Changes in Carbon Cycling during Development of Successional Agroforestry
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Net Biome Productivity
2.3. Aboveground Biomass Increase
2.4. Exported Fruit and Fruit Residue
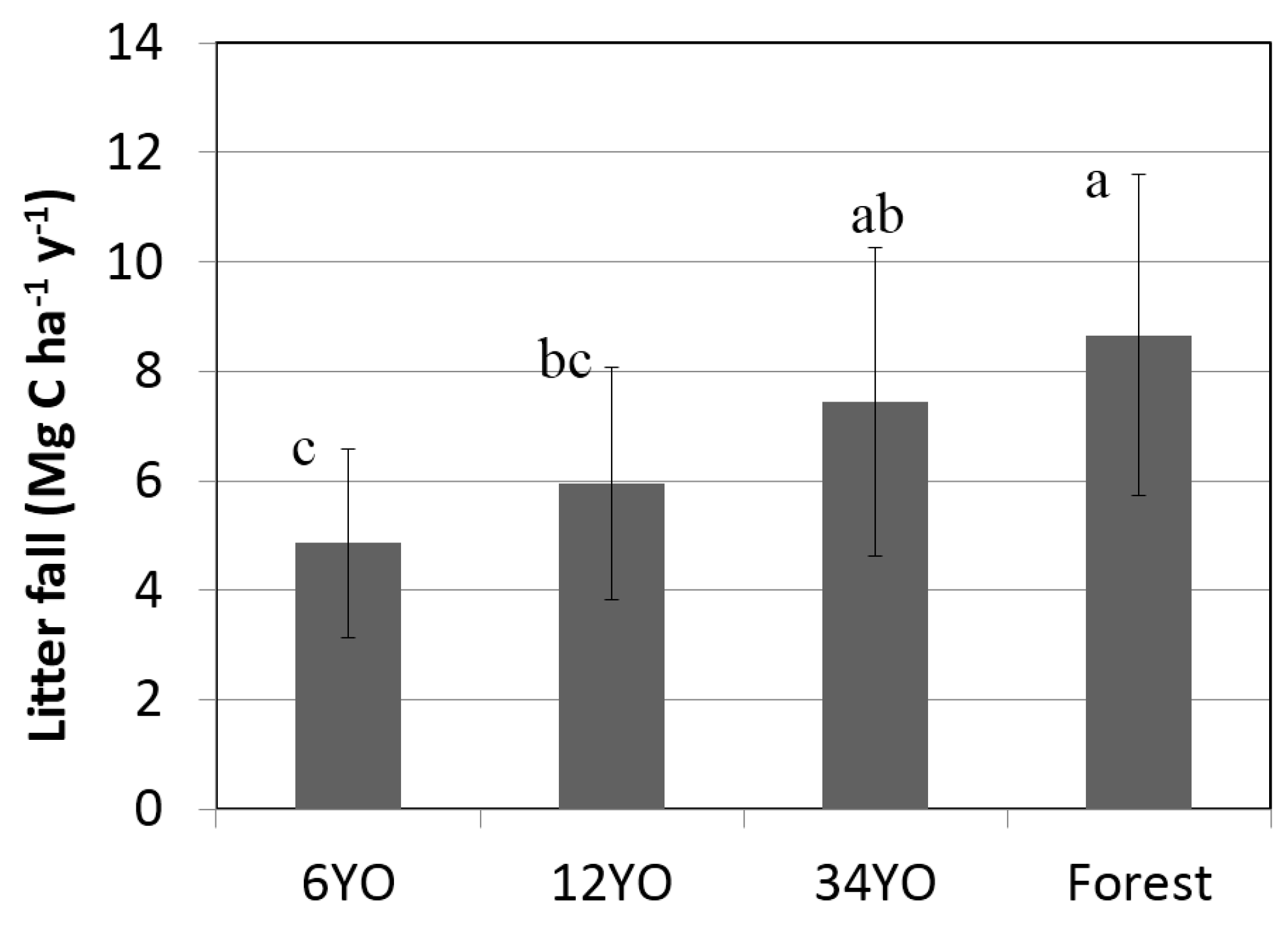
2.5. Litterfall
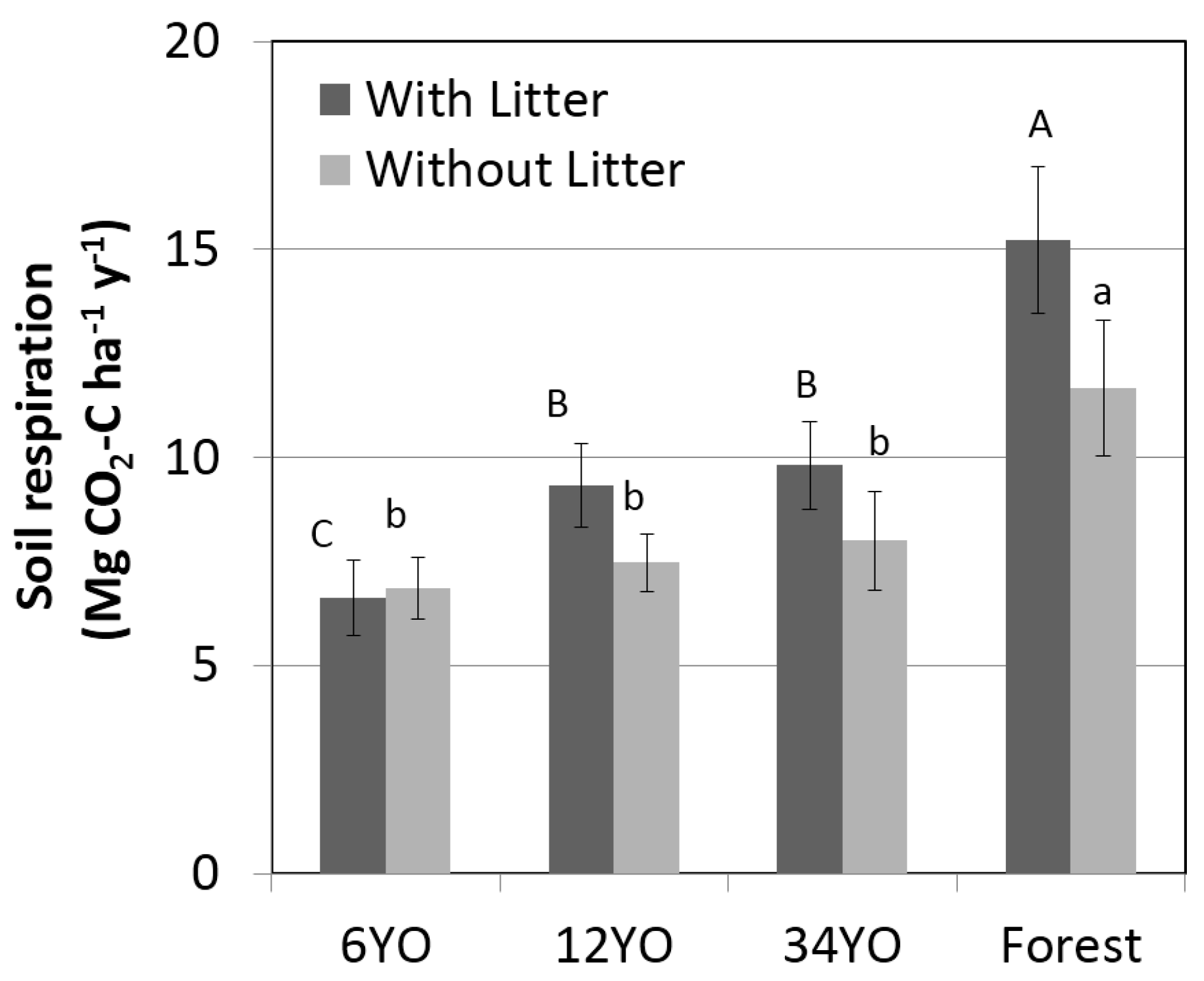
2.6. Soil Respiration
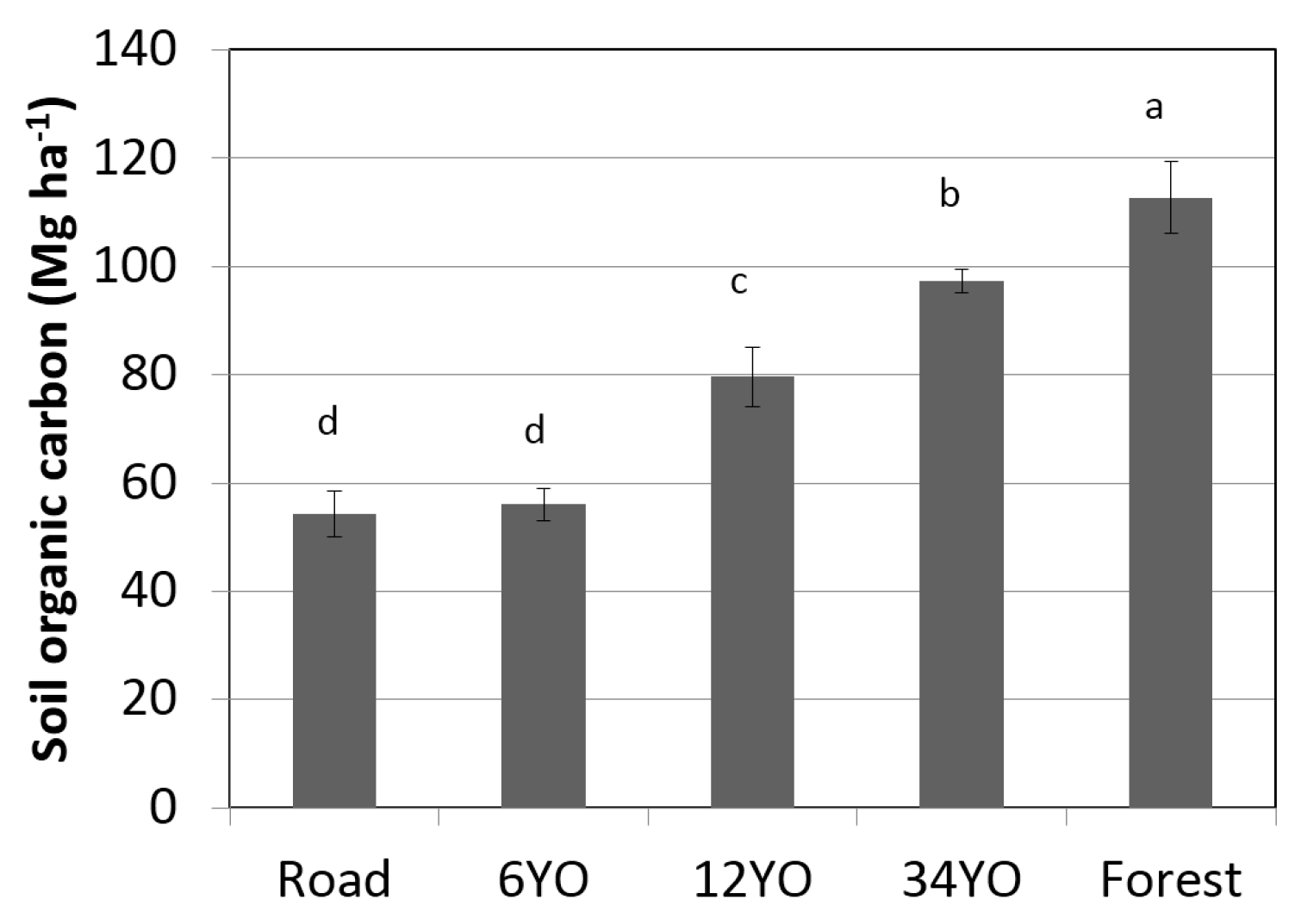
2.7. Soil Organic Carbon
2.8. Statistics
3. Results
4. Discussion
4.1. Changes in C Cycling during SAFS Development
4.2. Changes in Soil Organic Carbon during SAFS Development
4.3. Changes in C Sequestration during SAFS Development
5. Conclusions
Author Contributions
Conflicts of Interest
Abbreviations
| 6YO | 6-year-old system |
| 12YO | 12-year-old system |
| 34YO | 34-year-old system |
| AGB | above ground biomass (kg) |
| AGBI (2014–2013) | aboveground biomass increase from 2013 to 2014 |
| Ave | average |
| C | Carbon |
| CO2 | carbon dioxide |
| Db | diameter of the first biggest branch (cm) |
| DBH | diameter at breast height (cm) |
| Ds | stem diameter at 50 cm height (cm) |
| EF | exported fruit |
| FR | fruit residue |
| H | tree height (m) |
| LF | litterfall |
| NBP | net biome productivity |
| NPP | net primary productivity |
| SAFS | Successional agroforestry systems |
| SD | standard deviation |
| SOC | Soil organic carbon |
| SR | soil respiration |
References
- Glaser, B.; Haumaier, L.; Guggenberger, G.; Zech, W. The “Terra Preta” phenomenon: A model for sustainable agriculture in the humid tropics. Naturwissenschaften 2014, 88, 37–41. [Google Scholar] [CrossRef]
- Darkoh, M.B.K. The nature, causes and consequences of desertification in the drylands of Africa. Land Degrad. Dev. 1998, 9, 1–20. [Google Scholar] [CrossRef]
- Yamada, M.; Gholz, H.L. An evaluation of agroforestry systems as a rural development option for the Brazilian Amazon. Agrofor. Syst. 2002, 55, 81–87. [Google Scholar] [CrossRef]
- Vieira, D.L.; Holl, K.D.; Peneireiro, F.M. Agro-Successional Restoration as a Strategy to Facilitate Tropical Forest Recovery. Restor. Ecol. 2009, 17, 451–459. [Google Scholar] [CrossRef]
- Atangana, A.; Khasa, D.; Chang, S.; Degrande, A. Carbon Sequestration in Agroforestry Systems. In Tropical Agroforestry; Springer: Dordrecht, The Netherlands, 2014; pp. 217–225. [Google Scholar]
- Palm, C.; Tomich, T.; van Noordwijk, M.; Vosti, S.; Gockowski, J.; Alegre, J.; Verchot, L. Mitigating GHG emissions in the humid tropics: Case studies from the Alternatives to Slash-and-Burn Program (ASB). Environ. Dev. Sustain. 2004, 6, 145–162. [Google Scholar] [CrossRef]
- Kirby, K.R.; Potvin, C. Variation in carbon storage among tree species: Implications for the management of a small-scale carbon sink project. For. Ecol. Manag. 2007, 246, 208–221. [Google Scholar] [CrossRef]
- Reeves, D.W. The role of soil organic matter in maintaining soil quality in continuous cropping systems. Soil Tillage Res. 1997, 43, 131–167. [Google Scholar] [CrossRef]
- Brock, C.; Fließbach, A.; Oberholzer, H.-R.; Schulz, F.; Wiesinger, K.; Reinicke, F.; Koch, W.; Pallutt, B.; Dittman, B.; Zimmer, J.; et al. Relation between soil organic matter and yield levels of nonlegume crops in organic and conventional farming systems. J. Plant Nutr. Soil Sci. 2011, 174, 568–575. [Google Scholar] [CrossRef]
- Campbell, C.A. Soil organic carbon, nitrogen and fertility. Dev. Soil Sci. 1978, 8, 173–271. [Google Scholar]
- Karlen, D.L.; Mausbach, M.J.; Doran, J.W.; Cline, R.G.; Harris, R.F.; Schuman, G.E. Soil quality: A concept, definition, and framework for evaluation (a guest editorial). Soil Sci. Soc. Am. J. 1997, 61, 4–10. [Google Scholar] [CrossRef]
- Mills, A.J.; Fey, M.V. Declining soil quality in South Africa: Effects of land use on soil organic matter and surface crusting. S. Afr. J. Plant Soil 2004, 21, 388–398. [Google Scholar] [CrossRef]
- Brown, S.; Lugo, A.E. Effects of forest clearing and succession on the carbon and nitrogen content of soils in Puerto Rico and US Virgin Islands. Plant Soil 1990, 124, 53–64. [Google Scholar] [CrossRef]
- Mosquera-Losada, M.R.; Freese, D.; Rigueiro-Rodríguez, A. Carbon Sequestration in European Agroforestry Systems. In Carbon Sequestration Potential of Agroforestry Systems; Kumar, B.M., Nair, P.K.R., Eds.; Springer: Dordrecht, The Netherlands, 2011; Volume 8, pp. 43–59. [Google Scholar]
- Gupta, N.; Kukal, S.S.; Bawa, S.S.; Dhaliwal, G.S. Soil organic carbon and aggregation under poplar based agroforestry system in relation to tree age and soil type. Agrofor. Syst. 2009, 76, 27–35. [Google Scholar] [CrossRef]
- Jacobi, J.; Andres, C.; Schneider, M.; Pillco, M.; Calizaya, P.; Rist, S. Carbon stocks, tree diversity, and the role of organic certification in different cocoa production systems in Alto Beni, Bolivia. Agrofor. Syst. 2014, 88, 1117–1132. [Google Scholar] [CrossRef]
- Schulz, B.; Becker, B.; Götsch, E. Indigenous knowledge in a “modern” sustainable agroforestry system—A case study from eastern Brazil. Agrofor. Syst. 1994, 25, 59–69. [Google Scholar] [CrossRef]
- Peneireiro, F.M. Sistemas Agroflorestais Dirigidos Pela Sucessão Natural: Um Estudo de Caso; Universidade de São Paulo: Sao Paulo, Brazil, 1999. [Google Scholar]
- Quelca, A.S.; Gama, M.B.; Pastrana, A.; Ochoa, R. Percepciones y Valoración del Sistema Sucesional Multiestrato de los Productores Cacaoteros del Alto Beni, Bolivia. Agrofor. en las Am. 2005, 43–44, 77–80. [Google Scholar]
- Nair, P.K.R.; Mohan Kumar, B.; Nair, V.D. Agroforestry as a strategy for carbon sequestration. J. Plant Nutr. Soil Sci. 2009, 172, 10–23. [Google Scholar] [CrossRef]
- Baena, A.R.C.; Falesi, I.C. Avaliação do Potencial Químico e Físico dos Solos sob Diversos Sistemas de uso da Terra na Colônia Agrícola de Tomé-Açu, Estado do Pará; Embrapa Amazônia Oriental: Belém, Brazil, 1999; p. 23. [Google Scholar]
- Schulze, E.D.; Heimann, M. Carbon and water exchange of terrestrial ecosystems. In Asian Change in the Context of Global Climate Change: Impact of Natural and Anthropogenic Changes in Asia on Global Biogeochemical Cycles; Galloway, J., Melillo, J., Eds.; Cambridge University Press: Cambridge, UK, 1998; pp. 145–161. [Google Scholar]
- Subler, S. Allometric equations for estimating the above ground biosmass of cacao stands in the eastern Amazon Basin. Agrotrópica 1994, 6, 65–72. [Google Scholar]
- Brown, S.; Gillespie, A.J.R.; Lugo, A.E. Biomass estimation methods for tropical forests with applications to forest inventory data. For. Sci. 1989, 35, 881–902. [Google Scholar]
- Brown, S. Estimating biomass and biomass change of tropical forest: A primer; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy; pp. 1–55.
- Hanson, P.J.; Edwards, N.T.; Garten, C.T.; Andrews, J.A. Separating root and soil microbial contributions to soil respiration: A review of methods and observations. Biogeochemistry 2000, 48, 115–146. [Google Scholar] [CrossRef]
- Wendt, J.W.; Hauser, S. An equivalent soil mass procedure for monitoring soil organic carbon in multiple soil layers. Eur. J. Soil Sci. 2013, 64, 58–65. [Google Scholar] [CrossRef]
- Wendt, J.W. ESM Sample Spreadsheets. Xlsm. Available online: https://drive.google.com/file/d/0BzxNFfzLbFxjSG9RWlpwQ0FXc0k/view (accessed on 30 August 2016).
- Lorenz, K.; Lal, R. Carbon Sequestration in Forest Ecosystems; Springer: Dordrecht, The Netherlands, 2010. [Google Scholar]
- Lemieux, G. The Hidden World that feeds us: The living soil. Coord. Group Ramial Wood Laval Univ. Can. Publ. 1996, 46, 1–38. [Google Scholar]
- Grayston, S.J.; Vaughan, D.; Jones, D. Rhizosphere carbon flow in trees, in comparison with annual plants: The importance of root exudation and its impact on microbial activity and nutrient availability. Appl. Soil Ecol. 1997, 5, 29–56. [Google Scholar] [CrossRef]
- Silver, W.L.; Ostertag, R.; Lugo, A.E. The Potential for Carbon Sequestration through Reforestation of Abandoned Tropical Agricultural and Pasture Lands. Restor. Ecol. 2000, 8, 394–407. [Google Scholar] [CrossRef]
- Richter, D.D.; Markewitz, D.; Trumbore, S.E.; Wells, C.G. Rapid accumulation and turnover of soil carbon in a re-establishing forest. Nature 1999, 400, 56–58. [Google Scholar] [CrossRef]
- De Gryze, S.; Six, J.; Paustian, K.; Morris, S.J.; Paul, E.A.; Merckx, R. Soil organic carbon pool changes following land-use conversions. Glob. Chang. Biol. 2004, 10, 1120–1132. [Google Scholar] [CrossRef]
- Braimoh, A.K.; Vlek, P. Land Use and Soil Resources; Springer: Berlin/Heidelberg, Germany, 2008. [Google Scholar]
- IPCC. IPCC Guidelines for National Greenhouse Gas Inventories (3 Volumes); IPCC: Geneva, Switzerland, 1997. [Google Scholar]
- Dixon, R.K. Agroforestry systems: Sources or sinks of greenhouse gases? Agrofor. Syst. 1995, 31, 99–116. [Google Scholar] [CrossRef]
- Jobbagy, E.G.; Jackson, R.B. The Vertical Distribution of Soil Organic Carbon and Its Relation to Climate and Vegetation. Ecol. Appl. 2000, 10, 423. [Google Scholar] [CrossRef]
- Lutzow, M.V.; Kogel-Knabner, I.; Ekschmitt, K.; Matzner, E.; Guggenberger, G.; Marschner, B.; Flessa, H. Stabilization of organic matter in temperate soils: Mechanisms and their relevance under different soil conditions—A review. Eur. J. Soil Sci. 2006, 57, 426–445. [Google Scholar] [CrossRef]
- Perrott, K.W.; Ghani, A.; O’connor, M.B.; Waller, J.E.; Hawke, M.F. Tree stocking effects on soil chemical and microbial properties at the Tikitere Agroforestry Research Area. N. Z. J. For. Sci. 1999, 29, 116–130. [Google Scholar]
- Farley, K.A.; Kelly, E.F.; Hofstede, R.G.M. Soil Organic Carbon and Water Retention after Conversion of Grasslands to Pine Plantations in the Ecuadorian Andes. Ecosystems 2004, 7, 729–739. [Google Scholar] [CrossRef]
- Parfitt, R.L.; Percival, H.J.; Dahlgren, R.A.; Hill, L.F. Soil and solution chemistry under pasture and radiata pine in New Zealand. Plant Soil 1999, 191, 279–290. [Google Scholar] [CrossRef]
- Del Galdo, I.; Six, J.; Peressotti, A.; Francesca Cotrufo, M. Assessing the impact of land-use change on soil C sequestration in agricultural soils by means of organic matter fractionation and stable C isotopes. Glob. Chang. Biol. 2003, 9, 1204–1213. [Google Scholar] [CrossRef]
- Lemma, B.; Kleja, D.B.; Nilsson, I.; Olsson, M. Soil carbon sequestration under different exotic tree species in the southwestern highlands of Ethiopia. Geoderma 2006, 136, 886–898. [Google Scholar] [CrossRef]
- Grünzweig, J.M.; Gelfand, I.; Fried, Y.; Yakir, D. Biogeochemical factors contributing to enhanced carbon storage following afforestation of a semi-arid shrubland. Biogeosciences 2007, 4, 891–904. [Google Scholar] [CrossRef]
- Laganière, J.; Angers, D.A.; Paré, D. Carbon accumulation in agricultural soils after afforestation: A meta-analysis. Glob. Chang. Biol. 2010, 16, 439–453. [Google Scholar] [CrossRef]
- Edwards, N.T.; Harris, W.F. Carbon Cycling in a Mixed Deciduous Forest Floor. Ecology 1977, 58, 431–437. [Google Scholar] [CrossRef]
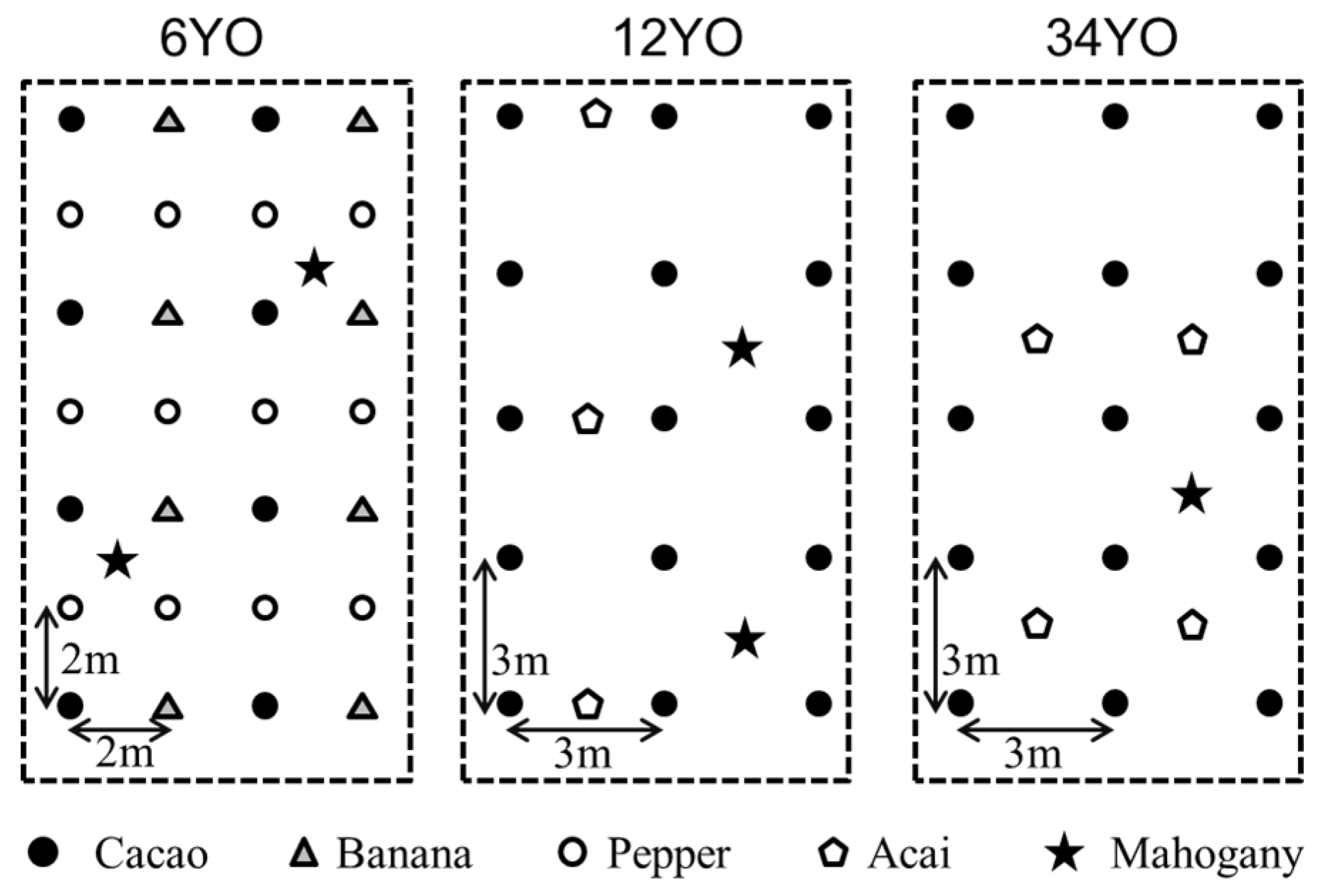
| Species | 6YO | 12YO | 34YO |
|---|---|---|---|
| Theobroma cacao (Cacao) | 625.0 | 1111.1 | 1111.1 |
| Swietenia macrophylla (Mahogany) | 208.3 | 277.8 | 123.5 * |
| Euterpe oleracea (Acai) | 277.8 | 277.8 | |
| Musa acuminata (Banana) | 625.0 | ||
| Piper nigrum (Pepper) | 1250.0 |
| Species | Formula | Source |
|---|---|---|
| Cacao | Log(Biomass) = −1.81 + 2.13log(Db + Ds) | [23] |
| Mahogany | AGB = exp(−3.1141 + 0.9719 × ln(DBH2 × H)) | [24] |
| Acai | AGB = 10 + 6.4 × H | [25] |
| 6YO | 12YO | 34YO | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ave | SD | Ave | SD | Ave | SD | ||||||
| Exported fruit and fruit residue | Cacao | 1.7 | ± | 0.4 | 3.7 | ± | 0.8 | 2.9 | ± | 0.7 | |
| Acai | 1.5 | ± | 0.6 | 1.6 | ± | 0.6 | |||||
| Pepper | 1.0 | ± | 0.2 | ||||||||
| Sum | 2.7 | ± | 0.5 | 5.2 | ± | 1.0 | 4.5 | ± | 0.9 | ||
| Exported fruit | Cacao | 1.3 | ± | 0.4 | 2.9 | ± | 0.6 | 2.3 | ± | 0.6 | |
| Acai | 1.1 | ± | 0.4 | 1.2 | ± | 0.4 | |||||
| Pepper | 1.0 | ± | 0.2 | ||||||||
| Sum | 2.3 | ± | 0.4 | 4.1 | ± | 0.8 | 3.5 | ± | 0.7 | ||
| 6YO | 12YO | 34YO | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Ave | SD | Ave | SD | Ave | SD | ||||
| Cacao | 0.9 | ± | 0.3 | 0.7 | ± | 0.3 | 0.9 | ± | 0.5 |
| Mahogany | 0.9 | ± | 0.2 | 0.8 | ± | 0.4 | 0.2 | ± | 0.1 |
| Acai | 0.8 | ± | 0.3 | 0.5 | ± | 0.2 | |||
| Banana | 0.5 | ± | 0.1 | ||||||
| Sum | 2.3 | ± | 0.4 | 2.3 | ± | 0.6 | 1.6 | ± | 0.5 |
| 6YO | 12YO | 34YO | Forest | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ave | SD | Ave | SD | Ave | SD | Ave | SD | |||||
| Above ground biomass increase (1) | 2.3 | ± | 0.4 | 2.3 | ± | 0.6 | 1.6 | ± | 0.5 | 5.0 | ~ | 10.0 |
| Exported fruit and fruit residue | 2.7 | ± | 0.5 | 5.2 | ± | 1.0 | 4.5 | ± | 0.9 | |||
| Litterfall | 4.9 | ± | 1.7 | 5.9 | ± | 2.1 | 7.4 | ± | 2.8 | 8.7 | ± | 2.9 |
| NPP | 9.8 | ± | 1.8 | 13.5 | ± | 2.4 | 13.5 | ± | 3.0 | 13.7 | ~ | 18.7 |
| Exported fruit | 2.3 | ± | 0.4 | 4.1 | ± | 0.8 | 3.5 | ± | 0.7 | |||
| Litter respiration | −0.2 | ± | 1.2 | 1.9 | ± | 1.2 | 1.8 | ± | 1.6 | 3.6 | ± | 2.4 |
| Soil heterotrophic respiration (2) | 1.4 | – | 5.5 | 1.5 | – | 6.0 | 1.6 | – | 6.4 | 2.3 | – | 9.3 |
| Total C output | 3.5 | – | 7.6 | 7.4 | – | 11.9 | 5.3 | – | 11.7 | 5.9 | – | 12.9 |
| NBP | 2.2 | – | 6.3 | 1.6 | – | 6.1 | 1.8 | – | 8.2 | 0.8 | – | 12.8 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Selecky, T.; Bellingrath-Kimura, S.D.; Kobata, Y.; Yamada, M.; Guerrini, I.A.; Umemura, H.M.; Dos Santos, D.A. Changes in Carbon Cycling during Development of Successional Agroforestry. Agriculture 2017, 7, 25. https://doi.org/10.3390/agriculture7030025
Selecky T, Bellingrath-Kimura SD, Kobata Y, Yamada M, Guerrini IA, Umemura HM, Dos Santos DA. Changes in Carbon Cycling during Development of Successional Agroforestry. Agriculture. 2017; 7(3):25. https://doi.org/10.3390/agriculture7030025
Chicago/Turabian StyleSelecky, Tomas, Sonoko D. Bellingrath-Kimura, Yuji Kobata, Masaaki Yamada, Iraê A. Guerrini, Helio M. Umemura, and Dinaldo A. Dos Santos. 2017. "Changes in Carbon Cycling during Development of Successional Agroforestry" Agriculture 7, no. 3: 25. https://doi.org/10.3390/agriculture7030025
APA StyleSelecky, T., Bellingrath-Kimura, S. D., Kobata, Y., Yamada, M., Guerrini, I. A., Umemura, H. M., & Dos Santos, D. A. (2017). Changes in Carbon Cycling during Development of Successional Agroforestry. Agriculture, 7(3), 25. https://doi.org/10.3390/agriculture7030025






