Scholar]
- Golubkina, N.A.; Kekina, H.G.; Engalichev, M.R.; Antoshkina, M.S.; Caruso, G. Genotypic effect on fruit production, quality, antioxidants and element composition of organically-grown Physalis angulata L. and Physalis pubescens L. Folia Hortic. 2018, 30, 367–374. [Google Scholar] [CrossRef]
- Rouphael, Y.; Giordano, M.; Cardarelli, M.; Cozzolino, E.; Mori, M.; Kyriacou, M.C.; Bonini, P.; Colla, G. Plant- and seaweed-based extracts increase yield but differentially modulate nutritional quality of greenhouse spinach through biostimulant action. Agronomy 2018, 8, 126. [Google Scholar] [CrossRef]
- De Sio, F.; Rapacciuolo, M.; De Giorgi, A.; Trifirò, A.; Giuliano, B.; Vitobello, L.; Cuciniello, A.; Caruso, G. Yield, quality and antioxidants of peeled tomato as affected by genotype and industrial processing in southern Italy. Adv. Hortic. Sci. 2018, 32, 379–387. [Google Scholar]
- Golubkina, N.A.; Kosheleva, O.V.; Krivenkova, L.V.; Dobrutskaya, H.G.; Nadezhkin, S.; Caruso, G. Intersexual differences in plant growth, yield, mineral composition and antioxidants of spinach (Spinacia oleracea L.) as affected by selenium form. Sci. Hortic. 2017, 225, 350–358. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of free radical method to evaluate antioxidant activity. LWT–Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Rafique, M.; Ortas, İ. Nutrient uptake-modification of different plant species in Mediterranean climate by arbuscular mycorrhizal fungi. Eur. J. Hortic. Sci. 2018, 83, 65–71. [Google Scholar] [CrossRef]
- Candido, V.; Campanelli, G.; D’Addabbo, T.; Castronuovo, D.; Perniola, M.; Camele, I. Growth and yield promoting effect of artificial mycorrhization on fieldtomato at different irrigation regimes. Sci. Hortic. 2015, 187, 35–43. [Google Scholar] [CrossRef]
- Balestrini, R.; Salvioli, A.; Dal Molin, A.; Novero, M.; Gabelli, G.; Paparelli, E.; Marroni, F.; Bonfante, P. Impact of an arbuscular mycorrhizal fungus versus a mixed microbial inoculum on the transcriptome reprogramming of grapevine roots. Mycorrhiza 2017, 27, 417–430. [Google Scholar] [CrossRef]
- Gutjahr, C.; Parniske, M. Cell and developmental biology of arbuscular mycorrhiza symbiosis. Annu. Rev. Cell Dev. Biol. 2013, 29, 593–617. [Google Scholar] [CrossRef] [PubMed]
- Bücking, H.; Shachar-Hill, Y. Phosphate uptake, transport and transfer by the arbuscular mycorrhizal fungus Glomus intraradices is stimulated by increased carbohydrate availability. New Phytol. 2005, 165, 899–912. [Google Scholar] [CrossRef]
- Munns, R. Comparative physiology of salt and water stress. Plant Cell Environ. 2002, 25, 239–250. [Google Scholar] [CrossRef]
- Sonneveld, C.; Vanderburg, A.M.M. Sodium-chloride salinity in fruit vegetable crops in soilless culture. Neth. J. Agric. Sci. 1991, 39, 115–122. [Google Scholar]
- Urrea-López, R.; Díaz de la Garza, R.I.; Valiente-Banuet, J.I. Effects of substrate salinity and nutrient levels on physiological response, yield, and fruit quality of habanero pepper. HortScience 2014, 49, 812–818. [Google Scholar] [CrossRef]
- Graham, M.H.; Haynes, R.J. Organic matter accumulation and fertilizer-induced acidification interact to affect soil microbial and enzyme activity on a long-term sugarcane management experiment. Biol. Fertil. Soils 2005, 41, 249–256. [Google Scholar] [CrossRef]
- García-Fraile, P.; Carro, L.; Robledo, M.; Ramírez-Bahena, M.-H.; Florés-Felix, J.-D.; Fernández, M.T.; Mateos, P.F.; Rivas, R.; Igual, J.M.; Martínez-Molina, E.; et al. Rhizobium promotes non-legumes growth and quality in several production steps: Towards a biofertilization of edible raw vegetables healthy for humans. PLoS ONE 2012, 7, e38122. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, Y.; Tamura, H.; Harada, T. Growth enhancement and verticillium wilt control by vesicular-arbuscular mycorrhizal fungus inoculation in eggplant. J. Jpn. Soc. Hortic. Sci. 1995, 64, 555–561. [Google Scholar] [CrossRef]
- Ludwig-Müller, J.; Güther, M. Auxins as signals in arbuscular mycorrhiza formation. Plant Signal. Behav. 2007, 2, 194–196. [Google Scholar] [CrossRef] [PubMed]
- Gemma, J.N.; Koske, R.E.; Habte, M. Mycorrhizal dependency of some endemic and endangered Hawaiian plant species. Am. J. Bot. 2002, 89, 337–345. [Google Scholar] [CrossRef]
- Plenchette, C.; Fortin, J.; Furlan, V. Growth responses of several plant species to mycorrhizae in a soil of moderate P-fertility. Plant Soil 1983, 70, 199–209. [Google Scholar] [CrossRef]
- Sainju, U.M.; Dris, R.; Singh, B. Mineral nutrition of tomato. Food Agric. Environ. 2003, 1, 176–183. [Google Scholar]
- Mahanta, D.; Rai, R.K.; Mishra, S.D.; Raja, A.; Purakayastha, T.J.; Varghese, E. Influence of phosphorus and biofertilizers on soybean and wheat root growth and properties. Field Crops Res. 2014, 166, 1–9. [Google Scholar] [CrossRef]
- Golubkina, N.; Zamana, S.; Seredin, T.; Poluboyarinov, P.; Sokolov, S.; Baranova, H.; Krivenkov, L.; Pietrantonio, L.; Caruso, G. Effect of selenium biofortification and beneficial microorganism inoculation on yield, quality and antioxidant properties of shallot bulbs. Plants 2019, 8, 102. [Google Scholar] [CrossRef] [PubMed]
- Adams, P.; Ho, L.C. Effects of constant and fluctuating salinity on the yield, quality and calcium status of tomatoes. J. Hortic. Sci. 1989, 64, 725–732. [Google Scholar] [CrossRef]
- Savvas, D.; Stamati, E.; Tsirogiannis, I.L.; Mantzos, N.; Barouchas, P.E.; Katsoulas, N.; Kittas, C. Interactions between salinity and irrigation frequency in greenhouse pepper grown in closed-cycle hydroponic systems. Agric. Water Manag. 2007, 91, 102–111. [Google Scholar] [CrossRef]
- Tadesse, T.; Nichols, M.A.; Fisher, K.J. Nutrient conductivity effects on sweet pepper plants grown using a nutrient film technique 1. Yield and fruit quality. N. Z. J. Crop. Hortic. Sci. 1999, 27, 141–148. [Google Scholar]
- Zouari, I.; Salvioli, A.; Chialva, M.; Novero, M.; Miozzi, L.; Tenore, G.C.; Bagnaresi, P.; Bonfante, P. From root to fruit: RNA-Seq analysis shows that arbuscular mycorrhizal symbiosis may affect tomato fruit metabolism. BMC Genom. 2014, 15, 221–239. [Google Scholar] [CrossRef] [PubMed]
- Giovannetti, M.; Avio, L.; Barale, R.; Ceccarelli, N.; Cristofani, R.; Iezzi, A.; Mignolli, F.; Picciarelli, P.; Pinto, B.; Reali, D.; et al. Nutraceutical value and safety of tomato fruits produced by mycorrhizal plants. Br. J. Nutr. 2012, 107, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Ndung’u Magiroi, K.W.; Hermann, L.; Okalebo, J.R.; Othieno, C.O.; Pypers, P.; Lesueur, D. Occurrence and genetic diversity of phosphate-solubilizing bacteria in soils of different chemical characteristics in Kenia. Ann. Microbiol. 2012, 62, 897–904. [Google Scholar] [CrossRef]
- Hamdali, H.; Hafidi, M.; Virolle, M.J.; Ouhdouch, Y. Growth promotion and protection against damping-off of wheat by two rock phosphate solubilizing actinomycetes in a P-deficient soil under greenhouse conditions. Appl. Soil Ecol. 2008, 40, 510–517. [Google Scholar] [CrossRef]
- Bücking, H.; Heyser, W. Uptake and transfer of nutrients in ectomycorrhizal associations: Interactions between photosynthesis and phosphate nutrition. Mycorrhiza 2003, 13, 59–68. [Google Scholar] [CrossRef]
- Thompson, J.P.; Clewett, T.G.; Fiske, M.L. Field inoculation with arbuscular-mycorrhizal fungi overcomes phosphorus and zinc deficiencies of linseed (Linum usitatissimum) in a vertisol subject to long-fallow disorder. Plant Soil 2013, 371, 117–137. [Google Scholar] [CrossRef]
- De Santiago, A.; García-López, A.M.; Quintero, J.M.; Avilés, M.; Delgado, A. Effect of Trichoderma asperellum strain T34 and glucose addition on iron nutrition in cucumber grown on calcareous soils. Soil Biol. Biochem. 2013, 57, 598–605. [Google Scholar] [CrossRef]
- Ochoa-Velasco, C.E.; Valadez-Blanco, R.; Salas-Coronado, R.; Sustaita-Rivera, F.; Hernández-Carlos, B.; García-Ortega, S.; Santos-Sánchez, N.F. Effects of nitrogen fertilization and Bacillus licheniformis biofertilizer addition on the antioxidants compounds and antioxidant activity of greenhouse cultivated tomato fruits (Solanum lycopersicum L. var. Sheva). Sci. Hortic. 2016, 201, 338–345. [Google Scholar] [CrossRef]
- Le Bot, J.; Bernard, C.; Robin, C.; Bourgaud, F.; Adamowicz, S. The ‘trade-off’ between synthesis of primary and secondary compounds in young tomato leaves is altered by nitrate nutrition: Experimental evidence and model consistency. J. Exp. Bot. 2009, 60, 4301–4314. [Google Scholar] [CrossRef]
- Seró, R.; Núñez, N.; Núñez, O.; Camprubí, A.; Grases, J.M.; Saurina, J.; Moyano, E.; Calvet, C. Modified distribution in the polyphenolic profile of rosemary leaves induced by plant inoculation with an arbuscular mycorrhizal fungus. J. Sci. Food Agric. 2019, 99, 2966–2973. [Google Scholar]
- Amanifar, S.; Khodabandeloo, M.; Fard, E.M.; Askari, M.S.; Ashrafi, M. Alleviation of salt stress and changes in glycyrrhizin accumulation by arbuscular mycorrhiza in liquorice (Glycyrrhiza glabra) grown under salinity stress. Environ. Exp. Bot. 2019, 160, 25–34. [Google Scholar] [CrossRef]
- Nzanza, B.; Marais, D.; Soundy, P. Yield and nutrient content of tomato (Solanum lycopersicum L.) as influenced by Trichoderma harzianum and Glomus mosseae inoculation. Sci. Hortic. 2012, 144, 55–59. [Google Scholar] [CrossRef]
- De Pascale, S.; Maggio, A.; Fogliano, V.; Ambrosino, P.; Ritieni, A. Irrigation with saline water improves carotenoids content and antioxidant activity of tomato. J. Hortic. Sci. Biotechnol. 2001, 76, 447–453. [Google Scholar] [CrossRef]
- Navarro, J.M.; Flores, P.; Garrido, C.; Martinez, V. Changes in the contents of antioxidant compounds in pepper fruits at different ripening stages, as affected by salinity. Food Chem. 2006, 96, 66–73. [Google Scholar] [CrossRef]
- Amalfitano, C.; Del Vacchio, L.; Somma, S.; Cuciniello, A.; Caruso, G. Effects of cultural cycle and nutrient solution electrical conductivity on plant growth, yield and fruit quality of “Friariello” pepper grown in hydroponics. Hortic. Sci. 2017, 44, 91–98. [Google Scholar]
| Precocity | Leaf Area | Dry Matter | Marketable Fruits | |||
|---|---|---|---|---|---|---|
| Experimental Treatment | Days from Transplant to First Harvest | m2 per Plant | g per Plant | Yield (g per Plant) | Number | Mean Weight (g) |
| Mycorrhizal-based formulate | ||||||
| Rizotech | 69.5 a | 0.37 a | 58.7 b | 520.8 a | 21.6 a | 24.0 a |
| Myco Apply | 69.5 a | 0.38 a | 88.8 a | 536.2 a | 22.0 a | 24.3 a |
| Non-inoculated control | 66.0 b | 0.34 b | 90.5 a | 395.8 b | 18.2 b | 21.7 b |
| Soil electrical conductivity | ||||||
| 1.5 mS·cm−1 | 71.3 a | 0.33 c | 47.7 c | 373.6 c | 17.5 c | 21.3 c |
| 3.0 mS·cm−1 | 69.7 ab | 0.37 ab | 79.1 b | 509.4 b | 21.2 b | 24.0 ab |
| 4.5 mS·cm−1 | 67.0 bc | 0.40 a | 107.2 a | 568.0 a | 22.8 a | 24.7 a |
| 6.0 mS·cm−1 | 65.3 c | 0.36 bc | 83.3 b | 485.9 b | 20.8 b | 23.2 b |
| Experimental Treatment | Dry Residue (%) | Soluble Solids (°Brix) | Organic Acids (g·kg−1 d.w.) | |||
|---|---|---|---|---|---|---|
| Malic | Oxalic | Citric | Isocitric | |||
| Mycorrhizal-based Formulate | ||||||
| Rizotech | 8.9 a | 7.6 a | 5.7 a | 2.2 a | 40.1 a | 0.71 a |
| Myco Apply | 8.9 a | 7.6 a | 6.0 a | 2.4 a | 41.6 a | 0.70 a |
| Non-inoculated control | 8.5 b | 7.3 b | 4.6 b | 1.5 b | 31.5 b | 0.49 b |
| Soil electrical Conductivity | ||||||
| 1.5 mS·cm−1 | 8.2 c | 7.0 d | 4.2 d | 1.2 d | 31.7 d | 0.49 c |
| 3.0 mS·cm−1 | 8.5 c | 7.3 c | 4.8 c | 1.7 c | 35.3 c | 0.55 c |
| 4.5 mS·cm−1 | 9.0 b | 7.7 b | 5.7 b | 2.2 b | 39.1 b | 0.66 b |
| 6.0 mS·cm−1 | 9.4 a | 8.0 a | 7.0 a | 2.9 a | 44.8 a | 0.83 a |
| Experimental Treatment | K | Ca | Mg | Na | P | S | NO3 | Cl |
|---|---|---|---|---|---|---|---|---|
| g·kg−1 d.w. | ||||||||
| Mycorrhizal-Based Formulate | ||||||||
| Rizotech | 32.4 a | 0.66 a | 1.31 a | 0.60 b | 1.06 a | 0.60 a | 0.17 a | 5.46 |
| Myco Apply | 32.5 a | 0.70 a | 1.32 a | 0.63 b | 1.00 a | 0.64 a | 0.18 a | 5.43 |
| Non-inoculated control | 28.8 b | 0.49 b | 1.06 b | 0.69 a | 0.64 b | 0.36 b | 0.09 b | 5.42 |
| n.s. | ||||||||
| Soil Electrical Conductivity | ||||||||
| 1.5 mS·cm−1 | 29.0 c | 0.52 d | 1.10 b | 0.75 a | 0.77 c | 0.42 c | 0.10 c | 5.40 |
| 3.0 mS·cm−1 | 30.4 bc | 0.59 c | 1.15 b | 0.69 b | 0.86 b | 0.53 bc | 0.11 c | 5.47 |
| 4.5 mS·cm−1 | 31.8 ab | 0.65 b | 1.32 a | 0.60 c | 0.93 b | 0.57 b | 0.17 b | 5.32 |
| 6.0 mS·cm−1 | 33.7 a | 0.72 a | 1.35 a | 0.52 d | 1.04 a | 0.62 a | 0.21 a | 5.57 |
| n.s. | ||||||||
| Lycopene mg·100 g−1 f.w. | Total Phenols mg Gallic Acid·100 g−1 d.w. | Ascorbic Acid mg·100 g−1 f.w. | Lipophilic Antioxidant Activity mmol Trolox eq·100 g−1 d.w. | Hydrophilic Antioxidant Activity mmol Ascorbic Acid eq·100 g−1 d.w. | |
|---|---|---|---|---|---|
| Mycorrhizal-based formulate | |||||
| Rizotech | 338.2 a | 1.93 a | 18.5 a | 10.2 a | 8.6 a |
| Myco Apply | 350.0 a | 2.04 a | 20.4 a | 11.3 a | 9.0 a |
| Non-inoculated control | 285.6 b | 1.63 b | 14.4 b | 7.5 b | 7.8 b |
| Soil electrical conductivity | |||||
| 1.5 mS·cm−1 | 207.7 d | 1.75 b | 11.0 d | 7.5 d | 7.4 c |
| 3.0 mS·cm−1 | 297.3 c | 1.80 b | 15.3 c | 9.1 c | 8.2 b |
| 4.5 mS·cm−1 | 360.6 b | 1.95 a | 20.6 b | 10.0 b | 8.8 ab |
| 6.0 mS·cm−1 | 432.9 a | 1.97 a | 24.2 a | 11.9 a | 9.4 a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sellitto, V.M.; Golubkina, N.A.; Pietrantonio, L.; Cozzolino, E.; Cuciniello, A.; Cenvinzo, V.; Florin, I.; Caruso, G. Tomato Yield, Quality, Mineral Composition and Antioxidants as Affected by Beneficial Microorganisms Under Soil Salinity Induced by Balanced Nutrient Solutions. Agriculture 2019, 9, 110. https://doi.org/10.3390/agriculture9050110
Sellitto VM, Golubkina NA, Pietrantonio L, Cozzolino E, Cuciniello A, Cenvinzo V, Florin I, Caruso G. Tomato Yield, Quality, Mineral Composition and Antioxidants as Affected by Beneficial Microorganisms Under Soil Salinity Induced by Balanced Nutrient Solutions. Agriculture. 2019; 9(5):110. https://doi.org/10.3390/agriculture9050110
Chicago/Turabian StyleSellitto, Vincenzo Michele, Nadezhda A. Golubkina, Laura Pietrantonio, Eugenio Cozzolino, Antonio Cuciniello, Vincenzo Cenvinzo, Imbrea Florin, and Gianluca Caruso. 2019. "Tomato Yield, Quality, Mineral Composition and Antioxidants as Affected by Beneficial Microorganisms Under Soil Salinity Induced by Balanced Nutrient Solutions" Agriculture 9, no. 5: 110. https://doi.org/10.3390/agriculture9050110
APA StyleSellitto, V. M., Golubkina, N. A., Pietrantonio, L., Cozzolino, E., Cuciniello, A., Cenvinzo, V., Florin, I., & Caruso, G. (2019). Tomato Yield, Quality, Mineral Composition and Antioxidants as Affected by Beneficial Microorganisms Under Soil Salinity Induced by Balanced Nutrient Solutions. Agriculture, 9(5), 110. https://doi.org/10.3390/agriculture9050110








