Gaseous Emissions after Soil Application of Pellet Made from Composted Pig Slurry Solid Fraction: Effect of Application Method and Pellet Diameter
Abstract
1. Introduction
2. Materials and Methods
2.1. Treatments and Experiment Setup
2.2. Gaseous Emissions Measurements
2.3. Statistical Analysis
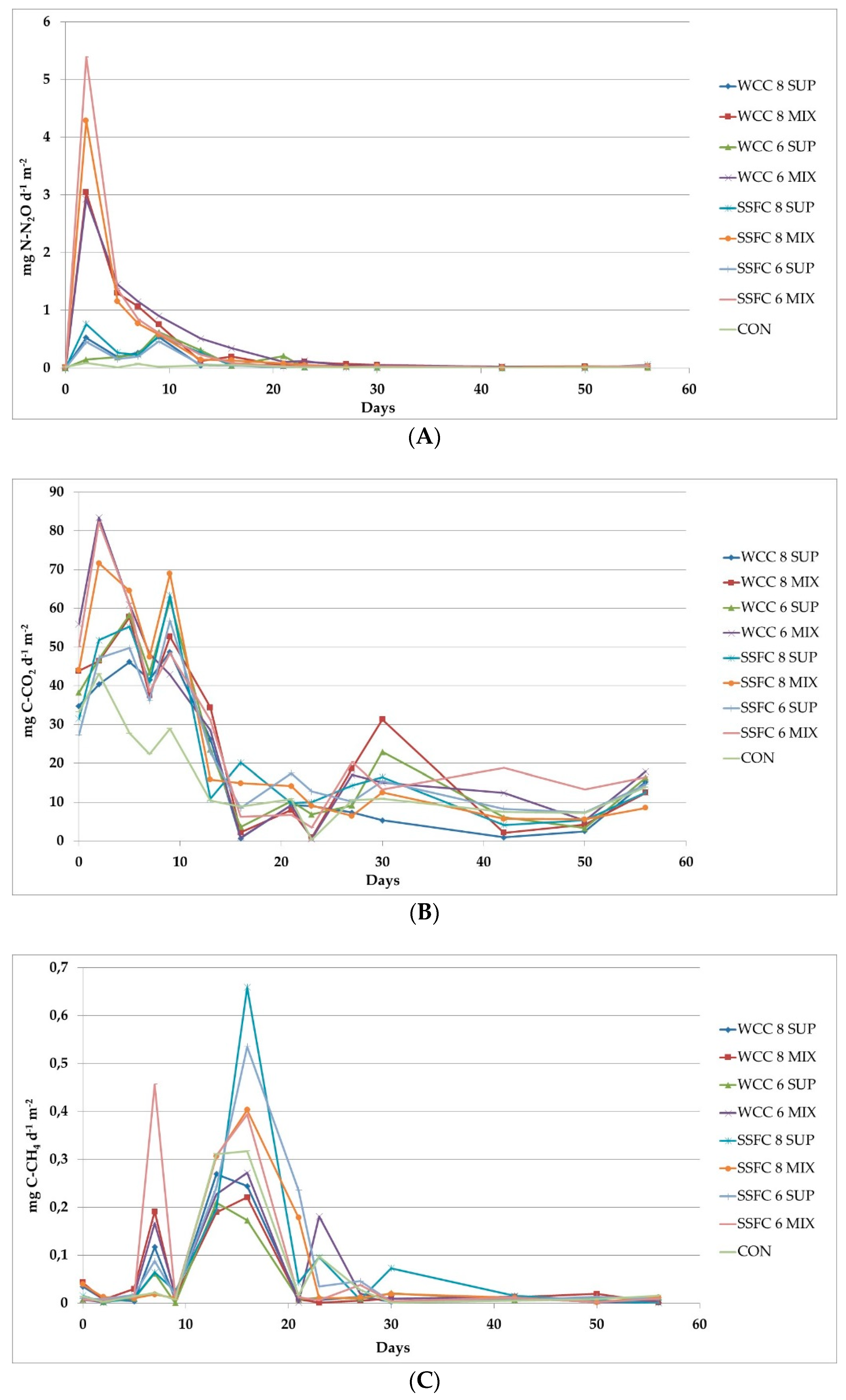
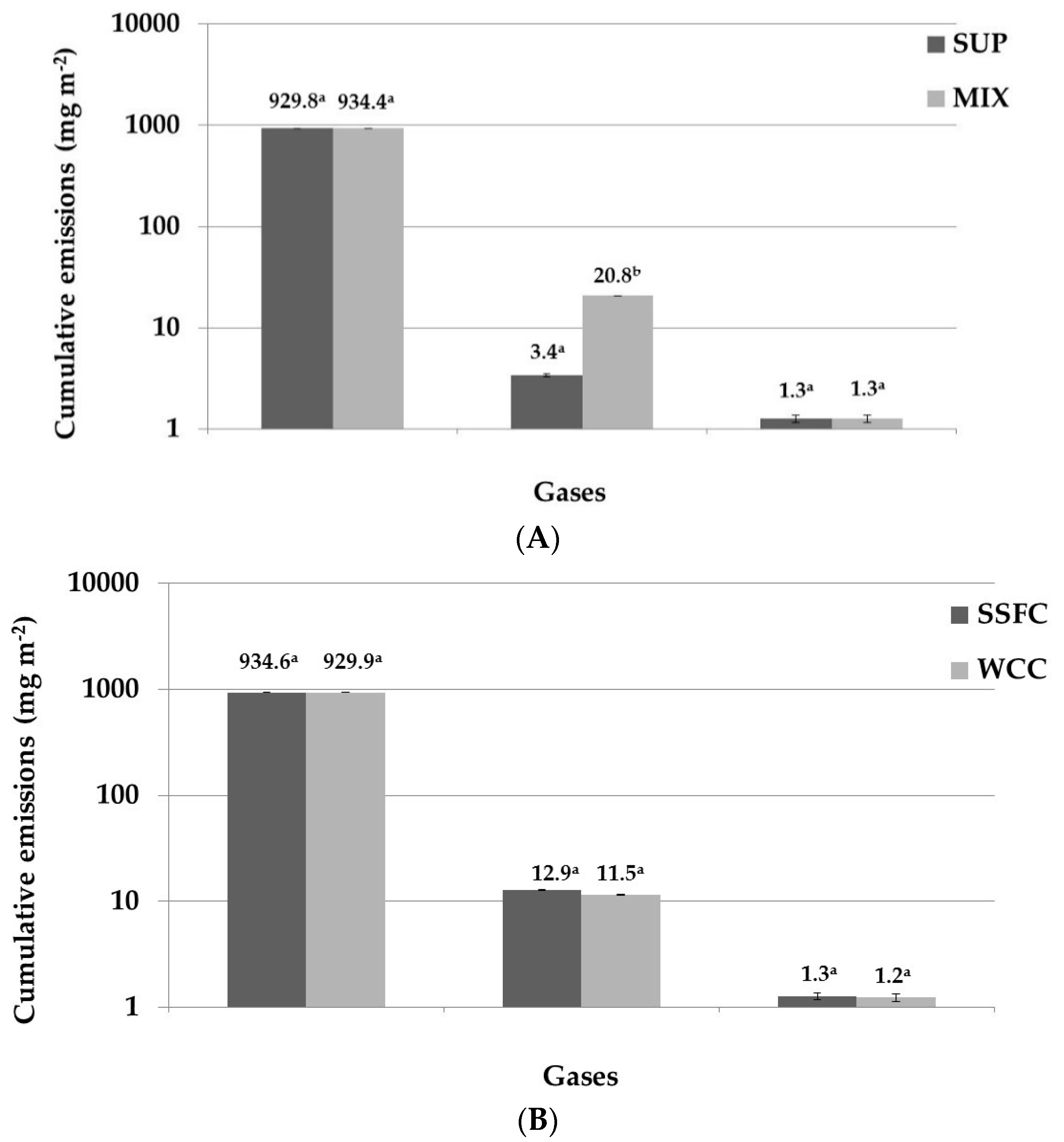
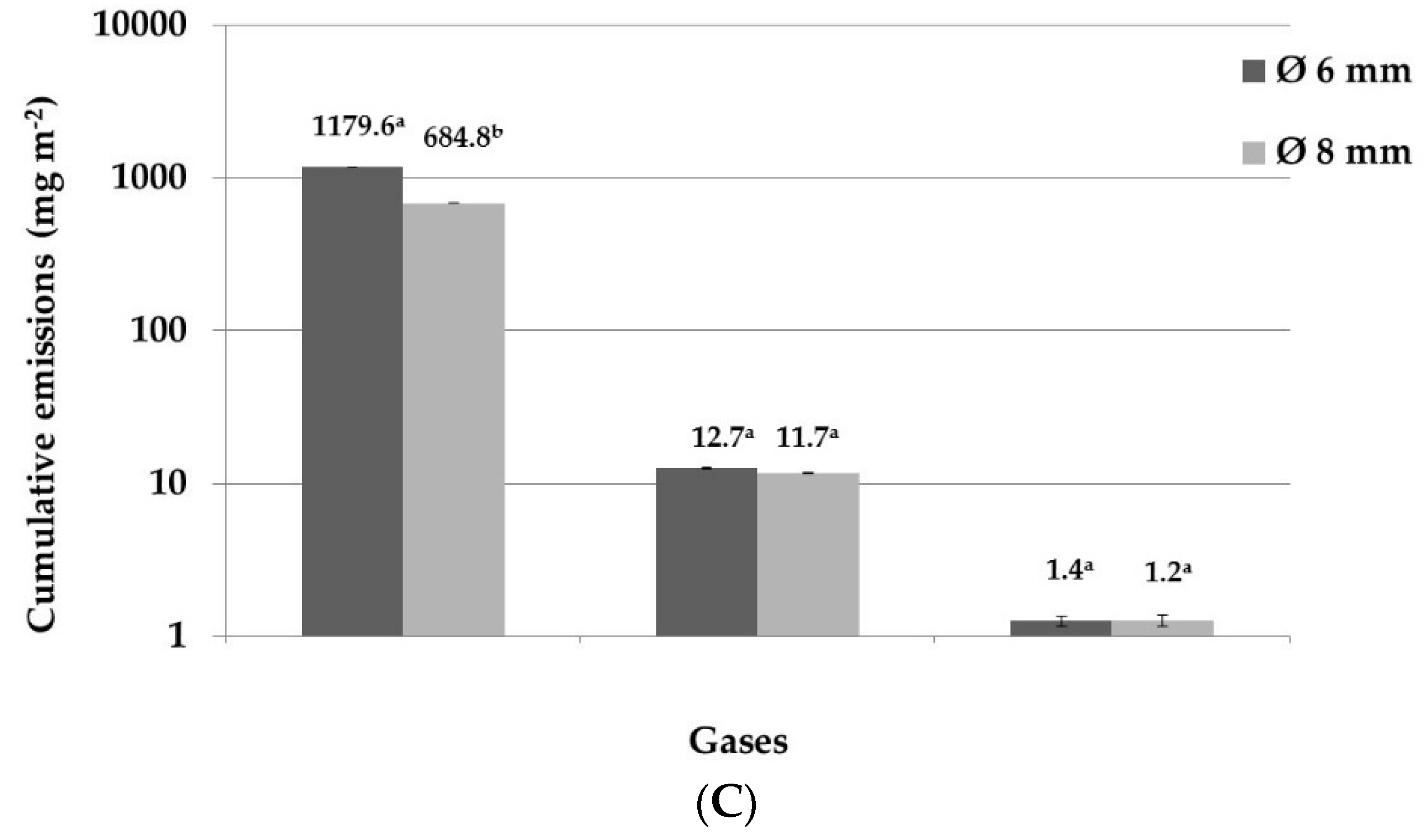
3. Results and Discussion
3.1. Nitrogen Emissions
3.2. Carbon Emissions
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- United States International Trade Commission. Pork and Swine. Industry & Trade Summary 2014. Publication ITS-11. Available online: http://www.usitc.gov/publications/332/pork_and_swine_summary_its_11.pdf (accessed on 11 May 2018).
- Segat, J.C.; Alves, P.R.L.; Baretta, D.; Cardoso, E.J.B.N. Ecotoxicological evaluation of swine manure disposal on tropical soils in Brazil. Ecotoxicol. Environ. Saf. 2015, 122, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Marquer, P.; Rabade, T.; Forti, R. Pig Farming Sector—Statistical Portrait 2014. Statistics in Focus. Available online: http://ec.europa.eu/eurostat/statistics-explained/index.php/Pig_farming_sector-statistical_portrait_2014 (accessed on 10 May 2018).
- Chen, K.; Wang, J. Hog farming in transition: The case of China. In Asian Livestock. Challenges opportunities and responses, Proceedings of the International Policy Forum, Bangkok, Thailand, 16–17 August 2012; FAO: Rome, Italy, 2012. [Google Scholar]
- ISTAT—Italian National Institute of Statistics. Preliminary Results of the 6th General Census of Agriculture. 2012. Available online: http://www.censimentoagricoltura.istat.it (accessed on 4 May 2018).
- Colonna, N.; Alfano, V. Quanto biogas si può produrre in Italia. L’Inf. Agrar.—Suppl. Energ. Rinnov. 2010, 22, 13–17. [Google Scholar]
- Kunz, A.; Miele, M.; Steinmetz, R.L.R. Advanced swine manure treatment and utilization in Brazil. Bioresour. Technol. 2009, 100, 5485–5489. [Google Scholar] [CrossRef] [PubMed]
- Salazar, F.J.; Chadwick, D.; Pain, B.F.; Hatch, D.; Owen, E. Nitrogen budgets for three cropping systems fertilized with cattle manure. Bioresour. Technol. 2005, 96, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Troy, S.M.; Lawlor, P.G.; O’Flynn, C.J.; Healy, M.G. Impact of biochar addition to soil on greenhouse gas emissions following pig manure application. Soil Biol. Biochem. 2013, 60, 173–181. [Google Scholar] [CrossRef]
- Vazquez, M.A.; De La Varga, D.; Plana, R.; Soto, M. Integrating liquid fraction of pig manure in the composting process for nutrient recovery and water re-use. J. Clean. Prod. 2015, 104, 80–89. [Google Scholar] [CrossRef]
- IPCC/WMO/UNEP. Climate Change 2007: Impacts, Adaptation, and Mitigation of Climate Change: Scientific-Technical Analyses; Prepared by IPCC Working Group III; Cambridge University Press: Cambridge, UK, 2007. [Google Scholar]
- Fangueiro, D.; Lopes, C.; Surgy, S.; Vasconcelos, E. Effect of the pig slurry separation techniques on the characteristics and potential availability of N to plants in the resulting liquid and solid fractions. Biosyst. Eng. 2012, 113, 187–194. [Google Scholar] [CrossRef]
- Pampuro, N.; Bisaglia, C.; Romano, E.; Brambilla, M.; Foppa Pedretti, E.; Cavallo, E. Phytotoxicity and chemical characterization of compost derived from pig slurry solid fraction for organic pellet production. Agriculture 2017, 7, 94. [Google Scholar] [CrossRef]
- Pampuro, N.; Facello, A.; Cavallo, E. Pressure and specific energy requirements for densification of compost derived from swine solid fraction. Span. J. Agric. Res. 2013, 11, 678–684. [Google Scholar] [CrossRef]
- Pampuro, N.; Bagagiolo, G.; Priarone, P.C.; Cavallo, E. Effects of pelletizing pressure and the addition of woody bulking agents on the physical and mechanical properties of pellets made from composted pig solid fraction. Powder Technol. 2017, 311, 112–119. [Google Scholar] [CrossRef]
- Kaliyan, N.; Vance Morey, R. Factors affecting strength and durability of densified biomass products. Biomass Bioenergy 2009, 33, 337–359. [Google Scholar] [CrossRef]
- Alemi, H.; Kianmehr, M.H.; Borghaee, A.M. Effect of pellet processing of fertilizer on slow-release nitrogen in soil. Asian J. Plant Sci. 2010, 9, 74–80. [Google Scholar] [CrossRef]
- Romano, E.; Brambilla, M.; Bisaglia, C.; Pampuro, N.; Foppa Pedretti, E.; Cavallo, E. Pelletization of composted swine manure solid fraction with different organic co-formulates: Effect of pellet physical properties on rotating spreader distribution patterns. Int. J. Recycl. Org. Waste Agric. 2014, 3, 101–111. [Google Scholar] [CrossRef]
- Pampuro, N.; Caffaro, F.; Cavallo, E. Reuse of Animal Manure: A Case Study on Stakeholders’ Perceptions about Pelletized Compost in Northwestern Italy. Sustainability 2018, 10, 2028. [Google Scholar] [CrossRef]
- Pampuro, N.; Dinuccio, E.; Balsari, P.; Cavallo, E. Gaseous emissions and nutrient dynamics during composting of swine solid fraction for pellet production. Appl. Math. Sci. 2014, 8, 6459–6468. [Google Scholar] [CrossRef]
- Pampuro, N.; Dinuccio, E.; Balsari, P.; Cavallo, E. Evaluation of two composting strategies for making pig slurry solid fraction suitable for pelletizing. Atmos. Pollut. Res. 2016, 7, 288–293. [Google Scholar] [CrossRef]
- Bernal, M.P.; Alburquerque, J.A.; Moral, R. Composting of animal manures and chemical criteria for compost maturity assessment. A review. Bioresour. Technol. 2009, 100, 5444–5453. [Google Scholar] [CrossRef] [PubMed]
- Amon, B.; Kryvoruchko, V.; Amon, T.; Zechmeister-Boltenstern, S. Methane, nitrous oxide and ammonia emissions during storage and after application of dairy cattle slurry and influence of slurry treatment. Agric. Ecosyst. Environ. 2006, 112, 153–162. [Google Scholar] [CrossRef]
- Fangueiro, D.; Senbayran, M.; Trindade, H.; Chadwick, D. Cattle slurry treatment by screw press separation and chemically enhanced settling: Effect on greenhouse gas emissions after land spreading and grass yield. Bioresour. Technol. 2008, 99, 7132–7142. [Google Scholar] [CrossRef] [PubMed]
- Zhu, K.; Christel, W.; Bruun, S.; Lensen, L.S. The different effects of applying fresh, composted or charred manure on soil N2O emissions. Soil Biol. Biochem. 2014, 74, 61–69. [Google Scholar] [CrossRef]
- Pampuro, N.; Preti, C.; Cavallo, E. Recycling pig slurry solid fraction compost as sound absorber. Sustainability 2018, 10, 277. [Google Scholar] [CrossRef]
- Navarro, A.F.; Cegarra, J.; Roig, A.; Garcia, D. Relationship between organic matter and carbon contents of organic wastes. Bioresour. Technol. 1993, 44, 203–207. [Google Scholar] [CrossRef]
- AOAC. Association of Official Analytical Chemist. Official Methods of Analysis, 15th ed.; Association of Official Analytical Chemist: Washington, DC, USA, 1990. [Google Scholar]
- Silber, A.; Bar-Yosef, B.; Levkovitch, I.; Soryano, S. pH-Dependent surface properties of perlite: Effects of plant growth. Geoderma 2010, 158, 275–281. [Google Scholar] [CrossRef]
- Garcia-Gomez, A.; Bernal, M.P.; Roig, A. Growth of ornamental plants in two composts prepared from agroindustrial wastes. Bioresour. Technol. 2002, 83, 81–87. [Google Scholar] [CrossRef]
- Subedi, R.; Kammann, C.; Pelissetti, S.; Sacco, D.; Grignani, C.; Monaco, S. Use of biochar and hydrochar to reduce ammonia emissions from soils fertilized with pig slurry. In Proceedings of the RAMIRAN 2013—15th International Conference, Versailles, France, 3–5 June 2013. [Google Scholar]
- Wu, Y.; Huang, M.; Warrington, D.N. Growth and transpiration of maize and winter wheat in response to water deficits in pots and plots. Environ. Exp. Bot. 2011, 71, 65–71. [Google Scholar] [CrossRef]
- Bateman, E.J.; Baggs, E.M. Contributions of nitrification and denitrification to N2O emissions. Biol. Fertil. Soils 2005, 41, 379–388. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Stocker, T.F., Qin, D., Plattner, G.-K., Tignor, M., Allen, S.K., Boschung, J., Nauels, A., Xia, Y., Bex, V., Midgley, P.M., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013; p. 1535. [Google Scholar]
- Fangueiro, D.; Pereira, J.; Chadwick, D.; Coutinho, J.; Moreira, N.; Trindade, H. Laboratory assessment of the effect of cattle slurry pre-treatment on organic N degradation after soil application and N2O and N2 emissions. Nutr. Cycl. Agroecosyst. 2008, 80, 107–120. [Google Scholar] [CrossRef]
- Sahrawat, K.L.; Keeney, D.R. Nitrous oxide emission from soils. Adv. Soil Sci. 1986, 4, 103–148. [Google Scholar]
- Bertora, C.; Alluvione, F.; Zavattaro, L.; van Groenigen, J.W.; Velthof, G.; Grignani, C. Pig slurry treatment modifies slurry composition, N2O and CO2 emissions after soil incorporation. Soil Biol. Biochem. 2008, 40, 1999–2006. [Google Scholar] [CrossRef]
- Ambus, P.; Jensen, E.S.; Robertson, G.P. Nitrous oxide and N-leaching losses from agricultural soil: Influence of crop residue particle size, quality and placement. Phyton Ann. Rei Bot. 2001, 41, 7–15. [Google Scholar]
- Calderon, F.J.; McCarty, G.W.; Van Kessel, J.A.S.; Reeves, J.B. Carbon and nitrogen dynamics in soil and plant as determined with N-15. Soil Sci. Soc. Am. J. 2004, 68, 1592–1599. [Google Scholar]
- Rochette, P.; Angers, D.A.; Cote, D. Soil carbon and nitrogen dynamics following application of pig slurry for the 19th consecutive year: I. Carbon dioxide fluxes and microbial biomass carbon. Soil Sci. Soc. Am. J. 2000, 64, 1389–1395. [Google Scholar] [CrossRef]
| Parameter | SSFC (Ø 6 mm and Ø 8 mm) | WCC (Ø 6 mm and Ø 8 mm) | ||
|---|---|---|---|---|
| Average | S.E. | Average | S.E. | |
| Dry Matter (%) | 85.4 | 0.7 | 84.6 | 0.4 |
| Moisture (%) | 14.6 | 0.7 | 15.4 | 0.4 |
| pH | 8.1 | 0.1 | 7.9 | 0.1 |
| TN (%) | 3.3 | 0.1 | 2.9 | 0.1 |
| NH4+-N (mg·kg−1) | 672.0 | 10.5 | 495.8 | 17.7 |
| NO3−-N (mg·kg−1) | 1460.0 | 13.8 | 2390.0 | 13.8 |
| TOC (%) | 36.9 | 0.4 | 38.1 | 0.2 |
| C/N | 11.2 | 0.3 | 13.2 | 0.3 |
| OM (%) | 63.6 | 1.5 | 65.7 | 0.5 |
| CEC (cmol·kg−1) | 70.9 | 1.7 | 79.5 | 4.2 |
| P2O5 (%) | 4.0 | 0.1 | 3.7 | 0.2 |
| K2O (%) | 1.0 | 0.1 | 1.6 | 0.1 |
| Parameter | Average | S.E. |
|---|---|---|
| pH | 8.55 | 0.01 |
| EC (dS m−1) | 0.18 | <0.01 |
| WHC (%) | 31.50 | 1.02 |
| CaCO3 (%) | 38.70 | 0.40 |
| CEC (cmol·kg−1) | 10.50 | 0.50 |
| OM (%) | 0.88 | 0.03 |
| TOC (%) | 0.51 | 0.01 |
| TN (%) | <0.01 | <0.01 |
| C/N | 7.29 | 0.09 |
| NH4+-N (mg·kg−1) | 10.80 | 0.80 |
| Available-P (mg·kg−1) | 27.70 | 0.50 |
| Gas | Units | SSFC | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Ø 6 mm | Ø 8 mm | ||||||||
| SUP | MIX | SUP | MIX | ||||||
| CO2 | mg CO2-C m−2 | 1170.34 a | (33.58) | 1101.55 a | (37.07) | 696.45 a | (25.68) | 770.11 a | (49.16) |
| mg CO2-eq m−2 (A) | 1170.34 | (33.58) | 1101.55 | (37.07) | 696.45 | (25.68) | 770.11 | (49.16) | |
| % TOC applied | 0.44 | (0.01) | 0.42 | (0.02) | 0.27 | (0.02) | 0.29 | (0.02) | |
| N2O | mg N2O-N m−2 | 2.70 a | (0.17) | 24.30 b | (1.19) | 4.56 a | (0.21) | 20.09 b | (0.62) |
| mg CO2-eq m−2 (B) | 715.50 | (30.73) | 6439.50 | (52.58) | 1208.40 | (37.36) | 5323.85 | (51.17) | |
| % TN applied | 0.01 | (<0.01) | 0.12 | (<0.01) | 0.02 | (<0.01) | 0.10 | (<0.01) | |
| CH4 | mg CH4-C m−2 | 1.28 a | (0.02) | 1.26 a | (0.02) | 1.31 a | (0.03) | 1.27 a | (0.03) |
| mg CO2-eq m−2 (C) | 35.84 | (1.31) | 35.28 | (1.32) | 36.68 | (1.39) | 35.56 | (1.24) | |
| % TOC applied | <0.01 | (<0.01) | <0.01 | (<0.01) | <0.01 | (<0.01) | <0.01 | (<0.01) | |
| Tot mg CO2-eq. m−2 (A + B + C) | 1921.68 a | (35.83) | 7576.33 b | (60.05) | 1941.53 a | (39.25) | 6129.52 b | (56.25) | |
| Gas | Units | WCC | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Ø 6 mm | Ø 8 mm | ||||||||
| SUP | MIX | SUP | MIX | ||||||
| CO2 | mg CO2-C m−2 | 1162.40 a | (32.25) | 1144.25 a | (31.17) | 670.90 a | (34.83) | 601.89 a | (39.02) |
| mg CO2-eq m−2 (A) | 1162.40 | (32.25) | 1144.25 | (31.17) | 670.90 | (35.68) | 601.89 | (39.02) | |
| % TOC applied | 0.37 | (0.02) | 0.36 | (0.02) | 0.22 | (0.02) | 0.19 | (0.02) | |
| N2O | mg N2O-N m−2 | 3.35 a | (0.17) | 20.36 b | (0.90) | 3.94 a | (0.11) | 18.30 b | (0.49) |
| mg CO2-eq m−2 (B) | 887.75 | (29.21) | 5395.40 | (42.23) | 1044.10 | (29.16) | 4849.50 | (49.82) | |
| % TN applied | 0.02 | (<0.01) | 0.10 | (<0.01) | 0.02 | (<0.01) | 0.09 | (<0.01) | |
| CH4 | mg CH4-C m−2 | 1.22 a | (<0.01) | 1.27 a | (0.02) | 1.25 a | (<0.01) | 1.23 a | (<0.01) |
| mg CO2-eq m−2 (C) | 34.16 | (1.12) | 35.56 | (1.28) | 35.00 | (1.09) | 34.44 | (1.18) | |
| % TOC applied | <0.01 | (<0.01) | <0.01 | (<0.01) | <0.01 | (<0.01) | <0.01 | (<0.01) | |
| Tot mg CO2-eq. m−2 (A + B + C) | 2084.31 a | (26.01) | 6575.21 b | (55.55) | 1750.00 a | (25.55) | 5485.83 b | (51.27) | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pampuro, N.; Busato, P.; Cavallo, E. Gaseous Emissions after Soil Application of Pellet Made from Composted Pig Slurry Solid Fraction: Effect of Application Method and Pellet Diameter. Agriculture 2018, 8, 119. https://doi.org/10.3390/agriculture8080119
Pampuro N, Busato P, Cavallo E. Gaseous Emissions after Soil Application of Pellet Made from Composted Pig Slurry Solid Fraction: Effect of Application Method and Pellet Diameter. Agriculture. 2018; 8(8):119. https://doi.org/10.3390/agriculture8080119
Chicago/Turabian StylePampuro, Niccolò, Patrizia Busato, and Eugenio Cavallo. 2018. "Gaseous Emissions after Soil Application of Pellet Made from Composted Pig Slurry Solid Fraction: Effect of Application Method and Pellet Diameter" Agriculture 8, no. 8: 119. https://doi.org/10.3390/agriculture8080119
APA StylePampuro, N., Busato, P., & Cavallo, E. (2018). Gaseous Emissions after Soil Application of Pellet Made from Composted Pig Slurry Solid Fraction: Effect of Application Method and Pellet Diameter. Agriculture, 8(8), 119. https://doi.org/10.3390/agriculture8080119







