1. Introduction
Antimicrobial resistance (AMR) is a well-documented risk to both animal and public health. Therefore, research into natural compounds with anti-inflammatory and antimicrobial properties has expanded steadily in recent years, reflecting their potential as supportive therapies for infectious and chronic diseases in veterinary medicine. Between 2011 and 2022, veterinary antimicrobial sales in Europe, expressed in mg per population correction unit (PCU), where 1 PCU corresponds to 1 kg of animal biomass, fell by 53% (from 161.2 to 75.8 mg/PCU)—an average decline of roughly 6–7% per year. The year 2022 alone brought an additional 12.7% reduction relative to 2021 [
1]. Data from the first ESUAvet report [
2] show that the EU has already reached half of the 50% reduction targeted for 2030 (88.5 mg/PCU versus the goal of 59.2 mg/PCU).
The regulatory changes related to AMR have also increased the research into non-antibiotic compounds that can be safely and effectively used to treat or prevent udder diseases, especially mastitis, a common and economically significant condition in dairy cattle. Mastitis is a severe inflammatory process affecting the mammary gland and tissues of the udder in dairy cattle, typically caused by bacterial infection, and it is characterized by an elevated somatic cell count (SCC), altered milk composition, and compromised animal welfare [
3,
4]. It is the most commonly recorded disease in dairy cows worldwide and has a negative impact on dairy farm profitability. In 2022, approximately 4458 tons (or 4,458,000 kg) of active antibiotic substances were sold for use in farmed animals and aquaculture in the European Economic Area countries, plus Switzerland and the UK. The total antimicrobial consumption for food-producing animals in 2022 was measured at 73.9 mg/PCU [
5]. Current treatment protocols still rely heavily on antibiotics, contributing to the development of AMR and necessitating milk withdrawal periods that reduce productivity and profitability.
Large-scale epidemiological data indicate that Gram-positive cocci remain the dominant cause of bovine mastitis. Across Europe, and in comparable datasets elsewhere,
Staphylococcus and
Streptococcus spp. dominate bovine mastitis, accounting for 77% of positive isolates in a 10-year Bavarian survey and 78% of clinical cases in a 97-herd UK-based study, while more recent large-scale surveys place their combined prevalence firmly in the ≈75–85% range [
6,
7,
8,
9,
10].
Therefore, any non-antibiotic intramammary strategy must primarily address infections caused by
Staphylococcus and
Streptococcus species. Among the alternative candidates for mastitis management are pectic polysaccharides, derived from plant cell walls [
11]. Intramammary use of chitosan gels, β-glucan nanoparticles and herbal extracts has shown mixed efficacy, but data on pectin are absent [
12,
13,
14,
15,
16]. Pectins have demonstrated both anti-inflammatory and antimicrobial properties, including disruption of bacterial membranes and anti-adhesive effects, particularly under acidic conditions, where protonation increases the electrostatic interactions with negatively charged bacterial surfaces [
17,
18,
19]. Additionally, pectins have been shown to modulate host immune responses through their interaction with Toll-like receptors (TLRs) and the regulation of cytokine production [
20]. Various forms of pectin exert direct and/or indirect immunomodulatory effects through multiple mechanisms [
21,
22].
Intervention studies in humans have shown that pectin supplementation can reduce the circulating levels of pro-inflammatory cytokines while simultaneously increasing the anti-inflammatory cytokine IL-10 [
23]. This supports the systemic anti-inflammatory potential of pectins, despite their primary site of action being the gastrointestinal tract. Importantly, pectins are not directly absorbed; due to their high molecular weight and complex structure, they do not cross the intestinal barrier into the systemic circulation [
24]. As such, their effects are exerted locally in the gut, rather than systemically like conventional anti-inflammatory drugs. Although there are no studies on intramammary administration of pectins, their known anti-inflammatory properties suggest their possible use in the treatment of mastitis.
While the systemic benefits of dietary pectins are well documented, considerably less is known about their potential for local application in the mammary gland. This study aimed to fill this gap by assessing the local tolerance and dose-dependent effects of pectin solutions administered intramammary to dairy cows with mastitis.
The bovine mammary epithelium expresses functional TLR-2 and TLR-4 receptors that sense staphylococcal and streptococcal ligands and trigger an NF-κB-dependent cytokine cascade [
25,
26]. Citrus and apple pectins have been shown to bind and sterically block the TLR2/1 heterodimer on intestinal epithelial cells, thereby attenuating downstream pro-inflammatory signaling [
27]. Local delivery of low methoxyl pectin (LM-pectin) into the gland therefore offers a plausible route to dampen TLR-mediated inflammation at its epithelial origin. In addition, milk provides the physicochemical milieu required for LM-pectin’s functionality. Normal bovine milk maintains a pH of 6.4–6.8, rising to ≈6.8–6.9 during mastitis, and contains free Ca
2+ [
28]. Pectin strands can rapidly cross-link with Ca
2+ within this pH window to form a porous hydrogel (“egg-box” junction zones) that (i) prolongs the residence time inside the teat cistern, (ii) acts as a physical barrier to bacterial colonization, and (iii) slowly releases protonated carboxyl groups able to electrostatically destabilize Gram-positive membranes [
29,
30,
31].
Despite its generally recognized as safe (GRAS) status and extensive use as a food hydrocolloid, pectin has seldom been explored for use in intramammary therapy. Two formulation hurdles help explain this gap. First, even modest concentrations (>1%
w/
v) of pectin display specific viscosities at 25 °C [
32], which complicates passage through teat cannulas. Second, the high Ca
2+ level in milk induces in situ gelation within seconds, risking cannula blockage and uneven distribution [
29]. Earlier work, therefore, focused on polymers with lower viscosity or thermogelling behavior, such as chitosan hydrogels that remain fluid at room temperature and set only at 37 °C [
13].
The present study deliberately addresses these formulation challenges by (i) selecting LM-pectin grades with reduced intrinsic viscosity, (ii) limiting the concentration to ≤10% w/v, and (iii) administering immediately after re-suspension to minimize premature cross-linking. This fills a clear knowledge gap, because no peer-reviewed intramammary trials with pectin have been reported to date.
Taken together, these features support the working hypothesis that intramammary LM-pectin can simultaneously regulate host TLR signaling and exert direct antibiofilm/antimicrobial pressure on Staphylococcus and Streptococcus spp.
Considering that pectins are not classical pharmaceuticals, although they are increasingly recognized as modulators of inflammation through TLR inhibition and subsequent downregulation of pro-inflammatory cytokines [
23], the hypothesis of this study is that intramammary administration of apple pectin solutions at a concentration of 10% is well tolerated by the mammary tissue and exerts a dose-dependent anti-inflammatory effect—leading to a reduction in the somatic cell counts and clinical signs of mastitis in dairy cows. This hypothesis was tested under real-world conditions using naturally occurring clinical mastitis cases.
This study aimed to evaluate the intramammary use of apple-derived pectin in dairy cows with clinical mastitis, focusing on preliminary evaluation of effects like local tolerability, dose-dependent effects, and impact on udder health indicators. Therefore, it was decided to compare directly with the reference therapy, i.e., an antibiotic.
2. Materials and Methods
2.1. Animals
This study was conducted on a group of Holstein–Friesian cows, weighing ≈ 650 ± 50 kg, aged 3 to 7 years. The study was conducted at a single farm (Liplas village, Lesser Poland Voivodeship, Poland; 49°57′ N 20°13′ E) between December 2024 and April 2025. The animals were fed a standard diet consisting of protein concentrates, mixed cereals, maize silage, grass silage, straw, and supplements. Unlimited access to water was provided.
2.2. Inclusion and Exclusion Criteria for the Study
The initial selection of cows for the experimental and control groups was based on monthly reports provided by the Polish Federation of Cattle Breeders and Dairy Farmers, which included data on the SCC, age, lactation stage, and individual milk yield. Only multiparous cows within the first 100 days of lactation were considered eligible. Only cows with inflammation limited to one quarter were enrolled in the experiment, and those with moderate mastitis. The main inclusion criterion was an SCC exceeding 400,000 cells/mL in one of the four udder quarters [
33].
Animals were excluded from the study if they showed acute and severe systemic signs of mastitis, signs of chronic disease such as hyperketonemia or hypocalcemia, nutritional deficiencies, or infectious diseases. Cows with advanced clinical symptoms, including skin cyanosis resulting from circulatory disturbances in the udder or pathological discharge from the affected quarter between milkings, were also excluded. Furthermore, udder palpation was performed to detect indurations or nodules (abscesses) within the mammary gland, which are typical of chronic Staphylococcus aureus infections and served as grounds for exclusion.
2.3. Pectin Preparation
Pectin from apple (#93854, Sigma-Aldrich, Merck Group, Darmstadt, Germany) was dissolved in phosphate-buffered saline (PBS; Sigma-Aldrich) to the desired concentration. The solution was prepared using a SONIC-2 ultrasonic bath (POLSONIC, Warsaw, Poland) and agitated for 30 min at 37 °C. The resulting pectin solution was transferred into custom-prepared 5 mL dermal tubing syringes. The final intramammary (IMM) dose was administered as 5 mL of the prepared pectin solution in PBS.
2.4. Pectin Dose Selection
In the preliminary experiment, pectin was prepared at concentrations of 2%, 4%, 10%, and 20%. However, the 20% concentration produced a suspension that was too viscous for IMM administration. Consequently, IMM administration of the 2%, 4%, and 10% pectin solutions was scheduled, with each formulation to be administered seven times, at 24 h intervals, to 10 cows per group diagnosed with clinical mastitis. A veterinarian conducted local tolerance assessments and clinical evaluations at 12 h intervals, with the mastitis scores determined at morning milking. It was anticipated that if the clinical signs worsened, the pectin treatment would be discontinued and standard systemic and intramammary antibiotic therapy, along with supportive care, would be initiated. However, following several days of administering 2% and 4% pectin, most animals exhibited worsening mastitis symptoms, necessitating the introduction of antibiotic treatment and the termination of further pectin administration in these groups. Therefore, the 2% and 4% concentrations were excluded from the study (
Supplementary Figure S1). Only the 10% pectin group, in which no animals required antibiotic treatment throughout the preliminary experiment, was included in the main experiment as the experimental pectin group.
2.5. Study Design
In the main study, 48 cows with clinical mastitis that met the predefined inclusion and exclusion criteria were enrolled. Prior to group allocation, all the animals underwent a three-day pre-treatment observation period (time points: −72 h, −48 h, and −24 h), during which milk samples were collected and analyzed for the SCC. The mean SCC across all the cows and all three time points was used to establish a global baseline for the study.
Following this period, cows were randomly assigned to one of two treatment groups (n = 24 per group). Post-randomization analysis confirmed that the SCC values measured at the −24 h time point did not differ significantly between the groups, ensuring baseline equivalence prior to treatment initiation.
The experimental group (the Pectin10 group, n = 24) received IMM administration of 10% pectin, applied seven times at 24 h intervals (time points: 0 h, 24 h, 48 h, 72 h, 96 h, 120 h, and 144 h). The animals were not given any other drugs.
The Control group (n = 24) was treated with the licensed multi-component formulation Mastijet Fort (Intervet International B.V., Boxmeer, the Netherlands), which is indicated for infectious and inflammatory conditions typical of acute bovine mastitis. One dose (8 g, containing tetracycline hydrochloride, 200 mg; neomycin sulphate, 250 mg; bacitracin, 2000 IU; and prednisolone, 10 mg) was infused IMM at time point 0 h, followed by a second dose 12 h later (total of two injections). In the Control group, two intramammary doses of a multi-component antibiotic (Mastijet Fort) were administered without prior pathogen-specific sensitivity testing, reflecting common field practice in which treatment is initiated immediately after diagnosis and culture results are used periodically to adjust herd-level therapy.
Both the pectin solution and the multi-component drug were administered into the affected quarter immediately after the morning milking. Before administration, the udder skin and teat orifice were disinfected with Oxy-Foam D (EcoLab Inc., Saint Paul, MN, USA) and dried with a disposable paper towel.
2.6. Physical Clinical Examinations
Throughout the entire duration of the experiment, the condition of the udder and the general health status of all the experimental animals were monitored by the same experienced veterinarian, who performed regular local tolerance assessments related to pectin or drug administration, as well as clinical examinations of each animal at 24 h intervals [
34].
The mastitis scores were recorded for the treated udder quarter during routine health monitoring conducted at morning milking, using a standardized 4-point scale. Score 0 indicated normal milk and udder appearance with no abnormalities; Score 1 reflected the presence of a small number of clots in the milk without visible swelling, redness, heat, or pain upon palpation; Score 2 indicated numerous clots, visible separation of milk and whey, and mild swelling, redness, heat, and pain; and Score 3 was assigned in cases of systemic illness (e.g., elevated body temperature), watery milk, and pronounced swelling, redness, heat, pain, and/or hardness of the affected quarter [
35,
36].
The mastitis scoring began three days before the initiation of treatment (at time points −72 h, −48 h, and −24 h), was conducted daily throughout the treatment period (time points 0 h to 144 h), and concluded one day after the final pectin dose (time point 168 h).
2.7. Milk and Blood Sample Collection
Milk samples were collected from the affected udder quarters after mastitis scoring was performed. A total of 15 milk samples per cow were collected throughout the study, starting three days prior to treatment initiation (time point −72 h) and ending 24 h after the final pectin administration (time point 168 h). All the milk samples were collected according to a standardized protocol, prior to pectin or multi-component drug administration. Before sampling, the udder quarters were thoroughly washed and dried, and the teat orifices were disinfected with 70% ethanol. The first 3–4 streams of milk were discarded to avoid contamination.
Milk samples for bacteriological culture, isolation, and identification of the causative bacterial agent were aseptically collected at time points −72 h and 168 h into sterile, labeled test tubes without preservatives. Milk samples for the somatic cell count (SCC) determination were collected in a volume of 30 mL into 50 mL Falcon tubes containing Broad Spectrum Microtabs II preservative tablets (Bentley Instruments, Chaska, MN, USA), consisting of bronopol (8 mg) and natamycin (0.3 mg), to prevent spoilage and stabilize the sample during transport. After collection, all the milk samples were stored on ice in a cooler box and transported under chilled conditions to the laboratory for further analysis.
The milk yield was continuously recorded, and data from the month preceding the mastitis diagnosis (before treatment) and the month following the end of treatment were used for analysis.
Blood samples were collected twice: immediately before the start of treatment (time point 0 h) and 24 h after the final pectin administration (time point 168 h). Blood was drawn from the tail vein into EDTA K2 tubes for basic hematological analysis and into clot activator tubes for biochemical analysis.
2.8. Analyses
2.8.1. Milk Analyses
The somatic cell count (SCC) levels in the milk were determined using a semi-automated BactoCount IBCm analyzer (Bentley Instruments, Chaska, MN, USA).
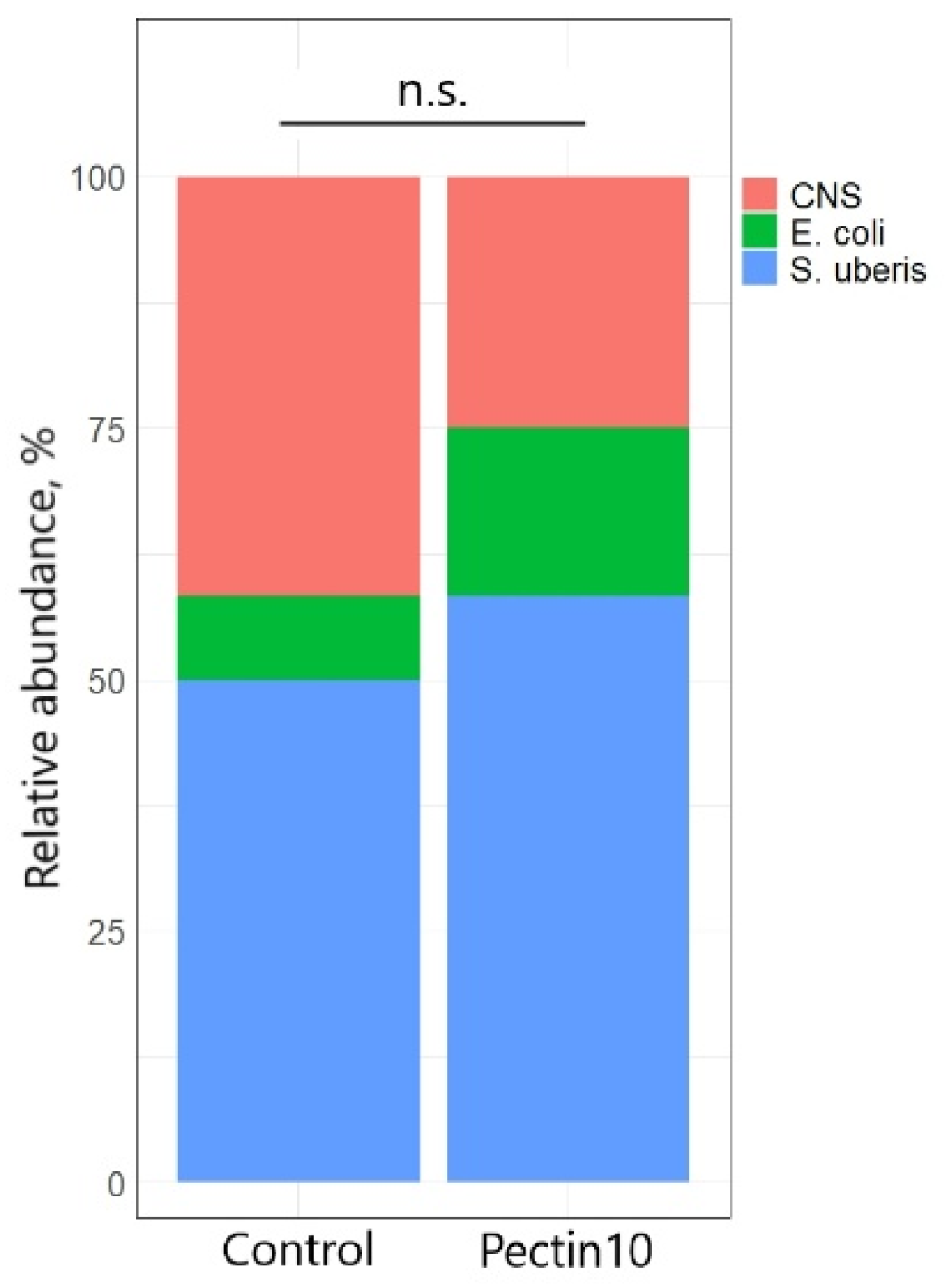
Analyses were performed in the microbiological laboratory, which routinely conducts diagnostic testing for mastitis-causing pathogens in samples from this dairy farm. Briefly, milk samples were cultured using standard bacteriological and mycological procedures. For bacterial isolation, samples were streaked on Columbia blood agar plates supplemented with 5% sheep blood and incubated aerobically at 36 °C for 24–48 h. For fungal detection, samples were inoculated onto Sabouraud dextrose agar containing chloramphenicol to suppress bacterial growth and incubated at 36 °C for up to 72 h. The colony morphology was assessed after 24, 48, and 72 h of incubation. Representative colonies were subjected to Gram staining and examined microscopically. Gram-positive cocci arranged in chains were classified as Streptococcus spp., while Gram-positive cocci in clusters were classified as Staphylococcus spp. Coagulase and catalase tests were performed to differentiate Staphylococcus aureus from coagulase-negative staphylococci (CNS). Gram-negative rods were classified as coliforms, predominantly Escherichia coli. When the morphological and basic biochemical characteristics were inconclusive, isolates were further identified using commercial biochemical identification systems (API 20E, API Staph, API Strep; bioMérieux, Marcy-l’Étoile, France), which assess the bacterial enzymatic activities and carbohydrate utilization patterns to enable species-level identification. No fungal colonies were detected on Sabouraud agar during the incubation period; therefore, no further identification of fungal isolates was conducted.
The cytokine profile analysis was intentionally limited to tumor necrosis factor-alpha (TNF-α), the most reliable marker of the initial phase of the inflammatory response. The TNF-α concentrations were measured using a commercially available, bovine-specific ELISA kit (LS-F5014, LSBio, Seattle, WA, USA), following the manufacturer’s instructions. The absorbance was read using a Benchmark Plus microplate reader (Bio-Rad Laboratories, Inc., Hercules, CA, USA).
All the measurements were performed in technical duplicates.
2.8.2. Blood Analyses
Hematological analyses were conducted using the Abacus Junior Vet automated hematology analyzer (Diatron, Budapest, Hungary). The following parameters were assessed: white blood cell count, red blood cell count, hemoglobin, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, red cell distribution width, and platelet count.
After serum separation, biochemical parameters, including cholesterol, urea, bilirubin, creatinine, calcium, and phosphorus, were measured using an automated biochemistry analyzer (Mindray BS-120, Bio-Medical Electronics, Shenzhen, China) with commercial, ready-to-use test kits (Alfa Diagnostics, Warsaw, Poland). All the measurements were validated using a multiparametric control serum (Alfa Diagnostics).
2.9. Statistical Analysis
All the analyses were performed in R (version 4.5.0). Statistical significance was defined as a p-value < 0.05 in all the analyses.
The required sample size was determined a priori based on an ANCOVA design using the pwr.f2.test function from the pwr package. Assuming a significance level (α) of 0.05 and targeting a moderate effect size (f2 = 0.18, equivalent to Cohen’s f ≈ 0.42), a sample of n = 24 animals per group (total n = 48) was deemed sufficient to achieve a statistical power of 0.80.
The relative abundance of the pathogen species isolated from the milk samples was compared using Fisher’s exact test.
The somatic cell counts were log2-transformed to obtain the somatic cell score (SCS) in order to normalize the data and meet the assumptions of the linear model [
37,
38]. To analyze the effect of the treatment and time on the SCS, a linear mixed-effects model was fitted with fixed effects for the group, the time, and their interaction, and a random intercept for the cow to account for repeated measures using the lme4 package. Post hoc pairwise comparisons between the groups at each time point, as well as between the baseline value and SCS recorded at each subsequent time point for each group, were performed using the estimated marginal means via the emmeans package. To control the false discovery rate associated with multiple testing, the
p-values for all the pairwise comparisons were adjusted using the Benjamini–Hochberg FDR method. The assumptions of the model were assessed by visual inspection of the residual plots, and the model was considered appropriate as the residuals were approximately normally distributed and homoscedastic.
The inflammation score was treated as an ordered factor. To assess the effects of the treatment and time on the inflammation score, a cumulative link mixed-effects model (CLMM) was fitted using the ordinal package, with fixed effects for the treatment, the time, and their interaction, and a random intercept for the cow to account for repeated measures. Time was defined as a categorical factor, with 0 h serving as the reference category. The estimated marginal means (emmeans) for each Treatment × Time combination were obtained and contrasted at each time point versus 0 h. The resulting p-values were adjusted for multiple comparisons using the Benjamini–Hochberg false discovery rate (FDR) method.
The biochemistry, hematology, and milk yield baseline characteristics (pre- and post-treatment) were summarized as the mean ± standard deviation (SD) for each treatment group. To evaluate the effect of treatment while adjusting for the baseline value, a one-way ANCOVA model was fitted with the post-treatment value as the dependent variable, treatment group as the categorical predictor, and baseline pre-treatment value as a continuous covariate. Statistical significance was calculated for the main effect of treatment on the data after adjustment for the pre-treatment values. The model residuals were inspected for normality (QQ-plot and Shapiro–Wilk test) and homogeneity of variance (Levene’s test). The adjusted least-squares means (lsmeans) for the post-treatment values in each group and their pooled standard errors (SEMs) were estimated from the ANCOVA model using the emmeans package.
4. Discussion
The study conducted is one of the few experimental works evaluating the effectiveness of IMM pectin application in dairy cows under production conditions. Previous publications have mostly focused on the role of pectins in gut models, calf nutrition, or in vitro studies, while data on their local use in mastitis treatment are very limited. In this experiment, the effectiveness of pectin was compared for the first time with antibiotic therapy when applied directly into the udder, making this study an important starting point for further clinical and practical investigations.
Mastitis is a complex disease that can present as either a monoinfection or a mixed form, and its development involves the gastrointestinal tract, microbial metabolites, microbiota, and nutritional deficiencies [
39]. The long-standing view of mastitis as solely an exogenous infection is incomplete, as increasing evidence supports the existence of an entero-mammary axis linking the gut and mammary gland health. Standard therapies for mastitis treatment are mainly based on antibiotics administered either intramammarily or systemically [
14]. Although antibiotics are effective against common mastitis pathogens, their overuse carries the risk of microbial resistance and requires strict adherence to milk withdrawal periods. In recent years, there has been growing interest in alternative mastitis therapies that reduce antibiotic use. In this study, conducted under real dairy farm conditions, three different concentrations of pectin were tested. The clinical changes, SCC, blood, and biochemical parameters were also analyzed between the group receiving a 10% pectin solution and the Control group treated with a standard multicomponent preparation. The product used in the Control group contained a combination of several active substances with broad-spectrum activity against typical mastitis pathogens.
4.1. Clinical and Cytological Effectiveness, Mechanisms of Action and Immune Modulation
In light of the problems related to antimicrobial resistance and milk withdrawal periods, this study aimed to evaluate an alternative, non-antibiotic mastitis therapy using pectins. Intramammary pectin treatment proved to be as effective as antibiotic therapy, which was confirmed by the lack of statistically significant differences in many evaluated parameters. This is a noteworthy result, especially considering that the antibiotic preparation used represents a well-established standard in mastitis treatment. Achieving a therapeutic effect with a non-antibiotic agent highlights the potential of pectins as an alternative treatment strategy.
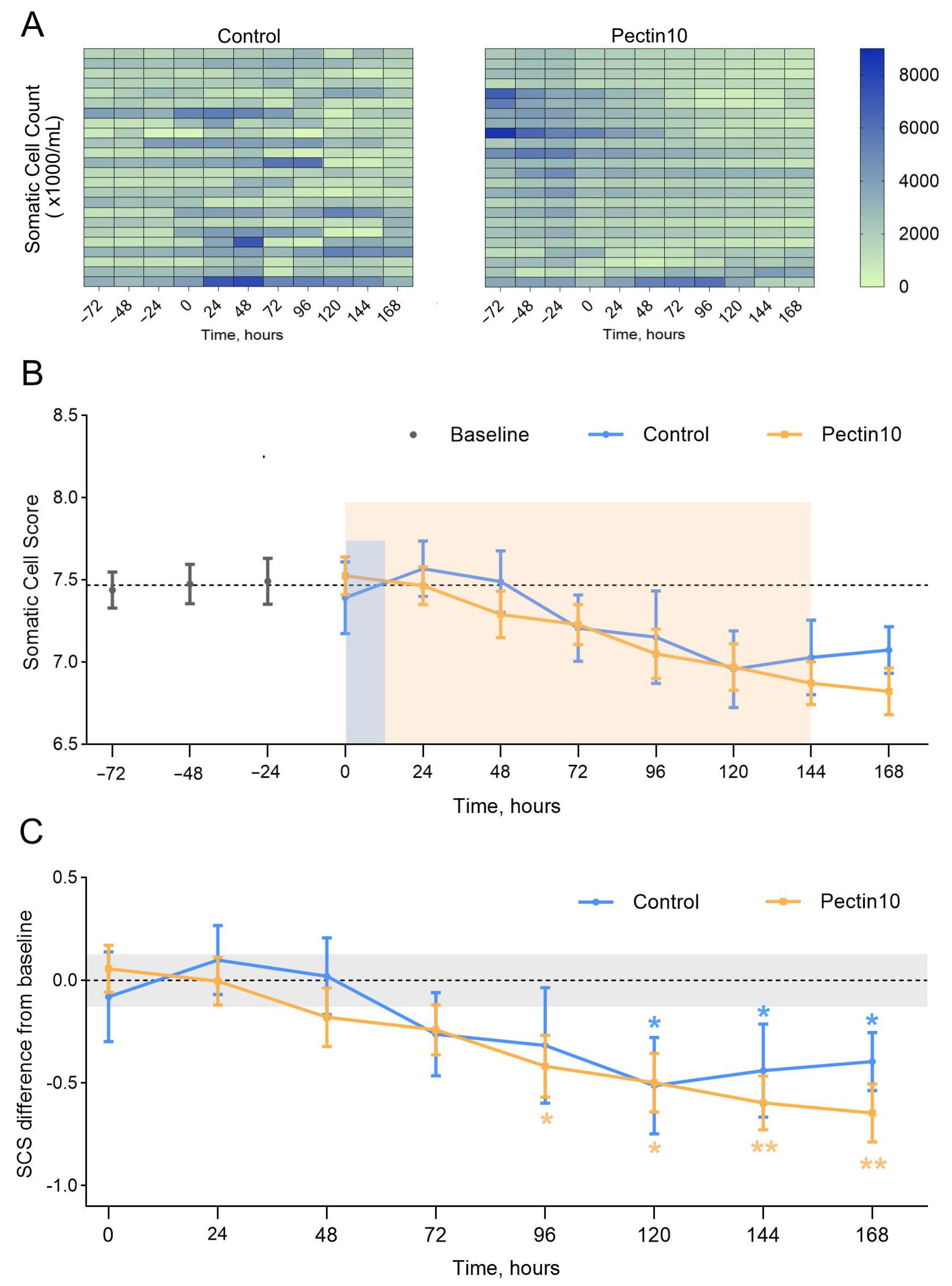
The results suggest that IMM administration of 10% pectin may be comparable to antibiotic therapy; however, this finding requires confirmation in studies with greater statistical power. The faster regression of clinical symptoms observed in the pectin group indicates that pectins may effectively modulate the inflammatory response in the udder. Specifically, the clinical signs resolved, on average, 1.6 days earlier in cows treated with the 10% pectin solution compared to the Control group (p < 0.05).
An objective confirmation of this difference in healing dynamics is the analysis of the changes in the SCC in milk. The SCC reflects the severity of inflammation in the mammary gland, where the higher the SCC, the stronger the inflammatory response and the higher the infiltration of leukocytes into the milk. In our study, a significantly faster decrease in the SCC was observed in the pectin-treated group compared to the Control group. As early as 48 h after the beginning of treatment, the average SCC in cows receiving 10% pectin began to drop more clearly than in cows treated with antibiotics (the difference between groups from day 2 onward was statistically significant). This indicates that in the pectin group, the inflammation subsided more quickly, while the udder responded with a reduction in inflammatory signs earlier than with standard treatment. In the Control group, the SCC also decreased, but the reduction was more gradual and delayed.
The faster normalization of the SCC in the milk of cows treated with pectin is consistent with the earlier resolution of clinical signs described above. Therefore, it can be assumed that IMM pectin application contributed to a more efficient reduction in the inflammatory response at the tissue level, leading to less leukocyte influx into the milk and quicker restoration of the udder’s physiological condition.
The therapeutic effects of pectin likely result from multiple overlapping mechanisms. As plant-derived polysaccharides, pectins possess immunomodulatory properties, including interaction with pattern recognition receptors such as TLR2 and TLR4. These interactions may downregulate the inflammatory cascade and result in a milder disease course [
24,
40]. While not directly bactericidal, this modulation may enhance host defense mechanisms and reduce tissue damage.
The anti-inflammatory effects of pectins have been documented beyond the context of mastitis. In human studies, citrus pectin supplementation significantly reduced the circulating levels of pro-inflammatory cytokines (TNF-α, IL-1β, IL-6, IFN-γ) and increased the IL-10 levels [
23]. Similar immunomodulatory effects were observed in animal models of colitis, allergy, and lung inflammation [
24,
40,
41], supporting the broader relevance of these findings.
In vitro studies also demonstrate direct pectin-induced modulation of immune cell activity, such as reduced cytokine and free radical production in chicken monocytes [
42]. While the data in bovines remain limited, these results support the rationale for pectin use in inflammatory conditions across species.
In addition to their biological activity, the physicochemical properties of pectins may play a complementary role. Their ability to form a gel-like, adhesive layer on mucosal surfaces could reduce bacterial adhesion, soothe local irritation, and facilitate mechanical clearance of pathogens during milking. Although in vitro gelation assays were not performed in this study, the capacity of pectins to undergo ionotropic gelation in the presence of divalent cations such as Ca2+ is well established. Milk and inflamed mammary tissues are rich in calcium ions, providing conditions that promote in situ gel formation following intramammary administration.
Studies in dermatological and mucosal models have demonstrated that pectin-based hydrogels form stable, bioadhesive films that protect epithelial surfaces, reduce transepithelial permeability, and support tissue regeneration [
43]. We assumed that similar gelation occurs IMM, enabling pectin to coat the epithelium, reduce bacterial adhesion, and enhance barrier function. This may partially explain the rapid clinical and cytological improvement observed in the pectin-treated group. This dual action, biological and mechanical, may help explain the favorable clinical and cytological outcomes observed, despite the use of a simple 10% solution without additional active ingredients.
Furthermore, pectins and related polysaccharides have been implicated in tissue regeneration processes, including autophagy activation and epithelial repair [
44]. Such mechanisms may have contributed to the faster stabilization of glandular function and the potentially lower risk of relapse observed in this study.
In summary, the observed efficacy of pectin appears to result from a synergistic combination of immunomodulatory effects, physical barrier formation, and support for tissue repair. This multi-level mechanism of action may offer significant advantages over conventional therapies that focus solely on pathogen eradication, particularly in light of increasing pressure to reduce antibiotic use in dairy production.
The data obtained provide a solid basis for planning larger-scale research. Pectin may prove to be a safe and effective alternative or complement to mastitis therapy, supporting efforts to reduce antibiotic use in veterinary medicine. Its potential economic benefits—no milk withdrawal period and fewer restrictions on milk sales—further enhance its attractiveness in farming practice.
4.2. Comparison with Other Polysaccharides (β-Glucan, Chitosan, Alginate)
Comparison of the obtained results with the effects found in other studies is very difficult, as intramammary use of pectins under farm or experimental conditions has not been thoroughly studied so far. However, there are studies in which immunomodulatory preparations, including natural polysaccharides (e.g., chitosan, β-glucans, alginate, pectins), were used intramammarily as anti-inflammatory and antibacterial agents or as drug carriers.
A number of studies have evaluated the effects of administering various polysaccharides directly into the mammary gland during mastitis. Even in earlier research, attempts were made to use β-1,3-glucans (e.g., from yeast) as immunostimulants in the udder. Persson Waller et al. [
15] administered β-1,3-glucan intramammarily to cows with chronic
S. aureus infections, achieving only a moderate improvement in the local immune response without significant bacterial eradication. In our study, we isolated
S. uberis and
E. coli—that is, both streptococci and coliforms. Pectin accelerated the decrease in the SCC and clinical remission regardless of the type of bacteria, indicating a broader spectrum and effectiveness in both acute and subclinical infections. Another study showed that intramammary administration of β-glucan to sheep with mastitis triggered a local response—increased migration of leukocytes (neutrophils, macrophages) into the udder [
45]. Our study shows that pectin significantly reduces the SCC levels within 48 h and accelerates the regression of clinical symptoms. This suggests that pectin induces faster and more effective inflammation control.
Although β-glucans exhibit some anti-inflammatory and immunostimulatory properties, their effectiveness in bacterial infections appears to depend on the experimental model. For example, better results were obtained in small ruminants in a fungal mastitis model, where IMM-applied β-glucan was as effective as conventional antifungal drugs [
46]. Our results refer to cattle and bacterial mastitis, where 10% pectin proved effective under farm conditions, both clinically and cytologically, without the need for additional antifungal drugs. In the case of bacterial infections, especially those caused by Gram-negative bacteria, pectin shows a clear advantage. β-glucans primarily stimulate innate immunity by activating macrophages and neutrophils, but this may be insufficient in chronic, deep-seated infections.
Pectin, in addition to immunostimulation (as mentioned above), forms a hydrogel barrier on the epithelial surface, which hinders bacterial adhesion and neutralizes toxins. This combination of a physicochemical mechanism and immunomodulation may explain the faster and more comprehensive response observed in our study.
In the treatment of acute bacterial mastitis, pectin appears to offer faster and more comprehensive effects than β-glucans, particularly in reducing the SCC and regressing clinical symptoms.
β-glucans may be best suited as immune support during the dry period or in prevention, whereas pectin may be a better candidate for treating existing bacterial infections. In the literature, chitosan, similarly to pectin, is investigated as a polysaccharide for intramammary application due to its anti-inflammatory and antimicrobial properties (
Staphylococcus,
Corynebacterium, and
Aerococcus) [
12,
44]. However, in practice, pectin turns out to be a simpler solution to apply, while being just as effective as chitosan. In our study, the rapid normalization of the SCC and regression of clinical symptoms suggest effective limitation of pathogen colonization or activity, confirming its therapeutic potential in mastitis treatment. Under farm conditions in cattle, 10% pectin alone proved to be clinically effective, achieving comparable cure rates and accelerated clinical remission compared to antibiotic therapy, without any additional compounds.
Chitosan acts mainly as an immunostimulant (stimulating phagocytes) and drug carrier (e.g., for matrine, berberine), which requires appropriate formulation and carrier stability [
47]. Pectin combines immunomodulation with ease of preparation (soluble solution), forming a hydrogel protective barrier.
Pectin alone may provide equally effective infection elimination and faster clinical improvement in cattle, without the need for additional substances or more complex formulations. It does not require extra carriers or active compounds.
Alginates (e.g., sodium alginate derived from algae) are polysaccharides used in gel formulations and nanoparticles due to their ability to gel in the presence of calcium ions. Alginates themselves have low toxicity—in in vitro studies on mammary gland epithelial cells, alginate solutions showed no cytotoxic effect (similar to other natural polymers, such as carboxymethylcellulose and xanthan gum) [
48]. Alginate is often combined with chitosan to create nanogels for controlled drug release. One example is a chitosan–alginate nanogel containing tilmicosin (a macrolide antibiotic). In an experiment on cows with staphylococcal mastitis, intramammary administration of this nanogel allowed for a 50% reduction in the antibiotic dose (150 vs. 300 mg/day) while achieving a higher cure rate (75% vs. 50%) in staphylococcal mastitis [
49]. This shows that alginate, in combination with chitosan, can effectively act as a drug carrier, prolonging its action and improving its therapeutic efficacy. Our 10% pectin solution worked independently, without additional carriers or antibiotics, achieving bacteriological effectiveness comparable to standard therapy and faster clinical improvement (a clear drop in the SCC after just 48 h). Alginate–chitosan nanogels allow for optimized antibiotic dosing, whereas pectin demonstrates sufficient standalone anti-inflammatory and antibacterial activity.
In contrast to β-glucans, chitosan, and alginates, which show limited effectiveness in bacterial infections, pectin provided rapid reduction of the SCC and better control of clinical symptoms. As mentioned, pectins have documented anti-inflammatory and immunomodulatory effects; their antibacterial activity and ability to modulate immune responses have been demonstrated in various models [
50]. In the context of mastitis treatment, it is worth noting that some plant-based intramammary preparations contain polysaccharides, which may play an active role. The rapid reduction in the SCC and resolution of clinical symptoms observed in this study after administration of a 10% pectin solution suggests the multi-level action of this polysaccharide in the mammary gland. As previously shown, pectins can modulate the host immune response by influencing the TLR2 and TLR4 receptors, thereby limiting the production of pro-inflammatory cytokines [
23,
24]. This effect may be particularly important in the early phase of inflammation, when excessive activation of the innate immune response exacerbates tissue damage.
Our observations also indicate local activation of defense mechanisms—a significant reduction in the SCC within 48 h after pectin infusion may suggest a rapid influx of leukocytes to the site of infection and enhanced phagocytic activity. A similar effect was previously noted with the use of β-glucans [
15] and chitosan during the dry period, where an increase in the SCC and lactoferrin indicated activation of protective mechanisms [
13].
Although polysaccharide therapies are more animal- and consumer-friendly (no withdrawal period, no risk of resistance), determining the effectiveness of polysaccharides such as pectin in acute mastitis cases requires further comparative studies with antibiotics. In our study, only the 10% pectin solution proved clinically effective—lower concentrations (2% and 4%) did not accelerate clinical remission, indicating that the effectiveness of these preparations depends on the concentration used.
4.3. Safety and Tolerance
An important observation was the absence of adverse effects related to pectin therapy during this study. None of the cows that received the 10% pectin solution intramammarily for 7 days showed any signs of local irritation in the treated quarters (e.g., increased pain, swelling not related to infection, or discharge suggesting an allergic reaction) or systemic symptoms indicating possible toxicity. No significant changes were observed in the hematological profiles, which may be attributed to the moderate nature of the inflammation and the absence of a systemic leukocyte shift typically seen in acute mastitis. Pectin application was well tolerated, during and after the treatment period, while no abnormal behavior or changes in milk quality were noted, apart from those related to the inflammatory process itself. No significant differences in the milk TNF-α levels were observed between the Pectin10 and Control groups. The lack of a TNF-α rise in the Pectin10 group may reflect a controlled local immune response, consistent with the immunomodulatory properties of pectin that support the resolution of inflammation without triggering excessive cytokine production.
This safety profile is in line with the general characteristics of pectins as natural substances long used in the pharmaceutical industry. Pectins are classified as dietary fibers and recognized as safe; moreover, studies on their use in especially sensitive populations (e.g., in infant formula) have confirmed their lack of harmful effects [
24]. The absence of adverse effects in this study indicates that IMM administration of pectins does not pose a health risk to cows and, at the same time, represents an attractive alternative in terms of food safety. Unlike antibiotics, pectins do not leave drug residues in milk and do not contribute to the selection of antimicrobial resistance. This safety aspect is particularly important in the context of efforts to reduce antibiotic use in food-producing animals.
An important aspect of evaluating alternative intramammary therapies is not only their clinical effectiveness but also their local tolerance and safety of use. Pectins, like other natural polysaccharides, show excellent local tolerance in the udder. In in vitro studies using mammary epithelial cell lines, pectin solutions did not exhibit cytotoxic effects, not even at high concentrations [
48]. Although direct reports on intramammary administration of pectins are limited, experience with gel formulations (e.g., hydroxypropyl methylcellulose, carboxymethylcellulose, alginate) indicates that these polysaccharides do not damage teat tissues and do not induce inflammation in ex vivo studies [
48]. In clinical practice, no irritations or discomfort were observed in cows after application of pectin solutions, which is consistent with the GRAS status of pectins commonly used in the food and pharmaceutical industries [
49]. This high safety profile confirms that a 10% pectin solution can be safely used intramammarily, minimizing the risk of adverse effects.
From a clinical practice perspective, the pharmacokinetic properties of pectins after intramammary administration are extremely important. Intramammary polysaccharides do not enter the systemic circulation and remain primarily within the udder tissue, which eliminates concerns about milk residues and the need for withdrawal periods. Pectins are locally biodegraded by enzymes (e.g., lysozyme) or phagocytosed by macrophages, and any potential trace presence in milk does not negatively affect its composition (lactose, fat, protein) or sensory qualities [
12]. Unlike antibiotics, pectins are not subject to residue limits, as they are classified as soluble dietary fiber with GRAS status. The intramammary pharmacokinetics of pectins are characterized by rapid transformation of the solution into a hydrogel, prolonged in situ retention, and minimal systemic absorption. This ensures a concentrated, long-lasting local effect without the risk of systemic toxicity [
14,
49].
4.4. Limitations of the Study and Directions for Further Research
The limitations of this study include its pilot nature, the relatively small number of animals, and the lack of a placebo group, which limit the interpretation of the absolute effectiveness of pectins. Another limitation is the absence of culture-based antimicrobial susceptibility testing. Without AST, we could not benchmark the efficacy against MDR/XDR phenotypes. Additionally, the pharmacokinetics of intramammary-administered pectin have not been established. Although pectin is not absorbed via the gastrointestinal tract and is generally considered safe, its persistence within the mammary gland and potential presence in milk remain unknown. These data are essential for estimating the minimum effective dose, establishing withdrawal times, and ensuring consumer safety. Due to the lack of pharmacokinetic information, the current dosing scheme (e.g., frequency of administration, solution concentration) was based primarily on preliminary assumptions and empirical experience.
Despite these limitations, this study confirms the promising therapeutic potential of pectins in mastitis treatment. In the context of increasing microbial resistance and the need to reduce antibiotic use in food-producing animals, non-antibiotic mastitis therapies such as pectin are particularly relevant. Our results suggest that pectins may be effective without the use of traditional antibacterial drugs; however, this requires further validation.
Future studies should aim to optimize the dosing protocols by determining the minimum effective concentrations, ideal dosing frequency, and pharmacokinetics and metabolism of pectins in the mammary gland. Additionally, broader immunological profiling should be included, encompassing both pro- and anti-inflammatory cytokines (e.g., IL-1β, IL-6, IL-10) and pattern recognition receptors such as TLR2 and TLR4 to better characterize pectin’s immunomodulatory effects.
Microbiome analysis before and after treatment could also provide insight into how pectin affects the intramammary microbial ecosystem and host–pathogen interactions. Moreover, given the observed need for daily pectin administration (seven doses) compared to only two doses in the antibiotic group, future work should explore improved delivery systems. These might include bioadhesive formulations, in situ gelling polymers, or nanoparticle-based carriers to reduce the treatment frequency while maintaining the therapeutic efficacy and minimizing the labor and compliance challenges.
Another mechanism worth exploring is pectin’s potential role in modulating bacterial biofilms and supporting epithelial repair. Pectins have demonstrated biofilm-disrupting and anti-adhesive properties in vitro. These features may be relevant in chronic mastitis caused by persistent biofilm-forming pathogens such as Staphylococcus aureus. Pectin has also been shown to stabilize the epithelial barriers in intestinal models via interactions with Toll-like receptors and mucosal immune pathways. Although these effects were not directly assessed in this study, future investigations should include histopathological evaluation of epithelial healing and post-treatment biofilm analysis.
Lastly, a formal economic assessment and evaluation of market acceptance, including consumer perception of “antibiotic-free” labeling, remain important areas for future research.
5. Conclusions
This pilot clinical trial showed that intramammary administration of a 10% pectin solution provided a similar level of clinical effectiveness, with no statistically significant difference compared to standard antibiotic treatment, while also potentially leading to faster clinical improvement and quicker normalization of inflammation markers (SCC), with no adverse effects observed.
The results also have broader significance, aligning with the principles of the One Health concept, which aims to integrate human, animal, and environmental health. Intramammary application of pectins may represent an important step toward reducing antibiotic use in dairy cattle farming, thereby helping to lower the risk of developing antimicrobial resistance.
The use of pectins, as natural and safe substances, fits into global strategies aimed at producing antibiotic-residue-free milk and supports the protection of public health and the environment. Pectin, as a natural, safe, and effective compound, may play a key role in the future in reducing antibiotic use in dairy cattle.