1. Introduction
The herbicide dicamba (3,6-dichloro-2-methoxybenzoic acid) belongs to the benzoic acid chemical group, and is a synthetic auxin [
1]. This postemergence herbicide has been used for a long time to control broadleaf weeds in winter cereals and in pastures in several countries around the globe. However, in recent years, a biotech trait has been introduced to allow the postemergence application of dicamba on soybean, corn and cotton. The Brazilian regulatory agency (CTNBio) has de-regulated dicamba-resistant soybean, corn and cotton in recent years [
2,
3]. The commercialization in Brazil of such crop trait products is planned to start in the 2021/2022 season. The introduction of such technology will serve as an important tool to sustainably manage herbicide-resistant weeds, and to protect the technology and selective herbicide chemistry currently available in the Brazilian market.
Since dicamba is a compound classified as moderately volatile, injuries after dicamba applications have been reported in nonresistant and nontarget plants [
4,
5]. Dicamba is a weak acid (pKa 1.87) that is formulated as a salt The acid form is the volatile form [
6,
7,
8] and the molecular state (whether protonated or deprotonated) may have a substantial impact on volatility [
8,
9]. For transgenic crops resistant to dicamba, new formulations with reduced volatility have been developed. Previous researchers have shown significant reductions in volatility for dicamba diglycolamine salt (DGA) when compared to dimethylamine (DMA) salt [
9]. In addition, a volatility reducer (VG) (acetic acid—VaporGrip
®) was also developed to further decrease the volatility profile of dicamba [
10], by scavenging free protons in the dicamba spray solution [
11]. The volatility of DGA dicamba is much lower than that of the DMA dicamba formulation, and VG technology further reduces volatility when present in the DGA formulation [
7]. However, there is no information in the literature on the use of the VG technology in a tank mix as compared to being part of the formulation itself.
Although the volatility of different dicamba salts in soybean and cotton has been previously investigated [
4,
9,
12,
13,
14,
15], few studies have evaluated the effect of different surfaces, such as soil, straw or inert materials. The amount of volatilized dicamba varies according to the characteristics of the surface onto which the spray droplets are deposited [
16]. The application of DMA dicamba on corn and soybean increased dicamba volatilization by 35% compared to the same product applied on silt loam soil [
8]. With the increased adoption of the no-till system in soybean, corn and cotton, and the double-crop system in the Cerrado region of Brazil (soybean/corn), a significant part of the applied dicamba will be deposited in the corn residue (straw). No-till crop production systems in Brazil occupy around 33 million hectares, and continue to expand [
17], and the interaction of the herbicide with the surface of the straw cover is different from with the soil or leaf surface [
18].
Dicamba is often applied in combination with glyphosate in tank mixtures to increase the efficacy or broaden the spectrum of weed control. Under controlled conditions, Mueller and Steckler [
9] evaluated the application of (1) DGA dicamba + glyphosate (potassium salt), (2) DGA + glyphosate + VG, and (3) DGA + VG in sandy soil at different temperatures. The authors observed that the volatilization of DGA + glyphosate and DGA + glyphosate + VG was higher than that of DGA + VG in all tested temperatures, indicating that the use of glyphosate in the mixture will likely increase overall dicamba volatilization. Thus, our hypotheses for the present study were that (1) the volatilization profile of DGA dicamba (alone or in mixture with glyphosate) changes according to the treated surface, and (2) the volatilization reducer (acetic acid—VaporGrip
®) alters the overall volatility profile of DGA dicamba. The objective of this study was to quantify the volatilization of standalone DGA dicamba or in mixture with glyphosate (potassium salt), with and without the volatilization reducer (acetic acid—VaporGrip
®) from different surfaces.
2. Materials and Methods
The experiment was conducted under laboratory conditions. DGA dicamba (Atectra
®, 480 g ae L
−1, BASF, Guaratinguetá, SP, Brazil) was applied at 720 g ae ha
−1 (highest label dose in Brazil) alone and in mixture with glyphosate (potassium salt) (Roundup Transorb R
®, 480 g ae L
−1, Monsanto, São José dos Campos, SP, Brazil) at 1440 g ae ha
−1 (intermediate label dose in Brazil) with 0.5%
v/
v of the volatility reducer VaporGrip
® (VG) (Monsanto, São José dos Campos, SP, Brazil) (
Table 1). The treatments were applied to different surfaces (glass, corn straw, dry soil, and moist soil).
For the glass surface, the herbicide was sprayed on a slide measuring 2.5 × 7.6 cm. For the straw surface, dry corn leaf cut into pieces of 6 × 8 cm was used; the application was made covering 1 cm of the edges with a rectangular frame, resulting in a useful area of 35 cm2. The soil used was collected from a herbicide-free area and had the following physicochemical characteristics: 95.8% sand, 2.9% clay, 1.3% silt, 6% organic matter, pH 7.0, and a cation exchange capacity (CEC) of 44 mmolc dm−3. The soil was placed in an aluminum tray with a volume of 12.25 cm3 (1.4 × 7.0 × 0.8 cm). At the time of application, the entire outer side of the tray was covered with adhesive tape to avoid contamination with the herbicide. For the moist soil treatment, 2 mL of distilled water was used to moisten the soil. The different surfaces were used without herbicide as control treatments.
The experiment was conducted in duplicate with a completely randomized design with three replications, and each experimental unit consisted of one surface. The products were applied using an automated sprayer located in a closed environment with a metal structure to support the 2 m long spray boom. Four XR 11002 VS nozzles (TeeJet Spraying Systems Company, Wheaton, IL, USA) were used, spaced 0.5 m apart and placed at a height of 0.5 m above the targets. The boom pressure was 200 kPa with a speed of 3.6 km h−1, generating a spray volume of 200 L ha−1.
The vapor collection system consisted of a closed pipe system with inputs to hold 24 cartridges and an outlet containing a capillary (3 mm) connected to a vacuum pump to create an air flow into the cartridge (30 mL min
−1). This system was placed inside a chromatograph oven, which was used as a precision chamber to maintain a constant temperature at 40 °C (
Figure 1). The cartridges containing the targets were 19.2 cm long with a volume of 132 cm
3. The cartridges were sealed with a lid that has a 3 mm diameter opening that served to ensure air passage through the system. Two filters (PVDF 0.20 μm–25 mm Chromafil Xtra) were used on the air exit of the cartridge to capture the dicamba vapor in the air. The filters were arranged in a series to ensure high vapor collection efficiency.
After application, the targets were left for 10 min to dry. At the end of this period, the protective material at the edges of the straws and the outer walls of the trays containing soil were removed. Each experimental unit was placed inside a sealed cartridge to prevent the loss of the volatilized herbicide (
Figure 1A). The cartridges containing the surfaces and the respective filters were placed in the vapor collection system and kept in the oven (
Figure 1B,C) at a constant temperature of 40 °C and relative humidity of 20% for 24 h.
After this 24 h period, dicamba was extracted from the surfaces, cartridges, and filters used. The straw and glass slides were removed from the cartridges and placed in a 50 mL centrifuge tube, to which 40 mL of methanol:distilled water (75:25 v/v) was added. The tubes were placed in an ultrasound bath for 30 min, and transferred to other containers.
The soil was placed in 10 mL plastic cartridges containing two fiberglass membranes in the bottom. The extraction was performed using a vacuum manifold system. Dicamba was eluted from soil with 40 mL of extraction solution (methanol:distilled water—75:25 v/v).
To determine the amount of dicamba that was retained on the cartridge walls, 10 mL of the extraction solution was added to the syringes, which were sealed at both ends for subsequent stirring for 5 min on a shaker table. To determine the amount of volatilized dicamba, the two filters were individually washed using 1.5 mL of extraction solution, generating samples for filter 1 (first filter of the series) and filter 2 (last filter of the series), which were stored at −20 °C. All extracted solutions were filtered using Millipore 0.45 μm syringe filters and transferred to 2 mL vials.
Dicamba was analyzed using a liquid chromatography–tandem mass spectrometry (LC-MS/MS) system composed of a high-performance liquid chromatograph (Prominence UFLC, Shimadzu, Kyoto, Japan) equipped with two LC-20AD pumps, a SIL-20AC autoinjector, a DGU-20A5 degasser, a CBM-20A controller system, and a CTO-20AC oven. The chromatograph was coupled to a Triple Quad 4500 mass spectrometer (Applied Biosystems, Foster City, CA, USA). Chromatographic analyses were performed with a C18 column (Phenomenex Gemini 5 μ C18RP 110Å) using an injection volume of 20 μL, with 5 mM ammonium acetate (Avantor Performance Materials, Inc., Center Valley, PA, USA) in water and 5 mM ammonium acetate in methanol (Merck KGaA, Darmstadt, Germany). The flow rate used was 1.0 mL min
−1, and the ratio of the solvents was gradually increased from the 80:20 (methanol/water) to the 95:5 range from 0 to 4 min, maintained at 95:5 range from 4 to 10 min, and returned to the initial condition. The total running time was 12 min. The retention time of dicamba in this system was 5.68 min. The electrospray ionization source (ESI) was used in the negative mode. Eight concentrations of the dicamba analytical standards with a certified purity level of 99.9% (Sigma Aldrich, St Louis, MO, USA) were used to construct the calibration curve. The limit of quantification (LOQ) for dicamba was 0.099 µg L
−1, determined according to ICH guidelines [
19].
The data obtained after the quantification of dicamba in the filters (ng) were converted to ng cm
−2 or percentage of the total deposition (sum of the total dicamba weight for each experimental unit). The data obtained in duplicate were submitted to analysis of variance independently, and the homogeneity test of residual variances (Fmax) was applied [
20]. Proving the homogeneity between the experiments, the data were grouped in an experiment with six replications. The 16 treatments were arranged in a factorial scheme with four herbicide treatments and four surfaces. The data were subjected to analysis of variance (ANOVA) by the F test at 5% probability, determining the effects of herbicide treatments, surfaces, and the interaction between them. Both the comparison of surfaces for each herbicide treatment and the comparison of herbicides for each surface are highly relevant in practical terms. The comparison between the herbicide treatments for each surface and between the surfaces for each herbicide treatment was done using the Tukey’s test at 5% probability using the statistical software Sisvar, version 5.6 (Sisvar
®, Lavras, MG, Brazil) [
21]. The confidence interval for each mean was established (values are reported as the mean ± confidence interval).
3. Results and Discussion
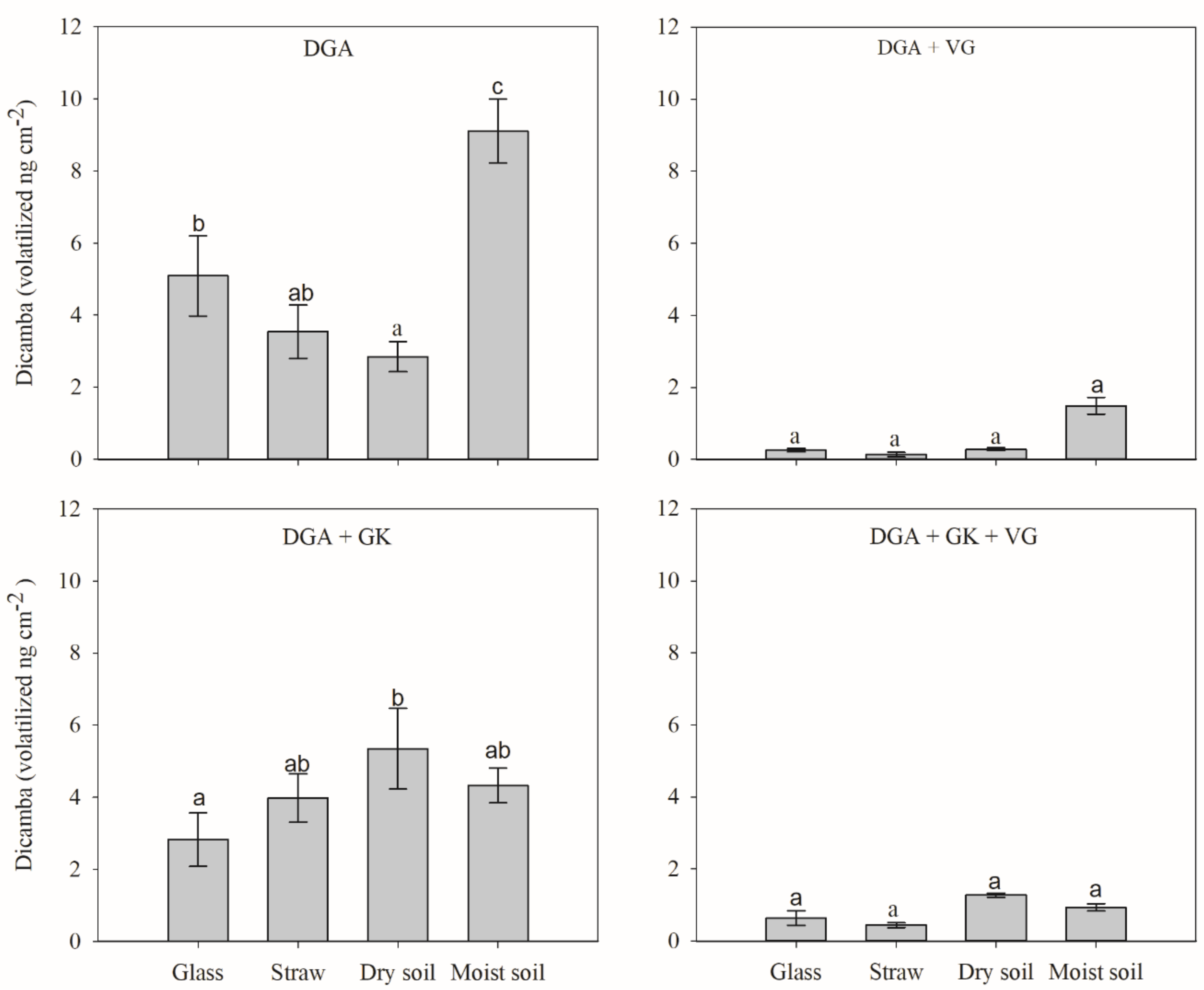
The interaction between surfaces and herbicide treatments was significant. The amount of volatilized dicamba was significantly influenced by the type of surface (
Figure 2). This was due to the differences in the physical and chemical properties of the surfaces whereon dicamba was applied. The glass surface used as reference was inert and smooth, which does not represent any surface that could be potentially sprayed with dicamba. In contrast, dicamba can interact with ions on other surfaces, such as soil, straw or a plant surface, and be absorbed or adsorbed [
22]. Information on the effect of the treated surface on dicamba volatilization is rare in the literature, and data on the effect of straw are very important, especially in Brazil, where most dicamba applications will be performed in the no-tillage system. Behrens and Leuschen [
8] reported that approximately more dicamba was volatilized on corn and soybean leaves than on a silt loam soil.
The volatility was much lower for the DGA + VG and DGA + glyphosate potassium salt (GK) + VG treatments when compared to the same treatments without VG, regardless of the surface tested. When VG was added to dicamba, with or without potassium glyphosate, no differences were observed between the different surfaces. For treatments without VG, different levels of volatility were quantified depending on the surfaces, with the highest amounts of volatilized dicamba for the standalone DGA in wet soil and for DGA + GK in dry soil, wet soil and straw.
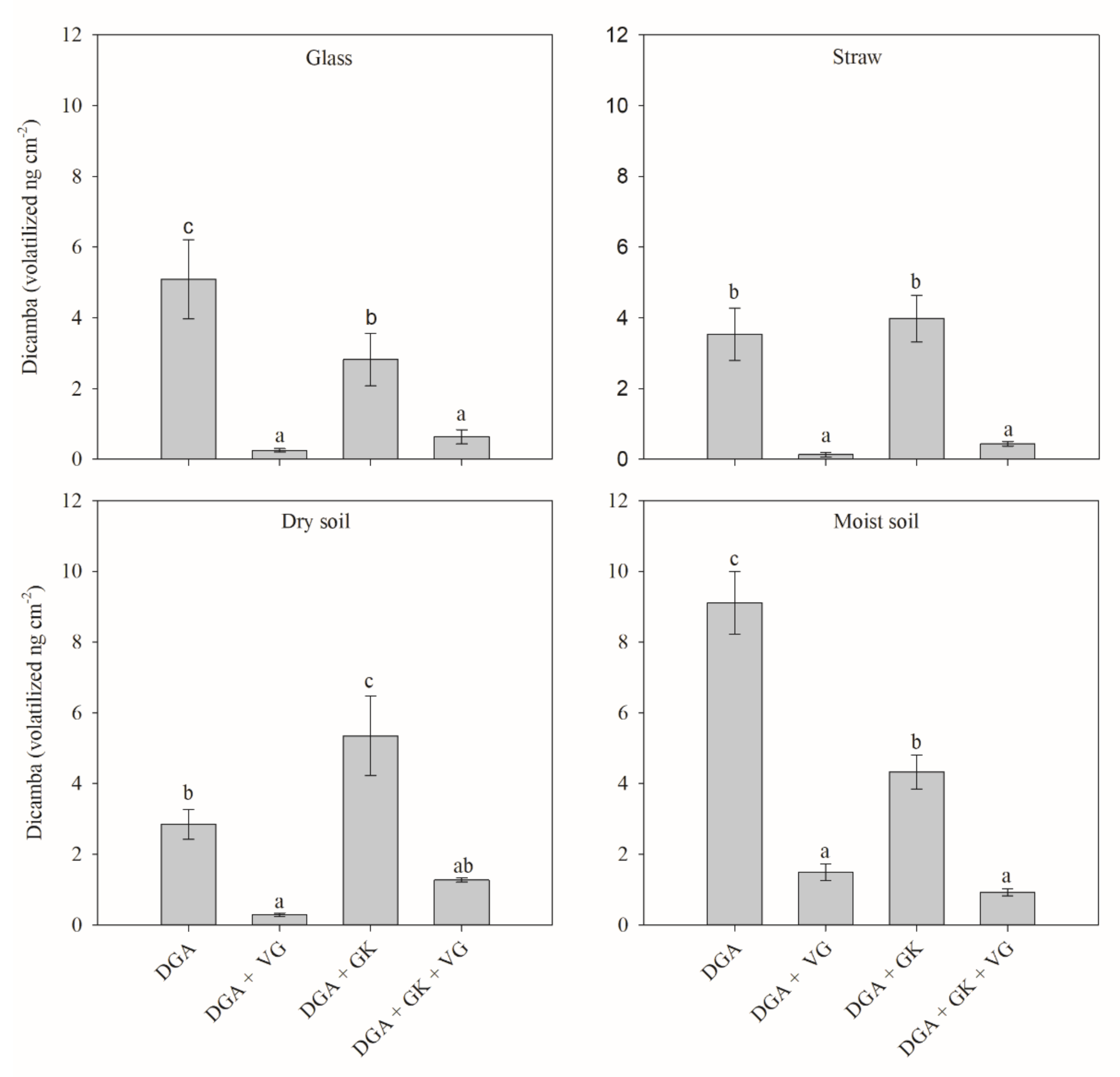
The volatilization of dicamba was also significantly influenced by the herbicide treatments (
Figure 3). The characteristics of each surface and the tank mixture components played an essential role in the volatility profile of dicamba [
16]. The data are discussed by surface to facilitate comprehension.
Glass. On the smooth glass surface, standalone DGA dicamba had the highest volatility (
Figure 3). The mixture of DGA dicamba with potassium glyphosate reduced the amount of volatilized dicamba by approximately 45%. When VG was used with DGA and DGA + glyphosate potassium salt (GK), the reductions in volatilization were 95% and 77%, respectively (compared to DGA and DGA + GK respectively).
Straw. When the herbicide treatments were applied over the straw, no differences were observed between standalone DGA dicamba and the mixture of DGA dicamba and glyphosate (
Figure 3). The addition of VG to the spray solution containing DGA dicamba alone and DGA dicamba plus glyphosate reduced the amount of volatilized dicamba by up to 96% and 88%, respectively.
Dry soil. Dry soil was the only surface for which the addition of glyphosate in the solution resulted in an increase of dicamba volatility when compared to dicamba DGA standalone. However, the addition of VG in the solution significantly decreased the amount of dicamba volatilization regardless of the herbicide treatment. Similar results were found by Mueller and Steckler [
9], where greater amounts of volatilized dicamba were observed when dicamba DGA was applied with glyphosate on dry soil, especially at higher temperatures. These same authors also reported a decrease in volatilization when Dicamba DGA with VaporGrip
® was in the solution, compared to dicamba DGA without VaporGrip
®.
Moist Soil. High concentrations of volatilized dicamba (9.11 ng cm
−2) were detected in the dicamba DGA standalone treatment (
Figure 3). This may have occurred due to the high water solubility of dicamba (400,000 mg L
−1) together with its low affinity with the solid soil phase (Koc = 2) [
23]. Similar to that of the glass surface, the addition of potassium glyphosate significantly decreased the amount of dicamba volatilized (52.5% reduction), and the inclusion of VG further reduced the volatility of dicamba significantly compared to the herbicide solutions without VG.
The off-target movement of dicamba can occur as physical particle drift, tank contamination, movement with dust, runoff or volatility [
24]. However, damage caused by dicamba volatility, with DGA salt, is considered low [
7]. According to Miller et al. [
25], the amount detected of volatilized dicamba from DGA salt formulation was half that of the DMA salt formulation. Furthermore, the addition of volatilization reducers further reduces the volatility potential of DGA dicamba [
7]. When applied alone, DGA volatilization was generally the lowest in dry soil, and when applied alone or with GK (without VG), the DGA volatilizations from dry soil and straw were similar. These results show that when the VG is used with DGA, the presence or absence of straw, or the condition of soil surface moisture, is not important, and the levels of volatility are similar. Overall, the largest volatilization problem occurred when DGA was applied on moist, bare soil (
Figure 2). This is an important finding for the Brazilian agricultural system, since the majority of the soybean production system is under no-till. Despite this, the small amounts of dicamba volatilized in all treatments and the best practices in the use of this herbicide must be carefully considered. Another point to be considered is that the application of dicamba on straw causes low volatility, as described, however the application of herbicides on straw can modify the degradation of these compounds in the environment, as well as their availability and persistence in the soil [
18,
26,
27]. This is a topic that deserves attention, since dicamba may have a short soil persistence and cause injury to the following crop.
Some researchers reported negative effects on soybean susceptible to dicamba at very low doses. They evaluated the response of soybean to low dicamba doses (2.18 and 8.75 g ha
−1), which represented 1/330 and 1/82 of the dose of DGA dicamba used in this study, and observed crop injury, albeit to a lesser extent or even nonexistent at the lowest dose, depending on the variable and variety evaluated [
28]. Greater reductions were observed at even lower doses, such as a 10% reduction in soybean yield with doses ranging from 0.32 to 0.63 g ha
−1 (~1/2250 to ~1/1140 times the maximum recommended dose) [
29]. Those results, as well as the those reported in the present experiment, demonstrate that the use of volatilization reducers combined with the use of dicamba in the DGA form considerably decreases the concentration of dicamba in the air, to levels that will likely not cause significant injury in nontarget crops, since the damage resulting from volatilized dicamba rates is minimal [
30,
31,
32].
High temperature and low humidity promote dicamba volatility [
9,
25], so the effects observed in the reduction of volatilization are very strong, since the tested condition was high temperature and low humidity. Twenty-three field experiments were carried out in six locations in the USA to provide an estimate of the volatility of dicamba in post-application [
33]. In these experiments, the volatilization of all formulations and conditions tested varied from 0.023 ± 0.003% to 0.302 ± 0.045% of the applied dicamba, and the volatilization peaks occurred within the first 24 h after application. This information shows the importance of the first 24 h in terms of the dicamba’s volatility, as quantified in this study.
Another important fact is that the addition of GK to DGA significantly reduced the volatilization from the inert surface (glass) and from bare, moist soil (
Figure 3). There is not enough information to explain this effect. However, for the two surfaces with greater dicamba retention (dry soil and dry straw), this effect was not observed. On the glass surface, in which there is no retention, and on the moist soil in which dicamba is more available, less volatilization occurred in the presence of glyphosate. It is not possible, with these results, to understand if the effect is related to glyphosate or other components of its formulation.
The method used in this study to quantify DGA dicamba volatility provides a rapid and consistent means of measuring volatilization. DGA dicamba volatilization was significantly influenced by the surface on which the product was applied, and the presence of potassium glyphosate and volatility reducer in the solution. Further research, with a focus on deeply understanding the reason for this interaction, shall be performed. Mixing dicamba and potassium glyphosate will likely be a common practice among Brazilian farmers. Furthermore, this tank mixture will most likely be applied on corn straw in Brazil in regions where thousands of hectares of corn are produced. The information in this research underlines the need for volatility reducers to minimize the risk of vapor drift from dicamba applications. The use of other possible tank partners, such as other herbicides, fungicides and insecticides, should be evaluated to avoid harmful volatilization levels of dicamba in the future.