Insect Meal as an Alternative to Protein Concentrates in Poultry Nutrition with Future Perspectives (An Updated Review)
Abstract
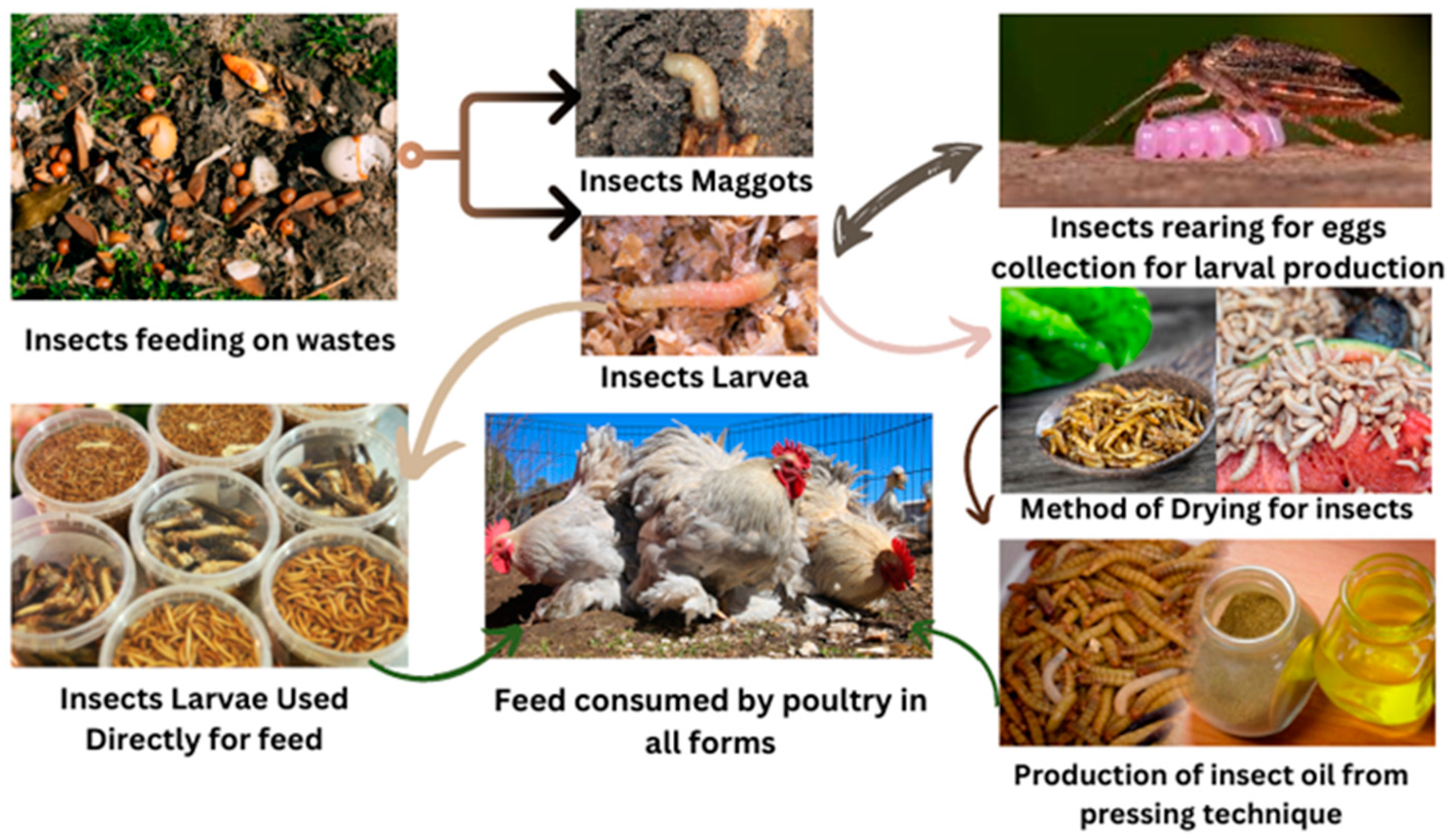
1. Introduction
2. Insect Meal as a Sustainable Nutrient Source in Poultry Diets
3. Digestibility of Insect-Based Feeds in Poultry Diets
4. Advantages of Using Insect Meal in Poultry Nutrition
5. Challenges and Limitations of Using Insect Meal in Poultry Nutrition
6. Future Prospects of Insect Meal as a Protein Concentrate in Poultry Nutrition
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Roser, M. Future Population Growth. Published Online at OurWorldInData. Org. Available online: https://ourworldindata.org/future-population-growth (accessed on 25 April 2023).
- Randive, K.; Raut, T.; Jawadand, S. An Overview of the Global Fertilizer Trends and India’s Position in 2020. Miner. Econ. 2021, 34, 371–384. [Google Scholar] [CrossRef]
- Asghar, M.U.; Doğan, S.C.; Wilk, M.; Korczyński, M. Effect of Dietary Supplementation of Black Cumin Seeds (Nigella sativa) on Performance, Carcass Traits, and Meat Quality of Japanese Quails (Coturnix coturnix japonica). Animals 2022, 12, 1298. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, I.; Inal, F.; Riaz, R.; Ahsan, U.; Kuter, E.; Ali, U. A Review of Black Soldier Fly (Hermetia illucens) as a Potential Alternative Protein Source in Broiler Diets. Ann. Anim. Sci. 2023. [Google Scholar] [CrossRef]
- Naımatı, S.; Doğan, S.C.; Asghar, M.U.; Wilk, M.; Korczyński, M. The Effect of Quinoa Seed (Chenopodium quinoa Willd.) Extract on the Performance, Carcass Characteristics, and Meat Quality in Japanese Quails (Coturnix coturnix japonica). Animals 2022, 12, 1851. [Google Scholar] [CrossRef] [PubMed]
- Thirumalaisamy, G.; Muralidharan, J.; Senthilkumar, S.; Sayee, R.H.; Priyadharsini, M. Cost-Effective Feeding of Poultry. Int. J. Sci. Environ. Technol. 2016, 5, 3997–4005. [Google Scholar]
- El-Sabrout, K.; Khalifah, A.; Mishra, B. Application of Botanical Products as Nutraceutical Feed Additives for Improving Poultry Health and Production. Vet. World 2023, 16, 369. [Google Scholar] [CrossRef]
- Khalifah, A.; Abdalla, S.; Rageb, M.; Maruccio, L.; Ciani, F.; El-Sabrout, K. Could Insect Products Provide a Safe and Sustainable Feed Alternative for the Poultry Industry? A Comprehensive Review. Animals 2023, 13, 1534. [Google Scholar] [CrossRef]
- FAO. The State of the World’s Animal Genetic Resources for Food and Agriculture; Food and Agriculture Organization of the United Nations: Rome, Italy, 2020. [Google Scholar]
- Chojnacka, K.; Mikula, K.; Izydorczyk, G.; Skrzypczak, D.; Witek-Krowiak, A.; Gersz, A.; Moustakas, K.; Iwaniuk, J.; Grzędzicki, M.; Korczyński, M. Innovative High Digestibility Protein Feed Materials Reducing Environmental Impact through Improved Nitrogen-Use Efficiency in Sustainable Agriculture. J. Environ. Manag. 2021, 291, 112693. [Google Scholar] [CrossRef]
- Liu, J.; Klebach, M.; Visser, M.; Hofman, Z. Amino Acid Availability of a Dairy and Vegetable Protein Blend Compared to Single Casein, Whey, Soy, and Pea Proteins: A Double-Blind, Cross-over Trial. Nutrients 2019, 11, 2613. [Google Scholar] [CrossRef]
- Gorissen, S.H.M.; Crombag, J.J.R.; Senden, J.M.G.; Waterval, W.A.H.; Bierau, J.; Verdijk, L.B.; van Loon, L.J.C. Protein Content and Amino Acid Composition of Commercially Available Plant-Based Protein Isolates. Amino Acids 2018, 50, 1685–1695. [Google Scholar] [CrossRef]
- Sanchez-Sabate, R.; Sabaté, J. Consumer Attitudes towards Environmental Concerns of Meat Consumption: A Systematic Review. Int. J. Environ. Res. Public Health 2019, 16, 1220. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Tang, J.; Yao, X.; Wu, Y.; Wang, X.; Feng, J. Effects of Dietary Inclusion of Fermented Cottonseed Meal on Growth, Cecal Microbial Population, Small Intestinal Morphology, and Digestive Enzyme Activity of Broilers. Trop. Anim. Health Prod. 2013, 45, 987–993. [Google Scholar] [CrossRef]
- Olukomaiya, O.; Fernando, C.; Mereddy, R.; Li, X.; Sultanbawa, Y. Solid-State Fermented Plant Protein Sources in the Diets of Broiler Chickens: A Review. Anim. Nutr. 2019, 5, 319–330. [Google Scholar] [CrossRef]
- Mariotti, F. 35—Plant Protein, Animal Protein, and Protein Quality. In Vegetarian and Plant-Based Diets in Health and Disease Prevention; Mariotti, F., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 621–642. ISBN 978-0-12-803968-7. [Google Scholar]
- Iji, P.A.; Toghyani, M.; Ahiwe, E.U.; Omede, A.A.; Applegate, T. Alternative Sources of Protein for Poultry Nutrition. Achiev. Sustain. Prod. Poult. Meat 2017, 2, 237–269. [Google Scholar]
- Olarotimi, O.J.; Adu, O.A. Potentials of Non-Conventional Protein Sources in Poultry Nutrition. Arch. Zootec. 2017, 66, 453–459. [Google Scholar]
- Moreki, J.C.; Tiroesele, B.; Chiripasi, S.C. Prospects of Utilizing Insects as Alternative Sources of Protein in Poultry Diets in Botswana: A Review. J. Anim. Sci. Adv. 2012, 2, 649–658. [Google Scholar]
- Khan, M.; Javed, M.M.; Zahoor, S.; Haq, U.-I. Kinetics and Thermodynamic Study of Urease Extracted from Soybeans. Biologia 2013, 59, 7–14. [Google Scholar]
- Veldkamp, T.; Meijer, N.; Alleweldt, F.; Deruytter, D.; Van Campenhout, L.; Gasco, L.; Roos, N.; Smetana, S.; Fernandes, A.; Van der Fels-Klerx, H.J. Overcoming Technical and Market Barriers to Enable Sustainable Large-Scale Production and Consumption of Insect Proteins in Europe: A susinchain Perspective. Insects 2022, 13, 281. [Google Scholar] [CrossRef]
- Van Huis, A. Edible Insects: Challenges and Prospects. Entomol. Res. 2022, 52, 161–177. [Google Scholar] [CrossRef]
- Demeke, S. Egg Production Performance of Local and White Leghorn Hens under Intensive and Rural Household Conditions in Ethiopia. Livest. Res. Rural. Dev. 2004, 16, 2004. [Google Scholar]
- Wu, D.; Cui, D.; Zhou, M.; Ying, Y. Information Perception in Modern Poultry Farming: A Review. Comput. Electron. Agric. 2022, 199, 107131. [Google Scholar] [CrossRef]
- Van Huis, A. Importance of Insects as Food in Africa. In African Edible Insects As Alternative Source of Food, Oil, Protein and Bioactive Components; Adam Mariod, A., Ed.; Springer International Publishing: Cham, Switzerland, 2020; pp. 1–17. ISBN 978-3-030-32952-5. [Google Scholar]
- Valdés, F.; Villanueva, V.; Durán, E.; Campos, F.; Avendaño, C.; Sánchez, M.; Domingoz-Araujo, C.; Valenzuela, C. Insects as Feed for Companion and Exotic Pets: A Current Trend. Animals 2022, 12, 1450. [Google Scholar] [CrossRef] [PubMed]
- Henchion, M.; Moloney, A.P.; Hyland, J.; Zimmermann, J.; McCarthy, S. Review: Trends for Meat, Milk and Egg Consumption for the next Decades and the Role Played by Livestock Systems in the Global Production of Proteins. Animal 2021, 15, 100287. [Google Scholar] [CrossRef]
- Gerber, P.; Opio, C.; Steinfeld, H. Poultry Production and the Environment—A Review; Animal Production and Health Division, Food and Agriculture Organization of the United Nations, Viale delle Terme di Caracalla: Roma, Italy, 2007; Volume 153, pp. 1–27. [Google Scholar]
- Asghar, M.U.; Rahman, A.; Hayat, Z.; Rafique, M.K.; Badar, I.H.; Yar, M.K.; Ijaz, M. Exploration of Zingiber Officinale Effects on Growth Performance, Immunity and Gut Morphology in Broilers. Braz. J. Biol. 2021, 83, e250296. [Google Scholar] [CrossRef]
- Adeniji, A.A. Effect of Replacing Groundnut Cake with Maggot Meal in the Diet of Broilers. Int. J. Poult. Sci. 2007, 6, 822–825. [Google Scholar] [CrossRef]
- Mugwanya, M.; Dawood, M.A.; Kimera, F.; Sewilam, H. Anthropogenic Temperature Fluctuations and Their Effect on Aquaculture: A Comprehensive Review. Aquac. Fish. 2022, 7, 223–243. [Google Scholar] [CrossRef]
- Oonincx, D.G.; De Boer, I.J. Environmental Impact of the Production of Mealworms as a Protein Source for Humans—A Life Cycle Assessment. PLoS ONE 2012, 7, e51145. [Google Scholar] [CrossRef] [PubMed]
- DeFoliart, G.R. Insects as a Global Food Resource: The History of Talking about It at the University of Wisconsin; University of Wisconsin: Madison, WI, USA, 2012. [Google Scholar]
- Ahmed, I.; Inal, F.; Kahraman, O.; Riaz, R. Insect Fat: An Alternative Source of Soybean Oil in Poultry Feed. In Proceedings of the 3rd International Eurasian Conference on Biological and Chemical Sciences, Ankara, Turkey, 19–20 March 2020; pp. 19–20. [Google Scholar]
- Li, Y.; Guo, B.; Wu, Z.; Wang, W.; Li, C.; Liu, G.; Cai, H. Effects of Fermented Soybean Meal Supplementation on the Growth Performance and Cecal Microbiota Community of Broiler Chickens. Animals 2020, 10, 1098. [Google Scholar] [CrossRef]
- Alves, J.F.; Mendes, S.; Alves da Silva, A.; Sousa, J.P.; Paredes, D. Land-Use Effect on Olive Groves Pest Prays Oleae and on Its Potential Biocontrol Agent Chrysoperla Carnea. Insects 2021, 12, 46. [Google Scholar] [CrossRef]
- Terova, G.; Gini, E.; Gasco, L.; Moroni, F.; Antonini, M.; Rimoldi, S. Effects of Full Replacement of Dietary Fishmeal with Insect Meal from Tenebrio Molitor on Rainbow Trout Gut and Skin Microbiota. J. Anim. Sci. Biotechnol. 2021, 12, 30. [Google Scholar] [CrossRef] [PubMed]
- Maulu, S.; Langi, S.; Hasimuna, O.J.; Missinhoun, D.; Munganga, B.P.; Hampuwo, B.M.; Gabriel, N.N.; Elsabagh, M.; Van Doan, H.; Kari, Z.A. Recent Advances in the Utilization of Insects as an Ingredient in Aquafeeds: A Review. Anim. Nutr. 2022, 11, 334–349. [Google Scholar] [CrossRef] [PubMed]
- Hadi, J.; Brightwell, G. Safety of Alternative Proteins: Technological, Environmental and Regulatory Aspects of Cultured Meat, Plant-Based Meat, Insect Protein and Single-Cell Protein. Foods 2021, 10, 1226. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.; Raai, M.N.; Zain, N.A.M.; Massawe, F.; Singh, A.; Wan-Mohtar, W.A.A.Q.I. In Search of Alternative Proteins: Unlocking the Potential of Underutilized Tropical Legumes. Food Sec. 2019, 11, 1205–1215. [Google Scholar] [CrossRef]
- Elsoud, M.M.A.; Elmansy, E.A.; Abdelhamid, S.A. Economic and Non-Seasonal Source for Production of Chitin and Chitosan. J. Chem. Rev. 2022, 4, 222–240. [Google Scholar]
- Čičková, H.; Newton, G.L.; Lacy, R.C.; Kozánek, M. The Use of Fly Larvae for Organic Waste Treatment. Waste Manag. 2015, 35, 68–80. [Google Scholar] [CrossRef]
- Finke, M.D. Complete Nutrient Content of Four Species of Commercially Available Feeder Insects Fed Enhanced Diets during Growth. Zoo Biol. 2015, 34, 554–564. [Google Scholar] [CrossRef]
- Rumpold, B.A.; Schlüter, O.K. Nutritional Composition and Safety Aspects of Edible Insects. Mol. Nutr. Food Res. 2013, 57, 802–823. [Google Scholar] [CrossRef]
- Schiavone, A.; Dabbou, S.; Marco, M.D.; Cullere, M.; Biasato, I.; Biasibetti, E.; Capucchio, M.T.; Bergagna, S.; Dezzutto, D.; Meneguz, M.; et al. Black Soldier Fly Larva Fat Inclusion in Finisher Broiler Chicken Diet as an Alternative Fat Source. Animal 2018, 12, 2032–2039. [Google Scholar] [CrossRef] [PubMed]
- Di Mattia, C.; Battista, N.; Sacchetti, G.; Serafini, M. Antioxidant Activities in Vitro of Water and Liposoluble Extracts Obtained by Different Species of Edible Insects and Invertebrates. Front. Nutr. 2019, 6, 106. [Google Scholar] [CrossRef]
- Makkar, H.P.S.; Tran, G.; Heuzé, V.; Ankers, P. State-of-the-Art on Use of Insects as Animal Feed. Anim. Feed. Sci. Technol. 2014, 197, 1–33. [Google Scholar] [CrossRef]
- Lähteenmäki-Uutela, A.; Hénault-Ethier, L.; Marimuthu, S.B.; Talibov, S.; Allen, R.N.; Nemane, V.; Vandenberg, G.W.; Józefiak, D. The Impact of the Insect Regulatory System on the Insect Marketing System. J. Insects Food Feed. 2018, 4, 187–198. [Google Scholar] [CrossRef]
- De Marco, M.; Martínez, S.; Hernandez, F.; Madrid, J.; Gai, F.; Rotolo, L.; Belforti, M.; Bergero, D.; Katz, H.; Dabbou, S. Nutritional Value of Two Insect Larval Meals (Tenebrio molitor and Hermetia illucens) for Broiler Chickens: Apparent Nutrient Digestibility, Apparent Ileal Amino Acid Digestibility and Apparent Metabolizable Energy. Anim. Feed. Sci. Technol. 2015, 209, 211–218. [Google Scholar] [CrossRef]
- Salomone, R.; Saija, G.; Mondello, G.; Giannetto, A.; Fasulo, S.; Savastano, D. Environmental impact of food waste bioconversion by insects: Application of life cycle assessment to process using Hermetia illucens. J. Clean. Prod. 2017, 140, 890–905. [Google Scholar] [CrossRef]
- WHO. Protein and Amino Acid Requirements in Human Nutrition; World Health Organization: Geneva, Switzerland, 2007; ISBN 978-92-4-120935-9. [Google Scholar]
- Triunfo, M.; Tafi, E.; Guarnieri, A.; Scieuzo, C.; Hahn, T.; Zibek, S.; Salvia, R.; Falabella, P. Insect Chitin-Based Nanomaterials for Innovative Cosmetics and Cosmeceuticals. Cosmetics 2021, 8, 40. [Google Scholar] [CrossRef]
- Ojha, S.; Bekhit, A.E.-D.; Grune, T.; Schlüter, O.K. Bioavailability of Nutrients from Edible Insects. Curr. Opin. Food Sci. 2021, 41, 240–248. [Google Scholar] [CrossRef]
- Weru, J.; Chege, P.; Kinyuru, J. Nutritional Potential of Edible Insects: A Systematic Review of Published Data. Int. J. Trop. Insect Sci. 2021, 41, 2015–2037. [Google Scholar] [CrossRef]
- Meyer-Rochow, V.B.; Gahukar, R.T.; Ghosh, S.; Jung, C. Chemical Composition, Nutrient Quality and Acceptability of Edible Insects Are Affected by Species, Developmental Stage, Gender, Diet, and Processing Method. Foods 2021, 10, 1036. [Google Scholar] [CrossRef]
- Selaledi, L.; Hassan, Z.; Manyelo, T.G.; Mabelebele, M. Insects’ Production, Consumption, Policy, and Sustainability: What Have We Learned from the Indigenous Knowledge Systems? Insects 2021, 12, 432. [Google Scholar] [CrossRef] [PubMed]
- Mlček, J.; Rop, O.; Borkovcová, M.; Bednářová, M. A Comprehensive Look at the Possibilities of Edible Insects as Food in Europe—A Review. Pol. J. Food Nutr. Sci. 2014, 64, 147–157. [Google Scholar] [CrossRef]
- Williams, J.P.; Williams, J.R.; Kirabo, A.; Chester, D.; Peterson, M. Chapter 3—Nutrient Content and Health Benefits of Insects. In Insects as Sustainable Food Ingredients; Dossey, A.T., Morales-Ramos, J.A., Rojas, M.G., Eds.; Academic Press: San Diego, CA, USA, 2016; pp. 61–84. ISBN 978-0-12-802856-8. [Google Scholar]
- Tzompa-Sosa, D.A.; Yi, L.; van Valenberg, H.J.F.; van Boekel, M.A.J.S.; Lakemond, C.M.M. Insect Lipid Profile: Aqueous versus Organic Solvent-Based Extraction Methods. Food Res. Int. 2014, 62, 1087–1094. [Google Scholar] [CrossRef]
- Zamudio-Flores, P.B.; Hernández -Gonzaléz, M.; Cano, V.G.G. Food Supplements from a Grasshopper: A Developmental Stage-Wise Evaluation of Amino Acid Profile, Protein and Vitamins in Brachystola Magna (Girard). Emir. J. Food Agric. 2019, 561–568. [Google Scholar] [CrossRef]
- Mwangi, M.N.; Oonincx, D.G.A.B.; Stouten, T.; Veenenbos, M.; Melse-Boonstra, A.; Dicke, M.; Loon, J.J.A. van Insects as Sources of Iron and Zinc in Human Nutrition. Nutr. Res. Rev. 2018, 31, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Zielińska, E.; Baraniak, B.; Karaś, M.; Rybczyńska, K.; Jakubczyk, A. Selected Species of Edible Insects as a Source of Nutrient Composition. Food Res. Int. 2015, 77, 460–466. [Google Scholar] [CrossRef]
- Ramos-Elorduy, J.; González, E.A.; Hernández, A.R.; Pino, J.M. Use of Tenebrio Molitor (Coleoptera: Tenebrionidae) to Recycle Organic Wastes and as Feed for Broiler Chickens. J. Econ. Entomol. 2002, 95, 214–220. [Google Scholar] [CrossRef]
- Rahimi, F. Feasibility of Hunting Invasive Locusts and Studying Its Economic, Social, and Environmental Effects (Approach: Preventing the Spraying of Locusts). Preprints.org 2023, 2023020029. [Google Scholar] [CrossRef]
- Ghosh, S.; Lee, S.-M.; Jung, C.; Meyer-Rochow, V.B. Nutritional Composition of Five Commercial Edible Insects in South Korea. J. Asia-Pac. Entomol. 2017, 20, 686–694. [Google Scholar] [CrossRef]
- Ham, Y.K.; Kim, S.W.; Song, D.H.; Kim, H.W.; Kim, I.S. Nutritional composition of white-spotted flower chafer (Protaetia brevitarsis) larvae produced from commercial insect farms in Korea. Food Sci. Anim. Resour. 2021, 41, 416. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, E.; de Vries, W. Potential benefits of using Hermetia illucens frass as a soil amendment on food production and for environmental impact reduction. Curr. Opin. Green Sustain. Chem. 2020, 25, 100335. [Google Scholar] [CrossRef]
- Maurer, V.; Holinger, M.; Amsler, Z.; Früh, B.; Wohlfahrt, J.; Stamer, A.; Leiber, F. Replacement of Soybean Cake by Hermetia Illucens Meal in Diets for Layers. J. Insects Food Feed. 2016, 2, 83–90. [Google Scholar] [CrossRef]
- Spranghers, T.; Michiels, J.; Vrancx, J.; Ovyn, A.; Eeckhout, M.; De Clercq, P.; De Smet, S. Gut Antimicrobial Effects and Nutritional Value of Black Soldier Fly (Hermetia Illucens L.) Prepupae for Weaned Piglets. Anim. Feed. Sci. Technol. 2018, 235, 33–42. [Google Scholar] [CrossRef]
- Uushona, T.; Simasiku, A.; Petrus, N.P. Evaluation of Musca Domestica (House Fly) Larvae Production from Organic Waste. Welwitschia Int. J. Agric. Sci. 2019, 1, 35–40. [Google Scholar] [CrossRef]
- Liu, C.; Lian, F.; Smith, D.E.; Russell, R.M.; Wang, X.-D. Lycopene Supplementation Inhibits Lung Squamous Metaplasia and Induces Apoptosis via Up-Regulating Insulin-like Growth Factor-Binding Protein 3 in Cigarette Smoke-Exposed Ferrets1. Cancer Res. 2003, 63, 3138–3144. [Google Scholar]
- Bahadori, Z.; Esmaielzadeh, L.; Karimi-Torshizi, M.A.; Seidavi, A.; Olivares, J.; Rojas, S.; Salem, A.Z.M.; Khusro, A.; López, S. The Effect of Earthworm (Eisenia foetida) Meal with Vermi-Humus on Growth Performance, Hematology, Immunity, Intestinal Microbiota, Carcass Characteristics, and Meat Quality of Broiler Chickens. Livest. Sci. 2017, 202, 74–81. [Google Scholar] [CrossRef]
- Kuntadi, K.; Adalina, Y.; Maharani, K.E. Nutritional compositions of six edible insects in java. Indones. J. For. Res. 2018, 5, 57–68. [Google Scholar] [CrossRef]
- Akande, O.A.; Falade, O.O.; Badejo, A.A.; Adekoya, I. Assessment of Mulberry Silkworm Pupae and African Palm Weevil Larvae as Alternative Protein Sources in Snack Fillings. Heliyon 2020, 6, e03754. [Google Scholar] [CrossRef]
- Kariuki, P.K. Diversity of Grasshoppers and Locusts, Life Parameters, Fat and Protein Content of Acanthacris Ruficornis in Nakuru County, Kenya. Ph.D. Thesis, University of Nairobi, Nairobi, Kenya, 2022. [Google Scholar]
- Oonincx, D.G.A.B.; Finke, M.D. Nutritional Value of Insects and Ways to Manipulate Their Composition. J. Insects Food Feed 2021, 7, 639–659. [Google Scholar] [CrossRef]
- Barroso, F.G.; Sánchez-Muros, M.-J.; Segura, M.; Morote, E.; Torres, A.; Ramos, R.; Guil, J.-L. Insects as Food: Enrichment of Larvae of Hermetia Illucens with Omega 3 Fatty Acids by Means of Dietary Modifications. J. Food Compos. Anal. 2017, 62, 8–13. [Google Scholar] [CrossRef]
- Anvo, M.P.M.; Toguyéni, A.; Otchoumou, A.K.; Zoungrana-Kaboré, C.Y.; Kouamelan, E.P. Nutritional Qualities of Edible Caterpillars Cirina Butyrospermi in Southwestern of Burkina Faso. Int. J. Innov. Appl. Stud. 2016, 18, 639–645. [Google Scholar]
- Sedgh-Gooya, S.; Torki, M.; Darbemamieh, M.; Khamisabadi, H.; Karimi Torshizi, M.A.; Abdolmohamadi, A. Yellow Mealworm, Tenebrio Molitor (Col: Tenebrionidae), Larvae Powder as Dietary Protein Sources for Broiler Chickens: Effects on Growth Performance, Carcass Traits, Selected Intestinal Microbiota and Blood Parameters. J. Anim. Physiol. Anim. Nutr. 2021, 105, 119–128. [Google Scholar] [CrossRef]
- Newton, G.L.; Booram, C.V.; Barker, R.W.; Hale, O.M. Dried Hermetia Illucens Larvae Meal as a Supplement for Swine. J. Anim. Sci. 1977, 44, 395–400. [Google Scholar] [CrossRef]
- Gasco, L.; Biasato, I.; Dabbou, S.; Schiavone, A.; Gai, F. Animals Fed Insect-Based Diets: State-of-the-Art on Digestibility, Performance and Product Quality. Animals 2019, 9, 170. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Hack, M.E.; Shafi, M.E.; Alghamdi, W.Y.; Abdelnour, S.A.; Shehata, A.M.; Noreldin, A.E.; Ashour, E.A.; Swelum, A.A.; Al-Sagan, A.A.; Alkhateeb, M. Black Soldier Fly (Hermetia illucens) Meal as a Promising Feed Ingredient for Poultry: A Comprehensive Review. Agriculture 2020, 10, 339. [Google Scholar] [CrossRef]
- Reilly, L.M.; Hu, Y.; von Schaumburg, P.C.; de Oliveira, M.R.D.; He, F.; Rodriguez-Zas, S.L.; Southey, B.R.; Parsons, C.M.; Utterback, P.; Lambrakis, L.; et al. Chemical Composition of Selected Insect Meals and Their Effect on Apparent Total Tract Digestibility, Fecal Metabolites, and Microbiota of Adult Cats Fed Insect-Based Retorted Diets. J. Anim. Sci. 2022, 100, skac024. [Google Scholar] [CrossRef]
- Mnisi, C.M.; Oyeagu, C.E.; Ruzvidzo, O. Mopane Worm (Gonimbrasia Belina Westwood) Meal as a Potential Protein Source for Sustainable Quail Production: A Review. Sustainability 2022, 14, 5511. [Google Scholar] [CrossRef]
- Pretorius, Q. The Evaluation of Larvae of Musca domestica (Common House Fly) as Protein Source for Broiler Production. Ph.D. Thesis, Stellenbosch University, Stellenbosch, South Africa, 2011. [Google Scholar]
- Areerat, S.; Chundang, P.; Lekcharoensuk, C.; Kovitvadhi, A. Possibility of Using House Cricket (Acheta domesticus) or Mulberry Silkworm (Bombyx mori) Pupae Meal to Replace Poultry Meal in Canine Diets Based on Health and Nutrient Digestibility. Animals 2021, 11, 2680. [Google Scholar] [CrossRef] [PubMed]
- Raju, M.; Rao, S.R.; Paul, S.S.; Prakash, B.; Reddy, M.R.; Kannan, A.; Shanmugam, M.; Kumar, P.S.P. Source Variation in Nutrient Profile and the Effects of Black Soldier Fly (Hermetia illucens) Larva Meal at Graded Levels in Diet on Performance, Carcass Traits, Serum Biochemical Profile and Cellular Immune Response in Broiler Chicken. Int. J. Trop. Insect Sci. 2023. [Google Scholar] [CrossRef]
- Attivi, K.; Mlaga, K.G.; Agboka, K.; Tona, K.; Kouame, Y.A.E.; Lin, H.; Tona, K. Effect of Fish Meal Replacement by Black Soldier Fly (Hermetia illucens) Larvae Meal on Serum Biochemical Indices, Thyroid Hormone and Zootechnical Performance of Laying Chickens. J. Appl. Poult. Res. 2022, 31, 100275. [Google Scholar] [CrossRef]
- Murawska, D.; Daszkiewicz, T.; Sobotka, W.; Gesek, M.; Witkowska, D.; Matusevičius, P.; Bakula, T. Partial and Total Replacement of Soybean Meal with Full-Fat Black Soldier Fly (Hermetia Illucens L.) Larvae Meal in Broiler Chicken Diets: Impact on Growth Performance, Carcass Quality and Meat Quality. Animals 2021, 11, 2715. [Google Scholar] [CrossRef]
- Dörper, A.; Veldkamp, T.; Dicke, M. Use of Black Soldier Fly and House Fly in Feed to Promote Sustainable Poultry Production. J. Insects Food Feed. 2021, 7, 761–780. [Google Scholar] [CrossRef]
- Kareem, K.Y.; Abdulla, N.R.; Foo, H.L.; Mohd, A.N.; Zamri, N.S.; Loh, T.C.; Alshelmani, M.I. Effect of Feeding Larvae Meal in the Diets on Growth Performance, Nutrient Digestibility and Meat Quality in Broiler Chicken. Indian J. Anim. Sci. 2018, 88, 1180–1185. [Google Scholar] [CrossRef]
- Kawasaki, K.; Hashimoto, Y.; Hori, A.; Kawasaki, T.; Hirayasu, H.; Iwase, S.; Hashizume, A.; Ido, A.; Miura, C.; Miura, T.; et al. Evaluation of Black Soldier Fly (Hermetia illucens) Larvae and Pre-Pupae Raised on Household Organic Waste, as Potential Ingredients for Poultry Feed. Animals 2019, 9, 98. [Google Scholar] [CrossRef] [PubMed]
- Barragan-Fonseca, K.B.; Dicke, M.; van Loon, J.J.A. Nutritional Value of the Black Soldier Fly (Hermetia illucens L.) and Its Suitability as Animal Feed—A Review. J. Insects Food Feed. 2017, 3, 105–120. [Google Scholar] [CrossRef]
- Dahiru, T.; Aliyu, A.A.; Shehu, A.U. A Review of Population-Based Studies on Diabetes Mellitus in Nigeria. Sub-Sahar. Afr. J. Med. 2016, 3, 59. [Google Scholar] [CrossRef]
- Khan, S.H. Recent Advances in Role of Insects as Alternative Protein Source in Poultry Nutrition. J. Appl. Anim. Res. 2018, 46, 1144–1157. [Google Scholar] [CrossRef]
- Okah, U.; Onwujiariri, E.B. Performance of Finisher Broiler Chickens Fed Maggot Meal as a Replacement for Fish Meal. J. Agric. Technol. 2012, 8, 471–477. [Google Scholar]
- Hwangbo, J.; Hong, E.C.; Jang, A.; Kang, H.K.; Oh, J.S.; Kim, B.W.; Park, B.S. Utilization of House Fly-Maggots, a Feed Supplement in the Production of Broiler Chickens. J. Environ. Biol. 2009, 30, 609–614. [Google Scholar]
- Ganda, H.; Zannou, E.T.; Kenis, M.; Abihona, H.A.; Houndonougbo, F.M.; Chrysostome, C.A.A.M.; Chougourou, D.C.; Mensah, G.A. Effect of Four Rearing Substrates on the Yield and the Chemical Composition of Housefly Larvae, Musca Domestica L. 1758 (Diptera: Muscidae). Int. J. Trop. Insect Sci. 2022, 42, 1331–1339. [Google Scholar] [CrossRef]
- Sanou, A.G.; Sankara, F.; Pousga, S.; Coulibaly, K.; Nacoulma, J.P.; Kenis, M.; Clottey, V.A.; Nacro, S.; Somda, I.; Ouédraogo, I. Indigenous Practices in Poultry Farming Using Maggots in Western Burkina Faso. J. Insects Food Feed. 2018, 4, 219–228. [Google Scholar] [CrossRef]
- Ndelekwute, E.K.; Essien, E.B.; Assam, E.D.; Ekanem, N.J. Potentials of Earthworm and Its By-Products in Animal Agriculture and Waste Management—A Review. Bangladesh J. Anim. Sci. 2016, 45, 2. [Google Scholar] [CrossRef]
- Abbas, G. Earthworm meal: A novel non-conventional feed ingredient for sustainable poultry production. Pak. J. Sci. 2022, 74, 4. [Google Scholar]
- Elahi, U.; Ma, Y.B.; Wu, S.G.; Wang, J.; Zhang, H.J.; Qi, G.H. Growth performance, carcass characteristics, meat quality and serum profile of broiler chicks fed on housefly maggot meal as a replacement of soybean meal. J. Anim. Physiol. Anim. Nutr. 2020, 104, 1075–1084. [Google Scholar] [CrossRef] [PubMed]
- Dillak, S.Y.F.G.; Suryatni, N.P.F.; Handayani, H.T.; Temu, S.T.; Nastiti, H.P.; Osa, D.B.; Henuk, Y.L. The effect of fed maggot meal as a supplement in the commercial diets on the performance of finisher broiler chickens. IOP Conf. Ser. Earth Environ. Sci. 2018, 260, 012056. [Google Scholar] [CrossRef]
- Akpodiete, O.J.; Inoni, O.E. Economics of production of broiler chickens fed maggot meal as replacement for fish meal. Niger. J. Anim. Prod. 2000, 27, 59–63. [Google Scholar] [CrossRef]
- Banday, M.T.; Adil, S.; Sheikh, I.U.; Hamadani, H.; Qadri, F.I.; Sahfi, M.E.; Sait, H.S.; Abd El-Mageed, T.A.; Salem, H.M.; Taha, A.E. The Use of Silkworm Pupae (Bombyx mori) Meal as an Alternative Protein Source for Poultry. Worlds Poult. Sci. J. 2023, 79, 119–134. [Google Scholar] [CrossRef]
- Khatun, R.; Howlider, M.A.R.; Rahman, M.M.; Hasanuzzaman, M.; Rahman, M.Z. Replacement of Fish Meal by Silkworm Pupae in Broiler Diets. Pak. J. Biol. Sci. 2003, 6, 955–958. [Google Scholar] [CrossRef]
- Khatun, R.; Azmal, S.A.; Sarker, M.S.K.; Rashid, M.A.; Hussain, M.A.; Miah, M.Y. Effect of Silkworm Pupae on the Growth and Egg Production Performance of Rhode Island Red (RIR) Pure Line. Int. J. Poult. Sci. 2005, 4, 718–720. [Google Scholar]
- Miah, M.Y.; Singh, Y.; Cullere, M.; Tenti, S.; Dalle Zotte, A. Effect of Dietary Supplementation with Full-Fat Silkworm (Bombyx mori L.) Chrysalis Meal on Growth Performance and Meat Quality of Rhode Island Red\times Fayoumi Crossbred Chickens. Ital. J. Anim. Sci. 2020, 19, 447–456. [Google Scholar] [CrossRef]
- Ijaiya, A.T.; Eko, E.O. Effect of Replacing Dietary Fish Meal with Silkworm (Anaphe Infracta) Caterpillar Meal on Performance, Carcass Characteristics and Haematological Parameters of Finishing Broiler Chicken. Pak. J. Nutr. 2009, 8, 850–855. [Google Scholar] [CrossRef]
- Biasato, I.; De Marco, M.; Rotolo, L.; Renna, M.; Lussiana, C.; Dabbou, S.; Capucchio, M.T.; Biasibetti, E.; Costa, P.; Gai, F. Effects of Dietary Tenebrio Molitor Meal Inclusion in Free-Range Chickens. J. Anim. Physiol. Anim. Nutr. 2016, 100, 1104–1112. [Google Scholar] [CrossRef]
- Marareni, M.; Mnisi, C.M. Growth Performance, Serum Biochemistry and Meat Quality Traits of Jumbo Quails Fed with Mopane Worm (Imbrasia belina) Meal-Containing Diets. Vet. Anim. Sci. 2020, 10, 100141. [Google Scholar] [CrossRef] [PubMed]
- Elahi, U.; Xu, C.; Wang, J.; Lin, J.; Wu, S.; Zhang, H.; Qi, G. Insect Meal as a Feed Ingredient for Poultry. Anim. Biosci. 2022, 35, 332. [Google Scholar] [CrossRef]
- Bovera, F.; Loponte, R.; Marono, S.; Piccolo, G.; Parisi, G.; Iaconisi, V.; Gasco, L.; Nizza, A. Use of Tenebrio Molitor Larvae Meal as Protein Source in Broiler Diet: Effect on Growth Performance, Nutrient Digestibility, and Carcass and Meat Traits. J. Anim. Sci. 2016, 94, 639–647. [Google Scholar] [CrossRef]
- Ballitoc, D.A.; Sun, S. Ground Yellow Mealworms (Tenebrio molitor L.) Feed Supplementation Improves Growth Performance and Carcass Yield Characteristics in Broilers. Open Sci. Repos. Agric. 2013, 18, e23050425. [Google Scholar]
- Kim, S.Y.; Park, J.B.; Lee, Y.B.; Yoon, H.J.; Lee, K.Y.; Kim, N.J. Growth Characteristics of Mealworm Tenebrio Molitor. J. Sericultural Entomol. Sci. 2015, 53, 1–5. [Google Scholar] [CrossRef]
- Abd Rahman Jabir, M.D. Evaluation of Superworm Meal (Zophobas morio) as a Dietary Protein Source for Red Tilapia (Oreochromis sp.) Diet/Abd Rahman Jabir Mohd Din. Ph.D. Thesis, University of Malaya, Kuala Lumpur, Malaysia, 2012. [Google Scholar]
- Hussain, I.; Khan, S.; Sultan, A.; Chand, N.; Khan, R.; Alam, W.; Ahmad, N. Meal Worm (Tenebrio molitor) as Potential Alternative Source of Protein Supplementation in Broiler. Int. J. Biosci. 2017, 10, 225–262. [Google Scholar]
- Marono, S.; Piccolo, G.; Loponte, R.; Di Meo, C.; Attia, Y.A.; Nizza, A.; Bovera, F. In Vitro Crude Protein Digestibility of Tenebrio Molitor and Hermetia Illucens Insect Meals and Its Correlation with Chemical Composition Traits. Ital. J. Anim. Sci. 2015, 14, 3889. [Google Scholar] [CrossRef]
- Bovera, F.; Piccolo, G.; Gasco, L.; Marono, S.; Loponte, R.; Vassalotti, G.; Mastellone, V.; Lombardi, P.; Attia, Y.A.; Nizza, A. Yellow Mealworm Larvae (Tenebrio molitor L.) as a Possible Alternative to Soybean Meal in Broiler Diets. Br. Poult. Sci. 2015, 56, 569–575. [Google Scholar] [CrossRef]
- Jang, W.-W.; Chung, T.-H.; Choi, I.-H. Growth Performance and Economic Evaluation of Insect Feed Powder-Fed Ducks. J. Environ. Sci. Int. 2019, 28, 709–712. [Google Scholar] [CrossRef]
- Ouko, K.O.; Mboya, J.B.; Obiero, K.O.; Ogello, E.O.; Mukhebi, A.W.; Muthoka, M.; Munguti, J.M. Determinants of Fish Farmers’ Awareness of Insect-Based Aquafeeds in Kenya; the Case of Black Soldier Fly Larvae Meal. Cogent Food Agric. 2023, 9, 2187185. [Google Scholar] [CrossRef]
- Muftau, M.A.; Olorede, B.R. Carcass Characteristics and Economics of Broiler Chickens Fed Different Levels of Grasshopper Meal in Place of Fish Meal. Anim. Prod. Res. Adv. 2009, 5, 248–251. [Google Scholar] [CrossRef]
- Ojianwuna, C.C.; Enwemiwe, V.N. Effects of Insect Formulated Feeds on Performance and Nutrient Retention Characteristics of Caged Cobb Broilers. Egypt. Acad. J. Biol. Sci. B. Zool. 2022, 14, 315–329. [Google Scholar] [CrossRef]
- Wang, H.; Guo, Y.; Shih, J.C. Effects of Dietary Supplementation of Keratinase on Growth Performance, Nitrogen Retention and Intestinal Morphology of Broiler Chickens Fed Diets with Soybean and Cottonseed Meals. Anim. Feed. Sci. Technol. 2008, 140, 376–384. [Google Scholar] [CrossRef]
- de Souza-Vilela, J.; Andrew, N.R.; Ruhnke, I.; de Souza-Vilela, J.; Andrew, N.R.; Ruhnke, I. Insect Protein in Animal Nutrition. Anim. Prod. Sci. 2019, 59, 2029–2036. [Google Scholar] [CrossRef]
- Phesatcha, B.; Phesatcha, K.; Viennaxay, B.; Matra, M.; Totakul, P.; Wanapat, M. Cricket Meal (Gryllus bimaculatus) as a Protein Supplement on In Vitro Fermentation Characteristics and Methane Mitigation. Insects 2022, 13, 129. [Google Scholar] [CrossRef] [PubMed]
- Permatahati, D.; Mutia, R.; Astuti, D.A. Effect of Cricket Meal (Gryllus bimaculatus) on Production and Physical Quality of Japanese Quail Egg. Trop. Anim. Sci. J. 2019, 42, 53–58. [Google Scholar] [CrossRef]
- Shah, A.A.; Wanapat, M. Gryllus Testaceus Walker (Crickets) Farming Management, Chemical Composition, Nutritive Profile, and Their Effect on Animal Digestibility. Entomol. Res. 2021, 51, 639–649. [Google Scholar] [CrossRef]
- Lieke, T.; Meinelt, T.; Hoseinifar, S.H.; Pan, B.; Straus, D.L.; Steinberg, C.E. Sustainable Aquaculture Requires Environmental-Friendly Treatment Strategies for Fish Diseases. Rev. Aquac. 2020, 12, 943–965. [Google Scholar] [CrossRef]
- Leiber, F.; Gelencsér, T.; Stamer, A.; Amsler, Z.; Wohlfahrt, J.; Früh, B.; Maurer, V. Insect and Legume-Based Protein Sources to Replace Soybean Cake in an Organic Broiler Diet: Effects on Growth Performance and Physical Meat Quality. Renew. Agric. Food Syst. 2017, 32, 21–27. [Google Scholar] [CrossRef]
- Allegretti, G.; Talamini, E.; Schmidt, V.; Bogorni, P.C.; Ortega, E. Insect as Feed: An Emergy Assessment of Insect Meal as a Sustainable Protein Source for the Brazilian Poultry Industry. J. Clean. Prod. 2018, 171, 403–412. [Google Scholar] [CrossRef]
- Ismail, B.P.; Senaratne-Lenagala, L.; Stube, A.; Brackenridge, A. Protein Demand: Review of Plant and Animal Proteins Used in Alternative Protein Product Development and Production. Anim. Front. 2020, 10, 53–63. [Google Scholar] [CrossRef]
- Imsland, A.K.; Reynolds, P.; Hangstad, T.A.; Jónsdóttir, Ó.D.B.; Noble, T.; Wilson, M.; Mackie, J.A.; Elvegård, T.A.; Urskog, T.C.; Mikalsen, B. Feeding Behaviour and Growth of Lumpfish (Cyclopterus lumpus L.) Fed with Feed Blocks. Aquac. Res. 2018, 49, 2006–2012. [Google Scholar] [CrossRef]
- IPIFF. The European Insect Sector Today: Challenges, Opportunities and Regulatory Landscape; IPIFF: Brussels, Belgium, 2018. [Google Scholar]
- Verbeke, W.; Sans, P.; Van Loo, E.J. Challenges and Prospects for Consumer Acceptance of Cultured Meat. J. Integr. Agric. 2015, 14, 285–294. [Google Scholar] [CrossRef]
- Sogari, G.; Amato, M.; Biasato, I.; Chiesa, S.; Gasco, L. The Potential Role of Insects as Feed: A Multi-Perspective Review. Animals 2019, 9, 119. [Google Scholar] [CrossRef] [PubMed]
- Onwezen, M.C.; Bouwman, E.P.; Reinders, M.J.; Dagevos, H. A Systematic Review on Consumer Acceptance of Alternative Proteins: Pulses, Algae, Insects, Plant-Based Meat Alternatives, and Cultured Meat. Appetite 2021, 159, 105058. [Google Scholar] [CrossRef] [PubMed]
- Park, K.; Goo, B.; Kim, Y.; Kim, E.; Park, J.Y.; Yun, J.S. Insect, potential source of animal feed. Food Sci. Ind. 2022, 55, 176–187. [Google Scholar] [CrossRef]
- de Koning, W.; Dean, D.; Vriesekoop, F.; Aguiar, L.K.; Anderson, M.; Mongondry, P.; Oppong-Gyamfi, M.; Urbano, B.; Luciano, C.A.G.; Jiang, B.; et al. Drivers and Inhibitors in the Acceptance of Meat Alternatives: The Case of Plant and Insect-Based Proteins. Foods 2020, 9, 1292. [Google Scholar] [CrossRef]
- Niva, M.; Vainio, A. Towards More Environmentally Sustainable Diets? Changes in the Consumption of Beef and Plant- and Insect-Based Protein Products in Consumer Groups in Finland. Meat Sci. 2021, 182, 108635. [Google Scholar] [CrossRef]
- Meyer-Rochow, V.B.; Jung, C. Insects used as food and feed: Isn’t that what we all need? Foods 2020, 9, 1003. [Google Scholar] [CrossRef] [PubMed]
- Meyer-Rochow, V.B. Can insects help to ease the problem of world food shortage? Search 1975, 6, 261–262. [Google Scholar]
- IPIFF (International Platform Insects for Food and Feed). An Overview of the European Market of Insects as Feed. Available online: https://ipiff.org/wp-content/uploads/2021/04/Apr-27-2021-IPIFF_The-European-market-of-insects-as-feed.pdf (accessed on 2 June 2023).
| Insects Species | Nutrient and DM % | Insect Life Stage | Method of Processing | Reference |
|---|---|---|---|---|
| Mealworm (Tenebrio molitor) | DM (97.02), CP (53.83), EE (28.03), Ash (6.99), CF (7.53), Chitin (5.6), GE (2.8), Ca (0.06), P (1.10) | Larvae | Degutted, freeze-dried | [63,64,65] |
| Field cricket (Gryllus bimaculatus) | CP (58.3), CF (9.5), EE (11.9), Ash (9.7) | Adult | Degutted, freeze-dried | [39,66] |
| Black soldier fly (Hermetia illucens) | CP (42.2), Chitin (5.6), EE (21.8) Ash (10.0), Ca (7.00), P(1.00) K (0.69), Na (0.13), Mg (0.39), Fe (0.14), Mn (246), Zn (108), Cu (6.0) | Prepupae | Whole, freeze-dried | [64,67,68,69] |
| House fly (Musca domestica) | CP (55.4), Chitin (6.2), EE (20.8), Ash (6.2) | Larvae | Meal, oven-dried for 2 days | [70] |
| Earthworm (Lumbricus terrestris) | CP (63.0), CF (5.9), Ash (8.9) Na (0.43), Ca (0.53), K (0.62) P (0.94), GE (1476kJ/100g) | Adult | direct heating or freezing before drying | [71,72] |
| Silkworm (Bombyx mori) | CP (23.1), CF (14.2), Moisture (60–70), Ash (1.5), GE (229 kcal/kg) | pupae | Sun dried, powdered | [73,74] |
| Grasshoppers (Caelifera (Suborder) | CP (28.13), CF (2.38), Ash (9.97) EE (4.18), GE (1618 kcal/g) | Larvae | Degutted, freeze-dried | [75] |
| Locust (Schistocerca gregaria) | CP (52.3), EE (12), CF (19), Ash (10.0) | Larvae | Degutted, freeze-dried | [76] |
| Crickets (Gryllus testaceus walker) | CP (58.3), EE (10.3) | Larvae | Degutted, freeze-dried | [77] |
| Westwood (Cirina forda) | CP (20.0), EE (12.5), Ash (8.7) carbohydrate (54.3), K (47.6) P (45.9), Na (44.4), Mg (43.8), Zn (24.1), Ca (12.8), Fe (1.2) | Larvae | Degutted, freeze-dried | [78] |
| Mopane worm (Gonimbrasia belina) | CP (55), Ash (5.8), Lignin (5.2), N (9.0), Fat (16.7), K (35.2), Ca (16.0), P (14.7), Mg (4.1, Fe (12.7) Zn (1.9), Na (33.3) | Larvae | Freeze-dried | [79] |
| Insect Species | References | Protein Sources for Poultry |
|---|---|---|
| Black Soldier Fly (Hermetia illucens) | [87] | BSFLM showed linear ↑ BWG and FI consumed up to 5% in the diet of broiler chickens during the initial 3 weeks of age. The serum biochemical profile and cellular immune response were unaffected, ↓ yield, ↓ breast weight, and ↑ abdominal fat content |
| [34,88] | Use of exogenous AAs with BSF ↑ BWG ↑ GP. Alternative options for replacing 2%, 4%, 6%, and 8% of FM in the diet with BSFMM in chickens from 14–56 days ↑ FE, ↑ BWG, and ↓ FI compared to the CG | |
| [89,90] | 17% BSFLM fed in 42 days ↑ FE of broiler chickens | |
| [91] | Use of dietary 2, 4, 6, 8, and 10% BSFLM in ↑ FE, ↑ BWG, and ↓ FI in poultry and 8% of BSFLM group | |
| [92] | BSF larvae could substitute SBM as an ingredient for poultry feed. FI and the laying of hens were examined, and no difference was observed; eggshell thickness and weight were ↑ in the pre-pupae-fed group compared to CG. | |
| [93] | BSFL led to ↑ protein level and other feed. It could only partially substitute traditional feedstuff because complete replacement leads to ↓ in performance. | |
| [94] | 5% BSFLM showed ↑ FI, BWG, and ↓ FE in comparison to CG, with exhibited ↓ FI and ↑ BWG mostly. | |
| Fly Maggots (Musca domestica) | [95] | SBM was substituted with a MM at rates of 0, 40, 50, and 60%. ↑ in BWG, ↑ in FI and FE were significantly ↓. Digestibility of DM, CP, EE, and ash were ↑ and CF was ↓ lower. |
| [96] | MM replaced FM at 0, 20, 30, 40, and 50% and showed ↓ feed and ↓ BWG in contrast with 40–50% CG | |
| [97] | 10 and 15% maggot diet instead of FM showed ↑ level of maggot supplementation caused ↑ BWG, while FE remained the same ↑ FI. | |
| [98] | MM provides high nutritive value with good protein and digestibility contents. | |
| [99] | The investigation highlighted the necessity of creating sustainable, efficient, safe, and effective production techniques, as well as the possibility of employing maggots as protein feed in traditional chicken farming. | |
| [100] | EWM is ↑ in protein and ↑ in AAs. EWM reportedly ↑ FI, supported growth, ↑ carcass quality in broiler chickens, and marginally ↑ egg size and hen day in layers at dietary inclusion levels of 0.2 to 0.6%. | |
| [101] | The study looked into the use of EWM in fish and poultry feed and found that it ↑ BWG, ↑ FE, and ↑ growth rates in broilers, as well as egg production in layers. | |
| [102] | Broilers were fed with 0, 4, and 8% MM to replace SBM, which showed ↑ FE, no significant effect on BWG, and FI in MM-fed group as compared to control group during the starter phase; 8% MM can be used as an alternative protein source without any adverse effects. | |
| [103] | Diets containing 0 CG, 25, 50, and 75% MM as broiler chicken feed. Results showed no effect on FI and ↑ BWG, which helped to lower the cost of commercial feed by up to 25%. | |
| [104] | FM replaced by MM in broilers at a rate of 75% on protein basis showed reduced feed cost and cost of meat per kg in chicken in finisher diet. This replacement resulted in 15.78%↑ in net return during starter, 12.27% in grower, and 13.62% in finisher phase of feeding. MM proved to be cost-effective as a substitute to FM. | |
| Silkworm (Bombyx mori) | [105] | ↑ SWPM in chicken feed resulted in an ↓ feed cost per unit. |
| [106] | ↑ in GR, FE, meat yield, and profitability by ↑ levels of SWPM. | |
| [107] | Efficiency, development, and laying were considerably↑ for diets with 6% SWPM in comparison with 0% and 8% SWPM. | |
| [108] | Broilers fed processed SWP at 25 and 50% ratios have ↑ BWG and ↑ FE over raw SWP. | |
| [109] | In poultry finisher diet, SWM was substituted for FM (25, 50, 75, and 100%). There were no substantial variations in FI, BWG, FE, or protein efficiency ratio amongst dietary treatments. | |
| Mealworm (Tenebrio molitor) | [110] | MW has the capacity to convert ↓ nutritive waste products into a ↑ protein diet. |
| [111] | If MWM levels ↑ in diet, the color of lean meat turns to yellow. An ↑ breast muscle weight and ↑ quality of meat were gained by using ↑ levels of MWM up to 6% in the broiler feed. | |
| [112] | MWM addition to broiler diets may be caused by a number of conditions, including the species and age of broilers, MWM doses, and source and replaced meal type. | |
| [110] | MWM in addition to broiler diets at 5–15% ↑ BWG and FI, while ↓ FE and intestinal anatomy have no influence on carcass hematochemical parameters. | |
| [113] | The substitution of 29.65% MWM for soybean meal ↑ FE, intestinal digestibility, and spleen weight. | |
| [49,114,115] | MWM inclusion in grill diets regulates meat quality and AAs and fatty acid contents. | |
| [116] | SWM ↓ CP over fish meal based on chemical constitution and nutritional digestibility. The SWM exhibited high values of CF as well as ↑ quality protein, making it a suitable substitute for fish meal. | |
| [117] | BWG increased with increasing MW levels (0.1, 0.2, and 0.3% MW, respectively). The supplemented groups had ↑ FE over the control group. The FI rates of the different groups did not differ significantly. | |
| [113,118,119] | ↑ BWG at a maximum level of 25% MW. | |
| In the broiler diet, SBM was completely replaced with MW larvae. Most growth performance, carcass characteristics, and meat chemical and physical attributes were unaffected by using MW as the major protein source in the poultry diet. However, the FE ↑ in the MW group over the entire experimental period compared to the SBM group. | ||
| [119] | MW larvae had no effect on the FI and BWG in relation to isoproteic and isoenergetic factors in diet of broilers aged 30–62 days. | |
| Grasshoppers (Caelifera (Suborder) | [71] | GHM was substituted for 20% and 40% FM in poultry diets with no impact on BWG or FI. |
| [120] | When GHM levels of 2.5–7.5% are introduced to broiler diets, BWG and FE are reduced, except for carcass protein content. | |
| [121] | 2.5% GHM in the grill diet was an appropriate and less expensive alternative to imported FM (100% replacement), while the overall diet contained slightly ↓ protein. | |
| [122] | The effect of replacing FM with GHM (0, 50, and 100%) on carcass characteristics and grill chicken production economics. Bird BWG ↑ as GHM levels ↑. GHM had a significantly ↓ FI over the CG. | |
| [95] | The results showed that during the starter phase, daily FI, BWG, and FE rates were statistically similar across diets. BWG tends to be increased after feeding with GHM as compared to FM as a control diet. | |
| Locust (Schistocerca gregaria) | [123] | The purpose of this study was to look into the chemical content, ↑ nutritional content, ↑ AAs, ↑ CP, ↑ CF, ↑ TC, ↑ CF, ↑ ash, ↑ gross energy, and ↑ minerals. |
| [47] | Locusts and grasshoppers, like other insects, are ↑ in protein and ↑ AAs. These insects can replace up to 25% of conventional protein-rich feed resources such as SBM and fish meal in poultry, pig, and fish diets. | |
| [123] | In grill starter diets, 50% FM was replaced with desert locust meal, which ↑ BWG, FI and FE as compared to ↑ levels of locust meals (3.4% and 6.8%) and CG. | |
| [64] | The results showed that insecticide-sprayed locust meal had a ↓ FI over non-sprayed locust meal and control. Furthermore, both types of locust meals reduced live BW and FE more than the CG. | |
| Crickets (Gryllus testaceus walker) | [124] | BWG, FI, and feed ratio levels of 5%, 10%, and 15% CM fed to poultry from eight to twenty days post-hatching were not extensively affected by diet. |
| [125] | CM ↑ FE, ↑ quality energy, ↑ protein and fat to poultry diet. | |
| [126] | It also has CP, CF, fat, and total digestible nutrients, as well as in addition of essential AAs. | |
| [127] | The use of CM in the quail ration ↑ egg production and the physical quality of eggs, such as egg WG, egg white weight, eggshell weight, and yolk score. It can be concluded that CM can partially or completely replace FM in layer diets. | |
| [128] | CM-fed group showed ↑ dry matter digestibility, ash content, and CF digestibility with ↓ FE and ↑ digestion over CG. | |
| Westwood (Cirina forda) | [25] | The ability of broiler chicks to replace FM (30, 50, 70, and 100%) with Cirina forda larvae was tested. The findings demonstrated that the FI and BWG of birds given larvae-containing diets did not differ substantially from the CG during the starter and finisher stages. |
| [129] | WWLM was investigated for its impact on laying performance and egg characteristics in hens, and it was revealed that it may replace up to 75% of FM in laying hen diets without influencing FI, BWG, egg production, FE, or egg quality features. The data indicated that with 100% replacement, the daily egg output, egg weight, and feed utilization efficiency all declined dramatically. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sajid, Q.U.A.; Asghar, M.U.; Tariq, H.; Wilk, M.; Płatek, A. Insect Meal as an Alternative to Protein Concentrates in Poultry Nutrition with Future Perspectives (An Updated Review). Agriculture 2023, 13, 1239. https://doi.org/10.3390/agriculture13061239
Sajid QUA, Asghar MU, Tariq H, Wilk M, Płatek A. Insect Meal as an Alternative to Protein Concentrates in Poultry Nutrition with Future Perspectives (An Updated Review). Agriculture. 2023; 13(6):1239. https://doi.org/10.3390/agriculture13061239
Chicago/Turabian StyleSajid, Qurat Ul Ain, Muhammad Umair Asghar, Haneef Tariq, Martyna Wilk, and Arkadiusz Płatek. 2023. "Insect Meal as an Alternative to Protein Concentrates in Poultry Nutrition with Future Perspectives (An Updated Review)" Agriculture 13, no. 6: 1239. https://doi.org/10.3390/agriculture13061239
APA StyleSajid, Q. U. A., Asghar, M. U., Tariq, H., Wilk, M., & Płatek, A. (2023). Insect Meal as an Alternative to Protein Concentrates in Poultry Nutrition with Future Perspectives (An Updated Review). Agriculture, 13(6), 1239. https://doi.org/10.3390/agriculture13061239







