Effects of Convective Drying of Quince Fruit (Cydonia oblonga) on Color, Antioxidant Activity and Phenolic Compounds under Various Fruit Juice Dipping Pre-Treatments
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Pre-Treatments of Samples
2.3. Cabinet Convective Dryer
2.4. Drying Tests
2.5. Color Measurement
2.6. Bioactive Compounds (Phenolic (TP), Trolox Equivalent Antioxidant Capacity (TEAC), Total Flavonoid (TF) Analysis)
2.7. Statistical Analysis
3. Results and Discussion
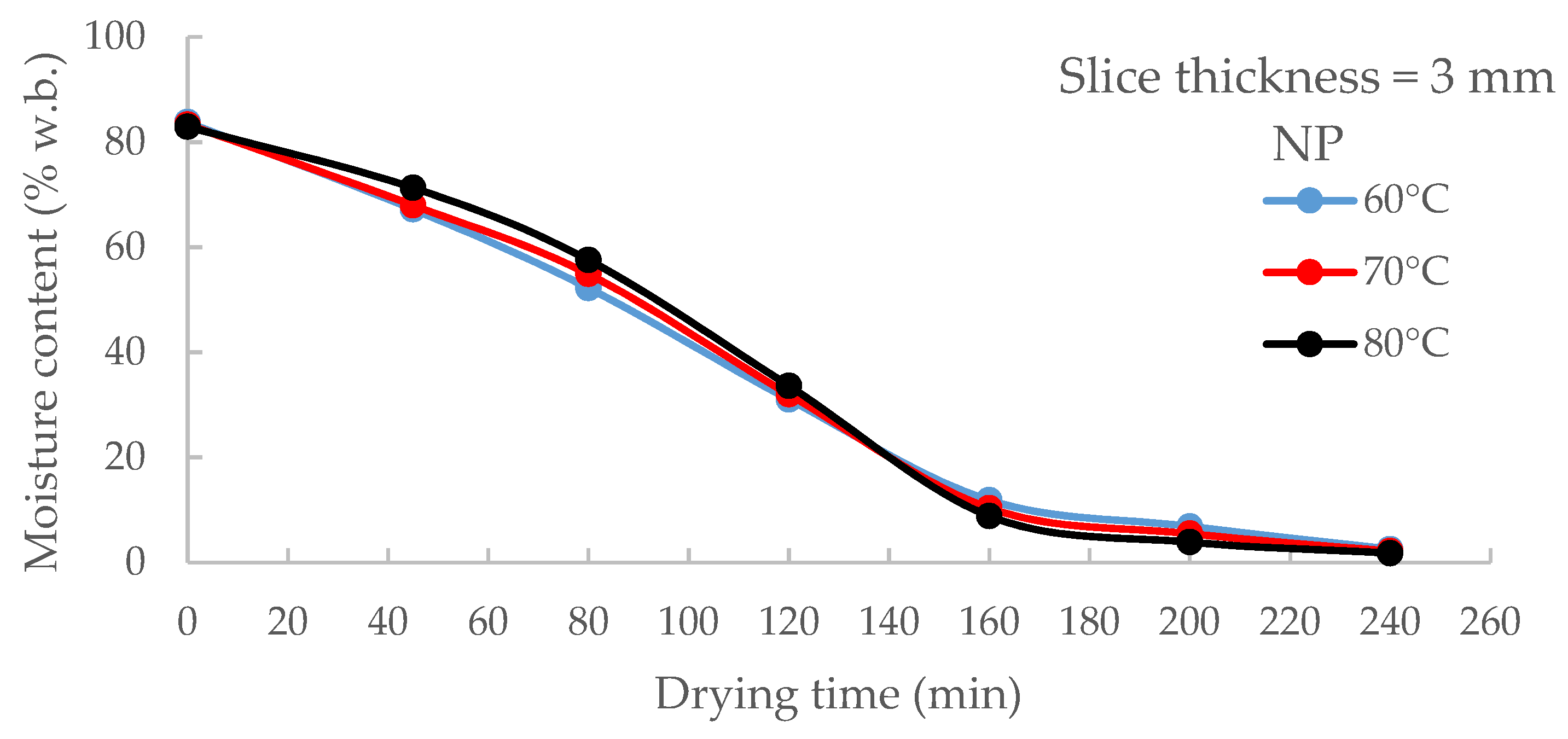
3.1. Effects of Drying Air Temperature on Drying Time
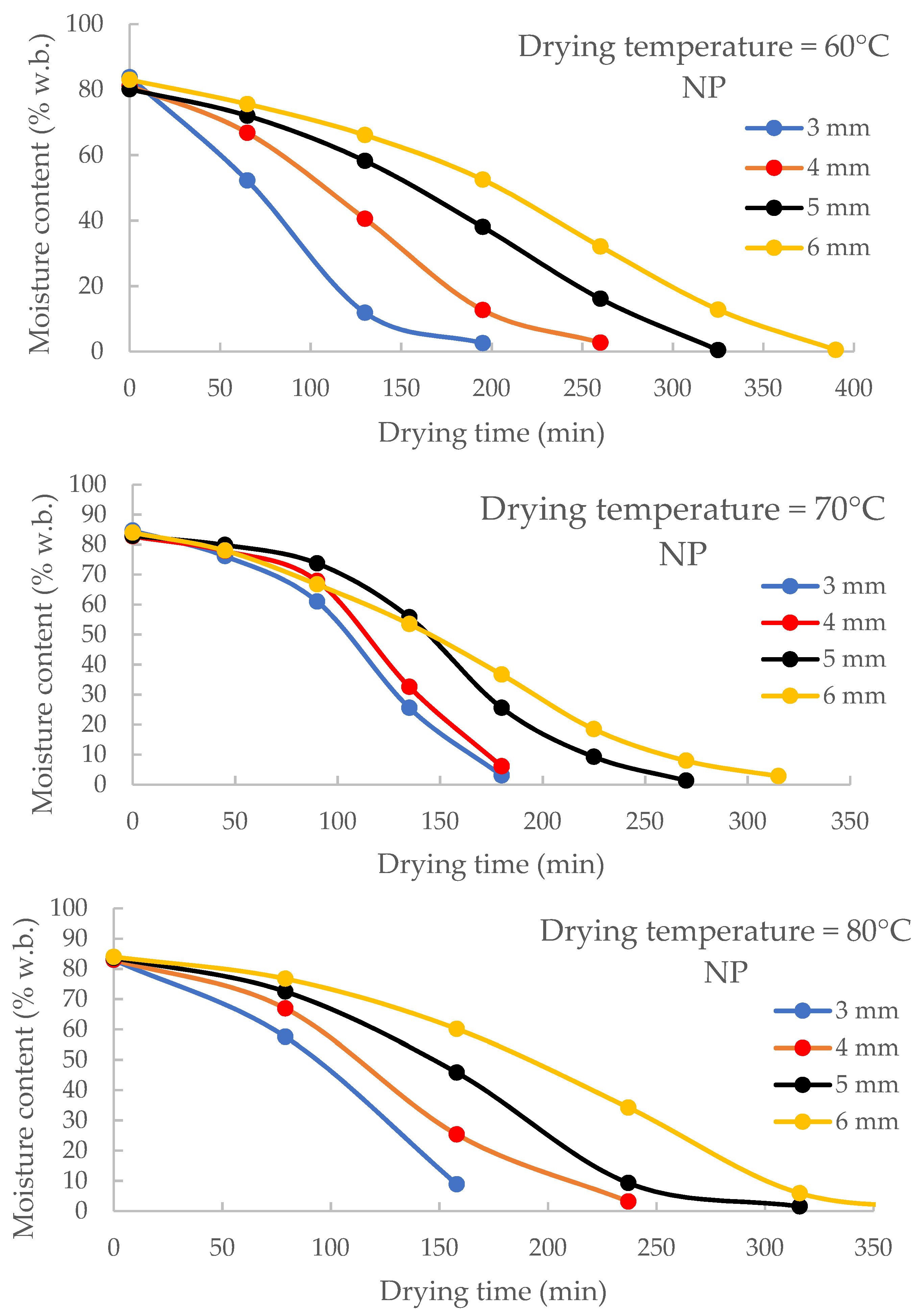
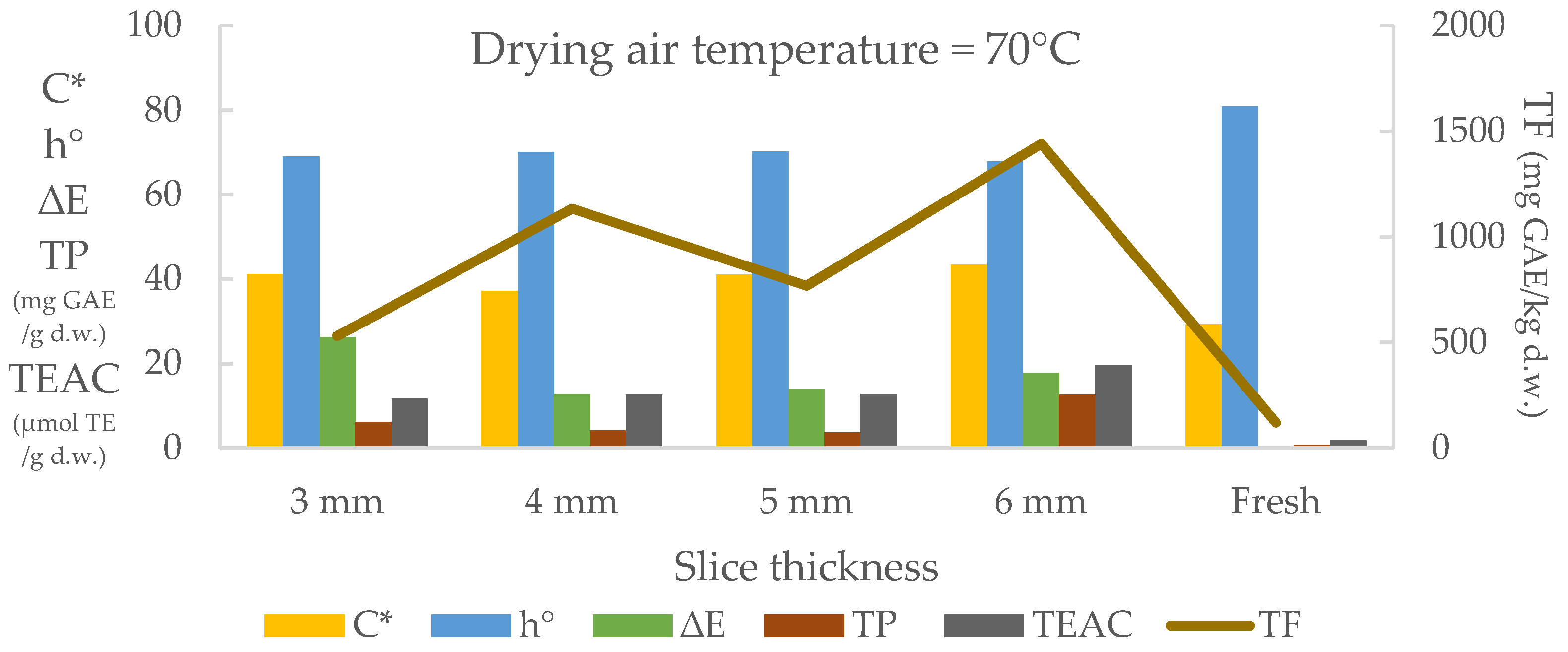
3.2. Effect of Thickness on Drying Time
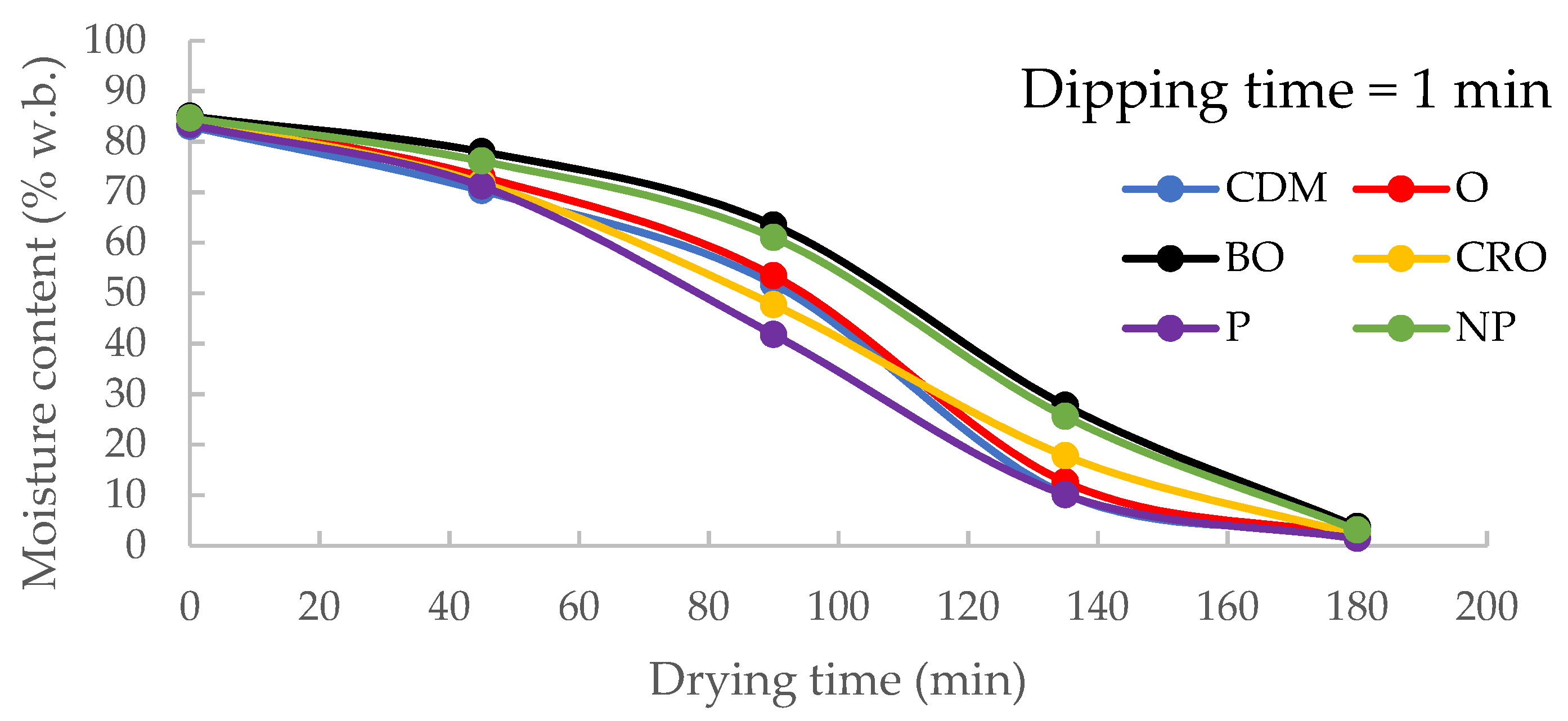
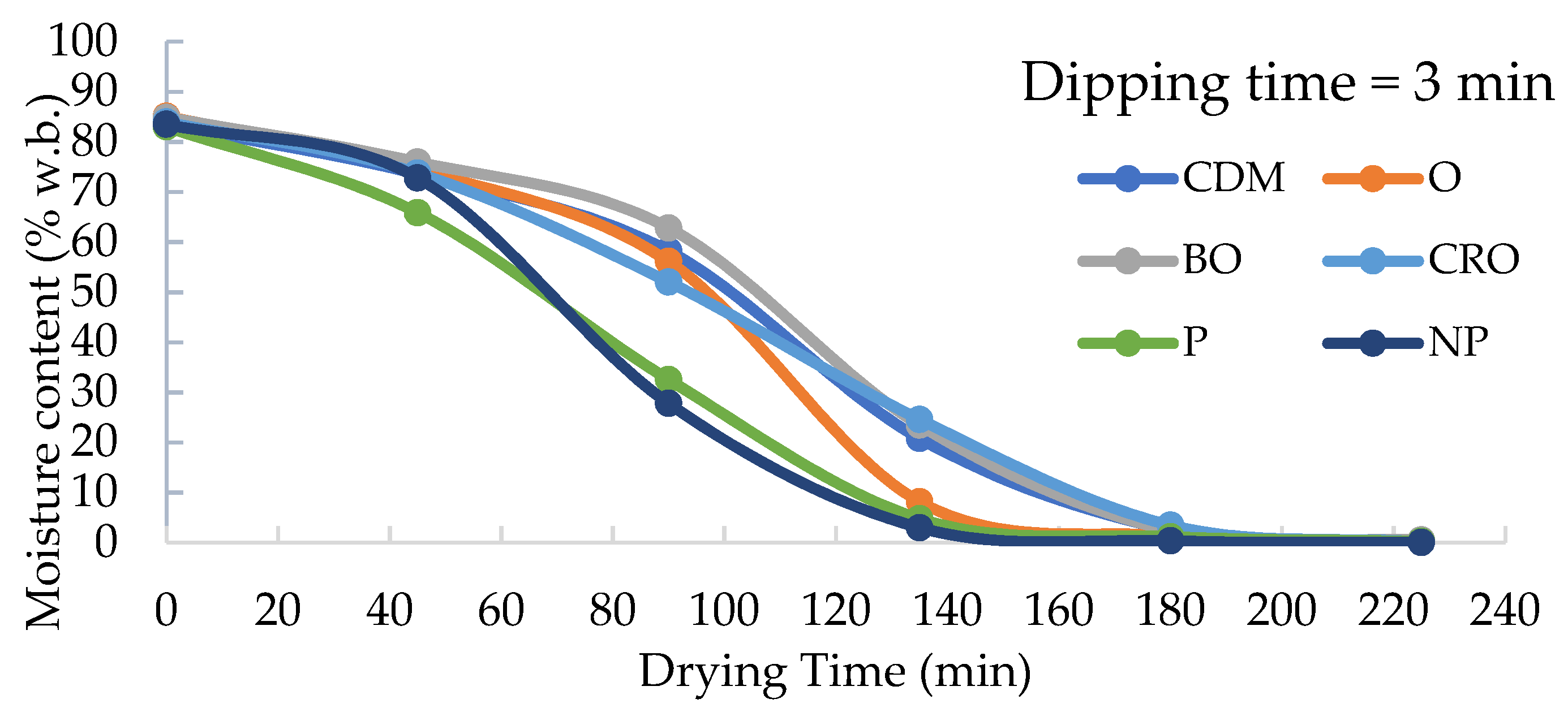
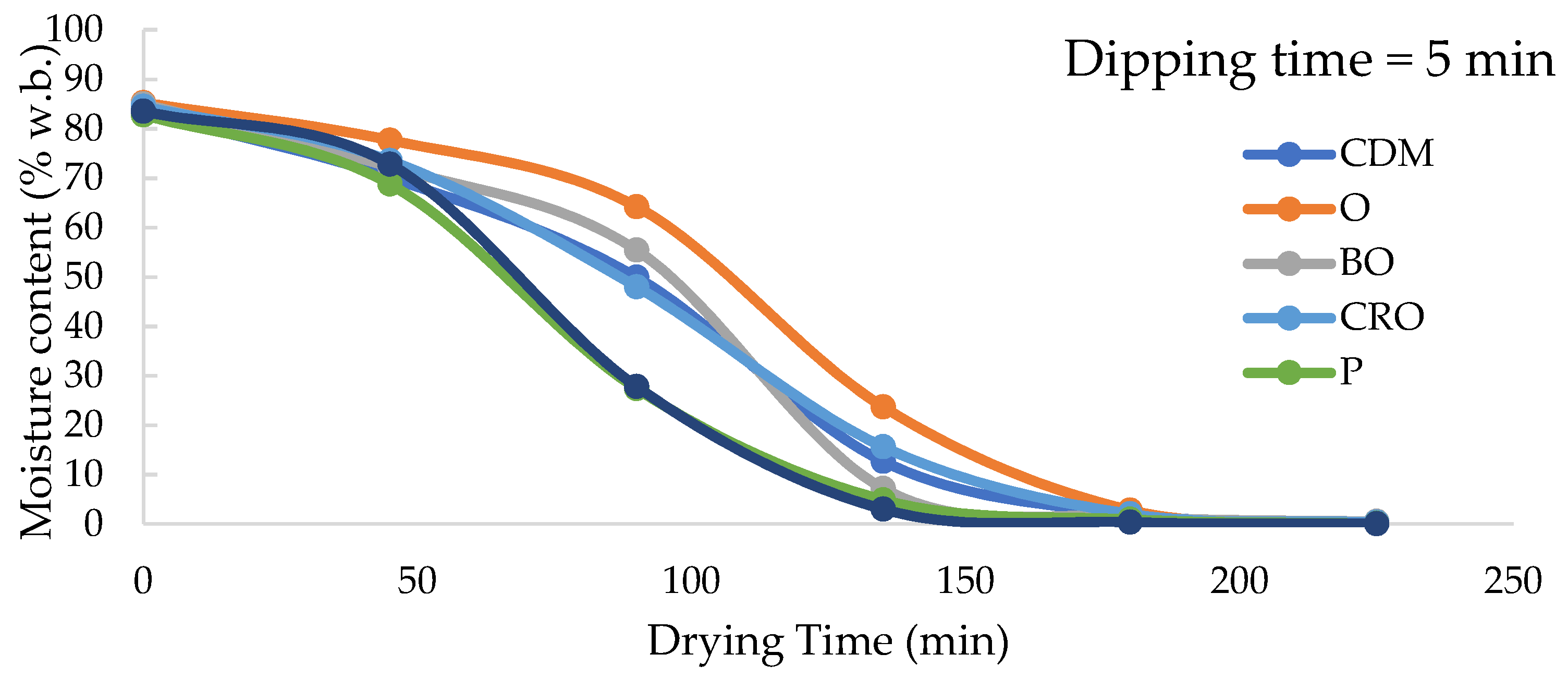
3.3. Effects of Dipping Pre-Treatments on Drying Time
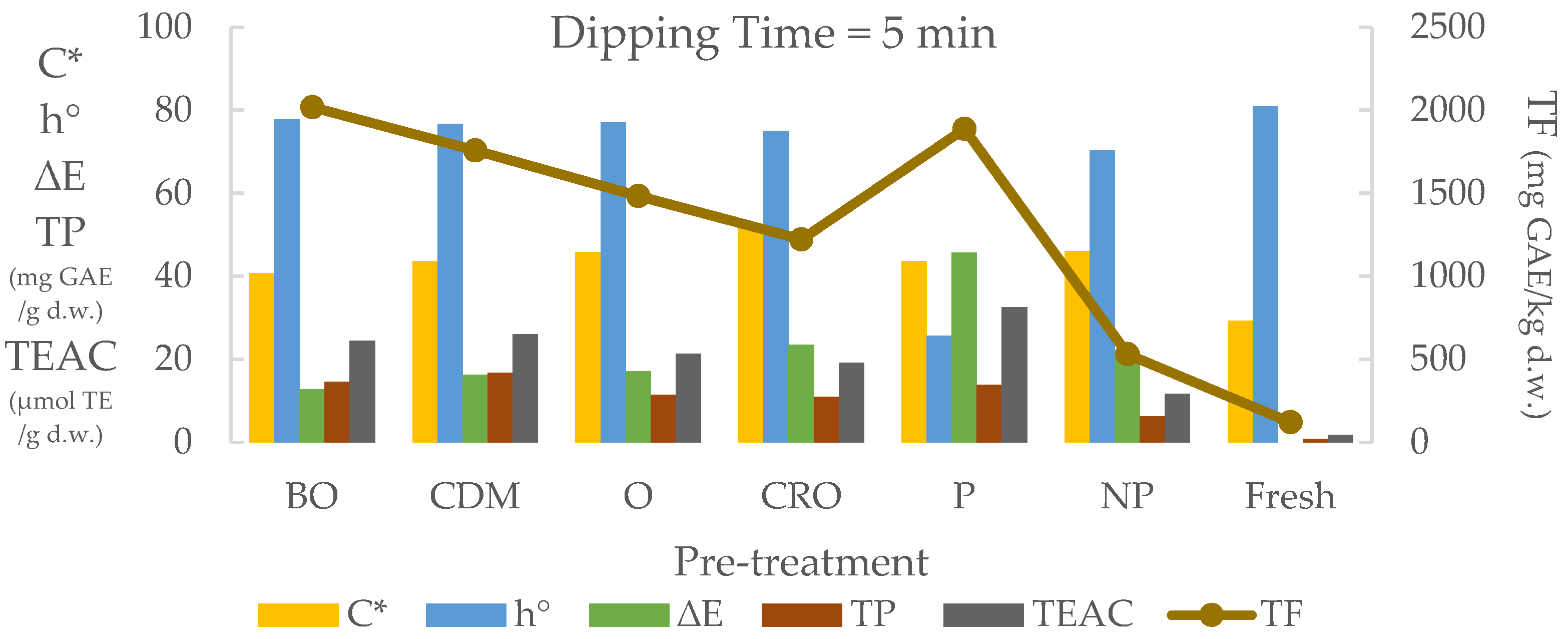
3.4. Dipping Pre-Treatment Effect on Color and Biocompound Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- SG. Tarım ve Orman Bakanlığı, Strateji Geliştirme Başkanlığı. Available online: https://arastirma.tarimorman.gov.tr/tepge/Belgeler/PDF%20Tar%C4%B1m%20%C3%9Cr%C3%BCnleri%20Piyasalar%C4%B1/2021-Ocak%20Tar%C4%B1m%20%C3%9Cr%C3%BCnleri%20Raporu/Ayva,%20Ocak-2021,%20Tar%C4%B1m%20%C3%9Cr%C3%BCnleri%20Piyasa%20Raporu.pdf (accessed on 8 October 2021).
- Antoniewska, A.; Rutkowska, J.; Adamska, A. Charakterystyka owoców pigwowca japońskiego oraz ich zastosowanie w przemyśle spożywczym. Żywność Nauka Technol. Jakość 2017, 24, 5–15. [Google Scholar] [CrossRef]
- Sharma, R.; Joshi, V.K.; Rana, J.C. Nutritional composition and processed products of quince (Cydonia oblonga Mill.). Indian J. Nat. Prod. Resour. 2011, 2, 354–357. [Google Scholar]
- Silva, B.M.; Andrade, P.B.; Martins, R.C.; Seabra, R.M.; Ferreira, M.A. Principal component analysis as tool of characterization of quince (Cydonia oblonga Miller) jam. Food Chem. 2006, 94, 504–512. [Google Scholar] [CrossRef]
- Ali, N.Y.; Abbas, A.; Ali, M.; Shahnawaz, N.; Hussain, A.; Hussain, A. Physico-chemical nutritional and sensory evaluation of local quince fruit of nomal village, Gilgit-Baltistan, Pakistan. Int. J. Nutr. Food Sci. 2015, 4, 600–608. [Google Scholar] [CrossRef][Green Version]
- Rodríguez-Guisado, I.; Hernández, F.; Melgarejo, P.; Legua, P.; Martínez, R.; Martínez, J.J. Chemical, morphological and organoleptical characterisation of five Spanish quince tree clones (Cydonia oblonga Miller). Sci. Hortic. 2009, 122, 491–496. [Google Scholar] [CrossRef]
- Grygorieva, O.; Klymenko, S.; Vergun, O.; Mňahončáková, E.; Brindza, J.; Terentjeva, M.; Ivanišová, E. Evaluation of the antioxidant activity and phenolic content of Chinese quince (Pseudocydonia sinensis Schneid.) fruit. Acta Sci. Pol. Technol. Aliment. 2020, 19, 25–36. [Google Scholar]
- Legua, P.; Serrano, M.; Melgarejo, P.; Valero, D.; Martínez, J.J.; Martínez, R.; Hernández, F. Quality parameters, biocompounds and antioxidant activity in fruits of nine quince (Cydonia oblonga Miller) accessions. Sci. Hortic. 2013, 154, 61–65. [Google Scholar] [CrossRef]
- Shinomiya, F.; Hamauzu, Y.; Kawahara, T. Anti-allergic effect of a hot-water extract of quince (Cydonia oblonga). Biosci. Biotechnol. Biochem. 2009, 73, 1773–1778. [Google Scholar] [CrossRef]
- Carvalho, M.; Silva, B.M.; Silva, R.; Valentao, P.; Andrade, P.B.; Basto, M.L. First report on Cydonia oblonga Miller anticancer potential: Differential antiproliferative effect against human kidney and colon cancer cells. J. Agric. Food Chem. 2010, 58, 3366–3370. [Google Scholar] [CrossRef]
- Hamauzu, Y.; Inno, T.; Kume, C.; Irie, M.; Hiramatsu, K. Antioxidant and antiulcerative properties of phenolics from Chinese quince, quince, and apple fruits. J. Agric. Food Chem. 2006, 54, 765–772. [Google Scholar] [CrossRef]
- Fattouch, S.; Caboni, P.; Coroneo, V.; Tuberoso, C.I.; Angioni, A.; Dessi, S.; Marzauki, N.; Cabras, P. Antimicrobial activity of Tunisian quince (Cydonia oblonga Miller) pulp and peel polyphenolic extracts. J. Agric. Food Chem. 2007, 55, 963–969. [Google Scholar] [CrossRef] [PubMed]
- İzli, N.; Polat, A. Ayvanın dondurularak ve konvektif kurutulması (Cydonia oblonga Miller): Kurutma kinetiği ve kalite özelliklerine etkileri. Isı Kütle Transf. 2019, 55, 1317–1326. [Google Scholar]
- Ochoa-Martínez, C.; Quintero, P.; Ayala, A.; Ortiz, M. Drying characteristics of mango slices using the Refractance Window™ technique. J. Food Eng. 2012, 109, 69–75. [Google Scholar] [CrossRef]
- Baltacioglu, C.; Uslu, N.; Ozcan, M.M. Optimization of microwave and air-drying conditions of quince (Cydonia oblonga Miller) using response surface methodology. Ital. J. Food Sci. 2015, 27, 1N. [Google Scholar]
- Mrad, N.D.; Boudhrioua, N.; Kechaou, N.; Courtois, F.; Bonazzi, C. Influence of air drying temperature on kinetics, physicochemical properties, total phenolic content and ascorbic acid of pears. Food Bioprod. Processing 2012, 90, 433–441. [Google Scholar] [CrossRef]
- Mercer, D.G. Meyve ve Sebzelerin Dehidrasyonu ve Kurutulmasına Giriş; Guelph Üniversitesi: Guelph, ON, Kanada, 2014; p. 166. [Google Scholar]
- Chua, K.J.; Chou, S.K. Low-Cost Drying Methods for Developing Countries. Trends Food Sci. Technol. 2003, 14, 519–528. [Google Scholar] [CrossRef]
- Çoklar, H.; Akbulut, M. Kara üzümün (Ekşikara) (Vitis vinifera L.) antosiyaninler, fenolik bileşikler ve antioksidan aktivitesi üzerine güneş, fırın ve dondurarak kurutmanın etkisi. S. Afr. J. Enol. Vitik. 2017, 38, 264–272. [Google Scholar]
- Krokida, M.K.; Maroulis, Z.B.; Saravacos, G.D. Kurutma yönteminin suyu alınmış ürünlerin rengine etkisi. Int. J. Gıda Bilimi Teknol. 2001, 36, 53–59. [Google Scholar]
- Ertekin, C.; Heybeli, N. Thin-layer infrared drying of mint leaves. J. Food Processing Preserv. 2014, 38, 1480–1490. [Google Scholar] [CrossRef]
- Adepoju, L.A.; Osunde, Z.D. Effect of pre-treatments and drying methods on some qualities of dried mango (Mangifera indica) fruit. Agric. Eng. Int. CIGR J. 2017, 19, 187–194. [Google Scholar]
- Landim, A.P.M.; Barbosa, M.I.M.J.; Junior, J.L.B. Influence of osmotic dehydration on bioactive compounds, antioxidant capacity, color and texture of fruits and vegetables: A review. Cienc. Rural 2016, 46, 1714–1722. [Google Scholar] [CrossRef]
- Hedayatizadeh, M.; Chaji, H. A review on plum drying. Renew. Sustain. Energy Rev. 2016, 56, 362–367. [Google Scholar] [CrossRef]
- Lewicki, P.P. Effect of pre-drying treatment, drying and rehydration on plant tissue properties: A review. Int. J. Food Prop. 1998, 1, 1–22. [Google Scholar] [CrossRef]
- Roueita, G.; Hojjati, M.; Noshad, M. Study of physicochemical properties of dried kiwifruits using the natural hypertonic solution in ultrasound-assisted osmotic dehydration as pretreatment. Int. J. Fruit Sci. 2020, 20, S491–S507. [Google Scholar] [CrossRef]
- Salehi, F.; Kashaninejad, M.; Jafarianlari, A. Drying kinetics and characteristics of combined infrared-vacuum drying of button mushroom slices. Heat Mass Transf. 2017, 53, 1751–1759. [Google Scholar] [CrossRef]
- Ding, P.; Ling, Y.S. Browning assessment methods and polyphenol oxidase in UV-C irradiated Berangan banana fruit. Int. Food Res. J. 2014, 21, 1667–1674. [Google Scholar]
- Menges, H.O.; Unver, A.; Ozcan, M.M.; Ertekin, C.; Sonmete, M.H. The Effects of Drying Parameters on Drying Characteristics, Colorimetric Differences, Antioxidant Capacity and Total Phenols of Sliced Kiwifruit. Erwerbs-Obstbau 2019, 61, 195–207. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.L. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Chang, C.C.; Yang, M.H.; Wen, H.M.; Chern, J.C. Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J. Food Drug Anal. 2002, 10, 178–182. [Google Scholar]
- Saracoglu, O. Phytochemical accumulation of anthocyanin rich mulberry (Morus laevigata) during ripening. J. Food Meas. Charact. 2018, 12, 2158–2163. [Google Scholar] [CrossRef]
- Denge, A. Ayvanın Kuruması Sırasında Yapısında Meydana Gelen Fiziksel Değişimlerin İncelenmesi. Master’s Thesis, Afyon Kocatepe University, Institute of Science, Food Engineering Department, Afyon, Turkey, 2011; pp. 10–30. [Google Scholar]
- Aktas, M.; İlbas, M.; Yalcin, A.; Sahin, M. Kızılötesi Işınımlı Bir Kurutucuda Kuruma Davranışlarının Deneysel İncelenmesi. Gazi Üniversitesi Mühendislik Mimar. Fakültesi Derg. 2013, 28, 767–775. [Google Scholar]
- Abbaspour-Gilandeh, Y.; Jahanbakhshi, A.; Kaveh, M. Prediction kinetic, energy and exergy of quince under hot air dryer using ANNs and ANFIS. Food Sci. Nutr. 2020, 8, 594–611. [Google Scholar] [CrossRef] [PubMed]
- Szychowski, P.J.; Lech, K.; Sendra-Nadal, E.; Hernández, F.; Figiel, A.; Wojdyło, A.; Carbonell-Barrachina, Á.A. Kinetics, biocompounds, antioxidant activity, and sensory attributes of quinces as affected by drying method. Food Chem. 2018, 255, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Celen, S.; Kuş, S. Mikrodalga Enerjisinin Ayva Dilimlerinin Kurutulmasina Etkisi. Electron. J. Vocat. Coll. 2016, 6, 30–40. [Google Scholar]
- Tzempelikos, D.A.; Vouros, A.P.; Bardakas, A.V.; Filios, A.E.; Margaris, D.P. Case studies on the effect of the air drying conditions on the convective drying of quinces. Case Stud. Therm. Eng. 2014, 3, 79–85. [Google Scholar] [CrossRef]
- Tzempelikos, D.A.; Vouros, A.P.; Bardakas, A.V.; Filios, A.E.; Margaris, D.P. Experimental study on convective drying of quince slices and evaluation of thin-layer drying models. Eng. Agric. Environ. Food 2015, 8, 169–177. [Google Scholar] [CrossRef]
- Karaaslan, S.; Erdem, T.; Sarı, M. Microwave Drying of Quince Slices. SDU J. Fac. Agric. 2016, 11, 27–33. [Google Scholar]
- Izli, N.; Polat, A. Freeze and convective drying of quince (Cydonia oblonga Miller.): Effects on drying kinetics and quality attributes. Heat Mass Transf. 2019, 55, 1317–1326. [Google Scholar] [CrossRef]
- Kaya, A.; Aydin, O.; Demirtas, C.; Akgün, M. An experimental study on the drying kinetics of quince. Desalination 2007, 212, 328–343. [Google Scholar] [CrossRef]
- Turkiewicz, I.P.; Wojdyło, A.; Lech, K.; Tkacz, K.; Nowicka, P. Influence of different drying methods on the quality of Japanese quince fruit. LWT 2019, 114, 108416. [Google Scholar] [CrossRef]
- Turkiewicz, I.P.; Wojdyło, A.; Tkacz, K.; Lech, K.; Nowicka, P. Osmotic dehydration as a pretreatment modulating the physicochemical and biological properties of the Japanese quince fruit dried by the convective and vacuum-microwave method. Food Bioprocess Technol. 2020, 13, 1801–1816. [Google Scholar] [CrossRef]
- Ertekin, C.; Yaldiz, O. Drying of eggplant and selection of a suitable thin layer drying model. J. Food Eng. 2004, 63, 349–359. [Google Scholar] [CrossRef]
- Mugodo, K.; Workneh, T.S. The kinetics of thin layer drying and modelling for mango slices and the influence of differing hot-air drying methods on quality. Heliyon 2021, 7, e07182. [Google Scholar] [CrossRef] [PubMed]
- Doymaz, I.; Ozdemir, O. Effect of air temperature, slice thickness and pretreatment on drying and rehydration of tomato. Int. J. Food Sci. Technol. 2014, 49, 558–564. [Google Scholar] [CrossRef]
- Dehghannya, J.; Hosseinlar, S.H.; Heshmati, M.K. Multi-stage continuous and intermittent microwave drying of quince fruit coupled with osmotic dehydration and low temperature hot air drying. Innov. Food Sci. Emerg. Technol. 2018, 45, 132–151. [Google Scholar] [CrossRef]
- Doymaz, İ.; Demir, H.; Yildirim, A. Drying of quince slices: Effect of pretreatments on drying and rehydration characteristics. Chem. Eng. Commun. 2015, 202, 1271–1279. [Google Scholar] [CrossRef]
- Icier, F.; Yildiz, H.; Eroglu, S.; Sabanci, S.; Eroglu, E. Ayva dilimlerinin ozmotik kurutulmasında elektriksel ve ultrasonik ön işlemlerin etkisi. Akad. Gıda 2013, 11, 60–69. [Google Scholar]
- Kalejahi, A.K.; Asefi, N. Influence of vacuum impregnation pretreatment combined with IR drying on quince quality with shrinkage modeling by ANN. Chem. Eng. Commun. 2019, 206, 1661–1675. [Google Scholar] [CrossRef]
- Elmizadeh, A.; Shahedi, M.; Hamdami, N. Quality assessment of electrohydrodynamic and hot air drying of quince slice. Ind. Crops Prod. 2018, 116, 35–40. [Google Scholar] [CrossRef]
- Taghinezhad, E.; Kaveh, M.; Jahanbakhshi, A.; Golpour, I. Use of artificial intelligence for the estimation of effective moisture diffusivity, specific energy consumption, color and shrinkage in quince drying. J. Food Process Eng. 2020, 43, e13358. [Google Scholar] [CrossRef]
- Wojdyło, A.; Oszmiański, L.; Bielicki, P. Polyphenolic Composition, Antioxidant Activity, and Polyphenol Oxidase (PPO) Activity of Quince (Cydonia oblonga Miller) Varieties. J. Agric. Food Chem. 2013, 61, 2762–2772. [Google Scholar] [CrossRef] [PubMed]
- Izli, G.; Yildiz, G. Evaluation of high intensity ultrasound pre-treatment effects on the physical properties and bioactive compounds of convective dried quince samples. Int. J. Fruit Sci. 2021, 21, 645–656. [Google Scholar] [CrossRef]
- Sohrabpour, S.; Yadegari, M.; Kenari, R.E. Effect of ultrasound and acid pretreatments during dehydration on quality properties of quince. Food Sci. Technol. 2020, 14, 88–97. [Google Scholar] [CrossRef]




| Fruit Weight (g) | Fruit Volume (cc, mL, cm3) | Fruit Length (mm) | Fruit Diameter (mm) | Fruit Shape Index | L* | a* | b* | Chroma (C*) | Hue (h°) |
|---|---|---|---|---|---|---|---|---|---|
| 461.09 | 565.42 | 111.74 | 95.69 | 0.86 | 72.44 | 9.04 | 65.45 | 66.13 | 82.20 |
| Acronym | Pre-Treatment | Dipping Time (min) | Soluble Solid Content (Brix) |
|---|---|---|---|
| NP | Non pre-treatment | - | 13.47 (Fresh) |
| BO | Bitter orange (Citrus aurantium) juice | 1-3-5 | 11.88 |
| CDM | Tangerine (Citrus deliciosa Mediterranean) juice | 1-3-5 | 13.28 |
| O | Orange (Navel citrus Sinensis) juice | 1-3-5 | 12.76 |
| CRO | Tangerine (Citrus reticulata Owari) juice | 1-3-5 | 11.82 |
| P | Pomegranate (Punica granatum) juice | 1-3-5 | 17.95 |
| Dose | Dipping Time (min) | L* | a* | b* | C* | h° | ∆E | BI | TP (mg GAE /g d.w.) | TEAC (µmol TE /g d.w.) | TF (mg GAE /kg d.w.) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| BO | 1 | 72.01 b | 8.33 a | 39.90 a,b,* | 40.77 a | 78.22 b | 12.16 a | 13.70 a | 10,800.44 b | 23.80 b | 1481.11 b |
| 3 | 73.27 b | 8.73 a | 39.25 a | 40.25 a | 77.40 b | 12.55 a | 13.79 a | 12,970.44 c | 26.21 b | 1644.07 c | |
| 5 | 72.79 b | 8.58 a | 39.73 a,b,* | 40.71 a | 77.80 b | 12.71 a | 13.81 a | 14,589.19 d | 24.48 b | 2018.15 d | |
| NP | - | 57.15 a | 15.77 b | 43.13 b | 45.98 b | 70.28 a | 22.03 b | 27.30 b | 6194.46 a | 11.63 a | 531.11 a |
| CDM | 1 | 68.61 c | 11.52 a | 43.18 a | 44.76 a | 75.32 b | 16.78 a,b,* | 18.42 a | 9006.69 b | 18.62 b | 1671.85 b |
| 3 | 62.35 a,b,* | 14.69 b | 50.74 b | 52.83 b | 73.85 b | 25.14 c | 24.69 b | 11,261.13 c | 24.21 c | 1757.04 c | |
| 5 | 68.01 b,c,* | 10.22 a | 42.44 a | 43.66 a | 76.63 b | 16.20 a | 17.22 a | 16,777.80 d | 26.04 d | 1757.03 c | |
| NP | - | 57.15 a | 15.77 b | 43.13 a | 45.98 a | 70.28 a | 22.03 b,c,* | 27.30 b | 6194.46 a | 11.63 a | 531.11 a |
| O | 1 | 67.14 b | 9.05 a,b,* | 40.25 a,b,* | 41.27 a | 77.40 b | 12.59 a,b,* | 15.47 a | 12,136.13 c | 21.38 b | 1807.03 c |
| 3 | 67.16 b | 7.34 a | 38.23 a | 38.94 a | 79.22 b | 10.19 a | 13.42 a | 12,077.80 c | 21.89 b | 2012.59 d | |
| 5 | 68.67 b | 10.26 b | 44.59 c | 45.82 b | 77.02 b | 17.08 b | 17.10 a | 11,402.80 b | 21.26 b | 1482.96 b | |
| NP | - | 57.15 a | 15.77 c | 43.13 b,c,* | 45.98 b | 70.28 a | 22.03 c | 27.30 b | 6194.46 a | 11.63 a | 531.11 a |
| CRO | 1 | 67.00 b | 15.13 b | 51.49 b | 53.68 b | 73.64 b | 25.12 b | 23.52 a,b,* | 8977.80 b | 18.50 b | 1155.18 c |
| 3 | 67.09 b | 11.59 a | 44.86 a | 46.35 a | 75.56 b | 17.79 a | 18.84 a | 8877.80 b | 18.70 b | 808.89 b | |
| 5 | 70.66 b | 13.65 a,b,* | 50.26 b | 52.11 b | 75.01 b | 23.45 b | 20.71 a | 10,944.46 c | 19.18 c | 1221.85 c | |
| NP | - | 57.15 a | 15.77 b | 43.13 a | 45.98 a | 70.28 a | 22.03 a,b,* | 27.30 b | 6194.46 a | 11.63 a | 531.11 a |
| P | 1 | 49.95 b | 32.55 b | 19.47 a | 38.12 a | 31.29 b | 35.23 b | 46.79 b | 12,438.91 b | 29.54 b | 1461.97 c |
| 3 | 45.24 a | 36.10 c | 16.11 a | 39.87 a,b,* | 23.88 a | 41.50 c | 53.66 b | 12,394.46 b | 32.17 c | 957.03 b | |
| 5 | 41.19 a | 39.24 c | 18.89 a | 43.64 b,c,* | 25.62 a | 45.61 c | 63.15 c | 13,852.80 c | 32.49 c | 1886.66 d | |
| NP | - | 57.15 c | 15.77 a | 43.13 b | 45.98 c | 70.28 c | 22.03 a | 27.30 a | 6194.46 a | 11.63 a | 531.11 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akman, H.E.; Boyar, I.; Gozlekci, S.; Saracoglu, O.; Ertekin, C. Effects of Convective Drying of Quince Fruit (Cydonia oblonga) on Color, Antioxidant Activity and Phenolic Compounds under Various Fruit Juice Dipping Pre-Treatments. Agriculture 2022, 12, 1224. https://doi.org/10.3390/agriculture12081224
Akman HE, Boyar I, Gozlekci S, Saracoglu O, Ertekin C. Effects of Convective Drying of Quince Fruit (Cydonia oblonga) on Color, Antioxidant Activity and Phenolic Compounds under Various Fruit Juice Dipping Pre-Treatments. Agriculture. 2022; 12(8):1224. https://doi.org/10.3390/agriculture12081224
Chicago/Turabian StyleAkman, Hasmet Emre, Ismail Boyar, Sadiye Gozlekci, Onur Saracoglu, and Can Ertekin. 2022. "Effects of Convective Drying of Quince Fruit (Cydonia oblonga) on Color, Antioxidant Activity and Phenolic Compounds under Various Fruit Juice Dipping Pre-Treatments" Agriculture 12, no. 8: 1224. https://doi.org/10.3390/agriculture12081224
APA StyleAkman, H. E., Boyar, I., Gozlekci, S., Saracoglu, O., & Ertekin, C. (2022). Effects of Convective Drying of Quince Fruit (Cydonia oblonga) on Color, Antioxidant Activity and Phenolic Compounds under Various Fruit Juice Dipping Pre-Treatments. Agriculture, 12(8), 1224. https://doi.org/10.3390/agriculture12081224







