Lethality of Three Phasmarhabditis spp. (P. hermaphrodita, P. californica, and P. papillosa) to Succinea Snails
Abstract
1. Introduction
2. Materials and Methods
2.1. Arena Design
2.2. Preparation of Nematode Inocula
2.3. Experimental Setup
2.4. Statistical Analysis
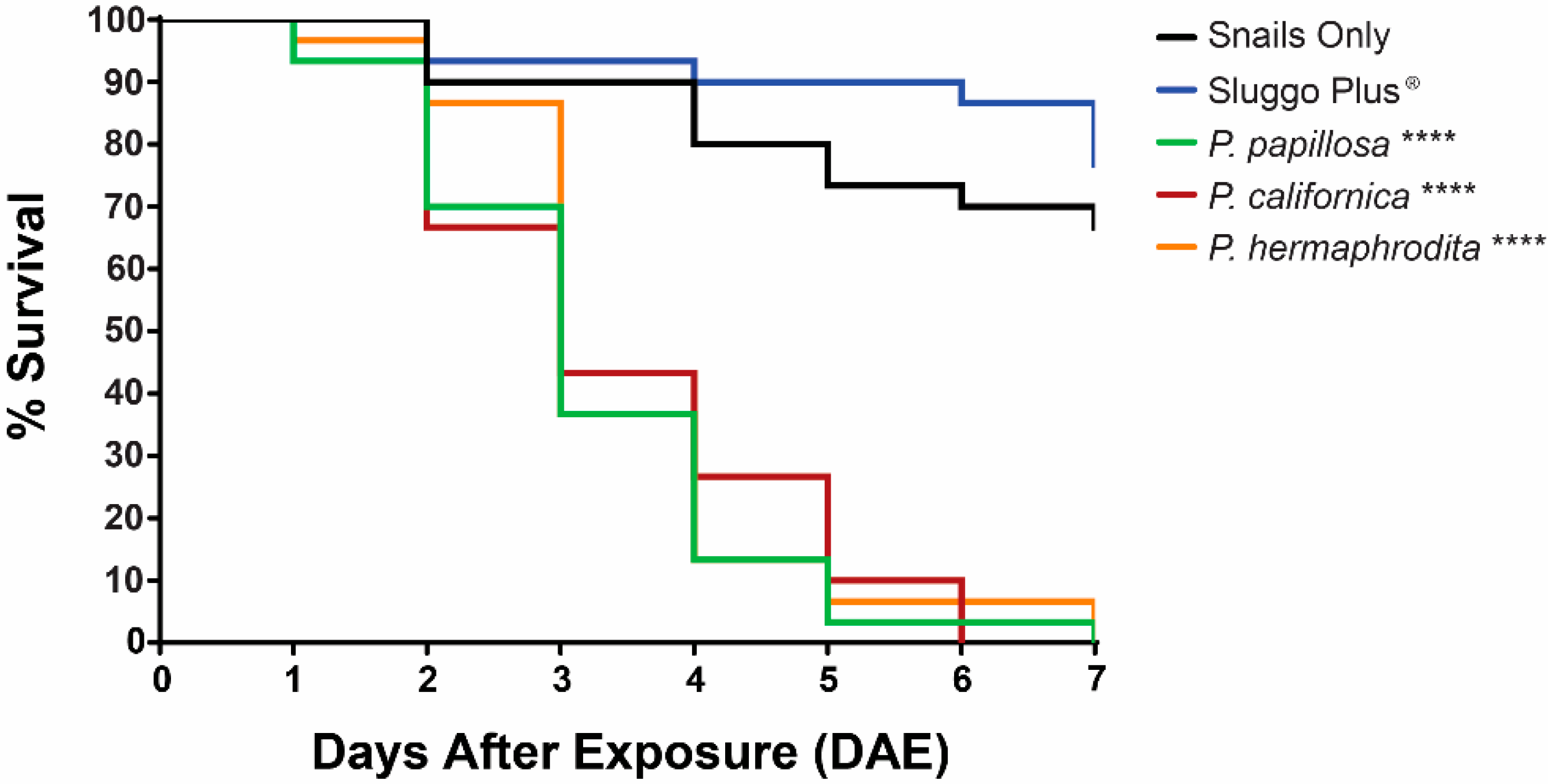
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sallam, A.; El-Wakeil, N. Biological and Ecological Studies on Land Snails and Their Control. In Pest Management and Pest Control-Current and Future Tactics; InTech Publishing: Shanghai, China, 2012; pp. 413–444. [Google Scholar]
- Jennings, T.J.; Barkham, J.P. Litter Decomposition by Slugs in Mixed Deciduous Woodland. Ecography 1979, 2, 21–29. [Google Scholar] [CrossRef]
- Prather, C.M.; Pelini, S.L.; Laws, A.; Rivest, E.; Woltz, M.; Bloch, C.P.; Del Toro, I.; Ho, C.K.; Kominoski, J.; Newbold, T.A.S.; et al. Invertebrates, Ecosystem Services and Climate Change. Biol. Rev. 2013, 88, 327–348. [Google Scholar] [CrossRef] [PubMed]
- Howlett, S.A. Terrestrial Slug Problems: Classical Biological Control and Beyond. CAB Rev. 2012, 7, 1–10. [Google Scholar] [CrossRef]
- Port, C.M. The Biology and Behavior of Slugs in Relation to Crop Damage and Control. Agric. Zool. Rev. 1986, 1, 255–299. [Google Scholar]
- Barker, G.M. Molluscs as Crop Pests; CABI Publishing: Wallingford, UK, 2002. [Google Scholar]
- Brooks, A.; Crook, M. The Use of Molluscicides in UK Agriculture and Their Effects on Non-Target Organisms. Malacol. Soc. Lond. Mini-Rev. 2003. Available online: https://malacsoc.org.uk/malacological_bulletin/Mini-Reviews/2Molluscicides/molluscicide.htm (accessed on 13 March 2022).
- Rose, J.H. Lungworms of the Domestic Pig and Sheep. Adv. Parasitol. 1973, 11, 559–599. [Google Scholar]
- Kim, J.R.; Hayes, K.A.; Yeung, N.W.; Cowie, R.H. Diverse Gastropod Hosts of Angiostrongylus cantonensis, the Rat Lungworm, Globally and with a Focus on the Hawaiian Islands. PLoS ONE 2014, 9, e94969. [Google Scholar] [CrossRef]
- Hasan, S.; Vago, C. Transmission of Alternaria brassicicola by Slugs. Plant Dis. Report. 1966, 50, 764–767. [Google Scholar]
- Turchetti, T.; Chelazzi, G. Possible Role of Slugs as Vectors of the Chestnut Blight Fungus. Eur. J. For. Pathol. 1984, 14, 125–127. [Google Scholar] [CrossRef]
- Wester, R.E.; Goth, R.W.; Webb, R.E. Transmission of Downy Mildew of Lima Beans by Slugs. Phytopathology 1964, 54, 749. [Google Scholar]
- Raloff, J. Lettuce Liability: Programs to Keep Salad Germ-Free, Raise Wildlife, and Conservation Concerns. Sci. News 2007, 172, 362–364. [Google Scholar] [CrossRef]
- Sproston, E.L.; Macrae, M.; Ogden, I.D.; Wilson, M.J.; Strachan, N.J.C. Slugs: Potential Novel Vectors of Escherichia coli O157. Appl. Environ. Microbiol. 2006, 72, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Tandingan De Ley, I.; Schurkman, J.; Wilen, C.; Dillman, A.R. Mortality of the Invasive White Garden Snail Theba pisana Exposed to Three US Isolates of Phasmarhabditis Spp. (P. hermaphrodita, P. californica, and P. papillosa). PLoS ONE 2020, 15, e0228244. [Google Scholar] [CrossRef] [PubMed]
- Schurkman, J.; Tandingan De Ley, I.; Dillman, A.R. Size and Dose Dependence of Phasmarhabditis Isolates (P. hermaphrodita, P. californica, P. papillosa) on the Mortality of Adult Invasive White Garden Snails (Theba pisana). PLoS ONE, 2022; in review. [Google Scholar]
- Hayes, K.; Tran, C.; Cowie, R. New Records of Alien Mollusca in the Hawaiian Islands: Non-Marine Snails and Slugs (Gastropoda) Associated with the Horticultural Trade; Bishop Museum Occasional Papers; Department of Biology Faculty Publications: Atlanta, GA, USA, 2007; p. 54. [Google Scholar]
- Robinson, D.G. Alien Invasions: The Effects of the Global Economy on Non Marine Gastropod Introductions into the United States. Malacologia 1999, 42, 413–438. [Google Scholar]
- Roth, B.; Sadeghian, P.S. Checklist of the Land Snails and Slugs of California; Santa Barbara Museum of Natural History: Santa Barbara, CA, USA, 2003; Volume 3, ISBN 0936494344. [Google Scholar]
- Schurkman, J.; Tandingan De Ley, I.; Anesko, K.; Paine, T.; Mc Donnell, R.; Dillman, A.R. Distribution of Phasmarhabditis (Nematoda: Rhabditidae) and Their Gastropod Hosts in California Plant Nurseries and Garden Centers. Front. Plant Sci. 2022, 811. [Google Scholar] [CrossRef]
- Abo-Zaid, A.; El-Hawary, I.S.; Mahrous, M.E.; El-Sheikh, M.F. Field Observation on Biology and Ecology of Terrestrial Snails Infesting Field and Vegetable Crops at Gharbia Governorate. Egypt. Acad. J. Biol. Sci. 2021, 13, 95–103. [Google Scholar] [CrossRef]
- Rosetta, R.; Coupland, J.; Wells, D.; Mack, C. Investigation of Amber Snail Management Treatments in Nursery Production Facilities; Oregon State University: Corvallis, OR, USA, 2011. [Google Scholar]
- Gurr, G.M.; Wratten, S.D.; Barbosa, P. Success in Conservation Biological Control of Arthropods. In Biological Control: Measures of Success; Springer: Singapore, 2000; pp. 105–132. [Google Scholar]
- South, A. Terrestrial Slugs: Biology, Ecology and Control; Chapman & Hall: London, UK, 1992. [Google Scholar]
- Salgado, V.L. Studies on the Mode of Action of Spinosad: Insect Symptoms and Physiological Correlates. Pestic. Biochem. Physiol. 1998, 60, 91–102. [Google Scholar] [CrossRef]
- Triebskorn, R.A.; Henderson, I.F.; Martin, A.P. Detection of Iron in Tissues from Slugs (Deroceras reticulatum Müller) after Ingestion of Iron Chelates, by Means of Energy-Filtering Transmission Electron Microscopy (EFTEM). Pestic. Sci. 1999, 55, 55–61. [Google Scholar] [CrossRef]
- Edwards, C.A.; Arancon, N.Q.; Vasko-Bennett, M.; Little, B.; Askar, A. The Relative Toxicity of Metaldehyde and Iron Phosphate Based Molluscicides to Earthworms. Crop Prot. 2009, 28, 289–294. [Google Scholar] [CrossRef]
- Langan, A.M.; Shaw, E.M. Responses of Earthworm Lumbricus terrestris (L.) to Iron Phosphate and Metaldehyde Slug Pellet Formulations. Appl. Soil Ecol. 1993, 34, 184–189. [Google Scholar] [CrossRef]
- Tandingan De Ley, I.; McDonnell, R.; Lopez, S.; Paine, T.D.; De Ley, P. Phasmarhabditis hermaphrodita (Nematoda: Rhabditidae), a Potential Biocontrol Agent Isolated for the First Time in North America. Nematology 2014, 16, 1129–1138. [Google Scholar] [CrossRef]
- Tandingan De Ley, I.; McDonnell, R.J.; Aronson, E.; Wilen, C. Discovery of Multiple Phasmarhabditis spp. in North America and Description of Phasmarhabditis californica n. sp. and First Report of P. papillosa (Nematoda: Rhabditidae) from Invasive Slugs in the USA. Nematology 2016, 18, 175–193. [Google Scholar] [CrossRef]
- Mc Donnell, R.J.; Lutz, M.S.; Howe, D.K.; Denver, D.R. First Report of the Gastropod-Killing Nematode, Phasmarhabditis hermaphrodita, in Oregon, USA. J. Nematol. 2018, 50, 77. [Google Scholar] [CrossRef]
- Coupland, J.B. Susceptibility of Helicid Snails to Isolates of the Nematode Phasmarhabditis hermaphrodita from Southern France. J. Invertebr. Pathol. 1995, 66, 207–208. [Google Scholar] [CrossRef]
- Rae, R.; Verdun, C.; Grewal, P.S.; Robertson, J.F.; Wilson, M.J. Biological Control of Terrestrial Molluscs Using Phasmarhabditis hermaphrodita Progress and Prospects. Pest Manag. Sci. 2007, 63, 1153–1164. [Google Scholar] [CrossRef] [PubMed]
- Schurkman, J.; Dodge, C.; McDonnell, R.; Tandingan De Ley, I.T.; Dillman, A.R. Lethality of Phasmarhabditis spp. (P. hermaphrodita, P. californica, and P. papillosa) Nematodes to the Grey Field Slug Deroceras reticulatum on Canna Lilies in a Lath House. Agronomy 2022, 12, 20. [Google Scholar] [CrossRef]
- Wilson, M.J.; Glen, S.K.; George, S.K.; Pearce, J.D.; Wiltshire, C.W. Biological Control of Slugs in Winter Wheat Using the Rhabditid Nematode Phasmarhabditis hermaphrodita. Ann. Appl. Biol. 1994, 125, 377–390. [Google Scholar] [CrossRef]
- Wilson, M.J.; Rae, R. Phasmarhabditis hermaphrodita as a Control Agent for Slugs. In Nematode Pathogenesis of Insects and Other Pests. Sustainability in Plant and Crop Protection; Campos-Herrera, R., Ed.; Springer: Cham, Switzerland, 2015. [Google Scholar]
- Grewal, S.K.; Grewal, P.S. Survival of Earthworms Exposed to the Slug-Parasitic Nematode Phasmarhabditis hermaphrodita. J. Invertebr. Pathol. 2003, 82, 72–74. [Google Scholar] [CrossRef]
- DeNardo, E.A.B.; Sindermann, A.B.; Grewal, S.K.; Grewal, P.S. Non-Susceptibility of Earthworm Eisenia fetida to the Rhabditid Nematode Phasmarhabditis hermaphrodita, a Biocontrol Agent of Slugs. Biol. Sci. Technol. 2004, 14, 93–98. [Google Scholar] [CrossRef]
- Rae, R.G.; Robertson, J.; Wilson, M.J.; Rae, R.G.; Robertson, J. Susceptibility of Indigenous UK Earthworms and an Invasive Pest Flatworm to the Slug Parasitic Nematode Phasmarhabditis hermaphrodita. Biocontrol Sci. Technol. 2007, 15, 623–626. [Google Scholar] [CrossRef]
- Wilson, M.J.; Hughes, L.A.; Hamacher, G.M.; Glen, D.M. Effects of Phasmarhabditis hermaphrodita on Non-Target Molluscs. Pest Manag. Sci. 2000, 56, 711–716. [Google Scholar] [CrossRef]
- Christensen, C.C.; Cowie, R.H.; Yeung, N.W.; Hayes, K.A. Biological control of pest non-marine molluscs: A pacific perspective on risks to non-target organisms. Insects 2021, 12, 583. [Google Scholar] [CrossRef] [PubMed]
- Mc Donnell, R.; Tandingan De Ley, I.; Paine, T.D. Susceptibility of Neonate Lissachatina fulica (Achatinidae: Mollusca) to a US Strain of the Nematode Phasmarhabditis hermaphrodita (Rhabditidae: Nematoda). Biocontrol Sci. Technol. 2018, 28, 1091–1095. [Google Scholar] [CrossRef]
- Mc Donnell, R.J.; Colton, A.J.; Howe, D.K.; Denver, D.R. Lethality of Four Species of Phasmarhabditis (Nematoda: Rhabditidae) to the Invasive Slug, Deroceras reticulatum (Gastropoda: Agriolimacidae) in Laboratory Infectivity Trials. Biol. Control. 2020, 150, 104349. [Google Scholar] [CrossRef]
- Matkin, O.A.; Chandler, P.A. The U.C.-Type Soil Mixes. In The UC System for Producing Healthy Container-Grown Plants through the Use of Clean Soil, Clean Stock, and Sanitation. Manual 23; Baker, K., Ed.; California Agricultural Experiment Station [and California Agricultural] Extension Service: Berkeley, CA, USA, 1957; pp. 68–85. [Google Scholar]
- Kaya, H.K.; Stock, S.P. Techniques in Insect Nematology. In Manual of Techniques in Insect Pathology; Lacey, L.A., Ed.; Academic Press: San Diego, CA, USA, 1997; pp. 281–324. [Google Scholar]
- Grannell, A.; Cutler, J.; Rae, R. Size-Susceptibility of Cornu aspersum Exposed to the Malacopathogenic Nematodes Phasmarhabditis hermaphrodita and P. californica. Biocontrol Sci. Technol. 2021, 31, 1149–1160. [Google Scholar] [CrossRef]
- Speiser, B.; Zaller, J.G.; Neudecker, A. Size-Specific Susceptibility of the Pest Slugs Deroceras reticulatum and Arion lusitanicus to the Nematode Biocontrol Agent Phasmarhabditis hermaphrodita. BioControl 2001, 46, 311–320. [Google Scholar] [CrossRef]

| Succinea sp. | County | Reference |
|---|---|---|
| S. california Groose and Fisher, 1878 | Monterey, San Diego | [19] |
| S. luteola Gould 1846 * | Solano | [19] |
| S. rusticana Gould 1846 | Sonoma, San Francisco, San Benito, Kern, San Luis Obispo | [19] |
| Succinea spp. | Orange, Sacramento, San Diego, Riverside, San Francisco, Solano, Los Angeles | Mc Donnell and Wilen, unpublished data (UCANR) ** |
| Succinea spp. | Humboldt, Stanislaus, Tulare, San Bernardino, San Diego | [20] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schurkman, J.; Tandingan De Ley, I.; Dillman, A.R. Lethality of Three Phasmarhabditis spp. (P. hermaphrodita, P. californica, and P. papillosa) to Succinea Snails. Agriculture 2022, 12, 837. https://doi.org/10.3390/agriculture12060837
Schurkman J, Tandingan De Ley I, Dillman AR. Lethality of Three Phasmarhabditis spp. (P. hermaphrodita, P. californica, and P. papillosa) to Succinea Snails. Agriculture. 2022; 12(6):837. https://doi.org/10.3390/agriculture12060837
Chicago/Turabian StyleSchurkman, Jacob, Irma Tandingan De Ley, and Adler R. Dillman. 2022. "Lethality of Three Phasmarhabditis spp. (P. hermaphrodita, P. californica, and P. papillosa) to Succinea Snails" Agriculture 12, no. 6: 837. https://doi.org/10.3390/agriculture12060837
APA StyleSchurkman, J., Tandingan De Ley, I., & Dillman, A. R. (2022). Lethality of Three Phasmarhabditis spp. (P. hermaphrodita, P. californica, and P. papillosa) to Succinea Snails. Agriculture, 12(6), 837. https://doi.org/10.3390/agriculture12060837







