Analysis of Linkage on Interaction of Main Aspects (Genotype by Environment Interaction, Stability and Genetic Parameters) of 1000 Kernels in Maize (Zea mays L.)
Abstract
1. Introduction
2. Materials and Methods
2.1. Field Trails
2.2. Statistical Analysis
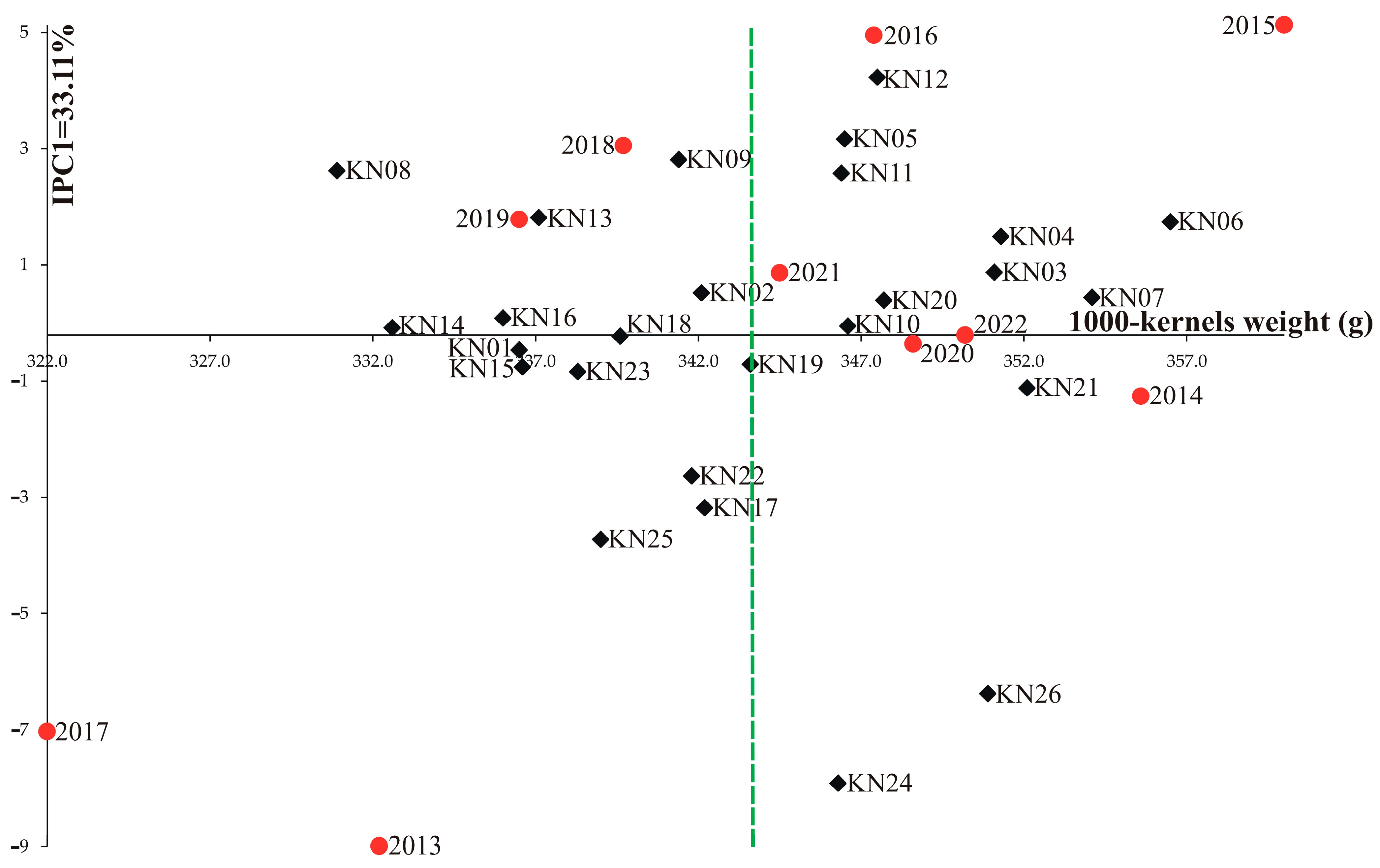
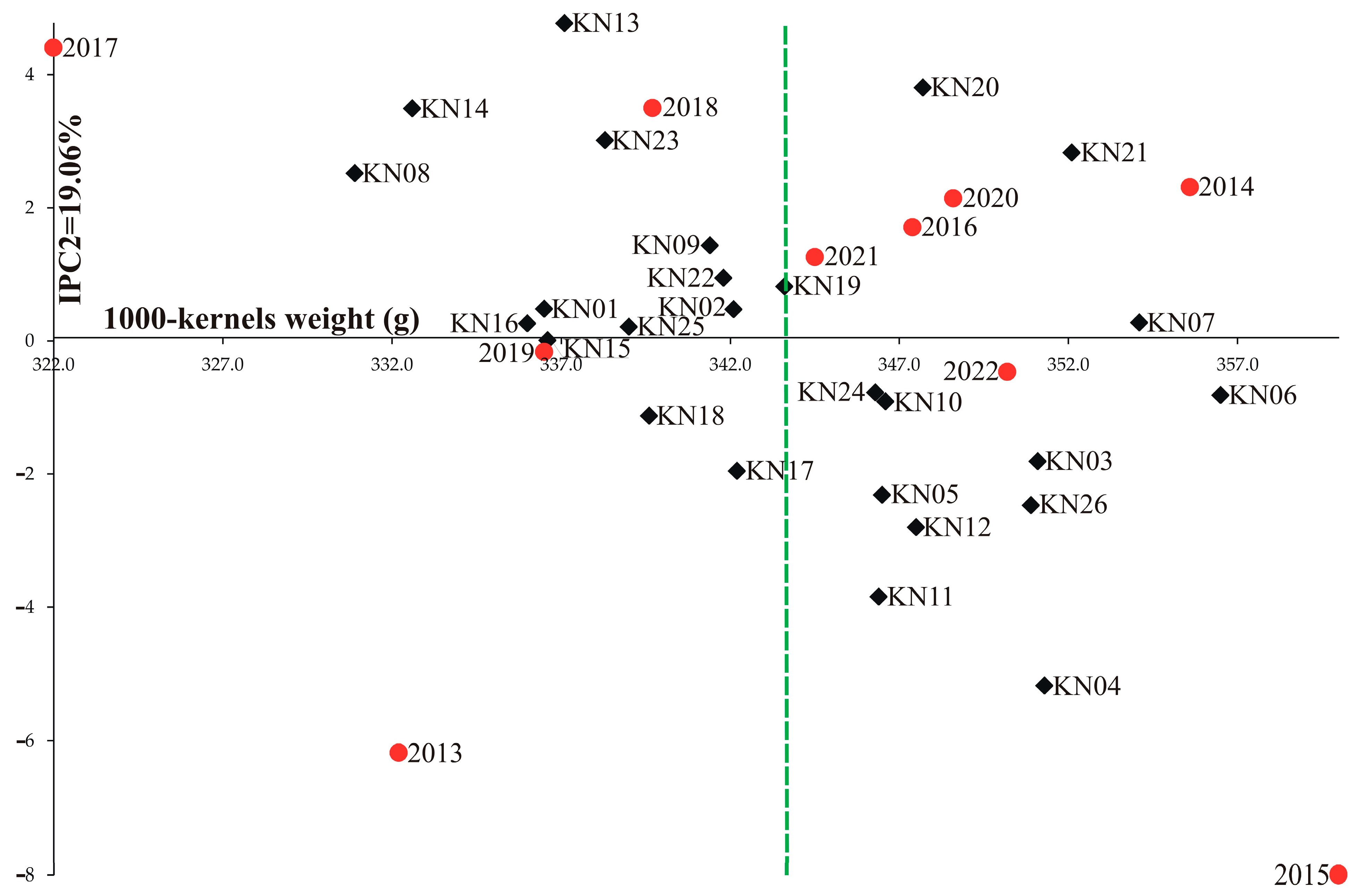
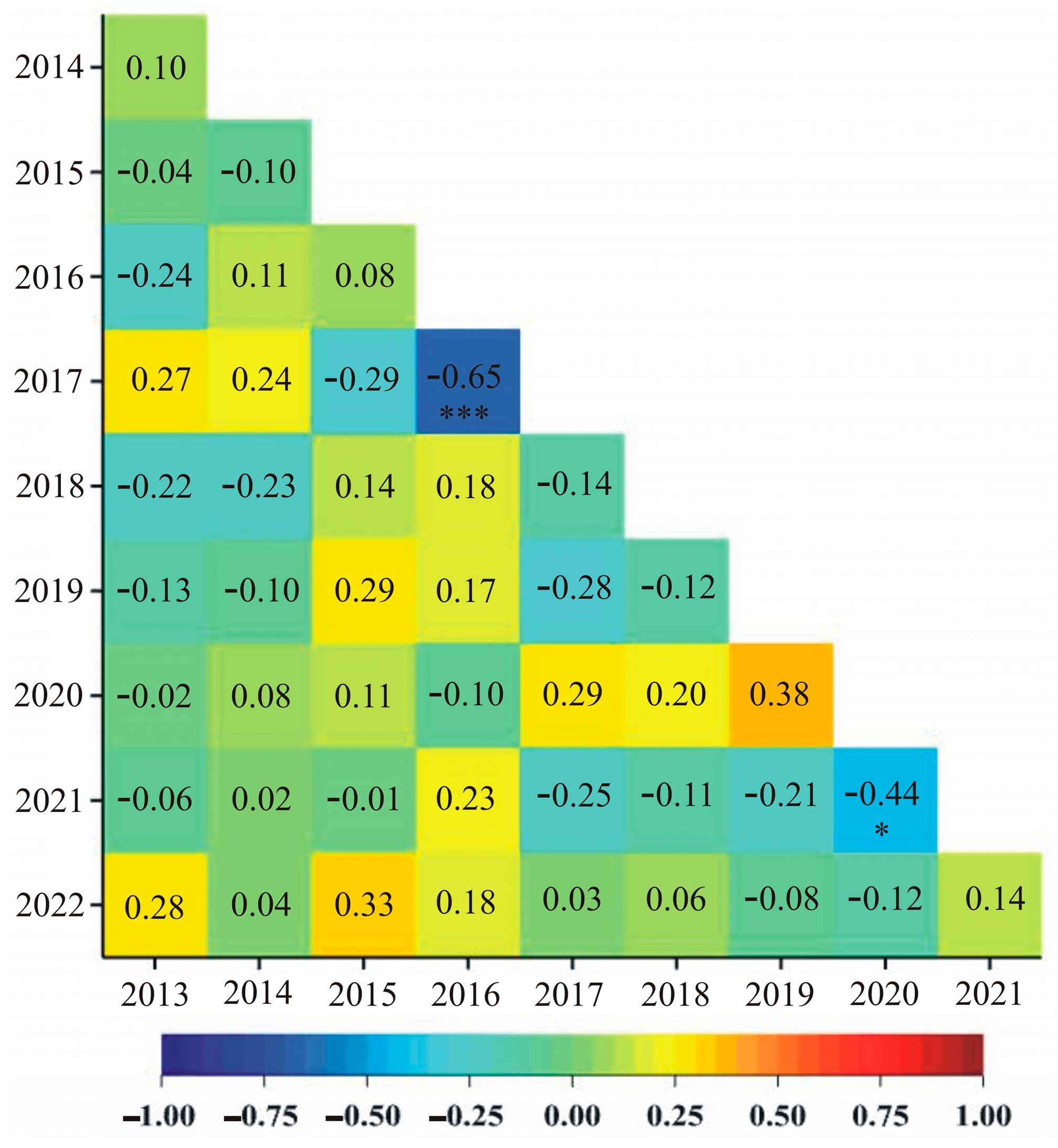
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Wang, Y.; Bi, Y.; Jiang, F.; Shaw, R.K.; Sun, J.; Hu, C.; Guo, R.; Fan, X. Mapping and Functional Analysis of QTL for Kernel Number per Row in Tropical and Temperate–Tropical Introgression Lines of Maize (Zea mays L.). Curr. Issues Mol. Biol. 2023, 45, 4416–4430. [Google Scholar] [CrossRef] [PubMed]
- Garg, M.; Sharma, N.; Sharma, S.; Kapoor, P.; Kumar, A.; Chunduri, V.; Arora, P. Biofortified Crops Generated by Breeding, Agronomy, and Transgenic Approaches Are Improving Lives of Millions of People around the World. Front. Nutr. 2018, 5, 12. [Google Scholar] [CrossRef] [PubMed]
- Farooq, A.; Farooq, N.; Akbar, H.; Hassan, Z.U.; Gheewala, S.H. A Critical Review of Climate Change Impact at a Global Scale on Cereal Crop Production. Agronomy 2023, 13, 162. [Google Scholar] [CrossRef]
- Albahri, G.; Alyamani, A.A.; Badran, A.; Hijazi, A.; Nasser, M.; Maresca, M.; Baydoun, E. Enhancing Essential Grains Yield for Sustainable Food Security and Bio-Safe Agriculture through Latest Innovative Approaches. Agronomy 2023, 13, 1709. [Google Scholar] [CrossRef]
- Campos, H.; Cooper, M.; Habben, J.E.; Edmeades, G.O.; Schussler, J.R. Improving drought tolerance in maize: A view from industry. Field Crops Res. 2004, 90, 19–34. [Google Scholar] [CrossRef]
- Ranum, P.; Peña-Rosas, J.P.; Garcia-Casal, M.N. Global maize production, utilization, and consumption. Ann. N.Y. Acad. Sci. 2014, 1312, 105–112. [Google Scholar] [CrossRef]
- Erenstein, O.; Jaleta, M.; Sonder, K.; Mottaleb, K.; Prasanna, B.M. Global maize production, consumption and trade: Trends and R&D implications. Food Sec. 2022, 14, 1295–1319. [Google Scholar] [CrossRef]
- Kashyap, S.; Kumar, R.; Ram, H.; Kumar, A.; Basak, N.; Sheoran, P.; Bhatacharjee, S.; Biswal, B.; Ali, G.; Kumar, B.; et al. Quantitative and Qualitative Response of Fodder Maize to Use of Bulk and Nano-fertilizers in North Western Plains of India. Agronomy 2023, 13, 1889. [Google Scholar] [CrossRef]
- Patel, S.; Hazel, C.M.; Winterton, A.G.M.; Gleadle, A.E. Surveillance of fumonisins in UK maize-based foods and other cereals. Food Addit. Contam. 1997, 14, 187–191. [Google Scholar] [CrossRef]
- Yadav, V.K.; Supriya, P. Value Addition in Maize. In Maize: Nutrition Dynamics and Novel Uses; Chaudhary, D., Kumar, S., Langyan, S., Eds.; Springer: New Delhi, India, 2014. [Google Scholar] [CrossRef]
- Kumar, K.; Singh, J.; Singh, B.R.; Chandra, S.; Chauhan, N.; Yadav, M.K.; Kumar, P. Consumption and processing patterns of maize (Zea mays): A review. Pharma Innov. J. 2022, 11, 51–57. [Google Scholar]
- Patzek, T.W. Thermodynamics of the Corn-Ethanol Biofuel Cycle. Crit. Rev. Plant Sci. 2004, 23, 519–567. [Google Scholar] [CrossRef]
- Groom, M.J.; Gray, E.M.; Townsend, P.A. Biofuels and Biodiversity: Principles for Creating Better Policies for Biofuel Production. Conserv. Biol. 2008, 22, 602–609. [Google Scholar] [CrossRef] [PubMed]
- Quintero, J.A.; Montoya, M.I.; Sánchez, O.J.; Giraldo, O.H.; Cardona, C.A. Fuel ethanol production from sugarcane and corn: Comparative analysis for a Colombian case. Energy 2008, 33, 385–399. [Google Scholar] [CrossRef]
- Hassan, M.H.; Kalam, A. An Overview of Biofuel as a Renewable Energy Source: Development and Challenges. Procedia Eng. 2013, 56, 39–53. [Google Scholar] [CrossRef]
- Ramos, J.L.; Pakuts, B.; Godoy, P.; García-Franco, A.; Duque, E. Addressing the energy crisis: Using microbes to make biofuels. Microb. Biotechnol. 2022, 15, 1026–1030. [Google Scholar] [CrossRef]
- Felten, D.; Fröba, N.; Fries, J.; Emmerling, C. Energy balances and greenhouse gas-mitigation potentials of bioenergy cropping systems (Miscanthus, rapeseed, and maize) based on farming conditions in Western Germany. Renew. Energy 2013, 55, 160–174. [Google Scholar] [CrossRef]
- Zhang, R.; Ma, S.; Li, L.; Zhang, M.; Tian, S.; Wang, D.; Liu, K.; Liu, H.; Zhu, W.; Wang, X. Comprehensive utilization of corn starch processing by-products: A review. Grain Oil Sci. Technol. 2021, 4, 89–107. [Google Scholar] [CrossRef]
- Parisi, G.; Tulli, F.; Fortina, R.; Marino, R.; Bani, P.; Zotte, A.D.; De Angelis, A.; Piccolo, G.; Pinotti, L.; Schiavone, A.; et al. Protein hunger of the feed sector: The alternatives offered by the plant world. Ital. J. Anim. Sci. 2020, 19, 1204–1225. [Google Scholar] [CrossRef]
- Suyker, A.E.; Verma, S.B. Coupling of carbon dioxide and water vapor exchanges of irrigated and rainfed maize–soybean cropping systems and water productivity. Agric. For. Meteorol. 2010, 150, 553–563. [Google Scholar] [CrossRef]
- Allen, L.H., Jr.; Kakani, V.G.; Vu, J.C.V.; Boote, K.J. Elevated CO2 increases water use efficiency by sustaining photosynthesis of water-limited maize and sorghum. J. Plant Physiol. 2011, 168, 1909–1918. [Google Scholar] [CrossRef]
- Panchasara, H.; Samrat, N.H.; Islam, N. Greenhouse Gas Emissions Trends and Mitigation Measures in Australian Agriculture Sector—A Review. Agriculture 2021, 11, 85. [Google Scholar] [CrossRef]
- Zeng, T.; Meng, Z.; Yue, R.; Lu, S.; Li, W.; Li, W.; Meng, H.; Sun, Q. Genome wide association analysis for yield related traits in maize. BMC Plant Biol. 2022, 22, 449. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Xu, W.; Velten, J.; Xin, Z.; Stout, J. Characterization of maize inbred lines for drought and heat tolerance. JSVC 2012, 67, 354–364. [Google Scholar] [CrossRef]
- Ahila Priyadharshini, R.; Arivazhagan, S.; Arun, M.; Mirnalini, A. Maize leaf disease classification using deep convolutional neural networks. Neural. Comput. Applic. 2019, 31, 8887–8895. [Google Scholar] [CrossRef]
- Li, X.Y.; Gong, J.D. Effects of different ridge:furrow ratios and supplemental irrigation on crop production in ridge and furrow rainfall harvesting system with mulches. Agric. Water Manag. 2002, 54, 243–254. [Google Scholar] [CrossRef]
- Ren, B.; Zhang, J.; Li, X.; Fan, X.; Dong, S.; Liu, P.; Zhao, B. Effects of waterlogging on the yield and growth of summer maize under field conditions. Can. J. Plant Sci. 2014, 94, 23–31. [Google Scholar] [CrossRef]
- Fang, H.; Niu, M.; Wang, X.; Zhang, Q. Effects of reduced chemical application by mechanical-chemical synergistic weeding on maize growth and yield in East China. Front. Plant Sci. 2022, 13, 1024249. [Google Scholar] [CrossRef]
- Han, X.; Dong, L.; Cao, Y.; Lyu, Y.; Shao, X.; Wang, Y.; Wang, L. Adaptation to Climate Change Effects by Cultivar and Sowing Date Selection for Maize in the Northeast China Plain. Agronomy 2022, 12, 984. [Google Scholar] [CrossRef]
- Tokatlidis, I.S.; Koutroubas, S.D. A review of maize hybrids’ dependence on high plant populations and its implications for crop yield stability. Field Crops Res. 2004, 88, 103–114. [Google Scholar] [CrossRef]
- Duvick, D.N. The Contribution of Breeding to Yield Advances in maize (Zea mays L.). Adv. Agron. 2005, 86, 83–145. [Google Scholar] [CrossRef]
- McMillen, M.S.; Mahama, A.A.; Sibiya, J.; Lübberstedt, T.; Suza, W.P. Improving drought tolerance in maize: Tools and techniques. Front. Genet. 2022, 13, 1001001. [Google Scholar] [CrossRef]
- Santos, T.d.O.; Amaral Junior, A.T.d.; Moulin, M.M. Maize Breeding for Low Nitrogen Inputs in Agriculture: Mechanisms Underlying the Tolerance to the Abiotic Stress. Stresses 2023, 3, 136–152. [Google Scholar] [CrossRef]
- Butts-Wilmsmeyer, C.J.; Seebauer, J.R.; Singleton, L.; Below, F.E. Weather during Key Growth Stages Explains Grain Quality and Yield of Maize. Agronomy 2019, 9, 16. [Google Scholar] [CrossRef]
- Altieri, M.A.; Nicholls, C.I. Soil fertility management and insect pests: Harmonizing soil and plant health in agroecosystems. Soil Tillage Res. 2003, 72, 203–211. [Google Scholar] [CrossRef]
- Munkvold, G. Crop Management Practices to Minimize the Risk of Mycotoxins Contamination in Temperate-Zone Maize. In Mycotoxin Reduction in Grain Chains; Leslie, J.F., Logrieco, A.F., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2014. [Google Scholar] [CrossRef]
- Rajcan, I.; Swanton, C.J. Understanding maize–weed competition: Resource competition, light quality and the whole plant. Field Crops Res. 2001, 71, 139–150. [Google Scholar] [CrossRef]
- Pierre, J.F.; Latournerie-Moreno, L.; Garruña, R.; Jacobsen, K.L.; Laboski, C.A.M.; Us-Santamaría, R.; Ruiz-Sánchez, E. Effect of Maize–Legume Intercropping on Maize Physio-Agronomic Parameters and Beneficial Insect Abundance. Sustainability 2022, 14, 12385. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, L.; Evers, J.; van der Werf, W.; Zhang, W.; Duan, L. Maize yield and quality in response to plant density and application of a novel plant growth regulator. Field Crops Res. 2014, 164, 82–89. [Google Scholar] [CrossRef]
- Jilo, T.; Tulu, L. Association and path coefficient analysis among grain yield and related traits in Ethiopian maize (Zea mays L.) inbred lines. Afr. J. Plant Sci. 2019, 13, 264–272. [Google Scholar] [CrossRef]
- Liu, D.-Y.; Zhang, W.; Liu, Y.-M.; Chen, X.-P.; Zou, C.-Q. Soil Application of Zinc Fertilizer Increases Maize Yield by Enhancing the Kernel Number and Kernel Weight of Inferior Grains. Front. Plant Sci. 2020, 11, 188. [Google Scholar] [CrossRef]
- Wang, F.; Xie, R.; Ming, B.; Wang, K.; Hou, P.; Chen, J.; Liu, G.; Zhang, G.; Xue, J.; Li, S. Dry matter accumulation after silking and kernel weight are the key factors for increasing maize yield and water use efficiency. Agric. Water Manag. 2021, 254, 106938. [Google Scholar] [CrossRef]
- Nurgia, N.; Tanab, T.; Dechassac, N.; Alemayehuc, Y.; Tessoc, B. Effects of planting density and variety on productivity of maize-faba bean intercropping system. Heliyon 2023, 9, e12967. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Messing, J. Proteome balancing of the maize seed for higher nutritional value. Front. Plant Sci. 2014, 5, 240. [Google Scholar] [CrossRef] [PubMed]
- Shiferaw, B.; Prasanna, B.M.; Hellin, J.; Bänziger, M. Crops that feed the world 6. Past successes and future challenges to the role played by maize in global food security. Food Sec. 2011, 3, 307–327. [Google Scholar] [CrossRef]
- Yan, Y.; Hou, P.; Duan, F.; Niu, L.; Dai, T.; Wang, K.; Zhao, M.; Li, S.; Zhou, W. Improving photosynthesis to increase grain yield potential: An analysis of maize hybrids released in different years in China. Photosynth. Res. 2021, 150, 295–311. [Google Scholar] [CrossRef]
- Mogesse, W.; Zeleke, H. Estimates of Combining Ability and Association among Morpho-Agronomic Traits of Single Cross Maize (Zea mays L.) Hybrids. J. Agric. Prod. 2022, 3, 78–87. [Google Scholar] [CrossRef]
- Ren, H.; Liu, M.; Zhang, J.; Liu, P.; Liu, C. Effects of agronomic traits and climatic factors on yield and yield stability of summer maize (Zea mays L) in the Huang-Huai-Hai Plain in China. Front. Plant Sci. 2022, 13, 1050064. [Google Scholar] [CrossRef]
- Mosisa, W.; Dechassa, N.; Kibret, K.; Zeleke, H.; Bekeko, Z. Response of Maize (Zea mays L.) Varieties to Rates of Mineral Nitrogen Fertilizer Application in Haramaya District, Eastern Highlands of Ethiopia. East Afr. J. Sci. 2023, 17, 1–18. [Google Scholar]
- Liu, Y.; Hou, P.; Zhang, W.; Xing, J.; Lv, T.; Zhang, C.; Wang, R.; Zhao, J. Drought resistance of nine maize cultivars released from the 1970s through the 2010s in China. Field Crops Res. 2023, 302, 109065. [Google Scholar] [CrossRef]
- Karayel, D.; Barut, Z.B.; Özmerzi, A. Mathematical Modelling of Vacuum Pressure on a Precision Seeder. Biosyst. Eng. 2004, 87, 437–444. [Google Scholar] [CrossRef]
- Mousavi, S.M.N.; Illés, Á.; Bojtor, C.; Nagy, J. The Impact of Different Nutritional Treatments on Maize Hybrids Morphological Traits Based on Stability Statistical Methods. Emir. J. Food Agric. 2020, 32, 666–672. [Google Scholar] [CrossRef]
- Kang, M.S.; Gorman, D.P. Genotype × Environment Interaction in Maize. Agron. J. 1989, 81, 662–664. [Google Scholar] [CrossRef]
- Crossa, J.; Vargas, M.; van Eeuwijk, F.; Jiang, C.; Edmeades, G.O.; Hoisington, D. Interpreting genotype × environment interaction in tropical maize using linked molecular markers and environmental covariables. Theor. Appl. Genet. 1999, 99, 611–625. [Google Scholar] [CrossRef]
- Signor, C.E.-L.; Dousse, S.; Lorgeou, J.; Denis, J.-B.; Bonhomme, R.; Carolo, P.; Charcosset, A. Interpretation of Genotype × Environment Interactions for Early Maize Hybrids over 12 Years. Crop Sci. 2001, 41, 663–669. [Google Scholar] [CrossRef]
- Singamsetti, A.; Shahi, J.P.; Zaidi, P.H.; Seetharam, K.; Vinayan, M.T.; Kumar, M.; Singla, S.; Shikha, K.; Madankar, K. Genotype × environment interaction and selection of maize (Zea mays L.) hybrids across moisture regimes. Field Crops Res. 2021, 270, 108224. [Google Scholar] [CrossRef]
- Mahajan, V.S.; Khehra, A.S. Stability analysis of kernel yield and its components in maize (Zea mays L.) in winter and monsoon seasons. Indian J. Genet. Plant Breed. 1992, 52, 63–67. [Google Scholar]
- Greveniotis, V.; Bouloumpasi, E.; Zotis, S.; Korkovelos, A.; Ipsilandis, C.G. Estimations on Trait Stability of Maize Genotypes. Agriculture 2021, 11, 952. [Google Scholar] [CrossRef]
- Mousavi, S.M.N.; Bojtor, C.; Illés, Á.; Nagy, J. Genotype by Trait Interaction (GT) in Maize Hybrids on Complete Fertilizer. Plants 2021, 10, 2388. [Google Scholar] [CrossRef]
- Gouesnard, B.; Zanetto, A.; Welcker, C. Identification of adaptation traits to drought in collections of maize landraces from southern Europe and temperate regions. Euphytica 2016, 209, 565–584. [Google Scholar] [CrossRef]
- Nagore, M.L.; Della Maggiora, A.; Andrade, F.H.; Echarte, L. Water use efficiency for grain yield in an old and two more recent maize hybrids. Field Crops Res. 2017, 214, 185–193. [Google Scholar] [CrossRef]
- Gauch, H.G. Model selection and validation for yield trials with interaction. Biometrics 1988, 44, 705–715. [Google Scholar] [CrossRef]
- Piepho, H.P. Methods for comparing the yield stability of cropping systems-a review. J. Agron. Crop Sci. 1998, 180, 193–213. [Google Scholar] [CrossRef]
- Mekbib, F. Yield stability in common bean (Phaseolus vulgaris L.) genotypes. Euphytica 2003, 130, 147–153. [Google Scholar] [CrossRef]
- Pacheco, R.M.; Duarte, J.B.; Vencovsky, R.; Pinheiro, J.B.; Oliveira, A.B. Use of supplementary genotypes in AMMI analysis. Theor. Appl. Genet. 2005, 110, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Motzo, R.; Giunta, F.; Deidda, M. Factors affecting the genotype × environment interaction in spring triticale grown in a Mediterranean environment. Euphytica 2001, 121, 317–324. [Google Scholar] [CrossRef]
- Yan, W.; Hunt, L.A. Interpretation of genotype × environment interaction for winter wheat yield in Ontario. Crop Sci. 2001, 41, 19–25. [Google Scholar] [CrossRef]
- Brancourt-Hulmel, M.; Lecomte, C. Effect of environmental varieties on genotype x environment interaction of winter wheat. A comparison of biadditive factorial regression to AMMI. Crop Sci. 2003, 43, 608–617. [Google Scholar] [CrossRef]
- Sinebo, W. Trade off between yield increase and yield stability in three decades of barley breeding in a tropical highland environment. Field Crops Res. 2005, 92, 35–52. [Google Scholar] [CrossRef]
- Rodriguez, M.; Rau, D.; Papa, R.; Attene, G. Genotype by environment interactions in barley (Hordeum vulgare L.): Different responses of landraces, recombinant inbred lines and varieties to Mediterranean environment. Euphytica 2008, 163, 231–247. [Google Scholar] [CrossRef]
- Balestre, M.; Von Pinho, R.G.; Souza, J.C.; Oliveira, R.L. Genotypic stability and adaptability in tropical maize based on AMMI and GGE biplot analysis. Genet. Mol. Res. 2009, 8, 1311–1322. [Google Scholar] [CrossRef]
- Pires, L.P.; Garcia Von Pinho, R.; Balestre, M. AMMI Bayesian Models to Study Stability and Adaptability in Maize. Agron. J. 2018, 110, 1765–1776. [Google Scholar] [CrossRef]
- Romagosa, I.; Fox, P.N. Genotype × environment interaction and adaptation. In Plant Breeding; Hayward, M.D., Bosemark, N.O., Romagosa, I., Cerezo, M., Eds.; Plant Breeding Series; Springer: Dordrecht, The Netherlands, 1993. [Google Scholar] [CrossRef]
- Nowosad, K.; Liersch, A.; Popławska, W.; Bocianowski, J. Genotype by environment interaction for seed yield in rapeseed (Brassica napus L.) using additive main effects and multiplicative interaction model. Euphytica 2016, 208, 187–194. [Google Scholar] [CrossRef]
- Mandel, J. A new analysis of variance model for non-additive data. Technometrics 1971, 13, 1–8. [Google Scholar] [CrossRef]
- Mandel, J. Non-additivity in two-way analysis of variance. J. Am. Stat. Assoc. 1961, 56, 878–888. [Google Scholar] [CrossRef]
- Mandel, J. The partitioning of interaction in analysis of variance. J. Res. Nat. Bureau. Stand. Sect. B 1969, 73, 309–328. [Google Scholar] [CrossRef]
- Gollob, H.F. A statistical model which combines features of factor analytic and analysis of variance techniques. Psychometrika 1968, 33, 73–155. [Google Scholar] [CrossRef] [PubMed]
- Cornelius, P.L. Statistical tests and retention of terms in the additive main effects and multiplicative interaction model for cultivar trials. Crop Sci. 1993, 33, 1186–1193. [Google Scholar] [CrossRef]
- Prigge, V.; Melchinger, A.E. Production of haploids and doubled haploids in maize. In Plant Cell Culture Protocols; Loyola-Vargas, V., Ochoa-Alejo, N., Eds.; Methods in Molecular Biology; Humana Press: Totowa, NJ, USA, 2012; Volume 877, pp. 161–172. [Google Scholar] [CrossRef]
- Gauch, H.G.; Zobel, R.W. Imputing missing yield trial data. Theor. Appl. Genet. 1990, 79, 753–761. [Google Scholar] [CrossRef]
- Purchase, J.L.; Hatting, H.; van Deventer, C.S. Genotype × environment interaction of winter wheat (Triticum aestivum L.) in South Africa: II. Stability analysis of yield performance. S. Afr. J. Plant Soil 2000, 17, 101–107. [Google Scholar] [CrossRef]
- Farshadfar, E.; Sutka, J. Locating QTLs controlling adaptation in wheat using AMMI model. Cereal Res. Commun. 2003, 31, 249–256. [Google Scholar] [CrossRef]
- Choo, T.M.; Reinbergs, E. Estimation of the number of genes in doubled haploid populations of barley (Hordeum vulgare). Can. J. Genet. Cytol. 1982, 24, 337–341. [Google Scholar] [CrossRef]
- Bocianowski, J.; Krajewski, P. Comparison of the genetic additive effect estimators based on phenotypic observations and on molecular marker data. Euphytica 2009, 165, 113–122. [Google Scholar] [CrossRef]
- Bocianowski, J. Analytical and numerical comparisons of two methods of estimation of additive × additive interaction of QTL effects. Sci. Agric. 2012, 69, 240–246. [Google Scholar] [CrossRef]
- VSN International Genstat for Windows, 23rd ed.; VSN International: Hemel Hempstead, UK, 2023.
- Peng, B.; Li, Y.; Wang, Y.; Liu, C.; Liu, Z.; Tan, W.; Zhang, Y.; Wang, D.; Shi, Y.; Sun, B.; et al. QTL analysis for yield components and kernel-related traits in maize across multi-environments. Theor. Appl. Genet. 2011, 122, 1305–1320. [Google Scholar] [CrossRef]
- Bocianowski, J.; Nowosad, K.; Tomkowiak, A. Genotype—Environment interaction for seed yield of maize hybrids and lines using the AMMI model. Maydica 2019, 64, M13. [Google Scholar]
- Mousavi, S.M.N.; Illés, A.; Szabó, A.; Shojaei, S.H.; Demeter, C.; Bakos, Z.; Vad, A.; Széles, A.; Nagy, J.; Bojtor, C. Stability yield indices on different sweet corn hybrids based on AMMI analysis. Braz. J. Biol. 2024, 84, e270680. [Google Scholar] [CrossRef] [PubMed]
- Matin, M.Q.I.; Rasul, M.G.; Islam, A.K.M.M.; Mian, M.A.K.; Ahmed, J.U.; Amiruzzaman, M. Stability analysis for yield and yield contributing characters in hybrid maize (Zea mays L.). Afr. J. Agric. Res. 2017, 12, 2795–2806. [Google Scholar] [CrossRef][Green Version]
- Bocianowski, J.; Tratwal, A.; Nowosad, K. Genotype by environment interaction for area under the disease-progress curve (AUDPC) value in spring barley using additive main effects and multiplicative interaction model. Australas. Plant Pathol. 2020, 49, 525–529. [Google Scholar] [CrossRef]
- Bocianowski, J.; Księżak, J.; Nowosad, K. Genotype by environment interaction for seeds yield in pea (Pisum sativum L.) using additive main effects and multiplicative interaction model. Euphytica 2019, 215, 191. [Google Scholar] [CrossRef]
- Presterl, T.; Seitz, G.; Landbeck, M.; Thiemt, E.M.; Schmidt, W.; Geiger, H.H. Improving Nitrogen-Use Efficiency in European Maize. Crop Sci. 2003, 43, 1259–1265. [Google Scholar] [CrossRef]
- Sang, Z.; Wang, H.; Yang, Y.; Zhang, Z.; Liu, X.; Li, Z.; Xu, Y. Epistasis Activation Contributes Substantially to Heterosis in Temperate by Tropical Maize Hybrids. Front. Plant Sci. 2022, 13, 921608. [Google Scholar] [CrossRef]
- Cyplik, A.; Sobiech, A.; Tomkowiak, A.; Bocianowski, J. Genetic Parameters for Selected Traits of Inbred Lines of Maize (Zea mays L.). Appl. Sci. 2022, 12, 6961. [Google Scholar] [CrossRef]
- Rio, S.; Charcosset, A.; Moreau, L.; Mary-Huard, T. Detecting directional and non-directional epistasis in bi-parental populations using genomic data. Genetics 2023, 224, iyad089. [Google Scholar] [CrossRef] [PubMed]
- Crow, J.F. On epistasis: Why it is unimportant in polygenic directional selection. Philos. Trans. R. Soc. B 2010, 365, 1241–1244. [Google Scholar] [CrossRef]
- Matsubara, K.; Yamamoto, E.; Mizobuchi, R.; Yonemaru, J.; Yamamoto, T.; Kato, H.; Yano, M. Hybrid breakdown caused by epistasisbased recessive incompatibility in a cross of rice (Oryza sativa L.). J. Hered. 2015, 106, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, V.; Gahlaut, V.; Meher, P.K.; Mir, R.R.; Jaiswal, J.P.; Rao, A.R.; Balyan, H.S.; Gupta, R.R. Genome wide single locus single trait, multilocus and multi-trait association mapping for some important agronomic traits in common wheat (T. aestivum L.). PLoS ONE 2016, 11, e0159343. [Google Scholar] [CrossRef] [PubMed]

| Year | April | May | June | July | August | September | October | November |
|---|---|---|---|---|---|---|---|---|
| 2013 | 32 | 89 | 108 | 47 | 42 | 111 | 13 | 26 |
| 2014 | 42 | 86 | 30 | 90 | 83 | 128 | 46 | 19 |
| 2015 | 27 | 35 | 58 | 41 | 14 | 38 | 39 | 54 |
| 2016 | 38 | 50 | 60 | 97 | 25 | 45 | 81 | 40 |
| 2017 | 47 | 27 | 80 | 83 | 69 | 70 | 78 | 37 |
| 2018 | 17 | 62 | 81 | 64 | 12 | 62 | 34 | 8 |
| 2019 | 14 | 56 | 19 | 44 | 40 | 55 | 17 | 38 |
| 2020 | 11 | 75 | 159 | 55 | 69 | 72 | 84 | 19 |
| 2021 | 43 | 84 | 39 | 101 | 95 | 17 | 10 | 29 |
| 2022 | 42 | 31 | 69 | 59 | 123 | 69 | 23 | 24 |
| Year | April | May | June | July | August | September | October | November |
|---|---|---|---|---|---|---|---|---|
| 2013 | 9.1 | 14.6 | 17.4 | 20.4 | 18.9 | 12.5 | 10.5 | 5.2 |
| 2014 | 10.8 | 13.5 | 16.4 | 21.3 | 18.4 | 15.9 | 11.7 | 6.8 |
| 2015 | 8.8 | 13.7 | 16.6 | 20.7 | 20.9 | 14.9 | 8.4 | 7.2 |
| 2016 | 9.1 | 14.9 | 18.8 | 19.8 | 18.3 | 16.8 | 8.3 | 3.9 |
| 2017 | 8.4 | 14.3 | 18.7 | 19.0 | 19.6 | 13.6 | 11.3 | 5.7 |
| 2018 | 14.0 | 16.5 | 18.5 | 19.9 | 21.5 | 16.3 | 11.7 | 5.5 |
| 2019 | 10.2 | 11.8 | 21.8 | 19.3 | 20.8 | 14.9 | 11.9 | 7.2 |
| 2020 | 9.8 | 12.1 | 17.7 | 18.7 | 20.5 | 16.0 | 11.3 | 6.3 |
| 2021 | 6.8 | 12.5 | 19.5 | 20.4 | 18.0 | 15.5 | 10.7 | 5.6 |
| 2022 | 7.5 | 14.9 | 19.5 | 19.2 | 20.3 | 13.0 | 12.6 | 5.4 |
| Year | Plant Height (cm) | Day to Flowering (Days) | Grain Yield (t ha–1) | |||
|---|---|---|---|---|---|---|
| Min | Max | Min | Max | Min | Max | |
| 2013 | 130 | 274 | 84.5 | 92.7 | 2.66 | 4.21 |
| 2014 | 149 | 269 | 88.5 | 92.5 | 4.40 | 6.07 |
| 2015 | 127 | 281 | 85.6 | 97.0 | 4.59 | 6.17 |
| 2016 | 125 | 277 | 85.1 | 94.5 | 3.50 | 5.97 |
| 2017 | 150 | 246 | 77.6 | 90.5 | 2.36 | 4.77 |
| 2018 | 135 | 273 | 88.2 | 93.1 | 1.66 | 5.71 |
| 2019 | 161 | 266 | 89.3 | 93.6 | 3.10 | 7.73 |
| 2020 | 145 | 257 | 89.7 | 94.4 | 4.22 | 5.89 |
| 2021 | 195 | 264 | 90.1 | 92.5 | 4.23 | 5.88 |
| 2022 | 166 | 268 | 89.5 | 94.9 | 3.90 | 6.01 |
| Source of Variation | d.f. | Sum of Squares | Mean Squares | F-Statistic | Variability Explained (%) |
|---|---|---|---|---|---|
| Total | 1039 | 1,368,792 | 1317 | ||
| Treatments | 259 | 652,863 | 2521 | 3.38 *** | 47.70 |
| Genotypes, G | 25 | 132,656 | 5306 | 7.12 *** | 9.69 |
| Environments, E | 9 | 192,945 | 21,438 | 28.76 *** | 14.10 |
| Block/Environment | 30 | 20,748 | 692 | 0.93 | 1.52 |
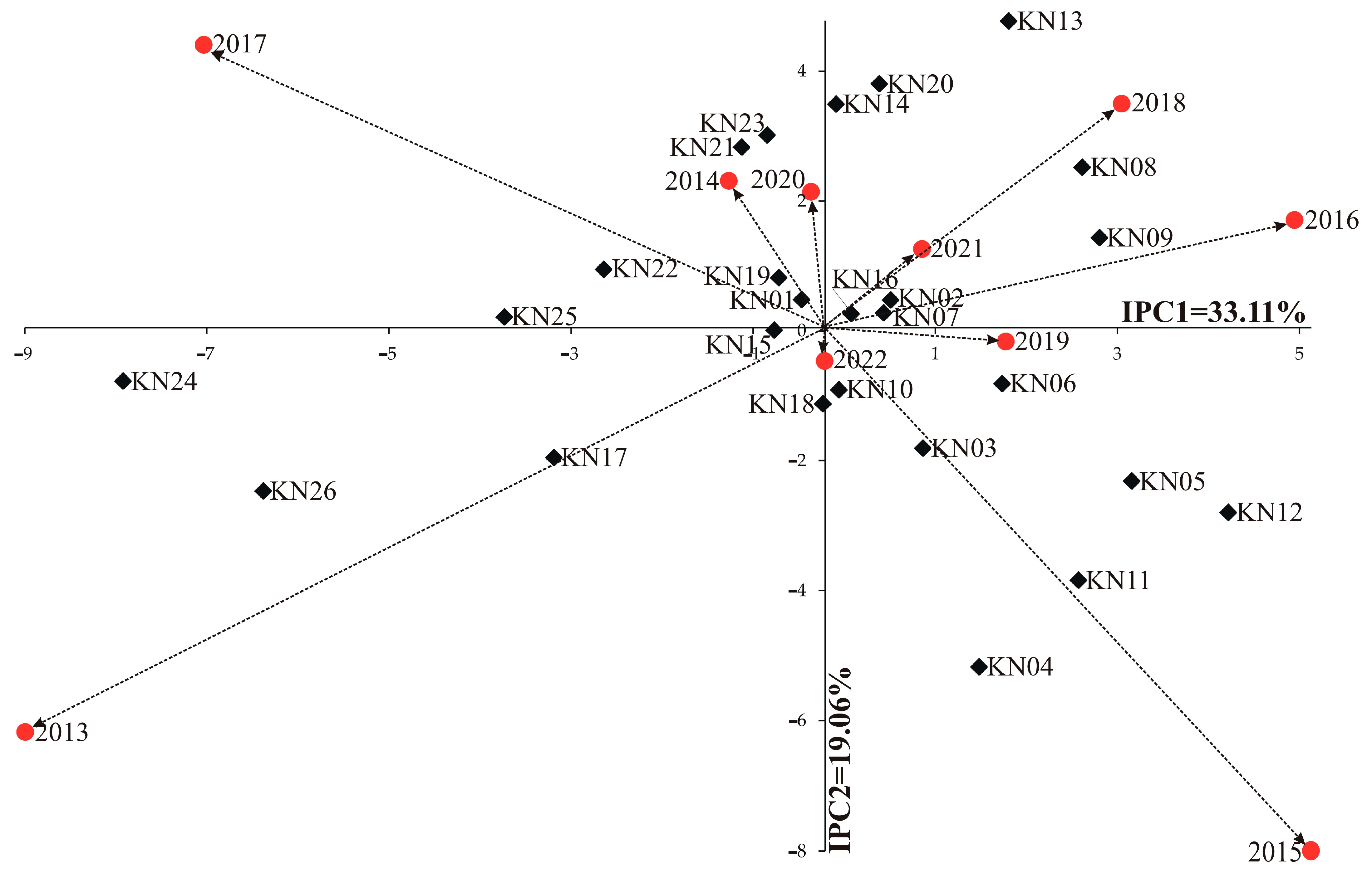
| GEIs | 225 | 462,458 | 2055 | 2.76 *** | 33.79 |
| IPCA 1 | 33 | 153,121 | 4640 | 6.23 *** | 33.11 |
| IPCA 2 | 31 | 88,129 | 2843 | 3.81 *** | 19.06 |
| IPCA 3 | 29 | 75,927 | 2618 | 3.51 *** | 16.42 |
| Residuals | 132 | 145,281 | 1101 | 1.48 *** | 31.41 |
| Error | 750 | 558,985 | 745 |
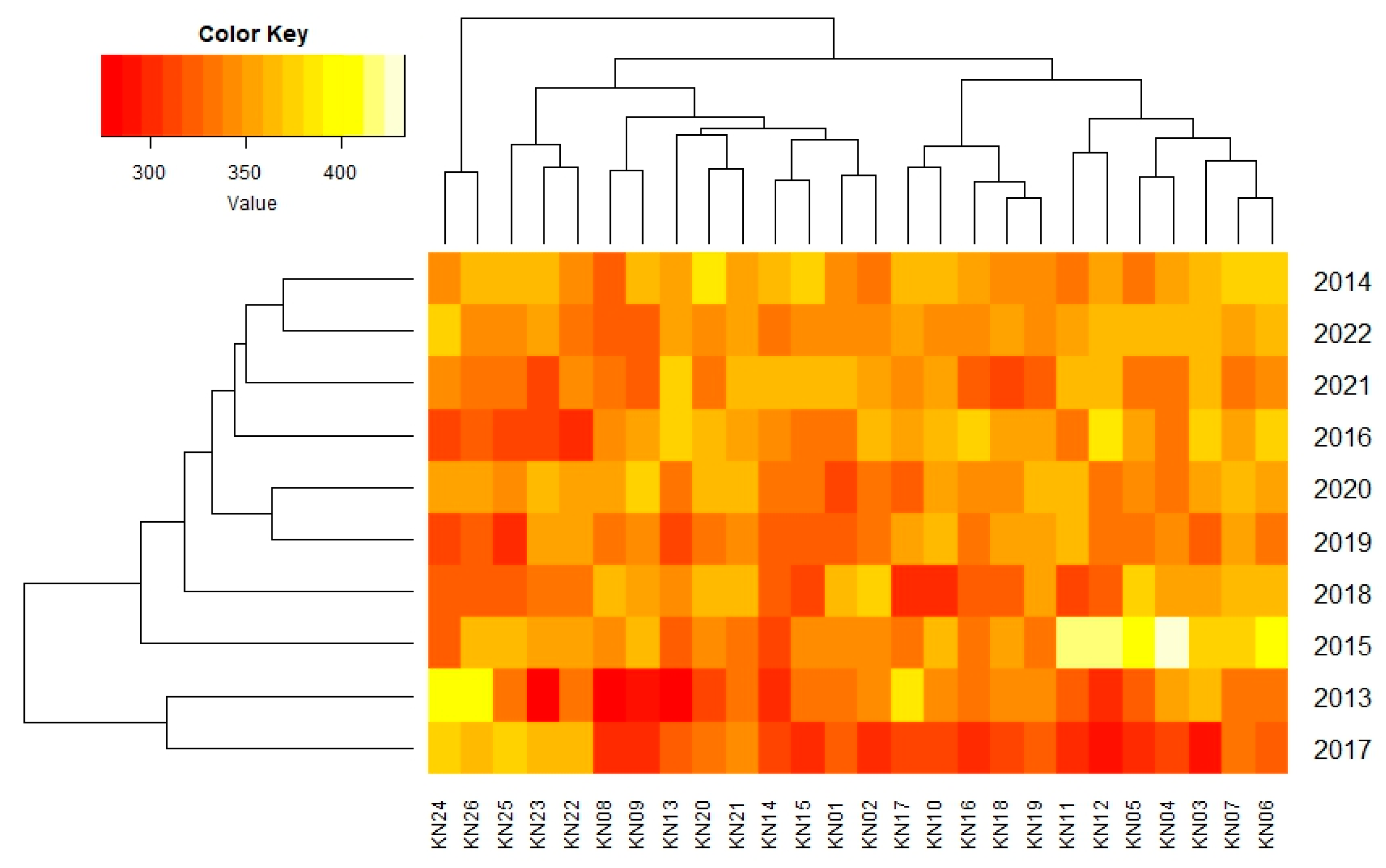
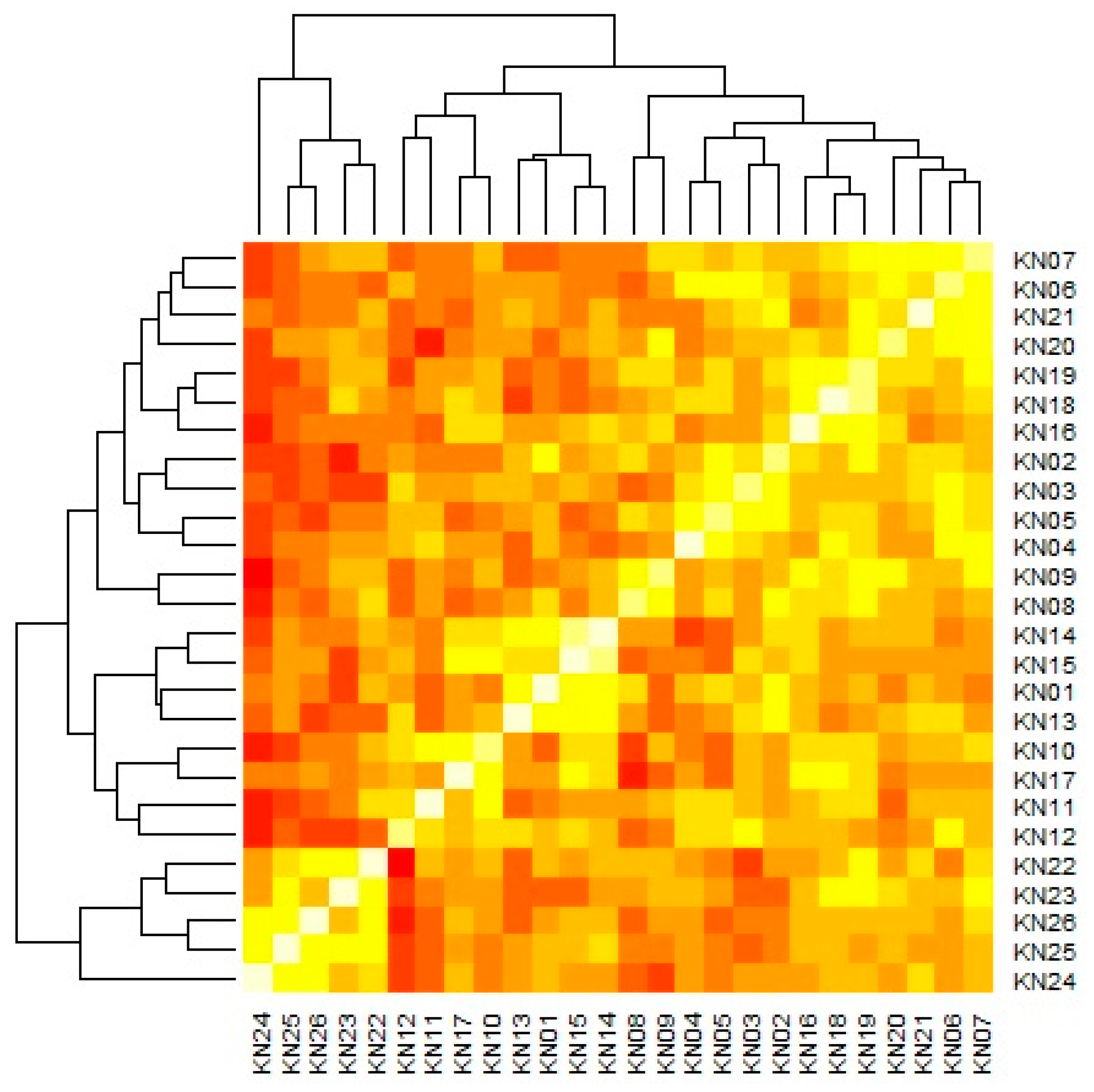
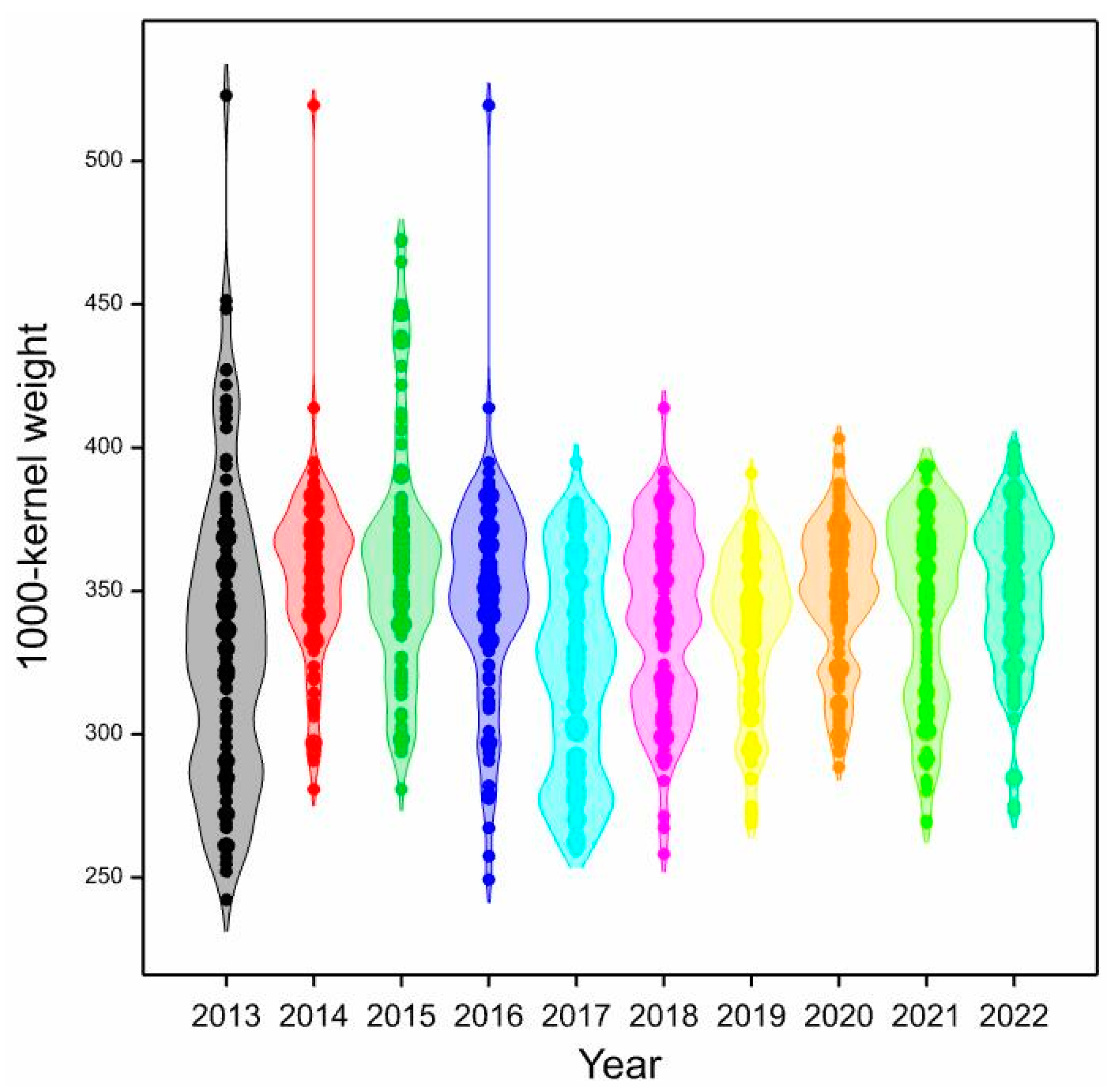
| Genotype | Years | IPCA g1 | IPCA g2 | IPCA g3 | RM | ASV | RA | GSI | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2013 | 2014 | 2015 | 2016 | 2017 | 2018 | 2019 | 2020 | 2021 | 2022 | Mean | ||||||||
| KN01 | 328.8 | 339.6 | 348.2 | 327.9 | 321.1 | 359.4 | 322.6 | 312.3 | 362.1 | 343.3 | 336.5 | −0.256 | 0.428 | 0.347 | 23 | 0.617 | 2 | 25 |
| KN02 | 345.1 | 336.3 | 340.1 | 363.5 | 299.0 | 371.8 | 329.1 | 337.1 | 352.6 | 346.4 | 342.1 | 0.723 | 0.423 | 2.499 | 15 | 1.325 | 8 | 23 |
| KN03 | 359.7 | 363.8 | 370.1 | 370.3 | 286.3 | 356.0 | 324.5 | 352.4 | 359.7 | 368.5 | 351.1 | 1.076 | −1.863 | 3.043 | 5 | 2.639 | 9 | 14 |
| KN04 | 349.7 | 353.2 | 433.0 | 333.2 | 306.8 | 356.6 | 344.9 | 337.9 | 334.3 | 363.7 | 351.3 | 1.693 | −5.229 | −2.697 | 4 | 6.000 | 20 | 24 |
| KN05 | 320.8 | 333.0 | 406.2 | 352.2 | 297.7 | 373.1 | 335.1 | 347.1 | 337.8 | 362.0 | 346.5 | 3.371 | −2.367 | −1.779 | 10 | 6.317 | 23 | 33 |
| KN06 | 332.9 | 371.3 | 398.8 | 370.3 | 325.3 | 369.5 | 337.8 | 350.1 | 345.6 | 363.2 | 356.5 | 1.946 | −0.867 | −0.817 | 1 | 3.491 | 13 | 14 |
| KN07 | 335.1 | 374.5 | 377.3 | 355.1 | 329.5 | 363.0 | 349.9 | 366.7 | 335.7 | 354.6 | 354.1 | 0.645 | 0.224 | −1.390 | 2 | 1.142 | 5 | 7 |
| KN08 | 282.9 | 322.7 | 345.4 | 340.6 | 302.6 | 367.3 | 328.9 | 354.9 | 336.5 | 327.2 | 330.9 | 2.827 | 2.468 | −1.587 | 26 | 5.497 | 17 | 43 |
| KN09 | 295.1 | 364.8 | 368.9 | 355.7 | 302.8 | 356.1 | 347.4 | 372.2 | 326.8 | 324.6 | 341.4 | 3.016 | 1.383 | −1.681 | 17 | 5.420 | 16 | 33 |
| KN10 | 339.1 | 365.5 | 360.4 | 369.1 | 314.0 | 298.2 | 363.1 | 352.8 | 359.1 | 345.3 | 346.6 | 0.153 | −0.961 | 2.377 | 9 | 0.998 | 4 | 13 |
| KN11 | 317.6 | 336.2 | 417.7 | 336.7 | 299.2 | 312.1 | 365.7 | 364.3 | 365.0 | 349.6 | 346.4 | 2.784 | −3.895 | −1.841 | 11 | 6.211 | 22 | 33 |
| KN12 | 305.9 | 353.6 | 415.1 | 387.5 | 295.5 | 318.2 | 334.2 | 331.6 | 367.1 | 366.3 | 347.5 | 4.431 | −2.853 | 1.398 | 8 | 8.211 | 24 | 32 |
| KN13 | 275.1 | 356.8 | 325.2 | 374.0 | 327.5 | 338.8 | 311.5 | 330.4 | 373.1 | 358.2 | 337.1 | 2.019 | 4.722 | 2.115 | 21 | 5.882 | 19 | 40 |
| KN14 | 297.6 | 367.1 | 315.6 | 343.8 | 316.3 | 322.8 | 327.2 | 334.2 | 363.6 | 337.6 | 332.6 | 0.122 | 3.441 | 1.950 | 25 | 3.448 | 12 | 37 |
| KN15 | 334.2 | 380.2 | 339.9 | 336.3 | 296.8 | 317.1 | 322.6 | 328.9 | 368.6 | 341.8 | 336.6 | −0.556 | −0.042 | 2.924 | 22 | 0.967 | 3 | 25 |
| KN16 | 333.4 | 352.2 | 337.6 | 374.6 | 302.1 | 321.0 | 332.1 | 344.9 | 322.3 | 339.7 | 336.0 | 0.289 | 0.209 | 2.507 | 24 | 0.544 | 1 | 25 |
| KN17 | 382.2 | 364.8 | 332.0 | 356.5 | 311.6 | 296.3 | 355.8 | 327.0 | 343.1 | 353.1 | 342.2 | −2.976 | −2.006 | 4.348 | 14 | 5.547 | 18 | 32 |
| KN18 | 339.0 | 341.6 | 355.2 | 350.4 | 308.7 | 327.4 | 352.6 | 348.6 | 314.7 | 358.1 | 339.6 | −0.021 | −1.177 | 0.172 | 18 | 1.178 | 7 | 25 |
| KN19 | 344.8 | 341.3 | 336.1 | 357.1 | 318.2 | 349.5 | 354.9 | 359.5 | 326.7 | 347.8 | 343.6 | −0.507 | 0.766 | 0.716 | 13 | 1.167 | 6 | 19 |
| KN20 | 310.8 | 382.1 | 340.1 | 369.7 | 337.6 | 364.3 | 333.4 | 363.9 | 328.0 | 347.6 | 347.7 | 0.597 | 3.753 | −0.373 | 7 | 3.894 | 14 | 21 |
| KN21 | 336.5 | 351.2 | 336.2 | 352.2 | 345.1 | 365.2 | 341.9 | 368.0 | 366.3 | 358.5 | 352.1 | −0.914 | 2.776 | 0.201 | 3 | 3.199 | 11 | 14 |
| KN22 | 329.9 | 348.4 | 354.2 | 305.0 | 359.9 | 331.9 | 353.6 | 357.6 | 342.6 | 334.5 | 341.8 | −2.429 | 0.893 | −3.497 | 16 | 4.314 | 15 | 31 |
| KN23 | 284.7 | 363.4 | 350.4 | 312.2 | 359.4 | 327.8 | 352.1 | 363.5 | 312.3 | 356.9 | 338.3 | −0.634 | 2.961 | −4.964 | 20 | 3.160 | 10 | 30 |
| KN24 | 411.0 | 347.7 | 324.9 | 308.9 | 370.7 | 325.3 | 307.2 | 352.8 | 344.1 | 370.0 | 346.3 | −7.709 | −0.828 | 0.307 | 12 | 13.419 | 26 | 38 |
| KN25 | 334.0 | 365.4 | 366.2 | 307.6 | 371.8 | 318.7 | 301.4 | 344.7 | 331.6 | 348.6 | 339.0 | −3.521 | 0.161 | −3.538 | 19 | 6.120 | 21 | 40 |
| KN26 | 410.7 | 368.4 | 363.9 | 322.4 | 365.9 | 323.6 | 318.9 | 359.2 | 337.3 | 339.1 | 350.9 | −6.169 | −2.521 | −0.739 | 6 | 11.011 | 25 | 31 |
| Mean | 332.2 | 355.6 | 360.0 | 347.4 | 322.0 | 339.7 | 336.5 | 348.6 | 344.5 | 350.2 | 343.7 | |||||||
| IPCA e1 | −8.782 | −1.056 | 5.339 | 5.159 | −6.822 | 3.260 | 1.989 | −0.153 | 1.068 | −0.002 | ||||||||
| IPCA e2 | −6.232 | 2.260 | −8.047 | 1.655 | 4.354 | 3.449 | −0.218 | 2.089 | 1.207 | −0.517 | ||||||||
| IPCA e3 | 3.851 | 1.136 | −5.213 | 6.259 | −5.027 | −2.194 | −0.649 | −2.879 | 3.999 | 0.718 | ||||||||
| Year | The Total Additive Effect, a | The Total Additive-by-Additive Interaction Effect, aa |
|---|---|---|
| 2013 | 67.95 *** | 10.85 *** |
| 2014 | 29.70 * | –3.20 |
| 2015 | 58.70 *** | 14.30 *** |
| 2016 | 41.25 * | –1.15 |
| 2017 | 42.75 ** | 7.05 ** |
| 2018 | 38.40 * | –5.00 * |
| 2019 | 32.15 * | –2.95 |
| 2020 | 29.95 * | –6.35 * |
| 2021 | 30.40 * | –1.80 |
| 2022 | 22.70 | –2.90 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nowosad, K.; Bocianowski, J.; Kianersi, F.; Pour-Aboughadareh, A. Analysis of Linkage on Interaction of Main Aspects (Genotype by Environment Interaction, Stability and Genetic Parameters) of 1000 Kernels in Maize (Zea mays L.). Agriculture 2023, 13, 2005. https://doi.org/10.3390/agriculture13102005
Nowosad K, Bocianowski J, Kianersi F, Pour-Aboughadareh A. Analysis of Linkage on Interaction of Main Aspects (Genotype by Environment Interaction, Stability and Genetic Parameters) of 1000 Kernels in Maize (Zea mays L.). Agriculture. 2023; 13(10):2005. https://doi.org/10.3390/agriculture13102005
Chicago/Turabian StyleNowosad, Kamila, Jan Bocianowski, Farzad Kianersi, and Alireza Pour-Aboughadareh. 2023. "Analysis of Linkage on Interaction of Main Aspects (Genotype by Environment Interaction, Stability and Genetic Parameters) of 1000 Kernels in Maize (Zea mays L.)" Agriculture 13, no. 10: 2005. https://doi.org/10.3390/agriculture13102005
APA StyleNowosad, K., Bocianowski, J., Kianersi, F., & Pour-Aboughadareh, A. (2023). Analysis of Linkage on Interaction of Main Aspects (Genotype by Environment Interaction, Stability and Genetic Parameters) of 1000 Kernels in Maize (Zea mays L.). Agriculture, 13(10), 2005. https://doi.org/10.3390/agriculture13102005







