Phenolic Compounds Characterization and Antioxidant Properties of Monocultivar Olive Oils from Northeast Algeria
Abstract
1. Introduction
2. Materials and Methods
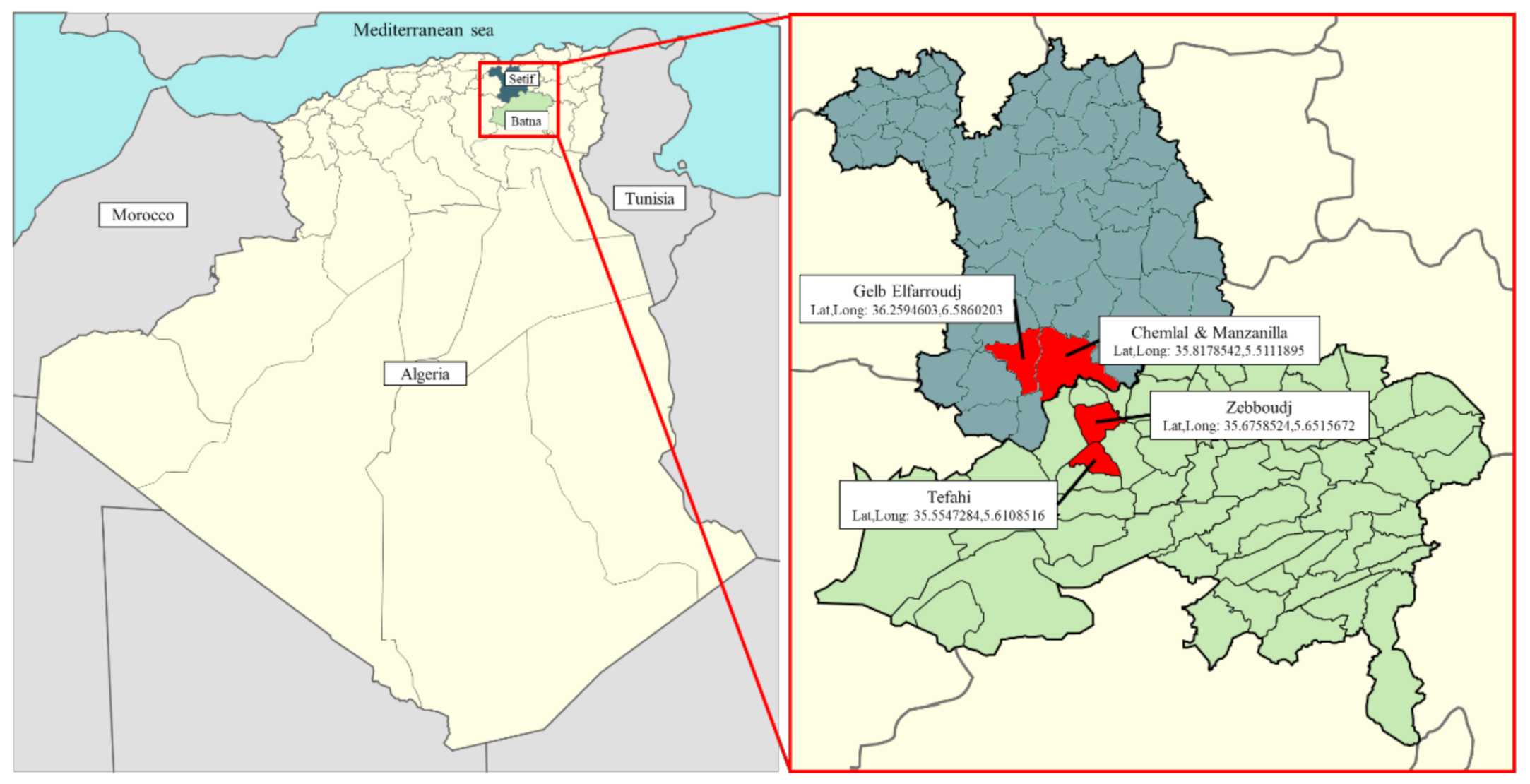
2.1. Plant Material and Olive Oil Samples
2.2. Reagents and Standards
2.3. Quality Parameters
2.4. FAMEs Composition
2.5. Individual Phenolic Compounds Characterization by UHPLC-HESI-MS
2.6. Olive Oil Extracts for Determination of Total Phenolic Content and for Antioxidant Assays
2.7. Total Phenols Content
2.8. Antioxidant Activity
2.8.1. 2,2-Diphenyl-1-Picrylhydrazyl Test
2.8.2. Trolox Equivalents Antioxidant Capacity
2.8.3. Ferric-Reducing/Antioxidant Power
2.8.4. Beta-Carotene Bleaching Assay
2.9. Statistical Analysis
3. Results
3.1. Quality Parameters
3.2. Fatty Acid Composition
3.3. Individual Phenolic Compounds Identification
3.4. Antioxidant Activity and Total Content of Phenolic Compounds
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gomez Caravaca, A.M.; Maggio, R.M.; Cerretani, L. Chemometric applications to assess quality and critical parameters of virgin and extra-virgin olive oil. Anal. Chim. Acta 2016, 913, 1–21. [Google Scholar] [CrossRef]
- Ouni, Y.; Taamalli, A.; Maria Gómez-Caravaca, A.; Segura-Carretero, A.; Fernández-Gutiérrez, A.; Zarrouk, M. Characterisation and quantification of phenolic compounds of extra-virgin olive oils according to their geographical origin by a rapid and resolutive LC-ESI-TOF MS method. Food Chem. 2011, 127, 1263–1267. [Google Scholar] [CrossRef]
- Deng, J.; Xu, Z.; Xiang, C.; Liu, J.; Zhou, L.; Li, T.; Yang, Z.; Ding, C. Comparative evaluation of maceration and ultrasonic-assisted extraction of phenolic compounds from fresh olives. Ultrason. Sonochem. 2017, 37, 328–334. [Google Scholar] [CrossRef]
- Genovese, A.; Yang, N.; Linforth, R.; Sacchi, R.; Fisk, I. The role of phenolic compounds on olive oil aroma release. Food Res. Int. 2018, 112, 319–327. [Google Scholar] [CrossRef]
- Ghanbari, R.; Anwar, F.; Alkharfy, K.M.; Gilani, A.H.; Saari, N. Valuable Nutrients and Functional Bioactives in Different Parts of Olive (Olea europaea L.). Int. J. Mol. Sci. 2012, 13, 3291–3340. [Google Scholar] [CrossRef]
- Reboredo-Rodríguez, P.; Varela-López, A.; Forbes-Hernández, T.Y.; Gasparrini, M.; Afrin, S.; Cianciosi, D.; Zhang, J.; Pia Manna, P.; Bompadre, S.; Quiles, J.L.; et al. Phenolic Compounds Isolated from Olive Oil as Nutraceutical Tools for the Prevention and Management of Cancer and Cardiovascular Diseases. J. Funct. Foods 2018, 19, 2305. [Google Scholar] [CrossRef]
- García-Rodríguez, R.; Belaj, A.; Romero-Segura, C.; Sanz, C.; Pérez, A.G. Exploration of genetic resources to improve the functional quality of virgin olive oil. J. Funct. Foods 2017, 38, 1–8. [Google Scholar] [CrossRef]
- Oueslati, I.; Krichene, D.; Manaï, H.; Taamalli, W.; Zarrouk, M.; Flamini, G. Monitoring the volatile and hydrophilic bioactive compounds status of fresh and oxidized Chemlali. Food Res. Int. 2018, 112, 425–433. [Google Scholar] [CrossRef]
- Jiménez, B.; Sánchez-Ortiz, A.; Lorenzo, M.L.; Rivas, A. Influence of fruit ripening on agronomic parameters, quality indices, sensory attributes and phenolic compounds of Picudo olive oils. Food Res. Int. 2013, 54, 1860–1867. [Google Scholar] [CrossRef]
- Ammar, S.; Kelebek, H.; Zribi, A.; Abichou, M.; Selli, S.; Bouaziz, M. LC-DAD/ESI-MS/MS characterization of phenolic constituents in Tunisian extra-virgin olive oils: Effect of olive leaves addition on chemical composition. Food Res. Int. 2017, 100, 477–485. [Google Scholar] [CrossRef]
- Natasa, P.K.; Reza, A.; Nikolaos, S.T. Application of an advanced and wide scope non-target screening workflow with LC-ESI-QTOF-MS and chemometrics for the classification of the Greek olive oil varieties. Food Chem. 2018, 256, 53–61. [Google Scholar]
- Salvo, A.; Rotondo, A.; La Torre, G.L.; Cicero, N.; Dugo, G. Determination of 1,2/1,3-diglycerides in Sicilian extra-virgin olive oils by 1H-NMR over a one-year storage period. Nat. Prod. Res. 2017, 31, 822–828. [Google Scholar] [CrossRef]
- Bakhouche, A.; Lozano Sánchez, J.; Bengana, M.; Fernández Gutiérrez, A.; Segura Carretero, A. Time course of Algerian Azeradj extra virgin olive oil quality during olive ripening. Eur. J. Lipid Sci. Technol. 2015, 117, 389–397. [Google Scholar] [CrossRef]
- Yakhlef, W.; Arhab, R.; Romero, C.; Brenes, M.; de Castro, A.; Medina, E. Phenolic composition and antimicrobial activity of Algerian olive products and by-products. LWT Food Sci. Technol. 2018, 93, 323–328. [Google Scholar] [CrossRef]
- Abdul-Hussain, K.H.; Abdul-Hussain, M.S. Influence of the gibberelic acid on the germination of the seeds of olive-tree Olea europaea L. J. Cent. Eur. Agric. 2004, 5, 1–4. [Google Scholar]
- International conventions and agreements-laws and decrees, judgments, decisions, notices, communications and announcements. Off. J. People’s Democr. Repub. Alger. 2008, 5, 46.
- Algerian Ministry of Agriculture. Fixation des Conditions D’éligibilité au Soutien sur le Compte D’affectation Spéciale no.302-139 Intitulé “Fonds National de Développement Agricole” ligne 1 “Développement de L’investissement Agricole”, Ainsi que les Modalités de Paiement des Subventions; Decision no. 414; 2014. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX:32016R2095 (accessed on 22 October 2020).
- European Commission. Regulation no. 2016/2095 amending regulation no. 2568/91 on the characteristics of olive oil and olive-residue oil and on the relevant methods of analysis. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX:32016R2095 (accessed on 1 December 2016).
- Di Stefano, V.; Melilli, M.G. Effect of storage on quality parameters and phenolic content of Italian extra-virgin olive oils. Nat. Prod. Res. 2020, 34, 78–86. [Google Scholar] [CrossRef]
- Grilo, F.; Novara, M.E.; D’Oca, M.C.; Rubino, S.; Lo Bianco, R.; Di Stefano, V. Quality evaluation of extra-virgin olive oils from Sicilian genotypes grown in a high-density system. Int. J. Food Sci. Nutr. 2020, 71, 397–409. [Google Scholar] [CrossRef]
- Di Stefano, V.; Avellone, G.; Bongiorno, D.; Indelicato, S.; Massenti, R.; Lo Bianco, R. Quantitative evaluation of the phenolic profile in fruits of six avocado (Persea americana) cultivars by ultra-high-performance liquid chromatography-heated electrospray-mass spectrometry. Int. J. Food Prop. 2017, 6, 1–27. [Google Scholar] [CrossRef]
- Napoli, E.; Siracusa, L.; Ruberto, G.; Carrubba, A.; Lazzara, S.; Speciale, A.; Cimino, F.; Saija, A.; Cristani, M. Phytochemical profiles, phototoxic and antioxidant properties of eleven Hypericum species. A comparative study. Phytochemistry. 2018, 152, 162–173. [Google Scholar] [CrossRef]
- Ouerghemmi, S.; Sebei, H.; Siracusa, L.; Ruberto, G.; Saija, A.; Cimino, F.; Cristani, M. Comparative study of phenolic composition and antioxidant activity of leaf extracts from three wild Rosa species grown in different Tunisia regions: Rosa canina L., Rosa moschata Herrm. And Rosa sempervirens L. Ind. Crops Prod. 2016, 94, 167–177. [Google Scholar] [CrossRef]
- Dehimi, K.; Speciale, A.; Saija, A.; Dahamna, S.; Raciti, R.; Cimino, F.; Cristani, M. Antioxidant and anti-inflammatory properties of Algerian Thymelaeamicrophylla Coss. And Dur. extracts. Pharmacogn. Mag. 2016, 12, 203–210. [Google Scholar]
- Boussahel, S.; Speciale, A.; Dahamna, S.; Amar, Y.; Bonaccorsi, I.; Cacciola, F.; Cimino, F.; Donato, P.; Ferlazzo, G.; Harzallah, D.; et al. Flavonoid profile, antioxidant and cytotoxic activity of different extracts from Algerian Rhamnus alaternus L. bark. Pharmacogn. Mag. 2015, 11, 102–109. [Google Scholar]
- Martorana, M.; Arcoraci, T.; Rizza, L.; Cristani, M.; Bonina, F.P.; Saija, A.; Trombetta, D.; Tomaino, A. In vitro antioxidant and in vivo photoprotective effect of pistachio (Pistacia vera L. variety Bronte) seed and skin extracts. Fitoterapia 2013, 85, 41–48. [Google Scholar] [CrossRef]
- International Olive Council. Trade Standard Applying to Olive Oils and Olive Pomace Oils COI/T.15/NC no. 3/Rev 13. Available online: https://www.internationaloliveoil.org/wp-content/uploads/2019/11/COI-T.15-NC.-No-3-Rev.-13-2019-Eng.pdf (accessed on 23 October 2020).
- Boudour Benrachou, N.; Plard, J.; Pinatel, C.; Artaud, J.; Dupuy, N. Fatty Acid Compositions of Olive Oils from Six Cultivars from East and South-Western Algeria. Adv. Food Technol. Nutri. Sci. 2017, 3, 1–5. [Google Scholar] [CrossRef]
- Laincer, F.; Iaccarino, N.; Amato, J.; Pagano, B.; Pagano, A.; Tenore, G.; Tamendjari, A.; Rovellini, P.; Venturini, S.; Bellan, G.; et al. Characterization of monovarietal extra virgin olive oils from the province of Béjaïa (Algeria). Food Res. Int. 2016, 89, 1123–1133. [Google Scholar] [CrossRef]
- Laincer, F.; Laribi, R.; Tamendjari, A.; Arrar, L.; Rovellini, P.; Venturini, S. Olive oils from Algeria: Phenolic compounds, antioxidant and antibacterial activities. Grasas Aceites 2014, 65, 1–10. [Google Scholar]
- Louadj, L.; Giuffre, A.M. Analytical characteristics of olive oil produced with three different processes in Algeria. Riv. Ital. Sostanze Grasse. 2010, 87, 186–195. [Google Scholar]
- Bengana, M.; Bakhouche, A.; Lozano Sánchez, J.; Amir, Y.; Youyou, A.; Segura Carretero, A.; Fernández Gutiérrez, A. Influence of olive ripeness on chemical properties and phenolic composition of “Chemlal” extra-virgin olive oil. Food Res. Int. 2013, 54, 1868–1875. [Google Scholar] [CrossRef]
- Yangui, A.; Costa-Font, M.; Gil, J.M. The effect of personality traits on consumers’ preferences for extra virgin olive oil. Food Qual Prefer. 2016, 51, 27–38. [Google Scholar] [CrossRef]
- McDonald, S.; Prenzler, P.D.; Antolovich, M.; Robards, K. Phenolic content and antioxidant activity of olive extracts. Food Chem. 2001, 73, 73–84. [Google Scholar] [CrossRef]
- Chou, T.H.; Ding, H.Y.; Hung, W.J.; Liang, C.H. Antioxidative characteristics and inhibition of α-melanocyte-stimulating hormone-stimulated melanogenesis of vanillin and vanillic acid from Origanumvulgare. Exp. Dermatol. 2010, 19, 742–750. [Google Scholar] [CrossRef] [PubMed]
- Visioli, F.; Poli, A.; Galli, C. Antioxidant and Other Biological Activities of Phenols from Olives and Olive Oil. Med. Res. Rev. 2002, 22, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Alowaiesh, B.; Singha, Z.; Fangc, Z.; Kailisd, S.G. Harvest time impacts the fatty acid compositions, phenolic compounds and sensory attributes of Frantoio and Manzanilla olive oil. Sci. Hortic. 2018, 234, 74–80. [Google Scholar] [CrossRef]
- Bouarroudja, K.; Tamendjaria, A.; Larbat, R. Quality, composition and antioxidant activity of Algerian wild olive (OleaeuropaeaL. subsp.Oleaster) oil. Ind. Crop. Prod. 2016, 83, 484–491. [Google Scholar] [CrossRef]
- Mailer, R.J.; Ayton, J.; Graham, K. The Influence of Growing Region, Cultivar and Harvest Timing on the Diversity of Australian Olive Oil. J. Am. Oil Chem. Soc. 2010, 87, 877–884. [Google Scholar] [CrossRef]
- Parenti, A.; Spugnoli, P.; Masella, P.; Calamai, L. The effect of malaxation temperature on the virgin olive oil phenolic profile under laboratory-scale conditions. Eur. J. Lipid Sci. Technol. 2008, 110, 735–741. [Google Scholar] [CrossRef]
| Cultivars | Free Acidity (%) | PV (meq O2/kg) | K232 | K270 | ΔK |
|---|---|---|---|---|---|
| Tefahi | 0.48 ± 0.03 c | 12.68 ± 1.23 b | 1.98 ± 0.17 b | 0.17 ± 0.02 a | −0.002 ± 0.01 c |
| Chemlal | 0.50 ± 0.05 c | 12.75 ± 1.13 b | 2.29 ± 0.28 a | 0.19 ± 0.01 a | 0.001 ± 0.01 b |
| Gelb Elfarroudj | 0.75 ± 0.06 b | 15.50 ± 1.53 a | 1.97 ± 0.26 b | 0.21 ± 0.03 a | 0.003 ± 0.03 b |
| Manzanilla | 0.80 ± 0.04 b | 15.50 ± 0.24 a | 1.47 ± 0.24 c | 0.12 ± 0.01 b | −0.007 ± 0.01 d |
| Zebboudj | 1.25 ± 0.11 a | 12.51 ± 0.32 b | 0.39 ± 0.03 d | 0.11 ± 0.05 b | 0.11 ± 0.05 a |
| Cultivars | |||||
|---|---|---|---|---|---|
| Fatty Acids | Tefahi | Chemlal | Gelb Elfarroudj | Manzanilla | Zebboudj |
| C16:0 | 12.27 ± 0.04 b | 15.75 ± 0.02 a | 12.70 ± 0.05 b | 9.97 ± 0.31 c | 11.59 ± 0.09 b |
| C16:1(ω-7) | 0.14 ± 0.01 b | 0.35 ± 1.5 a | 0.16 ± 0.01 b | 0.20 ± 0.01 b | nf |
| C16:1 (ω-9) | 1.12 ± 0.01 b | 1.05 ± 0.22 a | 1.21 ± 0.05 b | 0.57 ± 0.01 c | 0.10 ± 0.01 d |
| C17:0 | 0.03 ± 0.01 b | 0.91 ± 1.56 a | 0.08 ± 0.02 b | 0.05 ± 0.04 b | 0.08 ± 0.01 b |
| C17:1 | 0.05 ± 0.01 b | 0.15 ± 0.09 a | 0.10 ± 0.01 b | 0.08 ± 0.01 b | 0.05 ± 0.01 b |
| C18:0 | 2.46 ± 0.04 c | 4.95 ± 0.88 a | 2.68 ± 0.05 c | 2.39 ± 0.03 c | 4.27 ± 0.01 b |
| C18:1(ω-7) | 3.17 ± 0.11 a | 2.37 ± 0.55 b | 3.48 ± 0.39 a | 2.31 ± 0.11 b | 1.55 ± 0.03 c |
| C18:1(ω-9) | 66.93 ± 0.26 b | 59.97 ± 1.11 c | 66.03 ± 0.49 b | 74.9 ± 0.35 a | 26.65 ± 0.08 c |
| C18:2(ω-6) | 12.43 ± 0.17 b | 9.34 ± 0.33 c | 12.10 ± 0.09 b | 8.18 ± 0.06 c | 48.51± 0.02 a |
| C18:3(ω-3) | 0.84 ± 0.09 b | 0.83 ± 0.01 b | 0.97 ± 0.10 b | 0.81 ± 0.01 b | 6.35 ± 0.01 a |
| C20:0 | 0.26 ± 0.04 a | 0.32 ± 0.01 a | 0.33 ± 0.03 a | 0.27 ± 0.02 a | 0.33 ± 0.01 a |
| C20:1 (ω-9) | 0.27 ± 0.07 b | 0.25 ± 0.01 b | 0.24 ± 0.05 b | 0.31 ± 0.02 a | 0.16 ± 0.01 c |
| ∑SFA † % | 15.02 b | 21.93 a | 15.69 b | 12.58 c | 16.27 b |
| ∑MFA ‡ % | 71.7 a | 64.14 b | 71.2 a | 78.4 a | 28.5 c |
| ∑PFA § % | 13.27 b | 10.17 c | 13.07 b | 8.99 c | 54.86 a |
| Phenolic Compounds | Molecular Formula | Experimental m/z [M-H]− | Retention Time | Linear Regression | (r2) |
|---|---|---|---|---|---|
| Tyrosol | C8H10O2 | 137.05933 | 12.05 | Y= −124,926 + 8556 × X | 0.9937 |
| Hydroxytyrosol | C8H10O3 | 153.05438 | 9.15 | Y = −5.29 × 106 + 7.31 × 107 × X | 0.9974 |
| p-Coumaric Acid | C9H8O3 | 163.03847 | 18.30 | Y = −5.83 × 106 + 8.72 × 107 × X | 0.9959 |
| Caffeic Acid | C9H8O4 | 179.03392 | 14.90 | Y = −2.65 × 106 + 8.46 × 107 × X | 0.9750 |
| Ferulic Acid | C10H10O4 | 193.05026 | 19.10 | Y = −5.99 × 106 + 5.15 × 107 × X | 0.9983 |
| Vanillic Acid | C8H8O4 | 167.03388 | 14.30 | Y = −412,147 + 2.85 × 106 × X | 0.9812 |
| p-Hydroxybenzoic Acid | C7H6O3 | 137.02304 | 13.50 | Y = 648,298 + 6.49 × 107 × X | 0.9982 |
| Syringic Acid | C9H10O5 | 197.04519 | 14.56 | Y = 746,927 + 5.54 × 106 × X | 0.9852 |
| Cinnamic Acid | C9H802 | 147.04049 | 3.50 | Y = −1.69 × 106 + 2.42 × 106 × X | 0.9888 |
| Gallic Acid | C7H6O5 | 169.01319 | 7.45 | Y = −1.94 × 106 + 4.30 × 107 × X | 0.9968 |
| Protocatechuic Acid | C7H6O4 | 153.01763 | 10.35 | Y = −2.85 × 106 + 2.09 × 107 × X | 0.9929 |
| Oleocanthal | C17H20O5 | 303.12387 | 23.50 | Y = −3.19 × 106 + 3.09 × 107 × X | 0.9984 |
| Vanillin | C8H8O3 | 151.03867 | 17.22 | Y = −318,221 + 5.02 × 106 × X | 0.9994 |
| Pinoresinol | C20H22O6 | 357.13445 | 23.05 | Y = −61,525.2 + 634911 × X | 0.9988 |
| Oleuropein | C25H32O13 | 539.17732 | 20.60 | Y = −3.11 × 106 + 3.10 × 107 × X | 0.9985 |
| Luteolin | C15H10O6 | 285.04037 | 23.30 | Y = 3.61 × 106 + 1.24 × 108 × X | 0.9992 |
| Diosmetin | C16H12O6 | 299.05621 | 24.05 | Y = 1.435 × 108 × X | 0.9853 |
| Apigenin | C15H10O5 | 269.04565 | 24.00 | Y = 2.37 × 107 + 2.18 × 108 × X | 0.9989 |
| Apigenin-7-glucoside | C7H6O5 | 431.09854 | 22.90 | Y = 9.18 × 106 + 7.56 × 107 × X | 0.9954 |
| Phenolic Compound | Cultivars | |||||
|---|---|---|---|---|---|---|
| Tefahi | Chemlal | Gelb Elfarroudj | Manzanilla | Zebboudj | ||
| Phenolic alcohol | Hydroxytyrosol | 0.41 a | nf | nf | 0.24a | Nf |
| Phenolic acids | p-Coumaric acid | 0.54 b | nf | 0.61 a | nf | 0.41 b |
| Caffeic acid | 0.07 c | 0.98 c | 1.22 a | 0.8 b | 0.72 a | |
| Ferulic acid | 0.41 a | 0.55 c | 0.68 b | nf † | nf | |
| Vanillic acid | nf | nf | 0.70 c | nf † | 1.43 b | |
| p-Hydroxy benzoic acid | nf | nf | 0.42 a | 0.32 c | 0.45 a | |
| Secoiridoids | Oleacein (3,4-DHPEA-EDA) | 8.01 a | 2.50 b | 2.30 a | 25.16 b | nf |
| Oleocanthal (p-HPEA-EDA) | 2.07 b | 3.54 c | 1.13 b | 4.20 a | nf | |
| Oleuropein aglycon (3,4-DHPEA-EA) | 49.65 a | 7.28 a | 13.71 a | 62.67 a | 0.13 a | |
| Deacetoxy-10-hydroxy oleuropein aglycon (DAc-10-OH Ole Agly); | 0.61 a | 0.18 a | nf | 0.84 b | nf | |
| Elenolic acid | 16.88 b | 3.70 b | 1.72 a | 3.18 b | nf | |
| Ligstroside aglycon (p-HPEA-EA) | nf | 8.09 c | 3.46 c | 12.86 a | 0.22 a | |
| Flavonoids | Luteolin | 0.83 b | 0.54 a | 0.51 b | 0.41 a | 0.14 b |
| Varieties | Folin–Ciocalteu µg GAEmg−1 (±SD) | DPPH mmol TEmg−1 (±SD) | FRAP mmol Fe2+Emg−1 (±SD) | TEAC mM TEmg−1 (±SD) | BCB IC50% mgml−1 (CL95) |
|---|---|---|---|---|---|
| Tefahi | 237.19 ± 23.70 | 0.70 ± 0.08 | 0.84 ± 0.12 | 0.31 ± 0.07 | 1.84 (1.52–2.15) |
| Chemlal | 59.24 ± 8.99 ** | 0.05 ± 0.001 ** | 0.12 ± 0.02 ** | 0.008 ± 0.001 ** | 14.81 (12.74–17.21)* |
| Gelb Elfarroudj | 48.94 ± 5.65 ** | 0.03 ± 0.004 ** | 0.044 ± 0.002* | 0.020 ± 0.003 ** | 8.46 (7.41–9.66)* |
| Manzanilla | 46.32 ± 9.10 ** | 0.22 ± 0.06 ** | 0.34 ± 0.20 | 0.37 ± 0.02 | 6.54 (5.36–7.99) |
| Zebboudj | 37.34 ± 4.71 ** | - | - | - | 7.44 (5.62–9.84) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boussahel, S.; Di Stefano, V.; Muscarà, C.; Cristani, M.; Melilli, M.G. Phenolic Compounds Characterization and Antioxidant Properties of Monocultivar Olive Oils from Northeast Algeria. Agriculture 2020, 10, 494. https://doi.org/10.3390/agriculture10110494
Boussahel S, Di Stefano V, Muscarà C, Cristani M, Melilli MG. Phenolic Compounds Characterization and Antioxidant Properties of Monocultivar Olive Oils from Northeast Algeria. Agriculture. 2020; 10(11):494. https://doi.org/10.3390/agriculture10110494
Chicago/Turabian StyleBoussahel, Soulef, Vita Di Stefano, Claudia Muscarà, Mariateresa Cristani, and Maria Grazia Melilli. 2020. "Phenolic Compounds Characterization and Antioxidant Properties of Monocultivar Olive Oils from Northeast Algeria" Agriculture 10, no. 11: 494. https://doi.org/10.3390/agriculture10110494
APA StyleBoussahel, S., Di Stefano, V., Muscarà, C., Cristani, M., & Melilli, M. G. (2020). Phenolic Compounds Characterization and Antioxidant Properties of Monocultivar Olive Oils from Northeast Algeria. Agriculture, 10(11), 494. https://doi.org/10.3390/agriculture10110494







