Crop Systems, Quality and Protection of Diplotaxis tenuifolia
Abstract
1. Introduction
2. Notes on Taxonomy and Biology
3. Biochemical and Nutraceutical Properties
4. Uses of Product
5. Farming Practices
6. Diseases, Pests and Crop Protection
7. Industrial Processing
8. Conclusions
Author Contributions
Conflicts of Interest
References
- Parsons, W.T.; Cuthbertson, E.G. Noxious Weeds of Australia; Inkata Press: Melbourne, Australia, 1992; pp. 342–344. [Google Scholar]
- Bonasia, A.; Lazzizera, C.; Elia, A.; Conversa, G. Nutritional, biophysical and physiological characteristics of wild rocket genotypes as affected by soilless cultivation system, salinity level of nutrient solution and growing period. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Warwick, S.I.; Sauder, C.A. Phylogeny of tribe Brassiceae (Brassicaceae) based on chloroplast restriction site polymorphisms and nuclear ribosomal internal transcribed spacer and chloroplast trnL intron sequences. Can. J. Bot. 2005, 83, 467–483. [Google Scholar] [CrossRef]
- Martínez-Laborde, J.B. A brief account on the genus Diplotaxis. In Rocket: A Mediterranean Crop for the World; Padulosi, S., Pignone, D., Eds.; International Plant Genetic Resources Institute: Rome, Italy, 1996; pp. 13–22. [Google Scholar]
- Sobrino Vesperinas, E. Posición taxonómica de Diplotaxis cretacea Kotov (Cruciferae). Anales Jard. Bot. Madrid 1996, 54, 182–188. [Google Scholar]
- Martín, J.P.; Sánchez-Yélamo, M.D. Genetic relationships among species of the genus Diplotaxis (Brassicaceae) using inter-simple sequence repeat markers. Theor. Appl. Genet. 2000, 101, 1234–1241. [Google Scholar] [CrossRef]
- Bianco, V.V. Rocket, an ancient underutilized vegetable crop and its potential. In Rocket Genetic Resource Network; Padulosi, S., Ed.; International Plant Genetic Resources Institute: Rome, Italy, 1995; pp. 35–57. [Google Scholar]
- Eschmann-Grupe, G.; Neuffer, B.; Hurka, H. Extent and structure of genetic variation in two colonising Diplotaxis species (Brassicaceae) with contrasting breeding. Plant Syst. Evol. 2004, 244, 31–43. [Google Scholar] [CrossRef]
- Eschmann-Grupe, G.; Hurka, H.; Neuffer, B. Species relationships within Diplotaxis (Brassicaceae) and the phylogenetic origin of D. muralis. Plant Syst. Evol. 2004, 243, 13–29. [Google Scholar] [CrossRef]
- Koul, K.K.; Nagpal, R.; Raina, S.N. Seed coat microsculpturing in Brassica and allied genera (Subtribes Brassicinae, Raphaninae, Moricandiinae). Ann. Bot. 2000, 86, 385–397. [Google Scholar] [CrossRef]
- Ueno, O.; Wada, Y.; Wakai, M.; Bang, S.W. Evidence from photosynthetic characteristics for the hybrid origin of Diplotaxis muralis from a C3–C4 intermediate and a C3 species. Plant Biol. 2006, 8, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Apel, P.; Hillmer, S.; Pfeffer, M.; Muehle, K. Carbon metabolism type of Diplotaxis tenuifolia (L.) D.C. (Brassicaceae). Photosynthetica 1996, 32, 237–243. [Google Scholar]
- Pignone, D. Present status of rocket genetic resources and conservation activities. In Rocket: A Mediterranean Crop for the World. Report of a Workshop, Legnaro (Italy) 13–14 December 1996; Padulosi, S., Pignone, D., Eds.; International Plant Genetic Resources Institute: Rome, Italy, 1996; pp. 2–12. [Google Scholar]
- De Vos, A.C.; Broekman, R.; de Almeida Guerra, C.C.; van Rijsselberghe, M.; Rozema, J. Developing and testing new halophyte crops: A case study of salt tolerance of two species of the Brassicaceae, Diplotaxis tenuifolia and Cochlearia officinalis. Environ. Exp. Bot. 2013, 92, 154–164. [Google Scholar] [CrossRef]
- Choi, E.-Y.; McNeill, A.; Coventry, D.; Stangoulis, J. Whole plant response of crop and weed species to high subsoil boron. Aust. J. Agric. Res. 2006, 57, 761–770. [Google Scholar] [CrossRef]
- Hall, M.K.D.; Jobling, J.J.; Rogers, G.S. Factors affecting growth of perennial wall rocket and annual garden rocket. Int. J. Veg. Sci. 2012, 18, 393–411. [Google Scholar] [CrossRef]
- Erice, G.; Sanz-Sáez, A.; Aranjuelo, I.; Irigoyen, J.J.; Aguirreolea, J.; Avice, J.-C.; Sánchez-Díaz, M. Photosynthesis, N2 fixation and taproot reserves during the cutting regrowth cycle of alfalfa under elevated CO2 and temperature. J. Plant Physiol. 2011, 168, 2007–2014. [Google Scholar] [CrossRef] [PubMed]
- Hurka, H.; Bleeker, W.; Neuffer, B. Evolutionary processes associated with biological invasions in the Brassicaceae. Biol. Invasions 2003, 5, 281–292. [Google Scholar] [CrossRef]
- Giordano, S.; Molinaro, A.; Spagnuolo, V.; Muscariello, L.; Ferrara, R.; Cennamo, G.; Aliotta, G. In vitro allelopathic properties of wild rocket (Diplotaxis tenuifolia DC) extract and of its potential allelochemical S-glucopyranosyl thiohydroximate. J. Plant-Microbe Interact. 2005, 1, 51–60. [Google Scholar] [CrossRef]
- Caso, O. Fisiología de la regeneración de Diplotaxis tenuifolia (L.) D.C. B. Soc. Argent. Bot. 1972, 14, 335–346. [Google Scholar]
- Ziller, S.R.; Reaser, J.K.; Neville, L.E.; Brandt, K. Invasive Alien Species in South America: National Reports and Directory of Resources; Global Invasive Species Programme: Cape Town, South Africa, 2005. [Google Scholar]
- Brown, A. Ballast plants in New York City and its vicinity. Bull. Torrey Bot. Club 1879, 6, 353–360. [Google Scholar] [CrossRef]
- Rieger, M.A.; Preston, C.; Powles, S.B. Risks of gene flow from transgenic herbicide resistant canola (Brassica napus) to weedy relatives in southern Australia. Aust. J. Agric. Res. 1999, 50, 115–128. [Google Scholar] [CrossRef]
- Siemens, J. Interspecific hybridisation between wild relatives and Brassica napus to introduce new resistance traits into the oilseed rape gene pool. Czech J. Genet. Plant 2002, 38, 155–157. [Google Scholar] [CrossRef]
- Ozturk, M.; Sakcali, S.; Celik, A.A. Biomonitor of heavy metals on ruderal habitats in Turkey-Diplotaxis tenuifolia (L.) D.C. Sains Malays. 2013, 42, 1371–1376. [Google Scholar]
- Delaveau, P.; Paris, R. Sur la composition chimique de l’essence de Diplotaxis tenuifolia (L.) D.C. Ann. Pharm. Fr. 1958, 16, 81–86. [Google Scholar] [PubMed]
- Pasini, F.; Verardo, V.; Cerretani, L.; Caboni, M.F.; D’Antuono, L.F. Rocket salad (Diplotaxis and Eruca spp.) sensory analysis and relation with glucosinolate and phenolic content. J. Sci. Food Agric. 2011, 91, 2858–2864. [Google Scholar] [CrossRef] [PubMed]
- D’Antuono, L.F.; Elementi, S.; Neri, R. Exploring new potential health-promoting vegetables: Glucosinolates and sensory attributes of rocket salads and related Diplotaxis and Eruca species. J. Sci. Food Agric. 2009, 89, 713–722. [Google Scholar] [CrossRef]
- Larher, F.; Hamelin, J. 5-Dimethylsulfoniopentanoic acid from Diplotaxis tenuifolia. Phytochemistry 1979, 18, 1396–1397. [Google Scholar] [CrossRef]
- Hanson, A.D.; Huang, Z.H.; Gage, D.A. Evidence that the putative compatible solute 5-dimethylsulfoniopentanoate is an extraction artefact. Plant Physiol. 1993, 101, 1391–1393. [Google Scholar] [CrossRef] [PubMed]
- Fahey, J.W.; Zalemann, A.T.; Talalay, P. The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochemistry 2001, 56, 5–51. [Google Scholar] [CrossRef]
- Bell, L.; Wagstaff, C. Glucosinolates, myrosinase hydrolysis products, and flavonols found in rocket (Eruca sativa and Diplotaxis tenuifolia). J. Agric. Food Chem. 2014, 62, 4481–4492. [Google Scholar] [CrossRef] [PubMed]
- Deng, Q.; Zinoviadou, K.G.; Galanakis, C.M.; Orlien, V.; Grimi, N.; Vorobiev, E.; Lebovka, N.; Barba, F.J. The effects of conventional and non-conventional processing on glucosinolates and its derived forms, isothiocyanates: Extraction, degradation, and applications. Food Eng. Rev. 2015, 7, 357–381. [Google Scholar] [CrossRef]
- Omirou, M.; Papastylianou, I.; Iori, R.; Papastephanou, C.; Papadopoulou, K.K.; Ehaliotis, C.; Karpouzas, D.G. Microwave-assisted extraction of glucosinolates from Eruca sativa seeds and soil: Comparison with existing methods. Phytochem. Anal. 2009, 20, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Pedras, M.S.C.; Yaya, E.E. Tenualexin, other phytoalexins and indole glucosinolates from wild cruciferous species. Chem. Biodivers. 2014, 11, 910–918. [Google Scholar] [CrossRef] [PubMed]
- D’Antuono, L.F.; Elementi, S.; Neri, R. Glucosinolates in Diplotaxis and Eruca leaves: Diversity, taxonomic relations and applied aspects. Phytochemistry 2008, 69, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Nordmark, L.; Gertsson, U.; Olsson, K.; Olsson, M.E. Content of bioactive compounds in baby-leaves as affected by season and growth stage at harvest. In Proceedings of the III International Symposium on Human Health Effects of Fruits and Vegetables-FAVHEALTH, Avignon, France, 18–21 October 2009; Volume 1040, pp. 201–206. [Google Scholar]
- Bennett, R.N.; Rosa, E.A.; Mellon, F.A.; Kroon, P.A. Ontogenic profiling of glucosinolates, flavonoids, and other secondary metabolites in Eruca sativa (salad rocket), Diplotaxis erucoides (wall rocket), Diplotaxis tenuifolia (wild rocket), and Bunias orientalis (Turkish rocket). J. Agric. Food Chem. 2006, 54, 4005–4015. [Google Scholar] [CrossRef] [PubMed]
- Pasini, F.; Verardo, V.; Caboni, M.F.; D’Antuono, L.F. Determination of glucosinolates and phenolic compounds in rocket salad by HPLC-DAD–MS: Evaluation of Eruca sativa Mill. and Diplotaxis tenuifolia L. genetic resources. Food Chem. 2012, 133, 1025–1033. [Google Scholar] [CrossRef]
- Bell, L.; Oruna-Concha, M.J.; Wagstaff, C. Identification and quantification of glucosinolate and flavonol compounds in rocket salad (Eruca sativa, Eruca vesicaria and Diplotaxis tenuifolia) by LC–MS: Highlighting the potential for improving nutritional value of rocket crops. Food Chem. 2015, 172, 852–861. [Google Scholar] [CrossRef] [PubMed]
- Chun, J.H.; Arasu, M.V.; Lim, Y.P.; Kim, S.J. Variation of major glucosinolates in different varieties and lines of rocket salad. Hortic. Environ. Biotechnol. 2013, 54, 206–213. [Google Scholar] [CrossRef]
- Kovacic, M.; Veberic, R.; Ugrinovic, K.; Jakše, M. Glucosinolate analysis of wild rocket [Diplotaxis tenuifolia (L.) D.C.] from different Slovenian regions cultivated on two growing systems. Eur. J. Hortic. Sci. 2015, 80, 199–207. [Google Scholar] [CrossRef]
- Bennett, R.N.; Carvalho, R.; Mellon, F.A.; Eagles, J.; Rosa, E.A. Identification and quantification of glucosinolates in sprouts derived from seeds of wild Eruca sativa L. (salad rocket) and Diplotaxis tenuifolia L. (wild rocket) from diverse geographical locations. J. Agric. Food Chem. 2007, 55, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Talalay, P.; Cho, C.G.; Posner, G.H. A major inducer of anticarcinogenic protective enzymes from broccoli: Isolation and elucidation of structure. Proc. Natl. Acad. Sci. USA 1992, 89, 2399–2403. [Google Scholar] [CrossRef] [PubMed]
- Iori, R.; Bernardi, R.; Gueyrard, D.; Rollin, P.; Palmieri, S. Formation of glucoraphanin by chemoselective oxidation of natural glucoerucin: A chemoenzymatic route to sulforaphane. Bioorg. Med. Chem. Lett. 1999, 9, 1047–1048. [Google Scholar] [CrossRef]
- O’Hare, T.J.; Wong, L.S.; Irving, D.E. Asian and western horticultural species of the Brassica family with anti-cancer potential. Acta Hortic. 2005, 694, 457–462. [Google Scholar] [CrossRef]
- Cho, E.J.; Shin, J.S.; Chung, K.S.; Lee, Y.S.; Cho, Y.W.; Baek, N.I.; Chung, H.-G.; Lee, K.T. Arvelexin inhibits colonic inflammation by suppression of NF-kB activation in dextran sulfate sodium-induced mice and TNF-α-induced colonic epithelial cells. J. Agric. Food Chem. 2012, 60, 7398–7407. [Google Scholar] [CrossRef] [PubMed]
- Nitz, G.M.; Schnitzler, W.H. Variation der glucosinolatgehalte bei den rucolaarten Eruca sativa und Diplotaxis tenuifolia in abhängigkeit des ernteschnittes. J. Appl. Bot. 2002, 76, 82–86. [Google Scholar]
- Kirkegaard, J.A.; Sarwar, M. Biofumigation potential of brassicas. Plant Soil 1998, 201, 71–89. [Google Scholar] [CrossRef]
- Lu, P.; Gilardi, G.; Gullino, M.L.; Garibaldi, A. Biofumigation with Brassica plants and its effect on the inoculum potential of Fusarium yellows of Brassica crops. Eur. J. Plant Pathol. 2010, 126, 387–402. [Google Scholar] [CrossRef]
- Ntalli, N.; Caboni, P. A review of isothiocyanates biofumigation activity on plant parasitic nematodes. Phytochem. Rev. 2017, 16, 827–834. [Google Scholar] [CrossRef]
- Yulianti, T.; Sivasithamparam, K.; Turner, D.W. Response of different forms of propagules of Rhizoctonia solani AG2-1 (ZG5) exposed to the volatiles produced in soil amended with green manures. Ann. Appl. Biol. 2006, 148, 105–111. [Google Scholar] [CrossRef]
- Yulianti, T.; Sivasithamparam, K.; Turner, D.W. Saprophytic and pathogenic behaviour of R. solani AG2-1 (ZG-5) in a soil amended with Diplotaxis tenuifolia or Brassica nigra manures and incubated at different temperatures and soil water content. Plant Soil 2007, 294, 277–289. [Google Scholar] [CrossRef]
- Klein, E.; Katan, J.; Gamliel, A. Soil suppressiveness to Fusarium disease following organic amendments and solarization. Plant Dis. 2011, 95, 1116–1123. [Google Scholar] [CrossRef]
- Klein, E.; Katan, J.; Gamliel, A. Combining residues of herb crops with soil heating for control of soilborne pathogens in a controlled laboratory system. Crop Prot. 2011, 30, 368–374. [Google Scholar] [CrossRef]
- Klein, E.; Katan, J.; Gamliel, A. Soil suppressiveness to Meloidogyne javanica as induced by organic amendments and solarization in greenhouse crops. Crop Prot. 2012, 39, 26–32. [Google Scholar] [CrossRef]
- Pimpini, F.; Giannini, M.; Lazzarin, R. Ortaggi da Foglia e da Taglio; Veneto Agricoltura: Venezia, Italy, 2005; p. 118. [Google Scholar]
- Conforti, F.; Perri, V.; Menichini, F.; Marrelli, M.; Uzunov, D.; Statti, G.A.; Menichini, F. Wild Mediterranean dietary plants as inhibitors of pancreatic lipase. Phytother. Res. 2012, 26, 600–604. [Google Scholar] [CrossRef] [PubMed]
- Bruno, S.; Amico, A.; Stefanizzi, L. Vitamin C content of edible and medicinal plants of the Apulian region. Boll. Soc. Ital. Biol. Sper. 1980, 56, 2067–2070. [Google Scholar] [PubMed]
- Martínez-Sánchez, A.; Gil-Izquierdo, A.; Gil, M.I.; Ferreres, F. A comparative study of flavonoid compounds, vitamin C, and antioxidant properties of baby leaf Brassicaceae species. J. Agric. Food Chem. 2008, 56, 2330–2340. [Google Scholar] [CrossRef] [PubMed]
- National Research Council (US). Recommended Dietary Allowances, 10th ed.; National Academies Press: Washington, DC, USA, 1989; p. 285.
- Žnidarèiè, D.; Ban, D.; Šircelj, H. Carotenoid and chlorophyll composition of commonly consumed leafy vegetables in Mediterranean countries. Food Chem. 2011, 129, 1164–1168. [Google Scholar] [CrossRef] [PubMed]
- Salvatore, S.; Pellegrini, N.; Brenna, O.V.; Del Rio, D.; Frasca, G.; Brighenti, F.; Tumino, R. Antioxidant characterization of some Sicilian edible wild greens. J. Agric. Food Chem. 2005, 53, 9465–9471. [Google Scholar] [CrossRef] [PubMed]
- Durazzo, A.; Azzini, E.; Lazzè, M.C.; Raguzzini, A.; Pizzala, R.; Maiani, G. Italian wild rocket [Diplotaxis tenuifolia (L.) D.C.]: Influence of agricultural practices on antioxidant molecules and on cytotoxicity and antiproliferative effects. Agriculture 2013, 3, 285–298. [Google Scholar] [CrossRef]
- Maldoni, B.E.; Haurie, H.M.; Bartolome, E.R. Chemical study of weeds in the Buenos Aires province of Argentina. I. Diplotaxis tenuifolia. Anal. Assoc. Quím. Argent. 1988, 76, 237–240. [Google Scholar]
- Sánchez-Yélamo, M.D. A chemosystematic survey of flavonoids in the Brassicinae: Diplotaxis. Bot. J. Linn. Soc. 1994, 115, 9–18. [Google Scholar] [CrossRef]
- Heimler, D.; Isolani, L.; Vignolini, P.; Tombelli, S.; Romani, A. Polyphenol content and antioxidative activity in some species of freshly consumed salads. J. Agric. Food Chem. 2007, 55, 1724–1729. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Sánchez, A.; Llorach, R.; Gil, M.I.; Ferreres, F. Identification of new flavonoid glycosides and flavonoid profiles to characterize rocket leafy salads (Eruca vesicaria and Diplotaxis tenuifolia). J. Agric. Food Chem. 2007, 55, 1356–1363. [Google Scholar] [CrossRef] [PubMed]
- Truchado, P.; Tourn, E.; Gallez, L.M.; Moreno, D.A.; Ferreres, F.; Tomás-Barberán, F.A. Identification of botanical biomarkers in Argentinean Diplotaxis honeys: Flavonoids and glucosinolates. J. Agric. Food Chem. 2010, 58, 12678–12685. [Google Scholar] [CrossRef] [PubMed]
- Treutter, D. Significance of flavonoids in plant resistance and enhancement of their biosynthesis. Plant Biol. 2005, 7, 581–591. [Google Scholar] [CrossRef] [PubMed]
- Di Venere, D.; Calabrese, N.; Linsalata, V.; Cardinali, A.; Bianco, V.V. Influence of sowing time on phenolic composition of rocket. Acta Hortic. 2000, 533, 343–349. [Google Scholar] [CrossRef]
- Bouchereau, A.; Hamelin, J.; Lamour, I.; Renard, M.; Larher, F. Distribution of sinapine and related compounds in seeds of Brassica and allied genera. Phytochemistry 1991, 30, 1873–1881. [Google Scholar] [CrossRef]
- Brock, A.; Herzfeld, T.; Paschke, R.; Koch, M.; Draeger, B. Brassicaceae contain nortropane alkaloids. Phytochemistry 2006, 67, 2050–2057. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, S.A.; Vela Gurovic, M.S.; Mulet, M.C.; Murray, A.P. Diplotaxis tenuifolia (L.) D.C., a source of a potentially antifungal essential oil containing nitrile. Biochem. Syst. Ecol. 2006, 34, 353–355. [Google Scholar] [CrossRef]
- Dolya, V.S.; Koreshchuk, K.E.; Fursa, N.S.; Golodner, D.N.; Kaminskii, N.A. Oils from three representatives of Cruciferae family. Khimiya Prirodnykh Soedinenii 1972, 3, 386–387. [Google Scholar]
- Agullo, E.; Maldoni, B.E.; Rodriguez, M.S. Diplotaxis tenuifolia (wallrocket) seed. Extraction of the crude oil. Anal. Assoc. Quím. Argent. 1987, 75, 105–110. [Google Scholar]
- Tonguc, M.; Erbas, S. Evaluation of fatty acid compositions and some seed characters of common wild plant species of Turkey. Turk. J. Agric. For. 2012, 36, 673–679. [Google Scholar]
- Shoaib, A.; Saeed, G.; Ahmad, S. Antimicrobial activity and chemical analysis of some edible oils (Clove, Kalonji and Taramira). Afr. J. Biotechnol. 2014, 13, 4347–4354. [Google Scholar]
- De Feo, D.; Senatore, F.; De Feo, V. Medicinal plants and phytotherapy in the Amalfitan coast; Salerno province, Campania, southern Italy. J. Ethnopharmacol. 1993, 39, 39–51. [Google Scholar] [CrossRef]
- De Feo, V.; Aquino, R.; Menghini, A.; Ramundo, E.; Senatore, F. Traditional phytotherapy in the peninsula Sorrentina, Campania, Southern Italy. J. Ethnopharmacol. 1992, 36, 113–125. [Google Scholar] [CrossRef]
- Pieroni, A.; Quavec, C.L.; Santoro, R.F. Folk pharmaceutical knowledge in the territory of the Dolomiti Lucane, inland southern Italy. J. Ethnopharmacol. 2004, 95, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Caudron, D. Un mucolytique d’origine végétale Diplotaxis tenuifolia. Phytothérapie Européenne 2005, 25, 26–28. [Google Scholar]
- Nicola, S.; Hoeberechts, J.; Fontana, E. Comparison between traditional and soilless culture systems to produce rocket (Eruca sativa) with low nitrate content. Acta Hortic. 2005, 697, 549–555. [Google Scholar] [CrossRef]
- Cavarianni, R.L.; Cecílio Filho, A.B.; Cazetta, J.O.; May, A.; Corradi, M.M. Nutrient contents and production of rocket as affected by nitrogen concentrations in the nutritive solution. Sci. Agric. 2008, 65, 652–658. [Google Scholar] [CrossRef]
- Hall, M.; Jobling, J.; Rogers, G. Some perspectives on rocket as a vegetable crop: A review. Veg. Crops Res. Bull. 2012, 76, 21–41. [Google Scholar] [CrossRef]
- Schiattone, M.I.; Viggiani, R.; Di Venere, D.; Sergio, L.; Cantore, V.; Todorovic, M.; Perniola, M.; Candido, V. Impact of irrigation regime and nitrogen rate on yield, quality and water use efficiency of wild rocket under greenhouse conditions. Sci. Hortic. 2018, 229, 182–192. [Google Scholar] [CrossRef]
- Pimpini, F.; Enzo, M. Present status and prospects for rocket cultivation in the Veneto region. In Rocket: A Mediterranean Crop for the World. Report of a Workshop, Legnaro (Italy), 13–14 December 1996; Padulosi, S., Pignone, D., Eds.; International Plant Genetic Resources Institute: Rome, Italy, 1996; pp. 51–66. [Google Scholar]
- Bianco, V.V.; Boari, F. Up-to-date developments on wild rocket cultivation. In Rocket: A Mediterranean Crop for the World. Report of a Workshop, Legnaro (Italy) 13–14 December 1996; Padulosi, S., Pignone, D., Eds.; International Plant Genetic Resources Institute: Rome, Italy, 1996; pp. 41–49. [Google Scholar]
- Santamaria, P.; Gonnella, M.; Elia, A.; Parente, A.; Serio, F. Ways of reducing rocket salad nitrate content. Acta Hortic. 2001, 548, 529–536. [Google Scholar] [CrossRef]
- Caruso, G.; Conti, S.; La Rocca, G. Influence of crop cycle and nitrogen fertilizer form on yield and nitrate content in different species of vegetables. Adv. Hortic. Sci. 2011, 25, 81–89. [Google Scholar]
- Santamaria, P.; Elia, A.; Serio, F. Effect of solution nitrogen concentration on yield, leaf element content, and water and nitrogen use efficiency of three hydroponically-grown rocket salad genotypes. J. Plant Nutr. 2002, 25, 245–258. [Google Scholar] [CrossRef]
- Hu, Y.C.; Schmidhalter, U. Drought and salinity: A comparison of their effects on mineral nutrition of plants. J. Plant Nutr. Soil Sci. 2005, 168, 541–549. [Google Scholar] [CrossRef]
- Stefanelli, D.; Goodwin, I.; Jones, R. Minimal nitrogen and water use in horticulture: Effects on quality and content of selected nutrients. Food Res. Int. 2010, 43, 1833–1843. [Google Scholar] [CrossRef]
- Nicoletti, R.; Raimo, F.; Miccio, G.; Carella, A. Avversità biologiche della rucola selvatica (Diplotaxis tenuifolia). In Atti del Progetto ‘Analisi e Valutazione di Ordinamenti Colturali Alternativi Nelle Aree a Riconversione del Tabacco’; Consiglio per la Ricerca e la Sperimentazione in Agricoltura: Rome, Italy, 2007; pp. 298–304. [Google Scholar]
- Nicoletti, R.; Raimo, F.; Miccio, G. First report of Rhizoctonia solani on Diplotaxis tenuifolia in Italy. Plant Pathol. 2004, 53, 811. [Google Scholar] [CrossRef]
- Garibaldi, A.; Minuto, A.; Gullino, M.L. First report of Sclerotinia stem rot and watery soft rot caused by Sclerotinia sclerotiorum on sand rocket (Diplotaxis tenuifolia) in Italy. Plant Dis. 2005, 89, 1241. [Google Scholar] [CrossRef]
- Minuto, A.; Pensa, P.; Rapa, B.; Garibaldi, A. Sclerotinia sclerotiorum (Lib.) de Bary nuovo agente di alterazione basale e fogliare della Diplotaxis tenuifolia (L.) D.C. Inf. Fitopatol. 2005, 55, 43–45. [Google Scholar]
- Garibaldi, A.; Gilardi, G.; Gullino, M.L. Una tracheofusariosi su Eruca sativa e Diplotaxis sp. osservata per la prima volta in Europa. Inf. Fitopatol. 2002, 52, 57–59. [Google Scholar]
- Garibaldi, A.; Gilardi, G.; Gullino, M.L. First report of Fusarium oxysporum on Eruca vesicaria and Diplotaxis spp. in Europe. Plant Dis. 2003, 87, 201. [Google Scholar] [CrossRef]
- Garibaldi, A.; Gilardi, G.; Gullino, M.L. Evidence for an expanded host range of Fusarium oxysporum f. sp. raphani. Phytoparasitica 2006, 34, 115–121. [Google Scholar] [CrossRef]
- Catti, A.; Pasquali, M.; Ghiringhelli, D.; Garibaldi, A.; Gullino, M.L. Analysis of vegetative compatibility groups of Fusarium oxysporum from Eruca vesicaria and Diplotaxis tenuifolia. J. Phytopathol. 2007, 155, 61–64. [Google Scholar] [CrossRef]
- Gilardi, G.; Chen, G.; Garibaldi, A.; Zhiping, C.; Gullino, M.L. Resistance of different rocket cultivars to wilt caused by strains of Fusarium oxysporum under artificial inoculation conditions. J. Plant Pathol. 2007, 89, 113–117. [Google Scholar]
- Minuto, G.; Pensa, P.; Rapa, B.; Minuto, A.; Garibaldi, A. La peronospora della rucola selvatica [Diplotaxis tenuifolia (L.) D.C.] in Italia. Inf. Fitopatol. 2004, 54, 57–60. [Google Scholar]
- Garibaldi, A.; Minuto, A.; Gullino, M.L. First report of Peronospora parasitica on wild rocket (Diplotaxis tenuifolia) in Italy. Plant Dis. 2004, 88, 1381. [Google Scholar] [CrossRef]
- Choi, Y.-J.; Hong, S.-B.; Shin, H.-D. Genetic diversity within the Albugo candida complex (Peronosporales, Oomycota) inferred from phylogenetic analysis of ITS rDNA and COX2 mtDNA sequences. Mol. Phylogenet. Evol. 2006, 40, 400–409. [Google Scholar] [CrossRef] [PubMed]
- Ploch, S.; Choi, Y.J.; Rost, C.; Shin, H.D.; Schilling, E.; Thines, M. Evolution of diversity in Albugo is driven by high host specificity and multiple speciation events on closely related Brassicaceae. Mol. Phylogenet. Evol. 2010, 57, 812–820. [Google Scholar] [CrossRef] [PubMed]
- Gupta, K.; Prem, D.; Agnihotri, A. Role of biotechnology for incorporating white rust resistance in Brassica species. In Plant Biotechnology and Molecular Markers; Srivastava, P.S., Narula, A., Srivastava, S., Eds.; Kluwer Academic Publishers: Doordrecht, The Netherlands, 2004; pp. 156–168. [Google Scholar]
- Garibaldi, A.; Gilardi, G.; Bertoldo, C.; Gullino, M.L. First report of leaf spot of wild (Diplotaxis tenuifolia) and cultivated (Eruca vesicaria) rocket caused by Alternaria japonica in Italy. Plant Dis. 2011, 95, 1316. [Google Scholar] [CrossRef]
- Gilardi, G.; Demarchi, S.; Ortu, G.; Gullino, M.L.; Garibaldi, A. Occurrence of Alternaria japonica on seeds of wild and cultivated rocket. J. Phytopathol. 2015, 163, 419–422. [Google Scholar] [CrossRef]
- Klewer, A.; Mewes, S.; Mai, J.; Sacristán, M.D. Alternaria-resistenz in interspezifischen hybriden und deren rückkreuzungsnachkommenschaften im tribus Brassiceae. Vortr. Pflanzenschutzforschung-Zücht 2002, 54, 505–508. [Google Scholar]
- Sharma, G.; Dinesh Kumar, V.; Haque, A.; Bhat, S.R.; Prakash, S.; Chopra, V.L. Brassica coenospecies: A rich reservoir for genetic resistance to leaf spot caused by Alternaria brassicae. Euphytica 2002, 125, 411. [Google Scholar] [CrossRef]
- Garibaldi, A.; Gilardi, G.; Ortu, G.; Gullino, M.L. First report of Plectosphaerella cucumerina on greenhouse cultured wild rocket (Diplotaxis tenuifolia) in Italy. Plant Dis. 2012, 96, 1825. [Google Scholar] [CrossRef]
- Garibaldi, A.; Gilardi, G.; Ortu, G.; Gullino, M.L. First report of leaf spot of wild rocket (Diplotaxis tenuifolia) caused by Fusarium equiseti in Italy. Plant Dis. 2015, 99, 1183. [Google Scholar] [CrossRef]
- Gilardi, G.; Garibaldi, A.; Gullino, M.L. Seed transmission of Plectosphaerella cucumerina, causal agent of leaf spot of Diplotaxis tenuifolia in Italy. Phytoparasitica 2013, 41, 411–416. [Google Scholar] [CrossRef]
- Gilardi, G.; Pintore, I.; Gullino, M.L.; Garibaldi, A. Occurence of Fusarium equiseti as a contaminant of Diplotaxis tenuifolia seeds. J. Plant Pathol. 2017, 99, 245–248. [Google Scholar]
- Chen, C.Y.; Séguin-Swartz, G. Reaction of wild crucifers to Leptosphaeria maculans, the causal agent of blackleg of crucifers. Can. J. Plant Pathol. 1999, 21, 361–367. [Google Scholar] [CrossRef]
- Delourme, R.; Chévré, A.; Brun, H.; Rouxel, T.; Balesdent, M.; Dias, J.; Sailsbury, P.; Renard, M.; Rimmer, S. Major gene and polygenic resistance to Leptosphaeria maculans in oilseed rape (Brassica napus). Eur. J. Plant Pathol. 2006, 114, 41–52. [Google Scholar] [CrossRef]
- Voorips, R.E. Plasmodiophora brassicae: Aspects of pathogenesis and resistance in Brassica oleracea. Euphytica 1995, 83, 139–146. [Google Scholar] [CrossRef]
- Pennycook, S.R. Plant Diseases Recorded in New Zealand (2nd Vol); Plant Diseases Division, DSIR: Auckland, New Zealand, 1989; p. 502. [Google Scholar]
- Buser, H.; Heller, W.E. Falscher mehltau an rucola: Noch keine lösung des problems in sicht. Der Gemüsebau 2006, 69, 11–12. [Google Scholar]
- Westman, A.L.; Kresovich, S.; Dickson, M.H. Regional variation in Brassica nigra and other weedy crucifers for disease reaction to Alternaria brassicicola and Xanthomonas campestris pv. campestris. Euphytica 1999, 106, 253–259. [Google Scholar] [CrossRef]
- Raio, A.; Giorgini, M. Infezioni di Xantomonas campestris pv campestris in coltivazioni di rucola selvatica in Campania. Inf. Fitopatol. 2005, 55, 58–60. [Google Scholar]
- Pernezny, K.; Raid, R.N.; Jones, J.B.; Dickstein, E. First report of a leaf spot disease of wild rocket (Diplotaxis tenuifolia) in Florida caused by Xanthomonas campestris pv. raphani. Plant Dis. 2007, 91, 1360. [Google Scholar] [CrossRef]
- Amici, A.; Gracini, P.; Osler, R.; Refatti, E. Alterazioni a probabile eziologia da micoplasmi in varie specie di piante in Italia. Riv. Patol. Veg. 1972, 8, 21–50. [Google Scholar]
- Stavolone, L.; Alioto, D.; Ragozzino, A.; Laliberté, J.F. Variability among turnip mosaic potyvirus isolates. Phytopathology 1998, 88, 1200–1204. [Google Scholar] [CrossRef] [PubMed]
- Parrella, G.; Greco, B.; Troiano, E. First Report of Parietaria mottle virus associated with yellowing disease of Diplotaxis tenuifolia in Italy. Plant Dis. 2017, 101, 850. [Google Scholar] [CrossRef]
- Lupo, R.; Castellano, M.A.; Savino, V. Virus della Diplotaxis erucoides D.C. in Puglia. Inf. Fitopatol. 1991, 41, 42–44. [Google Scholar]
- Parrella, G.; Gognalons, P.; Gebre-Selassiè, K.; Vovlas, C.; Marchoux, G. An update of the host range of tomato spotted wilt virus. J. Plant Pathol. 2003, 85, 227–264. [Google Scholar]
- Gallitelli, D.; Rana, G.L.; Vovlas, C.; Martelli, G.P. Viruses of globe artichoke: An overview. J. Plant Pathol. 2004, 86, 267–281. [Google Scholar]
- Scholze, P.; Krämer, R.; Ryschka, U.; Klocke, E.; Schumann, G. Somatic hybrids of vegetable brassicas as source for new resistances to fungal and virus disease. Euphytica 2010, 176, 1–14. [Google Scholar] [CrossRef]
- Gómez-Merino, F.C.; Trejo-Téllez, L.I. Biostimulant activity of phosphite in horticulture. Sci. Hortic. 2015, 196, 82–90. [Google Scholar] [CrossRef]
- Gilardi, G.; Demarchi, S.; Gullino, M.L.; Garibaldi, A. Management of leaf spot of wild rocket using fungicides, resistance inducers and a biocontrol agent, under greenhouse conditions. Crop Prot. 2015, 71, 39–44. [Google Scholar] [CrossRef]
- Ciampolini, M.; Capella, A.; Farnesi, I. La tignola delle Crucifere insidia le coltivazioni di rucola. Inf. Agrar. 1998, 54, 69–72. [Google Scholar]
- Ciampolini, M.; Capella, A.; Farnesi, I.; Mozzo, G. Hellula undalis pericoloso fitofago della rucola. Inf. Agrar. 2001, 57, 69–73. [Google Scholar]
- Ciampolini, M.; Regalin, R.; Guarnone, A.; Farnesi, I. Diffusi e intensi attacchi di altiche (Phyllotreta spp.) su rucola. Inf. Agrar. 2001, 57, 87–91. [Google Scholar]
- Raimo, F.; Miccio, G.; Nicoletti, R.; Janssens, F. Danni da Collemboli su rucola selvatica (Diplotaxis tenuifolia). Inf. Fitopatol. 2005, 55, 44–47. [Google Scholar]
- Espinosa, B.; Sannino, L.; Conte, A.; Lista, A.; Del Grosso, M. Rucola non commerciabile per le mine di Scaptomyza flava. Inf. Agrar. 2010, 26, 60–61. [Google Scholar]
- Farnesi, I.; Massi, E.; Ciampolini, M. Tipici danni da sciaridi su colture di rucola in serra. Inf. Agrar. 2005, 16, 71–74. [Google Scholar]
- Solinas, M.; Bucci, M. An ecological investigation into flower galls on Diplotaxis muralis D.C. caused by the gall midge Paragephyraulus diplotaxis Solinas. Entomologica 1982, 17, 5–22. [Google Scholar]
- Skuhrava, M.; Skuhravy, V. Gall midges (Diptera: Cecidomyiidae) of Italy. Entomologica 1994, 28, 45–76. [Google Scholar]
- Shimat, J. Effects of insect growth regulators on Bagrada hilaris (Hemiptera: Pentatomidae). J. Econ. Entomol. 2017, 110, 2471–2477. [Google Scholar]
- Laffi, F. Infestations of the mite Bryobia praetiosa on Diplotaxis tenuifolia. Inf. Agrar. 2001, 57, 83–84. [Google Scholar]
- Giorgini, M. Un nuovo acaro dannoso alla coltura di rucola selvatica in Campania. Inf. Fitopatol. 2001, 51, 88–91. [Google Scholar]
- Vidovic, B.; Cvrkovic, T.; Maric, I.; Chetverikov, P.E.; Cristofaro, M.; Rector, B.G.; Petanovic, R. A new Metaculus species (Acari: Eriophyoidea) on Diplotaxis tenuifolia (Brassicaceae) from Serbia: A combined description using morphology and DNA barcode data. Ann. Entomol. Soc. Am. 2015, 108, 922–931. [Google Scholar] [CrossRef]
- Fuller, E.; Elderd, B.D.; Dwyer, G. Pathogen persistence in the environment and insect-baculovirus interactions: Disease-density thresholds, epidemic burnout and insect outbreaks. Am. Nat. 2012, 179, E70–E96. [Google Scholar] [CrossRef] [PubMed]
- Spadafora, N.D.; Amaro, A.L.; Pereira, M.J.; Müller, C.T.; Pintado, M.; Rogers, H.J. Multi-trait analysis of post-harvest storage in rocket salad (Diplotaxis tenuifolia) links sensorial, volatile and nutritional data. Food Chem. 2016, 211, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Cornacchia, R.; Rinaldi, R.; Quinto, M.; Colelli, G. Foglie di rucola: L’effetto sulla qualità della conservazione in AC. Colt. Prot. 2006, 35, 83–87. [Google Scholar]
- Luca, A.; Bach, V.; Edelenbos, M. Optimization of headspace solid-phase microextraction and static headspace sampling of low-boiling volatiles emitted from wild rocket (Diplotaxis tenuifolia L.). Food Anal. Methods 2015, 8, 1185–1196. [Google Scholar] [CrossRef]
- Luca, A.; Mahajan, P.V.; Edelenbos, M. Changes in volatile organic compounds from wild rocket (Diplotaxis tenuifolia L.) during modified atmosphere storage. Postharvest Biol. Technol. 2016, 114, 1–9. [Google Scholar] [CrossRef]
- Jin, J.; Koroleva, O.A.; Gibson, T.; Swanston, J.; Magan, J.; Zhang, Y.; Rowland, I.R.; Wagstaff, C. Analysis of phytochemical composition and chemoprotective capacity of rocket (Eruca sativa and Diplotaxis tenuifolia) leafy salad following cultivation in different environments. J. Agric. Food Chem. 2009, 57, 5227–5234. [Google Scholar] [CrossRef] [PubMed]
- Seefeldt, H.F.; Løkke, M.M.; Edelenbos, M. Effect of variety and harvest time on respiration rate of broccoli florets and wild rocket salad using a novel O2 sensor. Postharvest Biol. Technol. 2012, 69, 7–14. [Google Scholar] [CrossRef]
- Sinigaglia, M.; Albenzio, M.; Corbo, M.R. Influence of process operations on shelf-life and microbial population of fresh-cut vegetables. J. Ind. Microbiol. Biotechnol. 1999, 23, 484–488. [Google Scholar] [CrossRef]
- Galli, A.; Franzetti, L. La sicurezza igienico-sanitaria degli ortaggi di IV gamma. Inf. Agrar. 1998, 54, 46–47. [Google Scholar]
- Aruscavage, D.; Lee, K.; Miller, S.; Lejeune, J.T. Interactions affecting the proliferation and control of human pathogens on edible plants. J. Food Sci. 2006, 71, R89–R99. [Google Scholar] [CrossRef]
- Golberg, D.; Kroupitski, Y.; Belausov, E.; Pinto, R.; Sela, S. Salmonella typhimurium internalization is variable in leafy vegetables and fresh herbs. Int. J. Food Microbiol. 2011, 145, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Villari, P.; Crispino, M.; Montuori, P.; Stanzione, S. Prevalence and molecular characterization of Aeromonas spp. in ready-to-eat foods in Italy. J. Food Prot. 2000, 63, 1754–1757. [Google Scholar] [CrossRef] [PubMed]
- Marchetti, R.; Casadei, M.A.; Guerzoni, M.E. Microbial population dynamics in ready-to-use vegetable salads. Ital. J. Food Sci. 1992, 2, 97–108. [Google Scholar]
- Martínez-Sánchez, A.; Allende, A.; Bennett, R.N.; Ferreres, F.; Gil, M.I. Microbial, nutritional and sensory quality of rocket leaves as affected by different sanitizers. Postharvest Biol. Technol. 2006, 42, 86–97. [Google Scholar] [CrossRef]
- Kader, A.A. Biochemical and physiological basis for effect of controlled and modified atmosphere on fruit and vegetables. Food Technol. 1986, 40, 99–100. [Google Scholar]
- Martínez-Sánchez, A.; Marín, A.; Llorach, R.; Ferreres, F.; Gil, M.I. Controlled atmosphere preserves quality and phytonutrients in wild rocket (Diplotaxis tenuifolia). Postharvest Biol. Technol. 2006, 40, 26–33. [Google Scholar] [CrossRef]

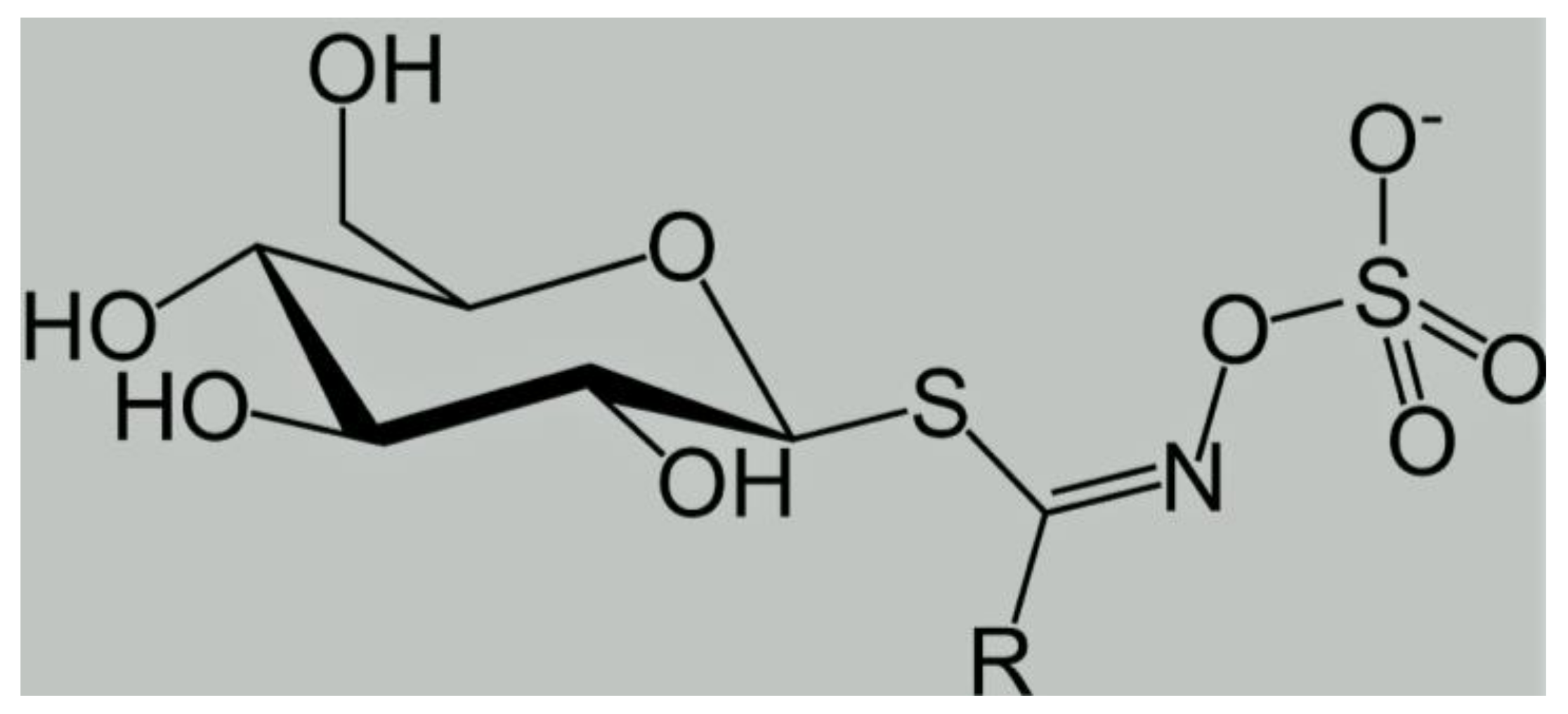
| Compound Name | Chemical Name (Based on the Aglycone Moiety) | References |
|---|---|---|
| Diglucothiobeinin | 4-glucopyranosyldisulfanylbutyl-GI | [39,41] |
| Glucoalyssin | 5-methylsulfinylpentyl-GI | [36,39,41,42] |
| 3-hydroxy-5-methylsulfinylpentyl-GI | [39] | |
| Glucoarmoracialafolicin | 4-phenylbutyl-GI | [36] |
| Glucobrassicanapin | 4-pentenyl-GI | [41] |
| Glucobrassicin | 3-indolylmethyl-GI | [35,36,39,41] |
| 4-hydroxy-3-indolylmethyl-GI | [36,39,40] | |
| 4-methoxy-3-indolylmethyl-GI | [35,36,38,41] | |
| Neoglucobrassicin | 1-methoxy-3-indolylmethyl-GI | [27,36] |
| Glucoerucin | 4-methylthiobutyl-GI | [36,38,39,40,41,42] |
| Glucoibarin | 7-methylsulfinylheptyl-GI | [40] |
| Glucoiberverin | 3-methylthiopropyl-GI | [40] |
| Glucolepidiin | ethyl-GI | [36,40] |
| Gluconapin | 3-butenyl-GI | [42] |
| Gluconasturtiin | 2-phenethyl-GI | [36,41] |
| Glucoputranjivin | 1-methylethyl-GI | [36] |
| Glucoraphanin | 4-methylsulfinylbutyl-GI | [36,38,39,40,41,42] |
| Glucoraphenin | 4-methylsulfinyl-3-butenyl-GI | [40] |
| Glucosativin | 4-mercaptobutyl-GI | [38,40,42,43] |
| dimeric 4-mercaptobutyl-desulfo-GI | [39,40] | |
| Glucotropaeolin | benzyl-GI | [36,40] |
| 2-benzoyloxyethyl-GI | [39] | |
| Progoitrin/epiprogoitrin | 2-hydroxy-3-butenyl-GI | [39] |
| Sinalbin, glucosinalbin | 4-hydroxybenzyl-GI | [36,38,39] |
| Sinigrin | 2-propenyl-GI (allyl-GI) | [43] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caruso, G.; Parrella, G.; Giorgini, M.; Nicoletti, R. Crop Systems, Quality and Protection of Diplotaxis tenuifolia. Agriculture 2018, 8, 55. https://doi.org/10.3390/agriculture8040055
Caruso G, Parrella G, Giorgini M, Nicoletti R. Crop Systems, Quality and Protection of Diplotaxis tenuifolia. Agriculture. 2018; 8(4):55. https://doi.org/10.3390/agriculture8040055
Chicago/Turabian StyleCaruso, Gianluca, Giuseppe Parrella, Massimo Giorgini, and Rosario Nicoletti. 2018. "Crop Systems, Quality and Protection of Diplotaxis tenuifolia" Agriculture 8, no. 4: 55. https://doi.org/10.3390/agriculture8040055
APA StyleCaruso, G., Parrella, G., Giorgini, M., & Nicoletti, R. (2018). Crop Systems, Quality and Protection of Diplotaxis tenuifolia. Agriculture, 8(4), 55. https://doi.org/10.3390/agriculture8040055








