Net Greenhouse Gas Budget and Soil Carbon Storage in a Field with Paddy–Upland Rotation with Different History of Manure Application
Abstract
:1. Introduction
2. Materials and Methods
2.1. Site Description and Plant Cultivation
2.2. CH4 and N2O Fluxes
2.3. Soil Carbon Storage and Decrease Rate
2.4. Net Greenhouse Gas Balance
2.5. Statistical Analyses
3. Results
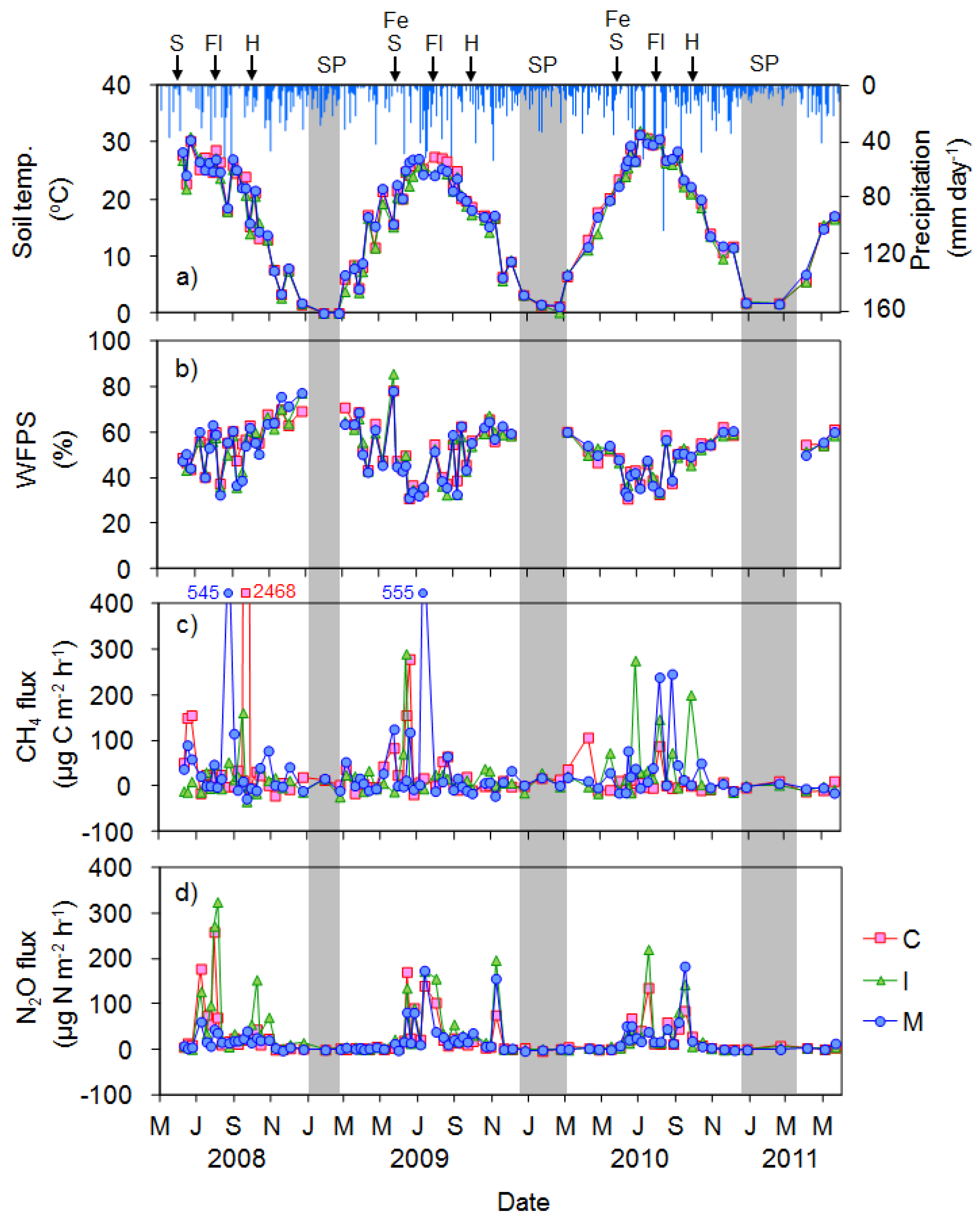
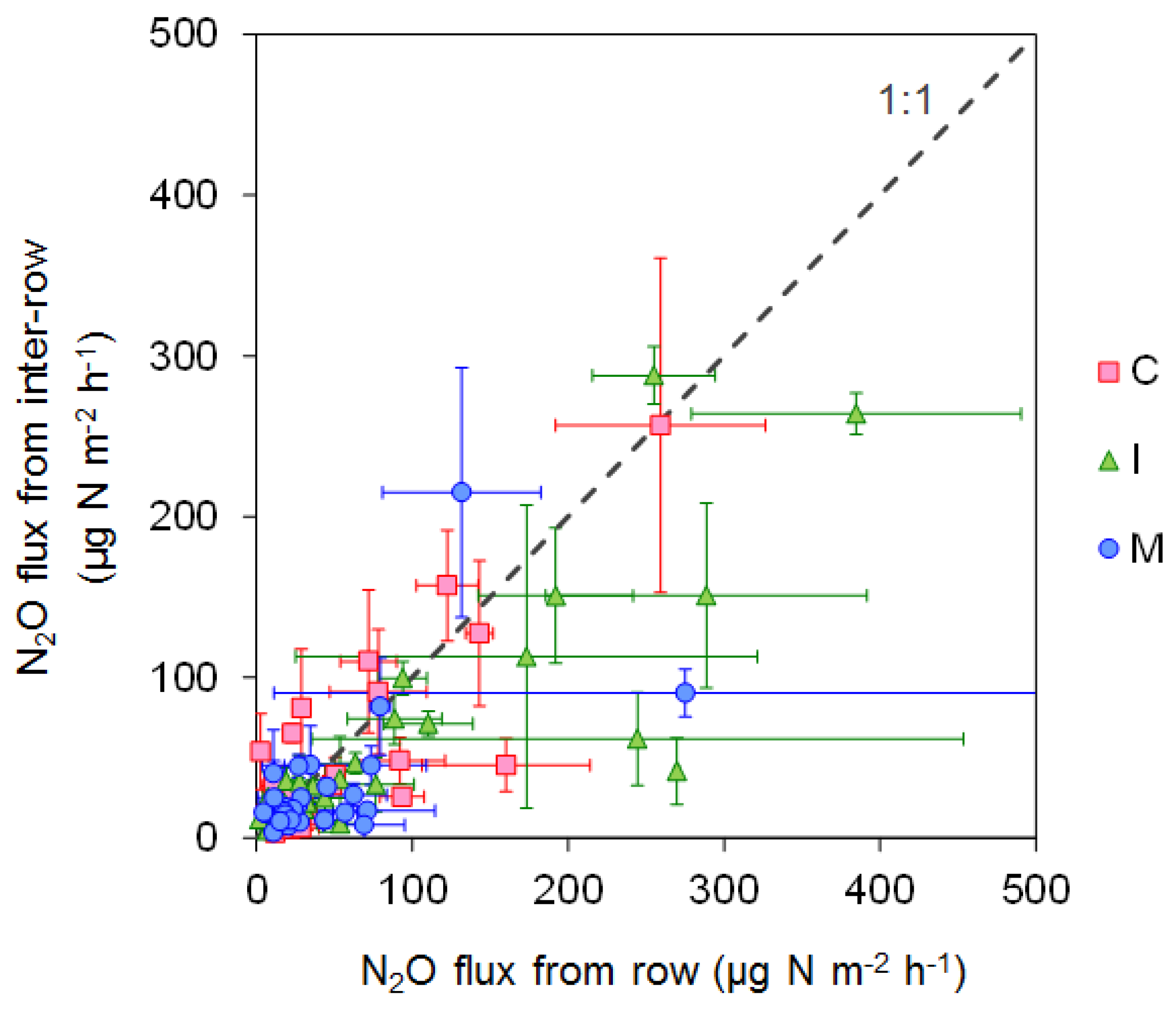
3.1. GHG Emissions from the Upland Soybean Field
3.2. GHG Emissions from the Rice Cultivated Paddy Field
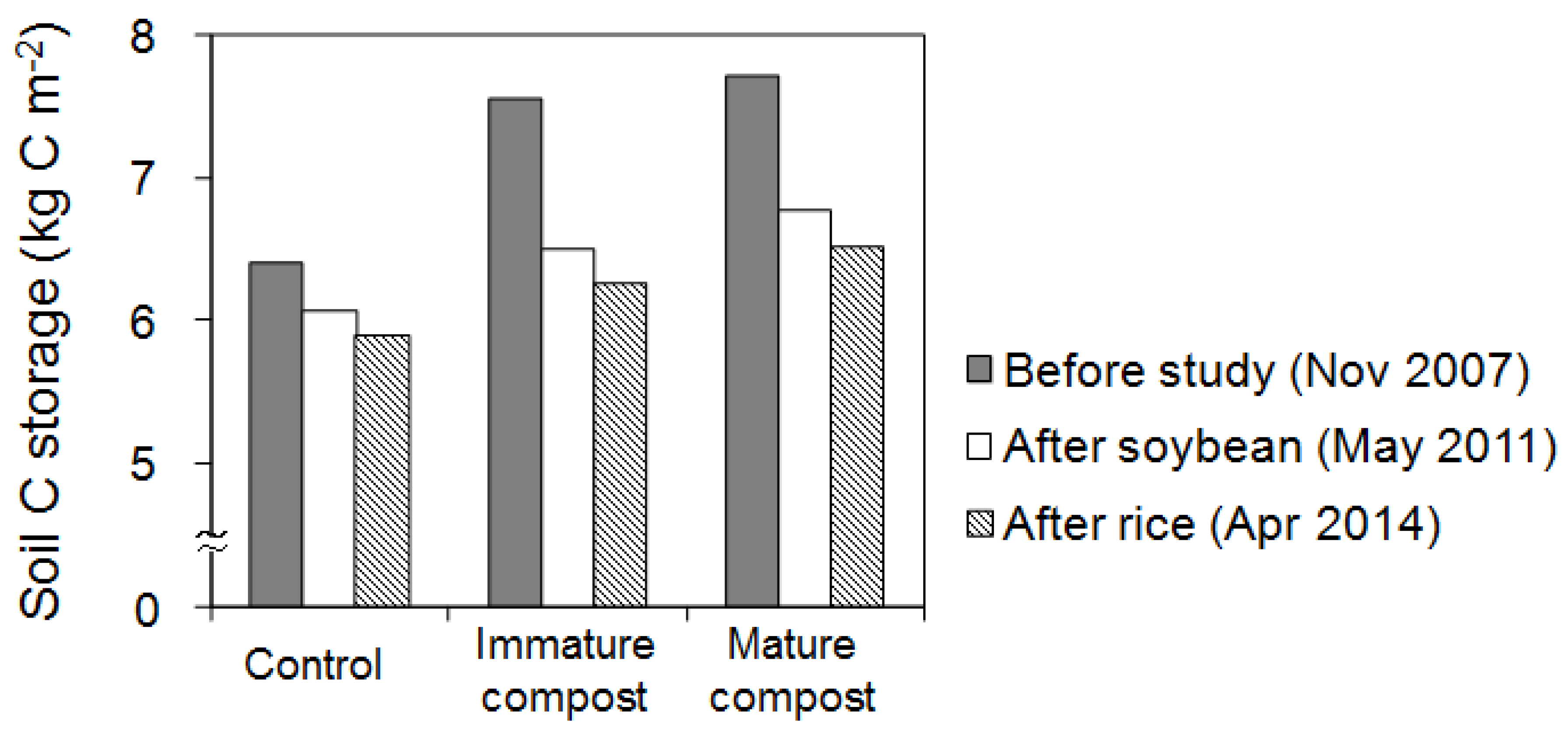
3.3. Changes in Soil Carbon Storage
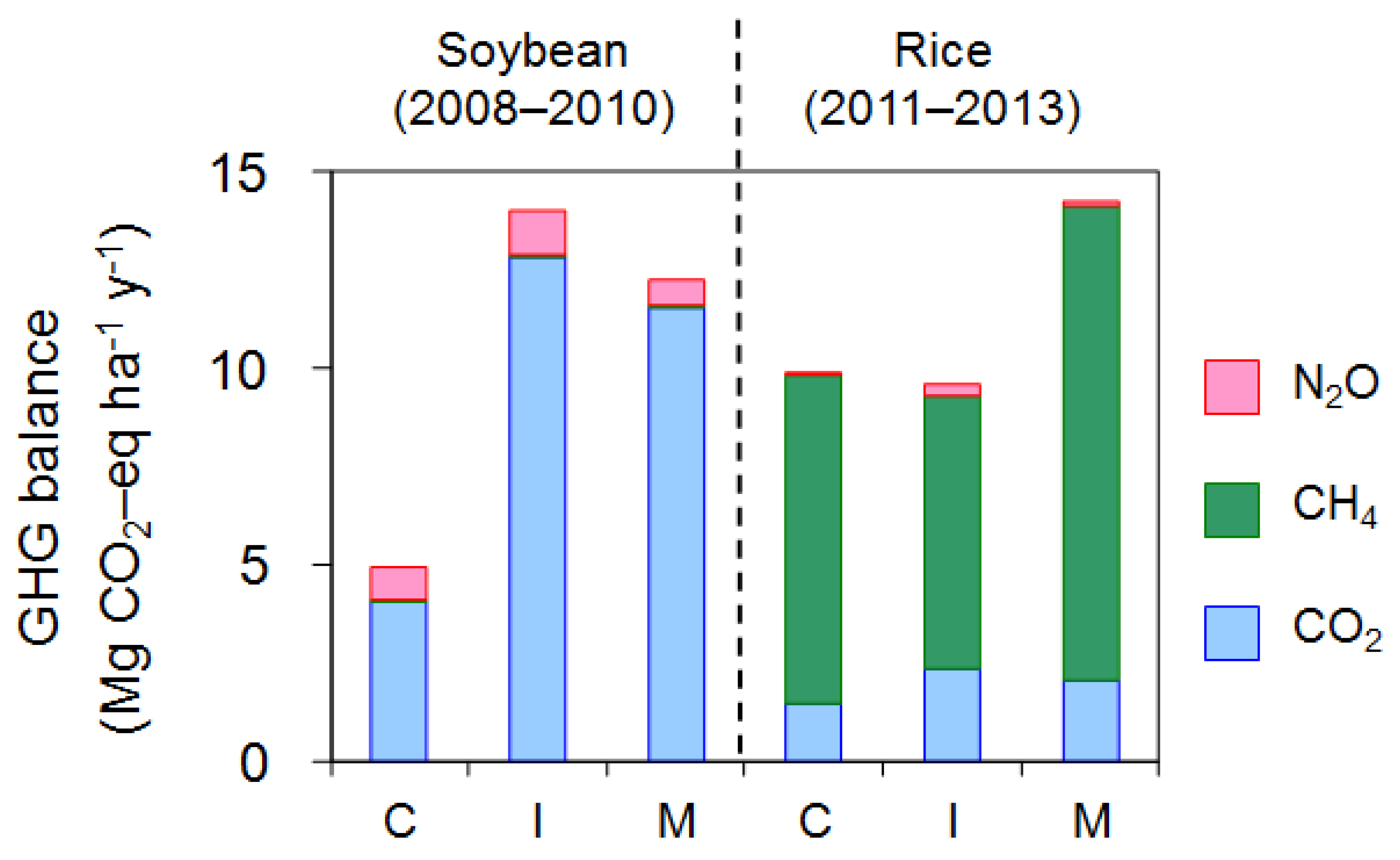
3.4. Net GHG Balance
4. Discussion
4.1. N2O Emissions from the Upland Soybean Field
4.2. CH4 and N2O Emissions from the Rice Paddy Field
4.3. CO2 Emission and Net GHG Balance
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Takahashi, T.; Sumida, H.; Nira, R. A new framework for study of irrigated paddy rice and upland crops rotation farming and its relation to soil and plant nutrition science. 1. Advances and perspectives in irrigated paddy rice and upland crops rotation farming. Jpn. J. Soil Sci. Plant Nutr. 2013, 84, 202–207. (In Japanese) [Google Scholar]
- Intergovernmental Panel on Climate Change (IPCC). The physical science basis: Anthropogenic and natural radiative forcing. In Climate Change 2013; Myhre, G., Shindell, D., Eds.; Cambridge University Press: Cambridge, UK, 2013. [Google Scholar]
- Paustian, K.; Andrén, O.; Janzen, H.; Lal, R.; Smith, P.; Tian, G.; Tiessen, H.; van Noordwijk, M.; Woomer, P. Agricultural soil as a C sink to offset CO2 emissions. Soil Use Manag. 1997, 13, 230–244. [Google Scholar] [CrossRef]
- Bouwman, A.F. Exchange of greenhouse gases between terrestrial ecosystems and the atmosphere. In Soils and the Greenhouse Effect; Bouwman, A.F., Ed.; John Wiley: New York, NY, USA, 1990; pp. 61–127. [Google Scholar]
- Mitsuchi, M. Characters of humus formed under rice cultivation. Soil Sci. Plant Nutr. 1974, 20, 249–259. [Google Scholar] [CrossRef]
- Moroyu, H. Changes of physical and chemical properties of paddy soils under the cultivation of upland crops. Jpn. J. Soil Sci. Plant Nutr. 1983, 54, 434–441. (In Japanese) [Google Scholar]
- Akiyama, H.; Yan, X.; Yagi, K. Estimations of emission factors for fertilizer-induced direct N2O emissions from agricultural soils in Japan: Summary of available data. Soil Sci. Plant Nutr. 2006, 52, 774–787. [Google Scholar] [CrossRef]
- Kumagai, K.; Konno, Y. Methane emission from rice paddy fields after upland farming. Jpn. J. Soil Sci. Plant Nutr. 1998, 69, 333–339, (In Japanese with English Summary). [Google Scholar]
- Nishimura, S.; Akiyama, H.; Sudo, S.; Fumoto, T.; Cheng, W.; Yagi, K. Combined emission of CH4 and N2O from a paddy field was reduced by preceding upland crop cultivation. Soil Sci. Plant Nutr. 2011, 57, 167–178. [Google Scholar] [CrossRef]
- Shiono, H.; Saito, H.; Nakagawa, F.; Nishimura, S.; Kumagai, K. Effects of crop rotation and rice straw incorporation in spring on methane and nitrous oxide emissions from an upland paddy field in a cold region of Japan. Jpn. J. Soil Sci. Plant Nutr. 2014, 85, 420–430, (In Japanese with English Summary). [Google Scholar]
- Sumida, H.; Kato, N.; Nishida, M. Depletion of soil fertility and crop productivity in succession of paddy rice-soybean rotation. Bull. Natl. Agric. Res. Cent. Tohoku Reg. 2005, 103, 39–52, (In Japanese with English Summary). [Google Scholar]
- Nishida, M.; Sekiya, H.; Yoshida, K. Status of paddy soils as affected by paddy rice and upland soybean rotation in northeast Japan, with special reference to nitrogen fertility. Soil Sci. Plant Nutr. 2013, 59, 208–217. [Google Scholar] [CrossRef]
- Shirato, Y.; Yagasaki, Y.; Nishida, M. Using different versions of the Rothamsted Carbon model to simulate soil carbon in long-term experimental plots subjected to paddy–upland rotation in Japan. Soil Sci. Plant Nutr. 2011, 57, 597–606. [Google Scholar] [CrossRef]
- Kumagai, K.; Shiono, H.; Morioka, M.; Nagasawa, K.; Nakagawa, F. Effect of application of livestock dung compost instead of spring incorporation of rice straw on methane emission from paddy fields in Yamagata. Bull. Agric. Res. Yamagata Prefect. 2010, 2, 1–18. [Google Scholar]
- Toma, Y.; Hatano, R. Effect of crop residue C:N ratio on N2O emissions from Gray Lowland soil in Mikasa, Hokkaido, Japan. Soil Sci. Plant Nutr. 2007, 53, 198–205. [Google Scholar] [CrossRef]
- Akiyama, H.; Tsuruta, H. Nitrous oxide, nitric oxide, and nitrogen dioxide fluxes from soils after manure and urea application. J. Environ. Qual. 2003, 32, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Takakai, F.; Takeda, M.; Kon, K.; Inoue, K.; Nakagawa, S.; Sasaki, K.; Chida, A.; Sekiguchi, K.; Takahashi, T.; Sato, T.; et al. Effects of preceding compost application on the nitrogen budget in an upland soybean field converted from a rice paddy field on gray lowland soil in Akita, Japan. Soil Sci. Plant Nutr. 2010, 56, 760–772. [Google Scholar] [CrossRef]
- Takakai, F.; Kikuchi, T.; Sato, T.; Takeda, M.; Sato, K.; Nakagawa, S.; Kon, K.; Sato, T.; Kaneta, Y. Changes in the nitrogen budget and soil nitrogen in a field with paddy–upland rotation with different histories of manure application. Agriculture 2017, 7, 39. [Google Scholar] [CrossRef]
- Department of Agriculture, Forestry and Fisheries, Akita Prefecture. Guidelines for Soybean Cultivation; Department of Agriculture, Forestry and Fisheries, Akita Prefecture: Akita, Japan, 2015; pp. 26–29. (In Japanese) [Google Scholar]
- Department of Agriculture, Forestry and Fisheries, Akita Prefecture. Guidelines for Rice Cultivation; Department of Agriculture, Forestry and Fisheries, Akita Prefecture: Akita, Japan, 2014; pp. 43–51. (In Japanese) [Google Scholar]
- Takakai, F.; Ichikawa, J.; Ogawa, M.; Ogaya, S.; Yasuda, K.; Kobayashi, Y.; Sato, T.; Kaneta, Y.; Nagahama, K. Suppression of CH4 emission by rice straw removal and application of bio-ethanol production residue in a paddy field in Akita, Japan. Agriculture 2017, 7, 21. [Google Scholar] [CrossRef]
- Koga, N.; Sawamoto, T.; Tsuruta, H. Life cycle inventory-based analysis of greenhouse gas emissions from arable land farming systems in Hokkaido, northern Japan. Soil Sci. Plant Nutr. 2006, 52, 564–574. [Google Scholar] [CrossRef]
- Uchida, Y.; Akiyama, H. Mitigation of postharvest nitrous oxide emissions from soybean ecosystems: A review. Soil Sci. Plant Nutr. 2013, 59, 477–487. [Google Scholar] [CrossRef]
- Minamikawa, K.; Nishimura, S.; Nakajima, Y.; Osaka, K.; Sawamoto, T.; Yagi, K. Upward diffusion of nitrous oxide produced by denitrification near shallow groundwater table in the summer: A lysimeter experiment. Soil Sci. Plant Nutr. 2011, 57, 719–732. [Google Scholar] [CrossRef]
- Davidson, E.A.; Keller, M.; Erickson, H.E.; Verchot, L.V.; Veldkamp, E. Testing a conceptual model of soil emissions of nitrous and nitric oxides. Bioscience 2000, 50, 667–680. [Google Scholar] [CrossRef]
- Toma, Y.; Kimura, S.D.; Yamada, H.; Hirose, Y.; Fujiwara, K.; Kusa, K.; Hatano, R. Effects of environmental factors on temporal variation in annual carbon dioxide and nitrous oxide emissions from an unfertilized bare field on Gray Lowland soil in Mikasa, Hokkaido, Japan. Soil Sci. Plant Nutr. 2010, 56, 663–675. [Google Scholar] [CrossRef]
- Eusufzai, M.K.; Tokida, T.; Okada, M.; Sugiyama, S.; Liu, G.C.; Nakajima, M.; Sameshima, R. Methane emission from rice fields as affected by land use change. Agric. Ecosyst. Environ. 2010, 139, 742–748. [Google Scholar] [CrossRef]
- Cheng, W.; Yagi, K.; Akiyama, H.; Nishimura, S.; Sudo, S.; Fumoto, T.; Hasegawa, T.; Hartley, A.E.; Megoniga, J.P. An empirical model of soil chemical properties that regulate methane production in Japanese rice paddy soils. J. Environ. Qual. 2007, 36, 1920–1925. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, H.; Yagi, K.; Yan, X.Y. Direct N2O emissions from rice paddy fields: Summary of available data. Glob. Biogeochem. Cycles 2005, 19. [Google Scholar] [CrossRef]
- Mu, Z.J.; Kimura, S.D.; Hatano, R. Estimation of global warming potential from upland cropping systems in central Hokkaido, Japan. Soil Sci. Plant Nutr. 2006, 52, 371–377. [Google Scholar] [CrossRef]
- Nishimura, S.; Yonemura, S.; Sawamoto, T.; Shirato, Y.; Akiyama, H.; Sudo, S.; Yagi, K. Effect of land use change from paddy rice cultivation to upland crop cultivation on soil carbon budget of a cropland in Japan. Agric. Ecosyst. Environ. 2008, 125, 9–20. [Google Scholar] [CrossRef]

| Year | Plot | CH4 | N2O | GWP (Mg CO2-eq ha−1 year−1) † | Measurement Period | ||
|---|---|---|---|---|---|---|---|
| (kg C ha−1 year−1) | (kg N ha−1 year−1) | CH4 | N2O | Total | |||
| 2008 | C | 5.51 | 1.73 | 0.25 | 0.81 | 1.06 | 10 Jun 2008 to 8 Jun 2009 |
| I | 0.89 | 2.70 | 0.04 | 1.26 | 1.30 | ||
| M | 3.14 | 0.88 | 0.14 | 0.41 | 0.56 | ||
| Average ‡ | 3.18 ± 1.33 a | 1.77 ± 0.52 a | 0.14 ± 0.06 | 0.83 ± 0.25 | 0.97 ± 0.22 a | ||
| 2009 | C | 1.96 | 1.85 | 0.09 | 0.87 | 0.96 | 8 June 2009 to 11 Jun 2010 |
| I | 1.35 | 2.73 | 0.06 | 1.28 | 1.34 | ||
| M | 2.49 | 1.90 | 0.11 | 0.89 | 1.00 | ||
| Average ‡ | 1.93 ± 0.33 a | 2.16 ± 0.28 a | 0.09 ± 0.01 | 1.01 ± 0.13 | 1.10 ± 0.12 a | ||
| 2010 | C | 0.15 | 1.44 | 0.01 | 0.67 | 0.68 | 11 Jun 2010 to 22 May 2011 |
| I | 1.94 | 1.75 | 0.09 | 0.82 | 0.91 | ||
| M | 1.73 | 1.29 | 0.08 | 0.61 | 0.68 | ||
| Average ‡ | 1.28 ± 0.57 a | 1.49 ± 0.13 a | 0.06 ± 0.03 | 0.70 ± 0.06 | 0.75 ± 0.07 a | ||
| Three years average ‡ | C | 2.54 ± 1.57 A | 1.68 ± 0.12 A,B | 0.12 ± 0.07 | 0.78 ± 0.06 | 0.90 ± 0.11 A,B | - |
| I | 1.40 ± 0.30 A | 2.39 ± 0.32 B | 0.06 ± 0.01 | 1.12 ± 0.15 | 1.18 ± 0.14 B | ||
| M | 2.45 ± 0.41 A | 1.36 ± 0.30 A | 0.11 ± 0.02 | 0.64 ± 0.14 | 0.75 ± 0.13 A | ||
| Year | Plot | CH4 | N2O | GWP (Mg CO2-eq ha−1 year−1) † | Measurement Period | ||
|---|---|---|---|---|---|---|---|
| (kg C ha−1 year−1) | (kg N ha−1 year−1) | CH4 | N2O | Total | |||
| 2011 | C | 48.7 | 0.11 | 2.21 | 0.05 | 2.26 | 2 Jun 2011 to 17 May 2012 |
| I | 71.5 | 0.37 | 3.24 | 0.17 | 3.41 | ||
| M | 83.5 | 0.18 | 3.78 | 0.08 | 3.87 | ||
| Average ‡ | 67.9 ± 10.2 a | 0.22 ± 0.08 a | 3.08 ± 0.46 | 0.10 ± 0.04 | 3.18 ± 0.48 a | ||
| 2012 | C | 106.9 | 0.28 | 4.84 | 0.13 | 4.97 | 27 May 2012 to 8 May 2013 |
| I | 113.7 | 0.50 | 5.15 | 0.23 | 5.39 | ||
| M | 133.9 | 0.25 | 6.07 | 0.12 | 6.19 | ||
| Average ‡ | 118.1 ± 8.1 a | 0.34 ± 0.08 a | 5.36 ± 0.37 | 0.16 ± 0.04 | 5.52 ± 0.36 a | ||
| 2013 | C | 364.0 | 0.03 | 16.50 | 0.01 | 16.52 | 30 May 2013 to 12 May 2014 |
| I § | 274.1 | 1.15 | 12.43 | 0.54 | 12.96 | ||
| M | 580.0 | 0.59 | 26.29 | 0.27 | 26.57 | ||
| Average ‡ | 406.1 ± 90.8 b | 0.59 ± 0.32 a | 18.41 ± 4.12 | 0.28 ± 0.15 | 18.68 ± 4.07 b | ||
| Three years average ‡ | C | 173.2 ± 96.9 A | 0.14 ± 0.07 A | 7.85 ± 4.39 | 0.07 ± 0.03 | 7.92 ± 4.37 A | - |
| I | 153.1 ± 61.7 A | 0.67 ± 0.24 A | 6.94 ± 2.80 | 0.32 ± 0.11 | 7.25 ± 2.91 A | ||
| M | 265.8 ± 157.8 A | 0.34 ± 0.13 A | 12.05 ± 7.15 | 0.16 ± 0.06 | 12.21 ± 7.21 A | ||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takakai, F.; Nakagawa, S.; Sato, K.; Kon, K.; Sato, T.; Kaneta, Y. Net Greenhouse Gas Budget and Soil Carbon Storage in a Field with Paddy–Upland Rotation with Different History of Manure Application. Agriculture 2017, 7, 49. https://doi.org/10.3390/agriculture7060049
Takakai F, Nakagawa S, Sato K, Kon K, Sato T, Kaneta Y. Net Greenhouse Gas Budget and Soil Carbon Storage in a Field with Paddy–Upland Rotation with Different History of Manure Application. Agriculture. 2017; 7(6):49. https://doi.org/10.3390/agriculture7060049
Chicago/Turabian StyleTakakai, Fumiaki, Shinpei Nakagawa, Kensuke Sato, Kazuhiro Kon, Takashi Sato, and Yoshihiro Kaneta. 2017. "Net Greenhouse Gas Budget and Soil Carbon Storage in a Field with Paddy–Upland Rotation with Different History of Manure Application" Agriculture 7, no. 6: 49. https://doi.org/10.3390/agriculture7060049
APA StyleTakakai, F., Nakagawa, S., Sato, K., Kon, K., Sato, T., & Kaneta, Y. (2017). Net Greenhouse Gas Budget and Soil Carbon Storage in a Field with Paddy–Upland Rotation with Different History of Manure Application. Agriculture, 7(6), 49. https://doi.org/10.3390/agriculture7060049






