Assessment of Morphological Diversity, Yield Components, and Seed Biochemical Composition in Common Bean (Phaseolus vulgaris L.) Landraces
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Field Environmental Conditions
2.2.1. Location and Soil Characteristics
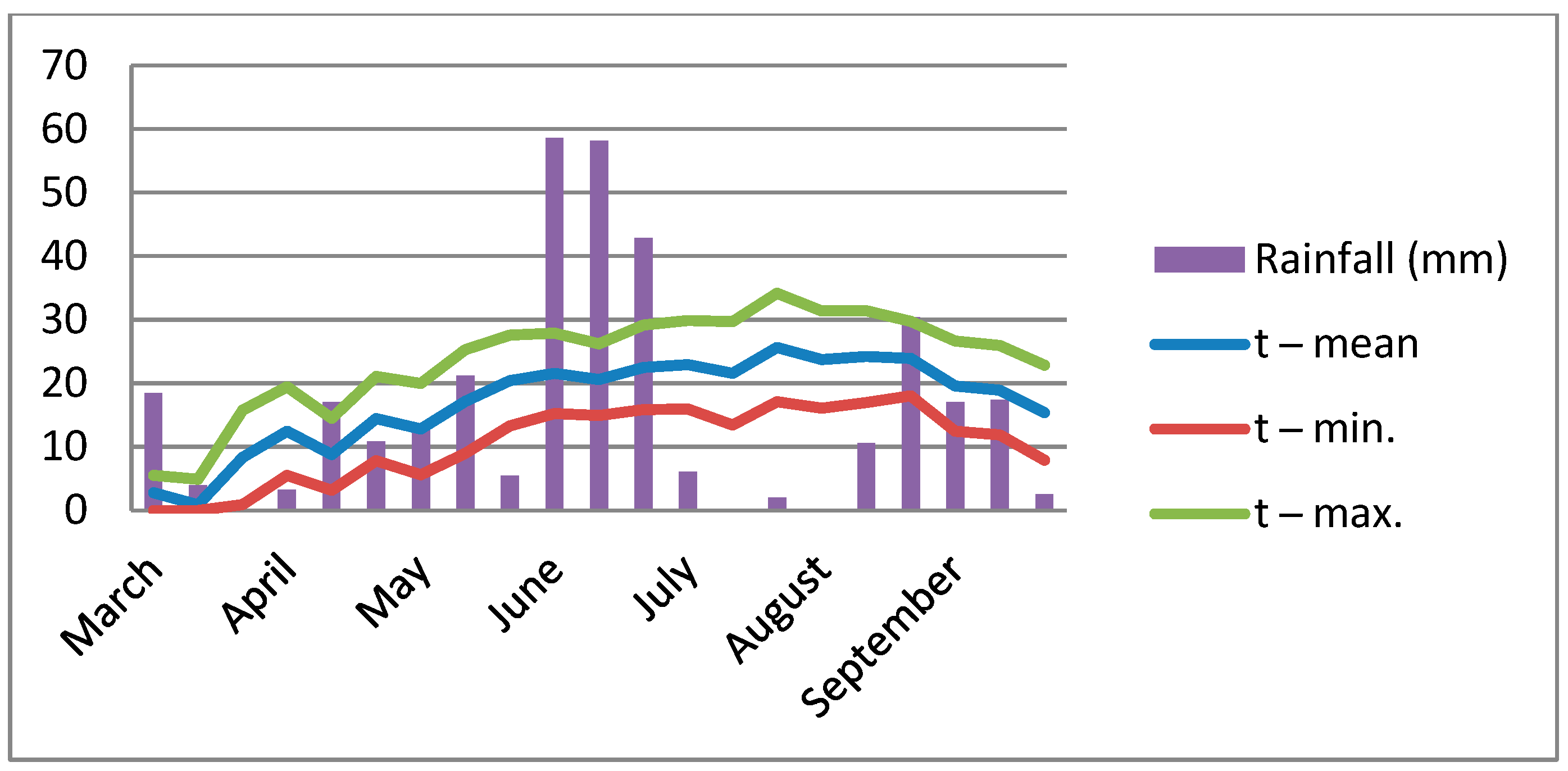
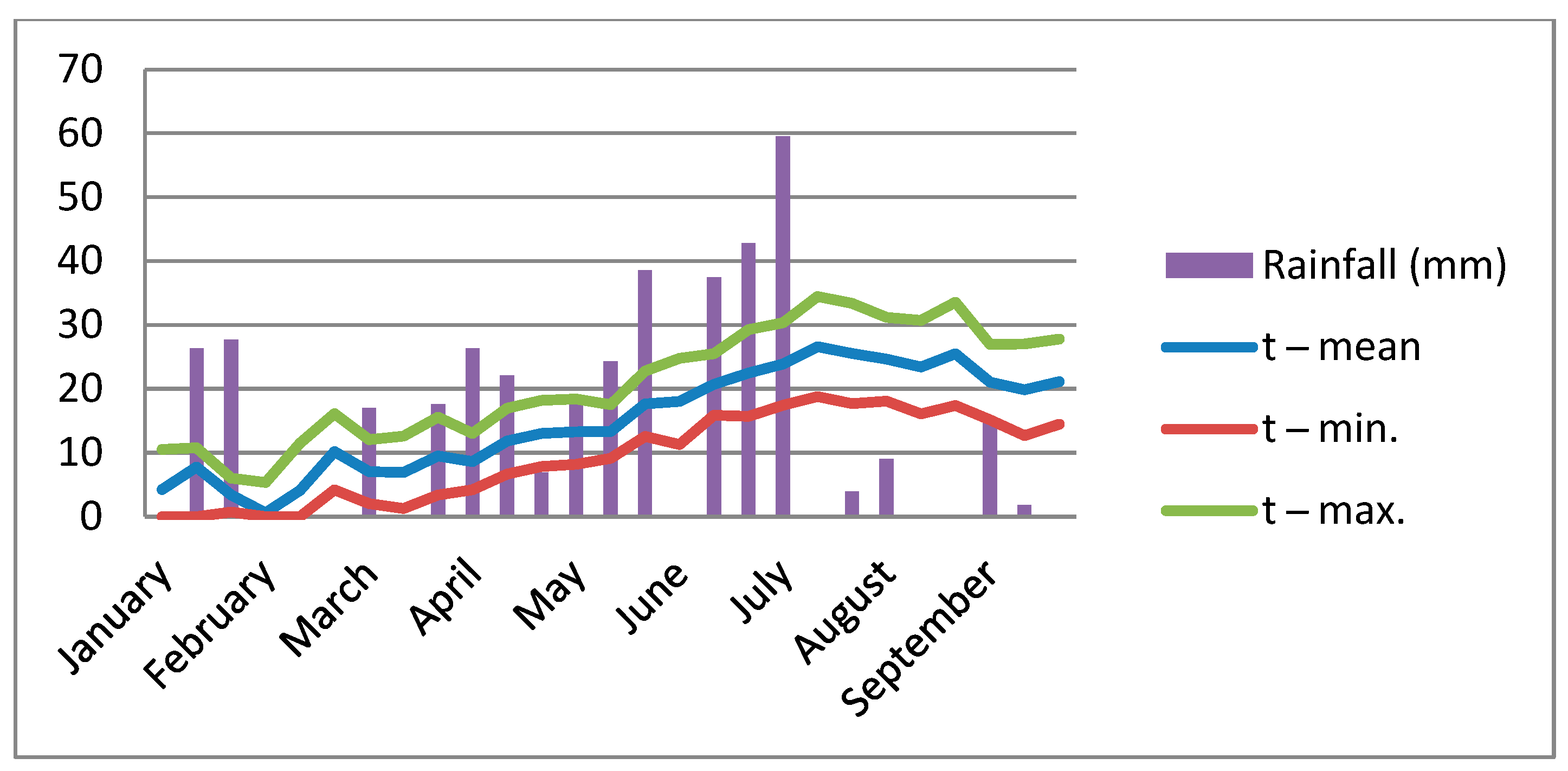
2.2.2. Weather Conditions
2.3. Experimental Design
2.4. Morphological and Agro-Biological Traits
2.5. Seed Biochemical Analyses
2.6. Statistical Analysis
3. Results
3.1. Agro-Biological Data
3.2. Variation in Agro-Morphological Traits
3.3. Phenotypic Correlation
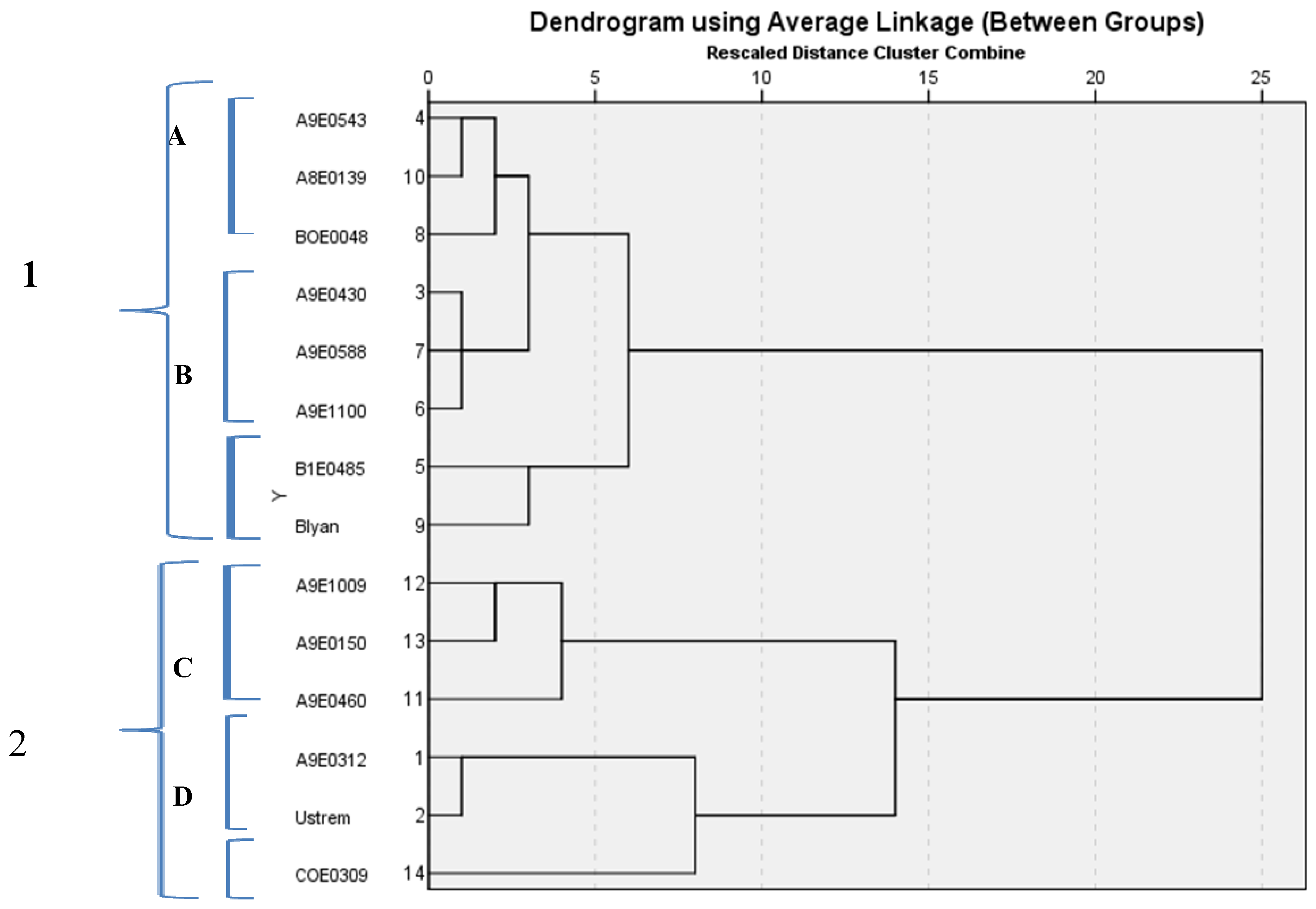
3.4. Hierarchical Cluster Analysis
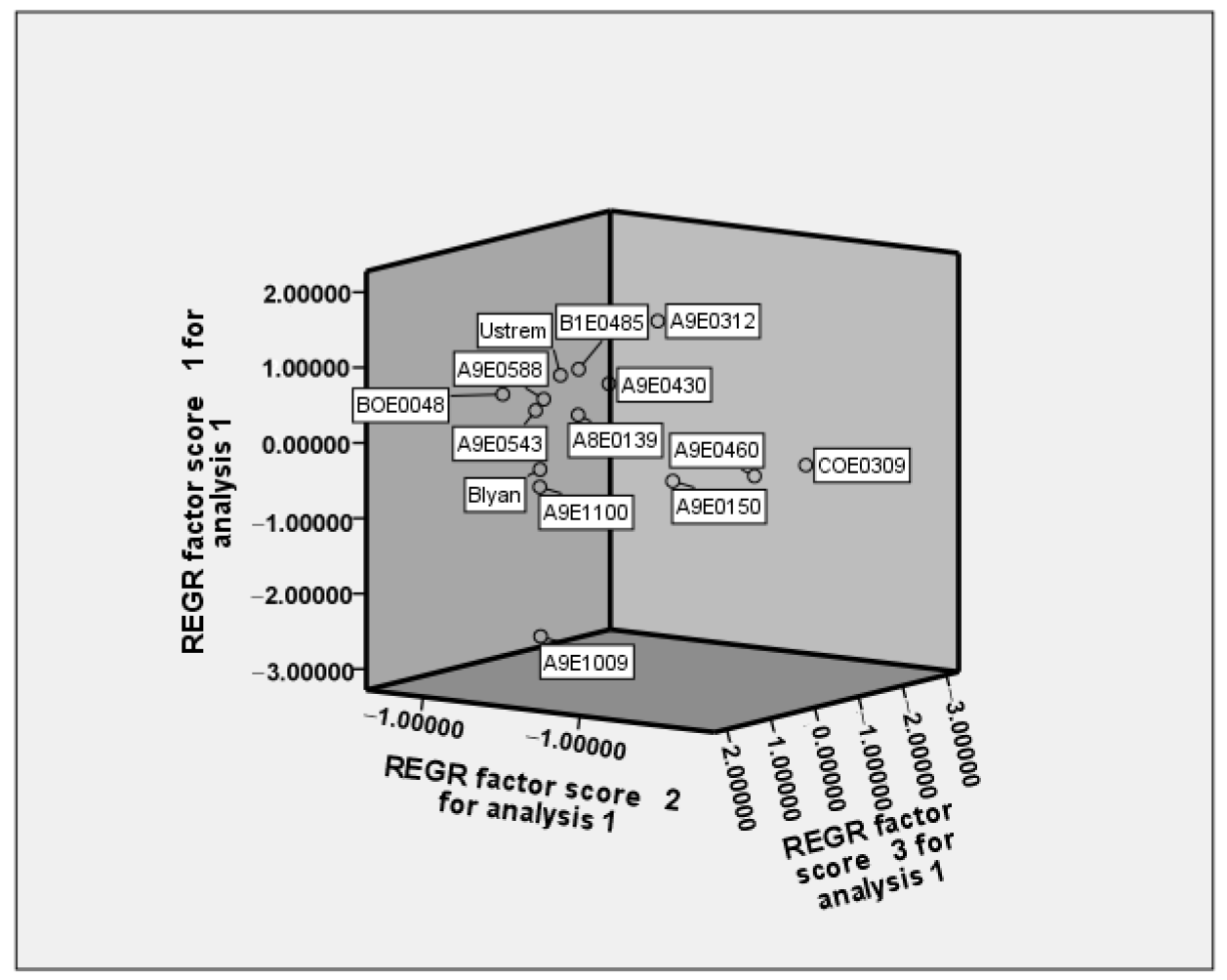
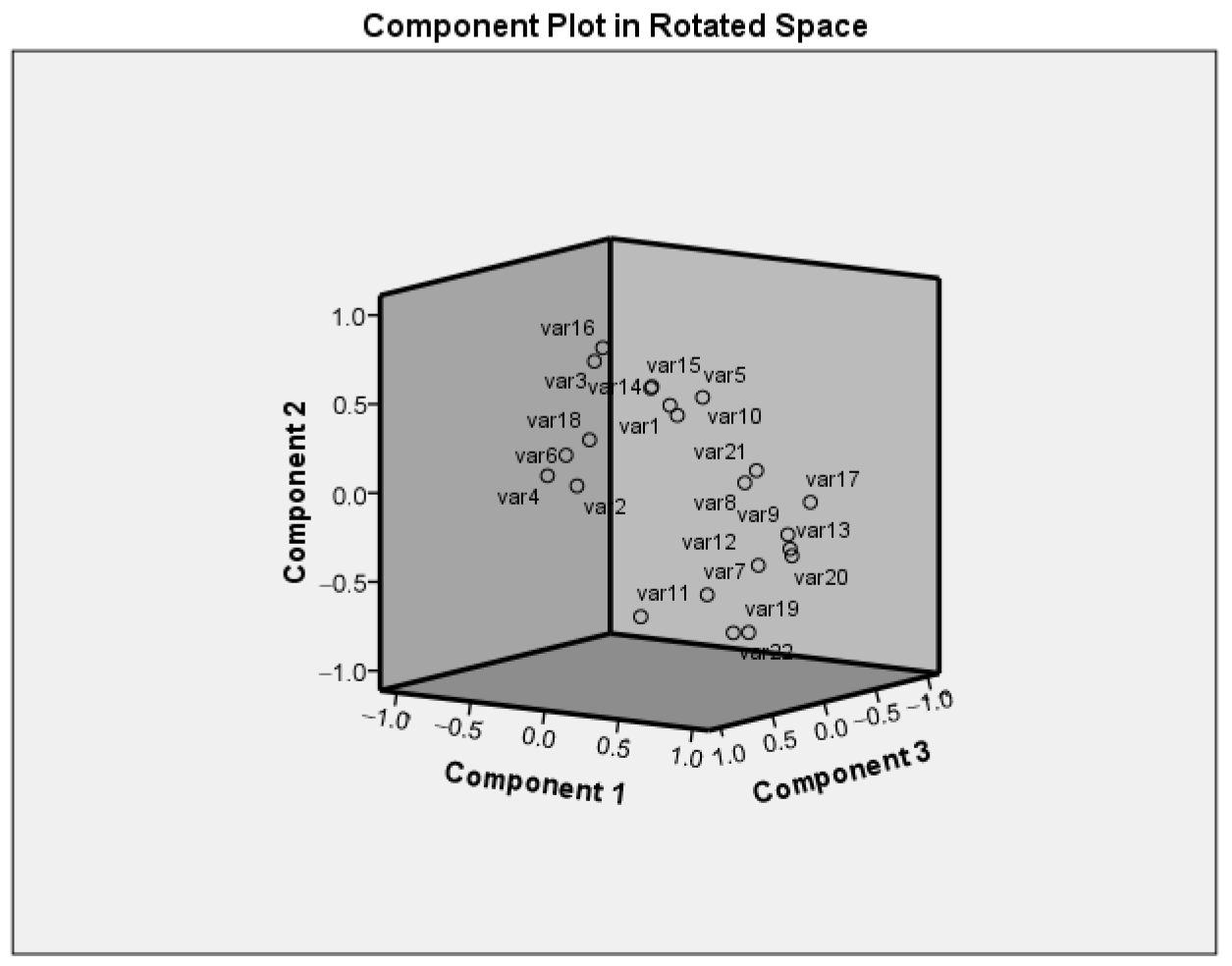
3.5. Principal Component Analysis
3.6. Seeds’ Biochemical Composition
4. Discussion
4.1. Morphological Trait Diversity
4.2. Seed Biochemical Composition
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Singh, S.P.; Gepts, P.; Debouck, D.G. Races of common bean (Phaseolus vulgaris, Fabaceae). Econ. Bot. 1991, 45, 379–396. [Google Scholar] [CrossRef]
- Nadeem, M.A.; Karaköy, T.; Yeken, M.Z.; Habyarimana, E.; Hatipoğlu, R.; Çiftçi, V.; Nawaz, M.A.; Sönmez, F.; Shahid, M.Q.; Yang, S.H. Phenotypic characterization of 183 Turkish common bean accessions for agronomic, trading, and consumer-preferred plant characteristics for breeding purposes. Agronomy 2020, 10, 272. [Google Scholar] [CrossRef]
- Nasar, S.; Shaheen, H.; Murtaza, G.; Tinghong, T.; Arfan, M.; Idrees, M. Socioeconomic Evaluation of Common Bean (Phaseolus vulgaris L.) Cultivation in Providing Sustainable Livelihood to the Mountain Populations of Kashmir Himalayas. Plants 2023, 12, 213. [Google Scholar] [CrossRef] [PubMed]
- FAO, Food and Agricultural Organization. Crops and Livestock Products. Available online: https://www.fao.org/faostat/en/#data/QCL (accessed on 2 May 2025).
- Naithani, S. The Origins of Crop Plants. In Hystory and Science of Cultivated Plants; Oregon State University: Corvallis, OR, USA, 2021; Available online: https://open.oregonstate.education/cultivatedplants/chapter/cropplants/ (accessed on 28 April 2025).
- Kaplan, L.; Kaplan, L. Genetic resources of Phaseolus beans: Their maintenance, domestication, evolution and utilisation. In Current Plant Science and Biotechnology in Agriculture; Gepts, P., Ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1988; pp. 125–142. [Google Scholar]
- Gepts, P.; Bliss, F.A. Phaseolin variability among wild and cultivated common beans (Phaseolus vulgaris) from Colombia. Econ. Bot. 1986, 40, 469–478. [Google Scholar] [CrossRef]
- Kwak, M.; Gepts, P.A.E. Structure of genetic diversity in the two major gene pools of common bean (Phaseolus vulgaris L., Fabaceae). Theor. Appl. Genet. 2009, 118, 979–992. [Google Scholar] [CrossRef]
- Sicard, D.; Nanni, L.; Porfiri, O.; Bulfon, D.; Papa, R. Genetic diversity of Phaseolus vulgaris L. and P. coccineus L. landraces in central Italy. Plant Breed. 2005, 124, 464–472. [Google Scholar] [CrossRef]
- Angioi, S.A.; Rau, D.; Attene, G.; Nanni, L.; Bellucci, E.; Logozzo, G.; Negri, V.; Zeuli, P.L.S.; Papa, R. Beans in Europe: Origin and structure of the European landraces of Phaseolus vulgaris L. Theor. Appl. Genet. 2010, 121, 829–843. [Google Scholar] [CrossRef]
- Monterroso, V.A.; Wien, H.C. Flower and pod abscission due to heat stress in beans. J. Amer. Soc. Hort. Sci. 1990, 115, 631–634. [Google Scholar] [CrossRef]
- Salcedo, J.M. Regeneration guidelines: Common bean. In Crop Specific Regeneration Guidelines [CD-ROM]; Dulloo, M.E., Thormann, I., Jorge, M.A., Hanson, J., Eds.; CGIAR System-wide Genetic Resource Programme (SGRP): Rome, Italy, 2008; p. 9. [Google Scholar]
- Magrini, M.-B.; Anton, M.; Cholez, C.; Corre-Hellou, G.; Duc, G.; Jeuffroy, M.-H.; Meynard, J.-M.; Pelzer, E.; Voisin, A.-S.; Walrand, S. Why are grain-legumes rarely present in cropping systems despite their environmental and nutritional benefits? Analyzing lock-in in the French agrifood system. Ecol. Econ. 2016, 126, 152–162. [Google Scholar] [CrossRef]
- Félix-Valencia, P.; Padilla-Valenzuela, I.; Ortiz-Enríquez, E.J.; Torres-Cruz, M.M.; Fuentes-Dávila, G. Effect of temperature, relative humidity and drip irrigation on productivity ofcommon bean (Phaseolus vulgaris L.) in the Yaqui Valley, Sonora, Mexico. GSC Adv. Res. Rev. 2023, 17, 010–020. [Google Scholar] [CrossRef]
- Lisciani, S.; Marconi, S.; Le Donne, C.; Camilli, E.; Aguzzi, A.; Gabrielli, P.; Gambelli, L.; Kunert, K.; Marais, D.; Vorster, B.J.; et al. Legumes and common beans in sustainable diets: Nutritional quality, environmental benefits, spread and use in food preparations. Front. Nutr. 2024, 11, 1385232. [Google Scholar] [CrossRef]
- Schneider, C.V.A. Overview of the market and consumption of pulses in Europe. Br. J. Nutr. 2003, 88 (Suppl. S3), S243–S250. [Google Scholar] [CrossRef] [PubMed]
- Basso Los, G.F.; Zielinski, F.A.c.A.; Wojeicchowski, P.J.; Nogueira, A.; Mottin Demiate, M.I. Beans (Phaseolus vulgaris L.): Whole seeds with complex chemical composition. Curr. Opin. Food Sci. 2018, 19, 63–71. [Google Scholar]
- Singh, P.R. Kidney bean starch: Composition, structure, properties, and modifications. In Non-Conventional Starch Sources, Properties, Functionality, and Applications, Chapter 15; Lorenzo, J.M., Bangar, S.P., Eds.; Academic Press, Science Direct: Cambridge, MA, USA, 2024; pp. 439–465. [Google Scholar] [CrossRef]
- Martino, H.S.D.; Bigonha, S.M.; Cardoso, L.D.M.; Rosa, C.D.O.B.; Costa, N.M.B.; Cárdenas, L.D.L.Á.R.; Ribeiro, S.M.R. Nutritional and bioactive compounds of bean: Benefits to human health. In Hispanic Foods: Chemistry and Bioactive Compounds; American Chemical Society: Washington, DC, USA, 2012; pp. 233–258. [Google Scholar] [CrossRef]
- Moussou, N.; Ouazib, M.; Wanasundara, J.; Zaidi, F.; Rubio, L.A. Nutrients and non-nutrients composition and in vitro starch digestibility of five Algerian legume seed flours. Internat. Food Res. J. 2019, 26, 1339–1349. [Google Scholar]
- Marathe, S.A.; Rajalakshmi, V.; Jamdar, S.N.; Sharma, A. Comparative study on antioxidant activity of different varieties of commonly consumed legumes in India. Food Chem. Toxicol. 2011, 49, 2005–2012. [Google Scholar] [CrossRef] [PubMed]
- Lioi, L.; Piergiovanni, A. Book chapter 2-European Common Bean. In Genetic and Genomic Resources of Grain Legume Improvement; Singh, M., Upadhyaya, H.D., Bisht, B.S., Eds.; Elsevier: Amsterdam, The Netherlands, 2013; pp. 11–40. [Google Scholar] [CrossRef]
- Mavromatis, A.G.; Arvanitoyannis, S.; Korkovelos, A.E.; Giakountis, A.; Chatzitheodorou, V.A.; Goulas, C.K. Genetic diversity among common bean (Phaseolus vulgaris L.) Greek landraces and commercial cultivars: Nutritional components, RAPD and morphological markers. Span. J. Agric. Res. 2010, 8, 986–994. [Google Scholar] [CrossRef]
- Ntuli, N.R. Morpho-Agronomic Variation among Phaseolus vulgaris Landraces: A Review. In Rediscovery of Landraces as a Resource for the Future; InTechOpen: London, UK, 2017. [Google Scholar] [CrossRef]
- Arteaga, S.; Yabor, L.; Torres, J.; Solbes, E.; Muñoz, E.; Díez, M.J.; Vicente, O.; Boscaiu, M. Morphological and agronomic characterization of Spanish landraces of Phaseolus vulgaris L. Agriculture 2019, 9, 149. [Google Scholar] [CrossRef]
- Mallor, C.; Barberán, M.; Aibar, J. Recovery of a common bean landrace (Phaseolus vulgaris L.) for commercial purposes. Front. Plant Sci. 2018, 9, 1440. [Google Scholar] [CrossRef]
- Lioi, L.; Piergiovanni, A.R.; Pignone, D.; Puglisi, S.; Santantonio, M.; Sonnante, G. Genetic diversity of some surviving on-farm Italian common bean (Phaseolus vulgaris L.) landraces. Plant Breed. 2005, 124, 576–581. [Google Scholar] [CrossRef]
- Chávez-Servia, J.L.; Heredia-García, E.; Mayek-Pérez, N.; Aquino-Bolaños, E.N.; Hernández-Delgado, S.; Carrillo-Rodríguez, J.C.; Gill-Langarica, H.R.; Vera-Guzmán, A.M. Diversity of common bean (Phaseolus vulgaris L.) landraces and the nutritional value of their grains. In Grain Legumes; IntechOpen: Rijeka, Croatia, 2016. [Google Scholar] [CrossRef]
- Stoilova, T.; Berova, M.; Kouzmova, K.; Stamatov, S. Study on diversity of Phaseolus spp. landraces with reference to global climate change. Afr. J. Agric. Res. 2014, 9, 2925–2935. [Google Scholar] [CrossRef]
- Bioversity Int. (IBPGR). Descriptors for Phaseolus vulgaris; IBPGR Secretariat: Rome, Italy, 1982. [Google Scholar]
- AOAC. Official Methods of Analysis, 16th ed.; Association of Official Analytical Chemists: Washington, DC, USA, 1995. [Google Scholar]
- AOAC. Kjeldahl’s method for protein determination in cereals and feed. In Official Methods of Analysis of AOAC International, Method 945, 18-B, 16th ed.; Association of Official Analytical Chemists: Washington, DC, USA, 1996. [Google Scholar]
- ISO 659; Animal and Vegetable Fats and Oils. Oilseeds—Determination of Oil Content (Reference Method). International Organization for Standardization: Geneva, Switzerland, 2009.
- Genadiev, A.; Kalcheva, D.; Nenchev, N.; Tevekelev, D.; Chadariva, N. Analysis of Food Products; Ed Technika: Sofia, Bulgaria, 1968; p. 695. (In Bulgarian) [Google Scholar]
- FAO. Food Energy—Methods of Analysis and Conversion Factors, Report of a Technical Workshop; FAO Food and Nutrition Paper 77; FAO: Rome, Italy, 2003. [Google Scholar]
- López-Hidalgo, C.; Meijón, M.; Lamelas, L.; Valledor, L. The rainbow protocol: A sequential method for quantifying pigments, sugars, free amino acids, phenolics, flavonoids and MDA from a small amount of sample. Plant Cell Environ. 2021, 44, 1977–1986. [Google Scholar] [CrossRef] [PubMed]
- Hansen, J.; Møller, I.B. Percolation of starch and soluble carbohydrates from plant tissue for quantitative determination with anthrone. Anal. Biochem. 1975, 68, 87–94. [Google Scholar] [CrossRef]
- Moore, S.; Stein, W.H. A modified ninhydrin reagent for the photometric determination of amino acids and related compounds. J. Biol. Chem. 1954, 211, 907–913. [Google Scholar] [CrossRef]
- Ainsworth, E.A.; Gillespie, K.M. Estimation of total phenolic content and other oxidation substrates in plant tissues using Folin-Ciocalteu reagent. Nat. Protoc. 2007, 2, 875–877. [Google Scholar] [CrossRef]
- Kakade, M.L.; Rackis, J.J.; McGhee, J.E.; Puski, G. Determination of trypsin inhibitor activity of soy products: A collaborative analysis of an improved procedure. Cereal Chem. 1974, 51, 376–382. [Google Scholar]
- IBM SPSS Statistics 19 Core System User’s Guide. Available online: https://ibm-spss-statistics.software.informer.com/download/#google_vignette (accessed on 16 December 2024).
- Lidansky, T. Statistical Methods in Biology and in Agriculture; Zemizdat: Sofia, Bulgaria, 1988; pp. 18–20. [Google Scholar]
- Ward, J.H. Hierarchical grouping to optimize an objective function. J. Amer. Stat. Assoc. 1963, 58, 236. [Google Scholar] [CrossRef]
- Angelova, S.; Stoilova, T. Maintenance, enrichment and utilization of grain legume collections in Bulgaria. Acta Horticult. 2009, 830, 695–700. [Google Scholar] [CrossRef]
- Casquero, A.P.; Lema, M.; Santalla, M.; De Ron, M.A. Performance of Common Bean (Phaseolus vulgaris L.) Landraces from Spain in the Atlantic and Mediterranean Environments. Genet. Res. Crop Evol. 2006, 53, 1021–1032. [Google Scholar] [CrossRef]
- Piergiovanni, P.A.; Villecco, D.; Lioi, L.; Zaccardelli, M. Comparison among recognized and non-recognized Phaseolus vulgaris L. landraces as traditional agro-food products of the Campania region in Italy. Genet. Res. Crop Evol. 2015, 62, 1009–1019. [Google Scholar] [CrossRef]
- Sincovoć, L.; Pipan, B.; Sincović, E.; Meglić, V. Morphological seed characterization of common (Phaseolus vulgaris L.) and runner (Phaseolus coccineus L.) bean germplasm: A Slovenian genebank example. BioMed. Res. Int. 2019, 2019, 6376948. [Google Scholar] [CrossRef]
- Mauceri, A.; Romeo, M.; Bacchi, M.; Prieti, G. Pheno-morphological, agronomic and genetic diversity in a common bean core collection from Calabria (Italy). Ital. J. Agron. 2025, 20, 100038. [Google Scholar] [CrossRef]
- Long, J.; Zhang, J.; Zhang, X.; Wu, J.; Chen, H.; Wang, P.; Wang, Q.; Du, C. Genetic diversity of common bean (Phaseolus vulgaris L.) germplasm resources in Chongqing, evidenced by morphological characterization. Front. Genet. 2020, 11, 697. [Google Scholar] [CrossRef]
- Jan, S.; Rather, I.A.; Sofi, P.A.; Wani, M.A.; Sheikh, F.A.; Bhat, M.A.; Mir, R.R. Characterization of common bean (Phaseolus vulgaris L.) germplasm for morphological and seed nutrient traits from Western Himalayas. Legume Sci. 2021, 3, e86. [Google Scholar] [CrossRef]
- Loha, G.; Silas, M.; Gidago, G. Effect of common bean (Phaseolus vulgaris L.) varieties and variable rates of potassium fertilizer on yield and yield related traits at Areca, Southern Etiopia. Appl. Environ. Soil Sci. 2023, 2023, 5996945. [Google Scholar] [CrossRef]
- Rodiño, A.; Santalla, M.; Montero, I.; Casquero, P.; De Ron, A. Diversity of common bean (Phaseolus vulgaris L.) germplasm from Portugal. Genet. Res. Crop Evol. 2001, 48, 409–417. [Google Scholar] [CrossRef]
- de Lima, M.S.; Carneiro, J.E.d.S.; Carneiro, P.C.S.; Pereira, C.S.; Vieira, R.F.; Cecon, P.R. Characterization of genetic variability among common bean genotypes by morphological descriptors. Crop Breed. Appl. Biotechnol. 2012, 12, 76–84. [Google Scholar] [CrossRef][Green Version]
- Reinprecht, Y.; Schram, L.; Perry, G.E.; Morneau, E.; Smith, T.H.; Pauls, K.P. Mapping yield and yield-related traits using diverse common bean germplasm. Front. Genet. 2024, 14, 1246904. [Google Scholar] [CrossRef]
- de Paula, E.; Almeida, R.N.d.; Santos, T.d.O.; Souza Neto, J.D.d.; Riva-Souza, E.M.; Posse, S.C.P.; Souza, M.N.; Madella de Oliveira, A.d.F.; Santos Júnior, A.C.; Santos, J.O.; et al. Genetic Diversity of Common Bean (Phaseolus vulgaris L.) Landraces Based on Morphological Traits and Molecular Markers. Plants 2024, 13, 2584. [Google Scholar] [CrossRef]
- Grela, E.R.; Günter, K.D. Fatty acid composition and tocopherol content of some legume seeds. Animal Feed Sci. Technol. 1995, 52, 325–331. [Google Scholar] [CrossRef]
- Guillon, F.; Champ, M.J. Carbohydrate fractions of legumes: Uses in human nutrition and potential for health. Brit. J. Nutr. 2002, 88 (Suppl. S3), 293–306. [Google Scholar] [CrossRef]
- Murube, E.; Beleggia, R.; Pacetti, D.; Nartea, A.; Frascarelli, G.; Lanzavecchia, G.; Bellucci, E.; Nanni, L.; Gioia, T.; Marciello, U.; et al. Characterization of nutritional quality traits of a common bean germplasm collection. Foods 2021, 10, 1572. [Google Scholar] [CrossRef]
- Chávez-Mendoza, C.; Sánchez, E. Antioxidant capacity and nutraceutical compounds content of six common bean (Phaseolus vulgaris L.) varieties harvested in Morelos, Mexico. Not. Sci. Biol. 2023, 15, 11353. [Google Scholar] [CrossRef]
- Rezende, A.A.; Pacheco, M.T.B.; Silva, V.S.N.D.; Ferreira, T.A.P.D.C. Nutritional and protein quality of dry Brazilian beans (Phaseolus vulgaris L.). Food Sci. Technol. 2017, 38, 421–427. [Google Scholar] [CrossRef]
- Pineda, M.I.; Galdón, B.R.; Álvarez, N.P.; Morales, D.A.; Mesa, D.R.; Romero, C.D.; Rodríguez-Rodríguez, E.M. Physico-chemical and nutritional characterization of Phaseolus vulgaris L. germplasm. Legume Res. 2023, 46, 273–280. [Google Scholar] [CrossRef]
- Escribano, M.R.; Santalla, M.; De Ron, A.M. Genetic diversity in pod and seed quality traits of common bean populations from northwestern Spain. Euphytica 1997, 93, 71–81. [Google Scholar] [CrossRef]
- Rivera, A.; Plans, M.; Sabaté, J.; Casañas, F.; Casals, J.; Rull, A.; Simó, J. The Spanish core collection of common beans (Phaseolus vulgaris L.): An important source of variability for breeding chemical composition. Front. Plant Sci. 2018, 9, 1642. [Google Scholar] [CrossRef]
- de Lima, J.; Rivadavea, W.; Kavalco, S.A.; Junior, A.C.; Lopes, A.; da Silva, G. Chemical and nutritional characterization of bean genotypes (Phaseolus vulgaris L.). AIMS Agric. Food 2021, 6, 932–944. [Google Scholar] [CrossRef]
- García-Díaz, Y.D.; Aquino-Bolaños, E.N.; Chávez-Servia, J.L.; Vera-Guzmán, A.M.; Carrillo-Rodríguez, J.C. Bioactive compounds and antioxidant activity in the common bean are influenced by cropping season and genotype. Chilean. J. Agric. Res. 2018, 78, 255–265. [Google Scholar] [CrossRef]
- Gorbe, E.; Morard, M.; Rausell-Moreno, A.; Calatayud, Á.; Penella, C. Exploring nutritional quality and bioactive compounds in Mediterranean bean landraces. J. Food Compos. Anal. 2025, 142, 107433. [Google Scholar] [CrossRef]
- Piergiovanni, A.R.; Galasso, I.; Lioi, L. Protease Inhibitors in Phaseolus spp. Seeds. In Seed Proteins: Biochemistry, Functional Properties and Health Benefits; Wilson, D.G., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2017; pp. 41–59. [Google Scholar]
- Florvil, F.; Márquez-Quiroz, C.; de-la-Cruz-Lázaro, E.; Osorio-Osorio, R.; Sánchez-Chávez, E. Bioactive compounds, antioxidant activity and mineral content of common bean varieties grown in Tabasco, Mexico. Indian J. Agricult. Res. 2022, 56, 368–372. [Google Scholar] [CrossRef]
- Cid-Gallegos, M.S.; Sánchez-Chino, X.M.; Corzo-Ríos, L.J.; del Rosario Moguel-Concha, D.; Borges-Martínez, J.E.; Jiménez-Martínez, C. Phaseolus vulgaris as a source of peptides and biofunctional compounds. In Improving Health and Nutrition Through Bioactive Compounds; Elsevier: Amsterdam, The Netherlands, 2025; pp. 79–91. [Google Scholar] [CrossRef]
- Kajiwara, V.; Moda-Cirino, V.; dos Santos Scholz, M.B. Studies on nutritional and functional properties of various genotypes of Andean beans. J. Food Sci. Technol. 2022, 59, 1468–1477. [Google Scholar] [CrossRef]
- León-Cortés, D.; Arce-Villalobos, K.; Bogantes-Ledezma, D.; Irías-Mata, A.; Chaves-Barrantes, N.; Vinas, M. Anti-aflatoxin potential of phenolic compounds from common beans (Phaseolus vulgaris L.). Food Chem. 2025, 469, 142597. [Google Scholar] [CrossRef]
- Doria, E.; Campion, B.; Sparvoli, F.; Tava, A.; Nielsen, E. Anti-nutrient components and metabolites with health implications in seeds of 10 common bean (Phaseolus vulgaris L. and Phaseolus lunatus L.) landraces cultivated in southern Italy. J. Food Comp. Anal. 2012, 26, 72–80. [Google Scholar] [CrossRef]
- Celmeli, T.; Sari, H.; Canci, H.; Sari, D.; Adak, A.; Eker, T.; Toker, C. The nutritional content of common bean (Phaseolus vulgaris L.) landraces in comparison to modern varieties. Agronomy 2018, 8, 166. [Google Scholar] [CrossRef]


| No | Accession | Origin | Biological Status | Source |
|---|---|---|---|---|
| 1 | A9E0312 | Silistra | Local population | IPGR genebank |
| 2 | Ustrem | DAI-Gen. Toshevo | Cultivar | DAI-Gen. Toshevo |
| 3 | A9E0430 | Obretenik village, Ruse | Local population | IPGR genebank |
| 4 | A9E0543 | Totleben village, Pleven | Local population | IPGR genebank |
| 5 | B1E0485 | Velingrad | Local population | IPGR genebank |
| 6 | A9E1100 | Bisser village, Haskovo | Local population | IPGR genebank |
| 7 | A9E0588 | Umarevtsi, village, Lovech | Local population | IPGR genebank |
| 8 | B0E0048 | Dimitrovgrad | Local population | IPGR genebank |
| 9 | Blyan | DAI-Gen. Toshevo | Cultivar | DAI-Gen. Toshevo |
| 10 | A8E0139 | Klisura | Local population | IPGR genebank |
| 11 | A9E0460 | Oryahovitsa village, Pleven | Local population | IPGR genebank |
| 12 | A9E1009 | Dragushinovo village, Samokov | Local population | IPGR genebank |
| 13 | A9E0150 | Sevlievo | Local population | IPGR genebank |
| 14 | C0E0309 | Sadovo | Local population | IPGR genebank |
| No | Character | Abbreviation | No | Character | Abbreviation |
|---|---|---|---|---|---|
| 1 | Days to emergence | DE | 13 | Weight of pods/plant | WPPl |
| 2 | Days to flowering | DFl | 14 | Pod length | PL |
| 3 | Duration of flowering | DurFl | 15 | Pod width | PW |
| 4 | Days to maturity | DMat | 16 | Pod thickness | PT |
| 5 | Plant height | PH | 17 | Number of seeds/pod | NSP |
| 6 | Length of medium leaf | LMl | 18 | Seed length | SL |
| 7 | Width of medium leaf | WMl | 19 | Seed width | SW |
| 8 | Weight of plant | WPl | 20 | Seed thickness | ST |
| 9 | Number of branches | NBr | 21 | Weight of seeds/plant | WSPl |
| 10 | Weight of plant without pods | WPlWP | 22 | Weight of 100 seeds | W100S |
| 11 | First pod height | FPH | 23 | Yield per plot | Yplot |
| 12 | Number of pods/plant | NPPl |
| Cat. No | H of Plant (cm) | Weight of Plant (g) | Number of Branches | Number of Pods/pl | Weight of Pods/pl (g) | Number of Seeds/Pod | Weight of Seeds/pl (g) | Size of Pod | Weight of 100 Seeds (g) | Harvest Index (%) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Length. (cm) | Width. (cm) | |||||||||||||||||||||
| M | ±D | M | ±D | M | ±D | M | ±D | M | ±D | M | ±D | M | ±D | M | ±D | M | ±D | M | ±D | M | ±D | |
| A9E0312 | 67.2 | 26.0 ** | 45.3 | 15.9 ** | 2.9 | 0.5 ** | 15.5 | 4.4 ** | 20.6 | 4.0 * | 5.4 | 0.9 ** | 13.8 | 1.7 | 10.5 | 1.4 ** | 0.9 | 0.1 ** | 22.5 | −11.1 ** | 30.6 | −8.5 ** |
| Ustrem St | 41.2 | 29.4 | 2.4 | 11.0 | 16.6 | 4.5 | 12.1 | 9.1 | 0.8 | 33.6 | 39.2 | |||||||||||
| A9E0430 | 55.9 | 14.7 ** | 28.9 | −0.5 | 2.5 | 0.1 | 12.0 | 1.0 | 13.9 | −2.7 | 5.4 | 0.9 ** | 10.4 | −1.7 | 10.1 | 0.9 ** | 0.9 | 0.1 ** | 24.9 | −8.7 ** | 35.8 | −3.3 |
| A9E0543 | 41.1 | −0.1 | 20.9 | −8.5 * | 2.3 | −0.1 | 8.0 | −3.1 ** | 12.3 | −4.3 ** | 3.6 | −0.9 ** | 8.7 | −3.4 ** | 10.6 | 1.4 ** | 0.9 | 0.0 | 33.7 | 0.1 | 41.5 | 2.3 |
| B1E0485 | 38.7 | −2.5 | 45.1 | 15.7 ** | 2.3 | −0.2 | 18.9 | 7.9 ** | 21.3 | 4.8 ** | 5.2 | 0.7 ** | 15.4 | 3.3 ** | 9.1 | −0.2 | 0.8 | 0.0 | 20.2 | −13.4 ** | 34.2 | −5.0 * |
| A9E1100 | 40.4 | −0.8 | 27.4 | −2.0 | 2.6 | 0.2 | 9.5 | −1.5 | 14.5 | −2.1 | 4.2 | −0.3 | 9.1 | −3.0 * | 10.7 | 1.5 ** | 0.9 | 0.0 | 33.7 | 0.1 | 32.8 | −6.3 ** |
| A9E0588 | 40.6 | −0.6 | 16.8 | −12.6 ** | 2.0 | −0.5 ** | 7.8 | −3.2 ** | 10.6 | −6.0 ** | 4.2 | −0.3 | 7.9 | −4.1 ** | 9.4 | 0.3 | 1.0 | 0.2 ** | 36.8 | 3.2 ** | 47.2 | 8.0 ** |
| BOE0048 | 51.8 | 10.6 ** | 51.5 | 22.1 ** | 2.9 | 0.5 ** | 18.8 | 7.7 ** | 21.9 | 5.4 ** | 5.7 | 1.2 ** | 15.4 | 3.4 ** | 9.4 | 0.3 | 0.7 | −0.1 ** | 20.0 | −13.6 ** | 30.6 | −8.5 ** |
| Blyan | 37.6 | −3.5 | 22.3 | −7.1 * | 20 | −0.4 ** | 8.0 | −3.1 ** | 11.4 | −5.2 ** | 3.9 | −0.7 * | 7.5 | −4.6 ** | 10.7 | 1.6 ** | 1.0 | 0.2 ** | 32.3 | −1.3 | 33.4 | −5.7 ** |
| A8E0139 | 47.6 | 6.4 * | 33.9 | 4.5 | 2.2 | −0.2 | 12.3 | 1.3 | 18.5 | 1.9 | 4.6 | 0.1 | 13.5 | 1.4 | 11.1 | 2.0 ** | 1.0 | 0.1 ** | 31.3 | −2.3 * | 40.2 | 1.0 |
| A9E0460 | 62.9 | 21.7 ** | 19.6 | −9.8 ** | 2.3 | −0.1 | 10.0 | −1.0 | 10.6 | −5.9 ** | 5.9 | 1.4 ** | 7.6 | −4.5 ** | 10.2 | 1.1 ** | 1.0 | −0.1 ** | 24.1 | −9.5 ** | 37.9 | −1.2 |
| A9E1009 | 35.3 | −5.8 | 11.4 | −18.0 ** | 2.2 | −0.2 | 6.0 | −5.1 ** | 5.8 | 10.8 ** | 4.7 | 0.2 | 4.1 | −7.9 ** | 10.8 | 1.7 ** | 0.8 | −0.0 | 20.2 | −13.4 ** | 36.1 | −3.1 |
| COE0309 | 42.1 | 0.9 | 26.6 | −2.8 | 2.1 | −0.3 ** | 6.3 | −4.7 ** | 8.8 | −7.7 ** | 5.7 | 1.2 ** | 6.6 | −5.4 ** | 11.8 | 2.7 ** | 0.9 | 0.1 ** | 36.6 | 3.0 ** | 25.1 | −14.0 ** |
| A9E0150 | 55.3 | 14.2 ** | 23.0 | −6.4 | 2.3 | −0.1 | 9.8 | −1.2 | 10.9 | −5.7 ** | 4.9 | 0.4 | 7.5 | −4.5 ** | 10.2 | 1.0 ** | 1.1 | 0.2 ** | 26.9 | −6.7 ** | 33.9 | −5.2 * |
| Min | 35.3 | 20.9 | 2.0 | 5.9 | 5.8 | 3.6 | 4.1 | 9.1 | 0.7 | 20.0 | 25.1 | |||||||||||
| Max | 67.2 | 51.5 | 2.9 | 18.9 | 21.3 | 5.7 | 15.4 | 11.8 | 1.1 | 36.8 | 47.2 | |||||||||||
| Mean | 48.3 | 29.8 | 2.4 | 11.3 | 14.4 | 4.9 | 11.3 | 10.3 | 0.9 | 28.3 | 35.6 | |||||||||||
| R | 31.9 | 30.6 | 0.9 | 9.0 | 15.5 | 2.1 | 11.3 | 2.7 | 0.4 | 16.8 | 22.1 | |||||||||||
| CV% | 13.7 | 18.6 | 7.8 | 25.9 | 24.9 | 9.0 | 26.9 | 7.2 | 10.4 | 14.8 | 15.2 | |||||||||||
| DE | DFl | DurFl | DMat | PH | LMl | WMl | WPl | NBr | WPlWP | FPH | NPPl | WPPl | PL | PW | PT | NSP | SL | SW | ST | WSPl | W100S | Yplot | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| DE | 1 | −0.47 | 0.51 | −0.5 | −0.4 | 0.49 | −0.3 | −0.536 * | −0.2 | −0.344 | −0.603 * | −0.577 * | −0.502 | 0.843 ** | −0.218 | 0.163 | 0.096 | 0.503 | −0.684 ** | 0.023 | −0.487 | 0.345 | −0.35 |
| DFl | 1 | −0.3 | 0.63 * | 0.41 | 0.29 | 0.06 | 0.357 | −0.16 | 0.491 | 0.051 | 0.278 | 0.072 | −0.571 * | 0.096 | 0.134 | −0.105 | −0.439 | 0.136 | 0 | 0.041 | −0.773 ** | −0.13 | |
| DurFl | 1 | 0.08 | −0.1 | 0.34 | −0.5 | −0.342 | −0.48 | 0.135 | −0.347 | −0.757 ** | −0.712 ** | 0.678 ** | 0.308 | 0.653 * | −0.546 * | 0.613 * | −0.788 ** | 0.01 | −0.715 ** | 0.216 | −0.715 ** | ||
| Dmat | 1 | 0.31 | 0.31 | −0.1 | 0.206 | −0.24 | 0.392 | 0.093 | 0.067 | −0.103 | −0.379 | 0.102 | 0.101 | −0.392 | −0.25 | −0.225 | −0.19 | −0.124 | −0.676 ** | −0.37 | |||
| PH | 1 | −0.3 | −0.2 | 0.602 * | 0.257 | 0.622 * | −0.239 | 0.317 | 0.375 | −0.271 | 0.558 * | 0.244 | 0.243 | −0.474 | 0.149 | −0.04 | 0.325 | −0.307 | −0.37 | ||||
| LMl | 1 | 0.03 | −0.208 | −0.19 | 0.096 | −0.132 | −0.328 | −0.447 | 0.229 | −0.304 | 0.255 | −0.252 | 0.292 | −0.686 ** | 0.095 | −0.48 | −0.275 | −0.37 | |||||
| WMl | 1 | 0.348 | 0.484 | 0.119 | 0.457 | 0.508 | 0.456 | −0.549 * | −0.378 | −0.540 * | 0.307 | −0.267 | 0.374 | 0.496 | 0.474 | 0.162 | 0.543 * | ||||||
| WPl | 1 | 0.623 * | 0.831 ** | 0.181 | 0.826 ** | 0.813 ** | −0.632 * | 0.222 | −0.152 | 0.443 | −0.783 ** | 0.346 | 0.118 | 0.787 ** | −0.155 | 0.198 | |||||||
| NBr | 1 | 0.288 | 0.337 | 0.682 ** | 0.762 ** | −0.322 | −0.277 | −0.466 | 0.701 ** | −0.489 | 0.294 | 0.168 | 0.755 ** | 0.159 | 0.417 | ||||||||
| WPlWP | 1 | −0.004 | 0.407 | 0.355 | −0.381 | 0.363 | 0.263 | 0.088 | −0.49 | −0.082 | 0.151 | 0.32 | −0.277 | −0.27 | |||||||||
| FPH | 1 | 0.343 | 0.277 | −0.607 * | −0.231 | −0.19 | −0.174 | −0.045 | 0.486 | 0.229 | 0.28 | 0.024 | 0.621 * | ||||||||||
| NPPl | 1 | 0.959 ** | −0.771 ** | −0.112 | −0.566 * | 0.627 * | −0.879 ** | 0.643 * | 0.035 | 0.962 ** | −0.152 | 0.617 * | |||||||||||
| WPPl | 1 | −0.643 * | −0.015 | −0.525 | 0.688 ** | −0.796 ** | 0.652 * | 0.048 | 0.993 ** | 0.035 | 0.607 * | ||||||||||||
| PL | 1 | −0.02 | 0.281 | −0.128 | 0.621 * | −0.697 ** | −0.22 | −0.633 * | 0.322 | −0.5 | |||||||||||||
| PW | 1 | 0.701 ** | −0.221 | 0.018 | 0.134 | 0.288 | −0.066 | 0.268 | −0.35 | ||||||||||||||
| PT | 1 | −0.558 * | 0.506 | −0.352 | 0.27 | −0.587 * | 0.104 | −0.614 * | |||||||||||||||
| NSP | 1 | −0.656 * | 0.298 | 0.135 | 0.717 ** | 0.162 | 0.371 | ||||||||||||||||
| SL | 1 | −0.457 | 0.093 | −0.814 ** | 0.338 | −0.33 | |||||||||||||||||
| SW | 1 | 0.182 | 0.658 * | 0.121 | 0.758 ** | ||||||||||||||||||
| ST | 1 | 0.044 | 0.551 * | 0.131 | |||||||||||||||||||
| WSPl | 1 | 0.05 | 0.640 * | ||||||||||||||||||||
| W100S | 1 | 0.243 | |||||||||||||||||||||
| Yplot | 1 |
| Source of Variation | SS | df | MS | F | F Crit | ŋ.% | Sign. |
|---|---|---|---|---|---|---|---|
| Year | 225.3 | 2 | 112.6 | 12.2 | 7.5 | 11.4 | *** |
| Genotype | 854.4 | 13 | 65.7 | 7.1 | 3.1 | 43.4 | *** |
| Year * Genotype | 113.3 | 26 | 4.4 | 0.5 | 2.5 | 5.8 | n.s. |
| Error | 776.5 | 84 | 9.2 | 39.4 | |||
| Total | 1969.5 | 125 | 100.0 |
| Source of Variation | SS | df | MS | F | F Crit | ŋ.% | Sign. |
|---|---|---|---|---|---|---|---|
| Year | 5,893,823.8 | 2 | 2,946,911.9 | 95.4 | 7.5 | 32.6 | *** |
| Genotype | 5,910,807.0 | 13 | 454,677.5 | 14.7 | 3.1 | 32.7 | *** |
| Year * Genotype | 3,696,186.5 | 26 | 142,161.0 | 4.6 | 2.5 | 20.4 | *** |
| Error | 2,595,728.5 | 84 | 30,901.5 | 14.3 | |||
| Total | 18,096,545.8 | 125 | 100.0 |
| Coefficients | Genetically Narrow | |
|---|---|---|
| 6.168 | A9E0543 | A8E0139 |
| 15.238 | A9E0430 | A9E0588 |
| 16.082 | A9E0430 | A9E1100 |
| Coefficients | Genetically Distance | |
|---|---|---|
| 606.0 | A9E0460 | BOE0048 |
| 580.7 | A9E0460 | A9E0543 |
| 557.8 | A9E0460 | A9E0430 |
| Component | Total | % of Variance | Cumulative % |
|---|---|---|---|
| 1 | 9.3 | 42.1 | 42.1 |
| 2 | 4.4 | 20.1 | 62.2 |
| 3 | 2.6 | 11.7 | 73.9 |
| Component | ||||
|---|---|---|---|---|
| Traits | var No | PC1 | PC2 | PC3 |
| DE | var1 | −0.668 | −0.387 | 0.367 |
| DFl | var2 | 0.228 | 0.777 | −0.373 |
| DurFl | var3 | −0.822 | 0.164 | 0.209 |
| DMat | var4 | 0.012 | 0.750 | −0.450 |
| PH | var5 | 0.296 | 0.691 | 0.473 |
| LMl | var6 | −0.424 | 0.220 | −0.270 |
| WMl | var7 | 0.607 | −0.246 | −0.226 |
| WPl | var8 | 0.760 | 0.461 | 0.321 |
| NBr | var9 | 0.748 | −0.158 | 0.323 |
| WPlWP | var10 | 0.312 | 0.745 | 0.300 |
| FPH | var11 | 0.464 | −0.171 | −0.579 |
| NPPl | var12 | 0.970 | 0.100 | 0.073 |
| WPPl | var13 | 0.944 | −0.016 | 0.269 |
| PL | var14 | −0.808 | −0.332 | 0.365 |
| PW | var15 | −0.123 | 0.531 | 0.310 |
| PT | var16 | −0.592 | 0.495 | 0.278 |
| NSP | var17 | 0.618 | −0.244 | 0.542 |
| SL | var18 | −0.804 | −0.302 | −0.091 |
| SW | var19 | 0.857 | −0.429 | −0.198 |
| WSPl | var20 | 0.951 | −0.064 | 0.251 |
| W100S | var21 | −0.104 | −0.625 | 0.470 |
| Yplot | var22 | 0.689 | −0.550 | −0.254 |
| Accession | Mois- ture % | Proteins % | Oils % | Ash % | Fibres % | Tannins % | Carbohyd- Rates % | kcal/ 100 g |
|---|---|---|---|---|---|---|---|---|
| A9E0312 | 11.0 ± 0.8 a | 25.6 ± 0.2 f | 2.2 ± 0.03 e | 5.1 ± 0.03 e | 5.4 ± 0.1 fg | 16.0 ± 0.1 b | 56.3 ± 0.7 bc | 347 ± 4 c |
| Ustrem | 12.1 ± 0.7 a | 23.4 ± 0.4 d | 2.4 ± 0.07 f | 4.8 ± 0.1 cd | 5.3 ± 0.1 d–f | 17.4 ± 0.1 cd | 57.4 ± 1.3 c–e | 345 ± 3 a–c |
| A9E0430 | 11.9 ± 0.7 a | 22.7 ± 0.1 bc | 2.5 ± 0.07 f | 4.8 ± 0.1 d | 3.3 ± 0.2 a | 19.1 ± 0.1 fg | 58.2 ± 0.5 d–g | 346 ± 3 bc |
| A9E0543 | 11.9 ± 0.7 a | 23.5 ± 0.2 d | 2.1 ± 0.1 de | 4.6 ± 0.1 ab | 4.8 ± 0.3 c–e | 18.0 ± 0.6 de | 58.1 ± 0.5 c–f | 344 ± 2 a–c |
| B1E0485 | 12.2 ± 0.5 a | 23.4 ± 0.3 d | 1.9 ± 0.1 bc | 5.1 ± 0.03 e | 5.7 ± 0.3 fg | 16.5 ± 0.5 bc | 57.6 ± 0.2 c–e | 340 ± 1 ab |
| A9E1100 | 11.9 ± 0.7 a | 24.2 ± 0.1 e | 2.2 ± 0.1 e | 4.6 ± 0.1 a | 4.8 ± 0.1 c | 21.5 ± 0.3 i | 57.2 ± 0.6 c–e | 345 ± 3 a–c |
| A9E0588 | 12.2 ± 0.7 a | 25.4 ± 0.2 f | 2.5 ± 0.1 f | 4.7 ± 0.01 a–c | 4.7 ± 0.1 cd | 17.1 ± 0.1 cd | 55.3 ± 0.4 ab | 345 ± 3 a–c |
| BOE0048 | 11.6 ± 0.6 a | 24.5 ± 0.2 e | 2.2 ± 0.1 e | 5.1 ± 0.03 e | 5.6 ± 0.3 fg | 20.2 ± 0.1 h | 56.7 ± 0.7 b–d | 344 ± 3 a–c |
| Blyan | 11.8 ± 0.5 a | 22.2 ± 0.2 ab | 1.7 ± 0.07 ab | 4.6 ± 0.03 a | 5.7 ± 0.3 fg | 13.6 ± 0.6 a | 59.9 ± 0.7 g | 343 ± 3 a–c |
| A8E0139 | 12.2 ± 0.5 a | 21.8 ± 0.2 a | 1.7 ± 0.07 ab | 4.7 ± 0.0 a–c | 5.3 ± 0.2 ef | 18.4 ± 0.2 ef | 59.7 ± 0.6 fg | 341 ± 2 a–c |
| A9E0460 | 11.7 ± 0.6 a | 27.0 ± 0.2 g | 1.9 ± 0.07 cd | 5.3 ± 0.1 f | 3.9 ± 0.1 b | 21.3 ± 0.2 i | 54.2 ± 0.7 a | 342 ± 3 a–c |
| A9E1009 | 11.9 ± 0.8 a | 23.0 ± 0.1 cd | 1.6 ± 0.03 a | 4.7 ± 0.03 a–c | 5.9 ± 0.1 g | 19.8 ± 0.1 gh | 58.9 ± 0.9 e–g | 341 ± 3 a–c |
| A9E0150 | 11.6 ± 1.0 a | 26.6 ± 0.2 g | 1.7 ± 0.03 a | 5.4 ± 0.1 f | 3.3 ± 0.3 a | 16.6 ± 0.4 bc | 54.4 ± 0.9 a | 339 ± 3 a |
| COE0309 | 11.7 ± 0.7 a | 22.2 ± 0.1 ab | 1.9 ± 0.03 cd | 4.7 ± 0.03 a–c | 3.6 ± 0.3 ab | 14.0 ± 0.2 a | 59.6 ± 0.6 fg | 345 ± 3 a–c |
| Accession | Sol.Sugars mg·g−1FW | Starch mg·g−1FW | Phenols mg·g−1FW | Amino Acids mg·g−1FW | TIA U·mg−1FW |
|---|---|---|---|---|---|
| A9E0312 | 49.9 ± 1.9 a–c | 580.5 ± 25.1 a | 1.3 ± 0.10 a | 3.6 ± 0.27 a–c | 1.2 a |
| Ustrem | 63.2 ± 10.7 a–f | 633.2 ± 70.1 ab | 1.6 ± 0.03 a | 3.3 ± 0.15 a–c | 2.4 bc |
| A9E0430 | 62.9 ± 6.9 b–f | 678.2 ± 30.50 a–d | 1.4 ± 0.03 a | 3.8 ± 0.62 a–c | 1.9 b |
| A9E0543 | 64.4 ± 6.2 b–f | 692.8 ± 58.30 a–d | 1.4 ± 0.01 a | 3.9 ± 0.45 bc | 2.3 bc |
| B1E0485 | 51.2 ± 5.1 a–d | 691.6 ± 48.0 a–d | 1.6 ± 0.14 a | 2.9 ± 0.34 ab | 1.5 a |
| A9E1100 | 76.4 ± 8.6 fg | 696.9 ± 45.50 a–d | 1.6 ± 0.09 a | 3.9 ± 0.68 bc | 2.4 bc |
| A9E0588 | 64.1 ± 5.4 b–f | 652.9 ± 39.3 a–c | 1.6 ± 0.05 a | 4.5 ± 0.14 c | 3.1 c |
| BOE0048 | 46.9 ± 5.4 ab | 686.2 ± 12.30 a–d | 1.6 ± 0.01 a | 3.3 ± 0.42 a–c | 1.7 ab |
| Blyan | 43.7 ± 5.7 a | 687.9 ± 41.10 a–d | 1.3 ± 0.15 a | 2.6 ± 0.25 a | 1.7 ab |
| A8E0139 | 56.8 ± 5.9 a–e | 770.8 ± 40.0 cd | 17.2 ± 0.1 d | 3.3 ± 0.48 a–c | 1.4 a |
| A9E0460 | 73.0 ± 0.6 e–g | 748.3 ± 16.5 bcd | 4.4 ± 0.35 b | 8.6 ± 2.13 d | 2.4 bc |
| A9E1009 | 72.2 ± 10.2 d–g | 681.3 ± 30.00 a–d | 6.1 ± 0.60 c | 7.4 ± 0.17 d | 1.4 a |
| A9E0150 | 89.1 ± 11.7 g | 795.7 ± 38.9 d | 2.2 ± 0.25 a | 7.4 ± 0.49 d | 2.1 b |
| COE0309 | 69.9 ± 1.5 c–g | 655.1 ± 34.30 a–d | 2.2 ± 0.11 a | 3.6 ± 0.05 a–c | 3.0 c |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stoilova, T.; Petrova, S.; Simova-Stoilova, L. Assessment of Morphological Diversity, Yield Components, and Seed Biochemical Composition in Common Bean (Phaseolus vulgaris L.) Landraces. Agriculture 2025, 15, 1856. https://doi.org/10.3390/agriculture15171856
Stoilova T, Petrova S, Simova-Stoilova L. Assessment of Morphological Diversity, Yield Components, and Seed Biochemical Composition in Common Bean (Phaseolus vulgaris L.) Landraces. Agriculture. 2025; 15(17):1856. https://doi.org/10.3390/agriculture15171856
Chicago/Turabian StyleStoilova, Tsvetelina, Sofiya Petrova, and Lyudmila Simova-Stoilova. 2025. "Assessment of Morphological Diversity, Yield Components, and Seed Biochemical Composition in Common Bean (Phaseolus vulgaris L.) Landraces" Agriculture 15, no. 17: 1856. https://doi.org/10.3390/agriculture15171856
APA StyleStoilova, T., Petrova, S., & Simova-Stoilova, L. (2025). Assessment of Morphological Diversity, Yield Components, and Seed Biochemical Composition in Common Bean (Phaseolus vulgaris L.) Landraces. Agriculture, 15(17), 1856. https://doi.org/10.3390/agriculture15171856






