1. Introduction
Legumes cultivation plays an important role in sustainable agriculture, since it contributes to improve the soil nitrogen content through the nitrogen-fixing bacteria that grow in symbiosis with legume roots. Thus, leguminous crops are often used in crop rotation with cereal crops for improving soil fertility [
1,
2]. Worldwide, legumes are primarily used as dry seeds (so-called pulses) for both human and animal nutrition. Pulses have a great importance for human nutrition considering their richness in proteins, carbohydrates, and minerals [
3]. In this context, it is interesting to highlight that the protein content in faba beans can exceed 30% (on dry basis) [
4], making this legume an excellent meat substitute. It is important to note that worldwide the legume production between 1961 and 2014 almost doubled, while the trend of legume production in the main agricultural countries of the European Union shows large differences. For example, France, Germany, the United Kingdom, and Poland show consistent increases (ranging from 29 to 420%), while Italy, Spain, Greece, and Romania show a decrease (ranging from 29 to 79%) [
5]. In particular, faba beans production in Italy ranging from just under 387,700 tons in 1961 to about 79,800 tons in 2015 [
5]. It should be considered that, in the last years, there was a new-found interest in legumes worldwide. Thus, to promote the unique quality of pulses, the year 2016 was declared as the International Year of Pulses (IYP) by the United Nations (UN) General Assembly. The IYP 2016 heightened public awareness of the nutritional benefits of pulses as part of sustainable food production aiming toward food security and nutrition. The IYP 2016 created a unique opportunity to encourage connections throughout the food chain that would better utilize pulse-based proteins, further global production of pulses, better utilize crop rotations and address the challenges in the trade of pulses [
6].
Apart the use as dry seeds, it should be considered that some legumes, such as green pea (
Pisum sativum L.), green bean (
Phaseolus vulgaris L.), and green cowpea (
Vigna unguiculata [L.] Walp. subsp.
unguiculata [L.] Walp.) are diffused also as vegetables for fresh consumption as well as for canned and frozen products. Differently from dry seeds, green legumes contain more water and lesser proteins and starch, but they are characterized by a greater amount of healthy compounds for humans such as phenols, chlorophylls, carotenoids, and vitamins [
7]. In addition to the previously described legumes also faba beans (
Vicia faba L. var.
major Harz) can be used as fresh, canned, or frozen vegetable [
8]. On the other hand, it must be considered that, currently, faba beans do not receive the merited consideration, probably due to the presence of vicine and convicine, which limit their consumption. Effectively, these anti-nutritional factors may cause an acute hemolysis in glucose-6-phosphate dehydrogenase deficient subjects [
9]. At the same time, faba beans show interesting characteristics also due to the presence of some nutraceutical compounds such as levo-dihydroxy phenylalanine (L-dopa), the precursor of the neurotransmitter dopamine [
10]. This compound is used for the treatment of some disease such as Parkinson, hypertension, renal failure, and liver cirrhosis [
10].
Faba beans as a vegetable for fresh consumption represent a traditional food in several Mediterranean countries. In Italy, this vegetable product named “fave fresche” (Italian for fresh faba beans) is listed in the “List of Traditional Agri-Food Products” of the Italian Ministry of Agricultural, Food and Forestry Policies, since its processing, preservation and aging methods is consolidated in time, harmonious, according to traditional rules, for a period not less than 25 years. In this context, it is important to highlight that the traditional food products are greatly appreciated for both their high nutritional and sensorial value, but also due to natural biodiversity of raw materials [
11]. Unfortunately, the production of faba beans for fresh seeds in Italy between 1961 and 2014 was almost halved ranging from about 109,000 tons to 52,000 tons [
5]. Furthermore, it should consider an estimated loss of 75% in the agro-biodiversity compared to 1900s, probably due to the wide use of high-yielding modern varieties [
12]. It is not a case that the loss of agro-biodiversity regards especially the landraces that are variable populations since they are not involved into programs of genetic improvements. Landraces are also known as local varieties, farmer’s varieties and folk varieties, and they are often characterized by a local name. This, considering their well adaptation to both specific environment and local cultural system as well as uses and knowledge of the local population that has developed their cultivation and valorization [
13]. In this context, it must be noted that the 28 European Union Countries as a whole have registered 115 varieties of broad bean in the common catalogue (by December 2018), of which only five varieties are hybrids (all Spanish). Italy has registered 15 varieties, including Aguadulce supersimonia and Extra-early purple used in our research [
14]. Although modern commercial varieties can show some desirable traits (e.g., a high yield), it should be highlighted that they can contain lower levels of appealing compounds than the older landraces [
13]. Thus, vegetable landraces represent an important germplasm resource for breeding and may be also considered as an agronomic valid choice due to their good quality and good adaptation to environment together to a lower needs of external input. [
15]. Therefore, the cultivation of faba beans landraces as a vegetable for fresh consumption together with the application of specific crop-management techniques may represent a useful strategy for linking sustainability and high-quality production.
The Puglia region (southern Italy) is a region very rich in vegetable landraces, which are largely used and requested by consumers. This, represents an example of sustainable food system in which modern vegetable production is strongly linked with local tradition and environment [
13,
16]. Unfortunately, the richness of local varieties in this region was progressively reduced in the last few decades due to several factors, such as reduction of seed savers, farmland abandonment, and loss of knowledge, generation after generation [
17]. In order to help conserve genetic resources in agriculture, the Puglia Regional Administration planned two specific actions under its 2007–2013 and 2014–2020 Rural Development Programmes. In this context, thanks to the project “Biodiversity of the Puglia’s vegetable crops (BiodiverSO)” [
17], several faba bean landraces were characterized for morphological, chemical and genetic traits.
To the best of our knowledge, there is a lack of information in literature with regard to faba beans as a vegetable for fresh consumption. Therefore, considering all the above remarks, the aims of the present study was to evaluate some quality traits as well as the yield response in some landraces of faba bean for fresh consumption in order to assess and distinguish the landraces also in comparison with two common commercial varieties.
2. Materials and Methods
2.1. Cropping Details
Six genotypes of broad beans were used for the experimentation: two commercial varieties (Aguadulce supersimonia and Extra-early purple) and four Puglia landraces (FV5, Cegliese, Iambola, and San Francesco—
Table 1).
The six genotypes were cultivated in open field at “La Noria” experimental farm of the Institute of Sciences of Food Production of the Italian National Research Council, located in Mola di Bari (Ba, Italy), 24 m above sea level, 41°03′ N, 17°04′ E. Sowing was carried out on autumn 2017 using 4.16 plants m
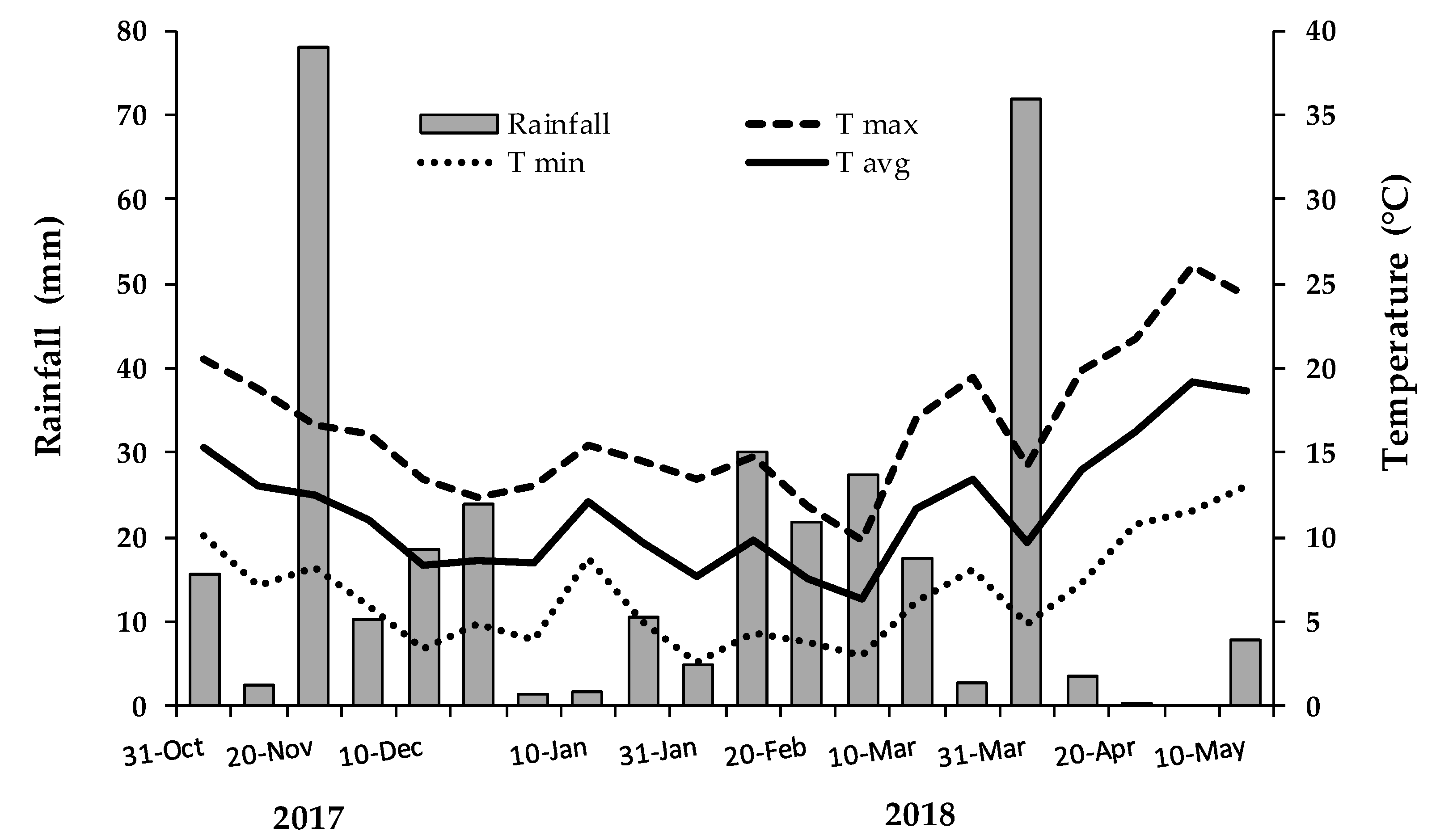
−2 (80 cm between rows and 30 cm within row). Seeds were sown in pockets using three seeds per pocket. After seedling emergence, thinning was carried out in order to obtain one plant per pocket. Every plot was of 30 plants, although plants on the border rows were not used for sampling. A randomized block design with three replications was adopted in the field. Plants were grown according to the agricultural practices of farmers specialized in the cultivation of local tomato landraces. No fertilization was carried out, while a drip irrigation system was set up to carry out supplemental irrigation between sowing and emergence of seedling. Temperatures and rainfall are reported in
Figure 1.
Harvesting was performed according to the progressive ripening of pods for fresh consumption, choosing those which appeared turgid to the touch and with well-formed seeds. Four harvest were carried out between April and May 2018. Harvesting was carried out simultaneously for all genotypes on 12 plants of the sampling area inside each plot. Quality analysis were carried out in triplicate for each plot at second harvest by using a representative portion of the entire plot harvest.
The harvested pods were transported to the laboratory of the Department of Agricultural and Environmental Science, University of Bari Aldo Moro, Italy, for performing both physical and chemical analysis. About 200 g of seeds for each replication were freeze-dried for the chemical analysis.
2.2. Physical Analysis
Before seeding for each genotype, the weight of 1000 dry seeds was measured in triplicate by using an analytical balance (precision 0.001 g). On harvested pods were measured as follows: weight (g), length (cm), number of seeds and seeds weight (g); yield (weigh and number of pods per both plant and area). Length, width, and thickness of the seed were also measured.
According to the Commission Internationale de l’Eclairage (CIE) color parameters (L, a*, b*) were detected by using the colorimeter CR-400 Chroma Meter (Minolta Co., Osaka, Japan). Each measurement was carried out in triplicate on 30 fresh seeds per sample. The colorimeter was calibrated with a standard reference (a white ceramic plate) with values L, a* and b* of 97.55, 1.32 and 1.41, respectively. Hue angle (h° = tan−1 (b*/a*)) and saturation or chroma (C = (a*2 + b*2)1/2) were then calculated from the primary L, a* and b* readings.
Total soluble solids (TSS) content were measured by using the refractometer DBR45 (XS Instruments, Carpi, Modena, Italy) on the liquid seeds extract obtained by homogenizing 100 g of sample in a blender (Sterilmimex lab, International PBI, Milan, Italy), and then filtering the juice; results were expressed in °Brix. For the measurement of dry weight (DW). Fresh seed samples were maintained in a forced draft oven at 70 °C until constant weight was reached.
2.3. Total Titratable Acidity, pH and Crude Protein Content
The titratable acidity (TA) was measured using a pH meter Model 507 (Crison, Milan, Italy) by potentiometric titration of seeds juice (10 mL) to pH 8.1 using NaOH (0.1 M). The results were expressed as percentage of citric acid equivalents in the juice. The pH values were determined with a pH meter Model 507 (Crison, Milan, Italy). The crude proteins content (N × 6.25) of the seeds was determined by Kjeldahl nitrogen, according to the AOAC method 955.04 [
18].
2.4. Vitamin C Content
Vitamin C was measured according to the method reported by Lee et al. [
19] with some modifications. Briefly, 5 g of samples were extracted by 50 mL of 3% (w/v) metaphosphoric acid followed by shaking for 45 min at 4 °C. Then, samples were centrifuged at 4000 r for 10 min and the supernatant (0.5 mL) was filtered with a 0.22 μm Nylon
® filter (Fisher Scientific
TM, Waltham, MA, USA). The High Performance Liquid Chromatography (HPLC) analysis was performed using an Agilent 1100 quaternary pump and an Agilent 1260 UV detector (Agilent Technologies, Santa Clara, CA, USA), and the absorbance was measured at 210 nm. To determine ascorbic acid content, a calibration curve of authentic L-ascorbic acid was used.
2.5. Total Phenols
For total phenols (TP) the extraction procedure reported by Renna et al. [
20] was used. A 0.45 g sample of lyophilized seeds was homogenized in methanol/water (80:20 v/v) for 1 min and then centrifuged at 6440 r for 5 min at 4 °C. TP were determined using a T60U spectrophotometer (PG Instruments, Woodway lane, Alma park, Leicestershire, UK) and the absorbance was measured at 765 nm; results were expressed as mg of gallic acid equivalents (GAE) per 100 g FW.
2.6. Chlorophylls a and b and Total Carotenoids Content
Chlorophylls a and b, and total carotenoids content were determined according to the method reported by Albanese et al. [
21]. Briefly, 2 g of sample were homogenized in 10 mL of acetone/water (80:20 v/v) and then centrifuged at 4000 r. The absorbance of the extract was measured at 646, 663, and 470 nm using a T60U spectrophotometer (PG Instruments, Leicestershire, UK).
2.7. Vicine and L-Dopa Content
Vicine and L-dopa content were determined as reported by Marquardt and Fröhlich [
22] modified by Rizzello et al. [
23]. Briefly, 5 g of samples were extracted by 50 mL of perchloric acid (5% w/v) by homogenizing for 5 minutes at 4 °C. The extract was centrifuged and filtered through a 0.22 μm filter (Millipore Co., Bedford, Massachusetts, USA) to remove suspended material before the analyses based on HPLC chromatograph (Agilent 1100 quaternary pump) equipped with a UV-visible detector (Agilent 1260—Agilent Technologies, Santa Clara, CA, USA) at 280 nm. The chromatographic conditions were as follows: column C18 (Supelcosil LC, 250 × 4.6 mm, 5 μm, Merck KGaA, Darmstadt, Germany) with binary elution gradient at 1.0 mL/min, based on water (H
2O) and acetonitrile (ACN), both containing 0.1% (v/v) of formic acid. The program adopted for the separation was: 0–2 min isocratic 0% ACN; 2–3 minutes from 0 to 10% ACN; 3–8 minutes isocratic at 10% ACN; 8–10 minutes from 10% to 90% ACN; 10–15 minutes at 90% ACN; 15–20 return to 0% ACN.
2.8. Statistical Analysis
A one-way analysis of variance (ANOVA) was performed to detect statistical significance using the GLM procedure (SAS software, Version 9.1, SAS Institute, Cary, North Carolina, USA) applying the statistical model for a randomized block design. The separation of means was obtained by the Student-Newman-Keuls (SNK) test.
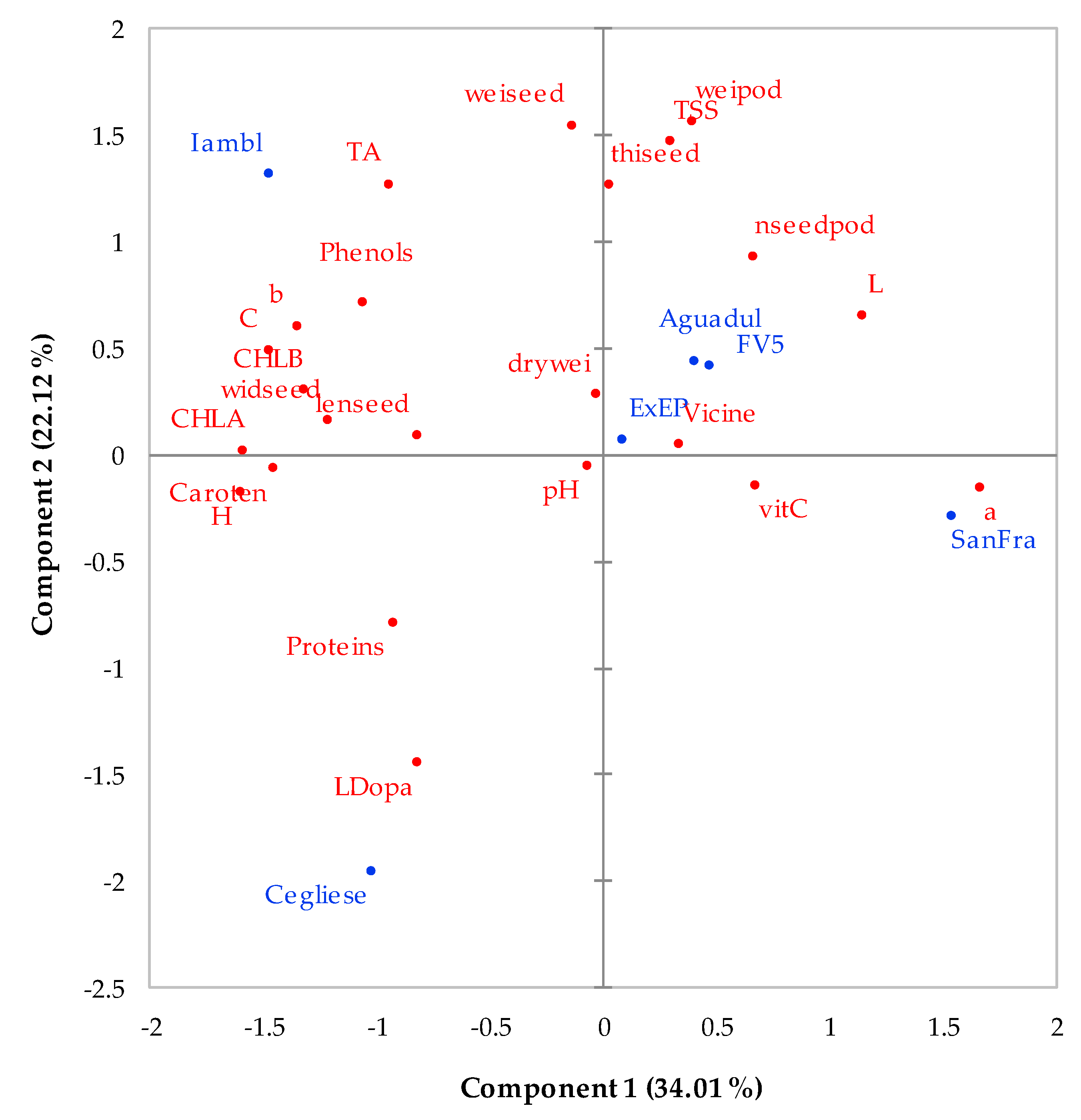
For a visual analysis of the data, principal component analysis (PCA) (PRINCOMP procedure, SAS software, Cary, NC, USA) was performed on mean centered and standardized (unit variance scaled) data prior to analysis. The data matrix submitted to PCA was made up of six observations (six genotypes—Aguadulce supersimonia, extra-early purple, Cegliese, San Francesco, Iambola, and FV5) and 23 variables (L, a*, b*, h°, C, dry weight, seed weight, pod weight, seed length, seed width, seed thickness, number of seeds per pod, pH, total phenols, crude protein, vitamin C, total carotenoids, chlorophyll a and b, vicine, L-dopa, titratable acidity, and total soluble solids). PCA was applied in order to obtain an interpretable overview of the data set variability. The results of the PCA are shown as biplot (XLStat, Addinsoft, Paris, France).
4. Discussion
In this study, an evaluation of some quality traits as well as the yield response in some landraces of faba bean for fresh consumption was carried out in order to assess and distinguish the landraces also in comparison with two common commercial varieties.
Results revealed that, independently of the genotype, it is possible to obtain the same pod yield without affecting the seeds size. In a study aimed to evaluate some genotypes of faba beans cultivated for fresh seed production, some authors [
24] showed that for the commercial variety Aquadulce by using about 9.5 plants m
−2 the pod yield was of about 2.8 kg m
−2, while the number of pods per plant was of about 10.7. In the present study, we used a plant density about 50% lower than that used by other authors [
24], and we obtained a pod yield of 2.1 kg m
−2 and about 22 pods plant
−1. Therefore, our results suggest the great influence of the plant density on pod yield, highlighting that a lower plant density causes a lower pod yield per area but a higher pod yield per plant. It is important to highlight that we decided to cultivate faba beans by using 4.16 plants m
−2 because this plant density is locally used for faba beans as a vegetable for fresh consumption. Moreover, it must be considered that this plant density allows farmers to better carry out the manual harvest and also promotes the optimal plant growth without using water as an external input for irrigation. Therefore, with the aim of producing faba beans as a vegetable for fresh consumption in a sustainable way, our results suggest the possibility to use local horticultural systems that are suited in optimizing resource utilization.
Color is an important quality trait of vegetables and also for faba beans. It is associated with the change in seeds from green to other colors, since chlorophyll breaks down and other pigments accumulate during ripening. Therefore, chlorophylls content can be considered the main factor affecting the color in fava beans for fresh consumption. In our study, we found a lower h° value for San Francesco in comparison with Aguadulce supersimonia and Iambola (
Figure 2). Hue angle represents the qualitative attribute according to which colors can be defined as green, yellow, etc. Hue angle values of 90 and 180 represent yellow and green hues, respectively. In our study, the h° value ranges between 111 and 113 (
Figure 2), so the seeds hues can be described between yellowish and slightly green. It is interesting to note that the trend of hue angle values was similar to the trend of chlorophyll a content, since, for this parameter, Iambola and Aguadulce supersimonia showed a higher value than other genotypes (
Table 6). Therefore, the lower h° value in San Francesco indicates a hue that is less green than other genotypes, probably due to its lower chlorophylls content. The trend for h° value and chlorophylls content was confirmed by PCA. Effectively,
Figure 3 shows that h° chlorophyll a and b were located close to each other and with the same positive direction as PC1, being located to the left in the biplot (
Figure 3). At the same time, Iambola and Cegliese, located both to the left in PC1 plot, were positively correlated with h° and chlorophylls, while San Francesco, located to the right in PC1 plot far from the origin, were inversely correlated with h° and chlorophylls (
Figure 3).
TSS and TA were distinctive traits for all the genotypes. Literature lacks information regarding these traits on faba beans as a vegetable. On the other hand, a high TSS value can be considered a positive qualitative trait which also may indicate the early ripening of the genotype. Effectively, for the commercial variety Extra-early purple, a specific genetic improvement program was probably conducted to obtain the early harvest of pods. In this context, it is interesting to note that the term “early” is inside the variety name and TSS value of this commercial variety is higher than Cegliese landrace (
Table 5). According to PCA, it is possible that the selection operated for Cegliese by farmers did not primarily consider this trait. Effectively, Cegliese is located in the lower left quadrant far from TSS, which is instead located in the upper right quadrant (
Figure 3). TA may be considered an important quality trait for processing faba beans because, like for other vegetables, the higher this value, the easier it is to control microbial deteriorations in processed products [
25] such as canned faba beans.
Table 5 shows a high TA value in Iambola, while PCA shows that this landrace is located close to TA parameter (
Figure 3). Therefore, our results suggest a good aptitude of Iambola as a processing vegetable.
From a nutritional point of view, our study shows that the Vitamin C content ranging between 386 and 703 mg 100 g
−1 FW (
Table 7). Vitamin C is an important antioxidant for human body and our results suggest that immature seeds of faba beans (for all genotypes) can be judged a very good source, considering its recommended daily intake of 45–70 mg [
26]. The ascorbic acid content in our study appear tens of times higher in comparison with that was reported from the National Nutrient Database of the United States Department of Agriculture (33 mg 100 g
−1 FW) [
27]. At the same time, the values of vitamin C found in our study are lower in comparison with results showed by other authors [
28]. These authors reported that, in
Vicia faba, during the seed development, great changes occur in the ascorbate system depending by embryogenesis stage. The activity of ascorbic peroxidase is high and remains constant when cells show both high mitotic activity and rapid cell elongation, but decreases when the water content in the seed begins to decrease until it disappears totally in the dried seed. Therefore, our results suggest the great importance of genotypes choose as well as the choose of optimal harvest time to obtain fava beans for fresh consumption with a high content of ascorbic acid.
Independently of the genotype, we found that the content of phenols, an important group of antioxidant compounds for human body, was of about 551 mg 100 g
−1 FW (
Table 7). In a study aimed to evaluate the phenols content in immature seeds from the 10 most widely consumed faba bean varieties in Chile, the authors [
29] measured a content of total phenols ranging from 82 to 134 mg 100 g
−1 FW. On the other hand, by measuring the total phenols content in mature dry seeds of
Vicia faba, other authors [
30] found an amount of about 49 mg 100 g
−1 (on dry weight basis). Our results highlight the high amount of total phenols in all studied genotypes, suggesting that the content of these compounds, which derived from the secondary plants metabolism, can be affected by the development stage (similarly to what reported for ascorbic acid content) and several other factors such as genotypes provenience and growing conditions.
Vicine is an antinutritional glucoside for human body, being a favism-inducing factor, and in our study its content was significantly different among the genotypes ranging from 366 to 509 mg 100 g
−1 FW (
Table 7). It is important to highlight that the amount of convicine, another favism-inducing factor, was not determined in this study, anyway it is well known that its content in faba beans is about half the vicine content [
31,
32]. Some authors [
32] reported a vicine content ranging from 65 to 759 mg 100 g
−1 DW in dry mature seeds of
Vicia faba, while other authors reported a vicine content ranging from 450 to 900 100 g
−1 DW on 78 faba bean genotypes [
33]. Moreover, in another study [
34] authors reported that the vicine content decreases rapidly during seed development, highlighting an inverse correlation between its content and dry matter. According to these authors, our results confirm the influence of the seed development stage on vicine content. At the same time, we observed a great variation in vicine content between commercial varieties as well as between some interesting differences between commercial varieties and two landraces. Effectively, although Aguadulce supersimonia showed the lowest vicine content, both Iambola and Cegliese showed a significant lower content of vicine compared to Extra-early purple. Therefore, our study suggests the great importance of genotype choice for obtaining fava beans as a vegetable for fresh consumption with a lower content of this favism-inducing factor.
As regards L-dopa, our study highlight that Cegliese was the genotype with the highest content (
Table 7). The ANOVA results regarding L-dopa content was confirmed by PCA. Effectively, both Cegliese and L-dopa were located with the same negative direction as both PC1 and PC2 (
Figure 3). Thus, it is possible to hypothesize that L-dopa was the leading factor affecting the separation between Cegliese and all other genotypes (
Figure 3). L-dopa, precursor of the dopamine, is a very important compound currently used in medicine for the Parkinson’s disease treatment [
10]. It is important to note that the use of natural sources of L-dopa is preferred instead of its synthesized counterpart, since the former allow to obtain the desired effects without side effects [
10]. In this context,
Vicia faba can be considered a very important species, given that it accumulates a large amount of L-dopa. Therefore, with the aim to obtain fava beans as a vegetable for fresh consumption with a high content of L-dopa, our results suggest the great importance to choose the optimal genotype.