Assessing Anti-Inflammatory Activities and Compounds in Switchgrass (Panicum virgatum)
Abstract
:1. Introduction
2. Materials & Methods
2.1. Switchgrass Collection
2.2. Sample Extraction
2.3. Identification of the Anti-Inflammatory Potential of Switchgrass
2.4. Identification and Qualification of Anti-Inflammatory Molecules in Switchgrass
2.5. Data Processing and Statistical Analysis
3. Results
3.1. Anti-Inflammatory Potential of Switchgrass
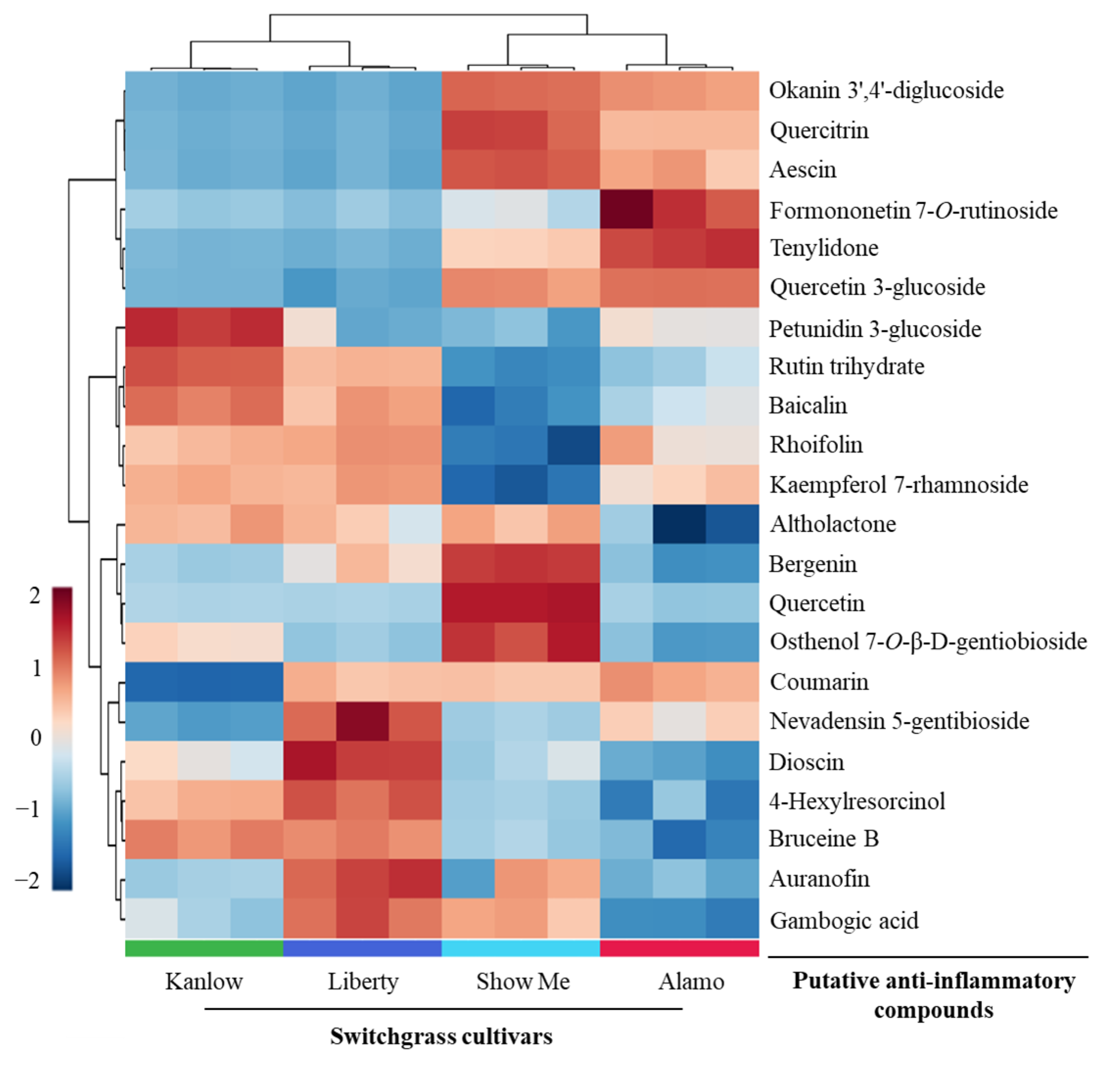
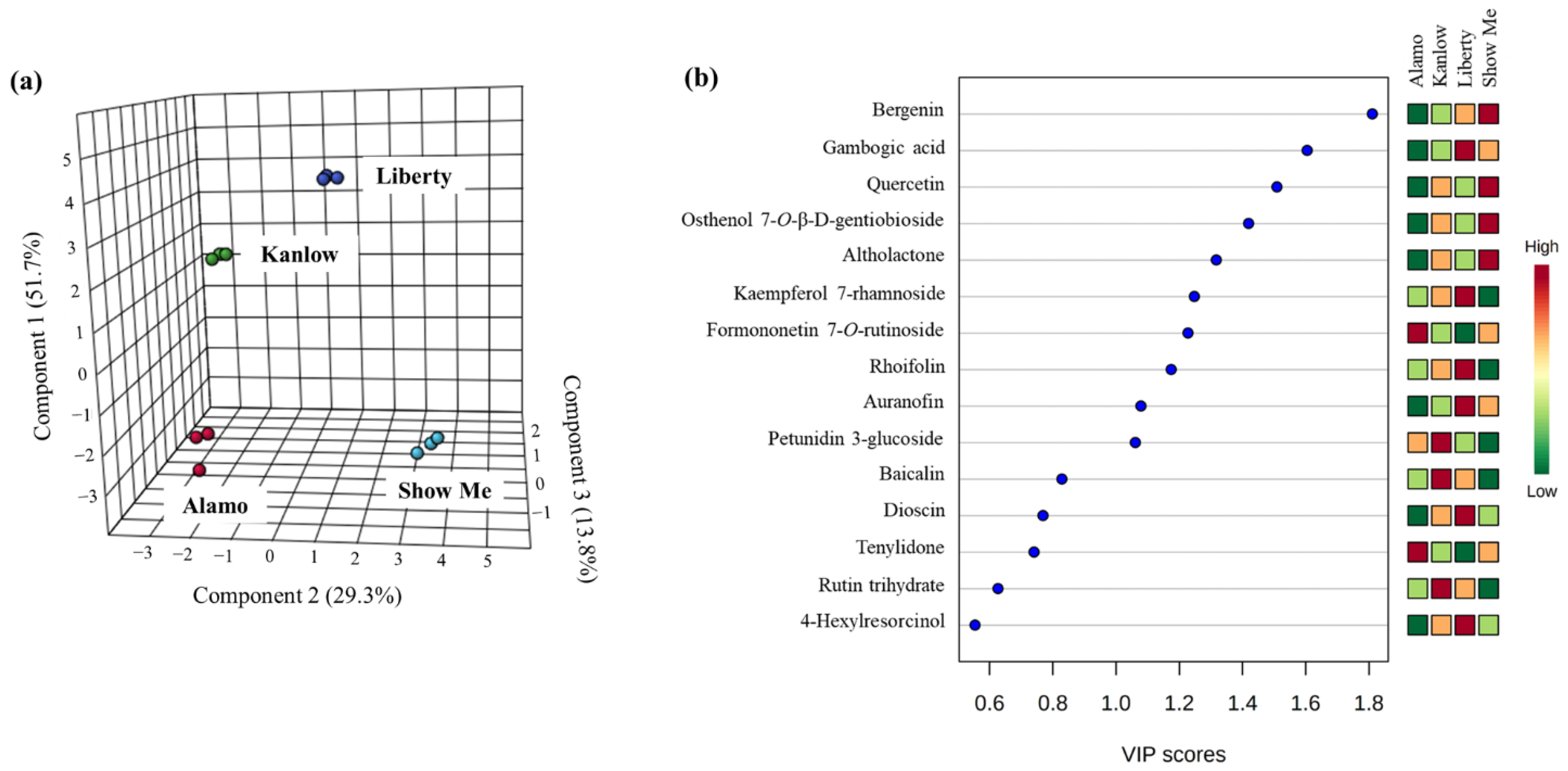
3.2. Anti-Inflammatory Compounds in Switchgrass
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| TNF-α | tumor necrosis factor alpha |
| IL-6 | interleukin-6 |
| IL-8 | interleukin-8 |
| IL-10 | interleukin-10 |
| PMA | phorbol 12-myristate 13-acetate |
| UHPLC | ultra-high-performance liquid chromatography |
| HRMS | high-resolution mass spectrometry |
| QTOF | quadrupole-time-of-flight |
| HPLC-MS/MS | liquid chromatography–tandem mass spectrometry |
| MTT | 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide |
References
- Parrish, D.J.; Fike, J.H. The biology and agronomy of switchgrass for biofuels. BPTS 2005, 24, 423–459. [Google Scholar] [CrossRef]
- Takemura, H.; Nagayoshi, H.; Matsuda, T.; Sakakibara, H.; Morita, M.; Matsui, A.; Ohura, T.; Shimoi, K. Inhibitory effects of chrysoeriol on DNA adduct formation with benzo [a] pyrene in MCF-7 breast cancer cells. Toxicology 2010, 274, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Larnaudie, V.; Ferrari, M.D.; Lareo, C. Switchgrass as an alternative biomass for ethanol production in a biorefinery: Perspectives on technology, economics and environmental sustainability. Renew. Sustain. Energy Rev. 2022, 158, 112115. [Google Scholar] [CrossRef]
- Shrestha, P.; Bellitürk, K.; Görres, J.H. Phytoremediation of heavy metal-contaminated soil by switchgrass: A comparative study utilizing different composts and coir fiber on pollution remediation, plant productivity, and nutrient leaching. Int. J. Environ. Res. Public Health 2019, 16, 1261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McKnight, A.M.; Gannon, T.W.; Yelverton, F. Phytoremediation potential of three terrestrial plant species for removal of atrazine, azoxystrobin, and imidacloprid. Int. J. Phytoremed. 2022, 24, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Kráľová, K.; Jampílek, J. Phytoremediation of Environmental Matrices Contaminated with Photosystem II-Inhibiting Herbicides. In Pesticides Bioremediation; Springer: Berlin/Heidelberg, Germany, 2022; pp. 31–80. [Google Scholar]
- Keshwani, D.R.; Cheng, J.J. Switchgrass for bioethanol and other value-added applications: A review. Bioresour. Technol. 2009, 100, 1515–1523. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wagner, C.; De Gezelle, J.; Komarnytsky, S. Celtic provenance in traditional herbal medicine of Medieval Wales and classical antiquity. Front. Pharmacol. 2020, 11, 105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, Z.; Sykes, R.; Davis, M.F.; Brummer, E.C.; Ragauskas, A.J. Chemical profiles of switchgrass. Bioresour. Technol. 2010, 101, 3253–3257. [Google Scholar] [CrossRef] [PubMed]
- Tao, J.; Rajan, K.; Ownley, B.; Gwinn, K.; D’Souza, D.; Moustaid-Moussa, N.; Tschaplinski, T.J.; Labbé, N. Natural variability and antioxidant properties of commercially cultivated switchgrass extractives. Ind. Crops Prod. 2019, 138, 111474. [Google Scholar] [CrossRef]
- Uppugundla, N.; Engelberth, A.; Vandhana Ravindranath, S.; Clausen, E.C.; Lay, J.O.; Gidden, J.; Carrier, D.J. Switchgrass water extracts: Extraction, separation and biological activity of rutin and quercitrin. J. Agric. Food Chem. 2009, 57, 7763–7770. [Google Scholar] [CrossRef] [PubMed]
- Lau, C.S.; Carrier, D.J.; Howard, L.R.; Lay, J.O.; Archambault, J.A.; Clausen, E.C. Extraction of antioxidant compounds from energy crops. Appl. Biochem. Biotechnol. 2004, 114, 569–583. [Google Scholar] [CrossRef]
- Medzhitov, R. Inflammation 2010: New adventures of an old flame. Cell 2010, 140, 771–776. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brenner, D.R.; Scherer, D.; Muir, K.; Schildkraut, J.; Boffetta, P.; Spitz, M.R.; Le Marchand, L.; Chan, A.T.; Goode, E.L.; Ulrich, C.M. A review of the application of inflammatory biomarkers in epidemiologic cancer research. Cancer Epidemiol. Biomark. Prev. 2014, 23, 1729–1751. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Labbe, N.; Ownley, B.H.; Gwinn, K.D.; Moustaid-Moussa, N.; D’souza, D.H. Antimicrobial and Anti-Inflammatory Activity of Switchgrass-Derived Extractives. U.S. Patent 9,282,747, 15 March 2016. [Google Scholar]
- Ho, K.-V.; Schreiber, K.L.; Vu, D.C.; Rottinghaus, S.M.; Jackson, D.E.; Brown, C.R.; Lei, Z.; Sumner, L.W.; Coggeshall, M.V.; Lin, C.-H. Black walnut (Juglans nigra) extracts inhibit proinflammatory cytokine production from lipopolysaccharide-stimulated human promonocytic cell line U-937. Front. Pharmacol. 2019, 10, 1059. [Google Scholar] [CrossRef] [PubMed]
- Ho, K.-V.; Lei, Z.; Sumner, L.; Coggeshall, M.; Hsieh, H.-Y.; Stewart, G.; Lin, C.-H. Identifying antibacterial compounds in black walnuts (Juglans nigra) using a metabolomics approach. Metabolites 2018, 8, 58. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ho, K.-V.; Schreiber, K.L.; Park, J.; Vo, P.; Lei, Z.; Sumner, L.W.; Brown, C.R.; Lin, C.-H. Identification and quantification of bioactive molecules inhibiting pro-inflammatory cytokine production in spent coffee grounds using metabolomics analyses. Front. Pharmacol. 2020, 11, 229. [Google Scholar] [CrossRef] [PubMed]
- Domingo-Almenara, X.; Siuzdak, G. Metabolomics data processing using XCMS. In Computational Methods and Data Analysis for Metabolomics; Springer: Berlin/Heidelberg, Germany, 2020; pp. 11–24. [Google Scholar]
- Pang, Z.; Chong, J.; Zhou, G.; de Lima Morais, D.A.; Chang, L.; Barrette, M.; Gauthier, C.; Jacques, P.-É.; Li, S.; Xia, J. MetaboAnalyst 5.0: Narrowing the gap between raw spectra and functional insights. Nucleic Acids Res. 2021, 49, W388–W396. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.-F.; Mowery, R.A.; Sevcik, R.S.; Scarlata, C.J.; Chambliss, C.K. Compositional analysis of water-soluble materials in switchgrass. J. Agric. Food Chem. 2010, 58, 3251–3258. [Google Scholar] [CrossRef]
- Yan, J.; Hu, Z.; Pu, Y.; Brummer, E.C.; Ragauskas, A.J. Chemical compositions of four switchgrass populations. Biomass Bioenergy 2010, 34, 48–53. [Google Scholar] [CrossRef]
- Hu, Z.; Foston, M.B.; Ragauskas, A.J. Biomass characterization of morphological portions of alamo switchgrass. J. Agric. Food Chem. 2011, 59, 7765–7772. [Google Scholar] [CrossRef] [PubMed]
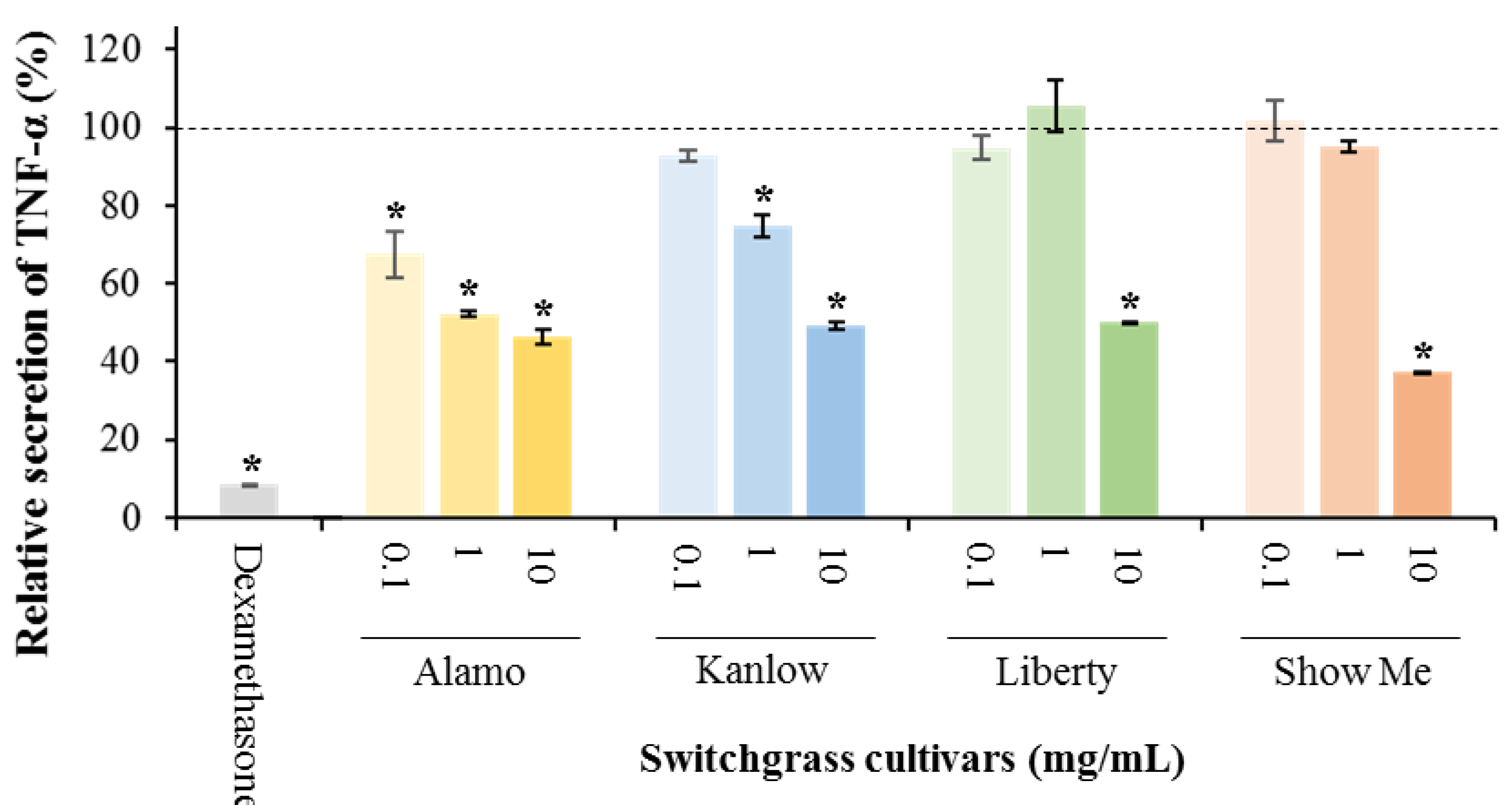
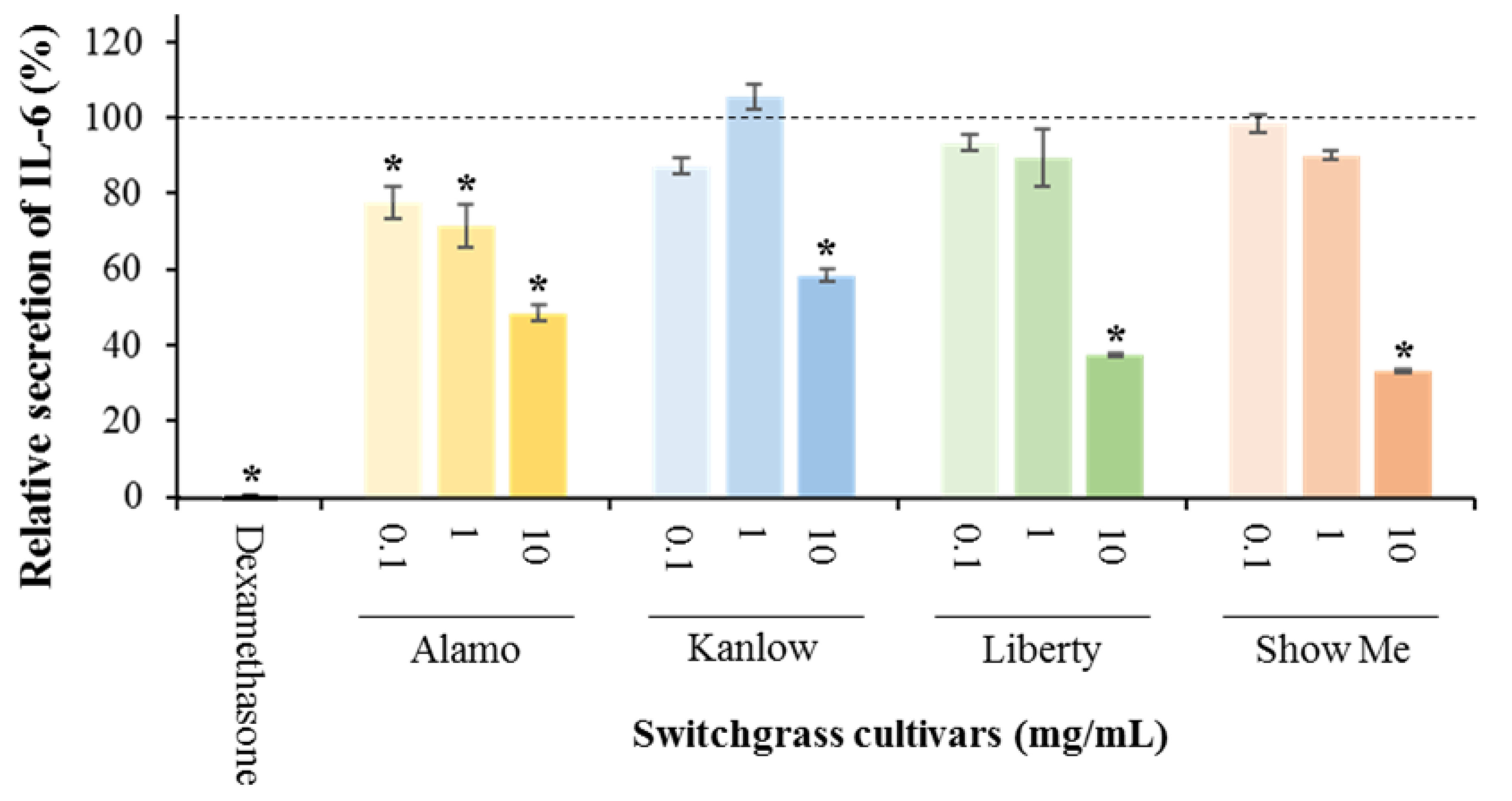
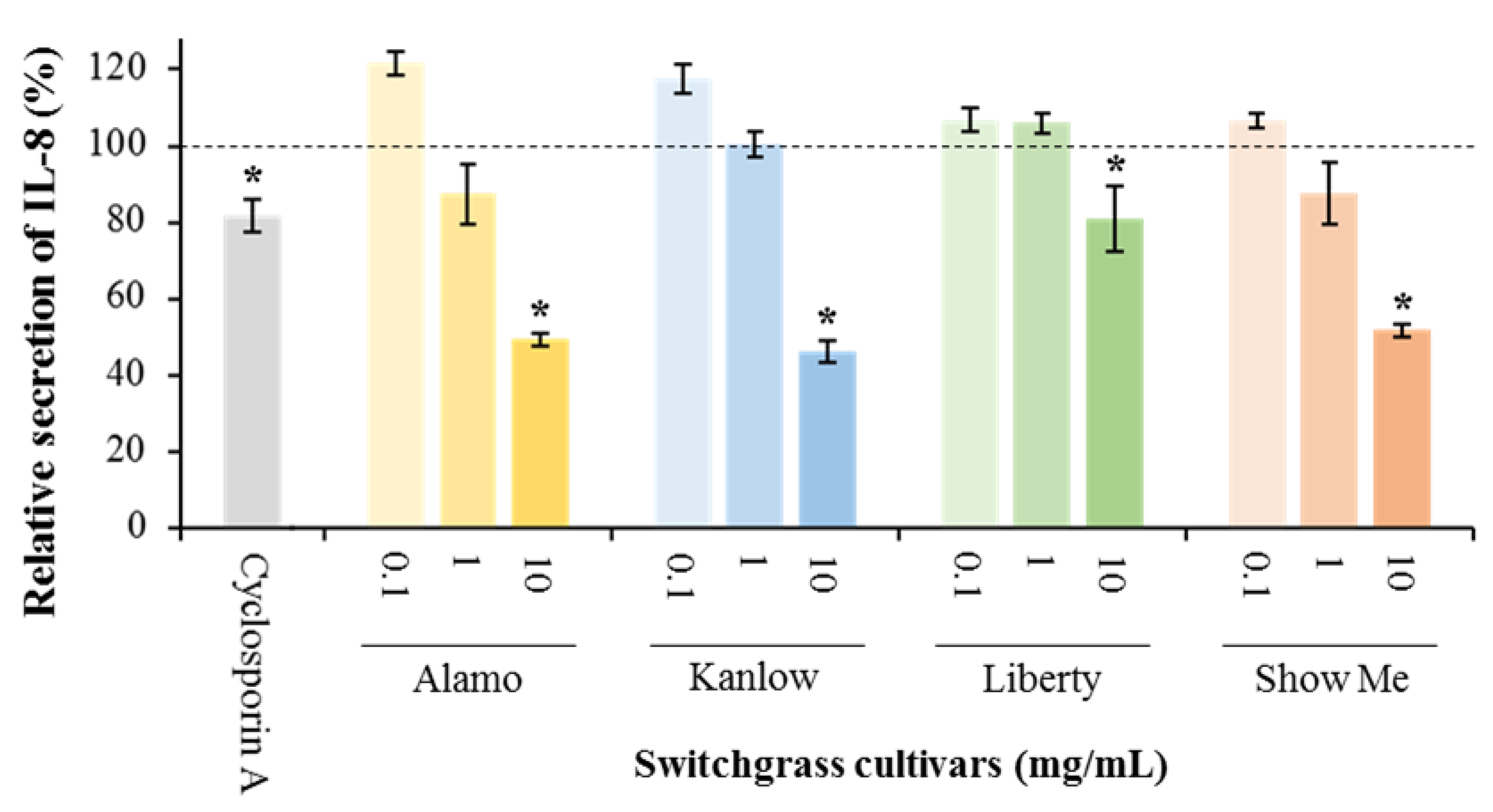
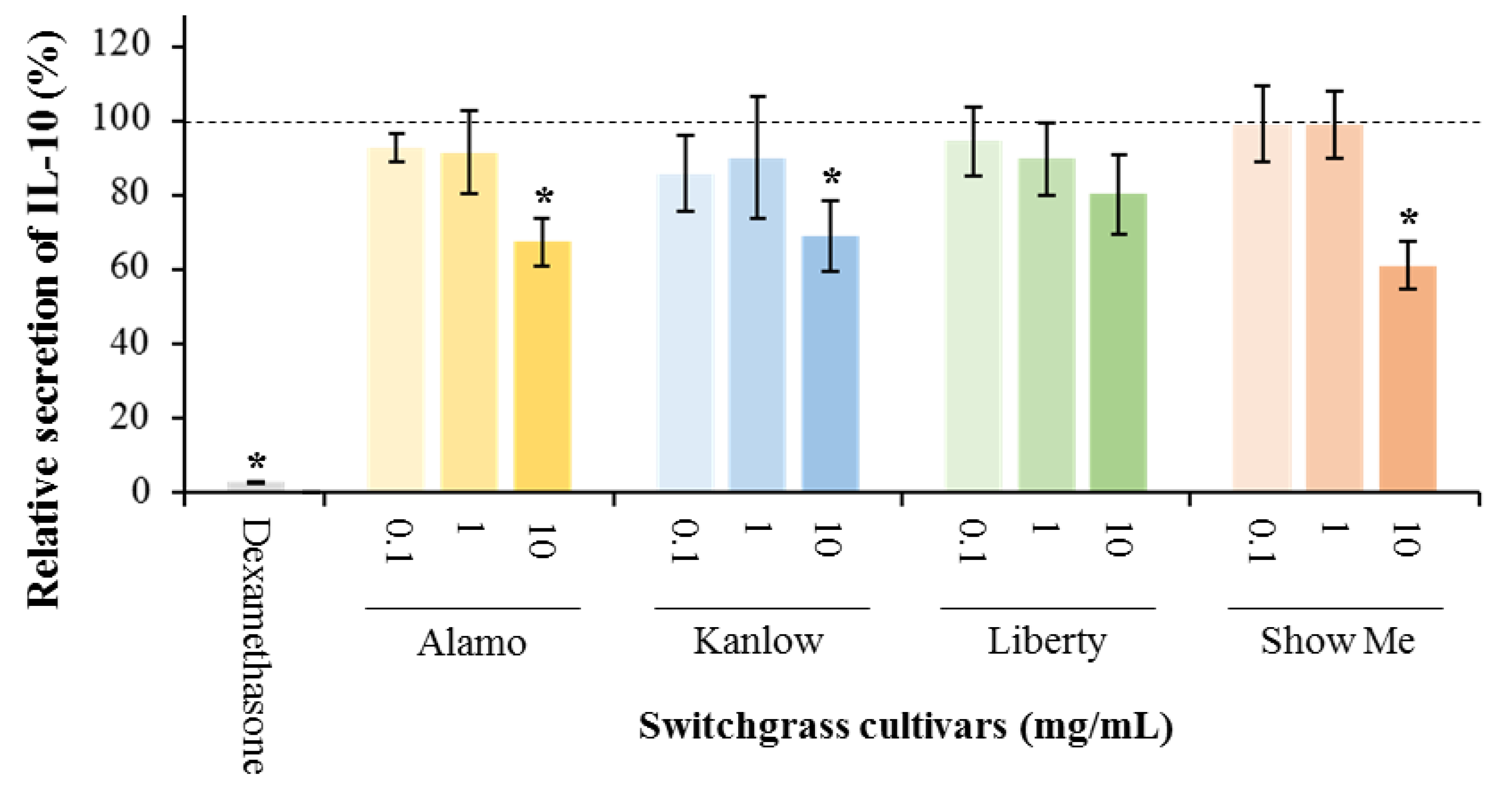
| Treatment | Cell Viability (%) |
|---|---|
| Vehicles/controls | |
| LPS (1 µg/mL) | 100.0 ± 4.6 |
| LPS (1 µg/mL) + DMSO (0.1%) | 101.9 ± 2.9 |
| LPS (1 µg/mL) + Ethanol (0.2%) | 99.1 ± 6.3 |
| Anti-inflammatory agents | |
| Dexamethasone (2 µg/mL) | 107.7 ± 3.6 |
| Cyclosporin A (2 µg/mL) | 99.1 ± 5.5 |
| Switchgrass cultivars | |
| Alamo (0.1 mg/mL) | 109.9 ± 6.7 |
| Alamo (1 mg/mL) | 95.5 ± 7.9 |
| Alamo (10 mg/mL) | 104.7 ± 3.0 |
| Kanlow (0.1 mg/mL) | 106.4 ± 2.7 |
| Kanlow (1 mg/mL) | 101.0 ± 4.3 |
| Kanlow (10 mg/mL) | 111.1 ± 8.9 |
| Liberty (0.1 mg/mL) | 127.2 ± 6.5 |
| Liberty (1 mg/mL) | 120.0 ± 14.8 |
| Liberty (10 mg/mL) | 107.1 ± 14.8 |
| Show Me (0.1 mg/mL) | 124.0 ± 7.3 |
| Show Me (1 mg/mL) | 111.0 ± 3.1 |
| Show Me (10 mg/mL) | 99.3 ± 2.3 |
| Compound | Retention Time (min) | Formula | Adducts | Theoretical Mass | Observed Mass | Δm (ppm) |
|---|---|---|---|---|---|---|
| 4-Hexylresorcinol | 5.75 | C12H18O2 | [M + H]+ | 194.1307 | 194.1302 | 2.68 |
| Aescin | 11.47 | C55H86O24 | [M − H]− | 1130.5509 | 1130.543 | 6.96 |
| Altholactone | 8.86 | C13H12O4 | [M + H]+ | 232.0736 | 232.0731 | 2.15 |
| Auranofin | 5.22 | C20H34AuO9PS | [M + H]+ | 678.1327 | 678.1335 | 1.2 |
| Baicalin | 6.24 | C21H18O11 | [M − H]− | 446.0849 | 446.0858 | 2.08 |
| Bergenin | 5.2 | C14H16O9 | [M + NH4]+ | 328.0794 | 328.0745 | 15.03 |
| Bruceine B | 4.56 | C23H28O11 | [M − H]− | 480.1632 | 480.1628 | 0.67 |
| Coumarin | 3.17 | C9H6O2 | [M + H]+ | 146.0368 | 146.0363 | 3.56 |
| Dioscin | 11.53 | C45H72O16 | [M + H]+ | 868.482 | 868.4838 | 1.98 |
| Formononetin 7-O-rutinoside | 8.49 | C30H36O17 | [M − H]− | 668.1952 | 668.1946 | 0.91 |
| Gambogic acid | 9.94 | C38H44O8 | [M − H]− | 628.3036 | 628.3045 | 1.47 |
| Kaempferol-7-rhamnoside | 7.34 | C21H20O10 | [M − H]− | 432.1056 | 432.1064 | 1.84 |
| Nevadensin 5-gentibioside | 2.29 | C30H36O17 | [M − H]− | 668.1952 | 668.1946 | 0.91 |
| Okanin 3′,4′-diglucoside | 6.96 | C27H32O16 | [M − H]− | 612.169 | 612.1704 | 2.3 |
| Osthenol-7-O-beta-D-gentiobioside | 4.64 | C26H34O13 | [M + Na]+ | 554.1999 | 554.1997 | 0.44 |
| Petunidin 3-glucoside | 6.47 | C22H23O12 | [M − H]− | 478.1111 | 478.112 | 1.91 |
| Quercetin | 6.22 | C15H10O7 | [M + H]+ | 302.0427 | 302.0423 | 1.3 |
| Quercetin-3-glucoside | 5.17 | C21H19O12 | [M − H]− | 464.0955 | 464.096 | 1.22 |
| Quercitrin | 7.17 | C21H20O11 | [M − H]− | 448.1006 | 448.1012 | 1.51 |
| Rhoifolin | 6.01 | C27H30O14 | [M + Na]+ | 578.1636 | 578.1643 | 1.29 |
| Rutin trihydrate | 4.65 | C27H36O19 | [M − H]− | 610.1534 | 610.1543 | 1.57 |
| Tenylidone | 6.09 | C16H14OS2 | [M + H]+ | 286.0486 | 286.0477 | 3.31 |
| Polyphenols | Switchgrass Cultivars | |||
|---|---|---|---|---|
| Alamo * | Kanlow | Liberty | Show Me | |
| Quercetin 3-glucoside | 346.3 ± 52.1 a | 260.6 ± 18.5 b | 215.8 ± 38.7 c | 155.9 ± 7.3 d |
| Quercetin | 0.2 ± 0.06 c | 0.7 ± 0.05 b | 0.1 ± 0.01 d | 0.8 ± 0.04 a |
| Rutin hydrate | 279.4 ± 17.0 a | 161.5 ± 11.9 c | 243.7 ± 13.7 b | 152.1 ± 1.1 c |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ho, K.-V.; Efrat, N.; Schreiber, K.L.; Vo, P.H.; De Canha, M.N.; van Staden, A.B.; Payne, B.D.; Oosthuizen, C.B.; Twilley, D.; Lei, Z.; et al. Assessing Anti-Inflammatory Activities and Compounds in Switchgrass (Panicum virgatum). Agriculture 2022, 12, 936. https://doi.org/10.3390/agriculture12070936
Ho K-V, Efrat N, Schreiber KL, Vo PH, De Canha MN, van Staden AB, Payne BD, Oosthuizen CB, Twilley D, Lei Z, et al. Assessing Anti-Inflammatory Activities and Compounds in Switchgrass (Panicum virgatum). Agriculture. 2022; 12(7):936. https://doi.org/10.3390/agriculture12070936
Chicago/Turabian StyleHo, Khanh-Van, Novianus Efrat, Kathy L. Schreiber, Phuc H. Vo, Marco N. De Canha, Analike Blom van Staden, Bianca D. Payne, Carel B. Oosthuizen, Danielle Twilley, Zhentian Lei, and et al. 2022. "Assessing Anti-Inflammatory Activities and Compounds in Switchgrass (Panicum virgatum)" Agriculture 12, no. 7: 936. https://doi.org/10.3390/agriculture12070936
APA StyleHo, K.-V., Efrat, N., Schreiber, K. L., Vo, P. H., De Canha, M. N., van Staden, A. B., Payne, B. D., Oosthuizen, C. B., Twilley, D., Lei, Z., Sumner, L. W., Brown, C. R., Lall, N., & Lin, C.-H. (2022). Assessing Anti-Inflammatory Activities and Compounds in Switchgrass (Panicum virgatum). Agriculture, 12(7), 936. https://doi.org/10.3390/agriculture12070936










