Sustainable Utilization Strategy of Organic Waste via Fabrication of Bioelastomer with Antibacterial and Antioxidant Activities Using Mandarin Peel Extracts
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Mandarin Peel Extracts
2.3. Fabrication of Bioelastomer
2.4. Antioxidant Activity of Bioelastomer
2.4.1. DPPH Radical Scavenging Activity
2.4.2. ABTS Radical Scavenging Activity
2.5. Antibacterial Activity of Bioelastomer
2.6. Characterization of Bioelastomer
2.7. Analytical Methods
3. Results and Discussion
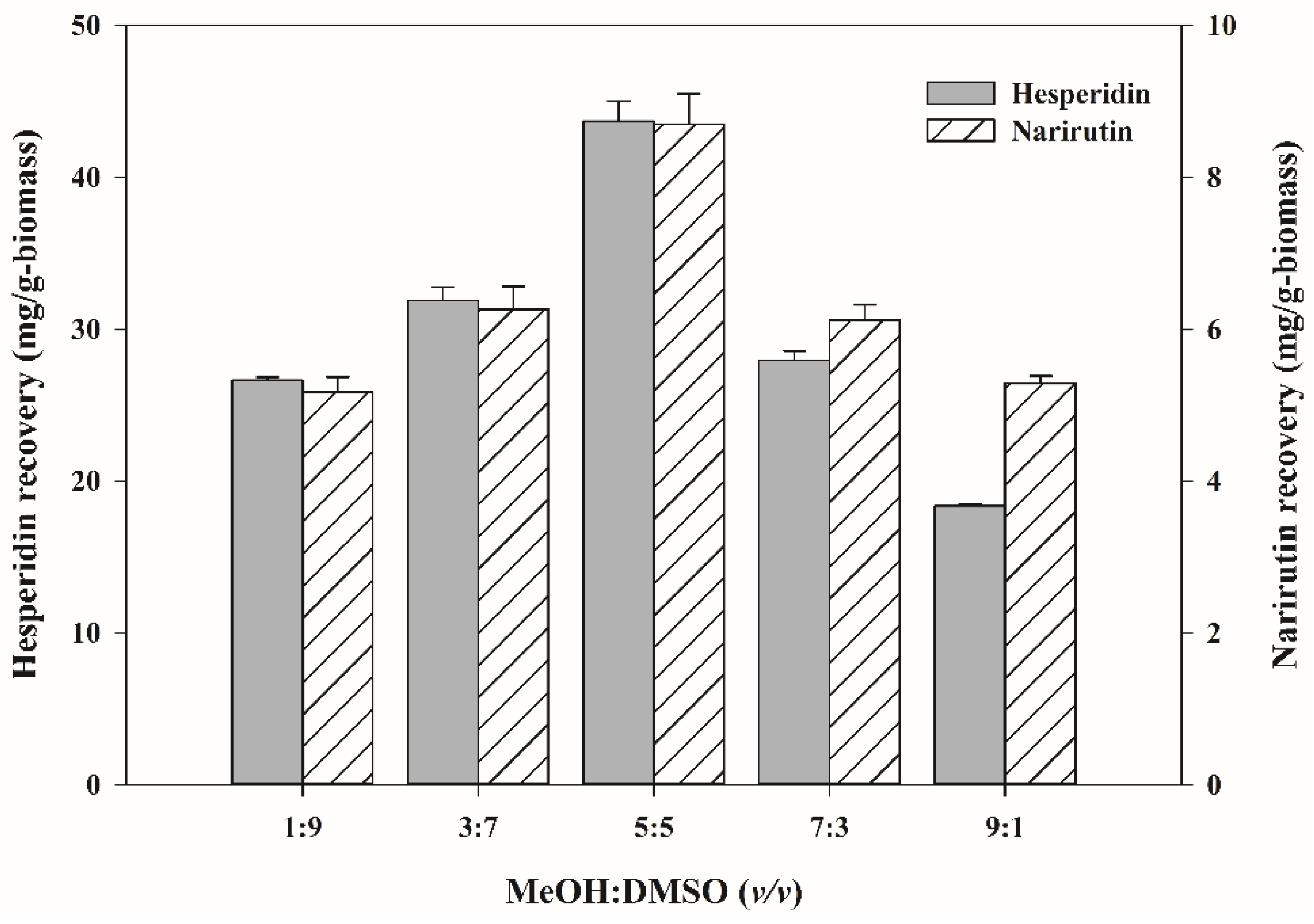
3.1. Selection of Extraction Solvent for Flavanone Recovery from Mandarin Peels
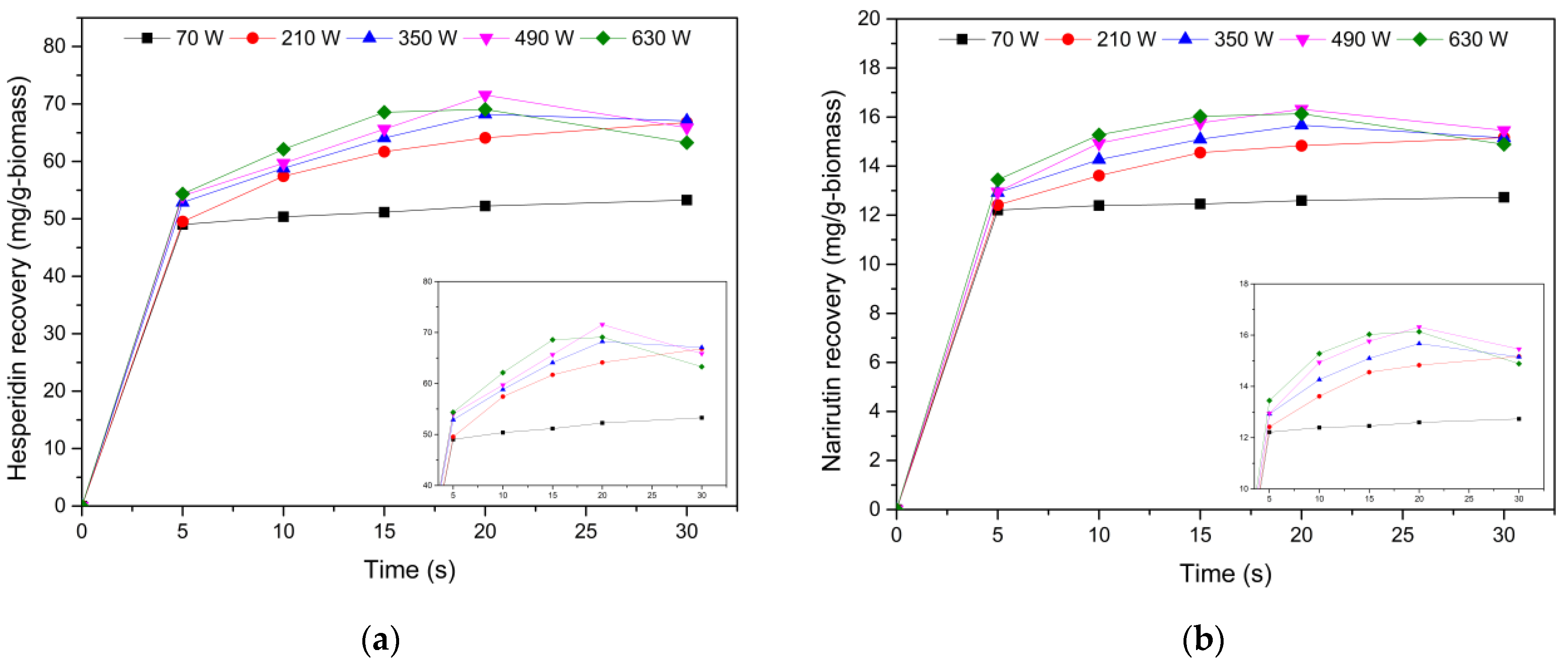
3.2. Determination of Microwave-Assisted Extraction Conditions
3.3. Antioxidant Activity of Bioelastomer
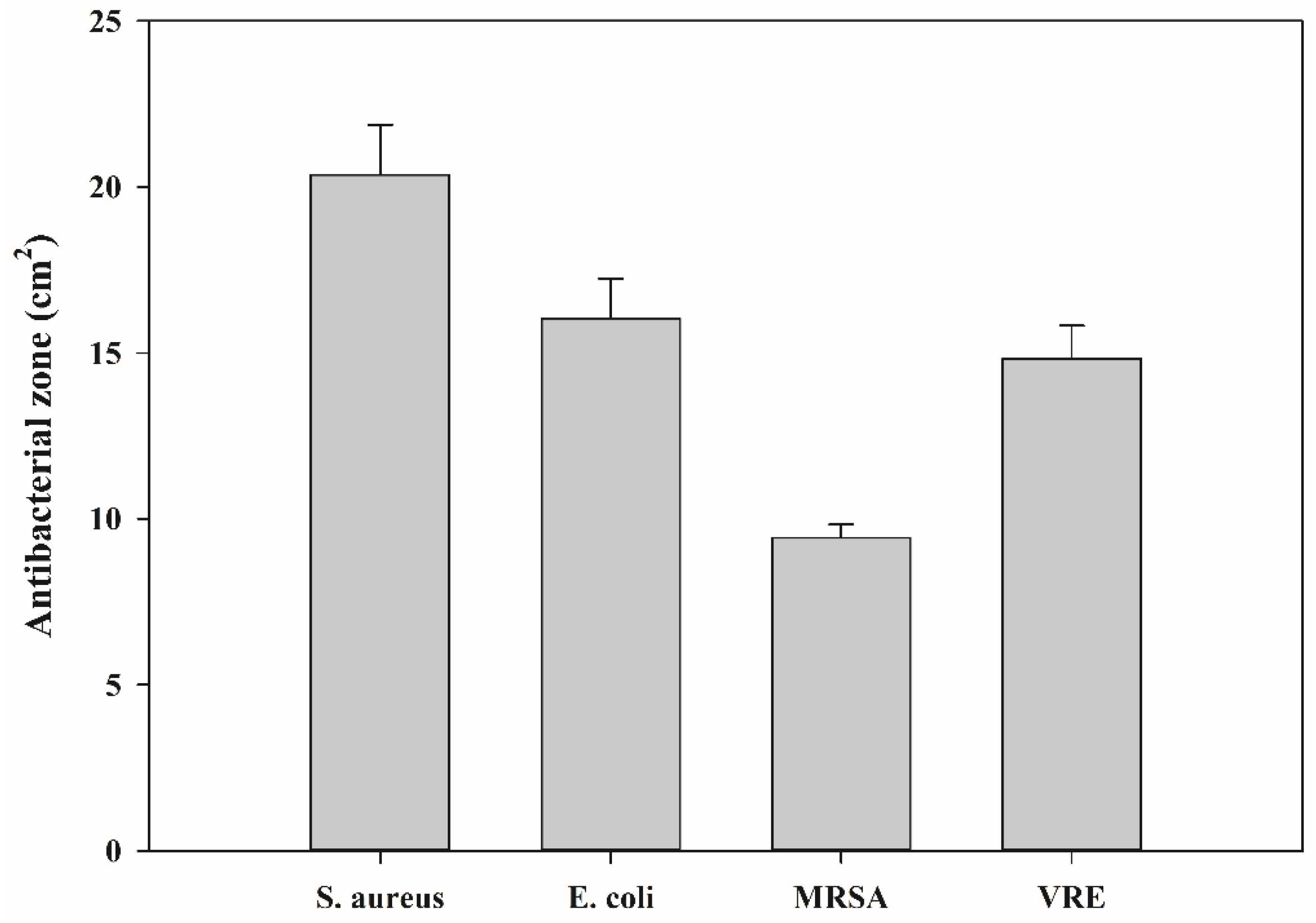
3.4. Antibacterial Activity of Bioelastomer
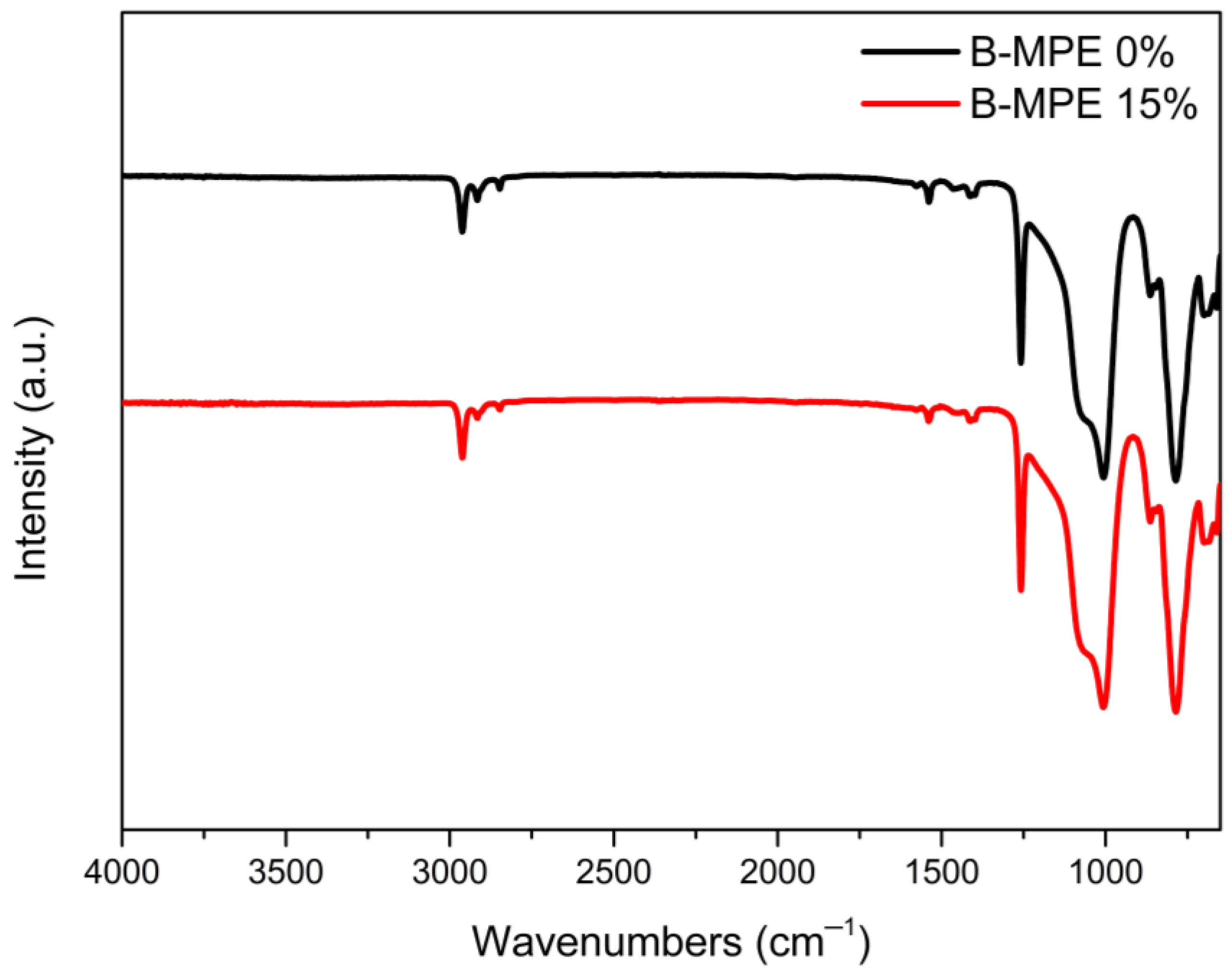
3.5. IR Analysis of Bioelastomer
3.6. SEM Image of Bioelastomer
3.7. Mechanical Strength of Bioelastomer
3.8. Water Vapor Transmission Rate of Bioelastomer
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Appendix A


References
- Jang, S.-K.; Jung, C.-D.; Seong, H.; Myung, S.; Kim, H. An integrated biorefinery process for mandarin peel waste elimination. J. Clean. Prod. 2022, 371, 133594. [Google Scholar] [CrossRef]
- Falcinelli, B.; Famiani, F.; Paoletti, A.; D’Egidio, S.; Stagnari, F.; Galieni, A.; Benincasa, P. Phenolic Compounds and Antioxidant Activity of Sprouts from Seeds of Citrus Species. Agriculture 2020, 10, 33. [Google Scholar] [CrossRef]
- Jeju special self-governing province. Citrus Annual Distribution Processing Analysis. Available online: http://citrus.or.kr/board/data/chang/2019.pdf (accessed on 21 March 2021).
- Lee, H.H.; Heo, D.Y.; Han, H.R.; Park, H.R.; Ruangcharus, C.; Kim, S.U.; Seo, D.C.; Oh, T.-K.; Hong, C.O. Evaluation of the effects of mandarin (Citrus reticulate) by-products containing citric acid on immobilization of cadmium in arable soils. Appl. Biol. Chem. 2019, 62, 45. [Google Scholar] [CrossRef]
- Choi, I.S.; Kim, J.-H.; Wi, S.G.; Kim, K.H.; Bae, H.-J. Bioethanol production from mandarin (Citrus unshiu) peel waste using popping pretreatment. Appl. Energy 2013, 102, 204–210. [Google Scholar] [CrossRef]
- Shin, D.Y.; Cho, H.U.; Utomo, J.C.; Choi, Y.N.; Xua, X.; Park, J.M. Biodiesel production from Scenedesmus bijuga grown in anaerobically digested food wastewater effluent. Bioresour. Technol. 2015, 184, 215–221. [Google Scholar] [CrossRef]
- Kadlimatti, H.M.; Raj Mohan, B.; Saidutta, M.B. Bio-oil from microwave assisted pyrolysis of food waste-optimization using response surface methodology. Biomass Bioenergy 2019, 123, 25–33. [Google Scholar] [CrossRef]
- Jang, Y.W.; Lee, K.H.; Yoo, H.Y. Improved sugar recovery from orange peel by statistical optimization of thermo-alkaline pretreatment. Processes 2021, 9, 409. [Google Scholar] [CrossRef]
- Kim, M.-H.; Kim, J.-W. Comparison through a LCA evaluation analysis of food waste disposal options from the perspective of global warming and resource recovery. Sci. Total Environ. 2010, 408, 3998–4006. [Google Scholar] [CrossRef]
- Saqib, N.U.; Sharma, H.B.; Baroutian, S.; Dubey, B.; Sarmah, A.K. Valorisation of food waste via hydrothermal carbonisation and techno-economic feasibility assessment. Sci. Total. Environ. 2019, 690, 261–276. [Google Scholar] [CrossRef]
- Wang, Y.; Tang, Y.; Li, M.; Yuan, Z. Aeration rate improves the compost quality of food waste and promotes the decomposition of toxic materials in leachate by changing the bacterial community. Bioresour. Technol. 2021, 340, 125716. [Google Scholar] [CrossRef]
- Ryen, E.G.; Babbitt, C.W. The role of US policy in advancing circular economy solutions for wasted food. J. Clean. Prod. 2022, 369, 133200. [Google Scholar] [CrossRef]
- Joshi, P.; Visvanathan, C. Sustainable management practices of food waste in Asia: Technological and policy drivers. J. Environ. Manag. 2019, 247, 538–550. [Google Scholar] [CrossRef] [PubMed]
- Velvizhi, G.; Balakumar, K.; Shetti, N.P.; Ahmad, E.; Kishore Pant, K.; Aminabhavi, T.M. Integrated biorefinery processes for conversion of lignocellulosic biomass to value added materials: Paving a path towards circular economy. Bioresour. Technol. 2022, 343, 126151. [Google Scholar] [CrossRef] [PubMed]
- Jia, F.R.; Jing, W.T.; Liu, G.X.; Yue, Q.; Wang, H.M.; Shi, L. Paraffin-based crude oil refining process unit-level energy consumption and CO2 emissions in China. J. Clean. Prod. 2020, 255, 120347. [Google Scholar] [CrossRef]
- Zang, Z. Are China’s climate commitments in a post-Paris agreement sufficiently ambitious? Wiley Interdiscip. Rev. Clim. Change 2017, 8, e443. [Google Scholar] [CrossRef]
- Lee, K.H.; Lee, S.K.; Lee, J.; Kim, S.; Kim, S.W.; Park, C.; Yoo, H.Y. Energy-efficient glucose recovery from chestnut shell by optimization of NaOH pretreatment at room temperature and application to bioethanol production. Environ. Res. 2022, 208, 112710. [Google Scholar] [CrossRef]
- Yoo, H.Y.; Kim, S.W. The next-generation biomass for biorefining. Bioresources 2021, 16, 2188–2191. [Google Scholar] [CrossRef]
- Mahato, N.; Sinha, M.; Sharma, K.; Koteswararao, R.; Cho, M.H. Modern extraction and purification techniques for obtaining high purity food-grade bioactive compounds and value-added co-products from citrus wastes. Foods 2019, 8, 523. [Google Scholar] [CrossRef]
- Hiasa, S.; Iwamoto, S.; Endo, T.; Edashige, Y. Isolation of cellulose nanofibrils from mandarin (Citrus unshiu) peel waste. Ind. Crops Prod. 2014, 62, 280–285. [Google Scholar] [CrossRef]
- Satari, B.; Karimi, K. Citrus processing wastes: Environmental impacts, recent advances, and future perspectives in total valorization. Resour. Conserv. Recycl. 2018, 129, 153–167. [Google Scholar] [CrossRef]
- John, I.; Pola, J.; Thanabalan, M.; Appusamy, A. Bioethanol production from Musambi peel by acid catalyzed steam pretreatment and enzymatic saccharification: Optimization of delignification using Taguchi design. Waste Biomass Valorization 2020, 11, 2631–2643. [Google Scholar] [CrossRef]
- Patsalou, M.; Samanides, C.G.; Protopapa, E.; Stavrinou, S.; Vyrides, I.; Koutinas, M. A Citrus peel waste biorefinery for ethanol and methane production. Molecules 2019, 24, 2451. [Google Scholar] [CrossRef] [PubMed]
- Manaf, I.S.A.; Embong, N.H.; Khazaai, S.N.M.; Rahim, M.H.A.; Yusoff, M.M.; Lee, K.T.; Maniam, G.P. A review for key challenges of the development of biodiesel industry. Energy Convers. Manag. 2019, 185, 508–517. [Google Scholar] [CrossRef]
- Koutra, E.; Economou, C.N.; Tsafrakidou, P.; Kornaros, M. Bio-based products from microalgae cultivated in digestates. Trends Biotechnol. 2018, 36, 819–833. [Google Scholar] [CrossRef]
- Zhang, H.J.; Cui, J.F.; Tian, G.F.; DiMarco-Crook, C.; Gao, W.; Zhao, C.Y.; Li, G.Y.; Lian, Y.H.; Xiao, H.; Zheng, J.K. Efficiency of four different dietary preparation methods in extracting functional compounds from dried tangerine peel. Food Chem. 2019, 289, 340–350. [Google Scholar] [CrossRef]
- Song, X.; Liu, T.; Wang, L.; Liu, L.; Li, X.; Wu, X. Antibacterial effects and mechanism of mandarin (Citrus reticulata L.) essential oil against Staphylococcus aureus. Molecules 2020, 25, 4956. [Google Scholar] [CrossRef]
- Khan, M.K.; Zill, E.H.; Dangles, O. A comprehensive review on flavanones, the major citrus polyphenols. J. Food Compos. Anal. 2014, 33, 85–104. [Google Scholar] [CrossRef]
- Smeriglio, A.; Cornara, L.; Denaro, M.; Barreca, D.; Burlando, B.; Xiao, J.; Trombetta, D. Antioxidant and cytoprotective activities of an ancient Mediterranean citrus (Citrus lumia Risso) albedo extract: Microscopic observations and polyphenol characterization. Food Chem. 2019, 279, 347–355. [Google Scholar] [CrossRef]
- Ernawita; Wahyuono, R.A.; Hesse, J.; Hipler, U.-C.; Elsner, P.; Böhm, V. In vitro lipophilic antioxidant capacity, antidiabetic and antibacterial activity of citrus fruits extracts from Aceh, Indonesia. Antioxidants 2017, 6, 11. [Google Scholar] [CrossRef]
- Gupta, A.; Jacobson, G.A.; Burgess, J.R.; Jelinek, H.F.; Nichols, D.S.; Narkowicz, C.K.; Al-Aubaidy, H.A. Citrus bioflavonoids dipeptidyl peptidase-4 inhibition compared with gliptin antidiabetic medications. Biochem. Biophys. Res. Commun. 2018, 503, 21–25. [Google Scholar] [CrossRef]
- Chen, X.M.; Tait, A.R.; Kitts, D.D. Flavonoid composition of orange peel and its association with antioxidant and anti-inflammatory activities. Food Chem. 2017, 218, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Bensid, A.; El Abed, N.; Houicher, A.; Regenstein, J.M.; Özogul, F. Antioxidant and antimicrobial preservatives: Properties, mechanism of action and applications in food—A review. Crit. Rev. Food Sci. Nutr. 2020, 32, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Singh, J.P.; Kaur, A.; Singh, N. Phenolic composition, antioxidant potential and health benefits of citrus peel. Food Res. Int. 2020, 132, 109114. [Google Scholar] [CrossRef] [PubMed]
- Bustamante, J.; Van Stempvoort, S.; García-Gallarreta, M.; Houghton, J.A.; Briers, H.K.; Budarin, V.L.; Matharu, A.S.; Clark, J.H. Microwave assisted hydro-distillation of essential oils from wet citrus peel waste. J. Clean. Prod. 2016, 137, 598–605. [Google Scholar] [CrossRef]
- Drinić, Z.; Vladić, J.; Koren, A.; Zeremski, T.; Stojanov, N.; Kiprovski, B.; Vidović, S. Microwave-assisted extraction of cannabinoids and antioxidants from Cannabis sativa aerial parts and process modeling. J. Chem. Technol. Biotechnol. 2020, 95, 831–839. [Google Scholar] [CrossRef]
- de Oliveira Filho, J.G.; Braga, A.R.C.; de Oliveira, B.R.; Gomes, F.P.; Moreira, V.L.; Pereira, V.A.C.; Egea, M.B. The potential of anthocyanins in smart, active, and bioactive eco-friendly polymer-based films: A review. Food Res. Int. 2021, 142, 110202. [Google Scholar] [CrossRef]
- Dordevic, S.; Dordevic, D.; Sedlacek, P.; Kalina, M.; Tesikova, K.; Antonic, B.; Tremlova, B.; Treml, J.; Nejezchlebova, M.; Vapenka, L.; et al. Incorporation of Natural Blueberry, Red Grapes and Parsley Extract By-Products into the Production of Chitosan Edible Films. Polymers 2021, 13, 3388. [Google Scholar] [CrossRef]
- Magaña, I.; López, R.; Enríquez-Medrano, F.J.; Kumar, S.; Aguilar-Sanchez, A.; Handa, R.; de León, R.D.; Valencia, L. Bioelastomers: Current state of development. J. Mater. Chem. A 2022, 10, 5019–5043. [Google Scholar] [CrossRef]
- Lee, K.H.; Chun, Y.; Jang, Y.W.; Lee, S.K.; Kim, H.R.; Lee, J.H.; Kim, S.W.; Park, C.; Yoo, H.Y. Fabrication of functional bioelastomer for food packaging from Aronia (Aronia melanocarpa) juice processing by-products. Foods 2020, 9, 1565. [Google Scholar] [CrossRef]
- Liu, J.; Cheng, Y.; Xu, K.; An, L.; Su, Y.; Li, X.; Zhang, Z. Effect of nano-silica filler on microstructure and mechanical properties of polydimethylsiloxane-based nanocomposites prepared by “inhibition-grafting” method. Compos. Sci. Technol. 2018, 167, 355–363. [Google Scholar] [CrossRef]
- Huang, N.; Zang, J.; Zhang, G.; Guan, L.; Li, S.; Zhao, L.; Tang, L. Efficient interfacial interaction for improving mechanical properties of polydimethylsiloxane nanocomposites filled with low content of graphene oxide nanoribbons. RSC Adv. 2017, 7, 22045–22053. [Google Scholar] [CrossRef]
- Periakaruppan, R.; Li, J.; Mei, H.; Yu, Y.; Hu, S.; Chen, X.; Li, X.; Guo, G. Agro–waste mediated biopolymer for production of biogenic nano iron oxide with superparamagnetic power and antioxidant strength. J. Clean. Prod. 2021, 311, 127512. [Google Scholar] [CrossRef]
- Lee, K.H.; Jang, Y.W.; Kim, H.; Ki, J.-S.; Yoo, H.Y. Optimization of lutein recovery from Tetraselmis suecica by response surface methodology. Biomolecules 2021, 11, 182. [Google Scholar] [CrossRef] [PubMed]
- Inoue, T.; Tsubaki, S.; Ogawa, K.; Onishi, K.; Azuma, J.I. Isolation of hesperidin from peels of thinned Citrus unshiu fruits by microwave-assisted extraction. Food Chem. 2010, 123, 542–547. [Google Scholar] [CrossRef]
- Magwaza, L.S.; Oara, U.L.; Cronje, P.J.R.; Landahl, S.; Ortiz, J.O.; Terry, L.A. Rapid methods for extracting and quantifying phenolic compounds in citrus rinds. Food Sci. Nutr. 2016, 4, 4–10. [Google Scholar] [CrossRef]
- Ballard, T.S.; Mallikarjunan, P.; Zhou, K.; O’Keefe, S. Microwave-assisted extraction of phenolic antioxidant compounds from peanut skins. Food Chem. 2010, 120, 1185–1192. [Google Scholar] [CrossRef]
- Ahmad, J.; Langrish, T.A.G. Optimisation of total phenolic acids extraction from mandarin peels using microwave energy: The importance of the Maillard reaction. J. Food Eng. 2012, 109, 162–174. [Google Scholar] [CrossRef]
- Chen, C.-W.; Xie, J.; Yang, F.-X.; Zhang, H.-L.; Xu, Z.-W.; Liu, J.-L.; Chen, Y.-J. Development of moisture-absorbing and antioxidant active packaging film based on poly (vinyl alcohol) incorporated with green tea extract and its effect on the quality of dried eel. J. Food Process. Preserv. 2018, 42, e13374. [Google Scholar] [CrossRef]
- Floegel, A.; Kim, D.-O.; Chung, S.-J.; Koo, S.I.; Chun, O.K. Comparison of ABTS/DPPH assays to measure antioxidant capacity in popular antioxidant-rich US foods. J. Food Compos. Anal. 2011, 24, 1043–1048. [Google Scholar] [CrossRef]
- Karaçelik, A.A.; Küçük, M.; Iskefiyeli, Z.; Aydemir, S.; De Smet, S.; Miserez, B.; Sandra, P. Antioxidant components of Viburnum opulus L. determined by on-line HPLC–UV–ABTS radical scavenging and LC–UV–ESI-MS methods. Food Chem. 2015, 175, 106–114. [Google Scholar] [CrossRef]
- Rodrigo, M.J.; Alquézar, B.; Alós, E.; Lado, J.; Zacarías, L. Biochemical bases and molecular regulation of pigmentation in the peel of Citrus fruit. Sci. Hortic. 2013, 163, 46–62. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, X.; Wang, Y.; Jiang, P.; Quek, S. Antibacterial activity and mechanism of cinnamon essential oil against Escherichia coli and Staphylococcus aureus. Food Control 2016, 59, 282–289. [Google Scholar] [CrossRef]
- Herraiz-Carboné, M.; Cotillas, S.; Lacasa, E.; Sainz de Baranda, C.; Riquelme, E.; Cañizares, P.; Rodrigo, M.; Sáez, C. Are We Correctly Targeting the Research on Disinfection of Antibiotic-Resistant Bacteria (ARB)? J. Clean. Prod. 2021, 320, 128865. [Google Scholar] [CrossRef]
- Shehata, M.G.; Awad, T.S.; Asker, D.H.; Abd El-Aziz, N.M.; Youssef, M.M. Antioxidant and antimicrobial activities and UPLC-ESI-MS/MS polyphenolic profile of sweet orange peel extracts. Curr. Res. Food Sci. 2021, 4, 326–335. [Google Scholar] [CrossRef] [PubMed]
- Alexandre, E.M.; Silva, S.; Santos, S.A.; Silvestre, A.J.; Duarte, M.F.; Saraiva, J.A.; Pintado, M. Antimicrobial activity of pomegranate peel extracts performed by high pressure and enzymatic assisted extraction. Food Res. Int. 2019, 115, 167–176. [Google Scholar] [CrossRef]
- Choi, S.-S.; Lee, S.-H.; Lee, K.-A. A comparative study of hesperetin, hesperidin and hesperidin glucoside: Antioxidant, anti-inflammatory, and antibacterial activities in vitro. Antioxidants 2022, 11, 1618. [Google Scholar] [CrossRef]
- Riaz, A.; Lagnika, C.; Luo, H.; Dai, Z.; Nie, M.; Hashim, M.M.; Liu, C.; Song, J.; Li, D. Chitosan-based biodegradable active food packaging film containing Chinese chive (Allium tuberosum) root extract for food application. Int. J. Biol. Macromol. 2020, 150, 595–604. [Google Scholar] [CrossRef]
- Shivangi, S.; Dorairaj, D.; Negi, P.S.; Shetty, N.P. Development and characterisation of a pectin-based edible film that contains mulberry leaf extract and its bio-active components. Food Hydrocoll. 2021, 121, 107046. [Google Scholar] [CrossRef]
- Abshirini, M.; Saha, M.C.; Altan, M.C.; Liu, Y. Synthesis and characterization of hierarchical porous structure of polydimethylsiloxane (PDMS) sheets via two-step phase separation method. Mater. Des. 2021, 212, 110194. [Google Scholar] [CrossRef]
- Abdelwahab, M.A.; Jacob, S.; Misra, M.; Mohanty, A.K. Super-tough sustainable biobased composites from polylactide bioplastic and lignin for bio-elastomer application. Polymer 2021, 212, 123153. [Google Scholar] [CrossRef]
- da Rosa, G.S.; Vanga, S.K.; Gariepy, Y.; Raghavan, V. Development of Biodegradable Films with Improved Antioxidant Properties Based on the Addition of Carrageenan Containing Olive Leaf Extract for Food Packaging Applications. J. Polym. Environ. 2020, 28, 123–130. [Google Scholar] [CrossRef]
- Song, Z.; Xiao, H.; Zhao, Y. Hydrophobic-modified nano-cellulose fiber/PLA biodegradable composites for lowering water vapor transmission rate (WVTR) of paper. Carbohydr. Polym. 2014, 111, 442–448. [Google Scholar] [CrossRef] [PubMed]
- El Bourakadi, K.; el Kacem Qaiss, A.; Bouhfid, R. Bio-films based on alginate/modified clay through spray drying: Mechanical, rheological, morphological, and transport properties for potential use as active food packaging. Int. J. Biol. Macromol. 2022, 210, 663–668. [Google Scholar] [CrossRef] [PubMed]
- Cerqueira, M.A.; Souza, B.; Martins, J.T.; Teixeira, J.A.; Vicente, A.A. Seed extracts of Gleditsia triacanthos: Functional properties evaluation and incorporation into galactomannan films. Food Res. Int. 2010, 43, 2031–2038. [Google Scholar] [CrossRef]

| Sample | PDMS (g) | MPE (g) |
|---|---|---|
| B–MPE 0% (control, PDMS) | 50 | 0 |
| B–MPE 1% (w/w) | 49.5 | 0.5 |
| B–MPE 3% | 48.5 | 1.5 |
| B–MPE 5% | 47.5 | 2.5 |
| B–MPE 7% | 46.5 | 3.5 |
| B–MPE 10% | 45 | 5 |
| B–MPE 15% | 42.5 | 7.5 |
| B–MPE 20% | 40 | 10 |
| Sample | Radical Scavenging Activity (%) | |
|---|---|---|
| DPPH | ABTS | |
| B–MPE 0% (control, PDMS) | 0 | 0 |
| B–MPE 1% | 1.0 ± 0.0 | 13.1 ± 0.3 |
| B–MPE 3% | 1.1 ± 0.1 | 16.8 ± 0.4 |
| B–MPE 5% | 1.2 ± 0.1 | 17.3 ± 0.6 |
| B–MPE 7% | 1.5 ± 0.1 | 18.8 ± 0.3 |
| B–MPE 10% | 1.8 ± 0.1 | 19.8 ± 0.6 |
| B–MPE 15% | 3.3 ± 0.2 | 20.8 ± 0.8 |
| B–MPE 20% | 4.8 ± 0.3 | 26.7 ± 1.2 |
| Hesperidin 100 ppm | 3.5 ± 0.4 | 17.8 ± 0.6 |
| Sample | Tensile Strength (N/mm2) | Elongation at Break (%) | Water vapor Transmission Rate (g/(m2 day)) |
|---|---|---|---|
| B–MPE 0% | 5.2 | 551 | 26.6 |
| B–MPE 15% | 5.1 | 649 | 33.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, K.H.; Chun, Y.; Lee, J.H.; Lee, J.U.; Lee, T.; Yoo, H.Y. Sustainable Utilization Strategy of Organic Waste via Fabrication of Bioelastomer with Antibacterial and Antioxidant Activities Using Mandarin Peel Extracts. Agriculture 2023, 13, 161. https://doi.org/10.3390/agriculture13010161
Lee KH, Chun Y, Lee JH, Lee JU, Lee T, Yoo HY. Sustainable Utilization Strategy of Organic Waste via Fabrication of Bioelastomer with Antibacterial and Antioxidant Activities Using Mandarin Peel Extracts. Agriculture. 2023; 13(1):161. https://doi.org/10.3390/agriculture13010161
Chicago/Turabian StyleLee, Kang Hyun, Youngsang Chun, Ja Hyun Lee, Jong Uk Lee, Taek Lee, and Hah Young Yoo. 2023. "Sustainable Utilization Strategy of Organic Waste via Fabrication of Bioelastomer with Antibacterial and Antioxidant Activities Using Mandarin Peel Extracts" Agriculture 13, no. 1: 161. https://doi.org/10.3390/agriculture13010161
APA StyleLee, K. H., Chun, Y., Lee, J. H., Lee, J. U., Lee, T., & Yoo, H. Y. (2023). Sustainable Utilization Strategy of Organic Waste via Fabrication of Bioelastomer with Antibacterial and Antioxidant Activities Using Mandarin Peel Extracts. Agriculture, 13(1), 161. https://doi.org/10.3390/agriculture13010161







