Effects of Rocket Seed Oil, Wheat Germ Oil, and Their Mixture on Growth Performance, Feed Utilization, Digestibility, Redox Status, and Meat Fatty Acid Profile of Growing Rabbits
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal Management and Feeding
2.2. Body Weight and Feed Intake
2.3. Digestibility Trial
2.4. Lipid Content and Fatty Acid Profile of Rabbit Meat
2.5. Serum Biochemical Parameters
2.6. Antioxidant Assays
2.7. Antibody Titers against SRBCs
2.8. Chemical Analysis
2.9. Statistical Analysis
3. Results
3.1. Body Weight, Feed Intake, and Feed Conversion Ratio
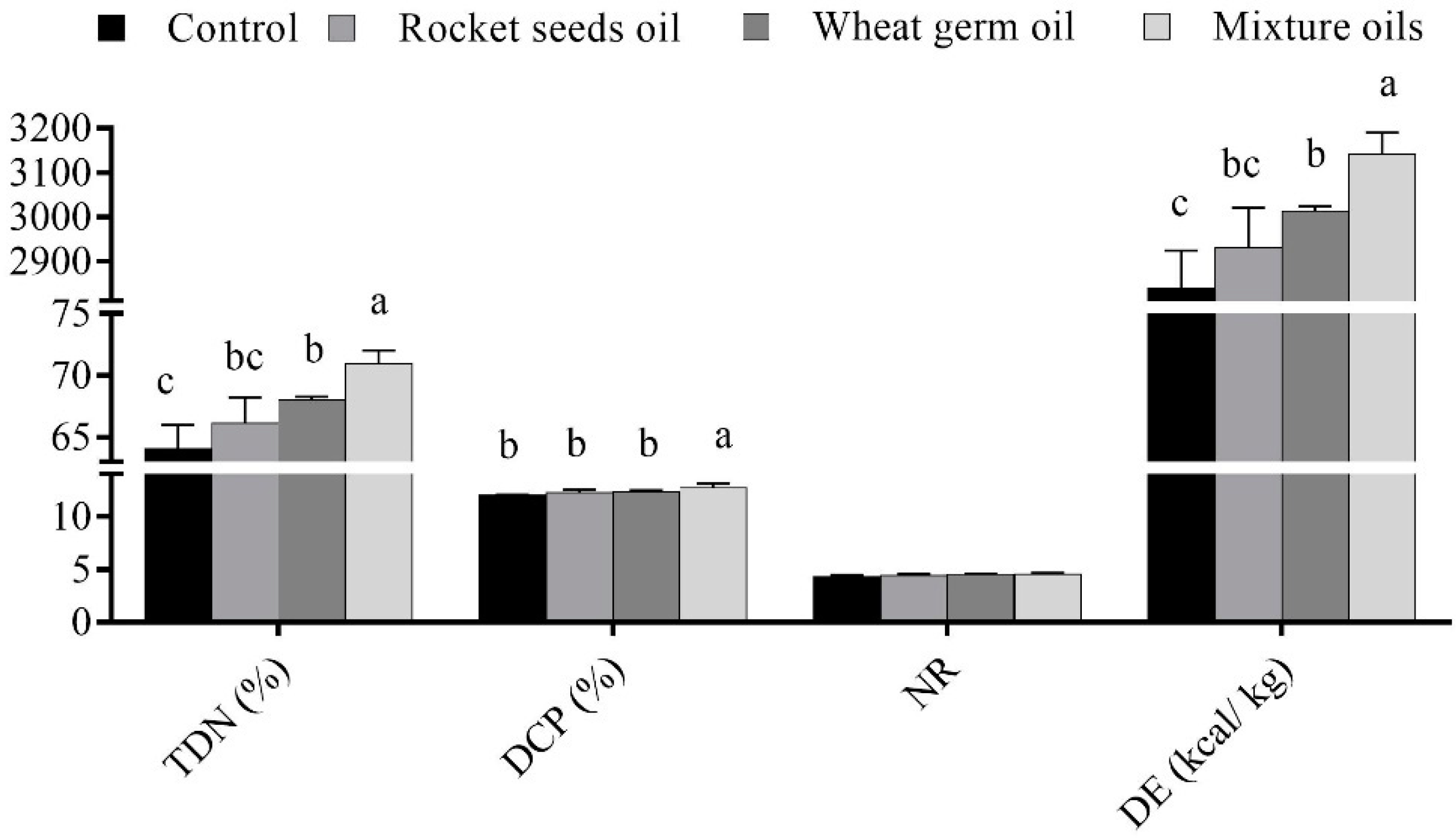
3.2. Digestion Coefficients of Nutrient, Nutritive Values, and Nitrogen Balance
3.3. Lipid Content and Fatty Acids Profile of Rabbit Meat
3.4. Serum Biochemical Parameters
3.5. Antioxidant Status
3.6. Antibody Titers against SRBCs
4. Discussion
4.1. Growth Performance and Feed Utilization
4.2. Digestion Coefficients of Nutrient, Nutritive Values, and Nitrogen Balance
4.3. Lipid Content and Fatty Acid Profile of Rabbit Meat
4.4. Serum Biochemical Parameters
4.5. Antioxidants
4.6. Antibody Titers against SRBCs
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Madhupriya, V.; Shamsudeen, P.; Manohar, G.R.; Senthilkumar, S.; Soundarapandiyan, V.; Moorthy, M. Phyto Feed Additives in Poultry Nutrition: A Review. Int. J. Sci. Environ. Technol. 2018, 7, 815–822. [Google Scholar]
- Mansour, A.T.; Miao, L.; Espinosa, C.; García-Beltrán, J.M.; Francisco, D.C.C.; Esteban, M.Á. Effects of dietary inclusion of Moringa oleifera leaves on growth and some systemic and mucosal immune parameters of seabream. Fish Physiol. Bioch. 2018, 44, 1223–1240. [Google Scholar] [CrossRef] [PubMed]
- Mansour, A.T.; Espinosa, C.; García-Beltrán, J.M.; Miao, L.; Francisco, D.C.C.; Alsaqufi, A.S.; Esteban, M.Á. Dietary supplementation of drumstick tree, Moringa oleifera, improves mucosal immune response in skin and gills of seabream, Sparus aurata, and attenuates the effect of hydrogen peroxide exposure. Fish Physiol. Bioch. 2020, 46, 981–996. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.-Z.; Dersjant-Li, Y.; Giannenas, I. Application of aromatic plants and their extracts in diets of broiler chickens. In Feed Additives; Elsevier: Amsterdam, The Netherlands, 2020; pp. 159–185. [Google Scholar] [CrossRef]
- Al-Suwaiegh, S.B.; Morshedy, S.A.; Mansour, A.T.; Ahmed, M.H.; Zahran, S.M.; Alnemr, T.M.; Sallam, S. Effect of an essential oil blend on dairy cow performance during treatment and post-treatment perioSds. Sustainability 2020, 12, 9123. [Google Scholar] [CrossRef]
- Tres, A.; Bou, R.; Codony, R.; Guardiola, F. Dietary n-6-or n-3-rich vegetable fats and α-tocopheryl acetate: Effects on fatty acid composition and stability of rabbit plasma, liver and meat. Animal 2009, 3, 1408–1419. [Google Scholar] [CrossRef]
- Niu, L.-Y.; Jiang, S.-T.; Pan, L.-J.; Pang, M. Characterization of wheat germ oil in terms of volatile compounds, lipid composition, thermal behavior, and structure. Int. J. Food Prop. 2013, 16, 1740–1749. [Google Scholar] [CrossRef]
- nez de la Ossa, E.M. Quality of wheat germ oil extracted by liquid and supercritical carbon dioxide. J. Am. Oil Chem. Soc. 2000, 77, 969–974. [Google Scholar]
- Taffner, J.; Cernava, T.; Erlacher, A.; Berg, G. Novel insights into plant-associated archaea and their functioning in arugula (Eruca sativa Mill.). J. Adv. Res. 2019, 19, 39–48. [Google Scholar] [CrossRef]
- Garg, G.; Sharma, V. Eruca sativa (L.): Botanical description, crop improvement, and medicinal properties. J. Herbs Spices Med. Plants 2014, 20, 171–182. [Google Scholar] [CrossRef]
- Nail, T.; Ali, M.; Salim, E. Phytochemical studies on Sudanese rocket (Eruca sativa) seeds and oil constituents. Am. J. Phytomed. Clin. Ther. 2017, 5, 1–5. [Google Scholar]
- El-Missiry, M.; El Gindy, A. Amelioration of alloxan induced diabetes mellitus and oxidative stress in rats by oil of Eruca sativa seeds. Ann. Nutr. Metab. 2000, 44, 97–100. [Google Scholar] [CrossRef] [PubMed]
- Khoobchandani, M.; Ganesh, N.; Gabbanini, S.; Valgimigli, L.; Srivastava, M. Phytochemical potential of Eruca sativa for inhibition of melanoma tumor growth. Fitoterapia 2011, 82, 647–653. [Google Scholar] [CrossRef]
- Gulfraz, M.; Sadiq, A.; Tariq, H.; Imran, M.; Qureshi, R.; Zeenat, A. Phytochemical analysis and antibacterial activity of Eruca sativa seed. Pak. J. Bot 2011, 43, 1351–1359. [Google Scholar]
- Hanafi, E.M.; Hegazy, E.M.; Riad, R.M.; Amer, H. Bio-protective effect of Eruca sativa seed oil against the hazardus effect of aflatoxin B1 in male-rabbits. Int. J. Acad. Res. 2010, 2, 67–74. Available online: https://www.semanticscholar.org/paper/BIO-PROTECTIVE-EFFECT-OF-ERUCA-SATIVA-SEED-OIL-THE-Hanafi-Hegazy/bced05dca22edbe48ed0208cb717442df5d76ab3 (accessed on 17 March 2021).
- El-Tohamy, M.M.; El-Nattat, W.; El-Kady, R. The beneficial effects of Nigella sativa, Raphanus sativus and Eruca sativa seed cakes to improve male rabbit fertility, immunity and production. Am. J. Sci. 2010, 6, 1247–1255. [Google Scholar]
- Ezzat, W.; Hamed, S. Influence of using rocket seed (Eruca sative) oil and onion seed (Allium cepa) oil on productive and physiological performance of growing rabbits under hot climate condition. J. Product Dev. (Agri. Res.) 2012, 17, 127–148. [Google Scholar]
- Alagawany, M.; El-Hack, A.; Mohamed, E.; Al-Sagheer, A.A.; Naiel, M.A.; Saadeldin, I.M.; Swelum, A.A. Dietary cold pressed watercress and coconut oil mixture enhances growth performance, intestinal microbiota, antioxidant status, and immunity of growing rabbits. Animals 2018, 8, 212. [Google Scholar] [CrossRef] [PubMed]
- Zeb, A.; Rahman, L. Eruca sativa seed oil: Characterization for potential beneficial properties. Pak. J. Pharm. Sci. 2018, 31, 1251–1258. [Google Scholar]
- Ge, Y.; Sun, A.; Ni, Y.; Cai, T. Some nutritional and functional properties of defatted wheat germ protein. J. Agric. Food Chem. 2000, 48, 6215–6218. [Google Scholar] [CrossRef]
- Brandolini, A.; Hidalgo, A. Wheat germ: Not only a by-product. Int. J. Food Sci. Nutr. 2012, 63, 71–74. [Google Scholar] [CrossRef]
- Hassanein, M.M.M.; Abedel-Razek, A.G. Chromatographic quantitation of some bioactive minor components in oils of wheat germ and grape seeds produced as by-products. J. Oleo Sci. 2009, 58, 227–233. [Google Scholar] [CrossRef]
- Alessandri, C.; Pignatelli, P.; Loffredo, L.; Lenti, L.; Del Ben, M.; Carnevale, R.; Perrone, A.; Ferro, D.; Angelico, F.; Violi, F. Alpha-linolenic acid–rich wheat germ oil decreases oxidative stress and CD40 ligand in patients with mild hypercholesterolemia. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 2577–2578. [Google Scholar] [CrossRef] [PubMed]
- Leenhardt, F.; Fardet, A.; Lyan, B.; Gueux, E.; Rock, E.; Mazur, A.; Chanliaud, E.; Demigné, C.; Rémésy, C. Wheat germ supplementation of a low vitamin E diet in rats affords effective antioxidant protection in tissues. J. Am. Coll. Nutr. 2008, 27, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Małecka, M. Antioxidant properties of the unsaponifiable matter isolated from tomato seeds, oat grains and wheat germ oil. Food Chem. 2002, 79, 327–330. [Google Scholar] [CrossRef]
- Vaher, M.; Matso, K.; Levandi, T.; Helmja, K.; Kaljurand, M. Phenolic compounds and the antioxidant activity of the bran, flour and whole grain of different wheat varieties. Procedia Chem. 2010, 2, 76–82. [Google Scholar] [CrossRef]
- Chadha, R.; Ram, H.; Purohit, A. Hypolipidemic effect of wheat germ oil in cholesterol fed Rabbits. Med. Drug Res. 2015, 3, 15–20. [Google Scholar]
- Zalatnai, A.; Lapis, K.; Szende, B.; Rásó, E.; Telekes, A.; Resetár, A.; Hidvégi, M. Wheat germ extract inhibits experimental colon carcinogenesis in F-344 rats. Carcinogenesis 2001, 22, 1649–1652. [Google Scholar] [CrossRef]
- Arshad, M.S.; Anjum, F.M.; Khan, M.I.; Shahid, M.; Akhtar, S.; Sohaib, M. Wheat germ oil enrichment in broiler feed with α-lipoic acid to enhance the antioxidant potential and lipid stability of meat. Lipids Health Dis. 2013, 12, 164. [Google Scholar] [CrossRef] [PubMed]
- NRC. Nutrient Requirements of Rabbits: 1977; National Academies Press: Washington, DC, USA, 1977. [Google Scholar]
- Cheeke, P.; Patton, N.; Tempelton, G. Rabbit Production, 5th ed.; FAO: Roma, Italy, 1982; ISBN 08-134-22221. [Google Scholar]
- Schneider, B.H.; Flatt, W.P. The Evaluation of Feeds through Digestibility Experiments; University of Georgia Press: Athens, GA, USA, 1975; p. 423. [Google Scholar]
- Folch, J.; Lees, M.; Stanley, G.S. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef]
- Pearson. Chemical Analysis of Food, 8th ed.; Church Hill Livingdtone: London, UK, 1981. [Google Scholar]
- Radwan, S. Coupling of two-dimensional thin-layer chromatography with gas chromatography for the quantitative analysis of lipid classes and their constituent fatty acids. J. Chromatogr. Sci. 1978, 16, 538–542. [Google Scholar] [CrossRef]
- Doumas, B.T.; Watson, W.A.; Biggs, H.G. Albumin standards and the measurement of serum albumin with bromcresol green. Clin. Chim. Acta 1971, 31, 87–96. [Google Scholar] [CrossRef]
- Coles, E.H. Kidney function tests In Vet Clin Pathol, 2nd ed.; W.B. Saunders Company: Philadelphia, PA, USA; London, UK, 1974. [Google Scholar]
- Zollner, N.; Kirsch, K. Colorimetric method for determination of total lipids. J. Exp. Med. 1962, 135, 545–550. [Google Scholar]
- Fossati, P.; Prencipe, L. Serum triglycerides determined colorimetrically with an enzyme that produces hydrogen peroxide. Clin. Chem. 1982, 28, 2077–2080. [Google Scholar] [CrossRef]
- Allain, C.C.; Poon, L.S.; Chan, C.S.G.; Richmond, W.; Fu, P.C. Enzymatic determination of total serum cholesterol. Clin. Chem. 1974, 20, 470–475. [Google Scholar] [CrossRef]
- Grove, T.H. Effect of reagent pH on determination of high-density lipoprotein cholesterol by precipitation with sodium phosphotungstate-magnesium. Clin. Chem. 1979, 25, 560–564. [Google Scholar] [CrossRef] [PubMed]
- Warnick, G.; Benderson, J.; Albers, J. Interlaboratory proficiency survey of high-density lipoprotein cholesterol measurement. Clin. Chem. 1983, 29, 516–519. [Google Scholar] [CrossRef]
- Chaney, A.L.; Marbach, E.P. Modified reagents for determination of urea and ammonia. Clin. Chem. 1962, 8, 130–132. [Google Scholar] [CrossRef] [PubMed]
- Kubasik, N.; Lundberg, P.; Brodows, R.; Hallauer, G.; Same, D.; Lindstedt, G.; Bengtsson, C.; Nyström, E. Free thyroxin by radioimmunoassay: Evaluation of a new direct method involving a radiolabeled thyroxin analog. Clin. Chem. 1983, 29, 1781–1786. [Google Scholar] [CrossRef]
- Tappel, A.L.; Zalkin, H. Inhibition of lipid peroxidation in mitochondria by vitamin E. Arch. Biochem. 1959, 80, 333–336. [Google Scholar] [CrossRef]
- Luck, H. Catalase. In Method of Enzymatic Analysis; Bergmayer, M.V., Ed.; Verlag Chemic/Academic Press: New York, NY, USA, 1974; p. 885. [Google Scholar]
- Misra, H.; Fridovich, I. The role of superoxide anion in the autooxidation of epinephrine and a simple assay for superoxide dismutase. J. Biol. Chem. 1972, 247, 3170–3175. [Google Scholar] [CrossRef]
- Koracevic, D.; Koracevic, G.; Djordjevic, V.; Andrejevic, S.; Cosic, V. Method for the measurement of antioxidant activity in human fluids. J. Clin. Pathol. 2001, 54, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Wegmann, T.G.; Smithies, O. A simple hemagglutination system requiring small amounts of red cells and antibodies. Transfusion 1966, 6, 67–73. [Google Scholar] [CrossRef]
- Nelson, N.; Lakshmanan, N.; Lamont, S. Sheep red blood cell and Brucella abortus antibody responses in chickens selected for multitrait immunocompetence. Poult. Sci. 1995, 74, 1603–1609. [Google Scholar] [CrossRef]
- AOAC. Association of Official Analytical Chemists. Official Method of Analysis; AOAC: Gaithersburg, MD, USA, 2006. [Google Scholar]
- NRC. Nutrient Requirements of Fish; National Academy Press: Washington, DC, USA, 1993. [Google Scholar]
- Cheeke, P. Rabbit Nutrition and Feeding; Academic Press Inc.: New York, NY, USA, 1987; p. 376. [Google Scholar]
- SPSS. Statistical Software Package for the Social Secinces; SPSS Inc.: Chicago, DE, USA, 2001. [Google Scholar]
- Duncan, D.B. Multiple range and multiple F tests. Biometrics 1955, 11, 1–42. [Google Scholar] [CrossRef]
- Pantsi, W.G.; Bester, D.J.; Esterhuyse, A.J.; Aboua, G. Dietary antioxidant properties of vegetable oils and nuts–the race against cardiovascular disease progression. In Antioxidant-Antidiabetic Agents and Human Health; Oguntibeju, O., Ed.; IntechOpen: London, UK, 2014; pp. 209–238. [Google Scholar] [CrossRef]
- Merghani, B.; Awadin, W.; Elseady, Y.; Abu-Heakal, S. Protective role of wheat germ oil against hyperglycemia and hyperlipidemia in streptozotocin induced diabetic rats. Asian J. Anim. Vet. Adv. 2015, 10, 852–864. [Google Scholar] [CrossRef]
- Martins, C.; Cullere, M.; Dalle Zotte, A.; Cardoso, C.; Alves, S.P.; Bessa, R.; Freire, J.P.B.; Falcao-e-Cunha, L. Incorporation of two levels of black soldier fly (Hermetia illucens L.) larvae fat or extruded linseed in diets of growing rabbits: Effects on growth performance and diet digestibility. Czech J. Anim. Sci. 2018, 63, 356–362. [Google Scholar]
- De Blas, C.; Wiseman, J. Nutrition of the Rabbit, 2nd ed.; CAB International: Wallingford, UK, 2010; p. 334. [Google Scholar]
- Fernández-Carmona, J.; Pascual, J.; Cervera, C. The use of fat in rabbit diets. World Rabbit. Sci. 2000, 8, 29–59. [Google Scholar]
- Ayyat, M.; Yamani, K.; Bassuny, M.; El-Gendy, K.; Abdalla, M. A study of using different energy levels for growing rabbits in Egypt. CIHEAM Options Méditerranéennes (Spain) 1992, 8, 131–139. [Google Scholar]
- Ayyat, M.; Marai, I. Effects of heat stress on growth, carcass traits and blood components of New Zealand White rabbits fed various dietary energy–fibre levels, under Egyptian conditions. J. Arid. Environ. 1997, 37, 557–568. [Google Scholar] [CrossRef]
- Barillari, J.; Canistro, D.; Paolini, M.; Ferroni, F.; Pedulli, G.F.; Iori, R.; Valgimigli, L. Direct antioxidant activity of purified glucoerucin, the dietary secondary metabolite contained in rocket (Eruca sativa Mill.) seeds and sprouts. J. Agric. Food Chem. 2005, 53, 2475–2482. [Google Scholar] [CrossRef]
- Khoobchandani, M.; Ojeswi, B.; Ganesh, N.; Srivastava, M.; Gabbanini, S.; Matera, R.; Iori, R.; Valgimigli, L. Antimicrobial properties and analytical profile of traditional Eruca sativa seed oil: Comparison with various aerial and root plant extracts. Food Chem. 2010, 120, 217–224. [Google Scholar] [CrossRef]
- Ebeid, T.; Zeweil, H.; Basyony, M.; Dosoky, W.; Badry, H. Fortification of rabbit diets with vitamin E or selenium affects growth performance, lipid peroxidation, oxidative status and immune response in growing rabbits. Livest. Sci. 2013, 155, 323–331. [Google Scholar] [CrossRef]
- Mustacich, D.J.; Leonard, S.W.; Patel, N.K.; Traber, M.G. α-tocopherol β-oxidation localized to rat liver mitochondria. Free Radical. Biol. Med. 2010, 48, 73–81. [Google Scholar] [CrossRef]
- Piras, A.; Rosa, A.; Falconieri, D.; Porcedda, S.; Dessì, M.A.; Marongiu, B. Extraction of oil from wheat germ by supercritical CO2. Molecules 2009, 14, 2573–2581. [Google Scholar] [CrossRef]
- Bradley, P. British Herbal Compendium. Volume 1. A Handbook of Scientific Information on Widely Used Plant Drugs. Companion to Volume 1 of the British Herbal Pharmacopoeia; British Herbal Medicine Association: Bournemouth, UK, 1992; p. 239. [Google Scholar]
- Leung, A.Y.; Foster, S. Encyclopedia of Common Natural Ingredients Used in Food, Drugs, and Cosmetics. J. Am. Chem. Soc. 1996, 118, 8988. [Google Scholar]
- Ezzat, W.; Hamed, S.; Abd El-Karim, R.; Shehata, M. Evaluation of adding moringa and rocket seeds oils in the diet on productive and reproductive performance of rabbits under hot climatic conditions. Egypt J. Rabbit Sci. 2014, 24, 375–393. [Google Scholar]
- Ahmed, A.S.; Fathey, I.; Bealish, A. Effect of using onion seed and moringa seed oil on productive and physiological performance of growing rabbits under hot climate conditions. Egypt J. Rabbit Sci. 2019, 29, 79–97. [Google Scholar] [CrossRef]
- Omer, H.; El-Nomeary, Y.; El-Kady, R.; Badr, A.M.; Ali, F.; Ahmed, S.M.; El-Allawy, H.; Ibrahim, S.A. Improving the utilization of rabbit diets containing vegetable oil by using fennel (Foeniculum vulgare) and oregano (Origanum vulgare L.) as feed additives. Life Sci. J. 2013, 10, 2625–2636. [Google Scholar]
- Šramková, Z.; Gregová, E.; Šturdík, E. Chemical composition and nutritional quality of wheat grain. Acta Chim. Slov. 2009, 2, 115–138. [Google Scholar]
- Dalle Zotte, A.; Szendrő, Z. The role of rabbit meat as functional food. Meat Sci. 2011, 88, 319–331. [Google Scholar] [CrossRef]
- Zsédely, E.; Tóth, T.; Eiben, C.; Virág, G.; Fábián, J.; Schmidt, J. Effect of dietary vegetable oil (sunflower, linseed) and vitamin E supplementation on the fatty acid composition, oxidative stability and quality of rabbit meat. In Proceedings of the 9th World Rabbit Congress, Verona, Italy, 10–13 June 2008; pp. 1473–1478. [Google Scholar]
- Maertens, L.; Huyghebaert, G.; Delezie, E. Fatty acid composition of rabbit meat when fed a linseed based diet during different periods after weaning. In Proceedings of the 9th World Rabbit Congress, Verona, Italy, 10–13 June 2008; pp. 1381–1386. [Google Scholar]
- Dal Bosco, A.; Castellini, C.; Bianchi, L.; Mugnai, C. Effect of dietary α-linolenic acid and vitamin E on the fatty acid composition, storage stability and sensory traits of rabbit meat. Meat Sci. 2004, 66, 407–413. [Google Scholar] [CrossRef]
- Lember, A.; Tikk, H.; Tikk, V.; Tamm, K.; Karus, A.; Kuusik, S.; Rei, M. The use of linseed oil in enriching the lipids of hen broiler, quail and rabbit meal with ω-3 Fatty acids. Agraarteadus J. Agric. Sci. 2006, 17, 45–67. Available online: https://agrt.emu.ee/pdf/2006_1_lemberik.pdf (accessed on 22 April 2021).
- Cortinas, L.; Villaverde, C.; Galobart, J.; Baucells, M.; Codony, R.; Barroeta, A. Fatty acid content in chicken thigh and breast as affected by dietary polyunsaturation level. Poult. Sci. 2004, 83, 1155–1164. [Google Scholar] [CrossRef]
- Villaverde, C.; Baucells, M.; Cortinas, L.; Barroeta, A. Effects of dietary concentration and degree of polyunsaturation of dietary fat on endogenous synthesis and deposition of fatty acids in chickens. Br. Poult. Sci. 2006, 47, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Zelenka, J.; Schneiderova, D.; Mrkvicova, E.; Dolezal, P. The effect of dietary linseed oils with different fatty acid pattern on the content of fatty acids in chicken meat. Vet. Med. (Praha) 2008, 53, 77. [Google Scholar] [CrossRef]
- Hashempour-Baltork, F.; Torbati, M.; Azadmard-Damirchi, S.; Savage, G.P. Vegetable oil blending: A review of physicochemical, nutritional and health effects. Trends Food Sci. Technol. 2016, 57, 52–58. [Google Scholar] [CrossRef]
- Hosseini, V.; Maroufi, N.F.; Saghati, S.; Asadi, N.; Darabi, M.; Ahmad, S.N.S.; Hosseinkhani, H.; Rahbarghazi, R. Current progress in hepatic tissue regeneration by tissue engineering. J. Transl. Med. 2019, 17, 1–24. [Google Scholar] [CrossRef]
- Kim, S.-J.; Jin, S.; Ishii, G. Isolation and structural elucidation of 4-(β-D-glucopyranosyldisulfanyl) butyl glucosinolate from leaves of rocket salad (Eruca sativa L.) and its antioxidative activity. Biosci. Biotechnol. Biochem. 2004, 68, 2444–2450. [Google Scholar] [CrossRef]
- Jonnala, R.; Dunford, N.; Irmak, S. Policosanol, tocopherol and phytosterol composition of wheat extract. In Proceedings of the IFT Annual Meeting, New Orleans, Louisiana, 15–20 July 2005; pp. 15–20. [Google Scholar]
- Sunmonu, T.; Oloyede, O. Biochemical assessment of the effects of crude oil contaminated catfish (Clarias gariepinus) on the hepatocytes and performance of rat. Afr. J. Biochem. Res. 2007, 1, 83–89. [Google Scholar]
- El-Kholy, K.; El-Damrawy, S.; Seleem, T. Improvement of rabbit’s productivity and immunity by probiotic bacteria isolated from suckling mother’s soft faeces. In Proceedings of the 5th Science Congress of Egyptian Society for Animal Management, Giza, Egypt, 18–22 September 2012; pp. 142–157. [Google Scholar]
- Abozid, M.M.; Ashoush, Y.A.; Sakr, A.A.; Taha, K.M.; Ayimba, E. Evaluation of Egyptian rocket seed oil as a source of essential fatty acids and its hypolipidemic effect in rats fed on high fat diet. Int. J. Adv. Res. 2014, 2, 434–441. [Google Scholar]
- de Jong, A.; Plat, J.; Mensink, R.P. Metabolic effects of plant sterols and stanols. J. Nutr. Biochem. 2003, 14, 362–369. [Google Scholar] [CrossRef]
- Halawa, E.H.; Radwan, A.A.; El-Sayed, A.I.M.; Farid, O.A.A. Effect of wheat germ oil and coenzyme Q10 on physiological performance and testicular oxidative stress markers in rabbit bucks. Ann. Agric. Sci. Moshtohor. 2019, 57, 47–58. [Google Scholar]
- Zacchi, P.; Daghero, J.; Jaeger, P.; Eggers, R. Extraction/fractionation and deacidification of wheat germ oil using supercritical carbon dioxide. Braz. J. Chem. Eng. 2006, 23, 105–110. [Google Scholar] [CrossRef]
- Irmak, S.; Dunford, N.T. Policosanol contents and compositions of wheat varieties. J. Agric. Food Chem. 2005, 53, 5583–5586. [Google Scholar] [CrossRef]
- Conti, V.; Izzo, V.; Corbi, G.; Russomanno, G.; Manzo, V.; De Lise, F.; Di Donato, A.; Filippelli, A. Antioxidant supplementation in the treatment of aging-associated diseases. Front Pharmacol. 2016, 7, 24. [Google Scholar] [CrossRef]
- Beneš, L.; Ďuračková, Z.; Ferenčik, M. Chemistry, physiology and pathology of free radicals. Life Sci. 1999, 65, 1865–1874. [Google Scholar]
- Kankofer, M.; Lipko, J.; Zdunczyk, S. Total antioxidant capacity of bovine spontaneously released and retained placenta. Pathophysiology 2005, 11, 215–219. [Google Scholar] [CrossRef]
- Shahidi, F. Antioxidants in food and food antioxidants. Food Nahrung 2000, 44, 158–163. [Google Scholar] [CrossRef]
- Fine, F.; Brochet, C.; Gaud, M.; Carre, P.; Simon, N.; Ramli, F.; Joffre, F. Micronutrients in vegetable oils: The impact of crushing and refining processes on vitamins and antioxidants in sunflower, rapeseed, and soybean oils. Eur. J. Lipid Sci. Technol. 2016, 118, 680–697. [Google Scholar] [CrossRef]
- El-Sisy, G.A.; Khalifa, W.H.; El-Nattat, W.S.; Abusinaa, G.E.; Maghraby, N.A. Effect of dietary supplementation of wheat germ on some reproductive performances and oxidative status of rabbit bucks under heat stress. Biosci. Res. 2018, 15, 4267–4273. [Google Scholar]
- Niu, L.-Y.; Jiang, S.-T.; Pan, L.-J. Preparation and evaluation of antioxidant activities of peptides obtained from defatted wheat germ by fermentation. J. Food Sci. Technol. 2013, 50, 53–61. [Google Scholar] [CrossRef]
- Paranich, V.; Cherevko, O.; Frolova, N.; Paranich, A. The effect of wheat germ oil on the antioxidant system of animals. Likars’ Ka Sprav. 2000, 2, 40–44. [Google Scholar]
- Chang, C.L.; Coudron, T.A.; Goodman, C.; Stanley, D.; An, S.; Song, Q. Wheat germ oil in larval diet influences gene expression in adult oriental fruit fly. J. Insect Physiol. 2010, 56, 356–365. [Google Scholar] [CrossRef]
- Bendich, A. Carotenoids and the immune response. J. Nutr. 1989, 119, 112–115. [Google Scholar] [CrossRef]
- Chew, B.P.; Park, J.S.; Wong, T.S.; Kim, H.W.; Weng, B.B.; Byrne, K.M.; Hayek, M.G.; Reinhart, G.A. Dietary β-carotene stimulates cell-mediated and humoral immune response in dogs. J. Nutr. 2000, 130, 1910–1913. [Google Scholar] [CrossRef]
- Sjövall, O.; Virtalaine, T.; Lapveteläinen, A.; Kallio, H. Development of rancidity in wheat germ analyzed by headspace gas chromatography and sensory analysis. J. Agric. Food Chem. 2000, 48, 3522–3527. [Google Scholar] [CrossRef]



| Ingredients | (g/kg) |
| Berseem hay | 280.00 |
| Barley | 173.00 |
| Corn yellow | 179.00 |
| Wheat bran | 120.00 |
| Soybean meal 44% | 200.00 |
| Molasses | 30.00 |
| Di-Ca-Ph | 10.00 |
| Salt | 3.00 |
| Vitamin premix 1 | 3.00 |
| Lysine | 1.00 |
| Methionine | 1.00 |
| Chemical analysis | (g/kg dry matter) |
| Organic matter | 912.50 |
| Ash | 87.50 |
| Crude protein | 174.04 |
| Ether extract | 84.09 |
| Crude fiber | 127.49 |
| Nitrogen-free extract | 526.88 |
| Neutral detergent fiber | 373.00 |
| acid detergent fiber | 210.59 |
| Hemicellulose | 162.41 |
| Energy value | |
| Gross energy (kcal/kg) | 3940.87 |
| Digestible energy (kcal/kg) | 2528.56 |
| Items | Control | Rocket Seed Oil | Wheat Germ Oil | Mixture Oils | p-Value |
|---|---|---|---|---|---|
| Initial body weight (g/rabbit) | 525.12 ± 10.17 | 540.43 ± 38.96 | 541.05 ± 26.33 | 535.81 ± 30.52 | 0.977 |
| Final body weight (g/rabbit) | 1995.71 ± 28.86 c | 2104.29 ± 27.42 b | 2237.57 ± 19.98 a | 2233.81 ± 43.72 a | 0.001 |
| Weight gain (g/rabbit) | 1470.60 ± 31.80 c | 1563.86 ± 30.76 b | 1696.52 ± 24.33 a | 1698.00 ± 32.43 a | 0.001 |
| Average daily gain (g/day) | 30.01 ± 0.65 c | 31.91 ± 0.63 b | 34.62 ± 0.50 a | 34.65 ± 0.66 a | 0.001 |
| Weight gain percent (%) | 280.99 ± 9.72 | 300.02 ± 24.83 | 318.15 ± 16.21 | 322.97 ± 19.10 | 0.372 |
| Feed intake (g/experimental period) | 5541.45 ± 25.16 | 5531.86 ± 14.09 | 5516.33 ± 11.23 | 5568.77 ± 21.40 | 0.275 |
| Daily feed intake (g/day) | 113.09 ± 0.51 | 112.89 ± 0.29 | 112.58 ± 0.23 | 113.65 ± 0.44 | 0.277 |
| Feed conversion ratio | 3.78 ± 0.08 a | 3.55 ± 0.07 b | 3.26 ± 0.04 c | 3.29 ± 0.07 c | 0.001 |
| Items | Control | Rocket Seed Oil | Wheat Germ Oil | Mixture Oils | p-Value |
|---|---|---|---|---|---|
| Digestion coefficient (%) | |||||
| Dry matter | 59.80 ± 1.09 c | 62.28 ± 0.91 b | 64.46 ± 0.04 b | 67.79 ± 0.45 a | 0.001 |
| Organic matter | 63.58 ± 1.10 c | 65.72 ± 0.86 bc | 67.64 ± 0.18 b | 70.57 ± 0.40 a | 0.001 |
| Crude protein | 69.01 ± 0.13 b | 70.39 ± 0.77 b | 70.84 ± 0.47 b | 73.21 ± 1.05 a | 0.008 |
| Ether extract | 52.16 ± 1.26 d | 58.77 ± 0.77 c | 62.47 ± 1.02 b | 67.11 ± 0.44 a | 0.001 |
| Crude fiber | 57.60 ± 0.64 | 58.69 ± 2.63 | 59.78 ± 0.53 | 62.12 ± 1.77 | 0.293 |
| NDF | 54.17 ± 0.84 d | 57.93 ± 0.87 c | 60.89 ± 0.21 b | 64.97 ± 0.45 a | 0.001 |
| ADF | 53.32 ± 0.91 d | 58.29 ± 0.82 c | 61.56 ± 0.55 b | 65.88 ± 0.39 a | 0.001 |
| Hemicellulose | 55.27 ± 1.04 c | 57.47 ± 0.95 c | 60.02 ± 0.28 b | 63.80 ± 0.59 a | 0.002 |
| Nitrogen free extract | 65.50 ± 2.00 b | 66.98 ± 1.02 b | 69.09 ± 0.48 ab | 71.89 ± 0.40 a | 0.012 |
| Nitrogen balance, NB | |||||
| N intake (g/day) | 2.97 ± 0.03 c | 3.15 ± 0.03 b | 3.20 ± 0.03 b | 3.35 ± 0.03 a | 0.001 |
| Feces N (g/day) | 0.92 ± 0.01 | 0.93 ± 0.02 | 0.94 ± 0.02 | 0.90 ± 0.03 | 0.670 |
| Absorbed N (g/day) | 2.05 ± 0.02 c | 2.21 ± 0.03 b | 2.27 ± 0.02 b | 2.46 ± 0.04 a | 0.001 |
| Urine N (g/day) | 0.53 ± 0.02 | 0.46 ± 0.02 | 0.48 ± 0.02 | 0.48 ± 0.04 | 0.315 |
| N balance (g/day) | 1.52 ± 0.02 c | 1.76 ± 0.02 b | 1.79 ± 0.03 b | 1.98 ± 0.07 a | 0.001 |
| NB intake (%) | 51.17 ± 0.46 b | 55.86 ± 0.74 a | 55.90 ± 0.81 a | 58.95 ± 2.13 a | 0.006 |
| NB absorption (%) | 74.15 ± 0.78 b | 79.37 ± 0.92 a | 78.90 ± 0.86 a | 80.45 ± 1.83 a | 0.012 |
| Items | Control | Rocket Seed Oil | Wheat Germ Oil | Mixture Oils | p-Value |
|---|---|---|---|---|---|
| Palmitic (c16:0,%) | 33.54 ± 0.56 a | 32.45 ± 1.74 ab | 30.79 ± 1.04 ab | 27.03 ± 2.47 b | 0.050 |
| Stearic (c18:0,%) | 6.78 ± 0.20 | 6.45 ± 0.15 | 6.49 ± 1.12 | 7.19 ± 1.08 | 0.902 |
| Palmitoleic (c16:1,%) | 4.42 ± 0.56 | 4.43 ± 0.83 | 3.18 ± 0.64 | 4.65 ± 0.29 | 0.372 |
| Oleic (c18:1,%) | 31.35 ± 0.43 a | 30.21 ± 0.96 ab | 28.30 ± 0.42 b | 28.64 ± 0.85 b | 0.050 |
| Linoleic (c18:2; n-6,%) | 19.31 ± 1.52 | 20.65 ± 3.20 | 24.13 ± 1.99 | 24.03 ± 1.08 | 0.330 |
| Linolenic (c18:3; n-3,%) | 3.12 ± 0.07 c | 4.32 ± 0.25 b | 5.29 ± 0.35 a | 5.59 ± 0.12 a | 0.001 |
| Items | Control | Rocket Seed Oil | Wheat Germ Oil | Mixture Oils | p-Value |
|---|---|---|---|---|---|
| Protein profile | |||||
| Protein (g/dL) | 5.37 ± 0.09 b | 5.39 ± 0.15 b | 5.70 ± 0.20 ab | 5.97 ± 0.05 a | 0.019 |
| Albumin (g/dL) | 3.06 ± 0.14 ab | 3.36 ± 0.09 a | 3.01 ± 0.06 b | 3.38 ± 0.12 a | 0.041 |
| Globulin (g/dL) | 2.31 ± 0.22 ab | 2.03 ± 0.21 b | 2.69 ± 0.19 a | 2.59 ± 0.16 a | 0.050 |
| A/G ratio | 1.41 ± 0.22 | 1.56 ± 0.20 | 1.15 ± 0.10 | 1.34 ± 0.13 | 0.215 |
| Kidney function | |||||
| Urea (mg/dL) | 49.50 ± 1.70 a | 44.30 ± 1.22 b | 45.70 ± 0.89 ab | 45.40 ± 1.12 b | 0.050 |
| Lipid profile | |||||
| Total lipid (mg/dL) | 433.00 ± 11.45 b | 470.00 ± 3.85 a | 424.70 ± 6.55 b | 424.50 ± 4.26 b | 0.001 |
| TG (mg/dL) | 61.60 ± 2.93 | 65.10 ± 4.89 | 69.10 ± 2.41 | 71.10 ± 5.67 | 0.411 |
| Cholesterol (mg/dL) | 63.00 ± 3.74 | 61.40 ± 4.48 | 62.00 ± 2.02 | 60.40 ± 1.89 | 0.955 |
| HDL-c (mg/dL) | 29.40 ± 1.29 b | 32.50 ± 0.92 ab | 34.00 ± 0.71 a | 33.20 ± 1.16 a | 0.033 |
| LDL-c (mg/dL) | 21.28 ± 3.40 a | 15.88 ± 3.74 b | 14.18 ± 1.97 b | 12.98 ± 2.00 c | 0.001 |
| vLDL-c (mg/dL) | 12.32 ± 0.59 | 13.02 ± 0.98 | 13.82 ± 0.48 | 14.22 ± 1.13 | 0.412 |
| HDL-c/LDL-c ratio | 1.61 ± 0.39 | 2.50 ± 0.53 | 2.56 ± 0.30 | 2.78 ± 0.40 | 0.238 |
| Hormone assay | |||||
| T4 (ng/mL) | 2.83 ± 0.10 | 3.48 ± 0.10 | 3.15 ± 0.15 | 3.46 ± 0.30 | 0.070 |
| T3 (ng/mL) | 1.04 ± 0.07 b | 1.34 ± 0.02 a | 1.36 ± 0.04 a | 1.42 ± 0.16 a | 0.035 |
| Items | Control | Rocket Seed Oil | Wheat Germ Oil | Mixture Oils | p-Value |
|---|---|---|---|---|---|
| SRBCs at 9 weeks of age | 0.74 ± 0.04 b | 0.84 ± 0.02 a | 0.87 ± 0.02 a | 0.84 ± 0.02 a | 0.010 |
| SRBCs at 10 weeks of age | 0.76 ± 0.03 b | 0.84 ± 0.03 ab | 0.85 ± 0.02 a | 0.76 ± 0.03 b | 0.044 |
| SRBCs at 11 weeks of age | 0.71 ± 0.04 | 0.76 ± 0.11 | 0.88 ± 0.02 | 0.71 ± 0.03 | 0.201 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morshedy, S.A.; Abdelmodather, A.M.; Basyony, M.M.; Zahran, S.A.; Hassan, M.A. Effects of Rocket Seed Oil, Wheat Germ Oil, and Their Mixture on Growth Performance, Feed Utilization, Digestibility, Redox Status, and Meat Fatty Acid Profile of Growing Rabbits. Agriculture 2021, 11, 662. https://doi.org/10.3390/agriculture11070662
Morshedy SA, Abdelmodather AM, Basyony MM, Zahran SA, Hassan MA. Effects of Rocket Seed Oil, Wheat Germ Oil, and Their Mixture on Growth Performance, Feed Utilization, Digestibility, Redox Status, and Meat Fatty Acid Profile of Growing Rabbits. Agriculture. 2021; 11(7):662. https://doi.org/10.3390/agriculture11070662
Chicago/Turabian StyleMorshedy, Sabrin Abdelrahman, Ahmed M. Abdelmodather, Mohamed M. Basyony, Soliman A. Zahran, and Mohamed A. Hassan. 2021. "Effects of Rocket Seed Oil, Wheat Germ Oil, and Their Mixture on Growth Performance, Feed Utilization, Digestibility, Redox Status, and Meat Fatty Acid Profile of Growing Rabbits" Agriculture 11, no. 7: 662. https://doi.org/10.3390/agriculture11070662
APA StyleMorshedy, S. A., Abdelmodather, A. M., Basyony, M. M., Zahran, S. A., & Hassan, M. A. (2021). Effects of Rocket Seed Oil, Wheat Germ Oil, and Their Mixture on Growth Performance, Feed Utilization, Digestibility, Redox Status, and Meat Fatty Acid Profile of Growing Rabbits. Agriculture, 11(7), 662. https://doi.org/10.3390/agriculture11070662








