Using Ecophysiology to Improve Farm Efficiency: Application in Temperate Dairy Grazing Systems
Abstract
:1. Introduction
2. Critical Ecophysiological Principles and Their Translation to Grazing Management Practice
3. Managing the Pasture Regrowth Curve
3.1. The Ecophysiological Basis of Current Grazing Management Recommendations for New Zealand Dairy Farms
3.2. Opportunities for Improving Pasture Harvest Rates
3.2.1. Targets for Residual Pasture State
3.2.2. Targets for Timing of Defoliation
3.2.3. Pasture Quality
3.3. Implementing Grazing Management on Farm
4. Future Opportunities for Application of Ecophysiological Information
4.1. The Extended Ryegrass Phenome and Its Implications for Grazing Management Practices
4.2. Pasture Persistence
5. Conclusions
Conflicts of Interest
References
- Da Silva, S.C.; Sbrissia, A.F.; Pereira, L.E.T. Ecophysiology of C4 forage grasses—Understanding plant growth for optimising their use and management. Agriculture 2015, 5, 598–625. [Google Scholar] [CrossRef]
- Hodgson, J. Grazing Management: Science into Practice; Longman Scientific and Technical: Harlow, Essex, UK, 1990. [Google Scholar]
- Lemaire, G.; Hodgson, J.; Moraes, A.; Nabinger, C.; Carvalho, P.C.F. (Eds.) Grassland Ecophysiology and Grazing Ecology; CAB International: Wallingford, UK, 2000.
- Bryant, A.M. Optimum stocking and feed management practices. In Proceedings of the 42nd Ruakura Farmers Conference, Hamilton, New Zealand, June 1990; MAF: Hamilton New Zealand, 1990; pp. 55–59. [Google Scholar]
- IFCN. Dairy Report 2015; International Farm Comparison Network: Kiel, Germany, 2015. [Google Scholar]
- McCarthy, S.; Hirst, C.; Donaghy, D.; Gray, D.; Wood, B. Opportunities to improve grazing management. Proc. N. Z. Grassl. Assoc. 2014, 76, 75–80. [Google Scholar]
- DairyNZ. Grazing Management: The Root to Success; Technical Series, Issue 23; DairyNZ: Hamilton, New Zealand, 2014. [Google Scholar]
- Savage, J.; Lewis, C. Applying science as a tool for dairy farmers. Proc. N. Z. Grassl. Assoc. 2005, 67, 61–66. [Google Scholar]
- Dillon, P.; Roche, J.R.; Shalloo, L.; Horan, B. Optimising financial returns from grazing in temperate pastures. In Utilisation of Grazed Grass in Temperate Animal Systems; Wageningen Academic Publishers: Wageningen, The Netherlands, 2005; pp. 131–147. [Google Scholar]
- Gastal, F.; Lemaire, G. Defoliation, shoot plasticity, sward structure and herbage utilization in pasture: Review of the underlying ecophysiological processes. Agriculture 2015, 5, 1146–1171. [Google Scholar] [CrossRef]
- Irving, J. Carbon assimilation, biomass partitioning and productivity in grasses. Agriculture 2015, 5, 1116–1134. [Google Scholar] [CrossRef]
- Lambers, H.; Chapin, F.S.; Pons, T.L. Plant Physiological Ecology; Springer-Verlag: New York, NY, USA, 1998. [Google Scholar]
- Chapman, D.F.; Parsons, A.J.; Cosgrove, G.P.; Barker, D.J.; Marotti, D.M.; Venning, K.J.; Rutter, S.M.; Hill, J.; Thompson, A.N. Impacts of spatial patterns in pasture on animal grazing behaviour, intake, and performance. Crop Sci. 2007, 47, 399–415. [Google Scholar] [CrossRef]
- Parsons, A.J.; Chapman, D.F. The principles of pasture growth and utilisation. In Grass: Its Production and Utilisation; Hopkins, A., Ed.; Blackwell Science: Oxford, UK, 2000; pp. 31–89. [Google Scholar]
- Clark, D.A.; Kanneganti, V.R. Grazing management systems for dairy cattle. In Grass for Dairy Cattle; Cherney, J.H., Cherney, D.J.R., Eds.; CABI: Wallingford, UK, 1998; pp. 311–334. [Google Scholar]
- Agricultural Development and Advisory Service. Grassland Management Calendar. Dairy Cows. Continuous Grazing; HMSO: London, UK, 1987. [Google Scholar]
- Milligan, K. Comments on the grazing management session. Proc. N. Z. Grassl. Assoc. 1984, 45, 207–208. [Google Scholar]
- Fulkerson, W.J.; Donaghy, D.J. Plant soluble carbohydrate reserves and senescence—Key criteria for developing an effective grazing management system for ryegrass-based pastures: A review. Aust. J. Exp. Agric. 2001, 41, 261–275. [Google Scholar] [CrossRef]
- MacDonald, K.A.; Glassey, C.B.; Rawnsley, R.P. The emergence, development and effectiveness of decision rules for pasture-based dairy systems. In Proceedings of the 4th Australasian Dairy Science Symposium, Lincoln, New Zealand, 31 August–2 September 2010; pp. 233–238.
- Brougham, R.W. Effect of intensity of defoliation on regrowth of pasture. Aust. J. Agric. Res. 1956, 7, 377–387. [Google Scholar] [CrossRef]
- Brougham, R.W. Pasture growth rate studies in relation to defoliation. Proc. N. Z. Soc. Ann. Prod. 1957, 17, 46–55. [Google Scholar]
- Bircham, J.S.; Hodgson, J. The influence of sward conditions on rate of herbage growth and senescence in mixed swards under continuous stocking management. Grass Forage Sci. 1983, 38, 323–331. [Google Scholar] [CrossRef]
- Parsons, A.J.; Johnson, I.R.; Harvey, A. Use of a model to optimize the interaction between frequency and severity of intermittent defoliation and to provide a fundamental comparison of the continuous and intermittent defoliation of grass. Grass Forage Sci. 1988, 43, 49–59. [Google Scholar] [CrossRef]
- Grant, S.A.; Barthram, G.T.; Torvell, L.; King, J.; Smith, H.K. Sward management, lamina turnover and tiller population density in continuously stocked Lolium perenne-dominated swards. Grass Forage Sci. 1983, 38, 333–344. [Google Scholar] [CrossRef]
- Parsons, A.J.; Leafe, E.L.; Collett, B.; Stiles, W. The physiology of grass production under grazing. 1. Characteristics of leaf and canopy photosynthesis of continuously grazed swards. J. Appl. Ecol. 1983, 20, 117–126. [Google Scholar] [CrossRef]
- Davies, A. Tissue turnover in the sward. In Sward Measurement Handbook, 2nd ed.; Hodgson, J., Baker, R.D., Davies, A., Laidlaw, A.S., Leaver, J.D., Eds.; The British Grassland Society: Reading, UK, 1981; pp. 183–215. [Google Scholar]
- Rawnsley, R.P.; Snare, T.A.; Lee, G.; Lane, P.A.; Turner, L.R. Effects of ambient temperature and osmotic stress on leaf appearance rate. In Proceedings of the 4th Australasian Dairy Science Symposium, Lincoln, New Zealand, 31 August–2 September 2010; pp. 345–350.
- Shannon, P. Feeding pastures for profit—An innovative and practical approach to understanding and managing grazing based feeding systems. In Proceedings of the 4th Australasian Dairy Science Symposium, Lincoln, New Zealand, 31 August–2 September 2010; pp. 185–189.
- Richards, J.H. Physiology of plants recovering from defoliation. In Proceedings of the XVII International Grassland Congress, Palmerston North, New Zealand, 8–21 February 1993; New Zealand Grassland Association: Palmerston North, New Zealand, 1993; pp. 85–94. [Google Scholar]
- Lee, J.; Hedley, P.; Roche, J. Grazing Management Guidelines for Optimum Pasture Growth and Quality; Technical Series, Issue 5; DairyNZ: Hamilton, New Zealand, 2011. [Google Scholar]
- Holmes, C.W.; Brookes, I.M.; Garrick, D.J.; MacKenzie, D.D.S.; Parkinson, T.J.; Wilson, G.F. Milk Production from Pasture. Principles and Practices; Massey University: Palmerston North, New Zealand, 2002. [Google Scholar]
- Chapman, D.; McCarthy, S.; Kay, J. Hidden dollars in grazing management: Getting the most profit from your pastures. In Proceedings of the South Island Dairy Event, Invercargill, New Zealand, 23–25 June 2014; pp. 21–36.
- Piggot, G.J.; Baars, J.A.; Waller, J.E.; Farrell, C.A. Initial development of the “pasture growth rate” concept for estimating pasture growth on farms across Northland. In Proceedings of the 16th Agronomy Society of New Zealand Conference, Auckland, New Zealand, 1–5 December 1986; Agronomy Society of New Zealand: Palmerston North, New Zealand, 1986; pp. 65–69. [Google Scholar]
- Piggott, G.J. Herbage accumulation patterns of pastures at various sites in Northland. In Proceedings of the 18th Agronomy Society of New Zealand Conference, Havelock North, New Zealand, 1988; Agronomy Society of New Zealand: Palmerston North, New Zealand, 1988; pp. 81–84. [Google Scholar]
- Chapman, D.F.; Tharmaraj, J.; Agnusdei, M.; Hill, J. Regrowth dynamics and grazing decision rules: Further analysis for dairy production systems based on perennial ryegrass (Lolium perenne L.) pastures. Grass Forage Sci. 2012, 67, 77–95. [Google Scholar] [CrossRef]
- Grant, S.A.; Barthram, G.T.; Torvell, L. Components of regrowth in grazed and cut Lolium perenne swards. Grass Forage Sci. 1981, 36, 155–168. [Google Scholar] [CrossRef]
- Gautier, H.; Varlet-Grancher, C.; Hazard, L. Tillering responses to the light environment and to defoliation in populations of perennial ryegrass (Lolium perenne L.) selected for contrasting leaf length. Ann. Bot. 1999, 83, 423–429. [Google Scholar] [CrossRef]
- Chapman, D.F.; Bryant, J.R.; Olayemi, M.E.; Edwards, G.R.; Thorrold, B.S.; McMillan, W.H.; Kerr, G.A.; Judson, G.; Cookson, T.; Moorhead, A.; et al. An economically based evaluation index for perennial and short-term ryegrasses in New Zealand dairy farm systems. Grass Forage Sci. 2016, in press. [Google Scholar] [CrossRef]
- Lee, J.M.; Donaghy, D.J.; Roche, J.R. Effect of defoliation severity on regrowth and nutritive value of perennial ryegrass (Lolium perenne L.) dominant swards. Agron. J. 2008, 100, 308–314. [Google Scholar] [CrossRef]
- Garcia, S.C.; Holmes, C.W. Seasonality of calving in pasture-based dairy systems: Its effects on herbage production, utilisation and dry matter intake. Aust. J. Exp. Agric. 2005, 45, 1–9. [Google Scholar] [CrossRef]
- Agnusdei, M.G.; Di Marco, O.N.; Nenning, F.R.; Aello, M.S. Leaf blade nutritional quality of rhodes grass (Chloris gayana) as affected by leaf age and length. Crop Pasture Sci. 2011, 62, 1098–1105. [Google Scholar] [CrossRef]
- Groot, J.C.J.; Neuteboom, J.H. Composition and digestibility during ageing of Italian ryegrass leaves of consecutive insertion levels. J. Sci. Food Agric. 1997, 75, 227–236. [Google Scholar] [CrossRef]
- Groot, J.C.J.; Neuteboom, J.H.; Deinum, B. Composition and digestibility during ageing of consecutive leaves on the main stem of Italian ryegrass plants, growing undisturbed or regrowing after cutting. J. Sci. Food Agric. 1999, 79, 1691–1697. [Google Scholar] [CrossRef]
- Wilson, J.R. Variation of leaf characteristics with level of insertion on a grass tiller. I. Development rate, chemical composition and dry matter digestibility. Aust. J. Agric. Res. 1976, 27, 343–354. [Google Scholar] [CrossRef]
- Minson, D.J. Forage in Ruminant Nutrition; California Academic Press: Millbrae, CA, USA, 1990. [Google Scholar]
- Sinclair, K.; Fulkerson, W.J.; Morris, S.G. Influence of regrowth time on the fresh quality of prairie grass, perennial ryegrass and tall fescue under non-limiting soil nutrient and moisture conditions. Aust. J. Exp. Agric. 2006, 46, 45–51. [Google Scholar] [CrossRef]
- Turner, L.R.; Donaghy, D.J.; Lane, P.A.; Rawnsley, R.P. Effect of defoliation management, based on leaf stage, on perennial ryegrass (Lolium perenne L.), prairie grass (Bromus willdenowii Kunth.) and cocksfoot (Dactylis glomerata L.) under dryland conditions. 2. Nutritive value. Grass Forage Sci. 2006, 61, 175–181. [Google Scholar] [CrossRef]
- Pacheco, D.; Waghorn, G.C. Dietary nitrogen—Definitions, digestion, excretion and consequences of excess for grazing ruminants. Proc. N. Z. Grassl. Assoc. 2008, 70, 107–116. [Google Scholar]
- Kebreab, E.; France, J.; Beever, D.E.; Castillo, A.R. Nitrogen pollution by dairy cows and its mitigation by dietary manipulation. Nut. Cycl. Agroecosys 2001, 60, 275–285. [Google Scholar] [CrossRef]
- Donaghy, D.J.; Fulkerson, W.J. Priority for allocation of water-soluble carbohydrate reserves during regrowth of Lolium perenne. Grass Forage Sci. 1998, 53, 211–218. [Google Scholar] [CrossRef]
- Donaghy, D.J.; Turner, L.R.; Adamczewski, K.A. Effect of defoliation management on water-soluble carbohydrate energy reserves, dry matter yields, and herbage quality of tall fescue. Agron. J. 2008, 100, 122–127. [Google Scholar] [CrossRef]
- Rawnsley, R.P.; Donaghy, D.J.; Fulkerson, W.J.; Lane, P.A. Changes in the physiology and feed quality of cocksfoot (Dactylis glomerata L.) during regrowth. Grass Forage Sci. 2002, 57, 203–211. [Google Scholar] [CrossRef]
- Miller, L.A.; Moorby, J.M.; Davies, D.R.; Humphreys, M.O.; Scollan, N.D.; MacRae, J.C.; Theodorou, M.K. Increased concentration of water-soluble carbohydrate in perennial ryegrass (Lolium perenne L.): Milk production from late-lactation dairy cows. Grass Forage Sci. 2001, 56, 383–394. [Google Scholar] [CrossRef]
- Turner, L.R.; Donaghy, D.J.; Pembleton, K.; Rawnsley, R.P. Longer defoliation interval ensures expression of the “high sugar” trait in perennial ryegrass cultivars in cool temperate Tasmania, Australia. J. Agric. Sci. 2015, 153, 995–1005. [Google Scholar] [CrossRef]
- Rasmussen, S.; Parsons, A.J.; Xue, H.; Newman, J.A. High sugar grasses—Harnessing the benefits of new cultivars through grazing management. Proc. N. Z. Grassl. Assoc. 2009, 71, 167–175. [Google Scholar]
- Gregorini, P.; Beukes, P.C.; Bryant, R.H.; Romera, A.J. A brief overview and simulation of the effects of some feeding strategies on nitrogen excretion and enteric methane emission from grazing dairy cows. In Proceedings of the 4th Australasian Dairy Science Symposium, Lincoln, New Zealand, 31 August–2 September 2010; pp. 29–43.
- Van Bysterveldt, A.; Christie, R. Dairy farmer adoption of science demonstrated by a commercially focussed demonstration farm. In Proceedings of the 3rd Australasian Dairy Science Symposium, Melbourne, Australia, November 2007; pp. 535–540.
- Beukes, P.C.; McCarthy, S.; Wims, C.M.; Romera, A.J. Regular estimates of paddock pasture mass can improve profitability on New Zealand dairy farms. J. N. Z. Grassl. 2015, 77, 29–34. [Google Scholar]
- Hodgson, J. Influence of sward characteristics on diet selection and herbage intake by the grazing animal. In Nutritional Limits to Animal Production from Pasture; Hacker, J.B., Ed.; CAB International: Wallingford, UK, 1982; pp. 153–166. [Google Scholar]
- Leuchtmann, A.; Bacon, C.W.; Schardl, C.L.; White, J.F., Jr.; Tadych, M. Nomenclatural realignment of Neotyphodium species with genus Epichloë. Mycologia 2014, 106, 202–215. [Google Scholar] [CrossRef] [PubMed]
- Hume, D.E.; Cooper, B.M.; Pankhurst, K.A. The role of endophyte in determining the persistence and productivity of perennial ryegrass, tall fescue and meadow fescue in Northland. Proc. N. Z. Grassl. Assoc. 2009, 71, 145–150. [Google Scholar]
- Thom, E.R.; Clark, D.A.; Waugh, C.D. Growth, persistence and alkaloid levels of endophyte-infected and endophyte-free ryegrass pastures grazed by dairy cows in northern New Zealand. N. Z. J. Agric. Res. 2009, 42, 241–253. [Google Scholar] [CrossRef]
- Popay, A.J.; Hume, D.E. Endophytes improve persistence by controlling insects. In Pasture Persistence Symposium; Mercer, C.F., Ed.; New Zealand Grassland Association: Dunedin, New Zealand, 2011; pp. 149–156. [Google Scholar]
- Thom, E.R.; Popay, A.J.; Hume, D.E.; Fletcher, L.R. Evaluating the performance of endophytes in farm systems to improve farmer outcomes—A review. Crop Pasture Sci. 2012, 63, 927–943. [Google Scholar] [CrossRef]
- Hume, D.E.; Sewell, J.C. Agronomic advantages conferred by endophyte infection of perennial ryegrass (Lolium perenne L.) and tall fescue (Festuca arundinaceae Schreb.) in Australia. Crop Pasture Sci. 2014, 65, 747–757. [Google Scholar] [CrossRef]
- Stewart, A.V. Genetic origins of perennial ryegrass (Lolium perenne) for New Zealand pastures. In Breeding for Success: Diversity in Action, Proceedings of the 13th Australasian Plant Breeding Conference, Christchurch, New Zealand, 18–21 April 2006; Mercer, C.F., Ed.; pp. 11–20.
- Lee, J.M.; Matthew, C.; Thom, E.R.; Chapman, D.F. Perennial ryegrass breeding in New Zealand: A dairy industry perspective. Crop Pasture Sci. 2012, 63, 107–127. [Google Scholar] [CrossRef]
- Gowen, N.; O’Donovan, M.; Casey, I.; Rath, M.; Delaby, L.; Stakelum, G. The effect of grass cultivars differing in heading date and ploidy on the performance and dry matter intake of spring calving dairy cows at pasture. Anim. Res. 2003, 52, 321–336. [Google Scholar] [CrossRef]
- O’Donovan, M.; Delaby, L. A comparison of perennial ryegrass cultivars differing in heading date and grass ploidy with spring calving dairy cows at two stocking rates. Anim. Res. 2005, 54, 337–350. [Google Scholar] [CrossRef]
- Chapman, D.F.; Edwards, G.R.; Stewart, A.V.; McEvoy, M.; O’Donovan, M.; Waghorn, G.C. Valuing forages for genetic selection: Which traits should we focus on? Anim. Prod. Sci. 2015, 55, 869–882. [Google Scholar] [CrossRef]
- Wims, C.M.; McEvoy, M.; Delaby, L.; Boland, T.M.; O’Donovan, M. Effect of perennial ryegrass (Lolium perenne L.) cultivars on the milk yield of grazing dairy cows. Animal 2013, 7, 410–421. [Google Scholar] [CrossRef] [PubMed]
- Mercer, C.F. (Ed.) Pasture Persistence Symposium. Grassland Research and Practice Series No. 15; New Zealand Grassland Association: Dunedin, New Zealand, 2011.
- Parsons, A.J.; Edwards, G.R.; Newton, P.C.D.; Chapman, D.F.; Caradus, J.R.; Rasmussen, S.; Rowarth, J.S. Past lessons and future prospects: Plant breeding for yield and persistence in cool-temperate pastures. Grass Forage Sci. 2011, 66, 153–172. [Google Scholar] [CrossRef]
- Popay, A.J.; Thom, E.R. Endophyte effects on major insect pests in Waikato dairy pasture. Proc. N. Z. Grassl. Assoc. 2009, 71, 121–126. [Google Scholar]
- Camlin, M.S.; Stewart, R.H. The assessment of persistence and its application to the evaluation of mid season and late perennial ryegrass cultivars. J. Br. Grassl. Soc. 1978, 33, 275–282. [Google Scholar] [CrossRef]
- Cashman, P.; O’Donovan, M.; McEvoy, M.; Shalloo, L. Quantifying ground score change on perennial ryegrass swards exposed to different grazing regimes. In Proceedings of the Agricultural Research Forum, Tullamore, Ireland, 10–11 March 2014; p. 89.
- McEvoy, M.; McHugh, N.; O’Donovan, M.; Grogan, D.; Shalloo, L. Pasture profit index: Updated economic values and inclusion of persistency. In EGF at 50: The Future of European Grasslands; Hopkins, A., Collins, R.P., Fraser, M.D., King, V.R., Lloyd, D.C., Moorby, J.M., Robson, P.R.H., Eds.; European Grassland Federation: Aberystwyth, UK, 2014; pp. 843–845. [Google Scholar]
- Chapman, D.F.; Muir, P.D.; Faville, M.J. Persistence of dry matter yield among New Zealand perennial ryegrass (Lolium perenne L.) cultivars: Insights from a long-term data set. J. N. Z. Grassl. 2015, 77, 177–184. [Google Scholar]
- Matthew, C.; Agnusdei, M.G.; Assuero, S.G.; Sbrissia, A.F.; Scheneiter, O.; da Silva, S.C. State of knowledge in tiller dynamics. In Proceedings of the XXII International Grassland Congress, Sydney, Australia, 15–19 September 2013; pp. 1041–1044.
- Edwards, G.R.; Chapman, D.F. Plant responses to defoliation and relationships with pasture persistence. In Pasture persistence symposium; Mercer, C.F., Ed.; New Zealand Grassland Association: Dunedin, New Zealand, 2011; pp. 39–46. [Google Scholar]
- Matthew, C.; Assuero, S.G.; Black, C.K.; Sackville-Hamilton, N.R. Tiller dynamics in grazed swards. In Grassland Ecophysiology and Grazing Ecology; Lemaire, G., Hodgson, J., Moraes, A., de Carvalho, P.C.F., Nabinger, C., Eds.; CAB International: Wallingford, UK, 2000; pp. 127–150. [Google Scholar]
- Chapman, D.F.; Edwards, G.R.; Nie, Z.N. Plant responses to climate and relationships with pasture persistence. In Pasture Persistence Symposium; Mercer, C.F., Ed.; New Zealand Grassland Association: Dunedin, New Zealand, 2011; pp. 99–107. [Google Scholar]
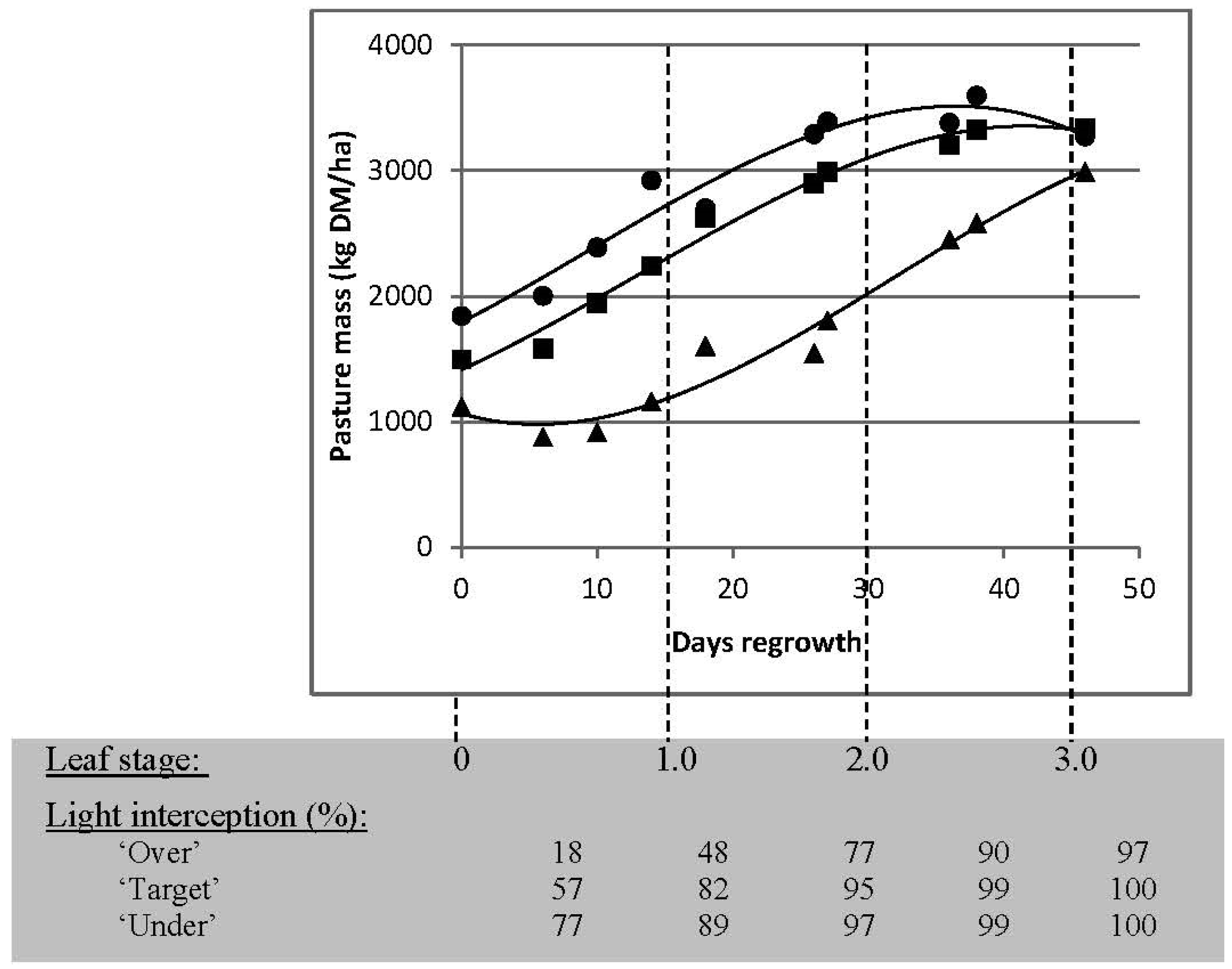
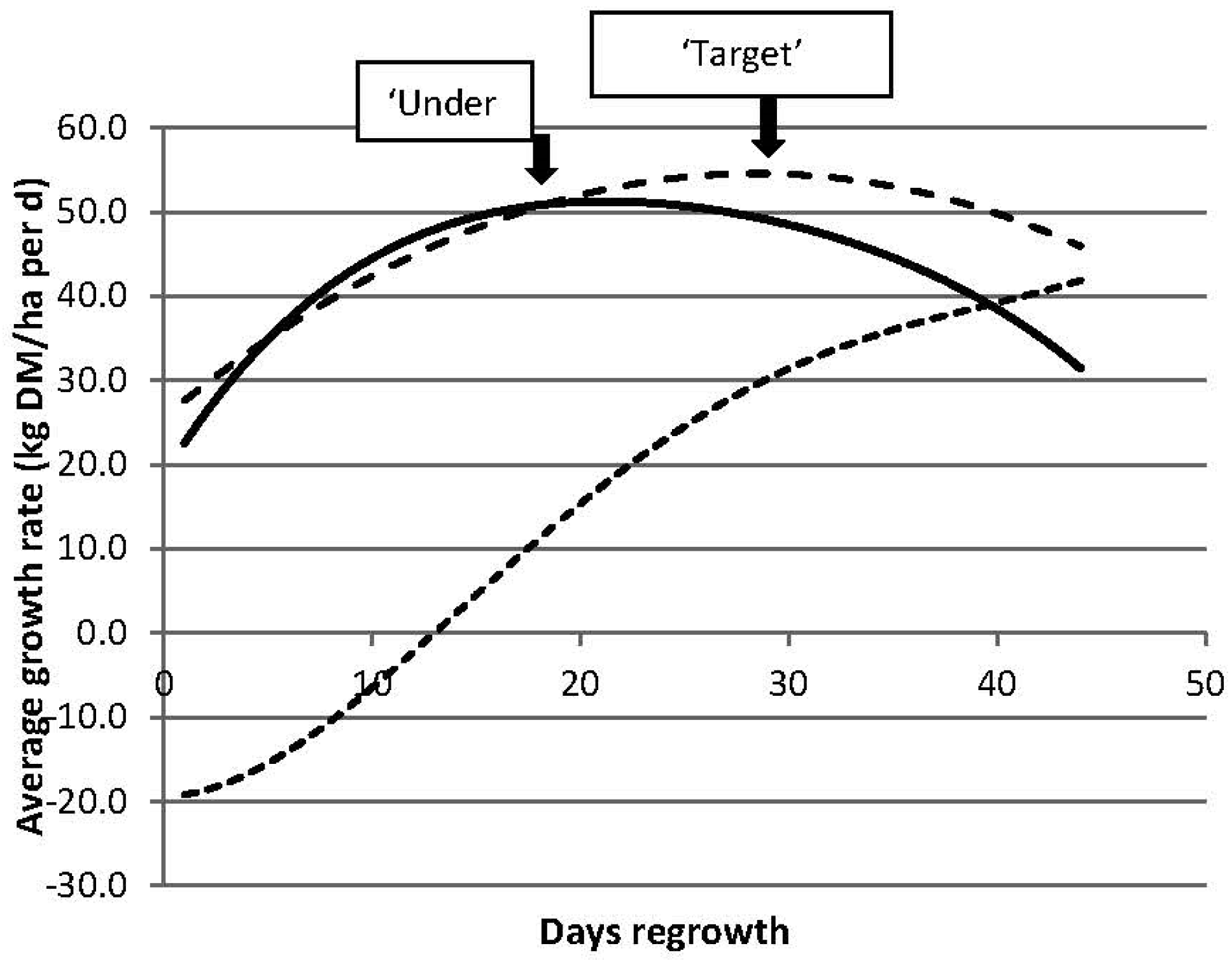
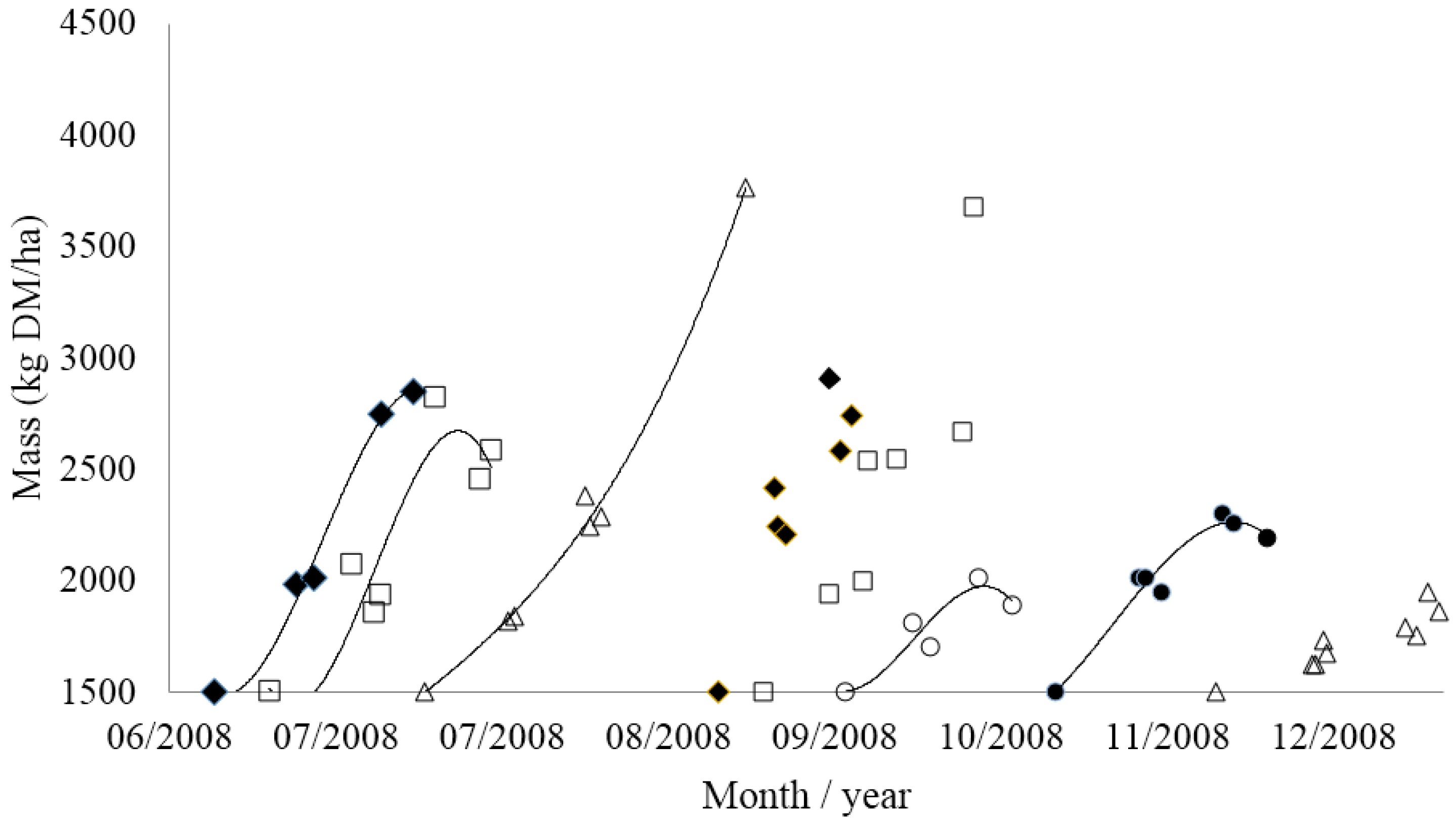
| Residual Pasture State | Timing of Next Grazing | ||
|---|---|---|---|
| 2 Leaves (30 Days) | 2.5 Leaves (37.5 Days) | 3 Leaves (45 Days) | |
| “Under” grazing | 2095 | 2260 | 2065 |
| “Target” grazing | 2100 | 2440 | 2595 |
| “Over” grazing | 1415 | 1945 | 2375 |
| Period | Growth Rate (kg·DM/ha/d) | Leaf Appearance Interval (d) | Contribution of 1st:2nd:3rd Leaves to Total DM (%) 1 | DM grown (kg/ha) When Grazed at | Difference in DM Grown (kg/ha) | EV for Season ($/kg·DM) 2 | EV of the Difference ($/ha) | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1.5-Leaf | 2-Leaf | 3-Leaf | 1.5- v. 3-Leaf | 2- v 3-Leaf | 1.5- v. 3-Leaf | 2- v 3-Leaf | |||||
| February–March | 55 | 10 | 25:35:40 | 2810 | 2970 | 3300 | −500 | −330 | 0.25 | −125 | −85 |
| April–May | 26 | 15 | 25:35:40 | 860 | 940 | 980 | −120 | −40 | 0.31 | −35 | −10 |
| June–July | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| August–September | 34 | 15 | 25:35:40 | 1120 | 1220 | 1280 | −150 | −50 | 0.42 | −65 | −20 |
| October–November | 95 | 7.5 | 30:35:35 | 7200 | 7410 | 7550 | −360 | −140 | 0.29 | −105 | −40 |
| December–January | 95 | 10 | 25:35:40 | 4850 | 5130 | 5700 | −860 | −570 | 0.18 | −155 | −105 |
| Totals | 16,840 | 17,670 | 18,810 | −1990 | −1130 | −485 | −260 | ||||
© 2016 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chapman, D.F. Using Ecophysiology to Improve Farm Efficiency: Application in Temperate Dairy Grazing Systems. Agriculture 2016, 6, 17. https://doi.org/10.3390/agriculture6020017
Chapman DF. Using Ecophysiology to Improve Farm Efficiency: Application in Temperate Dairy Grazing Systems. Agriculture. 2016; 6(2):17. https://doi.org/10.3390/agriculture6020017
Chicago/Turabian StyleChapman, David F. 2016. "Using Ecophysiology to Improve Farm Efficiency: Application in Temperate Dairy Grazing Systems" Agriculture 6, no. 2: 17. https://doi.org/10.3390/agriculture6020017
APA StyleChapman, D. F. (2016). Using Ecophysiology to Improve Farm Efficiency: Application in Temperate Dairy Grazing Systems. Agriculture, 6(2), 17. https://doi.org/10.3390/agriculture6020017





