Quantitative Analysis of Somatostatin and Dopamine Receptors Gene Expression Levels in Non-functioning Pituitary Tumors and Association with Clinical and Molecular Aggressiveness Features
Abstract
1. Introduction
2. Experimental Section
2.1. Patients and Samples
2.2. Histopathology and Immunohistochemistry
2.3. RNA Isolation, Reverse Transcription, and Analysis of Gene Expression by Quantitative Real-Time PCR
2.4. Statistical Analysis
3. Results
3.1. Patient and Sample Characteristics
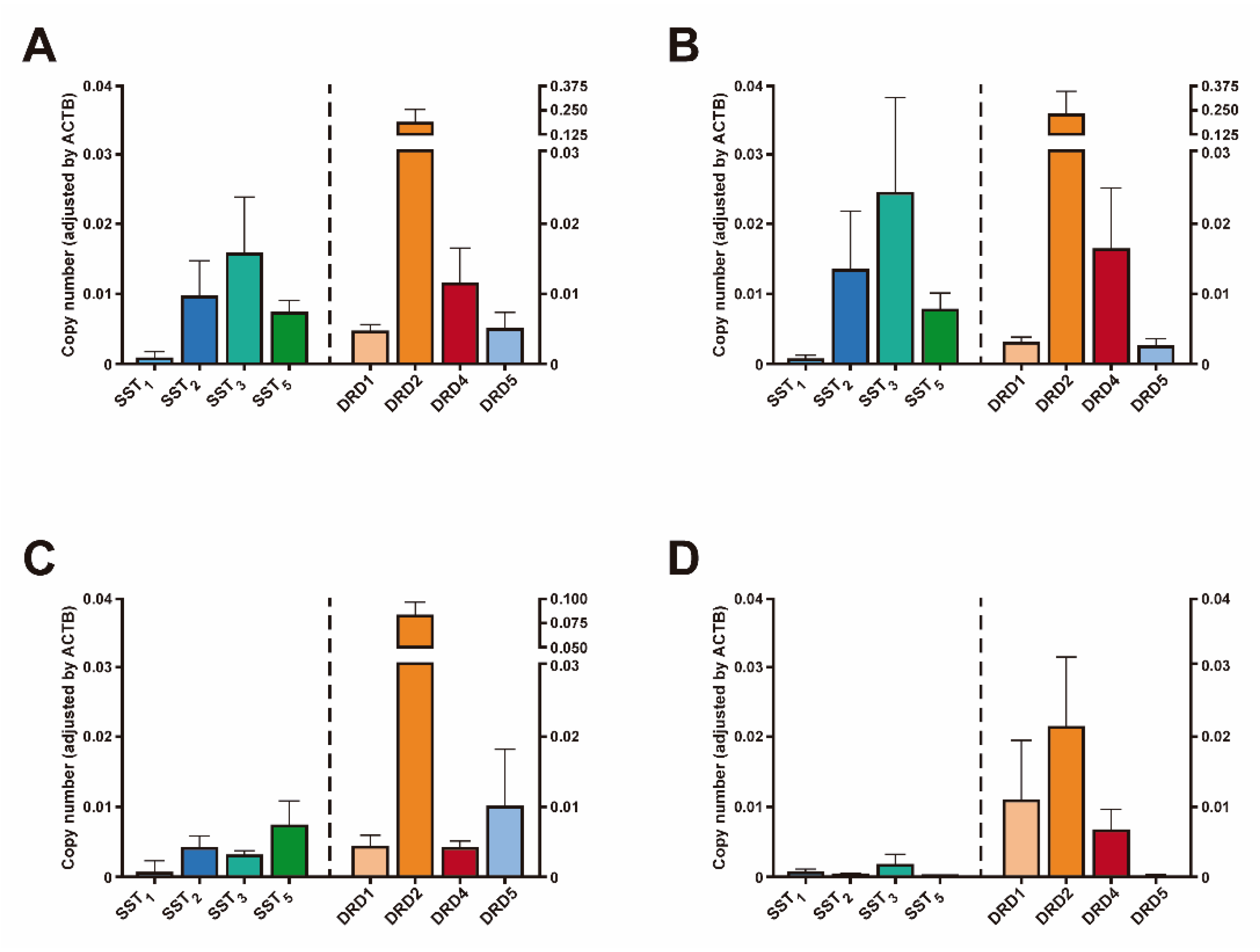
3.2. Receptor Expression Levels in NFPTs
3.3. Association between Clinical Characteristics of NFPTs and SSTs or DRDs mRNA Levels
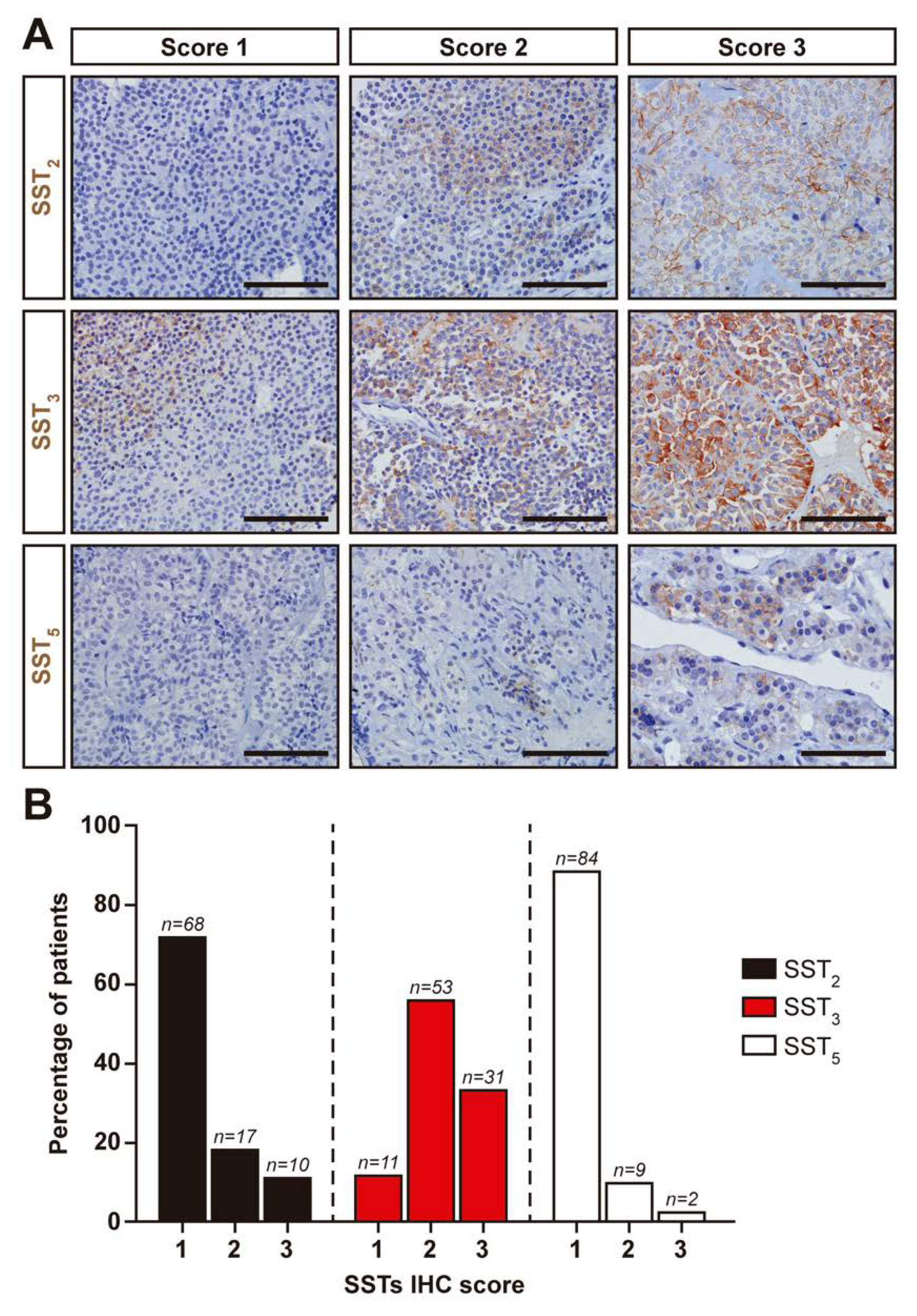
3.4. SSTs Expression in NFPTs as Assessed by Immunohistochemistry
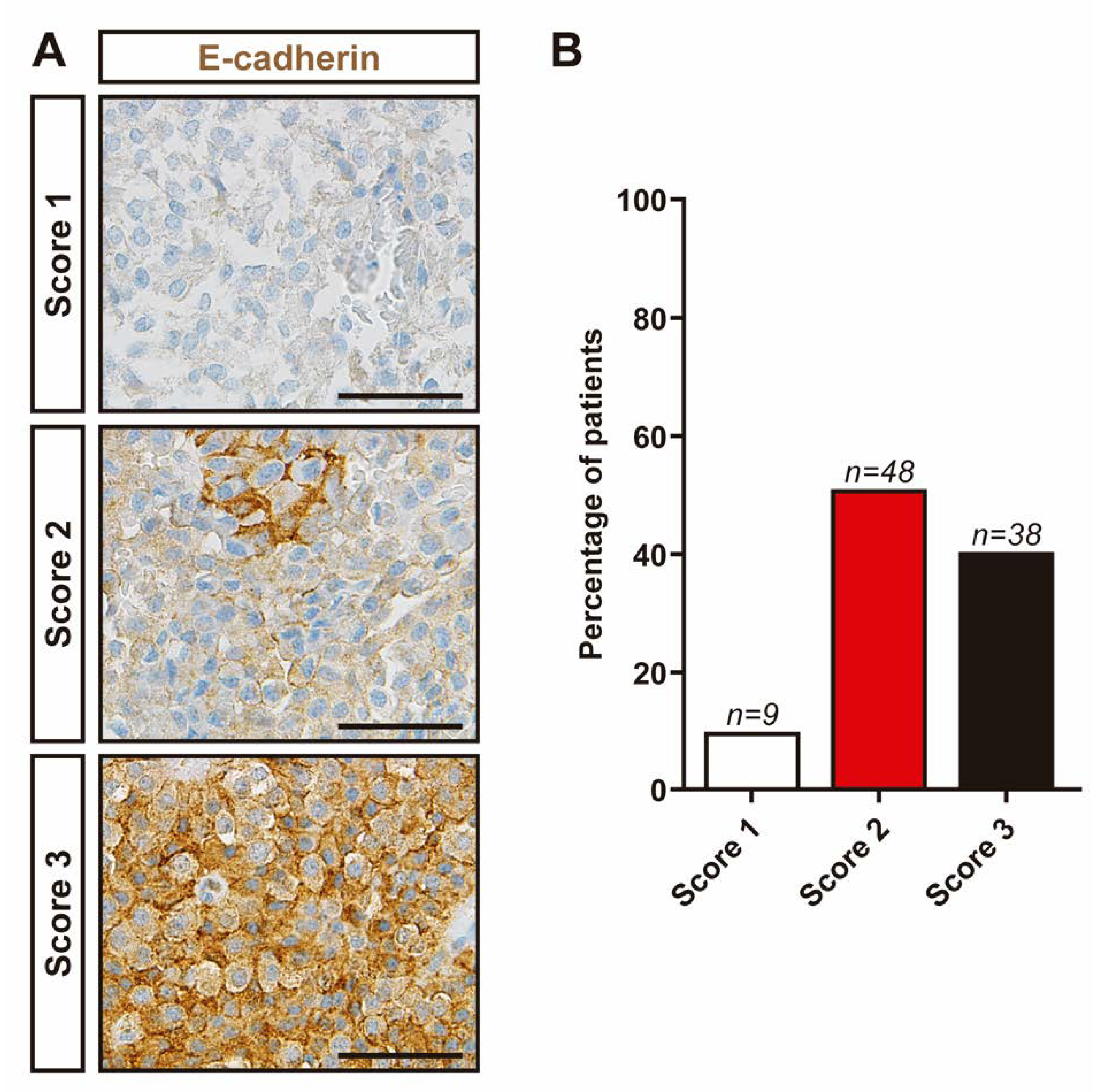
3.5. Evaluation of Molecular Markers Associated with Aggressive Features in NFPTs
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Melmed, S. Pituitary-tumor endocrinopathies. N. Engl. J. Med. 2020, 382, 937–950. [Google Scholar] [CrossRef] [PubMed]
- Ntali, G.; Wass, J.A. Epidemiology, clinical presentation and diagnosis of non-functioning pituitary adenomas. Pituitary 2018, 21, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Dworakowska, D.; Grossman, A.B. Aggressive and malignant pituitary tumours: State-of-the-art. Endocr. Relat. Cancer 2018, 25, R559–R575. [Google Scholar] [CrossRef]
- Øystese, K.A.; Evang, J.A.; Bollerslev, J. Non-Functioning pituitary adenomas: Growth and aggressiveness. Endocrine 2016, 53, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Zatelli, M.C. Pathogenesis of non-functioning pituitary adenomas. Pituitary 2018, 21, 130–137. [Google Scholar] [CrossRef]
- Penn, D.L.; Burke, W.T.; Laws, E.R. Management of non-functioning pituitary adenomas: Surgery. Pituitary 2018, 21, 145–153. [Google Scholar] [CrossRef]
- Greenman, Y. Management of endocrine disease: Present and future perspectives for medical therapy of nonfunctioning pituitary adenomas. Eur. J. Endocrinol. 2017, 177, R113–R124. [Google Scholar] [CrossRef]
- Even-Zohar, N.; Greenman, Y. Management of NFAs: Medical treatment. Pituitary 2018, 21, 168–175. [Google Scholar] [CrossRef]
- Greenman, Y.; Cooper, O.; Yaish, I.; Robenshtok, E.; Sagiv, N.; Jonas-Kimchi, T.; Yuan, X.; Gertych, A.; Shimon, I.; Ram, Z.; et al. Treatment of clinically nonfunctioning pituitary adenomas with dopamine agonists. Eur. J. Endocrinol. 2016, 175, 63–72. [Google Scholar] [CrossRef]
- Günther, T.; Tulipano, G.; Dournaud, P.; Bousquet, C.; Csaba, Z.; Kreienkamp, H.-J.; Lupp, A.; Korbonits, M.; Castaño, J.P.; Wester, H.-J.; et al. International union of basic and clinical pharmacology. CV. Somatostatin receptors: Structure, function, ligands, and new nomenclature. Pharmacol. Rev. 2018, 70, 763. [Google Scholar] [CrossRef]
- Vázquez-Borrego, M.C.; Gupta, V.; Ibáñez-Costa, A.; Gahete, M.D.; Venegas-Moreno, E.; Toledano-Delgado, Á.; Cano, D.A.; Blanco-Acevedo, C.; Ortega-Salas, R.; Japón, M.A.; et al. Somatostatin receptor subtype-3 (SST3) peptide agonist shows antitumor effects in experimental models of nonfunctioning pituitary tumors. Clin. Cancer Res. 2020, 26, 957–969. [Google Scholar] [CrossRef]
- Taboada, G.F.; Luque, R.M.; Bastos, W.; Guimarães, R.F.C.; Marcondes, J.B.; Chimelli, L.M.C.; Fontes, R.; Mata, P.J.P.; Filho, P.N.; Carvalho, D.P.; et al. Quantitative analysis of somatostatin receptor subtype (SSTR1-5) gene expression levels in somatotropinomas and non-functioning pituitary adenomas. Eur. J. Endocrinol. 2007, 156, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Tateno, T.; Kato, M.; Tani, Y.; Oyama, K.; Yamada, S.; Hirata, Y. Differential expression of somatostatin and dopamine receptor subtype genes in adrenocorticotropin (ACTH)-secreting pituitary tumors and silent corticotroph adenomas. Endocr. J. 2009, 56, 579–584. [Google Scholar] [CrossRef]
- Lee, M.; Lupp, A.; Mendoza, N.; Martin, N.; Beschorner, R.; Honegger, J.; Schlegel, J.; Shively, T.; Pulz, E.; Schulz, S.; et al. SSTR3 is a putative target for the medical treatment of gonadotroph adenomas of the pituitary. Endocr. Relat. Cancer 2015, 22, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Ramírez, C.; Cheng, S.; Vargas, G.; Asa, S.L.; Ezzat, S.; González, B.; Cabrera, L.; Guinto, G.; Mercado, M. Expression of Ki-67, PTTG1, FGFR4, and SSTR 2, 3, and 5 in nonfunctioning pituitary adenomas: A high throughput tma, immunohistochemical study. J. Clin. Endocrinol. Metab. 2012, 97, 1745–1751. [Google Scholar] [CrossRef] [PubMed]
- Manojlovic-Gacic, E.; Engström, B.E.; Casar-Borota, O. Histopathological classification of non-functioning pituitary neuroendocrine tumors. Pituitary 2018, 21, 119–129. [Google Scholar] [CrossRef]
- Venegas-Moreno, E.; Flores-Martinez, A.; Dios, E.; Vazquez-Borrego, M.C.; Ibañez-Costa, A.; Madrazo-Atutxa, A.; Japón, M.A.; Castaño, J.P.; Luque, R.M.; Cano, D.A.; et al. E-cadherin expression is associated with somatostatin analogue response in acromegaly. J. Cell. Mol. Med. 2019, 23, 3088–3096. [Google Scholar] [CrossRef]
- Venegas-Moreno, E.; Vazquez-Borrego, M.C.; Dios, E.; Gros-Herguido, N.; Flores-Martinez, A.; Rivero-Cortés, E.; Madrazo-Atutxa, A.; Japón, M.A.; Luque, R.M.; Castaño, J.P.; et al. Association between dopamine and somatostatin receptor expression and pharmacological response to somatostatin analogues in acromegaly. J. Cell. Mol. Med. 2018, 22, 1640–1649. [Google Scholar] [CrossRef]
- Luque, R.M.; Ibáñez-Costa, A.; Sánchez-Tejada, L.; Rivero-Cortés, E.; Robledo, M.; Madrazo-Atutxa, A.; Mora, M.; Álvarez, C.V.; Lucas-Morante, T.; Álvarez-Escolá, C.; et al. The molecular registry of pituitary adenomas (REMAH): A bet of Spanish endocrinology for the future of individualized medicine and translational research. Endocrinol. Nutr. 2016, 63, 274–284. [Google Scholar] [CrossRef]
- Neto, L.V.; Machado, E.d.O.; Luque, R.M.; Taboada, G.F.; Marcondes, J.B.; Chimelli, L.M.C.; Quintella, L.P.; Niemeyer, P.; de Carvalho, D.P.; Kineman, R.D.; et al. Expression analysis of dopamine receptor subtypes in normal human pituitaries, nonfunctioning pituitary adenomas and somatotropinomas, and the association between dopamine and somatostatin receptors with clinical response to octreotide-LAR in acromegaly. J. Clin. Endocrinol. Metab. 2009, 94, 1931–1937. [Google Scholar] [CrossRef]
- Ibáñez-Costa, A.; Gahete, M.D.; Rivero-Cortés, E.; Rincón-Fernández, D.; Nelson, R.; Beltrán, M.; de la Riva, A.; Japón, M.A.; Venegas-Moreno, E.; Gálvez, M.Á.; et al. In1-Ghrelin splicing variant is overexpressed in pituitary adenomas and increases their aggressive features. Sci. Rep. 2015, 5, 8714. [Google Scholar] [CrossRef] [PubMed]
- Ibáñez-Costa, A.; Rivero-Cortés, E.; Vázquez-Borrego, M.C.; Gahete, M.D.; Jiménez-Reina, L.; Venegas-Moreno, E.; de la Riva, A.; Arráez, M.Á.; González-Molero, I.; Schmid, H.A.; et al. Octreotide and pasireotide (dis)similarly inhibit pituitary tumor cells in vitro. J. Endocrinol. 2016, 231, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Luque, R.M.; Ibáñez-Costa, A.; Neto, L.V.; Taboada, G.F.; Hormaechea-Agulla, D.; Kasuki, L.; Venegas-Moreno, E.; Moreno-Carazo, A.; Gálvez, M.Á.; Soto-Moreno, A.; et al. Truncated somatostatin receptor variant sst5TMD4 confers aggressive features (proliferation, invasion and reduced octreotide response) to somatotropinomas. Cancer Lett. 2015, 359, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Ben-Shlomo, A.; Cooper, O. Silent corticotroph adenomas. Pituitary 2018, 21, 183–193. [Google Scholar] [CrossRef]
- Taboada, G.F.; Luque, R.M.; Neto, L.V.; Machado, E.d.O.; Sbaffi, B.C.; Domingues, R.C.; Marcondes, J.B.; Chimelli, L.M.C.; Fontes, R.; Niemeyer, P.; et al. Quantitative analysis of somatostatin receptor subtypes (1-5) gene expression levels in somatotropinomas and correlation to in vivo hormonal and tumor volume responses to treatment with octreotide LAR. Eur. J. Endocrinol. 2008, 158, 295–303. [Google Scholar] [CrossRef]
- Greenman, Y.; Melmed, S. Expression of three somatostatin receptor subtypes in pituitary adenomas: Evidence for preferential SSTR5 expression in the mammosomatotroph lineage. J. Clin. Endocrinol. Metab. 1994, 79, 724–729. [Google Scholar] [CrossRef]
- Saeger, W.; Lüdecke, D.K.; Buchfelder, M.; Fahlbusch, R.; Quabbe, H.-J.; Petersenn, S. Pathohistological classification of pituitary tumors: 10 years of experience with the German Pituitary Tumor Registry. Eur. J. Endocrinol. 2007, 156, 203–216. [Google Scholar] [CrossRef]
- Gabalec, F.; Beranek, M.; Netuka, D.; Masopust, V.; Nahlovsky, J.; Cesak, T.; Marek, J.; Cap, J. Dopamine 2 receptor expression in various pathological types of clinically non-functioning pituitary adenomas. Pituitary 2012, 15, 222–226. [Google Scholar] [CrossRef]
- Gagliano, T.; Filieri, C.; Minoia, M.; Buratto, M.; Tagliati, F.; Ambrosio, M.R.; Lapparelli, M.; Zoli, M.; Frank, G.; degli Uberti, E.; et al. Cabergoline reduces cell viability in non functioning pituitary adenomas by inhibiting vascular endothelial growth factor secretion. Pituitary 2013, 16, 91–100. [Google Scholar] [CrossRef]
- Florio, T.; Barbieri, F.; Spaziante, R.; Zona, G.; Hofland, L.J.; van Koetsveld, P.M.; Feelders, R.A.; Stalla, G.K.; Theodoropoulou, M.; Culler, M.D.; et al. Efficacy of a dopamine-somatostatin chimeric molecule, BIM-23A760, in the control of cell growth from primary cultures of human non-functioning pituitary adenomas: A multi-center study. Endocr. Relat. Cancer 2008, 15, 583–596. [Google Scholar] [CrossRef]
- Su, Z.; Wang, C.; Wu, J.; Jiang, X.; Chen, Y.; Chen, Y.; Zheng, W.; Zhuge, Q.; Wu, Z.; Zeng, Y. Expression of dopamine 2 receptor subtype MRNA in clinically nonfunctioning pituitary adenomas. Neurol. Sci. 2012, 33, 275–279. [Google Scholar] [CrossRef] [PubMed]
- Colao, A.; Ferone, D.; Lastoria, S.; Cerbone, G.; Di Sarno, A.; Di Somma, C.; Lucci, R.; Lombardi, G. Hormone levels and tumour size response to quinagolide and cabergoline in patients with prolactin-secreting and clinically non-functioning pituitary adenomas: Predictive value of pituitary scintigraphy with 123I-methoxybenzamide. Clin. Endocrinol. (Oxf.) 2000, 52, 437–445. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, S.; Mellemkjaer, S.; Rasmussen, L.M.; Ledet, T.; Olsen, N.; Bojsen-Møller, M.; Astrup, J.; Weeke, J.; Jørgensen, J.O. Expression of somatostatin receptors on human pituitary adenomas in vivo and ex vivo. J. Endocrinol. Investig. 2001, 24, 430–437. [Google Scholar] [CrossRef]
- Florio, T.; Thellung, S.; Arena, S.; Corsaro, A.; Spaziante, R.; Gussoni, G.; Acuto, G.; Giusti, M.; Giordano, G.; Schettini, G. Somatostatin and its analog lanreotide inhibit the proliferation of dispersed human non-functioning pituitary adenoma cells in vitro. Eur. J. Endocrinol. 1999, 141, 396–408. [Google Scholar] [CrossRef] [PubMed]
- Zatelli, M.C.; Piccin, D.; Bottoni, A.; Ambrosio, M.R.; Margutti, A.; Padovani, R.; Scanarini, M.; Taylor, J.E.; Culler, M.D.; Cavazzini, L.; et al. Evidence for differential effects of selective somatostatin receptor subtype agonists on alpha-subunit and chromogranin a secretion and on cell viability in human nonfunctioning pituitary adenomas in vitro. J. Clin. Endocrinol. Metab. 2004, 89, 5181–5188. [Google Scholar] [CrossRef]
- Vandeva, S.; Daly, A.F.; Petrossians, P.; Zacharieva, S.; Beckers, A. Somatic and germline mutations in the pathogenesis of pituitary adenomas. Eur. J. Endocrinol. 2019, 181, R235–R254. [Google Scholar] [CrossRef]
- Neou, M.; Villa, C.; Armignacco, R.; Jouinot, A.; Raffin-Sanson, M.-L.; Septier, A.; Letourneur, F.; Diry, S.; Diedisheim, M.; Izac, B.; et al. Pangenomic classification of pituitary neuroendocrine tumors. Cancer Cell 2020, 37, 123–134.e5. [Google Scholar] [CrossRef]
- Beckers, A.; Aaltonen, L.A.; Daly, A.F.; Karhu, A. Familial isolated pituitary adenomas (FIPA) and the pituitary adenoma predisposition due to mutations in the aryl hydrocarbon receptor interacting protein (AIP) gene. Endocr. Rev. 2013, 34, 239–277. [Google Scholar] [CrossRef]
- Elston, M.S.; Gill, A.J.; Conaglen, J.V.; Clarkson, A.; Cook, R.J.; Little, N.S.; Robinson, B.G.; Clifton-Bligh, R.J.; McDonald, K.L. Nuclear accumulation of E-cadherin correlates with loss of cytoplasmic membrane staining and invasion in pituitary adenomas. J. Clin. Endocrinol. Metab. 2009, 94, 1436–1442. [Google Scholar] [CrossRef]
- Fougner, S.L.; Lekva, T.; Borota, O.C.; Hald, J.K.; Bollerslev, J.; Berg, J.P. The expression of E-cadherin in somatotroph pituitary adenomas is related to tumor size, invasiveness, and somatostatin analog response. J. Clin. Endocrinol. Metab. 2010, 95, 2334–2342. [Google Scholar] [CrossRef]
- Zhou, W.; Song, Y.; Xu, H.; Zhou, K.; Zhang, W.; Chen, J.; Qin, M.; Yi, H.; Gustafsson, J.-A.; Yang, H.; et al. In nonfunctional pituitary adenomas, estrogen receptors and slug contribute to development of invasiveness. J. Clin. Endocrinol. Metab. 2011, 96, E1237–E1245. [Google Scholar] [CrossRef] [PubMed]
- Qian, Z.R.; Sano, T.; Yoshimoto, K.; Asa, S.L.; Yamada, S.; Mizusawa, N.; Kudo, E. Tumor-specific downregulation and methylation of the CDH13 (H-cadherin) and CDH1 (E-cadherin) genes correlate with aggressiveness of human pituitary adenomas. Mod. Pathol. 2007, 20, 1269–1277. [Google Scholar] [CrossRef] [PubMed]
- Chauvet, N.; Romanò, N.; Meunier, A.-C.; Galibert, E.; Fontanaud, P.; Mathieu, M.-N.; Osterstock, G.; Osterstock, P.; Baccino, E.; Rigau, V.; et al. Combining cadherin expression with molecular markers discriminates invasiveness in growth hormone and prolactin pituitary adenomas. J. Neuroendocrinol. 2016, 28. [Google Scholar] [CrossRef] [PubMed]
- Ongaratti, B.R.; Haag, T.; D’Ávila, M.F.; Trott, G.; Ferreira, N.P.; Rech, C.G.S.L.; Pereira-Lima, J.F.S.; da Costa Oliveira, M. Gene and protein expression of E-cadherin and NCAM markers in non-functioning pituitary adenomas. Ann. Diagn. Pathol. 2019, 38, 59–61. [Google Scholar] [CrossRef]
- Yamada, S.; Ohyama, K.; Taguchi, M.; Takeshita, A.; Morita, K.; Takano, K.; Sano, T. A Study of the correlation between morphological findings and biological activities in clinically nonfunctioning pituitary adenomas. Neurosurgery 2007, 61, 580–584, discussion 584–585. [Google Scholar] [CrossRef]
- Raverot, G.; Burman, P.; McCormack, A.; Heaney, A.; Petersenn, S.; Popovic, V.; Trouillas, J.; Dekkers, O.M.; European Society of Endocrinology. European Society of Endocrinology clinical practice guidelines for the management of aggressive pituitary tumours and carcinomas. Eur. J. Endocrinol. 2018, 178, G1–G24. [Google Scholar] [CrossRef]
- Ogawa, Y.; Jokura, H.; Niizuma, K.; Tominaga, T. Mid-term prognosis of non-functioning pituitary adenomas with high proliferative potential: Really an aggressive variant? J. Neurooncol. 2018, 137, 543–549. [Google Scholar] [CrossRef]
- Trouillas, J.; Roy, P.; Sturm, N.; Dantony, E.; Cortet-Rudelli, C.; Viennet, G.; Bonneville, J.-F.; Assaker, R.; Auger, C.; Brue, T. A new prognostic clinicopathological classification of pituitary adenomas: A multicentric case-control study of 410 patients with 8 years post-operative follow-up. Acta Neuropathol. 2013, 126, 123–135. [Google Scholar] [CrossRef]
- Grimm, F.; Maurus, R.; Beschorner, R.; Naros, G.; Stanojevic, M.; Gugel, I.; Giese, S.; Bier, G.; Bender, B.; Honegger, J. Ki-67 labeling index and expression of p53 are non-predictive for invasiveness and tumor size in functional and nonfunctional pituitary adenomas. Acta Neurochir. (Wien) 2019, 161, 1149–1156. [Google Scholar] [CrossRef]
- Zaidi, H.A.; Cote, D.J.; Dunn, I.F.; Laws, E.R. Predictors of aggressive clinical phenotype among immunohistochemically confirmed atypical adenomas. J. Clin. Neurosci. 2016, 34, 246–251. [Google Scholar] [CrossRef]
| Characteristics | |
|---|---|
| Sex (number and % female) | 40 (35.4) |
| Age at diagnosis (years, median, IQR) | 58 (44–70.5) |
| Maximum tumor diameter at diagnosis (mm, median, IQR) | 29.5 (22–39.5) |
| Invasiveness (% Knosp grade ≥ 3) | 52 (46) |
| Ki-67 index (%, median, IQR) | 0.54 (0.30–1.05) |
| Histological Subtype | Number (%) |
|---|---|
| Gonadotroph-storing tumor | 67 (59.3) |
| Null cell tumor | 32 (28.3) |
| Silent corticotroph tumor | 10 (8.8) |
| Plurihormonal tumor | 2 (1.8) |
| Silent somatotroph tumor | 2 (1.8) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Flores-Martinez, Á.; Venegas-Moreno, E.; Dios, E.; Remón-Ruiz, P.; Gros-Herguido, N.; Vázquez-Borrego, M.C.; Madrazo-Atutxa, A.; Japón, M.A.; Kaen, A.; Cárdenas-Valdepeñas, E.; et al. Quantitative Analysis of Somatostatin and Dopamine Receptors Gene Expression Levels in Non-functioning Pituitary Tumors and Association with Clinical and Molecular Aggressiveness Features. J. Clin. Med. 2020, 9, 3052. https://doi.org/10.3390/jcm9093052
Flores-Martinez Á, Venegas-Moreno E, Dios E, Remón-Ruiz P, Gros-Herguido N, Vázquez-Borrego MC, Madrazo-Atutxa A, Japón MA, Kaen A, Cárdenas-Valdepeñas E, et al. Quantitative Analysis of Somatostatin and Dopamine Receptors Gene Expression Levels in Non-functioning Pituitary Tumors and Association with Clinical and Molecular Aggressiveness Features. Journal of Clinical Medicine. 2020; 9(9):3052. https://doi.org/10.3390/jcm9093052
Chicago/Turabian StyleFlores-Martinez, Álvaro, Eva Venegas-Moreno, Elena Dios, Pablo Remón-Ruiz, Noelia Gros-Herguido, M. Carmen Vázquez-Borrego, Ainara Madrazo-Atutxa, Miguel A. Japón, Ariel Kaen, Eugenio Cárdenas-Valdepeñas, and et al. 2020. "Quantitative Analysis of Somatostatin and Dopamine Receptors Gene Expression Levels in Non-functioning Pituitary Tumors and Association with Clinical and Molecular Aggressiveness Features" Journal of Clinical Medicine 9, no. 9: 3052. https://doi.org/10.3390/jcm9093052
APA StyleFlores-Martinez, Á., Venegas-Moreno, E., Dios, E., Remón-Ruiz, P., Gros-Herguido, N., Vázquez-Borrego, M. C., Madrazo-Atutxa, A., Japón, M. A., Kaen, A., Cárdenas-Valdepeñas, E., Roldán, F., Castaño, J. P., Luque, R. M., Cano, D. A., & Soto-Moreno, A. (2020). Quantitative Analysis of Somatostatin and Dopamine Receptors Gene Expression Levels in Non-functioning Pituitary Tumors and Association with Clinical and Molecular Aggressiveness Features. Journal of Clinical Medicine, 9(9), 3052. https://doi.org/10.3390/jcm9093052








