Ecotone Dynamics and Stability from Soil Perspective: Forest-Agriculture Land Transition
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling and Processing
2.2. Physico-Chemical Analysis
- ΘMKK—maximum capillary water capacity (%)
- mMKK—weight of the artificially saturated sample after 2 h of draining off moisture (g)
- md—weight of the sample dried to the constant moisture (g)
- V—sample volume in Kopecky’s soil sample ring (cm3)
- AMKK = P − ΘMKK
- AMKK—minimum air capacity (%)
- P—porosity (%)
- ΘMKK—maximum capillary water capacity (%)
- P—porosity (%)
- —specific weight (g·cm3)
- —bulk density (g·cm3)
- —specific weight (g·cm3)
- m1—weight of soil sample dried to a constant weight (g)
- m2—weight of pycnometer with distilled water (g)
- m3—weight of pycnometer with the sample after boiling and distilled water (g)
- —bulk density (g·cm3)
- c—weight of Kopecky’s ring with lids and soil sample dried to a constant weight (g)
- a—weight of Kopecky’s soil sample ring with lids (g)
- V—volume of the Kopecky’s soil sample ring (cm3)
2.3. Statistical Analysis
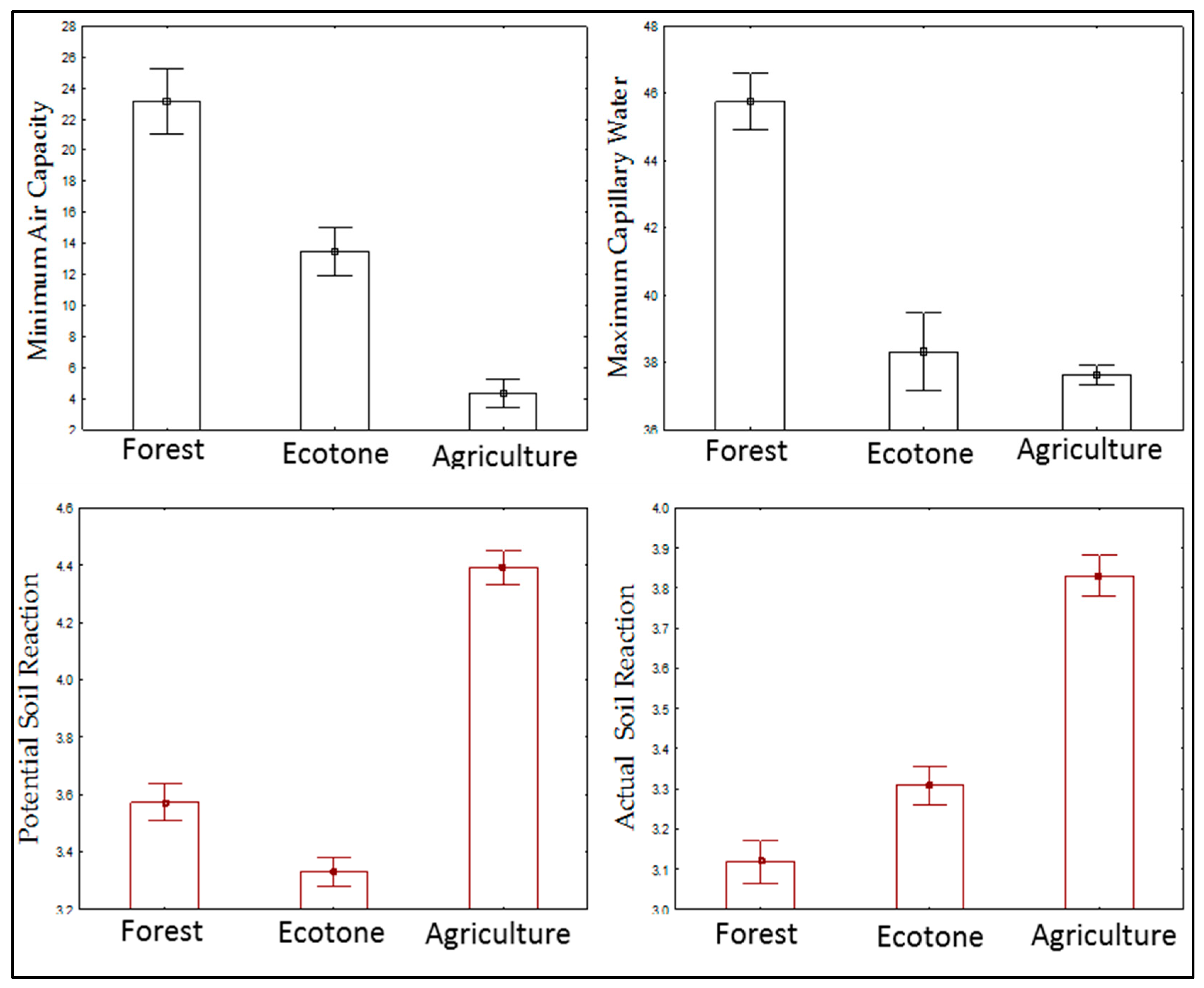
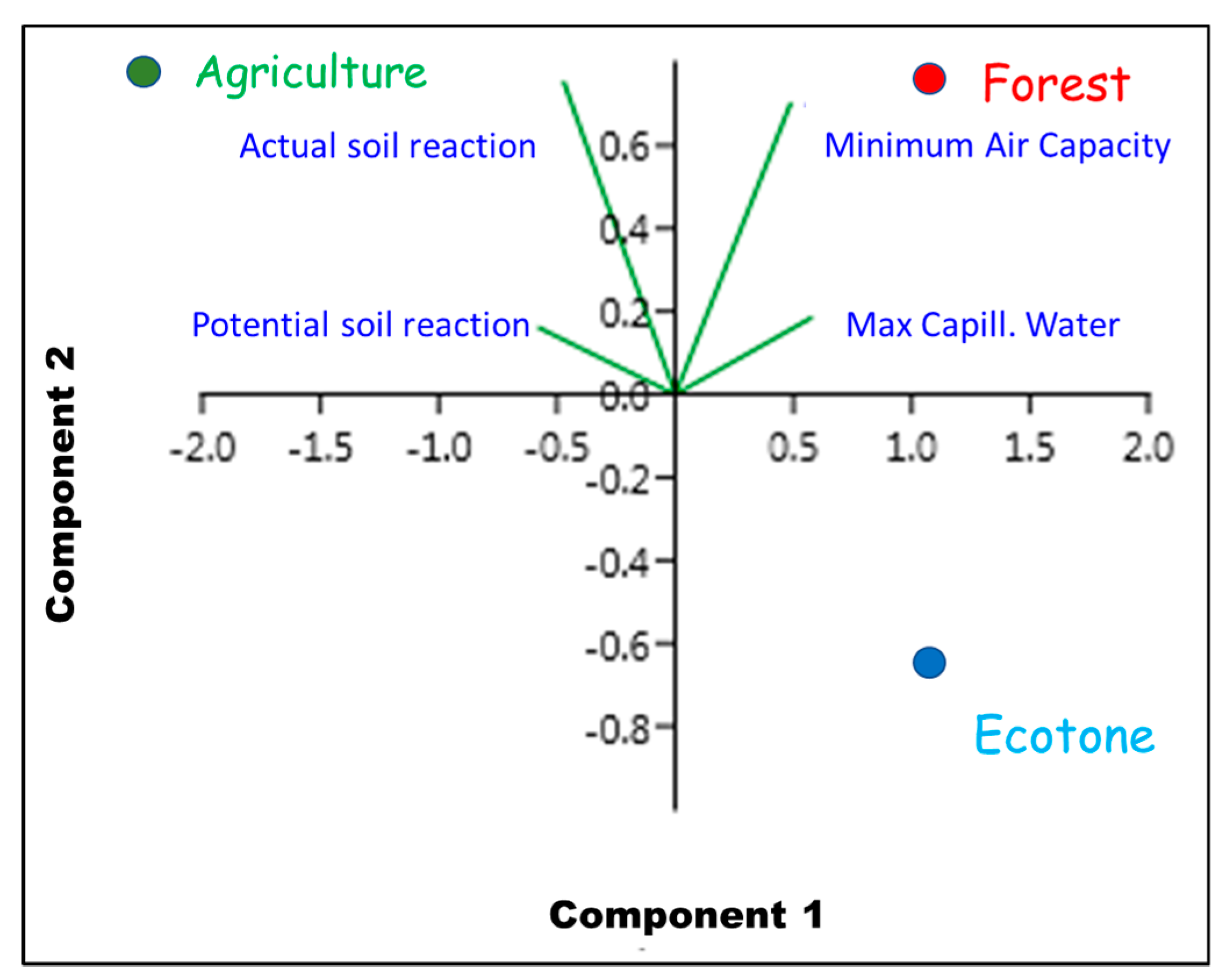
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ejsmont-Karabin, J. Is sandy beach of the lake an ecotone? Psammon Rotifera in a mesotrophic Lake Kuc (Masurian Lakeland, Northern Poland). Pol. J. Ecol. 2003, 51, 219–224. [Google Scholar]
- Hufkens, K.; Scheunders, P.; Ceulemans, R. Ecotones in vegetation ecology: Methodologies and definitions revisited. Ecol. Res. 2009, 24, 977–986. [Google Scholar] [CrossRef]
- Verry, E.S.; Dolloff, C.A.; Manning, M.E. Riparian ecotone: A functional definition and delineation for resource assessment. Water Air Soil Pollut. Focus 2004, 4, 67–94. [Google Scholar] [CrossRef]
- Bestelmeyer, B.T.; Ward, J.P.; Havstad, K.M. Soil-geomorphic heterogeneity governs patchy vegetation dynamics at an arid ecotone. Ecology 2006, 87, 963–973. [Google Scholar] [CrossRef]
- Casalini, A.I.; Bouza, P.J.; Bisigato, A.J. Geomorphology, soil and vegetation patterns in an arid ecotone. Catena 2019, 174, 353–361. [Google Scholar] [CrossRef]
- Marfo, T.D.; Datta, R.; Pathan, S.I.; Vranová, V. Ecotone Dynamics and Stability from Soil Scientific Point of View. Diversity 2019, 11, 53. [Google Scholar] [CrossRef]
- Marfo, T.D.; Resjek, K.; Vranova, V. Spatial variations in soil properties across ecotones: A short review. Bull. Geogr. Phys. Geogr. Ser. 2018, 14, 71–77. [Google Scholar] [CrossRef]
- Attua, E.M.; Pabi, O. Tree species composition, richness and diversity in the northern forest-savanna ecotone of Ghana. J. Appl. Biosci. 2013, 69, 5437–5448. [Google Scholar] [CrossRef]
- Hansen, A.J.; DiCastri, F. Landscape Boundaries: Consequences for Biotic Diversity and Ecological Flows; Springer Science & Business Media: Berlin, Germany, 2012; Volume 92. [Google Scholar]
- Hauck, M.; Javkhlan, S.; Lkhagvadorj, D.; Bayartogtokh, B.; Dulamsuren, C.; Leuschner, C. Edge and land-use effects on epiphytic lichen diversity in the forest-steppe ecotone of the Mongolian Altai. Flora-Morphol. Distrib. Funct. Ecol. Plants 2012, 207, 450–458. [Google Scholar] [CrossRef]
- Korpela, E.-L.; Hyvönen, T.; Kuussaari, M. Logging in boreal field-forest ecotones promotes flower-visiting insect diversity and modifies insect community composition. Insect Conserv. Divers. 2015, 8, 152–162. [Google Scholar] [CrossRef]
- Pe’er, G.; van Maanen, C.; Turbé, A.; Matsinos, Y.G.; Kark, S. Butterfly diversity at the ecotone between agricultural and semi-natural habitats across a climatic gradient. Divers. Distrib. 2011, 17, 1186–1197. [Google Scholar] [CrossRef]
- Ray, G.C.; Bruce, P.H. Coastal zone ecotones. In Landscape Boundaries; Springer: New York, NY, USA, 1992; pp. 403–420. [Google Scholar]
- Hansen, A.J.; Risser, P.G.; di Castri, F. Epilogue: Biodiversity and ecological flows across ecotones. In Landscape Boundaries; Springer: New York, NY, USA, 1992; pp. 423–438. [Google Scholar]
- Smith, T.B.; Wayne, R.K.; Girman, D.J.; Bruford, M.W. A role for ecotones in generating rainforest biodiversity. Science 1997, 276, 1855–1857. [Google Scholar] [CrossRef]
- Ward, J.V.; Tockner, K.; Schiemer, F. Biodiversity of floodplain river ecosystems: Ecotones and connectivity1. River Res. Appl. 1999, 15, 125–139. [Google Scholar] [CrossRef]
- Rhoades, C.C.; Miller, S.P.; Skinner, D.L. Forest vegetation and soil patterns across glade-forest ecotones in the Knobs region of northeastern Kentucky, USA. Am. Midl. Nat. 2005, 154, 1–11. [Google Scholar] [CrossRef]
- Burley, S.T.; Harper, K.A.; Lundholm, J.T. Vegetation composition, structure and soil properties across coastal forest-barren ecotones. Plant Ecol. 2010, 211, 279–296. [Google Scholar] [CrossRef]
- Alam, S.M.; Naqvi, S.S.M.; Ansari, R. Impact of soil pH on nutrient uptake by crop plants. Handb. Plant Crop Stress 1999, 2, 51–60. [Google Scholar]
- Schofield, R.K.; Taylor, A.W. The Measurement of Soil pH 1. SSSAJ 1955, 19, 164–167. [Google Scholar] [CrossRef]
- Harter, R.D. Effect of Soil pH on Adsorption of Lead, Copper, Zinc, and Nickel 1. SSSAJ 1983, 47, 47–51. [Google Scholar] [CrossRef]
- Bossuyt, B.; Hermy, M.; Deckers, J. Migration of herbaceous plant species across ancient-recent forest ecotones in central Belgium. J. Ecol. 1999, 87, 629–638. [Google Scholar] [CrossRef]
- Jiao, F. Changes in soil properties across chronosequence of vegetation restoration. Catena 2011, 82, 110–116. [Google Scholar] [CrossRef]
- Grable, A.R. Soil aeration and plant growth. In Advances in Agronomy; Academic Press: Cambridge, MA, USA, 1966; Volume 18, pp. 57–106. [Google Scholar]
- Lal, R. Soil structure and sustainability. J. Sustain. Agric. 1991, 1, 67–92. [Google Scholar] [CrossRef]
- Doran, J.W. Defining & assessing soil quality. In Defining Soil Quality for a Sustainable Environment Definingsoilqua; American Society of Agronomy: Madison, WI, USA, 1994; pp. 1–21. [Google Scholar]
- Holden, N.M. Description and classification of soil structure using distance transform data. Eur. J. Soil Sci. 2001, 52, 529–545. [Google Scholar] [CrossRef]
- Leopold, M.; Hürkamp, K.; Völkel, J.; Schmotz, K. Black soils, sediments and brown calcic luvisols: A pedological description of a newly discovered neolithic ring ditch system at Stephansposching, Eastern Bavaria, Germany. Quat. Int. 2011, 243, 293–304. [Google Scholar] [CrossRef]
- Schöning, I.; Kögel-Knabner, I. Chemical composition of young and old carbon pools throughout Cambisol and Luvisol profiles under forests. Soil Biol. Biochem. 2006, 38, 2411–2424. [Google Scholar] [CrossRef]
- Datta, R.; Vranová, V.; Pavelka, M.; Rejšek, K.; Formánek, P. Effect of soil sieving on respiration induced by low-molecular-weight substrates. Int. Agrophys. 2014, 28, 119–124. [Google Scholar] [CrossRef]
- ISO. ISO 10390: 2005 Soil Quality—Determination of pH 2005; ISO: Geneva, Switzerland, 2005. [Google Scholar]
- Winters, A.; Harland, M. Preparation of Soil Samples for Pipette Analysis. J. Am. Soc. Agron. 1930, 22, 771–780. [Google Scholar] [CrossRef]
- Folegatti, M.V.; Brasil, R.P.C.D.; Blanco, F.F. Sampling equipament for soil bulk density determination tested in a Kandiudalfic Eutrudox and a Typic Hapludox. Sci. Agric. 2001, 58, 833–838. [Google Scholar] [CrossRef][Green Version]
- Winters, A.; Harland, M.B. Preparation of Soil Samples for Pipette Analysis. 1930. Available online: https://dl.sciencesocieties.org/publications/aj/abstracts/22/9/AJ0220090771?access=0&view=pdf (accessed on 15 February 2019).
- Falkengren, G.U. Long-term changes in pH of forest soils in southern Sweden. Environ. Pollut. 1987, 43, 79–90. [Google Scholar] [CrossRef]
- Magiera, T.; Zawadzki, J. Using of high-resolution topsoil magnetic screening for assessment of dust deposition: Comparison of forest and arable soil datasets. Environ. Monit. Assess. 2007, 125, 19–28. [Google Scholar] [CrossRef]
- Balota, E.L.; Yada, I.F.U.; Amaral, H.F.; Nakatani, A.S.; Hungria, M.; Dick, R.P.; Coyne, M.S. Soil quality in relation to forest conversion to perennial or annual cropping in southern Brazil. Rev. Bras. Ciênc. Solo 2015, 39, 1003–1014. [Google Scholar] [CrossRef][Green Version]
- Rejšek, K. Lesnická Pedologie: Cvičení; Mendelova zemědělská a lesnická univerzita: Brně, Czechia, 1999; ISBN 978-80-7157-352-4. [Google Scholar]
- Lucas-Borja, M.E.; Zema, D.A.; Plaza-álvarez, P.A.; Zupanc, V.; Baartman, J.; Sagra, J.; González-Romero, J.; Moya, D.; de las Heras, J. Effects of different land uses (abandoned farmland, intensive agriculture and forest) on soil hydrological properties in Southern Spain. Water 2019, 11, 503. [Google Scholar] [CrossRef]
- Frazão, L.A.; de Cassia Piccolo, M.; Feigl, B.J.; Cerri, C.C.; Cerri, C.E.P. Inorganic nitrogen, microbial biomass and microbial activity of a sandy Brazilian Cerrado soil under different land uses. Agric. Ecosyst. Environ. 2010, 135, 161–167. [Google Scholar] [CrossRef]
- Molaei, A.; Lakzian, A.; Datta, R.; Haghnia, G.; Astaraei, A.; Rasouli-Sadaghiani, M.; Ceccherini, M.T. Impact of chlortetracycline and sulfapyridine antibiotics on soil enzyme activities. Int. Agrophys. 2017, 31, 499–505. [Google Scholar] [CrossRef]
- Molaei, A.; Lakzian, A.; Haghnia, G.; Astaraei, A.; Rasouli-Sadaghiani, M.; Ceccherini, M.T.; Datta, R. Assessment of some cultural experimental methods to study the effects of antibiotics on microbial activities in a soil: An incubation study. PLoS ONE 2017, 12, 180663. [Google Scholar] [CrossRef] [PubMed]
- Marfo, T.D.; Datta, R.; Lojkova, L.; Janous, D.; Pavelka, M.; Formanek, P. Limitation of Activity of Acid Phosphomonoesterase in Soils; Springer: Wien, Austria, 2015; Volume 47, p. 1691. [Google Scholar]
- Lojkova, L.; Datta, R.; Sajna, M.; Marfo, T.D.; Janous, D.; Pavelka, M.; Formanek, P. Limitation of Proteolysis in Soils of Forests and Other Types of Ecosystems by Diffusion of Substrate; Springer: Wien, Austria, 2015; Volume 47, pp. 1690–1691. [Google Scholar]
- Datta, R.; Anand, S.; Moulick, A.; Baraniya, D.; Pathan, S.I.; Rejsek, K.; Vranova, V.; Sharma, M.; Sharma, D.; Kelkar, A.; et al. How enzymes are adsorbed on soil solid phase and factors limiting its activity: A Review. Int. Agrophys. 2017, 31, 287–302. [Google Scholar] [CrossRef]
- Datta, R.; Kelkar, A.; Baraniya, D.; Molaei, A.; Moulick, A.; Meena, R.; Formanek, P. Enzymatic degradation of lignin in soil: A review. Sustainability 2017, 9, 1163. [Google Scholar] [CrossRef]
- Yadav, G.; Datta, R.; Imran Pathan, S.; Lal, R.; Meena, R.; Babu, S.; Das, A.; Bhowmik, S.; Datta, M.; Saha, P. Effects of Conservation Tillage and Nutrient Management Practices on Soil Fertility and Productivity of Rice (Oryza sativa L.)–Rice System in North Eastern Region of India. Sustainability 2017, 9, 1816. [Google Scholar] [CrossRef]
- Datta, R.; Baraniya, D.; Wang, Y.-F.; Kelkar, A.; Meena, R.; Yadav, G.; Teresa Ceccherini, M.; Formanek, P. Amino Acid: Its Dual Role as Nutrient and Scavenger of Free Radicals in Soil. Sustainability 2017, 9, 1402. [Google Scholar] [CrossRef]
- Munroe, J.S. Physical, chemical, and thermal properties of soils across a forest-meadow ecotone in the Uinta Mountains, Northeastern Utah, USA. Arct. Antarct. Alp. Res. 2012, 44, 95–106. [Google Scholar] [CrossRef]

| Factor | df | Potential Soil Reaction | Actual Soil Reaction | Maximum Capillary Water | Minimum Air Capacity |
|---|---|---|---|---|---|
| Time | 7 | 31.81*** | 31.07*** | 25.45*** | 61.24*** |
| Site | 2 | 396.51*** | 290.73*** | 950.10*** | 294.40*** |
| Time × Site | 14 | 1.87** | 6.21*** | 15.87*** | 17.15*** |
| Time | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| April | May | June | July | August | September | October | November | ||
| Potential Soil Reaction | Forest | 3.71 ± 0.05 | 3.63 ± 0.02 | 3.49 ± 0.03 | 3.55 ± 0.06 | 3.27 ± 0.04 | 3.45 ± 0.05 | 3.21 ± 0.01 | 4.24 ± 0.01 |
| Ecotone | 3.23 ± 0.07 | 3.43 ± 0.04 | 3.23 ± 0.06 | 3.33 ± 0.08 | 3.14 ± 0.04 | 3.36 ± 0.15 | 3.08 ± 0.04 | 3.81 ± 0.04 | |
| Agriculture Land | 4.44 ± 0.02 | 4.35 ± 0.09 | 4.29 ± 0.04 | 4.64 ± 0.10 | 4.1 ± 0.03 | 4.48 ± 0.2 | 4.01 ± 0.05 | 4.8 ± 0.03 | |
| Actual Soil Reaction | Forest | 2.66 ± 0.05 | 2.87 ± 0.06 | 3.1 ± 0.03 | 3.06 ± 0.05 | 2.98 ± 0.01 | 3.03 ± 0.05 | 2.94 ± 0.05 | 3.24 ± 0.05 |
| Ecotone | 2.48 ± 0.12 | 3.35 ± 0.07 | 3.15 ± 0.04 | 3.27 ± 0.03 | 3.16 ± 0.02 | 3.24 ± 0.13 | 3.07 ± 0.03 | 3.73 ± 0.04 | |
| Agriculture Land | 3.43 ± 0.02 | 3.75 ± 0.011 | 3.79 ± 0.06 | 3.75 ± 0.04 | 3.65 ± 0.02 | 3.75 ± 0.02 | 3.78 ± 0.05 | 3.71 ± 0.07 | |
| Minimum Air Capacity | Forest | 29.78 ± 1.2 | 22.25 ± 1.05 | 23.95 ± 2.9 | 28.4 ± 0.9 | 26.06 ± 0.17 | 17.07 ± 0.71 | 16.53 ± 0.52 | 21.1 ± 1.12 |
| Ecotone | 13.08 ± 0.46 | 18.96 ± 0.7 | 16.17 ± 0.52 | 12.65 ± 0.26 | 12.33 ± 0.48 | 6.33 ± 0.13 | 11.89 ± 0.14 | 16.51 ± 0.36 | |
| Agriculture Land | 4.42 ± 0.54 | 5.2 ± 0.022 | 3.22 ± 0.53 | 4.02 ± 0.28 | 2.21 ± 0.35 | 3.97 ± 0.40 | 3 ± 0.40 | 8.84 ± 0.92 | |
| Maximum Capillary Water | Forest | 45.73 ± 1.16 | 44.36 ± 0.34 | 48.93 ± 0.57 | 38.62 ± 0.21 | 40.79 ± 0.54 | 48.85 ± 0.49 | 50.01 ± 0.67 | 48.74 ± 1.52 |
| Ecotone | 33.57 ± 0.51 | 32.54 ± 1.60 | 44.61 ± 0.72 | 33.05 ± 0.52 | 32.77 ± 0.50 | 41.56 ± 0.32 | 44.15 ± 0.41 | 44.31 ± 0.62 | |
| Agriculture Land | 38.31 ± 1.17 | 38.3 ± 0.11 | 39.47 ± 0.42 | 35.63 ± 0.45 | 37.54 ± 0.74 | 38.03 ± 0.22 | 37.27 ± 0.83 | 36.53 ±0.63 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Danso Marfo, T.; Datta, R.; Vranová, V.; Ekielski, A. Ecotone Dynamics and Stability from Soil Perspective: Forest-Agriculture Land Transition. Agriculture 2019, 9, 228. https://doi.org/10.3390/agriculture9100228
Danso Marfo T, Datta R, Vranová V, Ekielski A. Ecotone Dynamics and Stability from Soil Perspective: Forest-Agriculture Land Transition. Agriculture. 2019; 9(10):228. https://doi.org/10.3390/agriculture9100228
Chicago/Turabian StyleDanso Marfo, Theodore, Rahul Datta, Valerie Vranová, and Adam Ekielski. 2019. "Ecotone Dynamics and Stability from Soil Perspective: Forest-Agriculture Land Transition" Agriculture 9, no. 10: 228. https://doi.org/10.3390/agriculture9100228
APA StyleDanso Marfo, T., Datta, R., Vranová, V., & Ekielski, A. (2019). Ecotone Dynamics and Stability from Soil Perspective: Forest-Agriculture Land Transition. Agriculture, 9(10), 228. https://doi.org/10.3390/agriculture9100228







